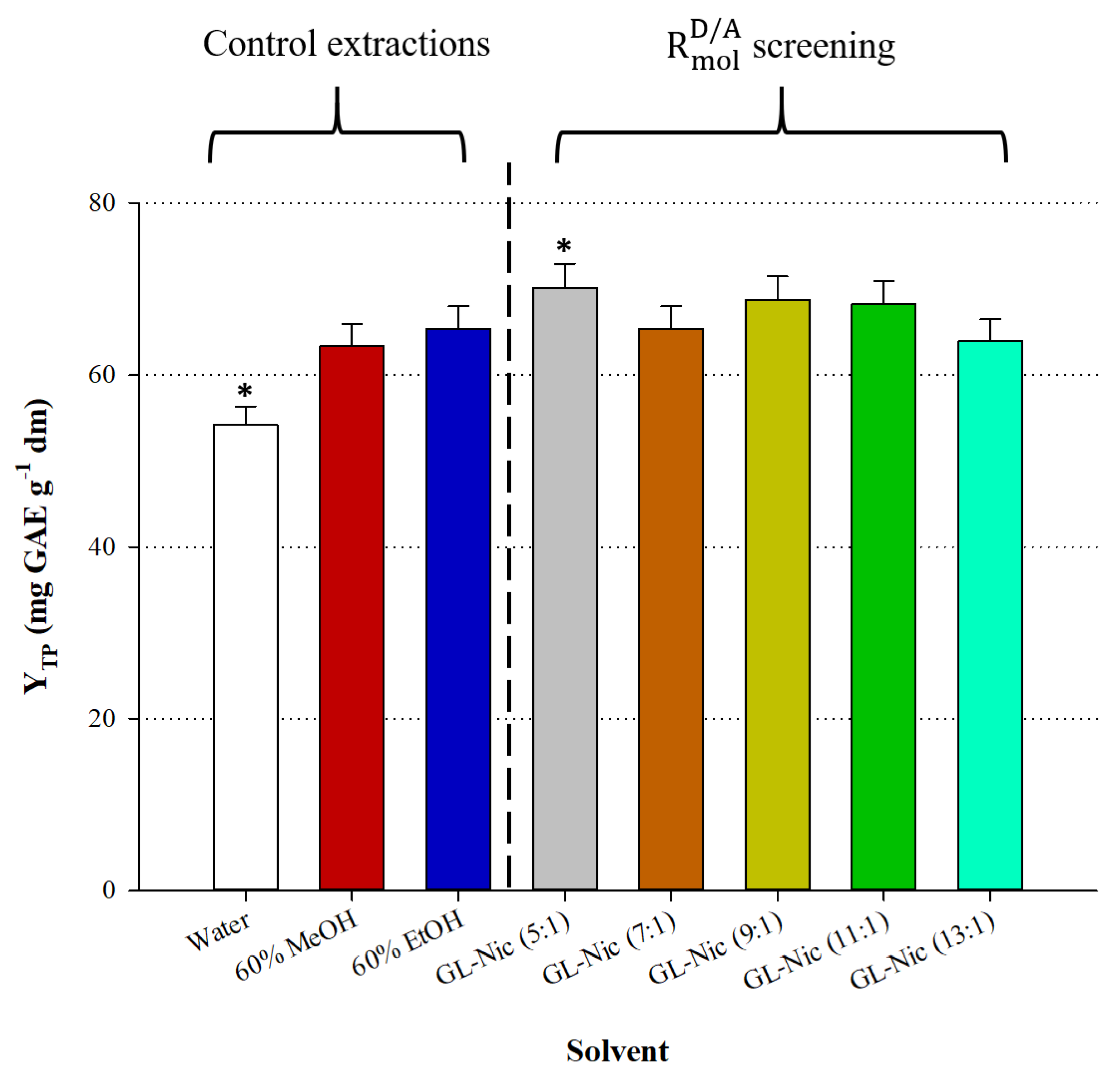
3.1. DES Synthesis and Examination of HBD: HBA Molar Ratio ()
The use of nicotinamide as HBA in L-lactic acid-based DES has been recently reported for the first time [
24]; however, the use of glycerol as HBD in combination with nicotinamide as HBA, to the best of the authors’ knowledge, is heretofore unreported. When designing a DES, the role of
in the polyphenol extraction performance is salient [
19,
25], and therefore, an initial screening of DES with varying
was deemed essential in identifying the highest-performing combination.
Attempts to combine glycerol and nicotinamide at
up to 4 were not met with success, since the DES synthesized tended to develop crystals within 24 h, at room temperature (23 ± 2 °C). Combination at
= 5 produced a DES, which remained stable for several weeks, and based on this finding, a series of DES with
ranging from 5 to 13 were synthesized and tested for their effectiveness in extracting polyphenols from
M. oleifera. All the DES assayed were used as 70% (
w/v) aqueous mixtures and the outcome of this screening is given in
Figure 1. The DES with
= 5 exhibited statistically higher extraction efficiency (
p < 0.05) compared with all other DES and also 60% (
v/v) aqueous ethanol and 60% (
v/v) aqueous methanol. Water, on the other hand, was the least efficient solvent (
p < 0.05). On this ground, the DES with
= 5, assigned as GL-Nic (5:1), was chosen for further examination.
