Thermal and Mechanical Characteristics of Okra (Abelmoschus esculentus) Fibers Obtained via Water- and Dew-Retting †
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Fiber Preparation Methodology
2.2.2. Water-Retting Method
2.2.3. Dew-Retting Method
2.2.4. Linear Density Measurements
2.2.5. Mechanical Properties Measurements
2.2.6. Thermogravimetric Analysis
2.2.7. Differential Scanning Calorimetry
3. Results
3.1. Fiber Yield
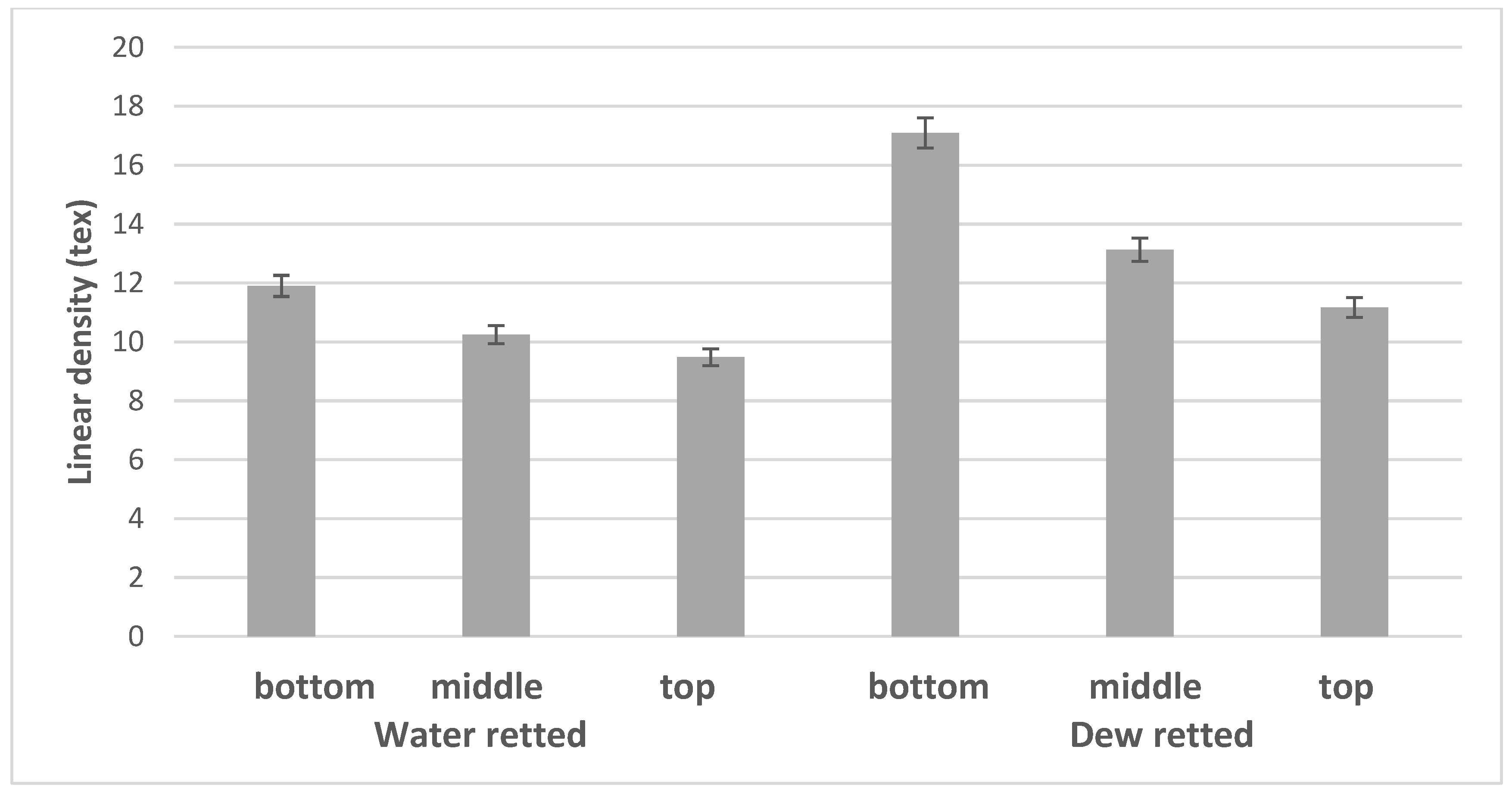
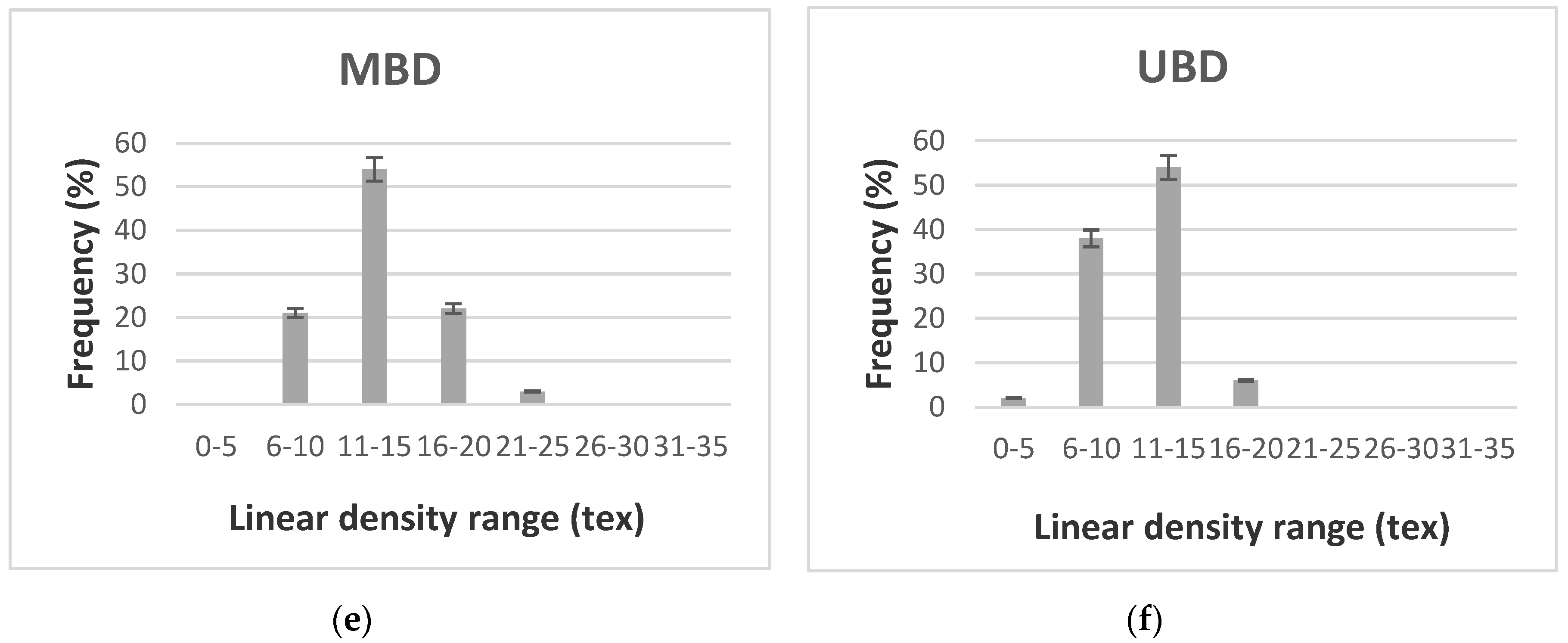
3.2. Linear Density of Fibers
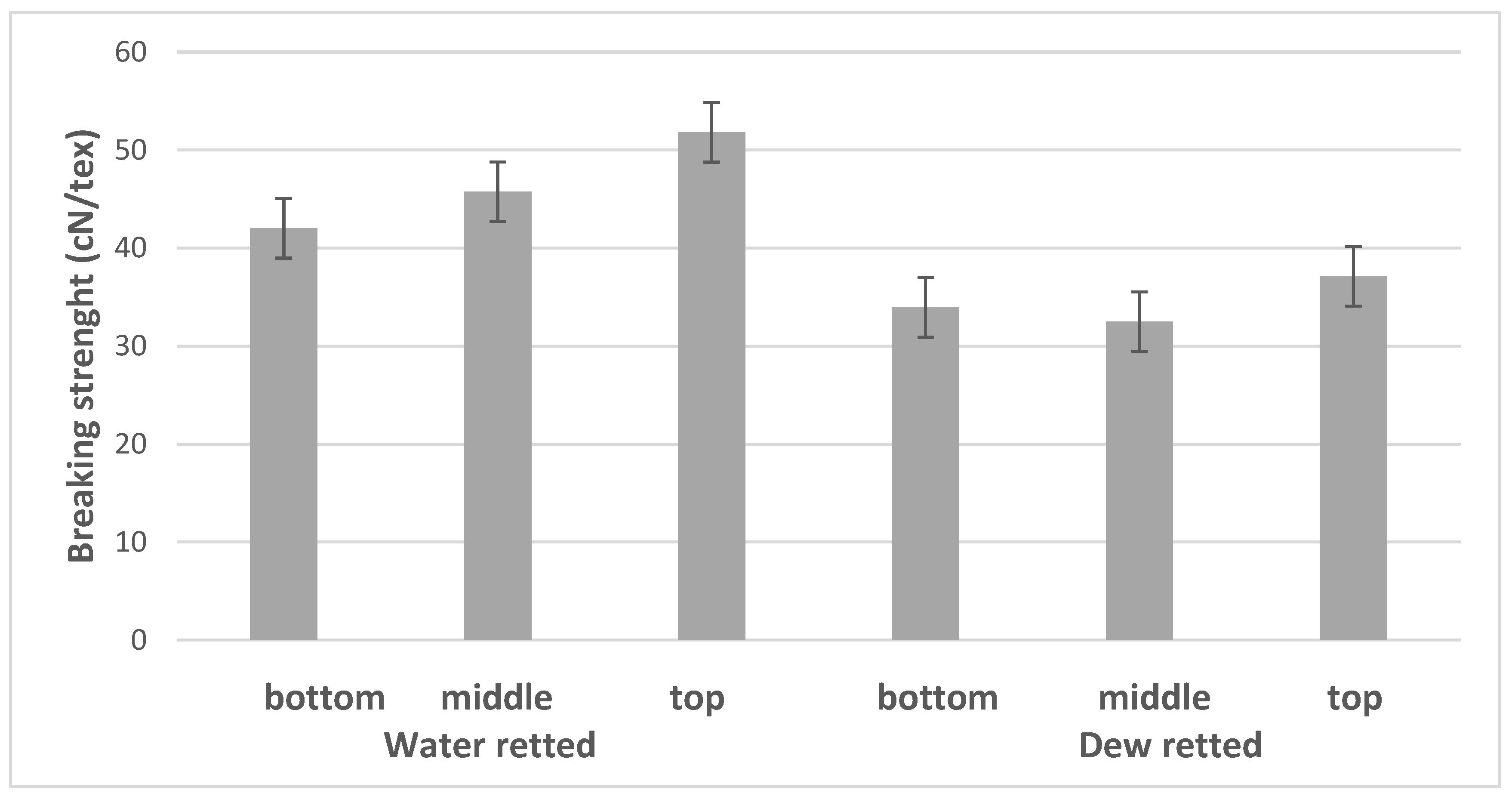
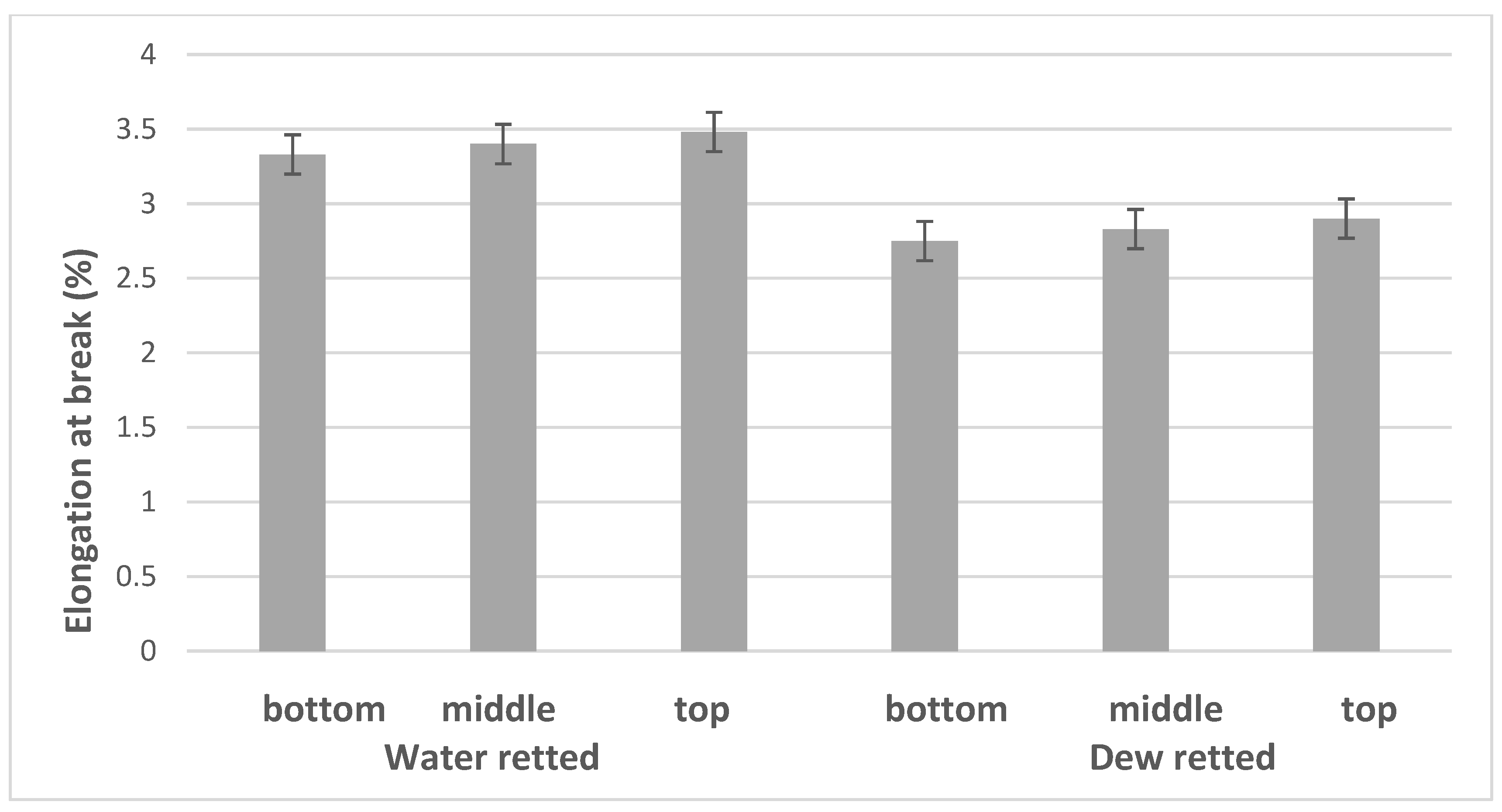
3.3. Mechanical Properties
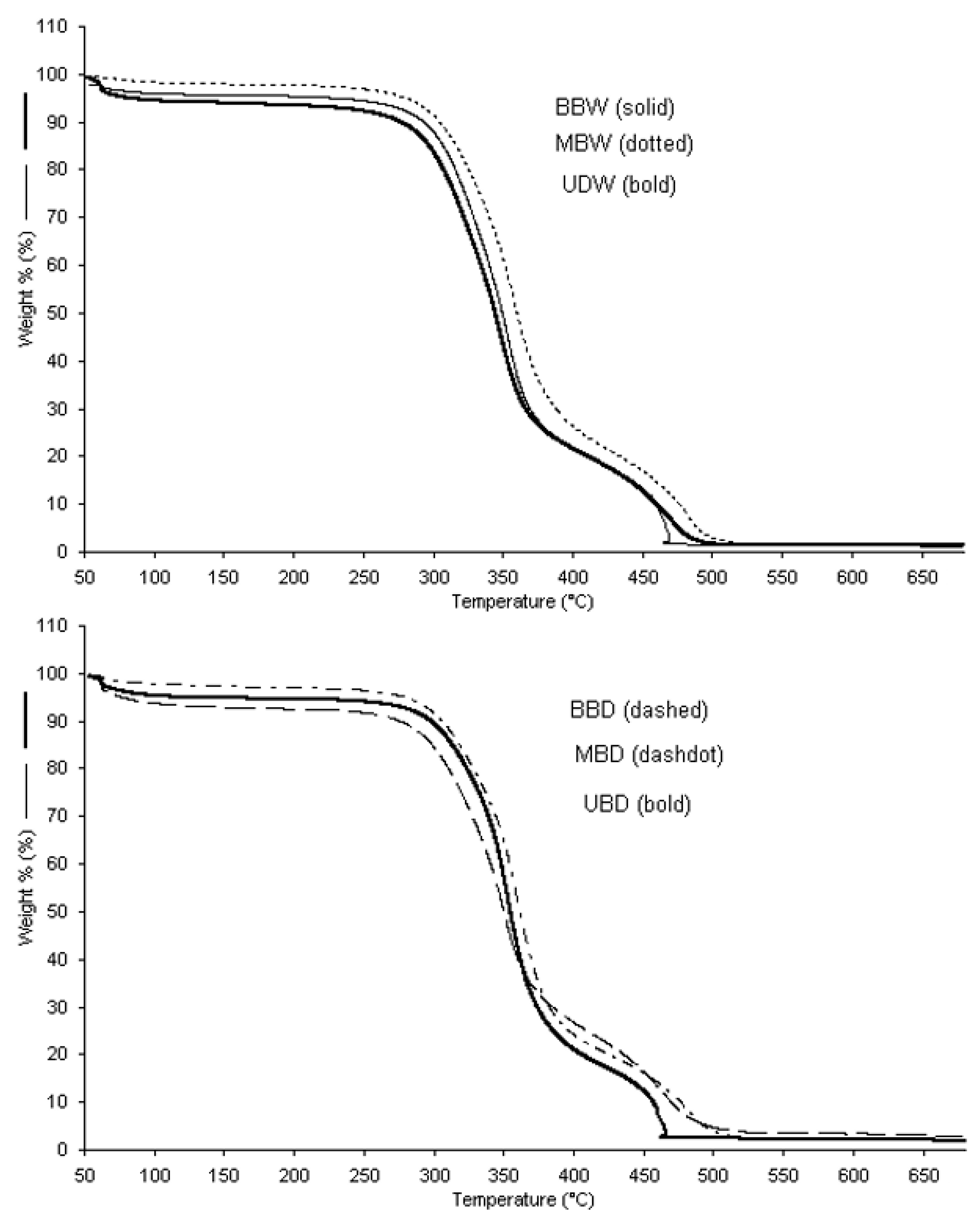
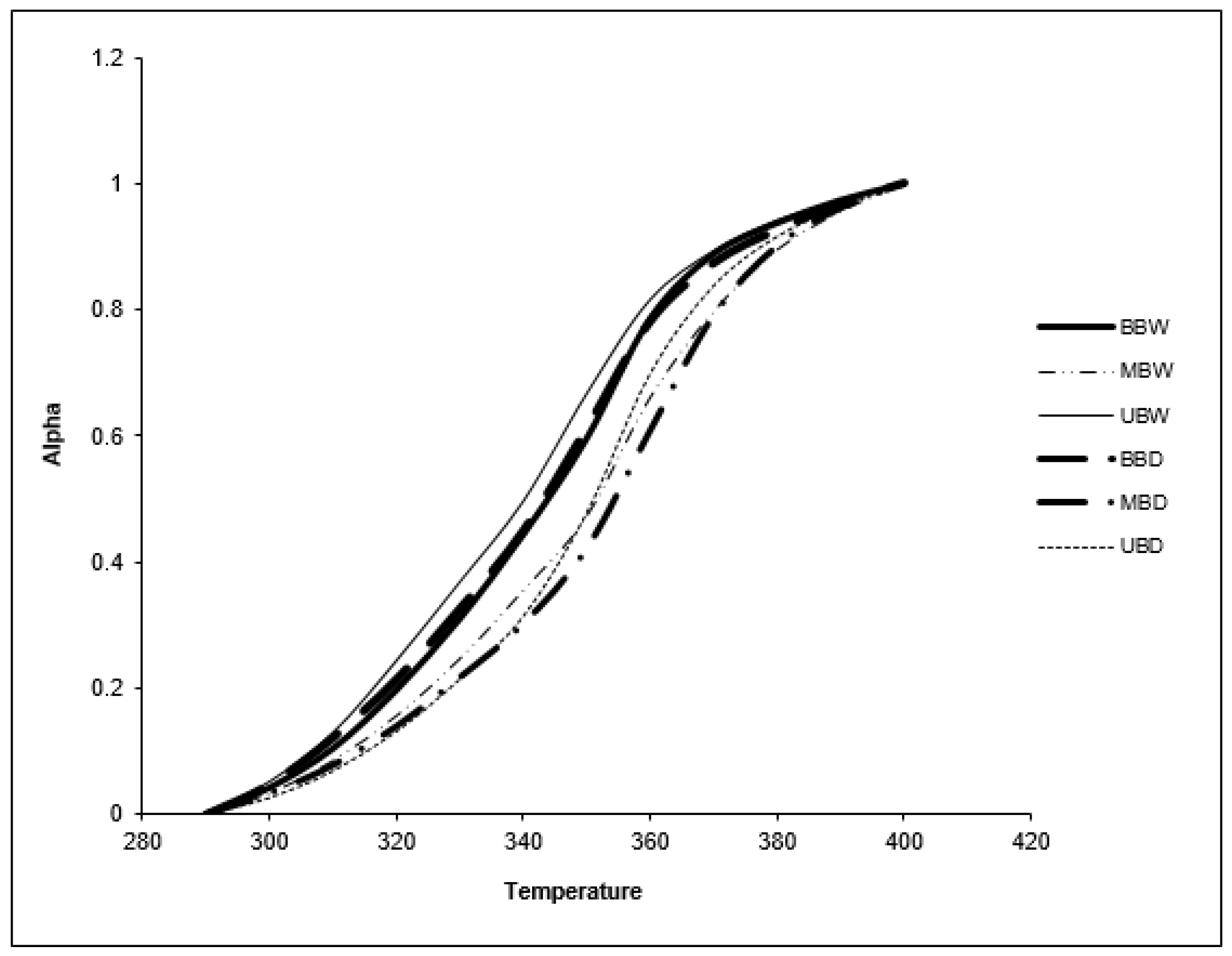
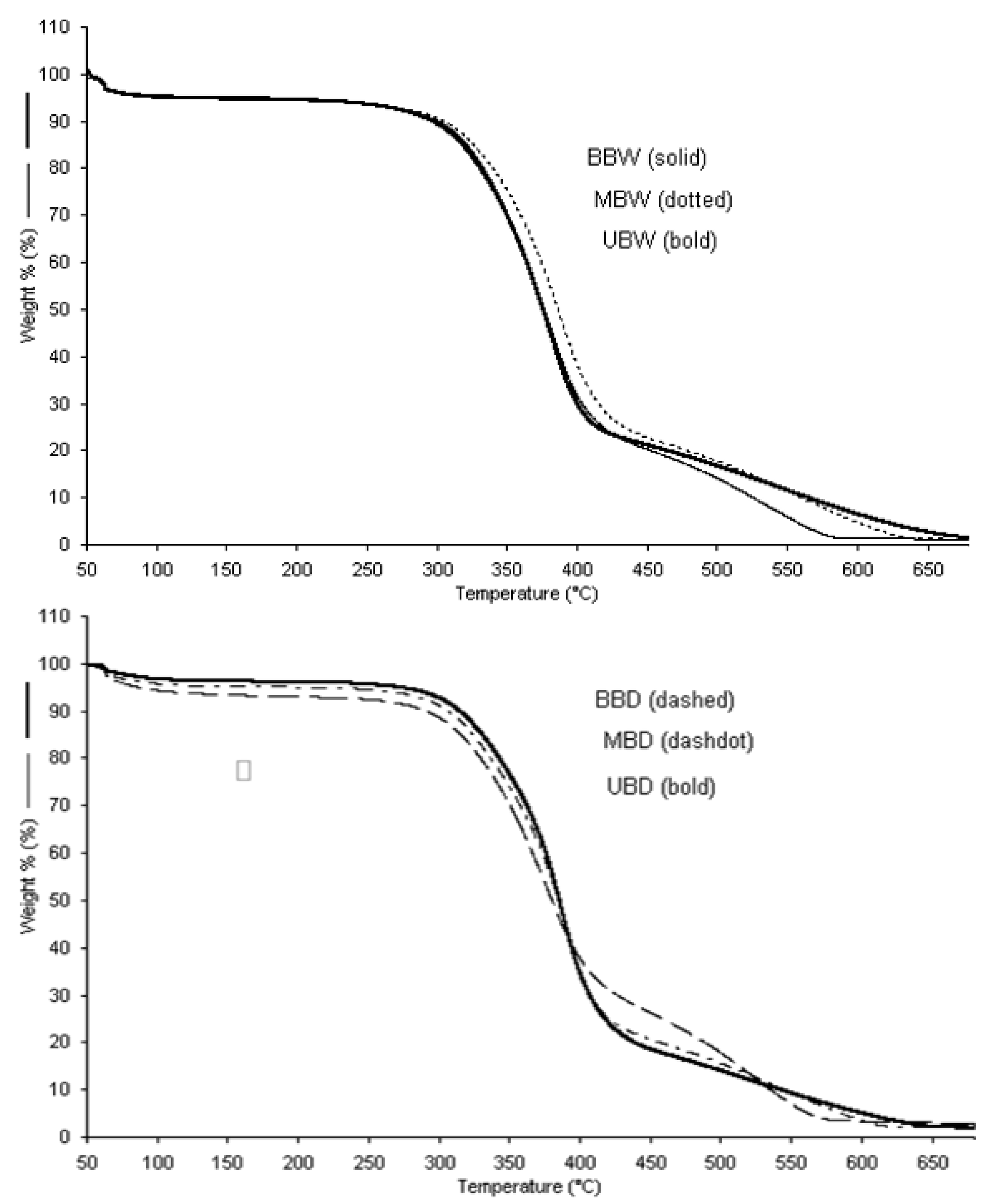
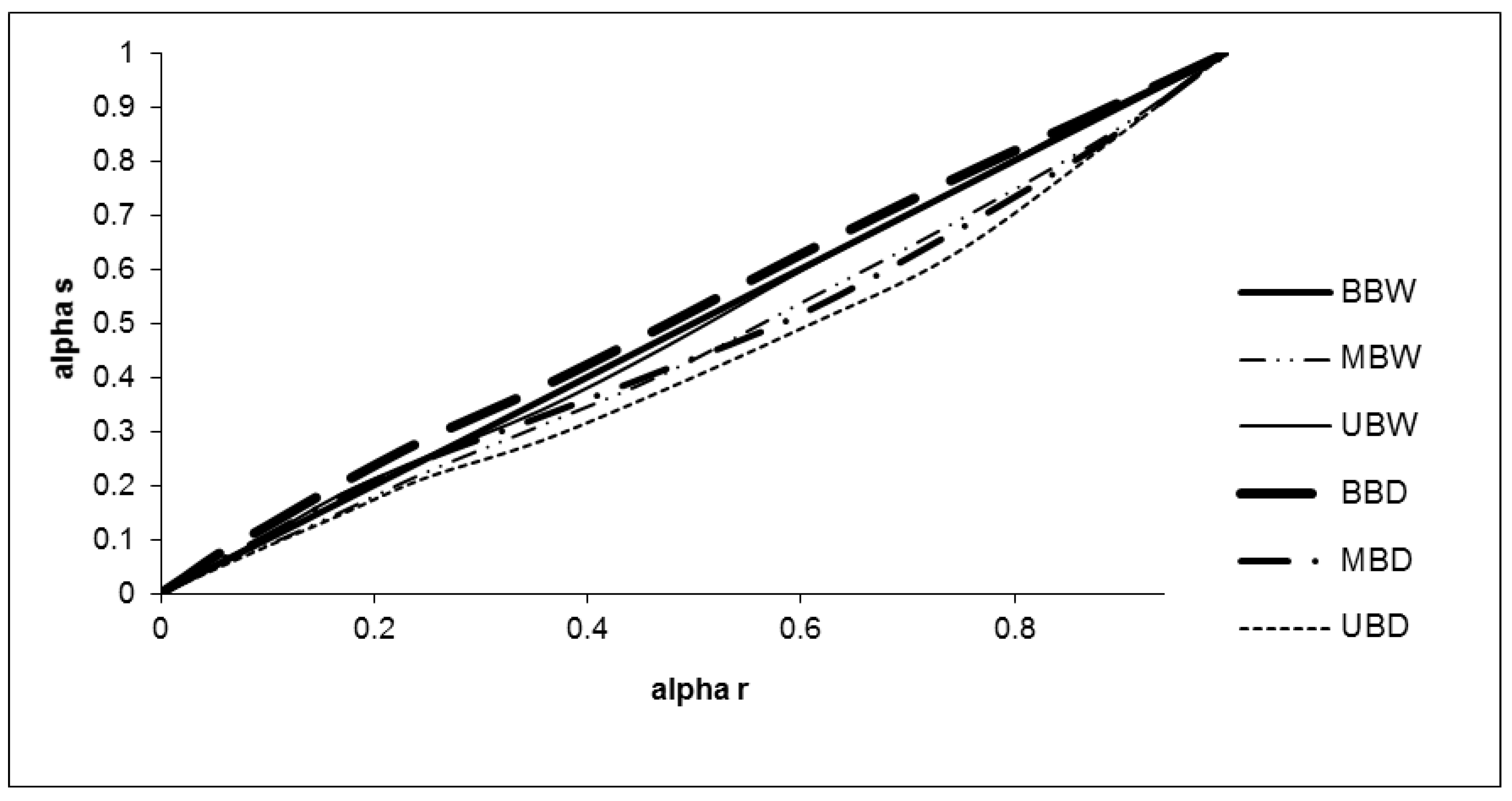
3.4. Thermal Properties
3.4.1. Thermo-Oxidation
3.4.2. Pyrolysis
