Improving Use of Social Communicative Gestures by Children with Autism
Abstract
1. Introduction
1.1. Gesture Development and Taxonomy
Pointing
1.2. Children with Autism and Gestures
1.3. The Present Study
2. Methods
2.1. Participants, Setting, and Materials
2.2. Design
2.3. Dependent Variables
2.4. Assent Measures
2.5. Interobserver Agreement and Treatment Integrity
2.6. General Procedures for Packaged Pointing Intervention
2.6.1. Baseline
2.6.2. Proximal Pointing Treatment
2.6.3. Distal Pointing Generalization
2.6.4. Maintenance
3. Results
3.1. Results by Participant
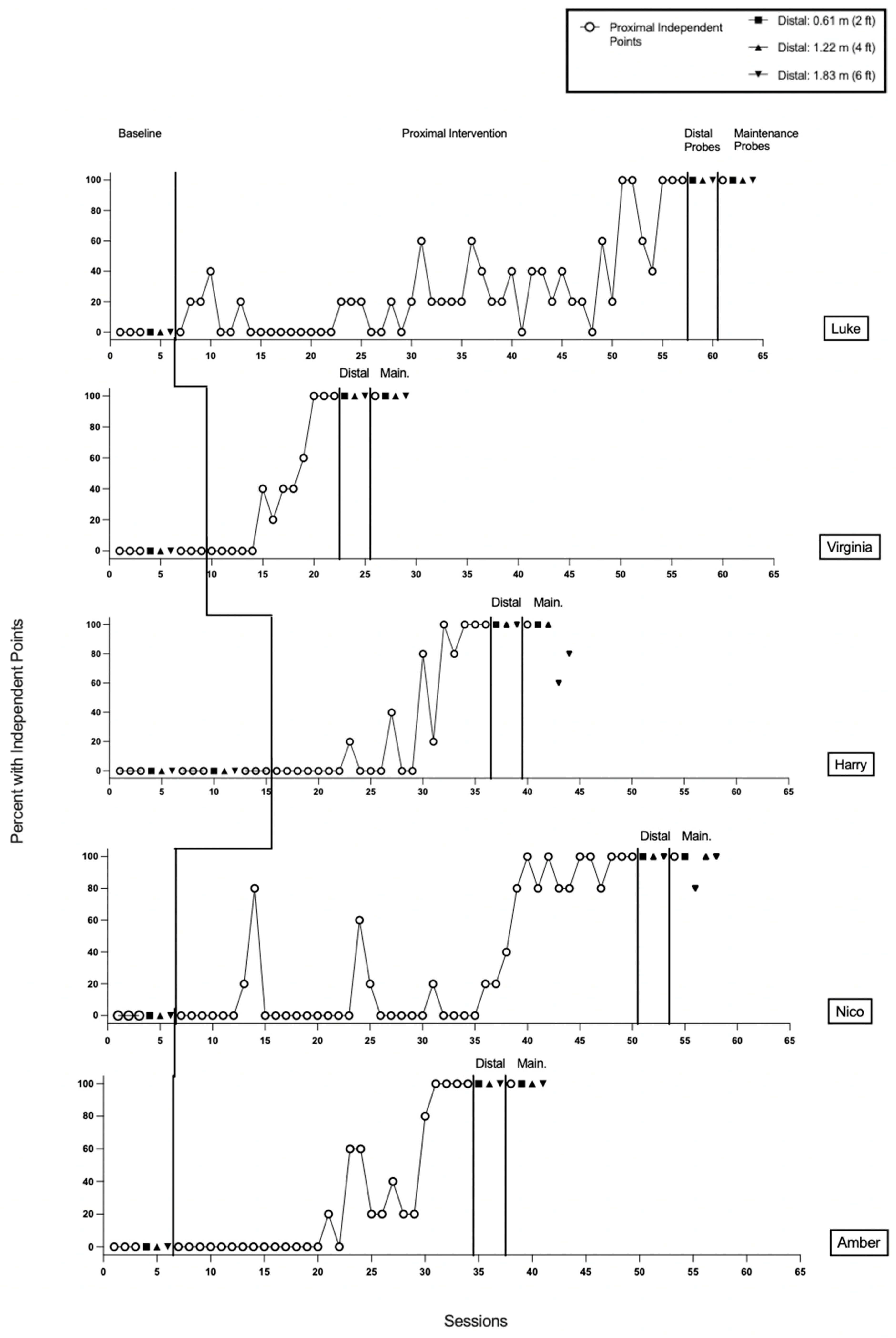
3.1.1. Luke
3.1.2. Virginia
3.1.3. Harry
3.1.4. Nico
3.1.5. Amber
3.1.6. Catherine
3.1.7. Gavin
3.1.8. Jasper
3.1.9. Hazel

3.1.10. Calvin
3.1.11. Esther
3.1.12. Peyton

4. Discussion
4.1. Summary of Findings
4.2. Limitations and Implications for Future Research
4.2.1. Experimental Control and Design Considerations
4.2.2. Session Design and Response Variability
4.2.3. Participant Characterization and Assessment Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABA | applied behavior analysis |
| ASD | autism spectrum disorder |
| BCBA | board-certified behavior analyst |
| CDI: WG | MacArthur–Bates Communicative Development Inventory: Words and Gestures |
| EO | establishing operation |
| ft | feet |
| IOA | interobserver agreement |
| JA | joint attention |
| m | meters |
| M | mean |
| MO | motivating operation |
| NR | not reported |
| TD | typically developing |
| TI | treatment integrity |
| VB-MAPP | Verbal Behavior Milestones Assessment and Placement Program |
| 1 | Individuals and communities may prefer person-first or identity-first language. This article uses both approaches to reflect and respect those preferences. |
References
- Acredolo, L. P., & Goodwyn, S. W. (1988). Symbolic gesturing in normal infants. Child Development, 59(2), 450–466. [Google Scholar] [CrossRef]
- Alcalay, A., Ferguson, J. L., Cihon, J. H., Torres, N., Leaf, J. B., Leaf, R., McEachin, J., Schulze, K. A., & Rudrud, E. H. (2019). Comparing multiple stimulus preference assessments without replacement to in-the-moment reinforcer analysis on rate of responding. Education and Training in Autism and Developmental Disabilities, 54(1), 69–82. [Google Scholar] [CrossRef]
- American Psychiatric Association. (2022). Diagnostic and statistical manual of mental disorders (5th ed., text rev.). American Psychiatric Association. [Google Scholar] [CrossRef]
- Anderson, D. K., Lord, C., Risi, S., DiLavore, P. S., Shulman, C., Thurm, A., Welch, K., & Pickles, A. (2007). Patterns of growth in verbal abilities among children with autism spectrum disorder. Journal of Consulting and Clinical Psychology, 75(4), 594–604. [Google Scholar] [CrossRef] [PubMed]
- Bal, V. H., Kim, S. H., Fok, M., & Lord, C. (2019). Autism spectrum disorder symptoms from ages 2 to 19 years: Implications for diagnosing adolescents and young adults. Autism Research, 12(1), 89–99. [Google Scholar] [CrossRef] [PubMed]
