Vibrotactile Stimulation of Nail of Hallux during Walking: Effect on Center-of-Mass Movement in Healthy Young Adults
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
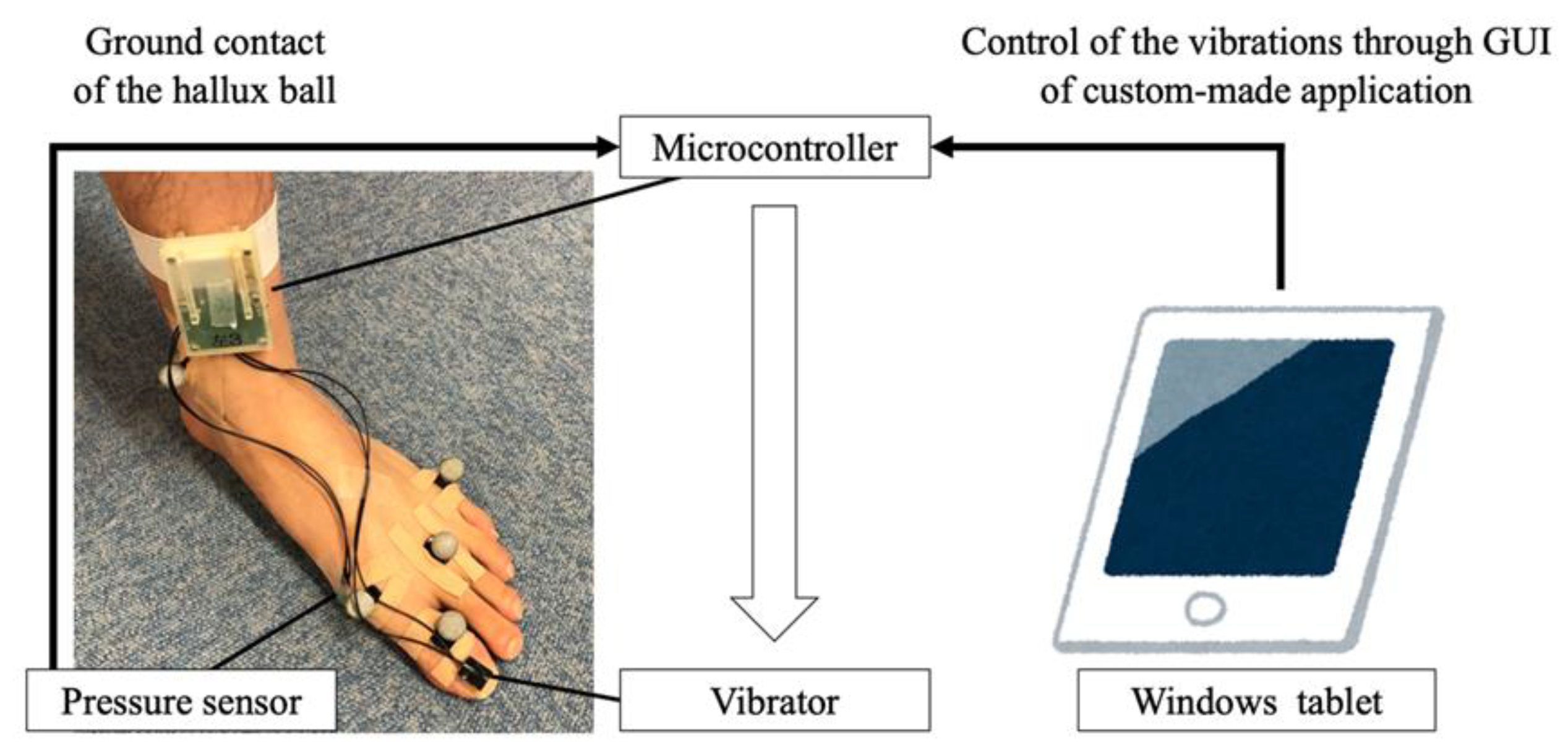
2.2. Vibration System
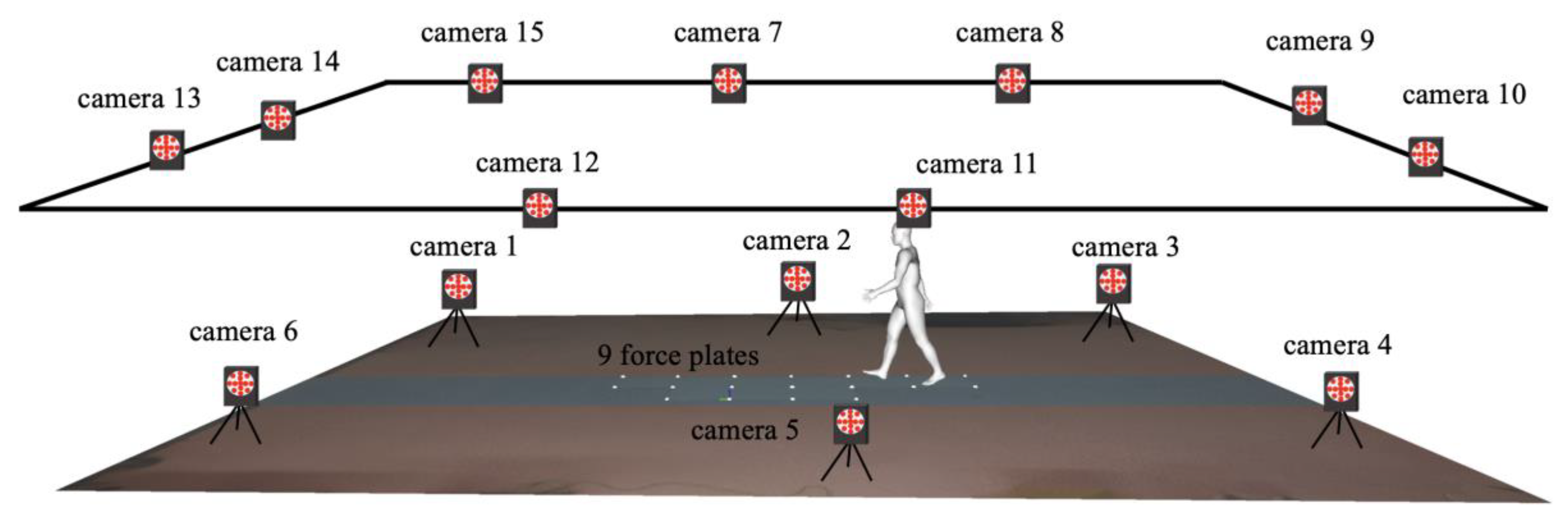
2.3. Experimental Setup
2.4. Data Collection
2.5. Data Analysis
2.6. Statistics
3. Results
3.1. Differences Between Variables Obtained with and Without Stimulation
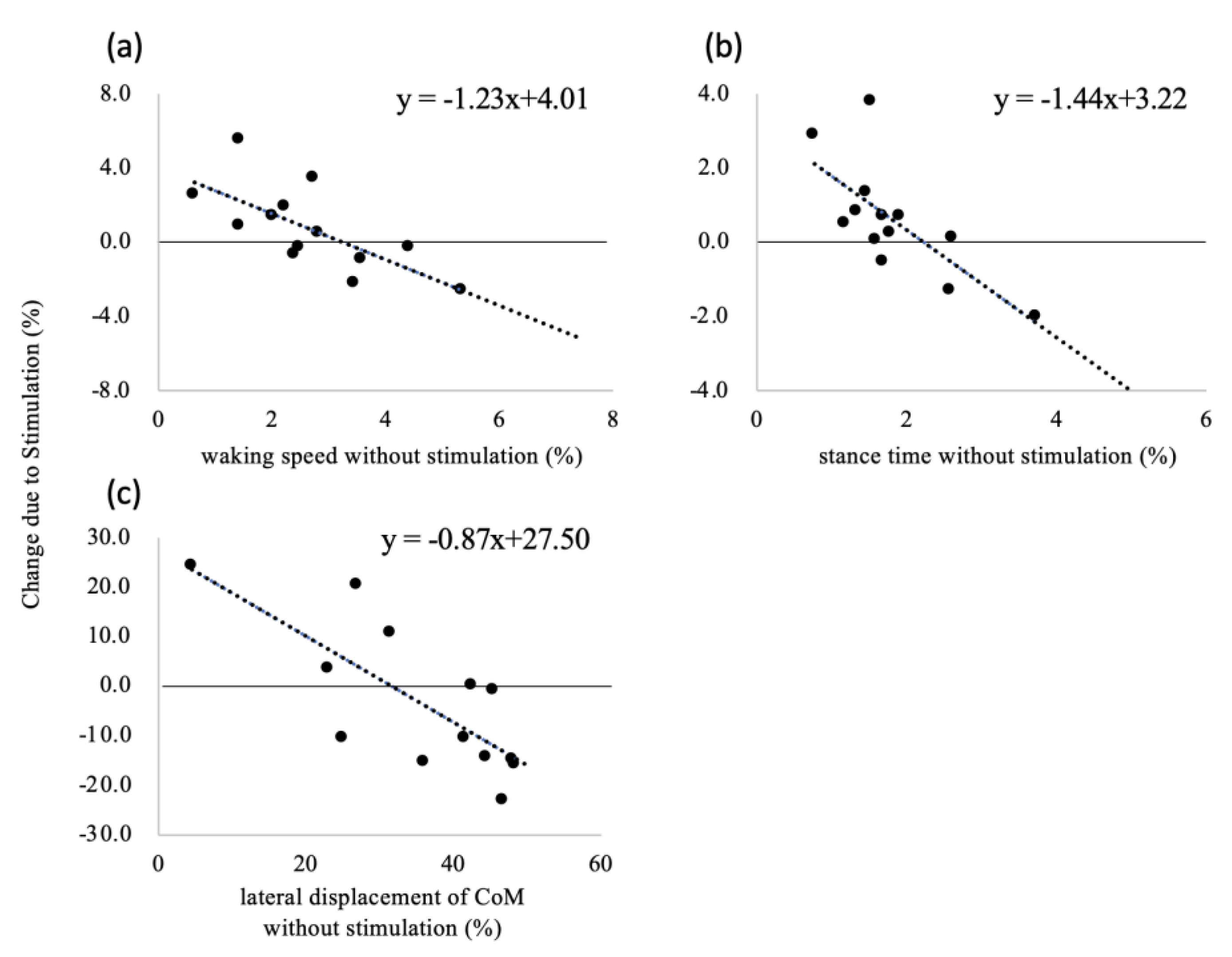
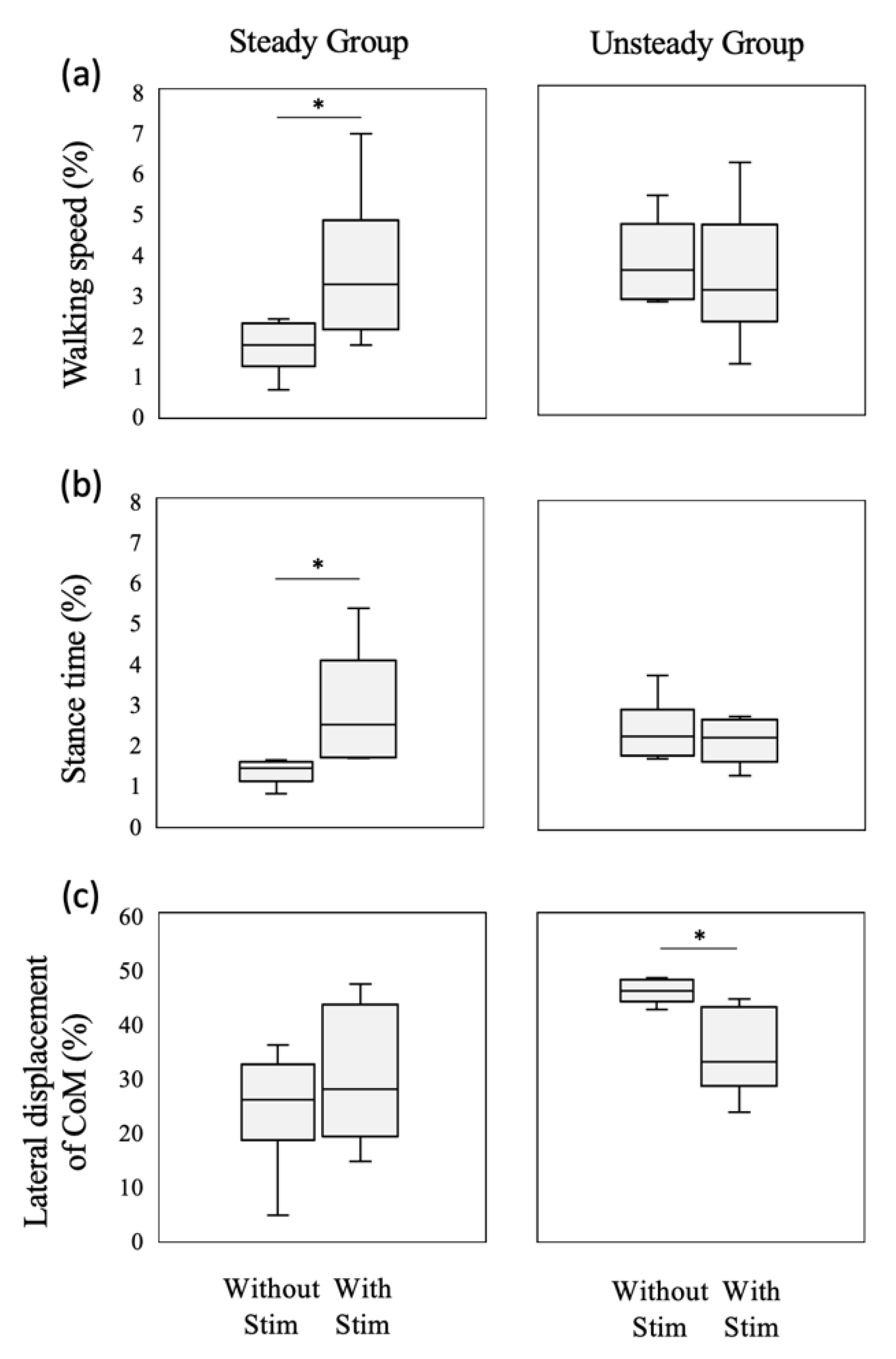
3.2. Baseline-Dependent Effect of Vibrotactile Stimulation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Taylor, A.J.; Menz, H.B.; Keenan, A.M. Effects of experimentally induced plantar insensitivity on forces and pressures under the foot during normal walking. Gait Posture 2004, 20, 232–237. [Google Scholar] [CrossRef]
- Sawa, R.; Doi, T.; Misu, S.; Tsutsumimoto, K.; Fujino, H.; Ono, R. Decreased skin temperature of the foot increases gait variability in healthy young adults. Gait Posture 2013, 38, 518–522. [Google Scholar] [CrossRef]
- Eils, E.; Behrens, S.; Mers, O.; Thorwesten, L.; Völker, K.; Rosenbaum, D. Reduced plantar sensation causes a cautious walking pattern. Gait Posture 2004, 20, 54–60. [Google Scholar] [CrossRef]
- Hohne, A.; Ali, S.; Stark, C.; Bruggemann, G.P. Reduced plantar cutaneous sensation modifies gait dynamics, lower-limb kinematics and muscle activity during walking. Eur. J. Appl. Physiol. 2012, 112, 3829–3838. [Google Scholar] [CrossRef] [PubMed]
- Eils, E.; Nolte, S.; Tewes, M.; Thorwesten, L.; Völker, K.; Rosenbaum, D. Modified pressure distribution patterns in walking following reduction of plantar sensation. J. Biomech. 2002, 35, 1307–1313. [Google Scholar] [CrossRef]
