Applicability of a Freundlich-Like Model for Plant Uptake at an Industrial Contaminated Site with a High Variable Arsenic Concentration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Sampling and Characterization
2.2. Mesocosm Experiment
2.3. Arsenic Quantification and Quality Control
2.4. Statistical Analysis
3. Results and Discussion
3.1. Soil Analysis
3.2. Mesocosm Experiment
3.2.1. Biomass Production
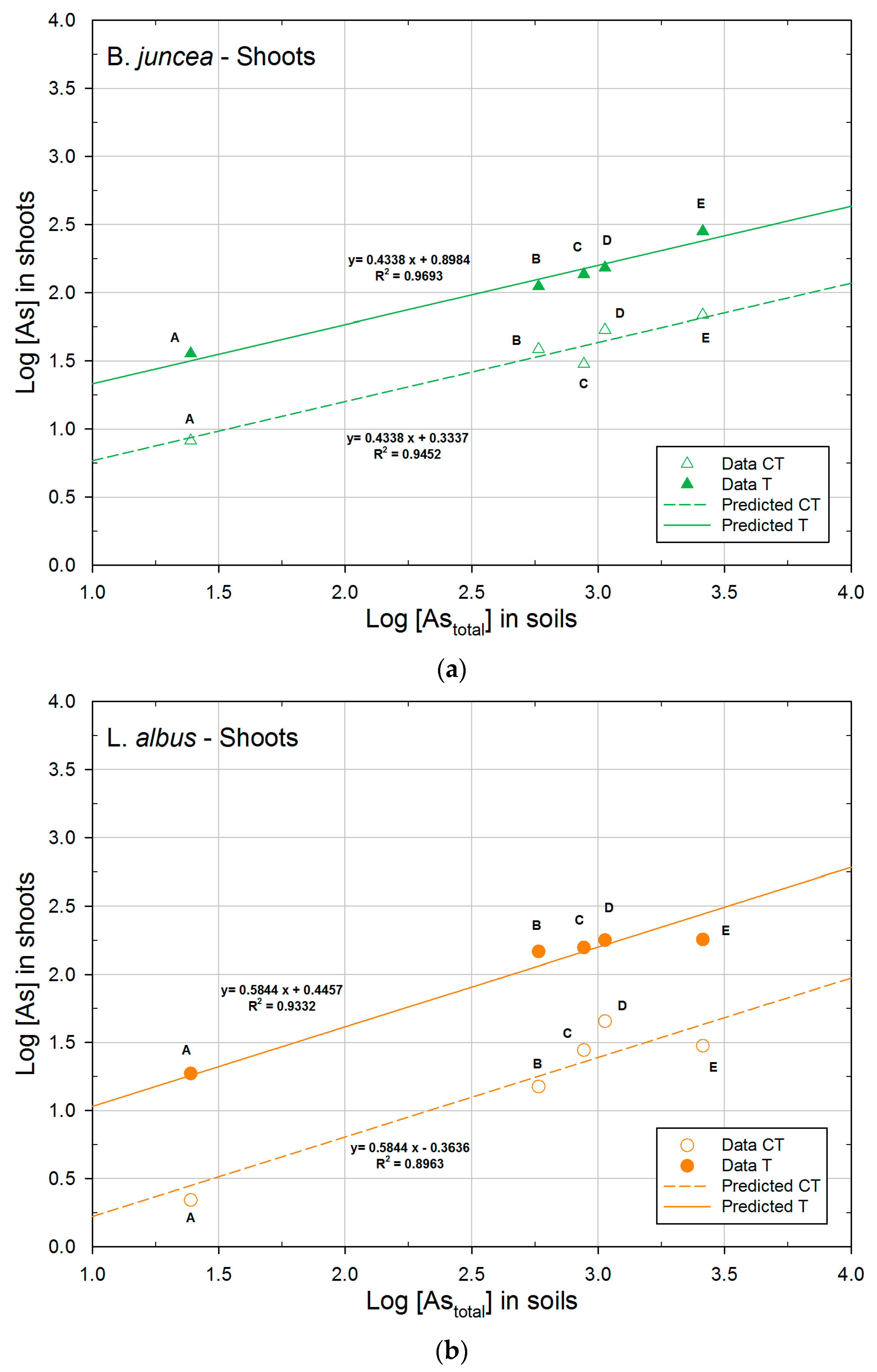
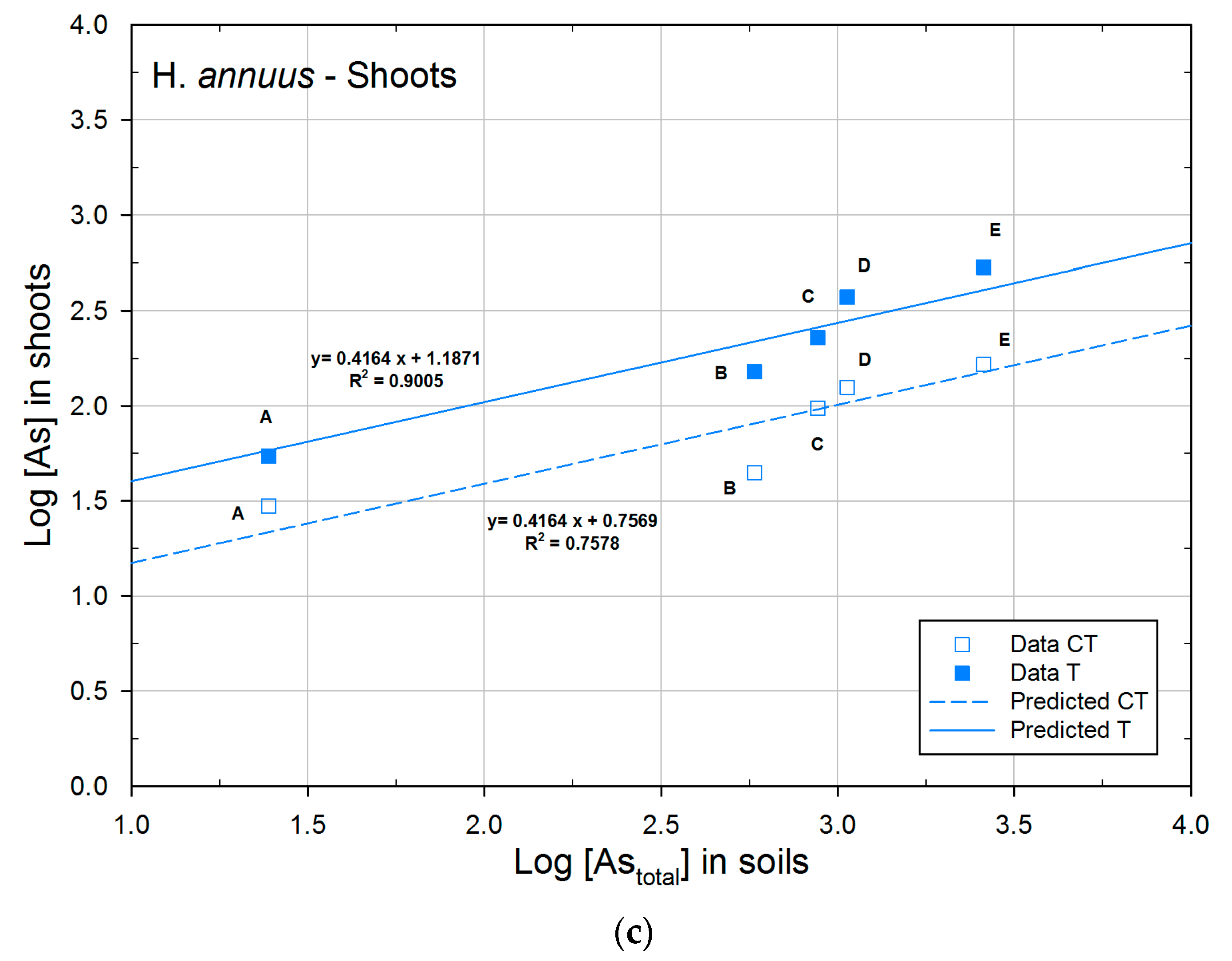
