Biochar for Wastewater Treatment—Conversion Technologies and Applications
Abstract
1. Introduction
2. Conventional Thermal Methods for the Conversion of Feedstock into Carbonaceous Materials
2.1. Pyrolysis
2.2. Hydrothermal Carbonization
2.3. Gasification
2.4. Torrefaction
3. Modification of Biochar
4. Biochar as an Efficient Adsorbent for Organic and Inorganic Pollutants
4.1. Organic Pollutants
4.2. Heavy Metals
4.3. Biochar Adsorption Mechanism
4.4. Parameters Affecting the Adsorption of Pollutants on Biochar
4.4.1. Biochar Characteristics
4.4.2. Solution pH
4.4.3. Adsorbent Dosage
4.4.4. Temperature
5. Environmental Application of Biochar for Wastewater Treatment
5.1. As a Support Media during Anaerobic Digestion
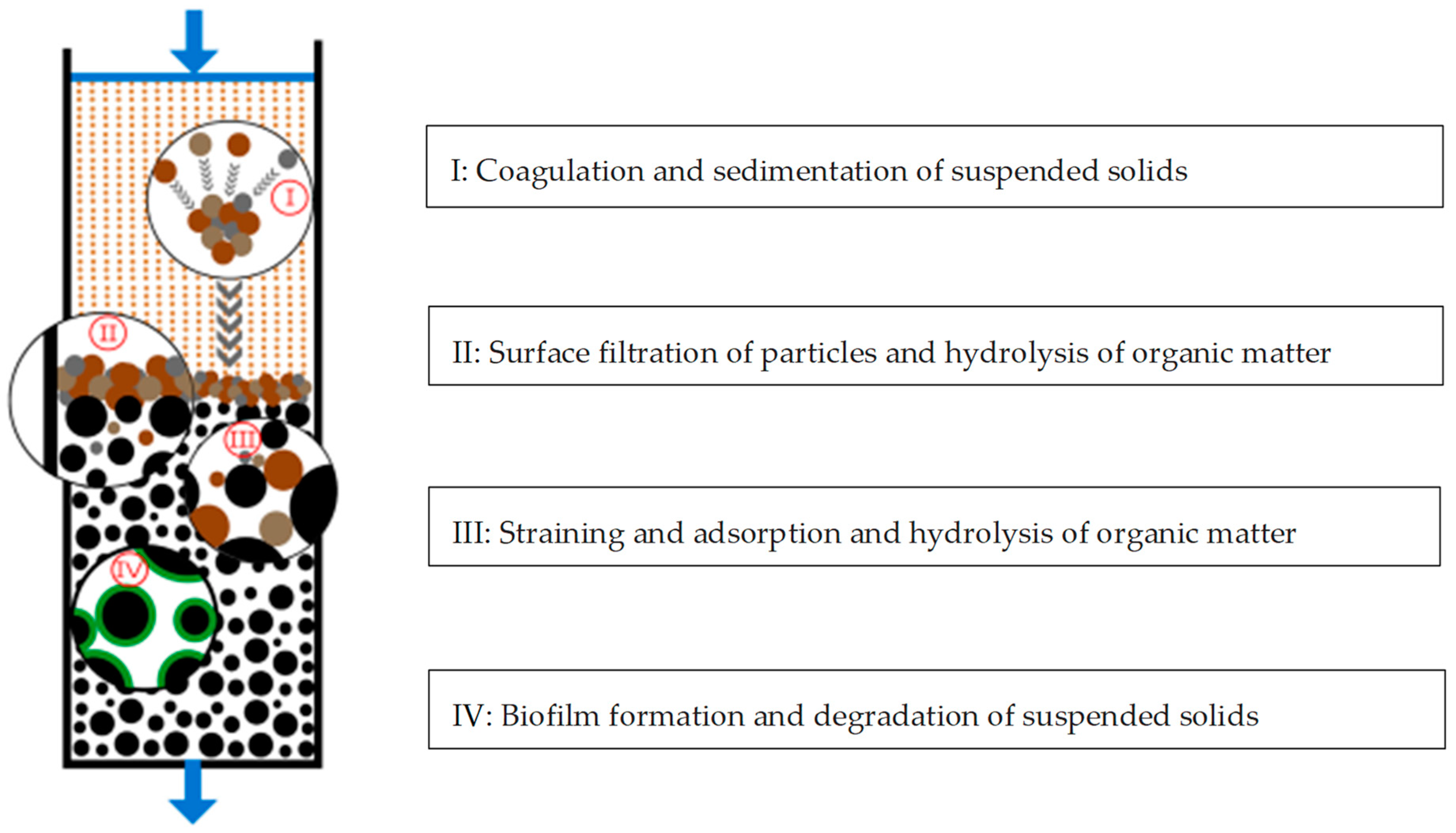
5.2. As a Filtration Support Media
5.3. As a Catalyst during Heterogenous Oxidation
6. Future Perspectives and Environmental Concern of Biochar
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zulfiqar, M.; Samsudin, M.F.R.; Sufian, S. Modelling and optimization of photocatalytic degradation of phenol via Tio2 nanoparticles: An insight into response surface methodology and artificial neural network. J. Photoch. Photobio. A 2019, 384, 112039. [Google Scholar] [CrossRef]
- Ayman, A.I.; Yahya, S.A.; Mohammad, A.A.; Amal, A.M.O. Studying competitive sorption behavior of methylene blue and malachite green using multivariate calibration. Chem. Eng. J. 2014, 240, 554–564. [Google Scholar]
- Bogusz, A.; Oleszczuk, P.; Dobrowolski, R. Application of laboratory prepared and commercially available biochars to adsorption of cadmium, copper and zinc ions from water. Bioresour. Technol. 2015, 196, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Sadat, M.; Hashemi, H.; Eslami, F.; Karimzadeh, R. Organic contaminants removal from industrial wastewater by CTAB treated synthetic zeolite Y. J. Environ. Manag. 2019, 233, 785–792. [Google Scholar]
- Vunain, E.; Masoamphambe, E.F.; Mpeketula, P.M.G.; Monjerezi, M.; Etale, A. Evaluation of coagulating efficiency and water borne pathogens reduction capacity of Moringa oleifera seed powder for treatment of domestic wastewater from Zomba, Malawi. J. Environ. Chem. Eng. 2019, 7, 103–118. [Google Scholar] [CrossRef]
- Guillossou, R.; Roux, J.L.; Mailler, R.; Pereira-Derome, C.S.; Varrault, G.; Bressy, A.; Vulliet, E.; Morlay, C.; Nauleau, F.; Rocher, V.; et al. Influence of dissolved organic matter on the removal of 12 organic micropollutants from wastewater effluent by powdered activated carbon adsorption. Water Res. 2020, 172, 115487. [Google Scholar] [CrossRef]
- Ejraei, A.; Aroon, M.A.; Saravani, A.Z. Wastewater treatment using a hybrid system combining adsorption, photocatalytic degradation and membrane filtration processes. J. Water Process. Eng. 2019, 28, 45–53. [Google Scholar] [CrossRef]
- Nharingo, T.; Moyo, M. Application of Opuntia ficus-indica in bioremediation of wastewaters. A critical review. J. Environ. Manag. 2016, 166, 55–72. [Google Scholar] [CrossRef]
- Cheng, Z.; Fu, F.; Dionysiou, D.D.; Tang, B. Adsorption, oxidation, and reduction behavior of arsenic in the removal of aqueous As(III) by mesoporous Fe/Al bimetallic particles. Water Res. 2016, 96, 22–31. [Google Scholar] [CrossRef]
- He, J.; Li, Y.; Cai, X.; Chen, K.; Zheng, H.; Wang, C.; Zhang, K.; Lin, D.; Kong, L.; Liu, J. Study on the removal of organic micropollutants from aqueous and ethanol solutions by HAP membranes with tunable hydrophilicity and hydrophobicity. Chemosphere 2017, 174, 380–389. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; Glaser, B.; Quicker, P. Technical, economical, and climate-related aspects of biochar production technologies: A literature review. Environ. Sci. Technol. 2011, 45, 9473–9483. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Ali, S.; Qayyum, M.F.; Ibrahim, M.; Ziaurrehman, M.; Abbas, T.; Ok, Y.S. Mechanisms of biochar-mediated alleviation of toxicity of trace elements in plants: A critical review. Environ. Sci. Pollut. Res. 2016, 23, 2230–2248. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.R.; Xie, T.; Dastgheibi, S. Evaluation of biochar as a potential filter media for the removal of mixed contaminants from urban storm water runoff. J. Environ. Eng. 2014, 140. [Google Scholar] [CrossRef]
- Molaei, R. Pathogen and Indicator Organisms Removal in Artificial Greywater Subjected to Aerobic Treatment. Master’s Thesis, Department of Energy and Technology, The Swedish University of Agricultural Science in Uppsala, Uppsala, Sweden, February 2014. [Google Scholar]
- Kaetzl, K.; Lübken, M.; Gehring, T.; Wichern, M. Efficient low-cost anaerobic treatment of wastewater using biochar and woodchip filters. Water 2018, 10, 818. [Google Scholar] [CrossRef]
- Kaetzl, K.; Lübken, M.; Nettmann, E.; Krimmler, S.; Wichern, M. Slow sand filtration of raw wastewater using biochar as an alternative filtration media. Sci. Rep. 2020, 10, 1229. [Google Scholar] [CrossRef]
- Yang, W.; Wang, Z.; Song, S.; Han, J.; Chen, H.; Wang, X.; Sun, R.; Cheng, J. Adsorption of copper(II) and lead(II) from seawater using hydrothermal biochar derived from Enteromorpha. Mar. Pollut. Bull. 2019, 149, 110586. [Google Scholar] [CrossRef]
- Gwenzi, W.; Musarurwa, T.; Nyamugafata, P.; Chaukura, N.; Chaparadza, A.; Mbera, S. Adsorption of Zn2+ and Ni2+ in a binary aqueous solution by biosorbants derived from sawdust and water hyacinth (Eichhornia crassipes). Water Sci. Technol. 2014, 70, 1419–1427. [Google Scholar] [CrossRef]
- Chen, Y.; Lin, Y.-C.; Ho, S.-H.; Zhouc, Y.; Ren, N. Highly efficient adsorption of dyes by biochar derived from pigments-extracted macroalgae pyrolyzed at different temperature. Bioresour. Technol. 2018, 259, 104–110. [Google Scholar] [CrossRef]
- Park, J.-H.; Wang, J.J.; Meng, Y.; Wei, Z.; DeLaune, R.D.; Seo, D.-C. Adsorption/desorption behavior of cationic and anionic dyes by biochars prepared at normal and high pyrolysis temperatures. Colloids Surf. A Physicochem. Eng. Asp. 2019, 572, 274–282. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U., Jr. Organic and inorganic contaminants removal from water with biochar, a renewable, low-cost and sustainable adsorbent: A critical review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Gai, X.; Wang, H.; Liu, J.; Zhai, L.; Liu, S.; Ren, T.; Liu, H. Effects of feedstock and pyrolysis temperature on biochar adsorption of ammonium and nitrate. PLoS ONE 2014, 9, e113888. [Google Scholar] [CrossRef]
- Joyce, S.C.; Suzanne, B.; Ted, M.K.; Joseph, M.; Cliff, T.J.; Brad, J. Initial biochar properties related to the removal of As, Se, Pb, Cd, Cu, Ni, and Zn from an acidic suspension. Chemosphere 2017, 170, 216–224. [Google Scholar]
- Brassard, P.; Godbout, S.; Raghavan, V. Soil biochar amendment as a climate change mitigation tool: Key parameters and mechanisms involved. J. Environ. Manag. 2016, 181, 484–497. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Ok, Y.S.; Choi, G.H.; Park, B.J. Residual perfluorochemicals in the biochar from sewage sludge. Chemosphere 2015, 134, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Li, Y.; Zhang, J.; Wu, S.; Cao, Y.; Liang, P.; Zhang, J.; Wong, M.W.M.H.; Shand, S.; Christie, P. Influence of pyrolysis temperature on properties and environmental safety of heavy metals in biochars derived from municipal sewage sludge. J. Hazard. Mater. 2016, 320, 417–426. [Google Scholar] [CrossRef]
- Enaime, G.; Ennaciri, K.; Ounas, A.; Baçaoui, A.; Seffen, M.; Selmi, T.; Yaacoubi, A. Preparation and characterization of activated carbons from olive wastes by physical and chemical activation: Application to indigo carmine adsorption. J. Mater. Environ. Sci. 2017, 8, 4125–4137. [Google Scholar]