- Menz, H.B.; Lord, S.R.; St George, R.; Fitzpatrick, R.C. Walking stability and sensorimotor function in older people with diabetic peripheral neuropathy. Arch. Phys. Med. Rehabil. 2004, 85, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Novak, P.; Novak, V. Effect of step-synchronized vibration stimulation of soles on gait in Parkinson’s disease: A pilot study. J. Neuroeng. Rehabil. 2006, 3, 9. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Cho, K.H.; Lee, W.H. Effect of a local vibration stimulus training programme on postural sway and gait in chronic stroke patients: A randomized controlled trial. Clin. Rehabil. 2013, 27, 921–931. [Google Scholar] [CrossRef]
- Stephen, D.G.; Wilcox, B.J.; Niemi, J.B.; Franz, J.R.; Kerrigan, D.; D’Andrea, S.E. Baseline-dependent effect of noise-enhanced insoles on gait variability in healthy elderly walkers. Gait Posture 2012, 36, 537–540. [Google Scholar] [CrossRef]
- Chien, J.H.; Ambati, V.N.P.; Huang, C.K.; Mukherjee, M. Tactile stimuli affect long-range correlations of stride interval and stride length differently during walking. Exp. Brain Res. 2017, 235, 1185–1193. [Google Scholar] [CrossRef]
- Sakai, K.; Hachisu, T.; Hashimoto, Y. Characteristics of illusory vibration in the toe pad induced by vibration applied to toenail and toe force to surface. In Proceedings of the 2017 IEEE World Haptics Conference (WHC), Munich, Germany, 5–9 June 2017; pp. 281–286. [Google Scholar] [CrossRef]
- Sakai, K.; Hachisu, T.; Hashimoto, Y. Sole tactile display using tactile illusion by vibration on toenail. In Haptic Interaction, Lecture Notes in Electrical Engineering, Proceedings of the International Asia Haptics Conference, Kashiwa, Japan, 29 November–1 December 2016; Hasegawa, S., Konyo, M., Kyung, K.U., Nojima, T., Kajimoto, H., Eds.; Springer: Singapore, 2016; Volume 432, pp. 95–97. [Google Scholar] [CrossRef]
