Pronounced Antagonism of Zinc and Arsenate on Toxicity to Barley Root Elongation in Soil
Abstract
1. Introduction
2. Materials and Methods
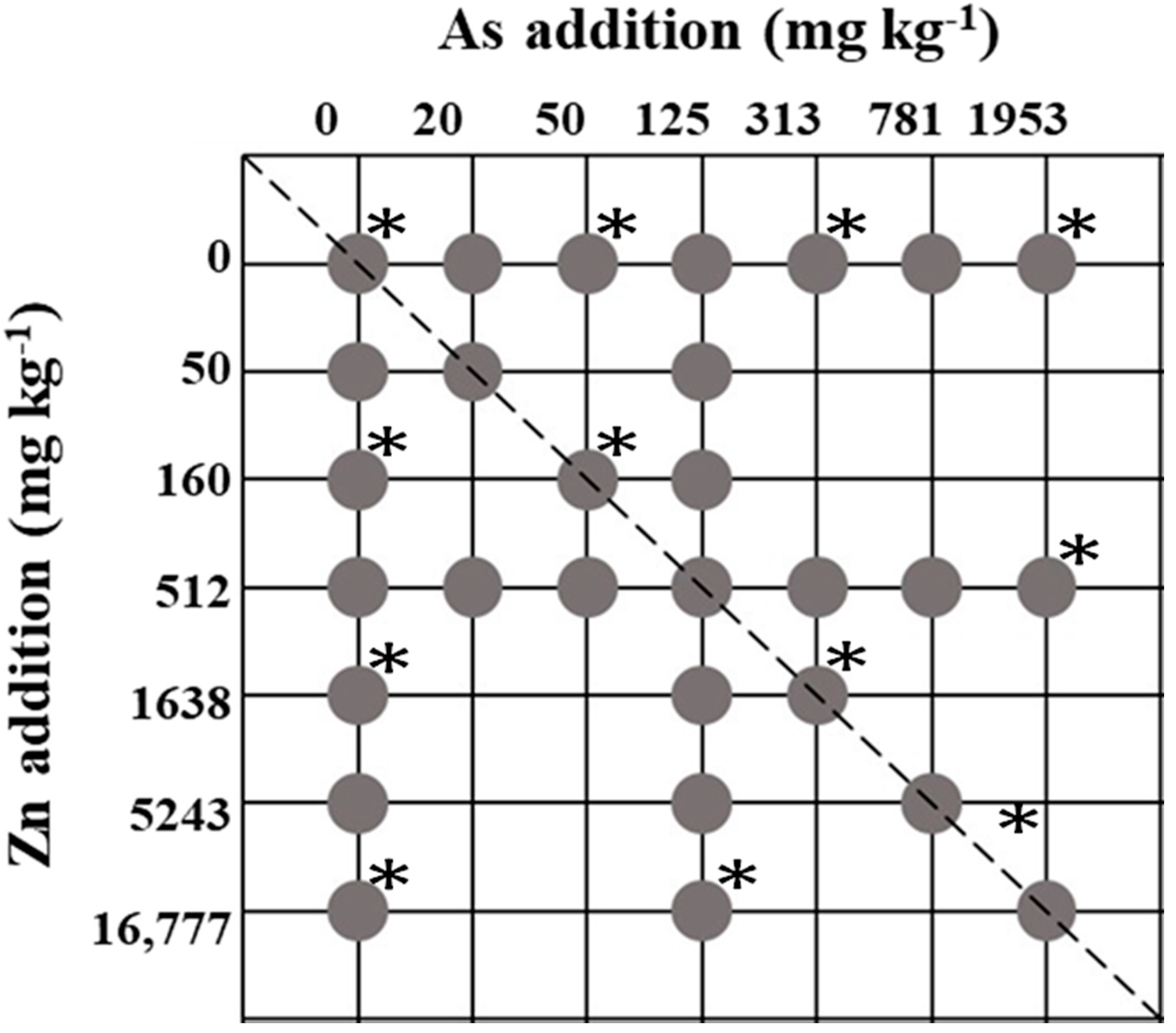
2.1. Experimental Design
2.2. Soil Samples
2.3. Stock Soils and Soil Treatments
2.4. Root Elongation Test
2.5. Soil Analyses
2.6. Single and Mixture Toxicity Models
3. Results
3.1. Soil Samples and Zn–As Treatments
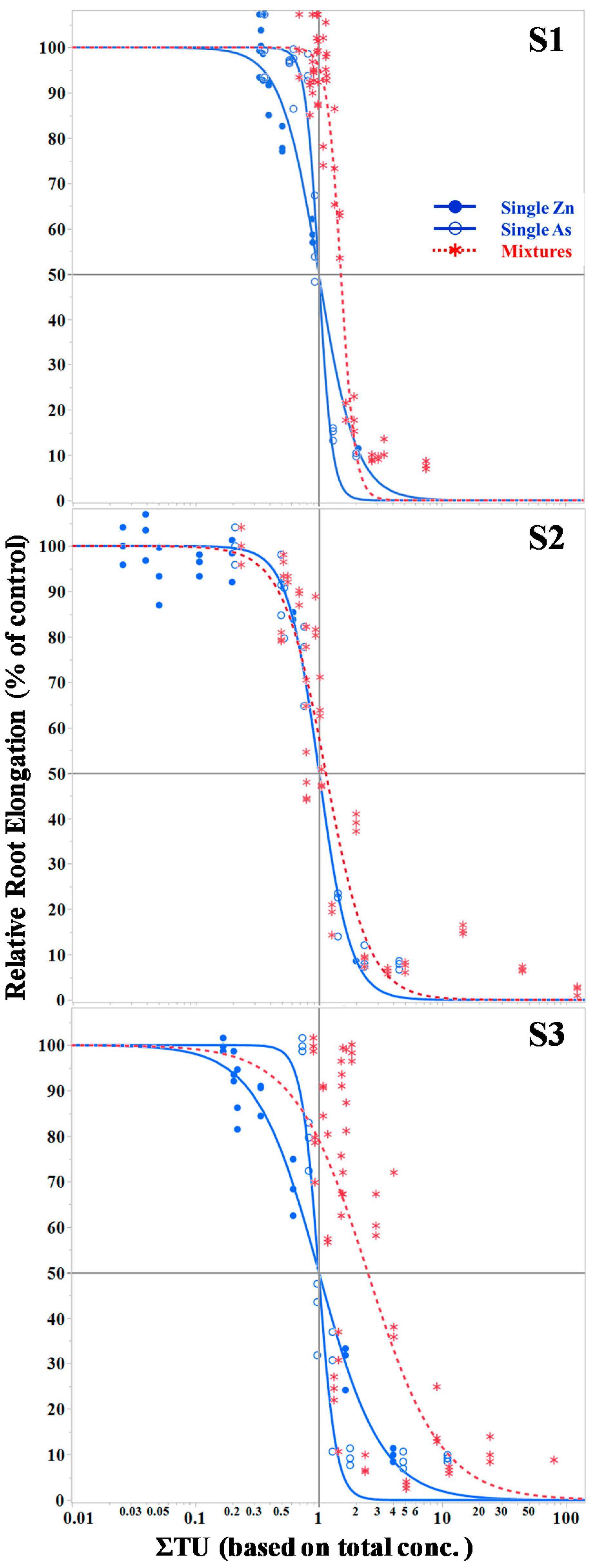
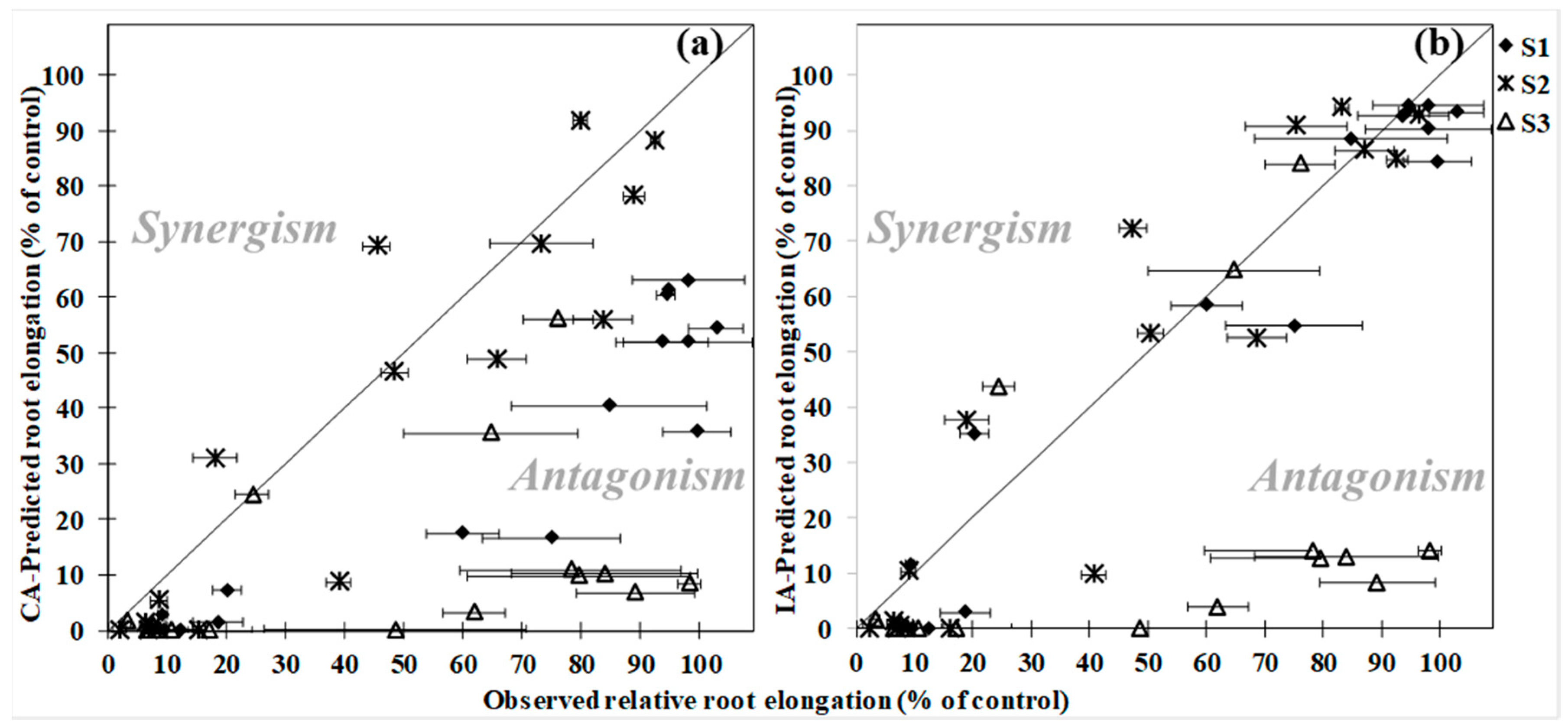
3.2. Root Elongation
3.3. Zinc and As Concentrations in Soil Solution
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alloway, B.J. Heavy Metals in Soils, 3rd ed.; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Nagajyoti, P.C.; Lee, K.D.; Sreekanth, T.V.M. Heavy metals, occurrence and toxicity for plants: A review. Environ. Chem. Lett. 2010, 8, 199–216. [Google Scholar] [CrossRef]
- Abbas, G.; Murtaza, B.; Bibi, I.; Shahid, M.; Niazi, N.K.; Khan, M.I.; Amjad, M.; Hussain, M. Arsenic Uptake, Toxicity, Detoxification, and Speciation in Plants: Physiological, Biochemical, and Molecular Aspects. Int. J. Environ. Res. Public Health 2018, 15, 59. [Google Scholar] [CrossRef] [PubMed]
- Reimann, C.; Fabian, K.; Birke, M.; Filzmoser, P.; Demetriades, A.; Négrel, P.; Oorts, K.; Matschullat, J.; de Caritat, P.; Albanese, S.; et al. GEMAS: Establishing geochemical background and threshold for 53 chemical elements in European agricultural soil. Appl. Geochem. 2018, 88, 302–318. [Google Scholar] [CrossRef]
- Teaf, C.M.; Covert, D.J.; Teaf, P.A.; Starks, M.J. Arsenic Cleanup Criteria for Soils in the US and Abroad: Comparing Guidelines and Understanding Inconsistencies. In Proceedings of the Annual International Conference on Soils, Sediments, Water and Energy, Amherst, MA, USA, 18–21 October 2010; Volume 15, p. 10. [Google Scholar]