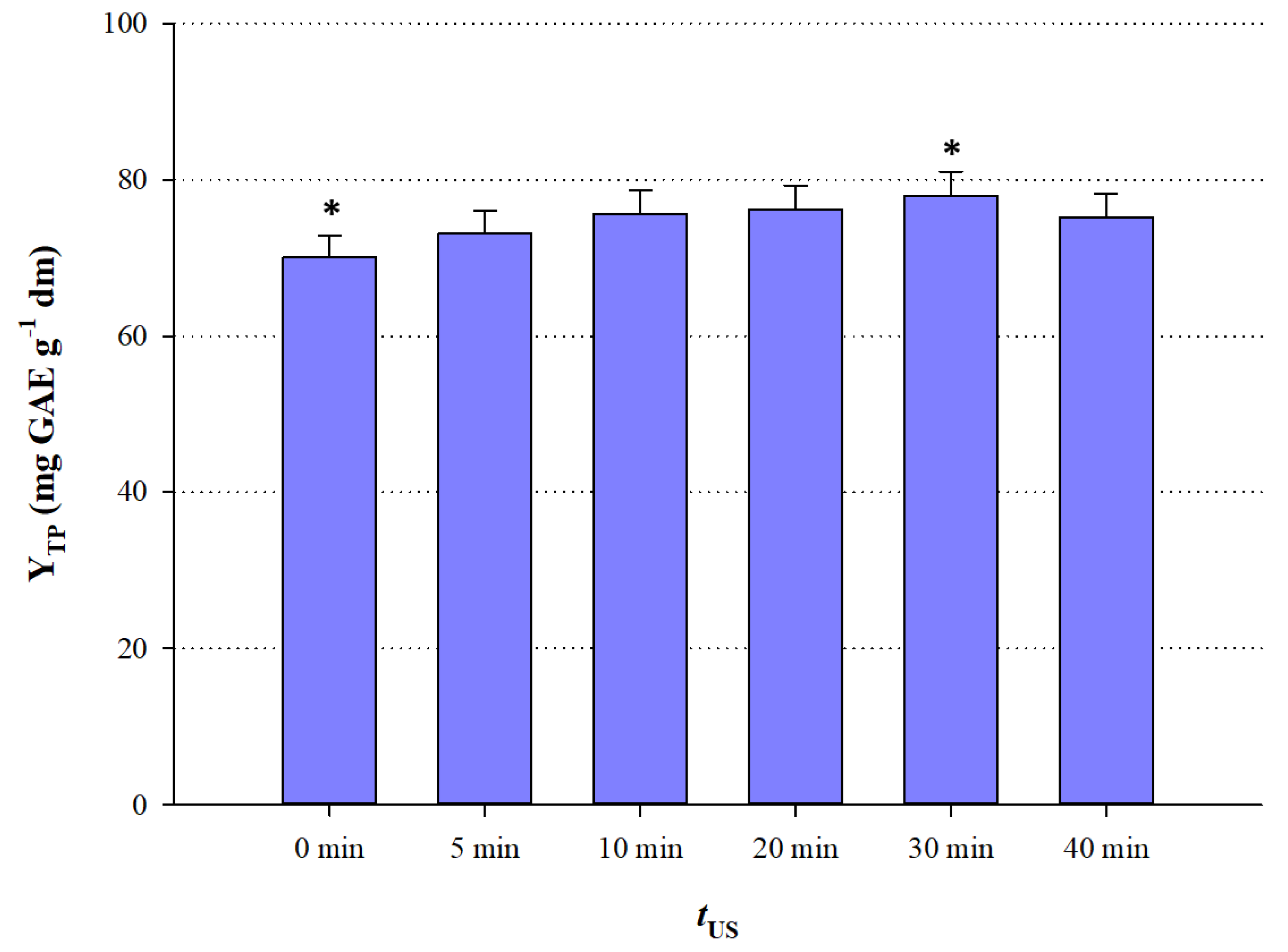
3.2. The Effect of Ultrasonication Pretreatment
To examine the effect of ultrasonication, GL-Nic (5:1) used as 70% (
w/v) aqueous solution, was mixed with dried powder of
M. oleifera leaves, shaken vigorously for a few seconds to form slurry, and then placed in the ultrasonication bath, at room temperature. Room temperature (23 ± 2 °C) was preferred, because at lower temperatures, ultrasonication results in better cavitation, which is the main effect involved in the ultrasound-assisted extraction. At higher temperatures, bubbles formed through cavitation collapse less violently, and thus the ultrasonication effect is less effective [
26]. Ultrasonication treatments were performed for 5, 10, 20, 30, and 40 min, followed by stirred-tank extraction. As can be seen in
Figure 2, a regime of 30-min ultrasonication gave statistically higher Y
TP (
p < 0.05), whereas extraction without ultrasonication pretreatment (0 min) was significantly less effective (
p < 0.05). Ultrasonication beyond 30 min resulted in decreased Y
TP, thus 30 min was chosen as the most appropriate pretreatment period.