- Selmi, T.; Sanchez-Sanchez, A.; Gadonneix, P.; Jagiello, J.; Seffen, M.; Sammouda, H.; Celzard, A.; Fierro, V. Tetracycline removal with activated carbons produced by hydrothermal carbonisation of Agave americana fibres and mimosa tannin. Ind. Crops Prod. 2018, 115, 146–157. [Google Scholar] [CrossRef]
- Islam, M.A.; Auta, M.; Kabir, G.; Hameed, B.H. A thermogravimetric analysis of the combustion kinetics of karanja (Pongamia pinnata) fruit hulls char. Bioresour. Technol. 2016, 200, 335–341. [Google Scholar] [CrossRef]
- Ahmad, M.; Lee, S.S.; Dou, X.; Mohan, D.; Sung, J.K.; Yang, J.E.; Ok, Y.S. Effects of pyrolysis temperature on soybean stover and peanut shell-derived biochar properties and TCE adsorption in water. Bioresour. Technol. 2012, 118, 536–544. [Google Scholar] [CrossRef]
- Suliman, W.; Harsh, J.B.; Abu-Lail, N.I.; Fortuna, A.M.; Dallmeyer, I.; Garcia-Perez, M. Influence of feedstock source and pyrolysis temperature on biochar bulk and surface properties. Biomass Bioenerg. 2016, 84, 37–48. [Google Scholar] [CrossRef]
- Brewer, C.E. Biochar Characterization and Engineering. Ph.D. Thesis, Iowa State University, Ames, IA, USA, 2012. [Google Scholar] [CrossRef]
- Windeatt, J.H.; Ross, A.B.; Williams, P.T.; Forster, P.M.; Nahil, M.A.; Singh, S. Characteristics of biochars from crop residues: Potential for carbon sequestration and soil amendment. J. Environ. Manag. 2014, 146, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Keiluweit, M.M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic molecular structure of plant biomassderived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.; Downie, A.; Hickey, M.; Jenkins, A.; Lines-Kelly, R.; McClintock, A.; van Zwieten, L. Biochar in Horticulture: Prospects for the Use of Biochar in Australian Horticulture; HAL Report; NSW Trade and Investment: Sydney, Australia, 2012; ISBN 9781742563497.
- Brewer, C.E.; Hu, Y.Y.; Schmidt-Rohr, K.; Loynachan, T.E.; Laird, D.A.; Brown, R.C. Extent of pyrolysis impacts on fast pyrolysis biochar properties. J. Environ. Qual. 2012, 41, 1115–1122. [Google Scholar] [CrossRef]
- Gul, S.; Whalen, J.K.; Thomas, B.W.; Sachdeva, V.; Deng, H. Physico-chemical properties and microbial responses in biochar-amended soils: Mechanisms and future directions. Agric. Ecosyst. Environ. 2015, 206, 46–59. [Google Scholar] [CrossRef]
- Ounas, A.; Aboulkas, A.; Harfi, K.E.; Bacaoui, A.; Yaacoubi, A. Pyrolysis of olive residue and sugar cane bagasse: Non-isothermal thermogravimetric kinetic analysis. Bioresour. Technol. 2011, 102, 11234–11238. [Google Scholar] [CrossRef]
- Inyang, M.; Dickenson, E. The potential role of biochar in the removal of organic and microbial contaminants from potable and reuse water: Review. Chemosphere 2015, 134, 232–240. [Google Scholar] [CrossRef]
- Greenhalf, C.E.; Nowakowski, D.J.; Harms, A.B.; Titiloye, J.O.; Bridgwater, A.V. A comparative study of straw, perennial grasses and hardwoods in terms of fast pyrolysis products. Fuel 2013, 108, 216–230. [Google Scholar] [CrossRef]
- Qian, K.; Kumar, A.; Zhang, H.; Bellmer, D.; Huhnke, R. Recent advances in utilization of biochar. Renew. Sustain. Energy Rev. 2015, 42, 1055–1064. [Google Scholar] [CrossRef]
- Arni, S.A. Comparison of slow and fast pyrolysis for converting biomass into fuel. Renew. Energy 2018, 124, 197–201. [Google Scholar] [CrossRef]
- Duku, M.H.; Gu, S.; BenHagan, E. Biochar production potential in Ghana—A review. Renew. Sust. Energ. Rev. 2011, 15, 3539–3551. [Google Scholar] [CrossRef]
- Libra, J.A.; Ro, K.S.; Kammann, C.; Funke, A.; Berge, N.D.; Neubauer, Y.; Titirici, M.-M.; Fühner, C.; Bens, O.; Kern, J.; et al. Hydrothermal carbonization of biomass residuals: A comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels 2011, 2, 71–106. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenerg. 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Liu, Z.; Quek, A.; Hoekman, S.K.; Balasubramanian, R. Production of solid biochar fuel from waste biomass by hydrothermal carbonization. Fuel 2013, 103, 943–949. [Google Scholar] [CrossRef]
- Mumme, J.; Eckervogt, L.; Pielert, J.; Diakité, M.; Rupp, F.; Kern, J. Hydrothermal carbonization of anaerobically digested maize silage. Bioresour. Technol. 2011, 102, 9255–9560. [Google Scholar] [CrossRef]
- Volpe, M.; Fiori, L. From olive waste to solid biofuel through hydrothermal carbonisation: The role of temperature and solid load on secondary char formation and hydrochar energy properties. J. Anal. Appl. Pyrolysis 2017, 124, 63–72. [Google Scholar] [CrossRef]
- Riling, M.C.; Wagner, M.; Salem, M.; Antunes, P.M.; George, C.; Ramke, H.G.; Titirici, M.M.; Antonietti, M. Material derived from hydrothermal carbonization: Effects on plant growth and arbuscular mycorrhiza. Appl. Soil Ecol. 2010, 45, 238–242. [Google Scholar]
- Cui, X.; Antonietti, M.; Yu, S.H. Structural effects of iron oxide nanoparticles and iron ions on the hydrothermal carbonization of starch and rice carbohydrates. Small 2006, 2, 756–759. [Google Scholar] [CrossRef]
- Zheng, J.; Liu, Z.; Liu, X.; Yan, X.; Li, D.; Chu, W. Facile hydrothermal synthesis and characteristics of B-doped TiO2 hybrid microspheres with higher photo-catalytic activity. J. Alloys Compd. 2011, 509, 3771–3776. [Google Scholar] [CrossRef]
- Danso-Boateng, E.; Holdich, R.G.; Shama, G.; Wheatley, A.D.; Sohail, M.; Martin, S.J. Kinetics of faecal biomass hydrothermal carbonisation for hydrochar production. Appl. Energy 2013, 111, 351–357. [Google Scholar] [CrossRef]
- Zhao, P.; Shen, Y.; Ge, S.; Yoshikawa, K. Energy recycling from sewage sludge by producing solid biofuel with hydrothermal carbonization. Energy Convers. Manag. 2014, 78, 815–821. [Google Scholar] [CrossRef]
- Oliveira, I.; Blöhse, D.; Ramke, H.G. Hydrothermal carbonization of agricultural residues. Bioresour. Technol. 2013, 142, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Funke, A.; Ziegler, F. Hydrothermal carbonization of biomass: A summary and discussion of chemical mechanisms for process engineering. Biofuel. Bioprod. Biorefin. 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Rodríguez-Jasso, R.M.; Fernandes, B.D.; Vicente, A.A.; Teixeira, J.A. Hydrothermal processing, as an alternative for upgrading agriculture residues and marine biomass according to the biorefinery concept: A review. Renew. Sustain. Energy Rev. 2013, 21, 35–51. [Google Scholar] [CrossRef]
- Roman, S.; Nabais, J.M.V.; Laginhas, C.; Ledesma, B.; Gonzalez, J.F. Hydrothermal carbonization as an effective way of densifying the energy content of biomass. Fuel Process. Technol. 2012, 103, 78–83. [Google Scholar] [CrossRef]
- Sabio, E.; Alvarez-Murillo, A.; Roman, S.; Ledesma, B. Conversion of tomato-peel waste into solid fuel by hydrothermal carbonization: Influence of the processing variables. Waste Manag. 2016, 47, 122–132. [Google Scholar] [CrossRef]
- Barin, G.B.; Gimenez, I.F.; Costa, L.P.; Filho, A.G.S.; Barreto, L.S. Hollow carbon nanostructures obtained from hydrothermal carbonization of lignocellulosic biomass. J. Mater. Sci. 2014, 49, 665–672. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A.B. Chemical and structural properties of carbonaceous products obtained by hydrothermal carbonization of saccharides. Chem. Eur. J. 2009, 15, 4195–4203. [Google Scholar] [CrossRef]
- Regmi, P.; Moscoso, J.L.G.; Kumar, S.; Cao, X.; Mao, J.; Schafran, G. Removal of copper and cadmium from aqueous solution using switchgrass biochar produced via hydrothermal carbonization process. J. Environ. Manag. 2012, 109, 61–69. [Google Scholar] [CrossRef]