- Bates, E., Camaioni, L., & Volterra, V. (1975). The acquisition of performatives prior to speech. Merrill-Palmer Quarterly, 21(3), 205–226. [Google Scholar]
- Behne, T., Liszkowski, U., Carpenter, M., & Tomasello, M. (2012). Twelve-month-olds’ comprehension and production of pointing. The British Journal of Developmental Psychology, 30(3), 359–375. [Google Scholar] [CrossRef] [PubMed]
- Bourret, J. C., Iwata, B. A., Harper, J. M., & North, S. T. (2012). Elimination of position-biased responding in individuals with autism and intellectual disabilities. Journal of Applied Behavior Analysis, 45(2), 241–250. [Google Scholar] [CrossRef]
- Breaux, C. A., & Smith, K. (2023). Assent in applied behaviour analysis and positive behaviour support: Ethical considerations and practical recommendations. International Journal of Developmental Disabilities, 69(1), 111–121. [Google Scholar] [CrossRef]
- Byiers, B. J., Reichle, J., & Symons, F. J. (2012). Single-subject experimental design for evidence-based practice. American Journal of Speech-Language Pathology, 21(4), 397–414. [Google Scholar] [CrossRef]
- Carpenter, M., Nagell, K., Tomasello, M., Butterworth, G., & Moore, C. (1998). Social cognition, joint attention, and communicative competence from 9 to 15 months of age. Monographs of the Society for Research in Child Development, 63(4), 1–174. [Google Scholar] [CrossRef]
- Chawarska, K., Klin, A., Paul, R., & Volkmar, F. (2007). Autism spectrum disorder in the second year: Stability and change in syndrome expression. Journal of Child Psychology and Psychiatry, 48(2), 128–138. [Google Scholar] [CrossRef]
- Choi, B., & Rowe, M. L. (2025). The role of gesture in language development for neurotypical children and children with or at increased likelihood of autism. Topics in Cognitive Science, 17, 527–544. [Google Scholar] [CrossRef]
- Choi, B., Wei, R., & Rowe, M. L. (2021). Show, give, and point gestures across infancy differentially predict language development. Developmental Psychology, 57(6), 851–862. [Google Scholar] [CrossRef]
- Cooper, J. O., Heron, T. E., & Heward, W. L. (2020). Applied behavior analysis (3rd ed.). Pearson. [Google Scholar]
- Demchak, M. (1990). Response prompting and fading methods: A review. American Journal on Mental Retardation, 94(6), 603–615. [Google Scholar]
- DiStefano, C., & Kasari, C. (2016). The window to language is still open: Distinguishing between preverbal and minimally verbal children with ASD. Perspectives of the ASHA Special Interest Groups, 1(1), 4–11. [Google Scholar] [CrossRef]
- Dube, W. V., MacDonald, R. P. F., Mansfield, R. C., Holcomb, W. L., & Ahearn, W. H. (2004). Toward a behavioral analysis of joint attention. The Behavior Analyst, 27(2), 197–207. [Google Scholar] [CrossRef] [PubMed]