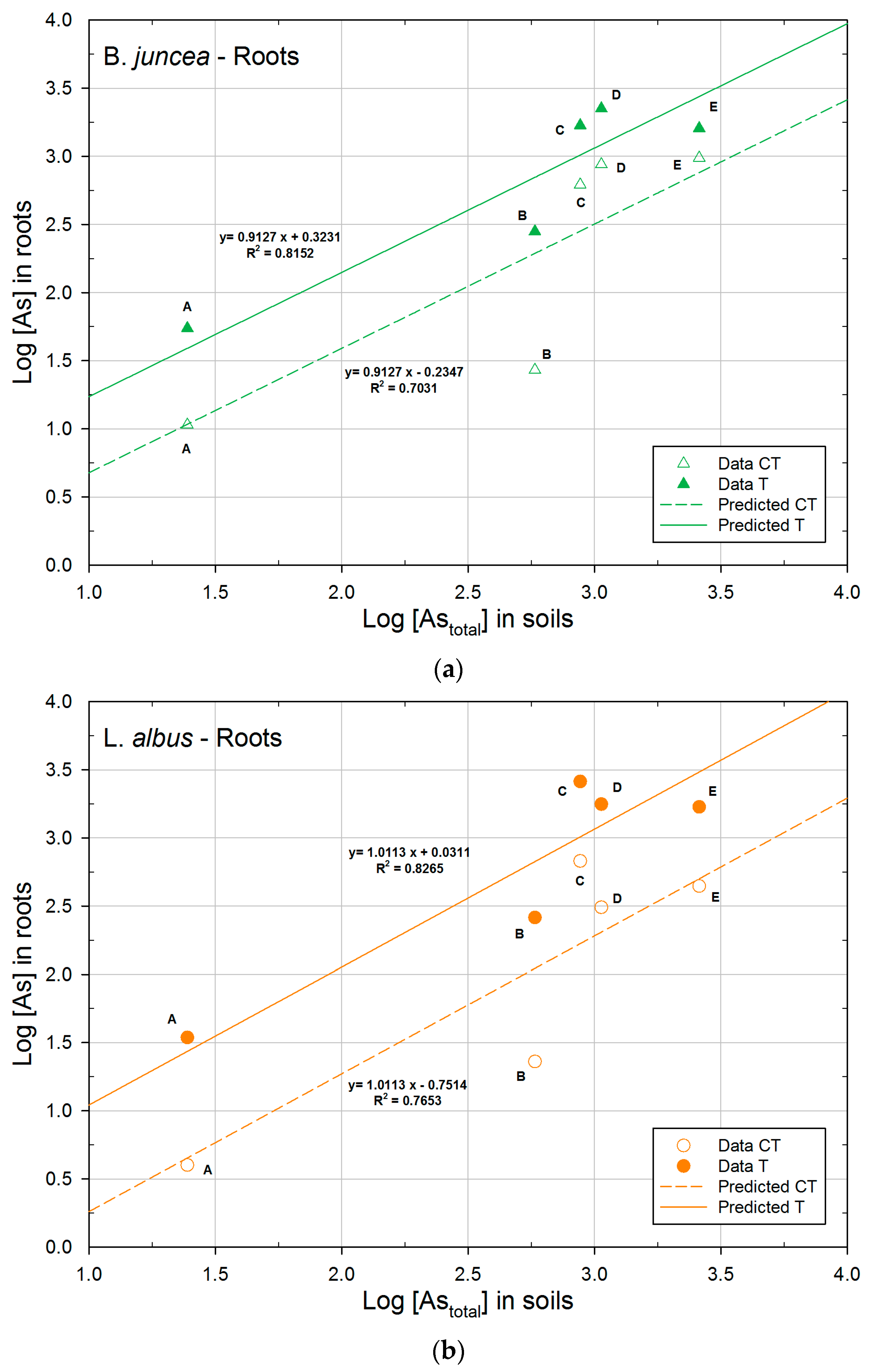
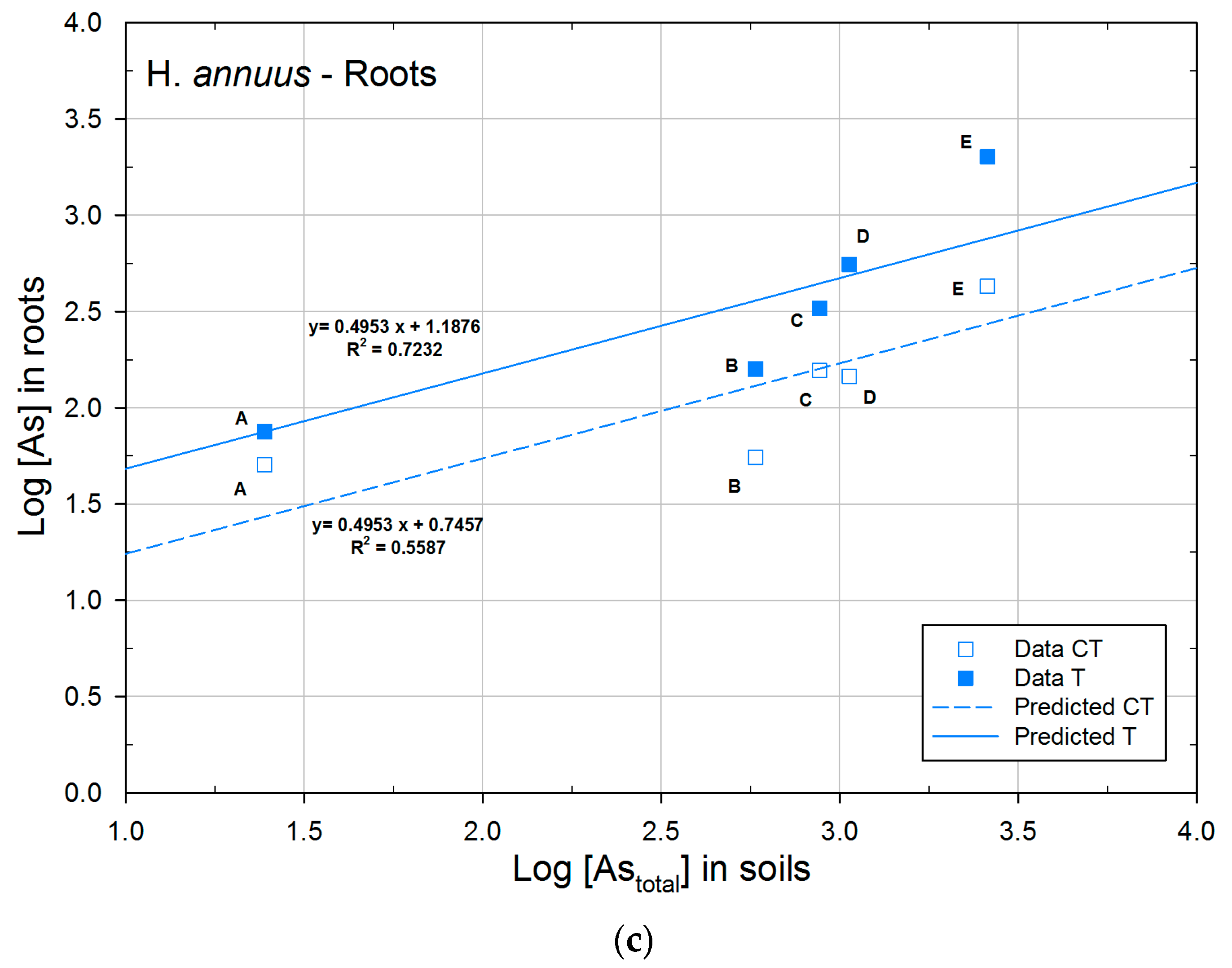
3.2.2. Concentration of Arsenic in Plants
3.3. Phytoextraction Ability
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Garelick, H.; Jones, H.; Dybowska, A.; Valsami-Jones, E. Arsenic Pollution Sources. In Arsenic Pollution and Remediation: An International Perspective; Garelick, H., Jones, H., Eds.; Springer: New York, NY, USA, 2009; pp. 17–60. [Google Scholar]
- Vocciante, M.; Caretta, A.; Bua, L.; Bagatin, R.; Ferro, S. Enhancements in electrokinetic remediation technology: Environmental assessment in comparison with other configurations and consolidated solutions. Chem. Eng. J. 2016, 289, 123–134. [Google Scholar] [CrossRef]
- EPA—United States Environmental Protection Agency. Green Remediation: Incorporating Sustainable Environmental Practices into Remediation of Contaminated Sites. 2008. Available online: https://www.epa.gov/sites/production/files/2015-04/documents/green-remediation-primer.pdf (accessed on 31 August 2017).