- Sun, K.; Ro, K.; Guo, M.; Novak, J.; Mashayekhi, H.; Xing, B. Sorption of bisphenol A, 17a ethinyl estradiol and phenanthrene on thermally and hydrothermally produced biochars. Bioresour. Technol. 2011, 102, 5757–5763. [Google Scholar] [CrossRef]
- Wu, J.; Yang, J.; Huang, G.; Xu, C.; Lin, B. Hydrothermal carbonization synthesis of cassava slag biochar with excellent adsorption performance for rhodamine b. J. Clean. Prod. 2020, 251, 119717. [Google Scholar] [CrossRef]
- Zhou, N.; Chen, H.; Xi, J.; Yao, D.; Zhou, Z.; Tian, Y.; Lu, X. Biochars with excellent Pb(II) adsorption property produced from fresh and dehydrated banana peels via hydrothermal carbonization. Bioresour. Technol. 2017, 232, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.-X.; Shen, F.; Qi, X.-H. Preparation of porous carbon directly from hydrothermal carbonization of fructose and phloroglucinol for adsorption of tetracycline. Chinese Chem. Lett. 2017, 28, 960–962. [Google Scholar] [CrossRef]
- Elaigwu, S.E.; Rocher, V.; Kyriakou, G.; Greenway, G.M. Removal of pb2+ and Cd2+ from aqueous solution using chars from pyrolysis and microwave-assisted hydrothermal carbonization of prosopis africana shell. J. Ind. Eng. Chem. 2014, 20, 3467–3473. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, F.-S.; Wu, J. Characterization and application of chars produced from pinewood pyrolysis and hydrothermal treatment. Fuel 2010, 89, 510–514. [Google Scholar] [CrossRef]
- Flora, J.F.R.; Lu, X.; Li, L.; Flora, J.R.V.; Berge, N.D. The effects of alkalinity and acidity of processwater and hydrochar washing on the adsorption of atrazine on hydrothermally produced hydrochar. Chemosphere 2013, 93, 1989–1996. [Google Scholar] [CrossRef]
- Liang, J.; Liu, Y.; Zhang, J. Effect of solution pH on the carbon microsphere synthesized by hydrothermal carbonization. Procedia Environ. Sci. C 2011, 11, 1322–1327. [Google Scholar] [CrossRef][Green Version]
- Reza, M.T.; Rottler, E.; Herklotz, L.; Wirth, B. Hydrothermal carbonization (HTC) of wheat straw: Influence of feedwater ph prepared by acetic acid and potassium hydroxide. Bioresour. Technol. 2015, 182, 336–344. [Google Scholar] [CrossRef]
- Skoulou, V.; Koufodimos, G.; Samaras, Z.; Zabaniotou, A. Low temperature gasification of olive kernels in a 5-kW fluidized bed reactor for H2-rich producer gas. Int. J. Hydrogen Energy 2008, 33, 6515–6524. [Google Scholar] [CrossRef]
- Hernández, J.J.; Lapuerta, M.; Monedero, E. Characterisation of residual char from biomass gasification: Effect of the gasifier operating conditions. J. Clean. Prod. 2016, 138, 83–93. [Google Scholar] [CrossRef]
- García-García, A.; Gregorio, A.; Franco, C.; Pinto, F.; Boavida, D.; Gulyurtlu, I. Unconverted chars obtained during biomass gasification on a pilot-scale gasifier as a source of activated carbon production. Bioresour. Technol. 2003, 88, 27–32. [Google Scholar] [CrossRef]
- Galhetas, M.; Mestre, A.S.; Pinto, M.L.; Gulyurtlu, I.; Lopes, H.; Carvalho, A.P. Chars from gasification of coal and pine activated with K2CO3: Acetaminophen and caffeine adsorption from aqueous solutions. J. Colloid Interf. Sci. 2014, 433, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Rousset, P.; Aguiar, C.; Labbé, N.; Commandré, J.-M. Enhancing the combustible properties of bamboo by torrefaction. Bioresour. Technol. 2011, 102, 8225–8231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Ho, S.H.; Chen, W.H.; Fu, Y.J.; Chang, J.S.; Bi, X.T. Oxidative torrefaction of biomass nutshells: Evaluations of energy efficiency as well as biochar transportation and storage. Appl. Energy 2019, 235, 428–441. [Google Scholar] [CrossRef]
- Li, L.; Yang, M.; Lu, Q.; Zhu, W.; Ma, H.; Dai, L. Oxygen-rich biochar from torrefaction: A versatile adsorbent for water pollution control. Bioresour. Technol. 2019, 294, 122142. [Google Scholar] [CrossRef]
- Salapa, I.; Haralampous, P.; Giakoumakis, G.; Nazos, A.; Sidiras, D. Torrefaction of barley straw for the co-production of energy and adsorbent materials. In Proceedings of the 4th World Congress on Mechanical, Chemical, and Material Engineering (MCM’18), Madrid, Spain, 16–18 August 2018. [Google Scholar]
- Michal, K.; Isabel, H.; Thomas, D.B.; Barbara, C.; Jadwiga, S.Z.; Patryk, O. Activated biochars reduce the exposure of polycyclic aromatic hydrocarbons in industrially contaminated soils. Chem. Eng. J. 2017, 310, 33–40. [Google Scholar]
- Vithanage, M.; Rajapaksha, A.U.; Dou, X.; Bolan, N.S.; Yang, J.E.; Ok, Y.S. Surface complexation modeling and spectroscopic evidence of antimony adsorption on ironoxide-rich red earth soils. J. Colloid Interf. Sci. 2013, 406, 217–224. [Google Scholar] [CrossRef]
- Liu, W.J.; Jiang, H.; Yu, H.Q. Development of biochar-based functional materials: Toward a sustainable platform carbon material. Chem. Rev. 2015, 115, 12251–12285. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Xing, Z.J.; Duan, Z.K.; Li, M.; Wang, Y. Effects of steam activation on the pore structure and surface chemistry of activated carbon derived from bamboo waste. Appl. Surf. Sci. 2014, 315, 279–286. [Google Scholar] [CrossRef]
- Xiong, Z.; Zhang, S.; Yang, H.; Shi, T.; Chen, Y.; Chen, H. Influence of NH3/CO2 modification on the characteristic of biochar and the CO2 capture. Bioenergy Res. 2013, 6, 1147–1153. [Google Scholar] [CrossRef]
- Zhang, J.; Fan, L.; Shao, L.; He, P. The use of biochar-amended composting to improve the humifianion and degradation of sewage sludge. Bioresour. Technol. 2014, 168, 252–258. [Google Scholar] [CrossRef]
- Peng, P.; Lang, Y.H.; Wang, X.M. Adsorption behavior and mechanism of pentachlorophenol on reed biochars: pH effect, pyrolysis temperature, hydrochloric acid treatment and isotherms. Ecol. Eng. 2016, 90, 225–233. [Google Scholar] [CrossRef]
- Mahmoud, D.K.; Salleh, M.A.M.; Karim, W.A.W.A.; Idris, A.; Abidin, Z.Z. Batch adsorption of basic dye using acid treated kenaf fibre char: Equilibrium, kinetic and thermodynamic studies. Chem. Eng. J. 2012, 181–182, 449–457. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, Y.; Liu, S.; Liu, H.; Zeng, G.; Tan, X.; Yang, C.; Ding, Y.; Yan, Z.; Cai, X. Sorption performance and mechanisms of arsenic(V) removal by magnetic gelatin-modified biochar. Chem. Eng. J. 2017, 314, 223–231. [Google Scholar] [CrossRef]
- Vithanage, M.; Rajapaksha, A.U.; Zhang, M.; Thielebruhn, S.; Lee, S.S.; Ok, Y.S. Acid activated biochar increased sulfamethazine retention in soils. Environ. Sci. Pollut. Control. Ser. 2015, 22, 2175–2186. [Google Scholar] [CrossRef]
- Jing, X.R.; Wang, Y.Y.; Liu, W.J.; Wang, Y.K.; Jiang, H. Enhanced adsorption performance of tetracycline in aqueous solutions by methanol-modified biochar. Chem. Eng. J. 2014, 248, 168–174. [Google Scholar] [CrossRef]
- Shen, Y.; Zhang, N. Facile synthesis of porous carbons from silica-rich rice husk char for volatile organic compounds (VOCs) sorption. Bioresour. Technol. 2019, 282, 294–300. [Google Scholar] [CrossRef]
- Cazetta, A.L.; Vargas, A.M.M.; Nogami, E.M.; Kunita, M.H.; Guilherme, M.R.; Martins, A.C.; Silva, T.L.; Moraes, J.C.G.; Almeida, V.C. NaOH-activated carbon of high surface area produced from coconut shell: Kinetics and equilibrium studies from the methylene blue adsorption. Chem. Eng. J. 2011, 174, 117–125. [Google Scholar] [CrossRef]
- Iriarte-Velasco, U.; Ayastuy, J.; Zudaire, L.; Sierra, I. An insight into the reactions occurring during the chemical activation of bone char. Chem. Eng. J. 2014, 251, 217–227. [Google Scholar] [CrossRef]