- Frampton, S. E., Axe, J. B., Davis, C. R., Meleshkevich, O., & Li, M.-H. (2024). A tutorial on indicating responses and their importance in mand training. Behavior Analysis in Practice, 17(4), 1238–1249. [Google Scholar] [CrossRef]
- Goldin-Meadow, S. (2009). How gesture promotes learning throughout childhood. Child Development Perspectives, 3(2), 106–111. [Google Scholar] [CrossRef]
- Goldin-Meadow, S., & Alibali, M. W. (2013). Gesture’s role in speaking, learning, and creating language. Annual Review of Psychology, 64, 257–283. [Google Scholar] [CrossRef] [PubMed]
- Iverson, J. M., & Goldin-Meadow, S. (2005). Gesture paves the way for language development. Psychological Science, 16(5), 367–371. [Google Scholar] [CrossRef] [PubMed]
- Iverson, J. M., & Thal, D. J. (1998). Communicative transitions: There’s more to the hand than meets the eye. In A. M. Wetherby, S. F. Warren, & J. Reichle (Eds.), Transitions in prelinguistic communication (pp. 59–86). Paul H. Brookes. [Google Scholar]
- Kasari, C., Brady, N., Lord, C., & Tager-Flusberg, H. (2013). Assessing the minimally verbal school-aged child with autism spectrum disorder. Autism Research, 6(6), 479–493. [Google Scholar] [CrossRef] [PubMed]
- Kazdin, A. E. (2019). Single-case experimental designs. Evaluating interventions in research and clinical practice. Behaviour Research and Therapy, 117, 3–17. [Google Scholar] [CrossRef]
- Laraway, S., Snycerski, S., Michael, J., & Poling, A. (2003). Motivating operations and some terms to describe them: Some further refinements. Journal of Applied Behavior Analysis, 36(4), 407–414. [Google Scholar] [CrossRef] [PubMed]
- LeBarton, E. S., Goldin-Meadow, S., & Raudenbush, S. (2015). Experimentally induced increases in early gesture lead to increases in spoken vocabulary. Journal of Cognition and Development, 16(2), 199–220. [Google Scholar] [CrossRef] [PubMed]
- Marchman, V. A., Dale, P. S., & Fenson, L. (Eds.). (2023). The MacArthur-Bates communicative development inventories: User’s guide and technical manual (3rd ed.). Brookes Publishing. [Google Scholar]
- Michael, J. (1982). Distinguishing between discriminative and motivational functions of stimuli. Journal of the Experimental Analysis of Behavior, 37(1), 149–155. [Google Scholar] [CrossRef]
- Mishra, A., Ceballos, V., Himmelwright, K., McCabe, S., & Scott, L. (2021). Gesture production in toddlers with autism spectrum disorder. Journal of Autism and Developmental Disorders, 51(5), 1658–1667. [Google Scholar] [CrossRef]
- Morris, C., Detrick, J. J., & Peterson, S. M. (2021). Participant assent in behavior analytic research: Considerations for participants with autism and developmental disabilities. Journal of Applied Behavior Analysis, 54(4), 1300–1316. [Google Scholar] [CrossRef]
- Morris, C., Oliveira, J. P., Perrin, J., Federico, C. A., & Martasian, P. J. (2024). Toward a further understanding of assent. Journal of Applied Behavior Analysis, 57(2), 304–318. [Google Scholar] [CrossRef]