- Rodríguez, L.; Ruiz, E.; Alonso-Azcárate, J.; Rincón, J. Heavy metal distribution and chemical speciation in tailings and soils around a Pb-Zn mine in Spain. J. Environ. Manag. 2009, 90, 1106–1116. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.S.; Huyck, H.L.O. An overview of the abundance, relative mobility, bioavailability, and human toxicity of metals. Rev. Econ. Geol. 1999, 6A–6B, 29–70. [Google Scholar]
- Backhaus, T.; Faust, M. Predictive Environmental Risk Assessment of Chemical Mixtures. Environ. Sci. Technol. 2012, 46, 2564–2573. [Google Scholar] [CrossRef] [PubMed]
- De Zwart, D.; Posthuma, L. Complex mixture toxicity for single and multiple species: Proposed methodologies. Environ. Toxicol. Chem. 2005, 24, 2665–2676. [Google Scholar] [CrossRef] [PubMed]
- Badot, P.-M.; Degiorgi, F.; Adam, O.; Ctini, G. Combined Exposure to Mixture of Chemicals. An Impossible Challenge? In Pesticides—The Impact of Pesticides Exposure; Stoycheva, M., Ed.; InTech Europe: London, UK, 2009; pp. 67–90. ISBN 978-953-307-531-0. [Google Scholar]
- Vijver, M.G.; Elliot, E.G.; Peijnenburg, W.J.G.M.; de Snoo, G.R. Response predictions for organisms water-exposed to metal mixtures: A meta-analysis. Environ. Toxicol. Chem. 2011, 30, 1482–1487. [Google Scholar] [CrossRef] [PubMed]
- Nys, C.; Versieren, L.; Cordery, K.I.; Blust, R.; Smolders, E.; de Schamphelaere, K.A.C. Systematic Evaluation of Chronic Metal-Mixture Toxicity to Three Species and Implications for Risk Assessment. Environ. Sci. Technol. 2017, 51, 4615–4623. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency (USEPA). Ecological Soil Screening Levels for Zinc Interim Final; USEPA: Washingtong, DC, USA, 2007.
- Arco-Lazaro, E.; Agudo, I.; Clemente, R.; Bernal, M.P. Arsenic (V) adsorption-desorption in agricultural and mine soils : Effects of organic matter addition and phosphate competition. Environ. Pollut. 2016, 216, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Sanchary, I.J.; Huq, S.M.I. Remediation of Arsenic Toxicity in the Soil-Plant System by Using Zinc Fertilizers. J. Agric. Chem. Environ. 2017, 6, 30–37. [Google Scholar] [CrossRef]