- Xue, Y.W.; Gao, B.; Yao, Y.; Inyang, M.; Zhang, M.; Zimmerman, A.R.; Ro, K.S. Hydrogen peroxide modification enhances the ability of biochar (hydrochar) produced from hydrothermal carbonization of peanut hull to remove aqueous heavy metals: Batch and column tests. Chem. Eng. J. 2012, 200, 673–680. [Google Scholar] [CrossRef]
- Tan, X.; Liu, S.; Liu, Y.; Gu, Y.; Zeng, G.; Hu, X.; Wang, X.; Liu, S.; Jiang, L. Biochar as potential sustainable precursors for activated carbon production: Multiple applications in environmental protection and energy storage. Bioresour. Technol. 2017, 227, 359–372. [Google Scholar] [CrossRef]
- Wang, H.; Gao, B.; Wang, S.; Fang, J.; Xue, Y.; Yang, K. Removal of Pb(II), Cu(II), and Cd(II) from aqueous solutions by biochar derived from KMnO4 treated hickory wood. Bioresour. Technol. 2015, 19, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Thang, P.Q.; Jitae, K.; Giang, N.M.V.B.L.; Huong, P.T. Potential application of chicken manure biochar towards toxic phenol and 2, 4-dinitrophenol in wastewaters. J. Environ. Manag. 2019, 251, 109556. [Google Scholar] [CrossRef]
- Mohammed, N.A.S.; Abu-Zurayk, R.A.; Hamadneh, I.; Al-Dujaili, A.H. Phenol adsorption on biochar prepared from the pine fruit shells: Equilibrium, kinetic and thermodynamics studies. J. Environ. Manag. 2018, 226, 377–385. [Google Scholar] [CrossRef]
- Oh, S.-Y.; Seo, Y.-D. Factors affecting the sorption of halogenated phenols onto polymer/ biomass derived biochar: Effects of pH, hydrophobicity, and deprotonation. J. Environ. Manag. 2019, 232, 145–152. [Google Scholar] [CrossRef]
- Qiu, Y.; Zheng, Z.; Zhou, Z.; Sheng, G.D. Effectiveness and mechanisms of dye adsorption on a strawbased biochar. Bioresour. Technol. 2009, 100, 5348–5351. [Google Scholar] [CrossRef]
- Ferreira, C.I.A.; Calisto, V.; Otero, M.; Nadais, H.; Esteves, V.I. Comparative adsorption evaluation of biochars from paper mill sludge with commercial activated carbon for the removal of fish anaesthetics from water in recirculating aquaculture systems. Aquac. Eng. 2016, 74, 76–83. [Google Scholar] [CrossRef]
- Jiang, S.; Huang, L.; Nguyen, T.A.H.; Ok, Y.S.; Rudolph, V.; Yang, H.; Zhang, D. Copper and zinc adsorption by softwood and hardwood biochars under elevated sulphate- induced salinity and acidic pH conditions. Chemosphere 2016, 142, 64–71. [Google Scholar] [CrossRef]
- Jin, H.; Hanif, M.; Capareda, S.; Chang, Z.; Huang, H.; Ai, Y. Copper (II) removal potential from aqueous solution by pyrolysis biochar derived from anaerobically digested algae-dairy manure and effect of KOH activation. J. Environ. Chem. Eng. 2016, 4, 365–372. [Google Scholar] [CrossRef]
- Ni, B.-J.; Huang, Q.-S.; Wang, C.; Ni, T.-Y.; Sun, J.; Wei, W. Competitive adsorption of heavy metals in aqueous solution onto biochar derived from anaerobically digested sludge. Chemosphere 2019, 219, 351–357. [Google Scholar] [CrossRef]
- Park, J.; Ok, Y.S.; Kim, S.; Cho, J.; Heo, J.; Delaune, R.D.; Seo, D. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, Y.; Wang, H.; Lu, W.; Zhou, Z.; Zhang, Y.; Ren, L. Influence of pyrolysis temperature on characteristics and heavy metal adsorptive performance of biochar derived from municipal sewage sludge. Bioresour. Technol. 2014, 164, 47–54. [Google Scholar] [CrossRef]
- Zhao, M.; Dai, Y.; Zhang, M.; Feng, C.; Qin, B.; Zhang, W.; Zhao, N.; Li, Y.; Ni, Z.; Xu, Z.; et al. Mechanisms of Pb and/or Zn adsorption by different biochars: Biochar characteristics, stability, and binding energies. Sci. Total Environ. 2020, 717, 136894. [Google Scholar] [CrossRef]
- Jingjian, P.; Jun, J.; Renkou, X. Adsorption of Cr(III) from acidic solutions by crop straw derived biochars. J. Environ. Sci. China 2013, 25, 1957–1965. [Google Scholar]
- Park, J.; Wang, J.; Kim, S.; Cho, J.; Kang, S.; Delaune, R.; Han, K.; Seo, D. Recycling of rice straw through pyrolysis and its adsorption behaviors for cu and zn ions in aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2017, 533, 330–337. [Google Scholar] [CrossRef]
- Xu, X.; Cao, X.; Zhao, L.; Zhou, H.; Luo, Q. Interaction of organic and inorganic fractions of biochar with pb(II) ion: Further elucidation of mechanisms for pb(II) removal by biochar. RSC Adv. 2014, 4, 44930. [Google Scholar] [CrossRef]
- Liu, L.; Fan, S. Removal of cadmium in aqueous solution using wheat straw biochar: Effect of minerals and mechanism. Environ. Sci. Pollut. Res. 2018, 25, 8688–8700. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Krukowska, J.; Thomas, P. Comparison of sorption and desorption studies of heavy metal ions from biochar and commercial active carbon. Chem. Eng. J. 2017, 307, 353–363. [Google Scholar] [CrossRef]
- Fang, J.; Gao, B.; Zimmerman, A.R.; Ro, K.S.; Chen, J. Physically (CO2) activated hydrochars from hickory and peanut hull: Preparation, characterization, and sorption of methylene blue, lead, copper, and cadmium. RSC Adv. 2016, 6, 24906–24911. [Google Scholar] [CrossRef]
- Samsuri, A.W.; Sadegh-Zadeh, F.; Seh-Bardan, B.J. Adsorption of As(III) and As(V) by Fe coated biochars and biochars produced from empty fruit bunch and rice husk. J. Environ. Chem. Eng. 2013, 1, 981–988. [Google Scholar] [CrossRef]
- Uchimiya, M.; Wartelle, L.H.; Klasson, K.T.; Fortier, C.A.; Lima, I.M. Influence of pyrolysis temperature on biochar property and function as a heavy metal sorbent in soil. J. Agric. Food Chem. 2011, 59, 2501–2510. [Google Scholar] [CrossRef] [PubMed]
- Gwenzi, W.; Nyamadzawo, G. Hydrological impacts of urbanization and urban roof water harvesting in water-limited catchments: A review. Environ. Process. 2014, 1, 573–593. [Google Scholar] [CrossRef]
- Rosales, E.; Meijide, J.; Pazos, M.; Sanroman, M.A. Challenges and recent advances in biochar as lowcost biosorbent: From batch assays to continuous-flow systems. Bioresour. Technol. 2017, 246, 176–192. [Google Scholar] [CrossRef]
- Pignatello, J.J. Interactions of Anthropogenic Organic Chemicals with Natural Organic Matter and Black Carbon in Environmental Particles. Biophysico-Chemical Processes of Anthropogenic Organic Compounds in Environmental Systems; John Wiley Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 1–50. [Google Scholar]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Qambrani, N.A.; Rahman, M.M.; Won, S.; Shim, S.; Ra, C. Biochar properties and ecofriendly applications for climate change mitigation, waste management, and wastewater treatment: A review. Renew. Sustain. Energy Rev. 2017, 79, 255–273. [Google Scholar] [CrossRef]
- Lu, H.; Zhang, W.; Yang, Y.; Huang, X.; Wang, S.; Qiu, R. Relative distribution of Pb2+ sorption mechanisms by sludge-derived biochar. Water Res. 2012, 46, 854–862. [Google Scholar] [CrossRef]
- Zazycki, M.A.; Godinho, M.; Perondi, D.; Foletto, E.L.; Collazzo, G.C.; Dotto, G.L. New biochar from pecan nutshells as an alternative adsorbent for removing reactive red 141 from aqueous solutions. J. Clean. Prod. 2018, 171, 57–65. [Google Scholar] [CrossRef]
- Mohamed, E.M.; Gehan, M.N.; Nabila, M.E.M.; Heba, I.B.; Sandeep, K.; Tarek, M.A.F. Kinetics, isotherm, and thermodynamic studies of the adsorption of reactive red 195 A dye from water by modified switchgrass biochar adsorbent. J. Ind. Eng. Chem. 2016, 37, 156–167. [Google Scholar]
- Sun, P.; Hui, C.; Khan, R.A.; Du, J.; Zhang, Q.; Zhao, Y.H. Efficient removal of crystal violet using Fe3O4-coated biochar: The role of the Fe3O4 nanoparticles and modelling study their adsorption behavior. Sci. Rep. 2015, 5, 12638. [Google Scholar] [CrossRef]