3.3. Optimisation of Extraction Performance
The evidence emerged from recent examinations on polyphenol extraction with DES suggested that the three process (independent) variables considered, namely
CDES, R
L/S, and S
S, are highly influential in solid/liquid extraction processes [
19,
25]. For this reason, these three parameters were used among several others to build a predictive extraction model. Two cases were examined, one with a 30-min ultrasonication regime prior to every extraction and one without ultrasonication. The purpose of such investigation was to clarify whether an ultrasonication step before batch stirred-tank extraction could affect the optimization settings and assist in achieving significantly higher extraction yield.
Appraisal of the fitted models and response surface suitability were based on the ANOVA test and lack-of-fit test. The
p-value for each equation term was calculated to examine the contribution of linear, interaction, and quadratic effects in the independent variables (
Table 2). Statistically non-significant terms were omitted from the mathematical models, which are given as polynomial equations in
Table 3. The predicted response values calculated by the models, along with the measured (actual) values for each design point, are shown in
Table 4. R
2 provides indication of the amount of total variability around the mean explained by the regression model. R
2 terms for both models were ≥0.95, so it would be argued that the estimation of the regression equations exhibited a good adjustment to the experimental data. The
p value for lack-of-fit (assuming a confidence interval of 95%) was >0.05 for both models (
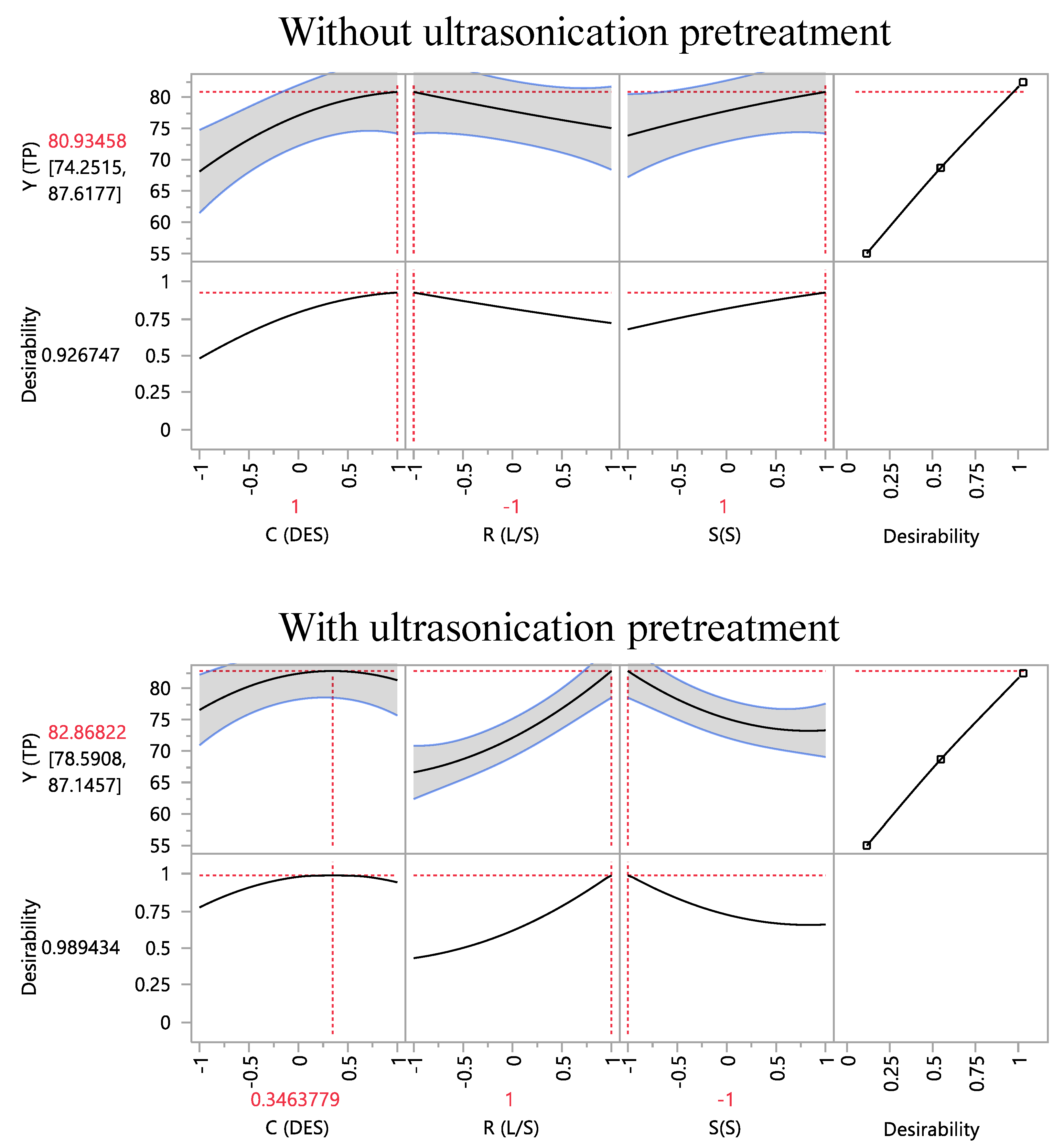
Table 4), suggesting that the fitted models may be reliable predictors. By setting the values of all three process variables at their optima, using the desirability function (
Figure 3), it was made possible to calculate the maximum predicted responses (
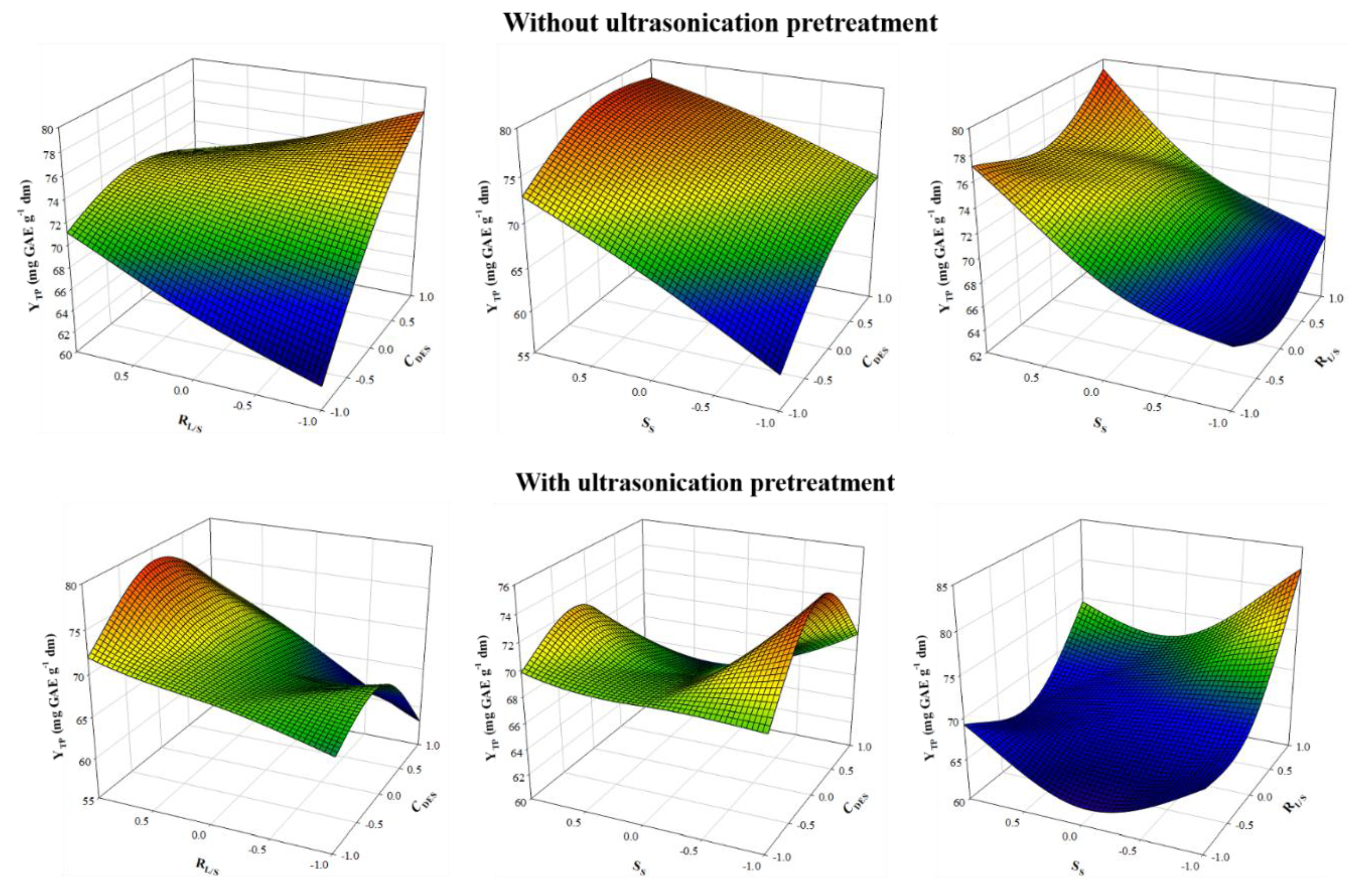
Table 5). The response surface plots (
Figure 4) were helpful in visualizing which variables were more influential to the response.