- Kader, M.; Lamb, D.T.; Wang, L.; Megharaj, M.; Naidu, R. Zinc-arsenic interactions in soil: Solubility, toxicity and uptake. Chemosphere 2017, 187, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Grafe, M.; Nachtegaal, M.; Sparks, D. Formation of Metal-Arsenate Precipitates at the Goethite-Water Interface. Environ. Sci. Technol. 2004, 38, 6561–6570. [Google Scholar] [CrossRef] [PubMed]
- Carabante, I.; Grahn, M.; Holmgren, A.; Kumpiene, J.; Hedlund, J. Influence of Zn(II) on the adsorption of arsenate onto ferrihydrite. Environ. Sci. Technol. 2012, 46, 13152–13159. [Google Scholar] [CrossRef] [PubMed]
- Das, D.K.; Garai, T.K.; Sarkar, S.; Sur, P. Interaction of Arsenic with Zinc and organics in a rice (Oryza sativa L.)—Cultivated field in India. Sci. World J. 2005, 5, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Das, I.; Sanyal, S.K.; Ghosh, K.; Das, D.K. Arsenic mitigation in soil-plant system through zinc application in West Bengal soils. Bioremediat. J. 2016, 20, 24–37. [Google Scholar] [CrossRef]
- Patel, H.V.; Parmar, S.R.; Chudasama, C.J.; Mangrola, A.V. Interactive studies of Zinc with Cadmium & Arsenic on seed germination and antioxidant properties of Phaseolus aureus Roxb. Int. J. Plant Anim. Environ. Sci. 2013, 3, 166–174. [Google Scholar]
- Guzmán-Rangel, G.; Versieren, L.; Qiu, H.; Smolders, E. Additive toxicity of zinc and arsenate on barley (Hordeum vulgare) root elongation. Environ. Toxicol. Chem. 2017, 36, 1556–1562. [Google Scholar] [CrossRef] [PubMed]
