Cognitive Development and Cannabis Use in Adolescents
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedures and Tasks
2.2.1. Procedures
2.2.2. Methods
3. Results
4. Discussion
5. Conclusions and Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Frolli, A.; Ricci, M.C.; Cavallaro, A.; Lombardi, A.; Pastorino, G.M.G.; Operto, F.F. Executive Functions and Cannabis Use in Adolescents. Acta Sci. Neurol. 2020, 3, 54–56. [Google Scholar]
- Giedd, J.N. Structural Magnetic Resonance Imaging of the Adolescent Brain. Ann. N. Y. Acad. Sci. 2004, 1021, 77–85. [Google Scholar] [CrossRef]
- Shaw, P.; Greenstein, D.; Lerch, J.; Clasen, L.; Lenroot, R.; Gogtay, N.; Evans, A.; Rapoport, J.; Giedd, J. Intellectual ability and cortical development in children and adolescents. Nat. Cell Biol. 2006, 440, 676–679. [Google Scholar] [CrossRef] [PubMed]
- Winters, K.C.; Lee, C.-Y.S. Likelihood of developing an alcohol and cannabis use disorder during youth: Association with recent use and age. Drug Alcohol Depend. 2008, 92, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Gogtay, N.; Giedd, J.N.; Lusk, L.; Hayashi, K.M.; Greenstein, D.; Vaituzis, A.C.; Nugent, T.F.; Herman, D.H.; Clasen, L.S.; Toga, A.W.; et al. Dynamic mapping of human cortical development during childhood through early adulthood. Proc. Natl. Acad. Sci. USA 2004, 101, 8174–8179. [Google Scholar] [CrossRef] [PubMed]
- Somerville, L.H.; Hare, T.A.; Casey, B.J. Frontostriatal Maturation Predicts Cognitive Control Failure to Appetitive Cues in Adolescents. J. Cogn. Neurosci. 2011, 23, 2123–2134. [Google Scholar] [CrossRef]
- Martins, S.S.; Storr, C.L.; Alexandre, P.K.; Chilcoat, H.D. Adolescent ecstasy and other drug use in the National Survey of Parents and Youth: The role of sensation-seeking, parental monitoring and peer’s drug use. Addict. Behav. 2008, 33, 919–933. [Google Scholar] [CrossRef] [PubMed]
- Reyna, V.F.; Farley, F. Risk and rationality in adolescent decision making: Implications for theory, practice, and public policy. Psychol. Sci. Public Interest 2006, 7, 1–44. [Google Scholar] [CrossRef]
- Romer, D.; Hennessy, M. A Biosocial-Affect Model of Adolescent Sensation Seeking: The Role of Affect Evaluation and Peer-Group Influence in Adolescent Drug Use. Prev. Sci. 2007, 8, 89–101. [Google Scholar] [CrossRef]
- Gardner, M.; Steinberg, L. Peer Influence on Risk Taking, Risk Preference, and Risky Decision Making in Adolescence and Adulthood: An Experimental Study. Dev. Psychol. 2005, 41, 625–635. [Google Scholar] [CrossRef]
- Rubino, T.; Zamberletti, E.; Parolaro, D.C. Uso di cannabis in adolescenza come fattore di rischio per le malattie psichiatriche e la dipendenza da altre droghe. Cannabis Danni Salute 2011. Available online: http://www.spaziosociale.it/public/allegati/CSAA0073.pdf (accessed on 17 March 2021).