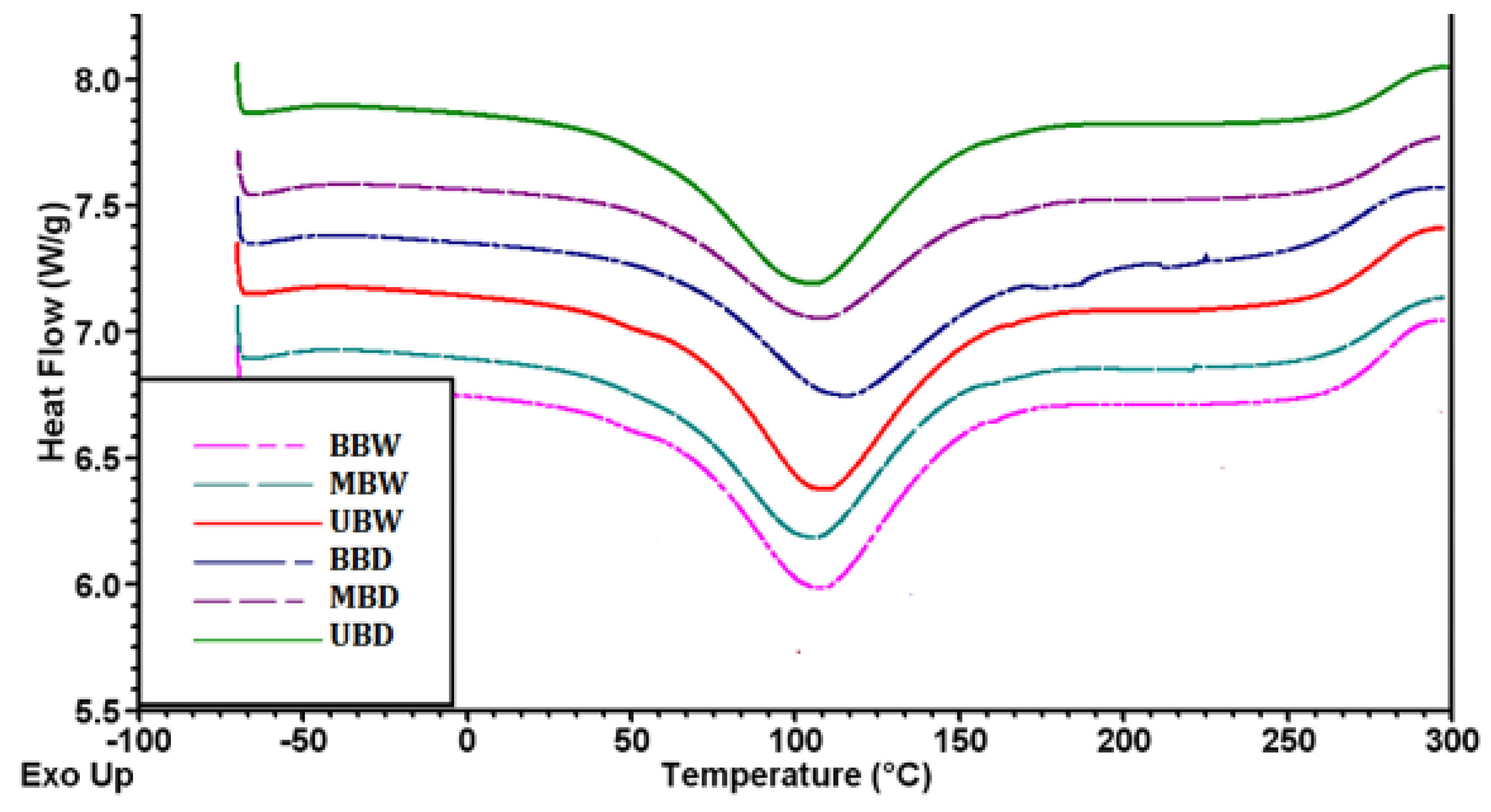
3.4.3. DSC Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- World Commission on Environment and Development. Our Common Future; Oxford University Press: Oxford, UK, 1987. [Google Scholar]
- Mohanty, A.K.; Misra, M.; Drzal, L.T.; Selke, S.E.; Harte, B.R.; Hinrichsen, G. Natural Fibers, Biopolymers, and Biocomposites: An Introduction. In Natural Fibers, Biopolymers, and Biocomposites; Mohanty, A.K., Misra, M., Drzal, L.T., Eds.; CRC Press: Boca Raton, FL, USA, 2005; pp. 1–36. [Google Scholar]
- Bismarck, A.; Mishra, S.; Lampke, T. Plant fibers as reinforcement for green composites. In Natural Fibers, Biopolymers, and Biocomposites; Mohanty, A.K., Misra, M., Drzal, L.T., Eds.; Taylor & Francis: Abingdon, UK, 2005; pp. 37–108. ISBN 9780203508206. [Google Scholar]