- Sakai, K.; Hashimoto, Y. Perceptual characteristics of a tactile illusion using toenail-mounted vibration. In Proceedings of the ICAT-EGVE 2015, Kyoto, Japan, 28–30 October 2015; p. 10. [Google Scholar]
- Hennig, E.M.; Sterzing, T. Sensitivity mapping of the human foot: Thresholds at 30 skin locations. Foot Ankle Int. 2009, 30, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, C.D.; Winter, D.A. Control of whole body balance in the frontal plane during human walking. J. Biomech. 1993, 26, 633–644. [Google Scholar] [CrossRef]
- Fujimoto, M.; Chou, L.S. Sagittal plane momentum control during walking in elderly fallers. Gait Posture 2016, 45, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Schneiders, A.G.; Sullivan, S.J.; O’Malley, K.J.; Clarke, S.V.; Knappstein, S.A.; Taylor, L.J. A valid and reliable clinical determination of footedness. PM&R 2010, 2, 835–841. [Google Scholar] [CrossRef]
- World Medical Association General Assembly. World medical association declaration of helsinki. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Ogata, T. Association between the gait pattern characteristics of older people and their two-step test scores. BMC Geriatr. 2018, 18, 101. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Tada, M.; Mochimaru, M. Dhaiba: Development of virtual ergonomic assessment system with human models. In Proceedings of the 3rd International Digital Human Modeling Symposium, Tokyo, Japan, 20–22 May 2014. [Google Scholar]
- Kobayashi, Y.; Hobara, H.; Matsushita, S.; Mochimaru, M. Key joint kinematic characteristics of the gait of fallers identified by principal component analysis. J. Biomech. 2014, 47, 2424–2429. [Google Scholar] [CrossRef]