- Smolders, E.; Oorts, K.; Van Sprang, P.; Schoeters, I.; Janssen, C.R.; McGrath, S.P.; McLaughlin, M.J. Toxicity of trace metals in soils as affected by soil type and aging after contamination: Using calibrated bioavailability models to set ecological soil standards. Environ. Toxicol. Chem. 2009, 28, 1633–1642. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Zhao, F.; McGrath, S.P.; Luo, Y. Influence of soil properties and aging on arsenic phytotoxicity. Environ. Toxicol. Chem. 2006, 25, 1663–1670. [Google Scholar] [CrossRef] [PubMed]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; FAO: Rome, Italy, 2014. [Google Scholar]
- Smolders, E.; Oorts, K. Development of a Predictive Model of Bioavailability and Toxicity of Copper in Soils: Microbial Toxicity; Final Report of ICA Projects; Laboratory of Soil and Water Management, Katholieke Universiteit: Leuven, Belgium, 2004. [Google Scholar]
- Di Bonito, M.; Breward, N.; Crout, N.; Smith, B.; Young, S. Overview of Selected Soil Pore Water Extraction Methods for the Determination of Potentially Toxic Elements in Contaminated Soils: Operational and Technical Aspects. In Site Characterization, Data Analysis and Case Histories; 208AD; Elsevier: London, UK, 2008; pp. 213–249. [Google Scholar]
- Seefeldt, S.S.; Jensen, J.E.; Fuerst, E.P. Log-logistic analysis of herbicide dose-response relationships. Weed Technol. 1995, 9, 218–227. [Google Scholar] [CrossRef]
- Backhaus, T.; Altenburger, R.; Bodeker, W.; Faust, M.; Scholze, M.; Grimme, L.H. Predictibility of the toxicity of a multiple mixture of dissimilary acting chemicals to Vibrio fischeri. Environ. Toxicol. Chem. 2000, 19, 2348–2356. [Google Scholar] [CrossRef]
- Jonker, M.J.; Svendsen, C.; Bedaux, J.J.M.; Bongers, M.; Kammenga, J.E. Significance testing of synergistic/antagonistic, dose level-dependent, or dose ratio-dependent effects in mixture dose-response analysis. Environ. Toxicol. Chem. 2005, 24, 2701–2713. [Google Scholar] [CrossRef] [PubMed]