Ultrasonication of samples prior to stirred-tank extraction had a very pronounced effect on the extraction models, as can be concluded by the equations derived (
Table 3). Thus, while
CDES (X
1) had a positive contribution in maximizing Y
TP in the extraction without ultrasonication pretreatment, it negatively affected the ultrasound-pretreated extraction. On the contrary, increased S
S (X
3) was significant for maximizing Y
TP in samples received no pretreatment, whereas ultrasound-pretreated samples required the lowest S
S used to give maximum Y
TP.
Furthermore, for the extractions without pretreatment, RL/S (X2) had no direct impact on YTP, but its cross effect (X1X2) with CDES was negative. For the ultrasound-pretreated extractions, quadratic effects of both RL/S (X2) and SS (X2) were positive and significant.
The impact of the ultrasonication pretreatment could be more characteristically depicted by the changes found in the optimization settings of all three variables. Pretreatment resulted in shifting the theoretically required DES amount from 85% to 75% (
w/v), R
L/S from 20 to 100 mL g
−1, and S
S from 800 to 200 rpm (
Table 5). This outcome suggested a profound influence of ultrasonication on the pattern through which the process variables can affect polyphenol extraction. On the other hand, it is to be emphasized that the ultrasonication pretreatment exerted virtually no effect on the extraction performance, since the optimal predicted values for Y
TP, for the extraction without and with pretreatment, had no statistically significant difference, being 80.93 ± 6.68 and 82.87 ± 4.28 mg GAE g
−1 dm, respectively (
Table 5).
A critical assessment of these data would point out that samples received no ultrasonication pretreatment required higher DES concentration and a relatively high speed of stirring to yield extracts with increased Y
TP. The optimum
CDES determined was 85% (
w/v), very close to the 80% (
w/v) found for
M. oleifera polyphenol extraction with a DES composed of glycerol and sodium acetate (6:1) [
13]. Likewise, the optimum S
S (800 rpm) was comparable to 900 rpm determined for polyphenol extraction from onion solid wastes, using a DES of glycerol/sodium propionate (8:1) [
25]. By contrast, the optimum R
L/S (20 mL g
−1) was rather low compared with those reported for polyphenol extraction with DES, ranging from 29 mL g
−1 [
27] to 100 mL g
−1 [
25].
This picture was fundamentally changed when samples were ultrasonicated prior to stirred-tank extraction. The optimum
CDES dropped to 75% (
w/v), while ultrasonication-pretreated samples required 5 times higher R
L/S but 4 times lower S
S to provide maximum Y
TP (
Table 5). Ultrasonication is known to contribute in intensification of extraction efficacy, owed to several phenomena that accompany irradiation with ultrasounds, including propagation of ultrasound pressure waves through the solvent and resulting cavitation effects [
28,
29]. The increased performance usually observed in the ultrasound-assisted extractions is generally attributed to mechanical, cavitation, and thermal effects, which can provoke cell wall disruption and reduction of particle size, resulting eventually in enhanced mass transfer across cell membranes. On such a theoretical background, it could be postulated that the reduced
CDES required for the ultrasonicated-pretreated samples to reach optimum Y
TP, could probably be ascribed to higher polyphenol diffusivity and solubilization, as a result of cell wall/membrane disruption. Such an event could facilitate leaching and dissolution of polyphenols into the liquid phase, an assumption corroborated by the drastic decrease in the optimum S
S required, from 800 to 200 rpm. It would appear that lower S
S (200 rpm) is sufficient to provide the turbulence necessary for effective diffusion, whereas higher S
S (800 rpm) might provoke adsorption effects, thereby hindering higher extraction yields.
On the other hand, the pronounced increase in the optimum R
L/S could be attributed to a rapid accumulation of polyphenols at the surface of the solid particles, due to cell breakdown and particle disintegration, that would cause extensive liberation of polyphenols from the interior of the solid particles. Such accumulation would presumably require a higher concentration gradient for effective diffusivity, hence the higher R
L/S. The parameter R
L/S is tightly associated with diffusion phenomena [
30,
31,
32], and it has been demonstrated that rising R
L/S could bring about a significant diffusivity increase [
33].