- Pickering, K.L.; Efendy, M.G.A.; Le, T.M. A review of recent developments in natural fibre composites and their mechanical performance. Compos. Part A Appl. Sci. Manuf. 2016, 83, 98–112. [Google Scholar] [CrossRef]
- Johnson, T. “History of Composites”. ThoughtCo. 11 February 2020. Available online: Thoughtco.com/history-of-composites-820404 (accessed on 24 June 2020).
- Rahman, M.R.; Hasan, M.; Huque, M.M.; Islam, M.N. Physico-mechanical properties of jute fiber reinforced polypropylene composites. J. Reinf. Plast. Compos. 2010, 29, 445–455. [Google Scholar] [CrossRef]
- Nair, G.R.; Singh, A.; Zimniewska, M.; Raghavan, V. Comparative evaluation of physical and structural properties of water retted and non-retted flax fibers. Fibers 2013, 1, 59–69. [Google Scholar] [CrossRef]
- Mathangadeera, R.W.; Hequet, E.F.; Kelly, B.; Dever, J.K.; Kelly, C.M. Importance of cotton fiber elongation in fiber processing. Ind. Crop. Prod. 2020, 147, 112217. [Google Scholar] [CrossRef]
- Ruan, P.; Du, J.; Raghavan, V.; Lyew, D.; Gariepy, Y.; Yang, H. Microwave pretreated enzymatic retting of flax stems and comparison with the effect of radio frequency pretreatment. Ind. Crop. Prod. 2020, 151, 112312. [Google Scholar] [CrossRef]
- Grégoire, M.; Barthod-Malat, B.; Labonne, L.; Evon, P.; De Luycker, E.; Ouagne, P. Investigation of the potential of hemp fibre straws harvested using a combine machine for the production of technical load-bearing textiles. Ind. Crop. Prod. 2020, 145, 111988. [Google Scholar] [CrossRef]
- Ivanovska, A.; Cerovic, D.; Tadic, N.; Castvan, I.J.; Asanovic, K.; Kostic, M. Sorption and dielectric properties of jute woven fabrics: Effect of chemical composition. Ind. Crop. Prod. 2019, 140, 111632. [Google Scholar] [CrossRef]
- Sun, Z.; Mingming, W. Effects of sol-gel modification on the interfacial and mechanical properties of sisal fiber reinforced polypropylene composites. Ind. Crop. Prod. 2019, 137, 89–97. [Google Scholar] [CrossRef]