- Dougherty, D.M.; Mathias, C.W.; Dawes, M.A.; Furr, R.M.; Charles, N.E.; Liguori, A.; Shannon, E.E.; Acheson, A. Impulsivity, attention, memory, and decision-making among adolescent marijuana users. Psychopharmacology 2013, 226, 307–319. [Google Scholar] [CrossRef]
- Jacobsen, L.K.; Mencl, W.E.; Westerveld, M.; Pugh, K.R. Impact of Cannabis Use on Brain Function in Adolescents. Ann. N. Y. Acad. Sci. 2004, 1021, 384–390. [Google Scholar] [CrossRef]
- Fried, P.; Watkinson, B.; Gray, R. Neurocognitive consequences of marihuana—A comparison with pre-drug performance. Neurotoxicology Teratol. 2005, 27, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Lisdahl, K.M.; Price, J.S. Increased Marijuana Use and Gender Predict Poorer Cognitive Functioning in Adolescents and Emerging Adults. J. Int. Neuropsychol. Soc. 2012, 18, 678–688. [Google Scholar] [CrossRef] [PubMed]
- Medina, K.L.; Hanson, K.L.; Schweinsburg, A.D.; Cohen-Zion, M.; Nagel, B.J.; Tapert, S.F. Neuropsychological functioning in adolescent marijuana users: Subtle deficits detectable after a month of abstinence. J. Int. Neuropsychol. Soc. 2007, 13, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Broyd, S.J.; Van Hell, H.H.; Beale, C.; Yücel, M.; Solowij, N. Acute and Chronic Effects of Cannabinoids on Human Cognition—A Systematic Review. Biol. Psychiatry 2016, 79, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Hindocha, C.; Freeman, T.P.; Xia, J.X.; Shaban, N.D.; Curran, H.V. Acute memory and psychotomimetic effects of cannabis and tobacco both ‘joint’ and individually: A placebo-controlled trial. Psychol. Med. 2017, 47, 2708–2719. [Google Scholar] [CrossRef]
- Fletcher, J.M.; Page, J.B.; Francis, D.J.; Copeland, K.; Naus, M.J.; Davis, C.M.; Morris, R.; Krauskopf, D.; Satz, P. Cognitive Correlates of Long-term Cannabis Use in Costa Rican Men. Arch. Gen. Psychiatry 1996, 53, 1051. [Google Scholar] [CrossRef]
- Pope, H.G.; Yurgelun-Todd, D. The residual cognitive effects of heavy marijuana use in college students. JAMA 1996, 275, 521–527. [Google Scholar] [CrossRef]
- Solowij, N.; Stephens, R.S.; Roffman, R.A.; Babor, T.; Kadden, R.; Miller, M.; Christiansen, K.; McRee, B.; Vendetti, J. Cognitive Functioning of Long-term Heavy Cannabis Users Seeking Treatment. JAMA 2002, 287, 1123–1131. [Google Scholar] [CrossRef]
- Porath-Waller, A.J.; Brown, J.E.; Frigon, A.P.; Clark, H. What Canadian Youth Think about Cannabis; Canadian Centre Substance Abuse: Ottawa, ON, Canada, 2013; p. 57. [Google Scholar]