3.4. Extraction Kinetics and Temperature Effects
The optimization of the extraction process revealed significant changes in the extraction variables as a consequence of ultrasonication pretreatment, but to portray the ultrasound effect on the process in an integrated frame, information pertaining to the kinetics of the extraction and the effect of temperature was indispensable. Towards this objective, extraction kinetics were traced over a wide breadth of temperatures, ranging from 40 to 80 °C (
Figure 5). In each case, the optimized conditions were used to carry out the extractions (
Table 5). A modification of a previously proposed kinetic model was employed for both non-pretreated and ultrasound-pretreated samples [
34]:
The term
YTP(t) represents the extraction yield in total polyphenols (mg GAE g
−1 dm) at any time
t,
YTP(0) is a fitting parameter,
YTP(s) the yield in total polyphenols at equilibrium (saturation), and
t0.5 half the time (min) required for the extraction to enter the regular regime. The phase of the regular regime refers to the period within which small increases in
YTP are achieved within relatively large
t [
35]. In all cases examined, fitting of the kinetic model to the experimental data gave R
2 > 0.98 (
p < 0.0001), suggesting that the model implemented could very effectively describe extraction kinetics. Determination of the initial rate of the extraction,
h, and the second-order extraction rate,
k, was accomplished using the following equations:
The data generated from the kinetic assay are analytically presented in
Table 6. The effect exerted by the ultrasonication pretreatment was shown to be temperature-dependent, since acceleration of the extraction (increased
k) was seen only at 70 and 80 °C. This trend was corroborated by the values determined for
h, which were increased for the ultrasound-pretreated samples at temperatures higher than 60 °C. Likewise,
t0.5 was shorter for the extractions received ultrasonication pretreatment at temperatures higher than 60 °C. Furthermore, for every temperature tested, Y
TP of the ultrasonication-pretreated samples was always significantly higher than that of the non-pretreated ones. However, in both cases, temperature had a non-significant effect on
YTP(s), since it varied between 82.03 to 83.90 mg GAE g
−1 dm for the non-pretreated extractions and from 91.24 to 93.97 mg GAE g
−1 dm for the ultrasound-pretreated extractions.
Pretreatment with ultrasonication is likely to solubilize all polyphenols occurring at and near the surface of the solid particles. This assumption could justify the fact that at temperatures 40–60 °C the stirred-tank extraction of the ultrasound-pretreated samples proceeded at a lower rate. On the other hand, the non-pretreated samples had a higher load of polyphenols at the exterior of the solid particles and exhibited apparently increased
k and
h. At higher temperatures, polyphenol extraction was more rapid for the ultrasonicated samples, most probably because the pretreatment brought about disruption of the cell walls of the plant material, which enabled more facile penetration of the solvent into the solid particles and easier entrainment of the solute (polyphenols) in the liquid phase. Concerning