- Kim, R.Y.; Yoon, J.K.; Kim, T.S.; Yang, J.E.; Owens, G.; Kim, K.R. Bioavailability of heavy metals in soils: definitions and practical implementation—A critical review. Environ. Geochem. Health 2015, 37, 1041–1061. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Cai, Y.; Tu, C.; Ma, L.Q. Arsenic speciation and distribution in an arsenic hyperaccumulating plant. Sci. Total Environ. 2002, 300, 167–177. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, F.-J.; Meharg, A.A.; Raab, A.; Feldmann, J.; McGrath, S.P. Mechanisms of arsenic hyperaccumulation in Pteris vittata. Uptake kinetics, interactions with phosphate, and arsenic speciation. Plant Physiol. 2002, 130, 1552–1561. [Google Scholar] [CrossRef] [PubMed]
- Lombi, E.; Sletten, R.S.; Wenzel, W.W. Sequentially extracted arsenic from different size fractions of contaminated soils. Water. Air. Soil Pollut. 2000, 124, 319–332. [Google Scholar] [CrossRef]
- Ferrucci, A.; Vocciante, M.; Bagatin, R.; Ferro, S. Electrokinetic remediation of soils contaminated by potentially toxic metals: Dedicated analytical tools for assessing the contamination baseline in a complex scenario. J. Environ. Manag. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.B.A.N.; Dushenkovn, V.; Motto, H.; Raskin, I. Phytoextraction: The use of plants to remove heavy metals from soils. Environ. Sci. Technol. 1995, 29, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Petruzzelli, G.; Pedron, F.; Rosellini, I.; Barbafieri, M. The bioavailability processes as a key to evaluate phytoremediation efficiency. In Phytoremediation; Ansari, A.A., Gill, S.S., Gill, R., Lanza, G.R., Newman, L., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 31–43. [Google Scholar]
- Yoon, J.; Cao, X.; Zhou, Q.; Ma, L.Q. Accumulation of Pb, Cu, and Zn in native plants growing on a contaminated Florida site. Sci. Total Environ. 2006, 368, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Ampiah-Bonney, R.J.; Tyson, J.F.; Lanza, G.R. Phytoextraction of arsenic from soil by Leersia Oryzoides. Int. J. Phytoremediat. 2007, 9, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Souza, L.A.; Piotto, F.A.; Nogueirol, R.C.; Azevedo, R.A. Use of non-hyperaccumulator plant species for the phytoextraction of heavy metals using chelating agents. Sci. Agric. 2013, 70, 290–295. [Google Scholar] [CrossRef]
- Grifoni, M.; Pedron, F.; Petruzzelli, G.; Rosellini, I.; Barbafieri, M.; Franchi, E.; Bagatin, R. Assessment of repeated harvests on mercury and arsenic phytoextraction in a multi-contaminated industrial soil. AIMS Environ. Sci. 2017, 4, 187–205. [Google Scholar] [CrossRef]