- Haas, C.N.; Cidambi, K.; Kersten, S.; Wright, K. Quantitative description of mixture toxicity: Effect of level of response on interactions. Environ. Toxicol. Chem. 1996, 15, 1429–1437. [Google Scholar] [CrossRef]
- Carter, W.H.; Gennings, C. Analysis of Chemical Combinations: Relating Isobolograms to Response. Surfaces. In Toxicology of Chemical Mixtures: Case Studies, Mechanisms, and Novel Approaches; Academic Press: London, UK, 1994; pp. 643–664. [Google Scholar]
- Hamels, F.; Malevé, J.; Sonnet, P.; Berggren-Kleja, D.; Smolders, E. Phytotoxicity of trace metals in spiked and field-contaminated soils: Linking soil-extractable metals with toxicity. Environ. Toxicol. Chem. 2014, 33, 2479–2487. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Miller, D.M. Growth and tissue composition of rice grown in soil treated with inorganic copper, nickel, and arsenic. Commun. Soil Sci. Plant Anal. 1991, 22, 2037–2045. [Google Scholar] [CrossRef]
- Cao, Q.; Hu, Q.H.; Khan, S.; Wang, Z.J.; Lin, A.J.; Du, X.; Zhu, Y.G. Wheat phytotoxicity from arsenic and cadmium separately and together in solution culture and in a calcareous soil. J. Hazard. Mater. 2007, 148, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, S.; Shan, X.; Christie, P. Combined toxicity of cadmium and arsenate to wheat seedlings and plant uptake and antioxidative enzyme responses to cadmium and arsenate co-contamination. Ecotoxicol. Environ. Saf. 2007, 68, 305–313. [Google Scholar] [CrossRef] [PubMed]
| Soil | pH | TOC a | eCEC b | Fe c | Al c | Zn d | As d |
|---|---|---|---|---|---|---|---|
| (CaCl2) | (%) | (cmolc kg−1) | (mg kg−1) | (mg kg−1) | |||
| S1 | 6.9 | 0.9 | 43 | 2200 | 1500 | 1400 | 240 |
| S2 | 6.9 | 3.1 | 18 | 920 | 1200 | 90 | 20 |
| S3 | 6.8 | 1.8 | 17 | 1700 | 870 | 350 | 70 |
| Soil | Soil Total Concentration Dose Expression (mg kg−1) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zn | As | |||||||||||