YTP(s), the variation as a response to
T was non-significant, a fact indicating the
T had virtually no effect on
YTP(s). This outcome is a paradox, in light of previous investigations, which demonstrated that increasing
T had a proportional effect on
YTP(s) [
36,
37]. However, earlier studies on polyphenol extraction from
M. oleifera leaves with a glycerol/sodium acetate DES revealed that switching
T from 50 to 80 °C resulted in a constant decline in
YTP(s) [
13]. The fact that increasing
T did not contribute in attaining higher
YTP(s) might suggest that extraction could reach equilibrium after a given period of time, irrespective of the
T.
The increase in
k as a response to increasing
T was found to obey the Arrhenius law:
where
k0,
R,
T, and
Ea correspond to the temperature-independent factor (min
−1), the universal gas constant (8.314 J K
−1 mol
−1), the absolute temperature (K), and the activation energy (J mol
−1). The linear expression of Equation (7) would be:
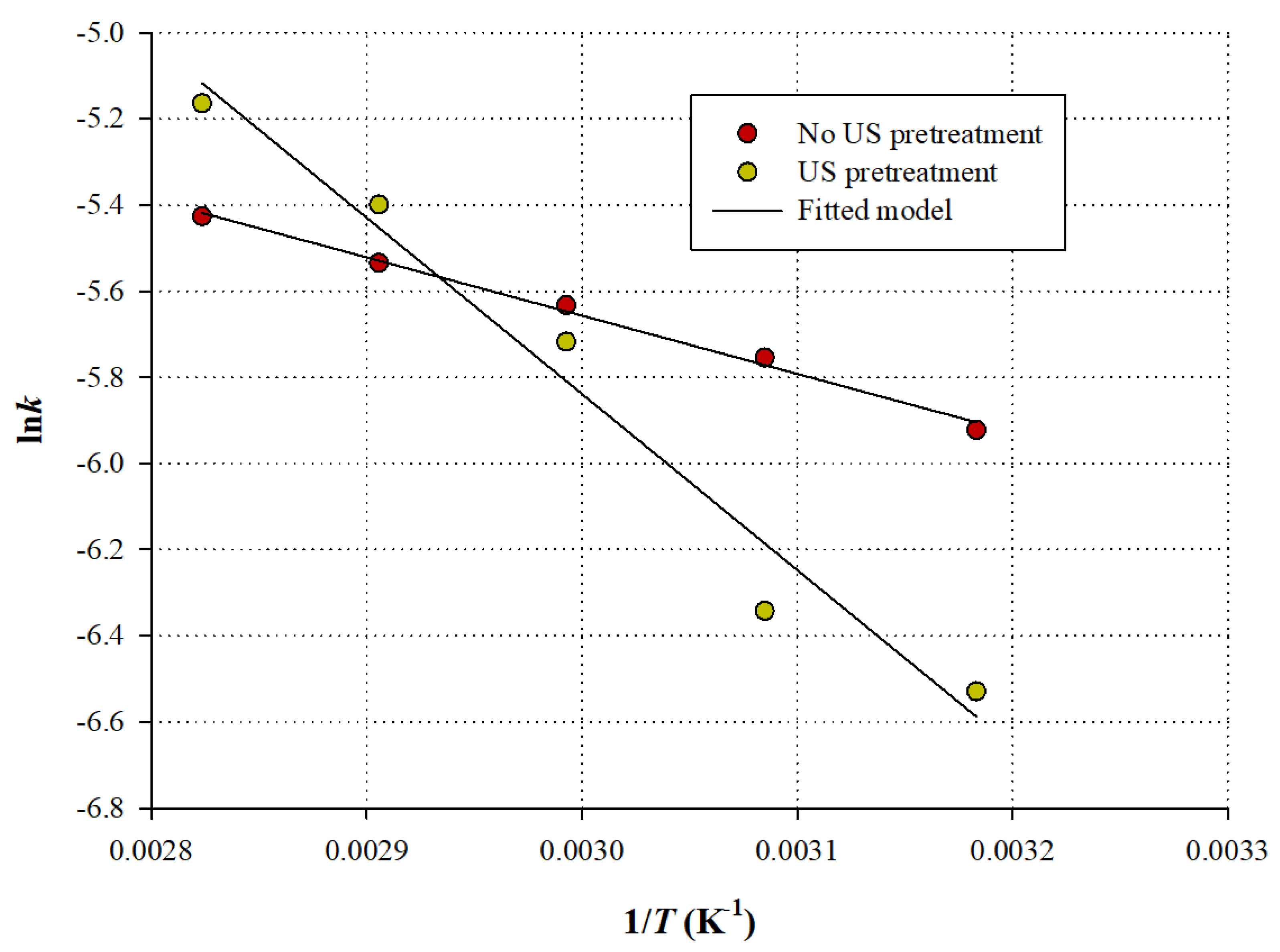
Thus,
Ea could be determined graphically, by the slope (
) of the straight line obtained by plotting
lnk as a function of 1/
T (
Figure 6).
The
Ea values for the extractions without and with ultrasonication pretreatment were 11.24 and 34.02 kJ mol
−1, respectively (
Table 6), clearly showing that the extraction of the ultrasonication-pretreated samples was more energy-demanding by almost 4 times. Based on this outcome, it would appear that ultrasonication prior to batch stirred-tank extraction does not lower the requirements in energy for accomplishing an efficient extraction of polyphenols from
M. oleifera leaves, but the energetic needs are even higher.
Yet, such observation could be rather misleading and the increased of
YTP(s) of the ultrasonication-pretreated samples should not be overlooked. The increased
Ea value found for the ultrasonication pretreated extraction was most probably because during pretreatment the most readily extracted polyphenols were dissolved in the liquid phase (solvent), leaving behind (inside the solid particles) polyphenols that were more difficult to extract, since solubilization of these polyphenols would depend on internal diffusion, which is the rate-determining step in diffusion-controlled extractions [
30,
38]. Therefore, the following stirred-tank extraction was apparently more energy-demanding. However, considering
YTP(s), it could be argued that ultrasonication facilitated the washing (initial) phase of the extraction, provoking extended release of superficial polyphenols, whereas the stirred-tank extraction recovered the remaining compounds, located deeper inside the particles. Indeed, just after ultrasonication the
YTP was approximately 29.70 mg GAE g
−1 dm, which was almost 30% of the
YTP(s) achieved with ultrasonication pretreatment (
Table 6). These data strongly supported that a large part of polyphenols was extracted during the ultrasonication step.