- Thines, K.R.; Abdullah, E.C.; Mubarak, N.M. Effect of process parameters for production of microporous magnetic biochar derived from agriculture waste biomass. Microporous Mesoporous Mater. 2017, 253, 29–39. [Google Scholar] [CrossRef]
- Yakout, S.M. Physicochemical characteristics of biochar produced from rice straw at different pyrolysis temperature for soil amendment and removal of organics. Proc. Natl. Acad. Sci. India A 2017, 87, 207–214. [Google Scholar] [CrossRef]
- Jian, X.; Zhuang, X.; Li, B.; Xu, X.; Wei, Z.; Song, Y.; Jiang, E. Comparison of characterization and adsorption of biochars produced from hydrothermal carbonization and pyrolysis. Environ. Technol. Innov. 2018, 10, 27–35. [Google Scholar] [CrossRef]
- Huff, M.D.; Marshall, S.; Saeed, H.A.; Lee, J.W. Surface oxygenation of biochar through ozonization for dramatically enhancing cation exchange capacity. Bioresour. Bioprocess. 2018, 5, 18. [Google Scholar] [CrossRef]
- Akl, M.A.A.; Dawy, M.B.; Serage, A.A. Efficient removal of phenol from water samples using sugarcane bagasse based activated carbon. J. Anal. Bioanal. Tech. 2014, 5, 1000189. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, F.-S. Removal of copper (II) and phenol from aqueous solution using porous carbons derived from hydrothermal chars. Desalination 2011, 267, 101–106. [Google Scholar] [CrossRef]
- Li, Y.; Tsend, N.; Li, T.; Liu, H.; Yang, R.; Gai, X.; Wang, H.; Shan, S. Microwave assisted hydrothermal preparation of rice straw hydrochars for adsorption of organics and heavy metals. Bioresour. Technol. 2019, 273, 136–143. [Google Scholar] [CrossRef]
- Li, Y.; Meas, A.; Shan, S.; Yang, R.; Gai, X. Production and optimization of bamboo hydrochars for adsorption of congo red and 2-naphthol. Bioresour. Technol. 2016, 207, 379–386. [Google Scholar] [CrossRef]
- Marfíl, A.P.; Ocampo-Pérez, R.; Collins-Martínez, V.H.; Flores-Vélez, L.M.; Gonzalez-Garcia, R.; Medellín-Castillo, N.A.; Labrada-Delgado, G.J. Synthesis and characterization of hydrochar from industrial capsicum annuum seeds and its application for the adsorptive removal of methylene blue from water. Environ. Res. 2020, 184, 109334. [Google Scholar] [CrossRef]
- Duan, X.; Hong, W.; Srinivasakannan, C.; Wang, X. Hydrochar silicate composite sorbent via simple hydrothermal carbonization and its application to methylene blue removal. Mater. Res. Express 2018, 6, 035601. [Google Scholar] [CrossRef]
- Nguyen, D.H.; Tran, H.N.; Chao, H.P.; Lin, C.C. Effect of nitric acid oxidation on the surface of hydrochars to sorb methylene blue: An adsorption mechanism comparison. Adsorpt. Sci. Technol. 2019, 37, 607–622. [Google Scholar] [CrossRef]
- Kılıc, M.; Mutlu, Ç.K.Ö.Ç.; Pütün, A.E. Adsorption of heavy metal ions from aqueous solutions by bio-char, a by-product of pyrolysis. Appl. Surf. Sci. 2013, 283, 856–862. [Google Scholar] [CrossRef]
- Wu, W.; Li, J.; Lan, T.; Müller, K.; Niazi, N.K.; Chen, X.; Xu, S.; Zheng, L.; Chu, Y.; Li, J.; et al. Unraveling sorption of lead in aqueous solutions by chemically modified biochar derived from coconut fiber: A microscopic and spectroscopic investigation. Sci. Total Environ. 2017, 576, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Shakya, A.; Agarwal, T. Removal of Cr(VI) from water using pineapple peel derived biochars: Adsorption potential and re-usability assessment. J. Mol. Liq. 2019, 293, 111497. [Google Scholar] [CrossRef]
- Arán, D.; Antelo, J.; Fiol, S.; Mac’ıas, F. Influence of feedstock on the copper removal capacity of wastederived biochars. Bioresour. Technol. 2016, 212, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Gao, B.; Zimmerman, A.R.; Li, Y.; Ma, L.; Harris, W.G.; Migliaccio, K.W. Physicochemical and sorptive properties of biochars derived from woody and herbaceous biomass. Chemosphere 2015, 134, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Bogusz, A.; Nowak, K.; Stefaniuk, M.; Dobrowolski, R.; Oleszczuk, P. Synthesis of biochar from residues after biogas production with respect to cadmium and nickel removal from wastewater. J. Environ. Manag. 2017, 201, 268–276. [Google Scholar] [CrossRef]
- Shah, G.M.; Nasir, M.; Imran, M.; Bakhat, H.F.; Rabbani, F.; Sajjad, M.; Farooq, A.B.U.; Ahmad, S.; Song, L. Biosorption potential of natural, pyrolysed and acid-assisted pyrolysed sugarcane bagasse for the removal of lead from contaminated water. PeerJ 2018, 6, e5672. [Google Scholar] [CrossRef]
- Abdelhafez, A.A.; Li, J. Removal of pb(II) from aqueous solution by using biochars derived from sugar cane bagasse and orange peel. J. Taiwan. Inst. Chem. Eng. 2016, 61, 367–375. [Google Scholar] [CrossRef]
- Deng, J.; Li, X.; Wei, X.; Liu, Y.; Liang, J.; Tanga, N.; Song, B.; Chen, X.; Cheng, X. Sulfamic acid modified hydrochar derived from sawdust for removal of benzotriazole and Cu(II) from aqueous solution: Adsorption behavior and mechanism. Bioresour. Technol. 2019, 290, 121765. [Google Scholar] [CrossRef]
- Runtti, H.; Tuomikoski, S.; Kangas, T.; Lassi, U.; Kuokkanena, T.; Rämö, J. Chemically activated carbon residue from biomass gasification as a sorbent for iron(II), copper(II) and nickel(II) ions. J. Water Process. Eng. 2014, 4, 12–24. [Google Scholar] [CrossRef]
- Lowell, S.; Shields, J.E.; Thomas, M.A.; Thommes, M. Characterisation of Porus Solids and Powders: Surface Area, Pore Size and Density, 4th ed.; Springer Science & Business Media: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Zabaniotou, A.; Stavropoulos, G.; Skoulou, V. Activated carbon from olive kernels in a two-stage process: Industrial improvement. Bioresour. Technol. 2008, 99, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Mosher, K. The Impact of Pore Size on Methane and CO2 Adsorption in Carbon. Master’s Thesis, Stanford University, Stanford, CA, USA, June 2011. [Google Scholar]
- Zhou, Z.; Shi, D.; Qiu, Y.; Sheng, G.D. Sorptive domains of pine chars as probed by benzene and nitrobenzene. Environ. Pollut. 2010, 158, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Daifullah, A.A.M.; Girgis, B.S. Removal of some substituted phenols by activated carbon obtained from agricultural waste. Water Res. 1998, 32, 1169–1177. [Google Scholar] [CrossRef]
- Gascó, G.; Paz-Ferreiro, J.; Álvarez, M.L.; Saa, A.; Méndez, A. Biochars and hydrochars prepared by pyrolysis and hydrothermal carbonisation of pig manure. J. Waste Manag. 2018, 79, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Castilla, C. Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon 2004, 42, 83–94. [Google Scholar] [CrossRef]
- Li, L.; Quinlivan, P.A.; Knappe, D.R.U. Effects of activated carbon surface chemistry and pore structure on the adsorption of organic contaminants from aqueous solution. Carbon 2002, 40, 2085–2100. [Google Scholar] [CrossRef]
- Kumar, P.S.; Ramalingam, S.; Sathishkumar, K. Removal of methylene blue dye from aqueous solution by activated carbon prepared from cashew nut shell as a new low-cost adsorbent. Korean J. Chem. Eng. 2011, 28, 149–155. [Google Scholar] [CrossRef]
- Kumar, S.; Loganathan, V.A.; Gupta, R.B.; Barnett, M.O. An assessment of U(VI) removal from groundwater using biochar produced from hydrothermal carbonization. J. Environ. Manag. 2011, 92, 2504–2512. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Sorption I: General introduction and sorption processes involving organic matter. In Environmental Organic Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; pp. 275–330. [Google Scholar]
- Kizito, S.; Wu, S.; Kirui, W.K.; Lei, Q.L.M.; Bah, H.; Dong, R. Evaluation of slow pyrolyzed wood and rice husks biochar for adsorption of ammonium nitrogen from piggery manure anaerobic digestate slurry. Sci. Total Environ. 2015, 505, 102–112. [Google Scholar] [CrossRef]