| EC50 | LCL a | UCL b | β | LCL | UCL | EC50 | LCL | UCL | β | LCL | UCL | |
| S1 | 4200 | 3800 | 4700 | 2.8 | 2.3 | 3.6 | 650 | 690 | 8 | 5.6 | ||
| S2 | 3400 | 3000 | 3800 | 3.4 | 2.8 | 4.5 | 110 | 98 | 110 | 3.4 | 2.8 | 4.2 |
| S3 | 2100 | 1900 | 2300 | 1.7 | 1.5 | 1.9 | 100 | 91 | 100 | 6.2 | 4.2 | 12 |
| Soil Solution Concentration Dose Expression (µmol L−1) | ||||||||||||
| Zn | As | |||||||||||
| EC50 | LCL | UCL | β | LCL | UCL | EC50 | LCL | UCL | β | LCL | UCL | |
| S1 | 180 | 400 | 0.8 | 0.6 | 110 | 73 | 190 | 0.9 | 0.6 | |||
| S2 | 280 | 150 | 800 | 1.0 | 0.8 | 1.8 | 40 | 31 | 52 | 1.2 | 1 | 1.5 |
| S3 | 27 | 20 | 1.4 | 2.4 | 3.5 | 2.8 | 1.8 | 1.2 | ||||
| Soil | Parameter a † | Interpretation of Interactions | |
|---|---|---|---|
| CA | IA | ||
| S1 | 2 ** | 10 * | Significant antagonism according to CA and IA |
| S2 | 0.3 | 0.0 | No significant interaction according to CA and IA |
| S3 | 11 * | 0.5 ** | Significant antagonism according to CA and IA |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzman-Rangel, G.; Montalvo, D.; Smolders, E. Pronounced Antagonism of Zinc and Arsenate on Toxicity to Barley Root Elongation in Soil. Environments 2018, 5, 83. https://doi.org/10.3390/environments5070083
Guzman-Rangel G, Montalvo D, Smolders E. Pronounced Antagonism of Zinc and Arsenate on Toxicity to Barley Root Elongation in Soil. Environments. 2018; 5(7):83. https://doi.org/10.3390/environments5070083
Chicago/Turabian StyleGuzman-Rangel, Georgina, Daniela Montalvo, and Erik Smolders. 2018. "Pronounced Antagonism of Zinc and Arsenate on Toxicity to Barley Root Elongation in Soil" Environments 5, no. 7: 83. https://doi.org/10.3390/environments5070083
APA StyleGuzman-Rangel, G., Montalvo, D., & Smolders, E. (2018). Pronounced Antagonism of Zinc and Arsenate on Toxicity to Barley Root Elongation in Soil. Environments, 5(7), 83. https://doi.org/10.3390/environments5070083