- Stokke, D.D. Alternative Low-Cost Biomass for the Biocomposites Industry. In Natural Fibers, Biopolymers, and Biocomposites; Mohanty, A.K., Misra, M., Drzal, L.T., Eds.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Yilmaz, N.D.; Konak, S.; Yilmaz, K.; Kartal, A.A.; Kayahan, E. Characterization, modification and use of biomass: Okra fibers. Bioinspired Biomim. Nanobiomaterials 2016, 5, 85–95. [Google Scholar] [CrossRef]
- Sain, M.; Panthapulakkal, S. Bioprocess preparation of wheat straw fibers and their characterization. Ind. Crop. Prod. 2006, 23, 1–8. [Google Scholar] [CrossRef]
- Bacci, L.; Baronti, S.; Predieri, S.; di Virgilio, N. Fiber yield and quality of fiber nettle (Urtica dioica L.) cultivated in Italy. Ind. Crop. Prod. 2009, 29, 480–484. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Properties and potential applications of natural cellulose fibers from cornhusks. Green Chem. 2005, 7, 190–195. [Google Scholar] [CrossRef]
- Subagyo, A.; Chafidz, A. Banana pseudo-stem fiber: Preparation, characteristics, and applications. In Banana Nutrition-Function and Processing Kinetics; Jideani, A.I.O., Anyasi, T.A., Eds.; IntechOpen: London, UK, 2018. [Google Scholar]
- Khan, G.M.A.; Yilmaz, N.D.; Yilmaz, K. Effects of chemical treatments and degumming methods on physical and mechanical properties of okra bast and corn husk fibers. J. Text. Inst. 2019, 1–18. [Google Scholar] [CrossRef]
- Ogaji, I.; Hoag, S. Novel extraction and application of okra gum as a film coating agent using theophylline as a model drug. J. Adv. Pharm. Technol. Res. 2014, 5, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.tilasto.com/en/topic/geography-and-agriculture/crop/okra/okra-production-quantity (accessed on 24 June 2020).