- Hu, X.; Zhang, X.; Ngo, H.H.; Guo, W.; Wen, H.; Li, C.; Zhang, Y.; Ma, C. Comparison study on the ammonium adsorption of the biochars derived from different kinds of fruit peel. Sci. Total Environ. 2020, 707, 135544. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.T.; Chen, H.R. Adsorption kinetics of herbicide paraquat in aqueous solution onto a low-cost adsorbent, swine-manure-derived biochar. Int. J. Environ. Sci. Technol. 2013, 10, 1349–1356. [Google Scholar] [CrossRef]
- Linville, J.L.; Shen, Y.; Ignacio-de Leon, P.A.; Schoene, R.P.; Urgun-Demirtas, M. In situ biogas upgrading during anaerobic digestion of food waste amended with walnut shell biochar at bench scale. Waste Manag. Res. 2017, 35, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Murray, B.C.; Galik, C.S.; Vegh, T. Biogas in the United States: An Assessment of Market Potential in a Carbon-Constrained Future; Nicholas Institute for Environmental Policy Solutions, Duke University: Durham, NC, USA, 2014. [Google Scholar]
- Fagbohungbe, M.O.; Herbert, B.M.J.; Hurst, L.; Ibeto, C.N.; Li, H.; Usmani, S.Q.; Semple, K.T. The challenges of anaerobic digestion and the role of biochar in optimizing anaerobic digestion. J. Waste Manag. 2017, 61, 236–249. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Lu, F.; Shao, L.; He, P. Application of eco-compatible biochar in anaerobic digestion to relieve acid stress and promote the selective colonization of functional microbes. Water Res. 2015, 68, 710–718. [Google Scholar] [CrossRef] [PubMed]
- Lü, F.; Luo, C.; Shao, L.; He, P. Biochar alleviates combined stress of ammonium and acids by firstly enriching methanosaeta and then methanosarcina. Water Res. 2016, 90, 34–43. [Google Scholar] [CrossRef]
- Torri, C.; Fabbri, D. Biochar enables anaerobic digestion of aqueous phase from intermediate pyrolysis of biomass. Bioresour. Technol. 2014, 172, 335–341. [Google Scholar] [CrossRef]
- Sunyoto, N.M.S.; Zhu, M.; Zhang, Z.; Zhang, D. Effect of biochar addition on hydrogen and methane production in two-phase anaerobic digestion of aqueous carbohydrates food waste. Bioresour. Technol. 2016, 219, 29–36. [Google Scholar] [CrossRef]
- Mumme, J.; Srocke, F.; Heeg, K.; Werner, M. Use of biochars in anaerobic digestion. Bioresour. Technol. 2014, 164, 189–197. [Google Scholar] [CrossRef]
- Cao, G.-L.; Guo, W.-Q.; Wang, A.-J.; Zhao, L.; Xu, C.-J.; Zhao, Q.-L.; Ren, N.-Q. Enhanced cellulosic hydrogen production from lime-treated cornstalk wastes using thermophilic anaerobic microflora. Int. J. Hydrogen Energy 2012, 37, 13161–13166. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Q.; Zheng, P.; Wang, Y. Anaerobic digestion of food waste stabilized by lime mud from papermaking process. Bioresour. Technol. 2014, 170, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.-H.; Xu, R.-K.; Zhang, H. The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour. Technol. 2011, 102, 3488–3497. [Google Scholar] [CrossRef] [PubMed]
- Lü, F.; Bize, A.; Guillot, A.; Monnet, V.; Madigou, C.; Chapleur, O.; Mazéas, L.; He, P.; Bouchez, T. Metaproteomics of cellulose methanisation under thermophilic conditions reveals a surprisingly high proteolytic activity. ISME J. 2014, 8, 88–102. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Zhang, Y.; Woodard, T.L.; Nevin, K.P.; Lovley, D.R. Enhancing syntrophic metabolism in up-flow anaerobic sludge blanket reactors with conductive carbon materials. Bioresour. Technol. 2015, 191, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Enaime, G.; Baçaoui, A.; Yaacoubi, A.; Berzio, S.; Wichern, M.; Lübken, M. Packed-bed biofilm reactor for semi-continuous anaerobic digestion of olive mill wastewater: Performances and COD mass balance analysis. Environ. Technol. 2019, 6, 1–13. [Google Scholar] [CrossRef]
- Gehring, T.; Klang, J.; Niedermayr, A.; Berzio, S.; Immenhauser, A.; Klocke, M.; Wichern, M.; Lübken, M. Determination of methanogenic pathways through carbon isotope analysis for the two-stage anaerobic digestion of high-solids substrates. Environ. Sci. Technol. 2015, 49, 4705–4714. [Google Scholar] [CrossRef]
- Gehring, T.; Niedermayr, A.; Berzio, S.; Immenhauser, A.; Wichern, M.; Lübken, M. Determination of the fractions of syntrophically oxidized acetate in a mesophilic methanogenic reactor trough an 12C and 13C isotope-based kinetic model. Water Res. 2016, 102, 362–373. [Google Scholar] [CrossRef]
- VandenBerg, L.; Kennedy, K.J.; Samson, R. Anaerobic downflow stationary fixed film reactor: Performance under steady-state and non-steady conditions. Water Sci. Technol. 1985, 17, 89–102. [Google Scholar] [CrossRef]
- Zheng, H.; Li, D.; Stanislaus, M.S.; Zhang, N.; Zhu, Q.; Hu, X.; Yang, Y. Development of a bio-zeolite fixed-bed bioreactor for mitigating ammonia inhibition of anaerobic digestion with extremely high ammonium concentration livestock waste. Chem. Eng. J. 2015, 280, 106–114. [Google Scholar] [CrossRef]
- Chauhan, A.; Ogram, A. Evaluation of support matrices for immobilization of anaerobic consortia for efficient carbon cycling in waste regeneration. Biochem. Biophys. Res. Commun. 2005, 327, 884–893. [Google Scholar] [CrossRef]
- Bertin, L.; Lampis, S.; Todaro, D.; Scoma, A.; Vallini, G.; Marchetti, L.; Majone, M.; Fava, F. Anaerobic acidogenic digestion of olive mill wastewaters in biofilm reactors packed with ceramic filters or granular activated carbon. Water Res. 2010, 44, 4537–4549. [Google Scholar] [CrossRef] [PubMed]
- Enaime, G.; Nettmann, E.; Berzio, S.; Baçaoui, A.; Yaacoubi, A.; Wichern, M.; Gehring, T.; Lübken, M. Performance and microbial analysis during long-term anaerobic digestion of olive mill wastewater in a packed-bed biofilm reactor. J. Chem. Technol. Biotechnol. 2020, 95, 850–861. [Google Scholar] [CrossRef]
- Sunyoto, N.M.S.; Zhu, M.; Zhang, Z.; Zhang, D. Effect of biochar addition and initial pH on hydrogen production from the first phase of two-phase anaerobic digestion of carbohydrates food waste. Energy Procedia 2017, 105, 379–384. [Google Scholar] [CrossRef]
- Li, Y.; McCarthy, D.T.; Deletic, A. Escherichia coli removal in copper-zeolite-integrated stormwater biofilters: Effect of vegetation, operational time, intermittent drying weather. Ecol. Eng. 2016, 90, 234–243. [Google Scholar] [CrossRef]
- Guest, R.M.; Schang, C.; Deletic, A.; McCarthy, D.T. Zinc-sulphate-heptahydrate coated activated carbon for microbe removal from stormwater. Water Sci. Technol. 2012, 66, 1582–1589. [Google Scholar] [CrossRef]
- Chandrasena, G.I.; Deletic, A.; Ellerton, J.; McCarthy, D.T. Evaluating escherichia coli removal performance in stormwater biofilters: A laboratory-scale study. Water Sci. Technol. 2012, 66, 1132–1138. [Google Scholar] [CrossRef]
- Kaetzl, K.; Lübken, M.; Uzuna, G.; Gehring, T.; Nettmann, E.; Stenchly, K.; Wichern, M. On-farm wastewater treatment using biochar from local agroresidues reduces pathogens from irrigation water for safer food production in developing countries. Sci. Total Environ. 2019, 682, 601–610. [Google Scholar] [CrossRef]
- Chen, G.; Walker, S.L. Fecal indicator bacteria transport and deposition in saturated and unsaturated porous media. Environ. Sci. Technol. 2012, 46, 8782–8790. [Google Scholar] [CrossRef]
- Stevik, T.K.; Aa, K.; Ausland, G.; Hanssen, J.F. Retention and removal of pathogenic bacteria in wastewater percolating through porous media: A review. Water Res. 2004, 38, 1355–1367. [Google Scholar] [CrossRef]