3.5. Polyphenolic Profile
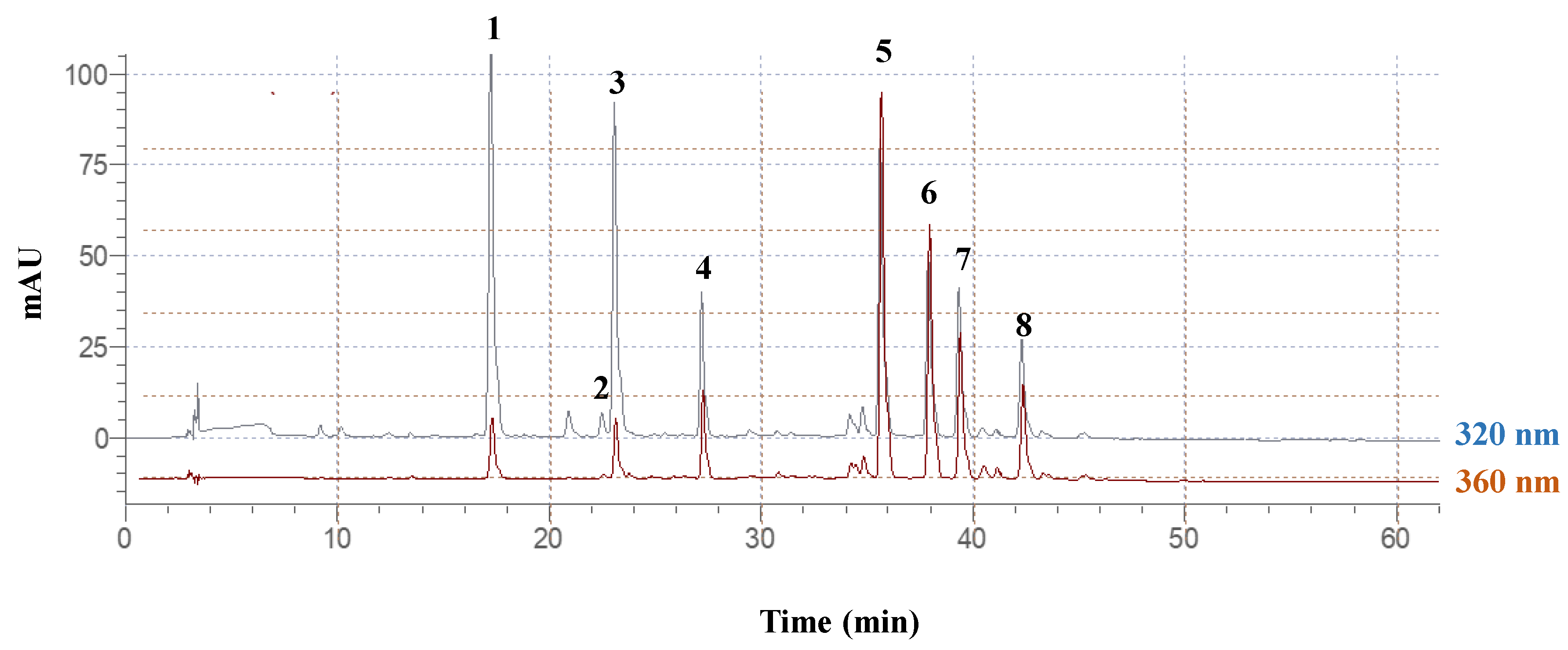
To illustrate the effect of ultrasonication pretreatment on the analytical polyphenolic profile, LC-MS was undertaken for the samples obtained after 180 min of extraction, at 80 °C, under optimal conditions. A typical chromatogram is given in
Figure 7, showing the principal substances detected at both 320 and 360 nm. It should be noted that for both ultrasonication-pretreated and non-pretreated samples, the chromatograms recorded were identical (data not shown), evidencing no effect of ultrasonication on the polyphenolic profile.
Peak #1 showed a pseudo-molecular ion at
m/
z = 355 and a derivative ion at
m/
z = 163. Considering the UV-vis characteristics, this structure was assigned to neochlorogenic acid. The identity of this peak was further confirmed by comparing the UV-vis spectrum and retention time (Rt) with an original standard. Likewise, peaks 2 and 3 were tentatively identified as chlorogenic acid and a chlorogenate isomer, respectively (
Table 7).
Peak #4 displayed a pseudo-molecular ion at
m/
z = 595, a sodium adduct at
m/
z = 617, and a diagnostic fragment (aglycone) at
m/
z = 287. This compound was tentatively assigned to multiflorin B (kaempferol 3-
O-rhamnosylglucoside). Similarly, peaks 7 and 8 were assigned to a kaempferol glucoside and kaempferol malonylglucoside. Peaks 5 and 6, which exhibited typical quercetin glycoside UV-vis pattern and diagnostic fragment at
m/
z = 303, were tentatively identified as quercetin glucoside and quercetin malonylglycoside derivative, respectively [
39]. All these compounds have been previously reported in
M. oleifera leaf extracts [
13]; however, the presence of a quercetin rhamnoside derivative claimed in an earlier study [
40] was not confirmed.
The information emerged from the quantitative data (
Table 8) showed that ultrasonication pretreatment afforded changes in the recovery of major polyphenols, but not all substances were equally affected.
For neochlorogenic acid and chlorogenic acid, ultrasonication pretreatment brought about an increase by 3.1% and 9.1%, but for the chlorogenic acid isomer, a decrease by almost 1.9% was observed. Overall, total chlorogenate content was by 1.1% increased, a difference that falls within the limits of statistical error. Thus, ultrasonication pretreatment had practically no effect on chlorogenate extractability. On the other hand, all flavonol glycosides had higher content in the ultrasonication-pretreated sample, the increases varying from 0.65% (quercetin glucoside) to 14.6% (quercetin manolyglycoside derivative). Overall, the increase in flavonol content caused by ultrasonication pretreatment was 6.4%, and for all polyphenols considered (chlorogenates + flavonols), an increase by 4.7% was determined.