- Cousijn, J.; Wiers, R.W.; Ridderinkhof, K.R.; Brink, W.V.D.; Veltman, D.J.; Goudriaan, A.E. Effect of baseline cannabis use and working-memory network function on changes in cannabis use in heavy cannabis users: A prospective fMRI study. Hum. Brain Mapp. 2013, 35, 2470–2482. [Google Scholar] [CrossRef]
- Solowij, N.; Battisti, R. Current drug abuse reviews. The Chronic Effects of Cannabis on Memory in Humans: A Review; Bentham Science Publishers: Sharjah, UAE, 2008; Volume 1, pp. 81–98. [Google Scholar]
- Battisti, R.A.; Roodenrys, S.; Johnstone, S.J.; Respondek, C.; Hermens, D.F.; Solowij, N. Chronic use of cannabis and poor neural efficiency in verbal memory ability. Psychopharmacology 2010, 209, 319–330. [Google Scholar] [CrossRef]
- Jager, G.; Kahn, R.S.; Brink, W.V.D.; Van Ree, J.M.; Ramsey, N.F. Long-term effects of frequent cannabis use on working memory and attention: An fMRI study. Psychopharmacology 2006, 185, 358–368. [Google Scholar] [CrossRef]
- Jager, G.; Van Hell, H.H.; De Win, M.M.L.; Kahn, R.S.; Brink, W.V.D.; Van Ree, J.M.; Ramsey, N.F. Effects of frequent cannabis use on hippocampal activity during an associative memory task. Eur. Neuropsychopharmacol. 2007, 17, 289–297. [Google Scholar] [CrossRef]
- Solowij, N.; Michie, P.T.; Fox, A.M. Effects of long-term cannabis use on selective attention: An event-related potential study. Pharmacol. Biochem. Behav. 1991, 40, 683–688. [Google Scholar] [CrossRef]
- Solowij, N.; Michie, P.T.; Fox, A.M. Differential impairments of selective attention due to frequency and duration of cannabis use. Biol. Psychiatry 1995, 37, 731–739. [Google Scholar] [CrossRef]
- Solowij, N.; Yücel, M.; Lorenzetti, V.; Lubman, D.I. Structural Brain Alterations in Cannabis Users: Association with Cognitive Deficits and Psychiatric Symptoms. In The Handbook of Neuropsychiatric Biomarkers, Endophenotypes and Genes; Metzler, J.B., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 215–225. [Google Scholar]
- Block, R.I.; O’Leary, D.S.; Hichwa, R.D.; Augustinack, J.C.; Ponto, L.L.B.; Ghoneim, M.; Arndt, S.; Hurtig, R.R.; Watkins, G.; Hall, J.A.; et al. Effects of frequent marijuana use on memory-related regional cerebral blood flow. Pharmacol. Biochem. Behav. 2002, 72, 237–250. [Google Scholar] [CrossRef]