- Krauss, M.; Wilcke, W.; Kobza, J.; Zech, W. Predicting heavy metal transfer from soil to plant: Potential use of Freundlich-type functions. J. Plant Nutr. Soil Sci. 2002, 165, 3–8. [Google Scholar] [CrossRef]
- Han, F.X.; Su, Y.; Monts, D.L.; Waggoner, C.A.; Plodinec, M.J. Binding, distribution, and plant uptake of mercury in a soil from Oak Ridge, Tennessee, USA. Sci. Total Environ. 2006, 368, 753–768. [Google Scholar] [CrossRef] [PubMed]
- Blanco Rodrı́guez, P.; Vera Tomé, F.; Lozano, J.C. About the assumption of linearity in soil-to-plant transfer factors for uranium and thorium isotopes and 226Ra. Sci. Total Environ. 2002, 284, 167–175. [Google Scholar] [CrossRef]
- Tuovinen, T.S.; Roivainen, P.; Makkonen, S.; Kolehmainen, M.; Holopainen, T.; Juutilainen, J. Soil-to-plant transfer of elements is not linear: results for five elements relevant to radioactive waste in five boreal forest species. Sci. Total Environ. 2011, 410–411, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Fantke, P.; Charles, R.; de Alencastro, L.F.; Friedrich, R.; Jolliet, O. Plant uptake of pesticides and human health: dynamic modeling of residues in wheat and ingestion intake. Chemosphere 2011, 85, 1639–1647. [Google Scholar] [CrossRef] [PubMed]
- Trapp, S.; Eggen, T. Simulation of the plant uptake of organophosphates and other emerging pollutants for greenhouse experiments and field conditions. Environ. Sci. Pollut. Res. 2013, 20, 4018–4029. [Google Scholar] [CrossRef] [PubMed]
- Molina, M.; Escudey, M.; Chang, A.C.; Chen, W.; Arancibia-Miranda, N. Trace element uptake dynamics for maize (Zea mays L.) grown under field conditions. Plant Soil 2013, 370, 471–483. [Google Scholar] [CrossRef]
- Liang, Z.; Ding, Q.; Wei, D.; Li, J.; Chen, S.; Ma, Y. Major controlling factors and predictions for cadmium transfer from the soil into spinach plants. Ecotoxicol. Environ. Saf. 2013, 93, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Sparks, D.L.; Page, A.L.; Helmke, P.A.; Loeppert, R.H. Methods of Soil Analysis, Part 3—Chemical Methods; Sparks, D.L., Ed.; Soil Science Ssociety of American: Madison, WI, USA, 1996; ISBN 978-0-89118-866-7. [Google Scholar]
- United States Environmental Protection Agency (EPA). Method 3051A: Microwave assisted acid digestion of sediments, sludges, soils, and oils. In Test Methods for Evaluating Solid Waste: Physical/Chemical Methods (SW-846); EPA: Washington, DC, USA, 1995; pp. 3051A-1–3051A-30. [Google Scholar]
- Wenzel, W.W.; Kirchbaumer, N.; Prohaska, T.; Stingeder, G.; Lombi, E.; Adriano, D.C. Arsenic fractionation in soils using an improved sequential extraction procedure. Anal. Chim. Acta 2001, 436, 309–323. [Google Scholar] [CrossRef]