- De Rosa, I.M.; Kenny, J.M.; Puglia, D.; Santulli, C.; Sarasini, F. Morphological, thermal and mechanical characterization of okra (Abelmoschus esculentus) fibres as potential reinforcement in polymer composites. Compos. Sci. Technol. 2010, 70, 116–122. [Google Scholar] [CrossRef]
- De Rosa, I.M.; Kenny, J.M.; Maniruzzaman, M.; Moniruzzaman, M.; Monti, M.; Puglia, D.; Santulli, C.; Sarasini, F. Effect of chemical treatments on the mechanical and thermal behaviour of okra (Abelmoschus esculentus) fibres. Compos. Sci. Technol. 2011, 71, 246–254. [Google Scholar] [CrossRef]
- Arifuzzaman Khan, G.M.; Yilmaz, N.D.; Yilmaz, K. Okra bast fiber as potential reinforcement element of biocomposites: Can it be the flax of the future. In Handbook of Composites from Renewable Materials; Wiley Scrivener: Hoboken, MA, USA, 2017; Volumes 1–8, pp. 379–405. ISBN 9781119441632. [Google Scholar]
- Zhu, J.; Zhu, H.; Njuguna, J.; Abhyankar, H. Recent development of flax fibres and their reinforced composites based on different polymeric matrices. Materials 2013, 6, 5171–5198. [Google Scholar] [CrossRef]
- Khan, G.M.A.; Shaheruzzaman, M.; Rahman, M.H.; Razzaque, S.M.A.; Islam, M.S.; Alam, M.S. Surface modification of okra bast fiber and its physico-chemical characteristics. Fibers Polym. 2009, 10, 65–70. [Google Scholar] [CrossRef]
- Nie, X.R.; Hong, Y.L.; Gang, D.; Lin, S.; Hu, R.; Li, H.Y.; Zhao, L.; Zhang, Q.; Chen, H.; Wu, D.T.; et al. Structural characteristics, rheological properties, and biological activities of polysaccharides from different cultivars of okra (Abelmoschus esculentus) collected in China. Int. J. Biol. Macromol. 2019, 139, 459–467. [Google Scholar] [CrossRef]
- Alam, M.S.; Khan, G.M.A. Chemical analysis of okra bast fiber (Abelmoschus esculentus) and its physico-chemical properties. J. Text. Apparel Technol. Manag. 2007, 5, 1–9. [Google Scholar]
- Tanaka, H. Characterization of Polymers. Kobunshi 1994, 43, 81. [Google Scholar] [CrossRef]
- Levi, D.W.; Reich, L.; Lee, H.T. Degradation of polymers by thermal gravimetric techniques. Polym. Eng. Sci. 1965, 5, 135–141. [Google Scholar] [CrossRef]
- Brown, M.E.; Maciejewski, M.; Vyazovkin, S.; Nomen, R.; Sempere, J.; Burnham, A.; Opfermann, J.; Strey, R.; Anderson, H.L.; Kemmler, A.; et al. Computational aspects of kinetic analysis Part A: The ICTAC Kinetics Project-data, methods and results. Thermochim. Acta 2000, 355, 125–143. [Google Scholar] [CrossRef]
- Marcilla, A.; García-Quesada, J.C.; Ruiz-Femenia, R. Additional considerations to the paper entitled: “Computational aspects of kinetic analysis. Part B: The ICTAC Kinetics Project—The decomposition kinetics of calcium carbonate revisited, or some tips on survival in the kinetic minefield”. Thermochim. Acta 2006, 445, 92–96. [Google Scholar] [CrossRef]
- Vyazovkin, S. Computational aspects of kinetic analysis. Part C. The ICTAC Kinetics Project-The light at the end of the tunnel? Thermochim. Acta 2000, 355, 155–163. [Google Scholar] [CrossRef]
- Burnham, A.K. Computational aspects of kinetic analysis. Part D: The ICTAC Kinetics Project-Multi-thermal-history model-fitting methods and their relation to isoconversional methods. Thermochim. Acta 2000, 355, 165–170. [Google Scholar] [CrossRef]
- Maciejewski, M. Computational aspects of kinetic analysis. Part B: The ICTAC Kinetics Project Ð the decomposition kinetics of calcium carbonate revisited, or some tips on survival in the kinetic minefield. Thermochim, Acta 2000, 355, 145–154. [Google Scholar] [CrossRef]
- Van Krevelen, D.W.; van Heerden, C.; Huntjes, F.J. Physicochemical aspects of the pyrolysis of coal and related organic compound. Fuel 1951, 30, 253–259. [Google Scholar] [CrossRef]
- Marini, A.; Berbenni, V.; Flor, G. Kinetic Parameters from Thermogravimetric Data. Z. Naturforsch.-Sect. A J. Phys. Sci. 1979, 34, 661–663. [Google Scholar] [CrossRef]
- Soares, S.; Camino, G.; Levchik, S. Effect of metal carboxylates on the thermal decomposition of cellkulose. Polym. Degrad. Stabil. 1998, 62, 25–31. [Google Scholar] [CrossRef]