- Morris, S. L., Conine, D., Sanz, C. M., Kronfli, F. R., & Mathieson, H. M. (2024). A survey of why and how clinicians change reinforcers during teaching sessions. Behavior Analysis in Practice, 17(4), 815–830. [Google Scholar] [CrossRef]
- Novack, M. A., & Goldin-Meadow, S. (2017). Gesture as representational action: A paper about function. Psychonomic Bulletin & Review, 24(3), 652–665. [Google Scholar] [CrossRef]
- Özçalışkan, Ş., Adamson, L. B., & Dimitrova, N. (2016). Early deictic but not other gestures predict later vocabulary in both typical development and autism. Autism: The International Journal of Research and Practice, 20(6), 754–763. [Google Scholar] [CrossRef]
- Özçalışkan, Ş., & Dimitrova, N. (2013). How gesture input provides a helping hand to language development. Seminars in Speech and Language, 34(4), 227–236. [Google Scholar] [CrossRef]
- Pelaez, M., Virues-Ortega, J., & Gewirtz, J. L. (2012). Acquisition of social referencing via discrimination training in infants. Journal of Applied Behavior Analysis, 45(1), 23–36. [Google Scholar] [CrossRef]
- Ramos-Cabo, S., Vulchanov, V., & Vulchanova, M. (2021). Different ways of making a point: A study of gestural communication in typical and atypical early development. Autism Research, 14(5), 984–996. [Google Scholar] [CrossRef]
- Rose, V., Trembath, D., Keen, D., & Paynter, J. (2016). The proportion of minimally verbal children with autism spectrum disorder in a community-based early intervention programme. Journal of Intellectual Disability Research, 60(5), 464–477. [Google Scholar] [CrossRef]
- Rowe, M. L., & Goldin-Meadow, S. (2009). Early gesture selectively predicts later language learning. Developmental Science, 12(1), 182–187. [Google Scholar] [CrossRef] [PubMed]
- Rowe, M. L., Özçalışkan, S., & Goldin-Meadow, S. (2008). Learning words by hand: Gesture’s role in predicting vocabulary development. First Language, 28(2), 182–199. [Google Scholar] [CrossRef] [PubMed]
- Shaw, K. A., Williams, S., Patrick, M. E., Valencia-Prado, M., Durkin, M. S., Howerton, E. M., Ladd-Acosta, C. M., Pas, E. T., Bakian, A. V., Bartholomew, P., Nieves-Muñoz, N., Sidwell, K., Alford, A., Bilder, D. A., DiRienzo, M., Fitzgerald, R. T., Furnier, S. M., Hudson, A. E., Pokoski, O. M., … Maenner, M. J. (2025). Prevalence and early identification of autism spectrum disorder among children aged 4 and 8 years—Autism and developmental disabilities monitoring network, 16 sites, United States, 2022. MMWR Surveillance Summaries, 74(2), 1–22. [Google Scholar] [CrossRef]
- Sigafoos, J., Woodyatt, G., Keen, D., Tait, K., Tucker, M., Roberts-Pennell, D., & Pittendreigh, N. (2000). Identifying potential communicative acts in children with developmental and physical disabilities. Communication Disorders Quarterly, 21(2), 77–86. [Google Scholar] [CrossRef]
- Skinner, B. F. (1957). Verbal behavior. Appleton-Century-Crofts. [Google Scholar]
- Sundberg, M. L. (2008). VB-MAPP: Verbal behavior milestones assessment and placement program. AVB Press. [Google Scholar]