- Barbafieri, M.; Pedron, F.; Petruzzelli, G.; Rosellini, I.; Franchi, E.; Bagatin, R.; Vocciante, M. Assisted phytoremediation of a multi-contaminated soil: Investigation on arsenic and lead combined mobilization and removal. J. Environ. Manage. 2017, 203, 316–329. [Google Scholar] [CrossRef] [PubMed]
- Raab, A.; Schat, H.; Meharg, A.A.; Feldmann, J. Uptake, translocation and transformation of arsenate and arsenite in sunflower (Helianthus annuus): Formation of arsenic-phytochelatin complexes during exposure to high arsenic concentrations. New Phytol. 2005, 168, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, S.; Moreno, E.; Carpena, R.O. Bioavailability of metals and As from acidified multicontaminated soils: Use of white lupin to validate several extraction methods. Environ. Geochem. Health 2008, 30, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Pedron, F.; Petruzzelli, G.; Rosellini, I.; Barbafieri, M.; Franchi, E.; Bagatin, R. Ammonium thiosulphate assisted phytoextraction of mercury and arsenic in multi-polluted industrial soil. Resour. Environ. 2015, 5, 173–181. [Google Scholar] [CrossRef]
- Pedron, F.; Petruzzelli, G.; Barbafieri, M.; Tassi, E. Remediation of a mercury-contaminated industrial soil using bioavailable contaminant stripping. Pedosphere 2013, 23, 104–110. [Google Scholar] [CrossRef]
- Tassi, E.; Pedron, F.; Barbafieri, M.; Petruzzelli, G. Phosphate-assisted phytoextraction in As-contaminated soil. Eng. Life Sci. 2004, 4, 341–346. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (EPA). Method 3052: Microwave assisted acid digestion of siliceous and organically based matrices. In Test Methods for Evaluating Soild Waste: Physical/Chemical Methods (SW-846); EPA: Washington, DC, USA, 1995; pp. 3052-1–3052-20. [Google Scholar]
- Smith, E.; Naidu, R.; Alston, A.M. Chemistry of inorganic arsenic in soils. J. Environ. Qual. 2002, 31, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Bolan, N.; Mahimairaja, S.; Kunhikrishnan, A.; Choppala, G. Phosphorus–arsenic interactions in variable-charge soils in relation to arsenic mobility and bioavailability. Sci. Total Environ. 2013, 463–464, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.; Ma, L.Q. Effects of arsenate and phosphate on their accumulation by an arsenic-hyperaccumulator Pteris vittata L. Plant Soil 2003, 249, 373–382. [Google Scholar] [CrossRef]
- Meharg, A.A. Mechanisms of plant resistance to metal and metalloid ions and potential biotechnological applications. Plant Soil 2005, 274, 163–174. [Google Scholar] [CrossRef]