- Poletto, M.; Zattera, A.J.; Forte, M.M.C.; Santana, R.M.C. Thermal decomposition of wood: Influence of wood components and cellulose crystallite size. Bioresour. Technol. 2012, 109, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liu, Q.; Luo, Z.; Wen, L.; Cen, K. Mechanism study on cellulose pyrolysis using thermogravimetric analysis coupled with infrared spectroscopy. Front. Energy Power Eng. China 2007, 1, 413–419. [Google Scholar] [CrossRef]
- Gao, M.; Wu, W.H.; Wu, F.C. Thermal degradation and smoke suspension of cotton cellulose modified with THPC and its lanthanide metal complexes. J. Therm. Anal. Calorim. 2009, 98, 245–252. [Google Scholar] [CrossRef]
- Gaan, S.; Rupper, P.; Salimova, V.; Heuberger, S.; Rabe, S.; Vogel, F. Thermal decomposition and burning behavior of cellulose treated with ethyl ester phosphoramidates: Effect of alkyl substituent on nitrogen atom. Polym. Degrad. Stab. 2009, 94, 1125–1134. [Google Scholar] [CrossRef]
- Aggarwal, P.; Dollimore, D. A comparative study of the degradation of different starches using thermal analysis. Talanta 1996, 43, 1527–1530. [Google Scholar] [CrossRef]
- Aggarwal, P.; Dollimore, D.; Kirn, Y.W. A comparative thermal analysis study of starch, cellulose and cationically modified products of these compounds. STP Pharma Sci. 1997, 7, 295–299. [Google Scholar]
- Stawski, D.; Jantas, R. Potato starch thermooxidation: Selection of the optimal calculation method for activation energy determination. Potato Res. 2009, 52, 355–365. [Google Scholar] [CrossRef]
- Lee, W.-F. Comparative study of various methods for thermal degradation of poly[3-dimethyl(methacryloyloxyethyl) ammonium propanesulfonate]. J. Appl. Polym. Sci. 1989, 37, 3263–3275. [Google Scholar] [CrossRef]




| Sample Source | Sample Code |
|---|---|
| Water-retted bottom okra bast fiber | BBW |
| Water-retted middle okra bast fiber | MBW |
| Water-retted upper okra bast fiber | UBW |
| Dew-retted bottom okra bast fiber | BBD |
| Dew-retted middle okra bast fiber | MBD |
| Dew-retted upper okra bast fiber | UBD |
| Okra Bast Fiber | Fiber Yield (%) | Linear Density (tex) | Breaking Strength (cN/tex) | Elongation at Break (%) | |||
|---|---|---|---|---|---|---|---|
| µ | σ | µ | σ | µ | σ | ||
| BBW | 14.47 | 11.90 | 4.30 | 42.01 | 17.40 | 3.33 | 1.07 |
| MBW | 14.33 | 10.25 | 2.66 | 45.76 | 16.29 | 3.40 | 0.92 |
| UBW | 12.65 | 9.45 | 2.79 | 51.80 | 18.30 | 3.48 | 1.30 |
| BBD | 14.73 | 17.09 | 6.95 | 33.93 | 18.99 | 2.75 | 0.98 |
| MBD | 10.85 | 13.13 | 3.68 | 32.50 | 14.09 | 2.83 | 0.87 |
| UBD | 14.25 | 11.17 | 3.07 | 37.12 | 16.71 | 2.90 | 0.89 |
| Sample | Tintit | T50% | Tfinal |
|---|---|---|---|
| BBW | 291.4 | 355.0 | 382.8 |
| MBW | 287.4 | 366.1 | 398.0 |
| UBW | 275.8 | 342.4 | 375.5 |
| BBD | 281.6 | 354.3 | 383.5 |
| MBD | 298.4 | 370.2 | 397.9 |
| UBD | 298.6 | 353.7 | 397.7 |
| Sample | Activation Energy (kJ/mol) |
|---|---|
| BBW | 84.1 |
| MBW | 69.9 |
| UBW | 41.9 |
| BBD | 46.6 |
| MBD | 36.4 |
| UBD | 39.3 |
| Sample | Tintit | T50% | Tfinal |
|---|---|---|---|
| BBW | 300.0 | 374.8 | 406.2 |
| MBW | 303.5 | 389.1 | 426.3 |
| UBW | 299.9 | 372.4 | 403.6 |
| BBD | 300.0 | 377.6 | 406.6 |
| MBD | 300.0 | 382.2 | 410.9 |
| UBD | 299.9 | 389.7 | 412.4 |
| Sample | Activation Energy (kJ/mol) |
|---|---|
| BBW | 60.4 |
| MBW | 50.9 |
| UBW | 50.4 |
| BBD | 42.8 |
| MBD | 54.3 |
| UBD | 62.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stawski, D.; Çalişkan, E.; Yilmaz, N.D.; Krucińska, I. Thermal and Mechanical Characteristics of Okra (Abelmoschus esculentus) Fibers Obtained via Water- and Dew-Retting. Appl. Sci. 2020, 10, 5113. https://doi.org/10.3390/app10155113
Stawski D, Çalişkan E, Yilmaz ND, Krucińska I. Thermal and Mechanical Characteristics of Okra (Abelmoschus esculentus) Fibers Obtained via Water- and Dew-Retting. Applied Sciences. 2020; 10(15):5113. https://doi.org/10.3390/app10155113
Chicago/Turabian StyleStawski, Dawid, Ebru Çalişkan, Nazire Deniz Yilmaz, and Izabella Krucińska. 2020. "Thermal and Mechanical Characteristics of Okra (Abelmoschus esculentus) Fibers Obtained via Water- and Dew-Retting" Applied Sciences 10, no. 15: 5113. https://doi.org/10.3390/app10155113
APA StyleStawski, D., Çalişkan, E., Yilmaz, N. D., & Krucińska, I. (2020). Thermal and Mechanical Characteristics of Okra (Abelmoschus esculentus) Fibers Obtained via Water- and Dew-Retting. Applied Sciences, 10(15), 5113. https://doi.org/10.3390/app10155113