- Andreopoulou, G.; Mahad, D.J.; Mercer, T.H.; Van Der Linden, M.L. Test-retest reliability and minimal detectable change of ankle kinematics and spatiotemporal parameters in MS population. Gait Posture 2019, 74, 218–222. [Google Scholar] [CrossRef]
- Haley, S.M.; Fragala-Pinkham, M.A. Interpreting change scores of tests and measures used in physical therapy. Phys. Ther. 2006, 86, 735–743. [Google Scholar] [CrossRef]
- Woolson, R.F. Wilcoxon signed-rank test. In Wiley Encyclopedia of Clinical Trials; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007; pp. 1–3. [Google Scholar] [CrossRef]
- Rogers, M.W.; Mille, M.-L. Lateral stability and falls in older people. Exerc. Sport Sci. Rev. 2003, 31, 182–187. [Google Scholar] [CrossRef]
- Pieruccini-Faria, F.; Montero-Odasso, M.; Hausdorff, J.M. Gait variability and fall risk in older adults: The role of cognitive function. In Falls and Cognition in Older Persons; Montero-Odasso, M., Camicioli, R., Eds.; Springer: Cham, Switzerland, 2019; pp. 107–138. [Google Scholar] [CrossRef]
- Bauby, C.E.; Kuo, A.D. Active control of lateral balance in human walking. J. Biomech. 2000, 33, 1433–1440. [Google Scholar] [CrossRef]
- Beilock, S.L.; Carr, T.H.; MacMahon, C.; Starkes, J.L. When paying attention becomes counterproductive: Impact of divided versus skill-focused attention on novice and experienced performance of sensorimotor skills. J. Exp. Psychol. Appl. 2002, 8, 6–16. [Google Scholar] [CrossRef]
- Moe-Nilssen, R.; Helbostad, J.L. Interstride trunk acceleration variability but not step width variability can differentiate between fit and frail older adults. Gait Posture 2005, 21, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Gabell, A.; Nayak, U. The effect of age on variability in gait. J. Gerontol. 1984, 39, 662–666. [Google Scholar] [CrossRef]