- Hartley-Whitaker, J.; Ainsworth, G.; Vooijs, R.; Bookum, W.T.; Schat, H.; Meharg, A.A. Phytochelatins are involved in differential arsenate tolerance in Holcus lanatus. Plant Physiol. 2001, 126, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Pigna, M.; Cozzolino, V.; Violante, A.; Meharg, A.A. Influence of phosphate on the arsenic uptake by wheat (Triticum durum L.) irrigated with arsenic solutions at three different concentrations. Water. Air. Soil Pollut. 2009, 197, 371–380. [Google Scholar] [CrossRef]
- Pickering, I.J.; Prince, R.C.; George, M.J.; Smith, R.D.; George, G.N.; Salt, D.E. Reduction and coordination of arsenic in Indian mustard. Plant Physiol. 2000, 122, 1171–1177. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, I. Effects of arsenic concentrations and forms on growth and arsenic uptake and accumulation by Indian mustard (Brassica juncea L.) genotypes. J. Cent. Eur. Agric. 2006, 7, 31–40. [Google Scholar]
- Jankong, P.; Visoottiviseth, P.; Khokiattiwong, S. Enhanced phytoremediation of arsenic contaminated land. Chemosphere 2007, 68, 1906–1912. [Google Scholar] [CrossRef] [PubMed]
- Memarian, R.; Ramamurthy, A.S. Modeling of lead and cadmium uptake by plants in the presence of surfactants. Environ. Monit. Assess. 2013, 185, 2067–2071. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Qu, R.; Zeng, S.; Ding, Q.; Liang, Z.; Wei, D.; Li, J.; Ma, Y. Factors and predictions for cadmium transfer from soils into tomato plants. Commun. Soil Sci. Plant Anal. 2016, 47, 1612–1621. [Google Scholar] [CrossRef]
- Petruzzelli, G.; Pedron, F.; Tassi, E.; Franchi, E.; Bagatin, R.; Agazzi, G.; Barbafieri, M.; Rosellini, I. The effect of thiosulphate on arsenic bioavailability in a multi contaminated soil. A novel contribution to phytoextraction. Res. J. Environ. Earth Sci. 2014, 6, 38–43. [Google Scholar]
- Jarrell, W.M.; Beverly, R.B. The Dilution Effect in Plant Nutrition Studies. Adv. Agron. 1981, 34, 197–224. [Google Scholar]

| Plant | Sample A | Sample B | Sample C | Sample D | Sample E | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | ||
| CT | B. juncea | 2.49b | 0.62c | 2.61b | 0.72c | 3.42c | 0.52b | 2.26b | 0.30b | 3.52b | 0.11a |
| L. albus | 3.20c | 0.43b | 0.81a | 0.21a | 0.71a | 0.11a | 1.22a | 0.11a | 1.82a | 0.52b | |
| H. annuus | 1.65a | 0.20a | 3.95c | 0.31b | 1.71b | 0.12a | 1.51a | 0.11a | 1.32a | 0.11a | |
| T | B. juncea | 2.32a | 0.57b | 2.40a | 0.41c | 3.24b | 0.52c | 3.18b | 0.46b | 3.79c | 0.17a |
| L. albus | 3.84b | 0.59b | 2.18a | 0.34b | 1.49a | 0.28b | 1.52a | 0.14a | 2.04a | 0.41b | |
| H. annuus | 2.74a | 0.28a | 4.59b | 0.27a | 1.73a | 0.11a | 1.44a | 0.11a | 2.59b | 0.21a | |
| Plant | logK | 1/n | R2 | |
|---|---|---|---|---|
| B. juncea | CT | 0.3337a | 0.4338 | 0.9452 |
| T | 0.8984b | 0.4338 | 0.9693 | |
| L. albus | CT | −0.3636a | 0.5844 | 0.8963 |
| T | 0.4457b | 0.5844 | 0.9332 | |
| H. annuus | CT | 0.7569a | 0.4164 | 0.7578 |
| T | 1.1871b | 0.4164 | 0.9005 | |