- Dahlgren, M.K.; Sagar, K.A.; Racine, M.T.; Dreman, M.W.; Gruber, S.A. Marijuana use predicts cognitive performance on tasks of executive function. J. Stud. Alcohol Drugs 2016, 77, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, L.K.; Pugh, K.R.; Constable, R.T.; Westerveld, M.; Mencl, W.E. Functional Correlates of Verbal Memory Deficits Emerging During Nicotine Withdrawal in Abstinent Adolescent Cannabis Users. Biol. Psychiatry 2007, 61, 31–40. [Google Scholar] [CrossRef]
- Solowij, N.; Edwards, G. Cannabis and Cognitive Functioning; Amsterdam University Press: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Orsini, A.; Pezzuti, L.; Picone, L. WISC-IV: Contributo Alla Taratura Italiana; (Italian Edition); Giunti, O.S., Ed.; Organizzazioni Speciali: Firenze, Italy, 2012. [Google Scholar]
- Gugliotta, M.; Bisiacchi, P.S.; Cendron, M.; Tressoldi, P.E.; Vio, C. BVN 12-18-Batteria per la Valutazione Neuropsicologica per l’adolescenza; Edizioni Erickson: Trento, Italy, 2009. [Google Scholar]
- Cornoldi, C.; Baldi, A.P.; Giofrè, D. Prove MT Avanzate-3-Clinica; Giunti Edu: Firenze, Italy, 2017. [Google Scholar]
- Corp, IBM. Statistiche IBM SPSS per Windows, versione 26.0; IBM Corp: Armonk, NY, USA, 2019. [Google Scholar]
- Harvey, M.A.; Sellman, J.D.; Porter, R.J.; Frampton, C.M. The relationship between non-acute adolescent cannabis use and cognition. Drug Alcohol Rev. 2007, 26, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.C.; Wolf, D.H.; Calkins, M.E.; Bach, E.C.; Weidner, J.; Ruparel, K.; Moore, T.M.; Jones, J.D.; Jackson, C.T.; Gur, R.E.; et al. Cognitive functioning of adolescent and young adult cannabis users in the Philadelphia Neurodevelopmental Cohort. Psychol. Addict. Behav. 2017, 31, 423–434. [Google Scholar] [CrossRef]
- Kang-Park, M.-H.; Wilson, W.A.; Kuhn, C.M.; Moore, S.D.; Swartzwelder, H.S. Differential Sensitivity of GABAA Receptor-Mediated IPSCs to Cannabinoids in Hippocampal Slices from Adolescent and Adult Rats. J. Neurophysiol. 2007, 98, 1223–1230. [Google Scholar] [CrossRef]
- Smith, M.J.; Cobia, D.J.; Reilly, J.L.; Gilman, J.M.; Roberts, A.G.; Alpert, K.I.; Wang, L.; Breiter, H.C.; Csernansky, J.G. Cannabis-related episodic memory deficits and hippocampal morphological differences in healthy individuals and schizophrenia subjects. Hippocampus 2015, 25, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Lisdahl, K.M.; Wright, N.E.; Medina-Kirchner, C.; Maple, K.E.; Shollenbarger, S. Considering Cannabis: The Effects of Regular Cannabis Use on Neurocognition in Adolescents and Young Adults. Curr. Addict. Rep. 2014, 1, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Rosser, R.; Stevens, S.; Ruiz, B. Cognitive markers of adolescent risk taking: A correlate of drug abuse in at-risk individuals. Prison J. 2005, 85, 83–96. [Google Scholar] [CrossRef]