- Taga, G.; Yamaguchi, Y.; Shimizu, H. Self-organized control of bipedal locomotion by neural oscillators in unpredictable environment. Biol. Cybern. 1991, 65, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Kerrigan, D.C.; Todd, M.K.; Croce, U.D. Gender differences in joint biomechanics during walking; Normative study in young adults. Am. J. Phys. Med. Rehabil. 1998, 77, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Aboutorabi, A.; Arazpour, M.; Bahramizadeh, M.; Farahmand, F.; Fadayevatan, R. Effect of vibration on postural control and gait of elderly subjects: A systematic review. Aging Clin. Exp. Res. 2018, 30, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Pandy, M.G.; Lin, Y.-C.; Kim, H.J. Muscle coordination of mediolateral balance in normal walking. J. Biomech. 2010, 43, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
| Variable | Without Stim | With Stim | MDC | P-Value | ||
|---|---|---|---|---|---|---|
| Mean | Walking speed (m/s) | 1.32 ± 0.15 | 1.29 ± 0.14 | 0.10 | 0.03 * | |
| Stance time (s) | 0.61 ± 0.05 | 0.61 ± 0.05 | 0.03 | 0.38 | ||
| CoM (cm) | Vertical | 92.66 ± 2.49 | 92.65 ± 2.46 | 1.06 | 0.97 | |
| Progression | 81.22 ± 6.93 | 79.42 ± 7.54 | 5.69 | 0.02 * | ||
| Lateral | 1.92 ± 0.67 | 1.75 ± 0.73 | 2.17 | 0.97 | ||
| CV (%) | Walking speed | 2.69 ± 1.28 | 3.37 ± 1.65 | 0.38 | ||
| Stance time | 1.84 ± 0.76 | 2.39 ± 1.12 | 0.15 | |||
| CoM | Vertical | 0.35 ± 0.20 | 0.36 ± 0.25 | 0.86 | ||
| Progression | 2.48 ± 0.95 | 2.61 ± 1.40 | 0.75 | |||
| Lateral | 35.69 ± 13.00 | 32.16 ± 9.81 | 0.38 | |||