| Plant | logK | 1/n | R2 | |
|---|---|---|---|---|
| B. juncea | CT | −0.2347a | 0.9127 | 0.7031 |
| T | 0.3231b | 0.9127 | 0.8152 | |
| L. albus | CT | −0.7514a | 1.0113 | 0.7653 |
| T | 0.0311b | 1.0113 | 0.8265 | |
| H. annuus | CT | 0.7457a | 0.4953 | 0.5587 |
| T | 1.1876b | 0.4953 | 0.7232 | |
| Plant | logK | 1/n | R2 | |
|---|---|---|---|---|
| B. juncea | CT | 1.2418a | 0.3348 | 0.7072 |
| T | 1.8065b | 0.3348 | 0.8184 | |
| L. albus | CT | 0.8379a | 0.4786 | 0.8731 |
| T | 1.6472b | 0.4786 | 0.7226 | |
| H. annuus | CT | 1.5868a | 0.3737 | 0.9320 |
| T | 2.0170b | 0.3737 | 0.8857 | |
| Plant | logK | 1/n | R2 | |
|---|---|---|---|---|
| B. juncea | CT | 1.5219a | 0.8979 | 0.9566 |
| T | 2.0796b | 0.8979 | 0.9560 | |
| L. albus | CT | 1.2142a | 0.9705 | 0.9709 |
| T | 1.9966b | 0.9705 | 0.9635 | |
| H. annuus | CT | 1.7300a | 0.4482 | 0.7064 |
| T | 2.1719b | 0.4482 | 0.7402 | |
| Plant | logK | 1/n | R2 | |
|---|---|---|---|---|
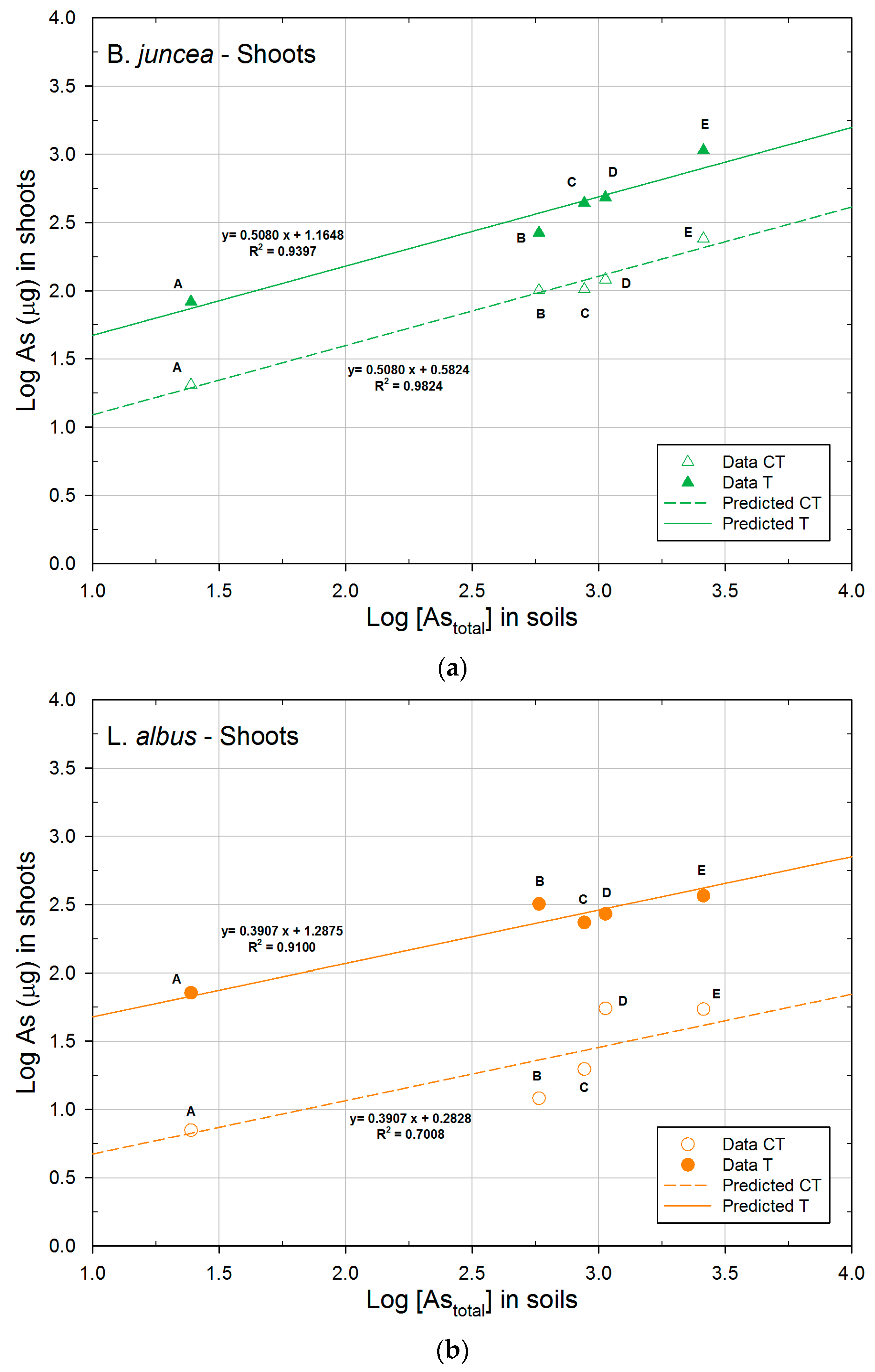
| B. juncea | CT | 0.5824a | 0.5080 | 0.9824 |
| T | 1.1648b | 0.5080 | 0.9397 | |
| L. albus | CT | 0.2828a | 0.3907 | 0.7008 |
| T | 1.2875b | 0.3907 | 0.9100 | |
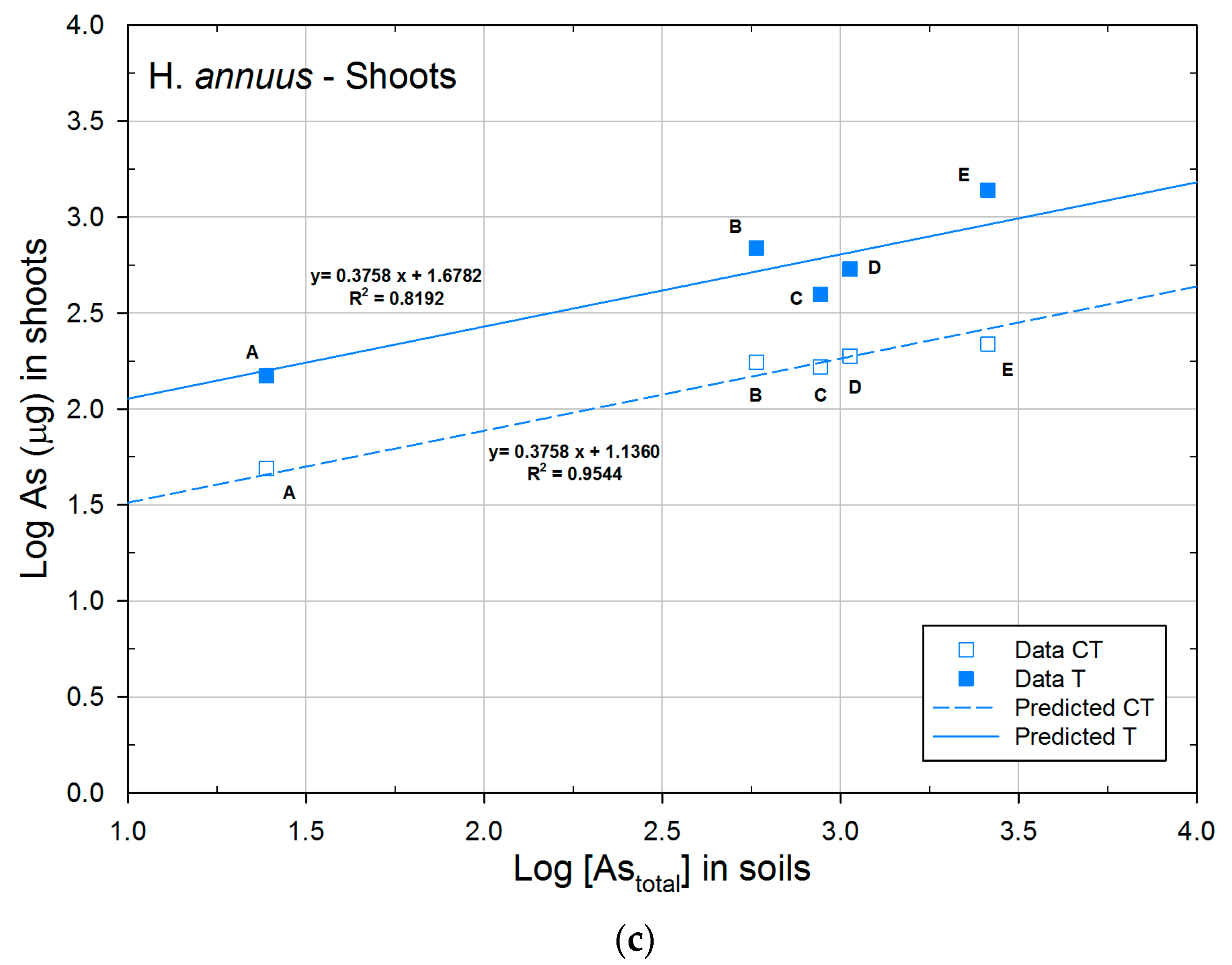
| H. annuus | CT | 1.1360a | 0.3758 | 0.9544 |
| T | 1.6782b | 0.3758 | 0.8192 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pedron, F.; Grifoni, M.; Barbafieri, M.; Petruzzelli, G.; Rosellini, I.; Franchi, E.; Bagatin, R.; Vocciante, M. Applicability of a Freundlich-Like Model for Plant Uptake at an Industrial Contaminated Site with a High Variable Arsenic Concentration. Environments 2017, 4, 67. https://doi.org/10.3390/environments4040067
Pedron F, Grifoni M, Barbafieri M, Petruzzelli G, Rosellini I, Franchi E, Bagatin R, Vocciante M. Applicability of a Freundlich-Like Model for Plant Uptake at an Industrial Contaminated Site with a High Variable Arsenic Concentration. Environments. 2017; 4(4):67. https://doi.org/10.3390/environments4040067
Chicago/Turabian StylePedron, Francesca, Martina Grifoni, Meri Barbafieri, Gianniantonio Petruzzelli, Irene Rosellini, Elisabetta Franchi, Roberto Bagatin, and Marco Vocciante. 2017. "Applicability of a Freundlich-Like Model for Plant Uptake at an Industrial Contaminated Site with a High Variable Arsenic Concentration" Environments 4, no. 4: 67. https://doi.org/10.3390/environments4040067
APA StylePedron, F., Grifoni, M., Barbafieri, M., Petruzzelli, G., Rosellini, I., Franchi, E., Bagatin, R., & Vocciante, M. (2017). Applicability of a Freundlich-Like Model for Plant Uptake at an Industrial Contaminated Site with a High Variable Arsenic Concentration. Environments, 4(4), 67. https://doi.org/10.3390/environments4040067