- Ganzer, F.; Bröning, S.; Kraft, S.; Sack, P.-M.; Thomasius, R. Weighing the Evidence: A Systematic Review on Long-Term Neurocognitive Effects of Cannabis Use in Abstinent Adolescents and Adults. Neuropsychol. Rev. 2016, 26, 186–222. [Google Scholar] [CrossRef] [PubMed]
- Rubino, T.; Vigano’, D.; Realini, N.; Guidali, C.; Braida, D.; Capurro, V.; Castiglioni, C.; Cherubino, F.; Romualdi, P.; Candeletti, S.; et al. Chronic delta (9) -tetrahydrocannabinol during adolescence provokes sex-dependent changes in the emotional profile in adult rats: Behavioral and biochemical correlates. Neuropsychopharmacology 2008, 33, 2760–2771. [Google Scholar] [CrossRef]
- Cohen-Zion, M.; Drummond, S.P.; Padula, C.B.; Winward, J.; Kanady, J.; Medina, K.L.; Tapert, S.F. Sleep architecture in adolescent marijuana and alcohol users during acute and extended abstinence. Addict. Behav. 2009, 34, 976–979. [Google Scholar] [CrossRef]
- Batalla, A.; Bhattacharyya, S.; Yücel, M.; Fusar-Poli, P.; Crippa, J.A.; Nogué, S.; Torrens, M.; Pujol, J.; Farré, M.; Martin-Santos, R. Structural and Functional Imaging Studies in Chronic Cannabis Users: A Systematic Review of Adolescent and Adult Findings. PLoS ONE 2013, 8, e55821. [Google Scholar] [CrossRef]
- Volkow, N.D.; Baler, R.D.; Compton, W.M.; Weiss, S.R. Adverse Health Effects of Marijuana Use. New Engl. J. Med. 2014, 370, 2219–2227. [Google Scholar] [CrossRef] [PubMed]
- Di Forti, M.; Marconi, A.; Carra, E.; Fraietta, S.; Trotta, A.; Bonomo, M.; Bianconi, F.; Gardner-Sood, P.; O’Connor, J.; Russo, M.; et al. Proportion of patients in south London with first-episode psychosis attributable to use of high potency cannabis: A case-control study. Lancet Psychiatry 2015, 2, 233–238. [Google Scholar] [CrossRef]
- Chan, G.C.-K.; Hinds, T.R.; Impey, S.; Storm, D.R. Hippocampal Neurotoxicity of Δ9-Tetrahydrocannabinol. J. Neurosci. 1998, 18, 5322–5332. [Google Scholar] [CrossRef] [PubMed]
- Lawston, J.; Borella, A.; Robinson, J.K.; Whitaker-Azmitia, P.M. Changes in hippocampal morphology following chronic treatment with the synthetic cannabinoid WIN 55,212-2. Brain Res. 2000, 877, 407–410. [Google Scholar] [CrossRef]