- Tager-Flusberg, H., & Kasari, C. (2013). Minimally verbal school-aged children with autism spectrum disorder: The neglected end of the spectrum. Autism Research, 6(6), 468–478. [Google Scholar] [CrossRef] [PubMed]
- Toth, K., Dawson, G., Meltzoff, A. N., Greenson, J., & Fein, D. (2007). Early social, imitation, play, and language abilities of young non-autistic siblings of children with autism. Journal of Autism and Developmental Disorders, 37(1), 145–157. [Google Scholar] [CrossRef]
- Touchette, P. E., & Howard, J. S. (1984). Errorless learning: Reinforcement contingencies and stimulus control transfer in delayed prompting. Journal of Applied Behavior Analysis, 17(2), 175–188. [Google Scholar] [CrossRef]
- U.S. Census Bureau. (n.d.). Available online: https://data.census.gov (accessed on 22 January 2024).
- Watson, L. R., Crais, E. R., Baranek, G. T., Dykstra, J. R., & Wilson, K. P. (2013). Communicative gesture use in infants with and without autism: A retrospective home video study. American Journal of Speech-Language Pathology, 22(1), 25–39. [Google Scholar] [CrossRef] [PubMed]
- Wetherby, A. M., Watt, N., Morgan, L., & Shumway, S. (2007). Social communication profiles of children with autism spectrum disorders late in the second year of life. Journal of Autism and Developmental Disorders, 37(5), 960–975. [Google Scholar] [CrossRef] [PubMed]
- Wodka, E. L., Mathy, P., & Kalb, L. (2013). Predictors of phrase and fluent speech in children with autism and severe language delay. Pediatrics, 131(4), e1128–e1134. [Google Scholar] [CrossRef] [PubMed]
| Participant | Age (Years) | Sex | Race and Ethnicity | VB-MAPP Overall Score (/170) | Social Domain Score (/30) | Mand Domain Score (/25) | Vocal Domain Score (/20) | Initial Controlling Prompt Level |
|---|---|---|---|---|---|---|---|---|
| Luke | 5 | Male | White | 44.5 | 6 | 5 | 4.5 | Full physical |
| Virigina | 9 | Female | NR | 46.5 | 6 | 5 | 4 | Model |
| Harry | 3 | Male | White | 12 | 1 | 0 | 1 | Full physical |
| Nico | 4 | Male | White | 13 | NR | NR | NR | Full physical |
| Amber | 4 | Female | Hispanic or Latino | 24.5 | 4.5 | 0.5 | 2 | Full physical |
| Catherine | 4 | Female | White | 15 | 2 | 0 | 2.5 | Full physical |
| Gavin | 11 | Male | Hispanic or Latino and White | 109.5 | 9.5 | 7 | 7 | Model |
| Jasper | 5 | Male | White | 10 | 2 | 0 | 1 | Full physical |
| Hazel | 7 | Female | NR | 30 | 5 | 5 | 3.5 | Full physical |
| Calvin | 9 | Male | NR | 21.5 | 2 | 2 | 2 | Full physical |
| Esther | 3 | Female | Native Hawaiian/Other Pacific Islander | 34 | 3 | 4 | 3 | Full physical |
| Peyton | 3 | Male | Black or African American | 67.5 | 6 | 8.5 | 3 | Full physical |
| Term | Definition |
|---|---|
| Proximal index finger pointing | The child refers to an object that is within 0.30 m (1 ft) by touching it or hovering an extended index finger while the other fingers are curled down and separated from the index finger. Use of the index finger to operate a toy or manipulate an item is not considered a proximal point. |