- Werner, S.; Katzl, K.; Wichern, M.; Buerkert, A.; Steiner, C.; Marschner, B. Agronomic benefits of biochar as a soil amendment after its use as waste water filtration medium. Environ. Pollut. 2018, 233, 561–568. [Google Scholar] [CrossRef]
- Dalahmeh, S.; Ahrens, L.; Gros, M.; Wiberg, K.; Pell, M. Potential of biochar filters for onsite sewage treatment: Adsorption and biological degradation of pharmaceuticals in laboratory filters with active, inactive and no biofilm. Sci. Total Environ. 2018, 612, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Perez-Mercado, L.F.; Lalander, C.; Berger, C.; Dalahmeh, S.S. Potential of biochar filters for onsite wastewater treatment: Effects of biochar type, physical properties and operating conditions. Water 2018, 10, 1835. [Google Scholar] [CrossRef]
- Lau, A.Y.T.; Tsang, D.C.W.; Graham, N.; Ok, Y.; Yang, X.; Li, X. Surface-modified biochar in a bioretention system for Escherichia coli removal from stormwater. Chemosphere 2017, 169, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.K.; Boehm, A.B. Escherichia coli removal in biochar-augmented biofilter: Effect of infiltration rate, initial bacterial concentration, biochar particle size, and presence of compost. Environ. Sci. Technol. 2014, 48, 11535–11542. [Google Scholar] [CrossRef] [PubMed]
- Moussavi, G.; Khosravi, R. Preparation and characterization of a biochar from pistachio hull biomass and its catalytic potential for ozonation of water recalcitrant contaminants. Bioresour. Technol. 2012, 119, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Zhu, R.; Liu, J.; Zhou, Q.; Zhu, J.; Liang, X.; Xi, Y.; He, H. Fullerol modification ferrihydrite for the degradation of acid red 18 under simulated sunlight irradiation. J. Mol. Catal. A Chem. 2016, 424, 393–401. [Google Scholar] [CrossRef]
- Qin, Y.; Li, G.; Gao, Y.; Zhang, L.; Ok, Y.S.; An, T. Persistent free radicals in carbon-based materials on transformation of refractory organic contaminants (rocs) in water: A critical review. Water Res. 2018, 137, 130–143. [Google Scholar] [CrossRef]
- Yang, S.; Xiao, T.; Zhang, J.; Chen, Y.; Li, L. Activated carbon fiber as heterogeneous catalyst of peroxymonosulfate activation for efficient degradation of acid orange 7 in aqueous solution. Sep. Purif. Technol. 2015, 143, 19–26. [Google Scholar] [CrossRef]
- Fang, G.; Zhu, C.; Dionysiou, D.D.; Gao, J.; Zhou, D. Mechanism of hydroxyl radical generation from biochar suspensions: Implications to diethyl phthalate degradation. Bioresour. Technol. 2015, 176, 210–217. [Google Scholar] [CrossRef]
- Qin, Y.; Zhang, L.; An, T. Hydrothermal carbon-mediated fenton-like reaction mechanism in the degradation of alachlor: Direct electron transfer from hydrothermal carbon to Fe(III). ACS Appl. Mater. Interfaces 2017, 9, 17115–17124. [Google Scholar] [CrossRef]
- Izghri, Z.; Enaime, G.; Louarrat, M.; Gaini, L.E.; Baçaoui, A.; Yaacoubi, A. Novel catalyst from two-phase olive mill wastes using hydrothermal carbonisation for the removal of methylene blue by heterogeneous fenton-like oxidation. Int. J. Environ. Anal. Chem. 2019. [Google Scholar] [CrossRef]
- Varjani, S.; Joshi, R.; Srivastava, V.K.; Ngo, H.H.; Guo, W. Treatment of wastewater from petroleum industry: Current practices and perspectives. Environ. Sci. Pollut. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Wiedemeier, D.B.; Abiven, S.; Hockaday, W.C.; Keiluweit, M.; Kleber, M.; Masiello, C.A.; McBeath, A.V.; Nico, P.S.; Pyle, L.A.; Schneider, M.P.W.; et al. Aromaticity and degree of aromatic condensation of char. Org. Geochem. 2015, 78, 135–143. [Google Scholar] [CrossRef]
- Huang, M.; Li, Z.; Luo, N.; Yang, R.Y.; Wen, J.; Huang, B.; Zeng, G. Application potential of biochar in environment: Insight from degradation of biocharderived dom and complexation of dom with heavy metals. Sci. Total Environ. 2019, 646, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Luo, J.W.; Deng, H.; Huang, P.; Ge, C.J.; Yu, H.M.; Xu, W. Effect of cassava waste biochar on sorption and release behaviour of atrazine in soil. Sci. Total Environ. 2018, 644, 1617–1624. [Google Scholar] [CrossRef]

| Process | Temperature | Heating Rate | Residence Time | By-Products [%] | ||
|---|---|---|---|---|---|---|
| Biochar | Bio-Oil | Syngas | ||||
| Fast pyrolysis | 400–1000 °C | Very fast (~1000 °C/s) | <2 s | 12 | 75 | 13 |
| Slow pyrolysis | 350–980 °C | Slow (<10 °C/min) | 1 h | 27–37 | 19–29 | 25–41 |
| HTC | 180–300 °C | Slow | 1–16 h | 50–80 | 5–20 | 2–5 |
| Gasification | 700–1500 °C | Moderate-very fast | 10–20 s | 10 | 5 | 85 |
| Torrefaction | 200–300 °C | Slow, (<10 °C/min) | ~10–60 min | 80 | 0 | 20 |
| Feedstock | Preparation Conditions | Yield [%] | SAA [m2/g] | Vt [cm3/g] | Ash [%] | CEC [cmol/kg] | pH | Elemental Analysis [%] | Organic Pollutants | pH a | T [°C] | Qmax (mg/g) | Ref. | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | H | N | O | |||||||||||||
| Pecan nutshell | Pyrolysis, 800 °C, 60 min | 30 | 93 | 0.055 | - | - | - | - | - | - | - | Reactive Red 141 | 3 | 35 | 130 | [122] |
| Switchgrass | Pyrolysis, 450 °C, 20 min | - | - | - | - | - | - | - | - | - | - | Reactive red 195A | 5 | - | 1288.4 | [123] |
| Corn stalks | Pyrolysis, 400 °C, 120 min | - | - | - | - | - | - | - | - | - | - | Crystal violet | 6 | 40 | 278.5 | [124] |
| Macroalgae residue | Pyrolysis, 800 °C, 90 min | 22.62 | 133.2 | - | 53.59 | 12.31 | 42.39 | 0.46 | 1.43 | 2.13 | Malachite green | - | 35 | 5306.2 | [20] | |
| Crystal violet | 1222.5 | |||||||||||||||
| Congo red | 345.2 | |||||||||||||||
| Durian rind | Pyrolysis, 800 °C, 25 min | - | 820 | - | - | - | - | - | - | - | - | Congo red | - | - | 87.3 | [125] |
| Switchgrass | Pyrolysis, 900 °C, 60 min | 14.8 | 641.6 | 0.058 | 7.54 | - | 10.7 | 85.5 | 0.6 | 1.5 | 2.6 | Methylene Blue | - | - | 196.1 | [21] |
| Orange G | - | - | 38.2 | |||||||||||||
| Congo Red | - | - | 22.6 | |||||||||||||
| Chicken manure | Pyrolysis, 500 °C, 120 min | - | 68.8 | - | - | - | - | 54.3 | 10.5 | 4.5 | 18.8 | Phenol | 7 | 22 | 106.2 | [97] |
| 2,4-Dinitrophenol | 148.1 | |||||||||||||||
| Pine fruit shell | Slow pyrolysis, 550 °C, 60 min | 29.23 | 228.11 | 0.148 | 3.23 | 8.7 | 83.27 | 3.69 | 0.00 | 13.04 | Phenol | 6.5 | 25 | 26.73 | [98] | |
| Rice straw | Pyrolysis, 500 °C, 60 min | 28.0 | 96.0 | 0.06 | 50.1 | - | - | - | - | - | - | Phenol | 7 | 25 | 80.5 | [126] |
| Rice husk | Pyrolysis, 650 °C, 60 min | 34.1 | 21.7 | - | 36.0 | - | - | 60.3 | 0.1 | 0.4 | 37.6 | Iodine | - | 20 | 120.7 | [127] |
| Methylene blue | 3.8 | |||||||||||||||
| Shore pine | Pyrolysis, 400 °C, 30 min | - | - | - | - | 15.39 | 7.3 | 73.9 | 3.32 | <0.5 | - | Methylene blue | - | - | 1.79 | [128] |
| Sugarcane bagasse | Pyrolysis, 600 °C, 2 h and steam activation, 750 °C, 36 min | - | 347.54 | - | 7.21 | - | - | - | - | - | - | phenol | - | - | 46.43 | [129] |
| Pinewood sawdust | HTC, 300 °C, 20 min and activation, 800 °C, 1 h | 53.8 | 425 | - | 2.90 | - | - | - | - | - | - | phenol | - | - | 83.88 | [130] |
| Rice husk | 80.5 | 358 | - | 59.96 | - | - | - | - | - | - | - | - | 39.30 | |||
| Barely straw | Torrefaction, 220 ℃, 20 min | 81.0 | - | - | - | - | - | - | - | - | - | Methylene blue | - | 23 | 11.65 | [79] |
| Rice straw | Microwave assisted HTC, 180 °C, 50 min | 42.31 | 15.9 | - | 22.13 | - | - | 40.06 | 5.52 | 0.48 | 31.79 | Congo red | - | - | 222.1 | [131] |
| Berberine hydrochloride | - | - | 174.0 | |||||||||||||
| Rice straw | Microwave assisted HTC 200 °C, 60 min | 37.84 | 18.9 | - | 14.27 | - | - | 40.69 | 5.12 | 0.81 | 39.07 | 2-naphthol | - | - | 48.7 | [131] |