- Wilson, W.; Mathew, R.; Turkington, T.; Hawk, T.; Coleman, R.E.; Provenzale, J. Brain morphological changes and early marijuana use: A magnetic resonance and positron emission tomography study. J. Addict. Dis. 2000, 19, 1–22. [Google Scholar] [CrossRef]
- Yücel, M.; Solowij, N.; Respondek, C.; Whittle, S.; Fornito, A.; Pantelis, C.; Lubman, D.I. Regional Brain Abnormalities Associated with Long-term Heavy Cannabis Use. Arch. Gen. Psychiatry 2008, 65, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Lubman, D.I.; Cheetham, A.; Yücel, M. Cannabis and adolescent brain development. Pharmacol. Ther. 2015, 148, 1–16. [Google Scholar] [CrossRef]
- Kolb, B.; Gorny, G.; Limebeer, C.L.; Parker, L.A. Chronic treatment with Δ-9-tetrahydrocannabinol alters the structure of neurons in the nucleus accumbens shell and medial prefrontal cortex of rats. Synapse 2006, 60, 429–436. [Google Scholar] [CrossRef]
- Downer, E.; Boland, B.; Fogarty, M.; Campbell, V. Δ9-Tetrahydrocannabinol induces the apoptotic pathway in cultured cortical neurones via activation of the CB1 receptor. NeuroReport 2001, 12, 3973–3978. [Google Scholar] [CrossRef]
- Lorenzetti, V.; Solowij, N.; Yücel, M. The Role of Cannabinoids in Neuroanatomic Alterations in Cannabis Users. Biol. Psychiatry 2016, 79, e17–e31. [Google Scholar] [CrossRef]
- Lorenzetti, V.; Solowij, N.; Fornito, A.; Lubman, D.I.; Yucel, M. The association between regular cannabis exposure and alterations of human brain morphology: An updated review of the literature. Curr. Pharm. Des. 2014, 20, 2138–2167. [Google Scholar] [CrossRef] [PubMed]
- Hyman, S.E.; Malenka, R.C.; Nestler, E.J. Neural Mechanisms of Addiction: The Role of Reward-Related Learning and Memory. Annu. Rev. Neurosci. 2006, 29, 565–598. [Google Scholar] [CrossRef]
- Ramus, S.J.; Davis, J.B.; Donahue, R.J.; Discenza, C.B.; Waite, A.A. Interactions between the Orbitofrontal Cortex and the Hippocampal Memory System during the Storage of Long-Term Memory. Ann. N. Y. Acad. Sci. 2007, 1121, 216–231. [Google Scholar] [CrossRef]
- Rueda, M.R.; Posner, M.I.; Rothbart, M.K. The Development of Executive Attention: Contributions to the Emergence of Self-Regulation. Dev. Neuropsychol. 2005, 28, 573–594. [Google Scholar] [CrossRef]
- Bressler, S.L.; Menon, V. Large-scale brain networks in cognition: Emerging methods and principles. Trends Cogn. Sci. 2010, 14, 277–290. [Google Scholar] [CrossRef]