| Distal index finger pointing | The child refers to a distant object (i.e., more than 0.30 m (1 ft) from the referent) using an extended index finger. The other fingers must be clearly curled down and separated from the index finger. |
| Assent | The child approaches a location and remains within 1.83 m (6 ft) of it, associated with an activity, and/or uses their communication system, vocal or non-vocal, to indicate “yes” to access the presented activity. |
| Withdrawal of assent | The child refuses to enter a location, leaves a location (i.e., exits the door or moves away from the area associated with an activity), and/or uses their communication system, vocal or non-vocal, to indicate “no” or reject the activity. |
| Prompt Type | Prompt |
|---|---|
| Full physical | Adult provides full hand-over-hand physical guidance, isolating the child’s index finger and physically lifting the arm, wrist, or hand to point toward the item. |
| Partial physical | Adult guides part of the child’s upper limb to initiate a pointing gesture with index finger isolation. This may include hand-over-wrist, hand-over-arm, or hand-over-elbow guidance, and/or a shoulder tap to prompt forward arm movement. |
| Model + Partial physical | Adult models the pointing gesture with index finger isolation while simultaneously delivering a partial physical prompt (e.g., light guidance on wrist or elbow). |
| Model | Adult models a pointing gesture with index finger isolation to prompt imitation. |
| Independent | The child initiates and completes the pointing gesture without any physical or modeled prompts from the adult. |
| Participant | Initial Assent (% of Sessions) | Assent Withdrawal (% of Sessions) |
|---|---|---|
| Luke | 100 | 0 |
| Virginia | 100 | 0 |
| Harry | 100 | 0 |
| Nico | 100 | 1.64 |
| Amber | 100 | 0 |
| Catherine | 100 | 0 |
| Gavin | 100 | 0 |
| Jasper | 100 | 0 |
| Hazel | 100 | 0 |
| Calvin | 100 | 0 |
| Esther | 100 | 0 |
| Peyton | 100 | 0 |
| Mean ± SD | 100 ± 0 | 0.92 ± 2.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Barall, R.J.; Shillingsburg, M.A. Improving Use of Social Communicative Gestures by Children with Autism. Behav. Sci. 2026, 16, 401. https://doi.org/10.3390/bs16030401
Barall RJ, Shillingsburg MA. Improving Use of Social Communicative Gestures by Children with Autism. Behavioral Sciences. 2026; 16(3):401. https://doi.org/10.3390/bs16030401
Chicago/Turabian StyleBarall, Rebecca J., and M. Alice Shillingsburg. 2026. "Improving Use of Social Communicative Gestures by Children with Autism" Behavioral Sciences 16, no. 3: 401. https://doi.org/10.3390/bs16030401
APA StyleBarall, R. J., & Shillingsburg, M. A. (2026). Improving Use of Social Communicative Gestures by Children with Autism. Behavioral Sciences, 16(3), 401. https://doi.org/10.3390/bs16030401