| Variable | Unstandardized Coefficients (95% CI) | Standardized Coefficients | Adjusted R2 | P-Value | ||
|---|---|---|---|---|---|---|
| Mean | Walking speed | −0.10 (−0.36 to 0.15) | −0.27 | 0.01 | 0.38 | |
| Stance time | −0.01 (−0.22 to 0.18) | −0.06 | 0.08 | 0.84 | ||
| CoM | Vertical | −0.02 (−0.09 to 0.05) | −0.18 | 0.05 | 0.56 | |
| Progression | −0.02 (−0.34 to 0.29) | −0.05 | 0.08 | 0.84 | ||
| Lateral | −0.63 (−1.32 to 0.05) | −0.52 | 0.21 | 0.06 | ||
| CV | Walking speed | −1.23 (−2.08 to −0.39) | −0.70 | 0.44 | 0.008 * | |
| Stance time | −1.44 (−2.37 to 0.52) | −0.71 | 0.47 | 0.006 * | ||
| CoM | Vertical | −0.01 (−0.52 to 0.49) | −0.02 | 0.09 | 0.95 | |
| Progression | −0.64 (−1.59 to 0.30) | −0.41 | 0.09 | 0.16 | ||
| Lateral | −0.87 (−1.36 to −0.37) | −0.76 | 0.54 | 0.003 * | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toda, H.; Hashimoto, Y.; Tada, M. Vibrotactile Stimulation of Nail of Hallux during Walking: Effect on Center-of-Mass Movement in Healthy Young Adults. Appl. Sci. 2020, 10, 4562. https://doi.org/10.3390/app10134562
Toda H, Hashimoto Y, Tada M. Vibrotactile Stimulation of Nail of Hallux during Walking: Effect on Center-of-Mass Movement in Healthy Young Adults. Applied Sciences. 2020; 10(13):4562. https://doi.org/10.3390/app10134562
Chicago/Turabian StyleToda, Haruki, Yuki Hashimoto, and Mitsunori Tada. 2020. "Vibrotactile Stimulation of Nail of Hallux during Walking: Effect on Center-of-Mass Movement in Healthy Young Adults" Applied Sciences 10, no. 13: 4562. https://doi.org/10.3390/app10134562
APA StyleToda, H., Hashimoto, Y., & Tada, M. (2020). Vibrotactile Stimulation of Nail of Hallux during Walking: Effect on Center-of-Mass Movement in Healthy Young Adults. Applied Sciences, 10(13), 4562. https://doi.org/10.3390/app10134562