| Bamboo sawdust | HTC, 240 °C, 30 min | 49.2 | 19.77 | 0.10 | 0.54 | 58.57 | 4.32 | 0.53 | 36.51 | Congo red | - | 25 | 96.9 | [132] | ||
| 2-naphthol | - | 462.6 | ||||||||||||||
| Chili seeds | HTC, 215 °C, 8 h | 57.8 | 0.5 | 0.00457 | - | - | - | 64.04 | 7.58 | 1.70 | 26.60 | Methylene blue | 7 | 25 | 145 | [133] |
| Walnut shell | HTC, 180 °C, 24 h | - | 52.23 | - | - | - | - | - | - | - | - | Methylene blue | 25 | 68 | [134] | |
| Orange peles | HTC, 190 °C, 24 h | 51.58 | 34.06 | 0.0474 | - | - | - | 78.85 | - | 1.23 | 19.92 | Methylene blue | 7 | 30 | 59.6 | [135] |
| Rice husk | HTC, 260 °C, 60 min | 28.1 | 3.5 | - | 2.7 | - | - | 73.3 | 4.9 | 2.7 | 19.0 | Methylene blue | - | 20 | 9.7 | [127] |
| Iodine | - | 173.1 | ||||||||||||||
| Corn stover | Torrefaction, 250 °C, 0.5 h | - | - | - | 30.38 | 42.92 | 2.39 | 1.55 | 22.25 | Methylene blue | 349.74 | [78] | ||||
| Olive wastes | Pyrolysis, 300 °C, 1 h and KOH activation, 850 °C, 1 h. | - | 1375 | 0.84 | - | - | - | - | - | - | - | Methylene blue | 2 | - | 536 | [28] |
| Iodine | 1136 | |||||||||||||||
| Indigo carmine | 598.8 | |||||||||||||||
| Coconut shell | Pyrolysis, 500 °C, 2 h and NaOH activation, 700 °C, 1.5 h. | 18.8 | 2825 | 1.498 | - | - | - | - | - | - | - | Methylene blue | - | - | 916.26 | [92] |
| Feedstock | Preparation Conditions | Yield [%] | SSA [m2/g] | Vt [cm3/g] | Ash [%] | CEC [cmol/kg] | pH | Elemental Analysis [%] | Metal | pH a | T [°C] | Qmax (mg/g) | Ref. | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | H | N | O | |||||||||||||
| Rice straw | Pyrolysis 550 °C, 2 h | 33.6 | 4.15 | - | 42.9 | - | 10.3 | 45.7 | 2.13 | 1.17 | - | Pb2+ | 5 | 25 | 0.85 b | [107] |
| Zn2+ | 0.61 b | |||||||||||||||
| Chicken manure | Pyrolysis 550 °C, 2 h | 77.5 | 7.09 | 87.2 | - | 9.95 | 7.49 | 0.43 | 0.76 | - | Pb2+ | 5 | 25 | 0.58 b | [107] | |
| Zn2+ | 0.17 b | |||||||||||||||
| Sewage sludge | Pyrolysis 550 °C, 2 h | 79.0 | 8.20 | 68.7 | - | 7.62 | 18.9 | 1.70 | 2.92 | - | Pb2+ | 5 | 25 | 0.11 b | [107] | |
| Zn2+ | 0.07 b | |||||||||||||||
| Chestnut shell | Pyrolysis 450 °C, 2 h | - | - | - | - | - | - | - | - | - | - | As(V) | 7 | 25 | 17.5 | [88] |
| Canola straw | Pyrolysis 400 °C, 3 h 45 min | - | - | - | - | 316.9 | - | - | - | - | - | Cr(III) | 4 | 25 | 0.28 b | [108] |
| Rice straw | - | - | - | - | 483.1 | - | - | - | - | - | 0.27 b | |||||
| Soybean straw | - | - | - | - | 229.5 | - | - | - | - | - | 0.33 b | |||||
| Peanut straw | - | - | - | - | 196.2 | - | - | - | - | - | 0.48 b | |||||
| Almond shell | Pyrolysis 650 °C | - | 145 | - | - | - | - | - | - | - | - | Ni(II) | 7 | 40 | 22.2 | [136] |
| Co(II) | 7 | 20 | 28.09 | |||||||||||||
| Coconut fiber | Pyrolysis, 300 °C, 6 h | - | 4.495 | - | 3.76 | 72.86 | 7.41 | - | - | - | - | Pb2+ | 6 | 25 | 49.5 | [137] |
| Pineapple peels | Pyrolysis, 350 °C, 1 h | - | - | - | - | - | - | - | - | - | - | Cr(VI) | 2 | 30 | 41.7 | [138] |
| Hardwood (Acacia) | Pyrolysis, 300–400 °C, 2 h | - | 1.30 | - | 3.5 | - | 7.62 | 69.6 | 4.3 | 0.6 | 22.0 | Cu2+ | 5 | - | 3.48 | [139] |
| Hardwood (Citrus) | Pyrolysis, 300 °C | 41.6 | 0.8 | - | 8.43 | - | 7.76 | 60.37 | 4.43 | 1.43 | 25.34 | Pb2+ | - | 22 | 7.153 | [140] |
| Wheat Straw | Pyrolysis, 600 °C, 5 h | - | 26.3 | - | 41.1 | 6.12 c | 9.9 | 54 | 2.3 | 0.9 | 2.3 | Cd2+ | 5.5 | 22 | 17.92 | [141] |
| Ni2+ | 16.26 | |||||||||||||||
| Rice Straw | Pyrolysis, 600 °C, 4 h | 21.2 | 162.6 | - | - | - | 9.89 | 68.7 | 2.3 | 3.0 | 25.9 | Zn2+ | 5 | 25 | 38.6 | [109] |
| Cu2+ | 56.5 | |||||||||||||||
| Rice husk | Pyrolysis, 650 °C, 1 h | 34.1 | 21.7 | - | 36.0 | - | - | 60.3 | 0.1 | 0.4 | 37.6 | Cu2+ | 4.8 | 20 | 48.9 | [127] |
| Anaerobically digested sludge | Pyrolysis, 600 °C, 2 h | 52.6 | 162.7 | 0.04 | - | - | - | 27.8 | 1.3 | - | 9.6 | Pb | 7 | 22 | 0.61 b | [104] |
| Sugarcane bagasse | Pyrolysis, 300 °C, 20 min | - | - | - | - | - | - | - | - | - | - | Pb | 6.3 | 25 | 53.48 | [142] |
| Sugarcane bagasse | Pyrolysis, 500 °C | - | 92.30 | 0.04531 | 12.21 | - | 9.63 | 74.02 | 2.61 | 1.00 | 22.37 | Pb(II) | 5 | 30 | 86.96 | [143] |
| Orange peel | - | 0.21 | 0.00016 | 11.17 | - | 8.75 | 66.36 | 3.60 | 2.13 | 28.09 | 27.86 | |||||
| Macroalgae (Enteromorpha) | Hydrothermal liquefaction, 250 °C, 40 min | 20.5 | 29.7 | - | - | - | - | 70.2 | 4.5 | - | 23.1 | Cu2+ | 8.1 | - | 0.254 | [18] |
| Pb2+ | 0.098 | |||||||||||||||
| Sawdust | HTC, 190 °C, 12 h | - | 7.86 | 0.61 | - | - | - | 60.78 | 6.02 | 0.94 | 32.26 | Cu2+ | 5 | 25 | 298.9 | [144] |
| Rice husk | HTC, 260 °C, 1 h | 28.1 | 3.5 | - | 2.7 | - | - | 73.3 | 4.9 | 2.7 | 19.0 | Cu+2 | 4.8 | 20 | 68.2 | [127] |
| Banana peels | HTC, 230 °C, 2 h | 15.6 | 31.65 | - | 0.53 | - | - | 71.38 | 6.34 | 0.57 | 19.78 | Pb2+ | - | - | 315.16 | [65] |
| Rice Straw | Microwave assisted HTC 160 °C, 60 min | 42.53 | 8.21 | - | 17.56 | - | - | 40.34 | 5.11 | 1.15 | 35.76 | Cu2+ | - | - | 144.9 | [131] |
| Rice Straw | Microwave assisted HTC, 200 °C, 50 min | 36.22 | 25.4 | - | 19.53 | - | - | 37.44 | 4.85 | 0.75 | 37.4 | Zn2+ | - | - | 112.8 | [131] |
| Wood chips (pine and spruce) | Gasification at 1000 °C | - | 14.4 | 0.03 | - | - | - | - | - | - | - | Fe | 4 | - | 24.1 | [145] |
| Cu | 5 | 11.1 | ||||||||||||||
| Ni | 8 | 5.6 | ||||||||||||||
| Wood chips (pine and spruce) | Gasification at 1000 °C and ZnCl2 activation, 500 °C, 1 h | - | 259 | 0.26 | - | - | - | - | - | - | - | Fe | - | - | 20.5 | [145] |
| Cu | 23.1 | |||||||||||||||
| Ni | 18.2 | |||||||||||||||
| Pinewood sawdust | HTC, 300 °C, 20 min and CO2 activation, 800 °C, 1 h | 53.8 | 425 | - | 2.90 | - | - | - | - | - | - | Cu | - | - | 25.18 | [130] |
| Rice husk | 80.5 | 358 | - | 59.96 | - | - | - | - | - | - | - | - | 22.62 | |||
| Feedstock | Preparation Conditions | Biochar Characteristics | Removal Efficiency | Ref. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SSA [m2/g] | Porosity [%] | C [%] | H [%] | O [%] | COD [%] | Nutrients [%] | TSS/Turbidity [%] | HM [%] | E. coli [%] | |||
| Waste wood pellets | Gasification, 520 °C | - | - | - | - | - | - | NO3-N: 86 Tot-P: 47 | 86 | 17–75 | 27 | [14] |
| Miscanthus | Pyrolysis, 850 °C, 30 min | 500 | - | 80.0 | 1.3 | 8.1 | 74 | NH4-N: ≈ 7 Tot-P: 35 | 31 | - | 23 | [17] |
| Hard wood | - | 184 | 72–74 | - | - | - | 95 | Tot-N: 52 Tot-P:57 | - | - | - | [190] |
| Pine-spruce | - | 170–200 | 72–74 | - | - | - | > 90 | Tot-N: 50–52 Tot-P: 60–93 | - | - | - | [191] |
| Forestry wood waste | Pyrolysis, 700 °C, 15 h | 137.0 | - | - | - | - | - | - | - | - | 92–99 | [192] |
| Rice husk | Gasification | 143 | - | - | - | - | 94 | Tot-N: 10.8 Tot-P: 5.3 | 63 | - | 55 | [186] |
| Softwood | Pyrolysis, 815–1315 °C, 1–3 s | - | - | 79 | - | - | - | - | - | - | ~96 | [193] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Enaime, G.; Baçaoui, A.; Yaacoubi, A.; Lübken, M. Biochar for Wastewater Treatment—Conversion Technologies and Applications. Appl. Sci. 2020, 10, 3492. https://doi.org/10.3390/app10103492
Enaime G, Baçaoui A, Yaacoubi A, Lübken M. Biochar for Wastewater Treatment—Conversion Technologies and Applications. Applied Sciences. 2020; 10(10):3492. https://doi.org/10.3390/app10103492
Chicago/Turabian StyleEnaime, Ghizlane, Abdelaziz Baçaoui, Abdelrani Yaacoubi, and Manfred Lübken. 2020. "Biochar for Wastewater Treatment—Conversion Technologies and Applications" Applied Sciences 10, no. 10: 3492. https://doi.org/10.3390/app10103492
APA StyleEnaime, G., Baçaoui, A., Yaacoubi, A., & Lübken, M. (2020). Biochar for Wastewater Treatment—Conversion Technologies and Applications. Applied Sciences, 10(10), 3492. https://doi.org/10.3390/app10103492