- Vincent, J.L.; Kahn, I.; Snyder, A.Z.; Raichle, M.E.; Buckner, R.L. Evidence for a Frontoparietal Control System Revealed by Intrinsic Functional Connectivity. J. Neurophysiol. 2008, 100, 3328–3342. [Google Scholar] [CrossRef] [PubMed]
- Solowij, N.; Jones, K.A.; Rozman, M.E.; Davis, S.M.; Ciarrochi, J.; Heaven, P.C.L.; Lubman, D.I.; Yücel, M. Verbal learning and memory in adolescent cannabis users, alcohol users and non-users. Psychopharmacology 2011, 216, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Takagi, M.; Yücel, M.; Cotton, S.M.; Baliz, Y.; Tucker, A.; Elkins, K.; Lubman, D.I. Verbal memory, learning, and executive functioning among adolescent inhalant and cannabis users. J. Stud. Alcohol Drugs 2011, 72, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Takagi, M.; Lubman, D.I.; Cotton, S.; Fornito, A.; Baliz, Y.; Tucker, A.; Yűcel, M. Executive control among adolescent inhalant and cannabis users. Drug Alcohol Rev. 2010, 30, 629–637. [Google Scholar] [CrossRef]
- Morin, J.F.G.; Afzali, M.H.; Bourque, J.; Stewart, S.H.; Séguin, J.R.; O’Leary-Barrett, M.; Conrod, P.J. A population-based analysis of the relationship between substance use and adolescent cognitive development. Am. J. Psychiatry 2019, 176, 98–106. [Google Scholar] [CrossRef]
- Schweinsburg, A.D.; Nagel, B.J.; Schweinsburg, B.C.; Park, A.; Theilmann, R.J.; Tapert, S.F. Abstinent adolescent marijuana users show altered fMRI response during spatial working memory. Psychiatry Res. Neuroimaging 2008, 163, 40–51. [Google Scholar] [CrossRef]
- Medina, K.L.; Tapert, S.F. Effetti cronici dell’uso di marijuana sul cervello e sulle capacità cognitive dell’adolescente. In Cannabis e Danni alla Salute—Aspetti Tossicologici, Neuropsichici, Medici, Sociali e Linee di Indirizzo per la Prevenzione e il Trattamento; Serpelloni, G., Diana, M., Gomma, M., Rimondo, C., Eds.; (a cura di) Cierre Grafica: Verona, Italy, 2011. [Google Scholar]
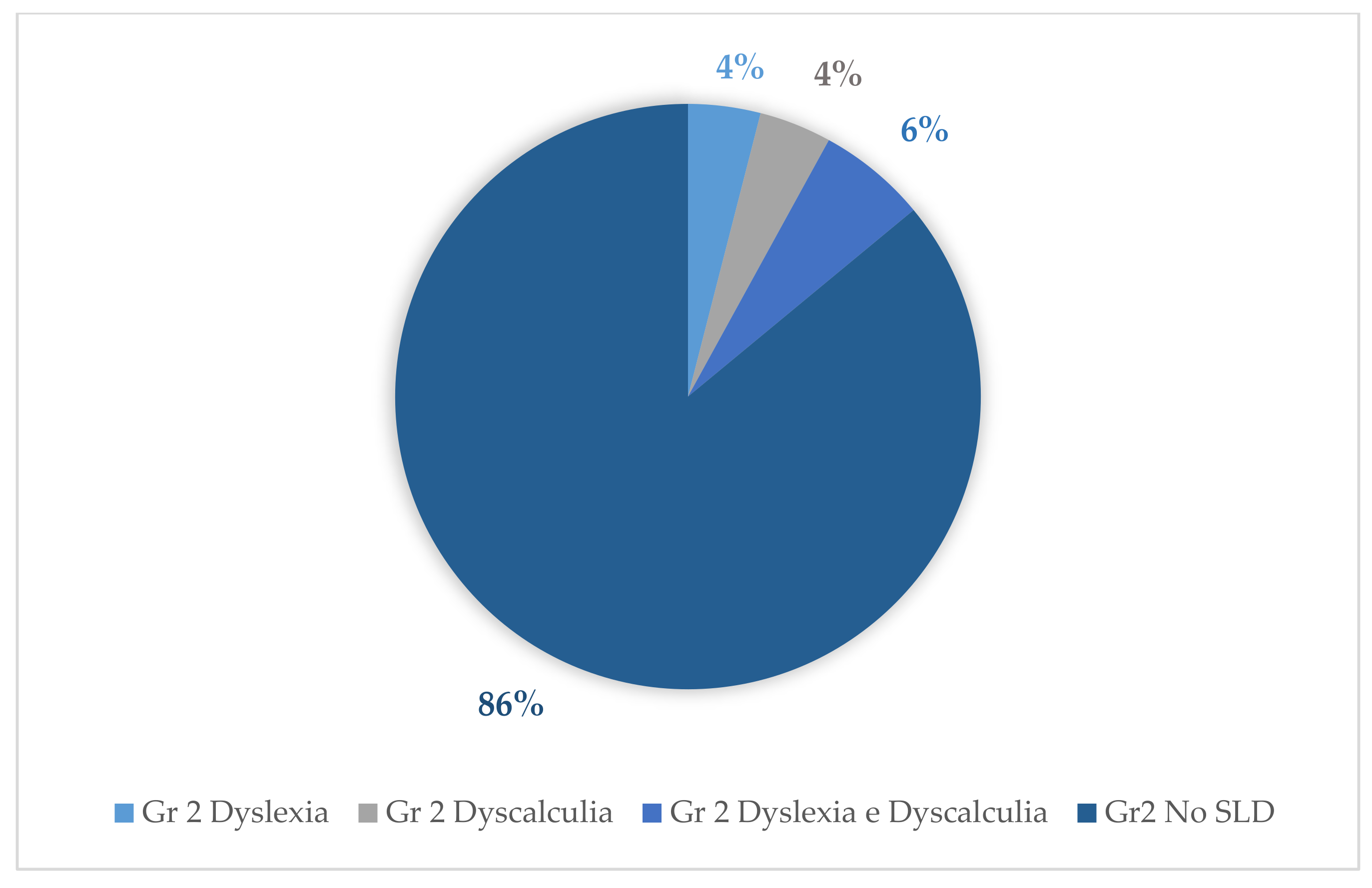
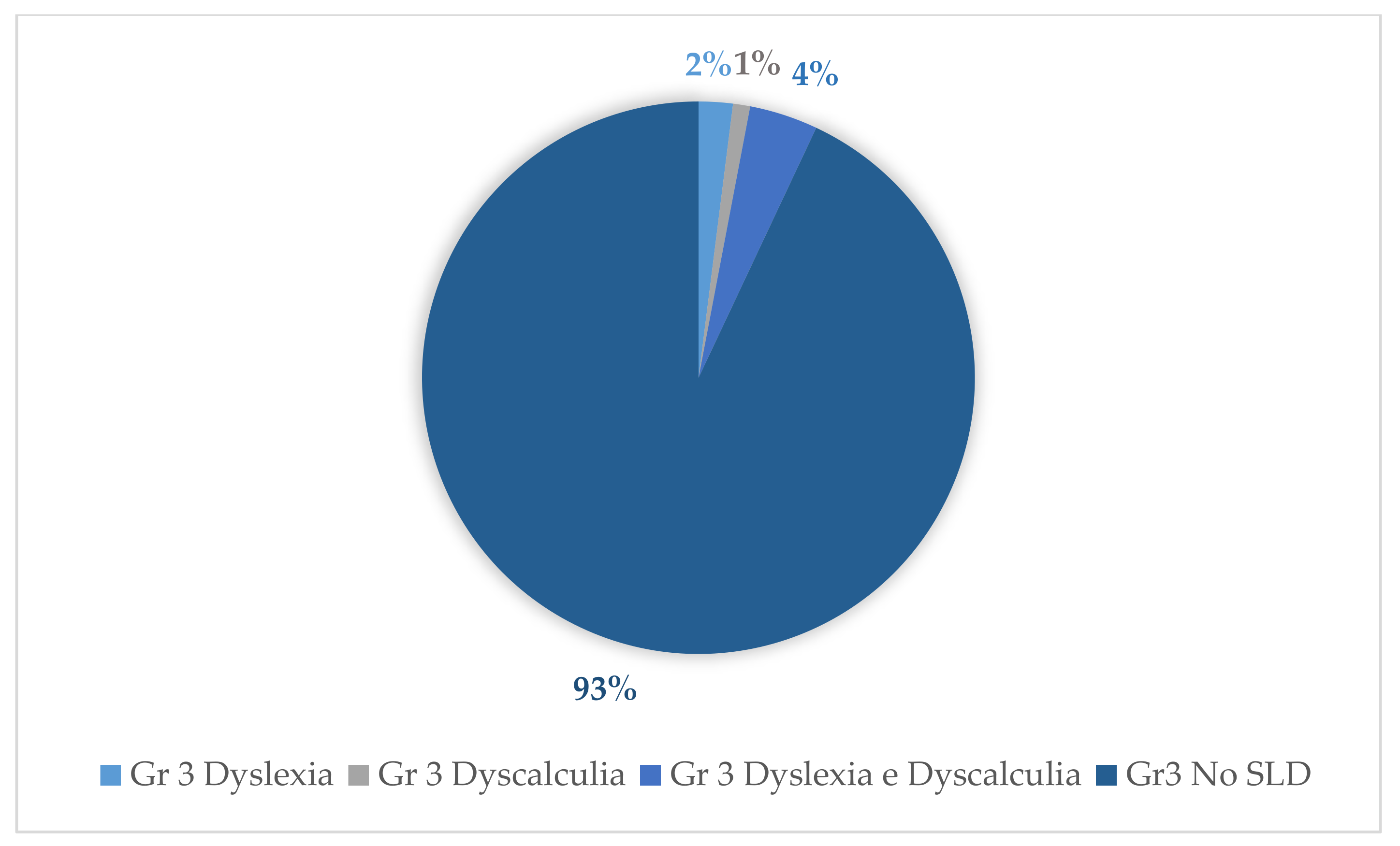
| Group 1 | Group 2 | Group 3 | ||||||
|---|---|---|---|---|---|---|---|---|
| Mage | SD | Gender | Mage | SD | Gender | Mage | SD | Gender |
| 15.2 | 0.25 | M/F 60/40 | 15.5 | 0.14 | M/F 70/30 | 15.3 | 0.12 | M/F 65/35 |
| Indices | Group1 | Group2 | Group3 | F | P | |||
|---|---|---|---|---|---|---|---|---|
| Means | SD | Means | SD | Means | SD | |||
| VCI | 98.76 | 4.57 | 101.70 | 6.30 | 101.78 | 5.33 | 9.968 | <0.05 * |
| PRI | 99.12 | 6.61 | 102.50 | 5.88 | 102.89 | 4.88 | 12.588 | <0.05 * |
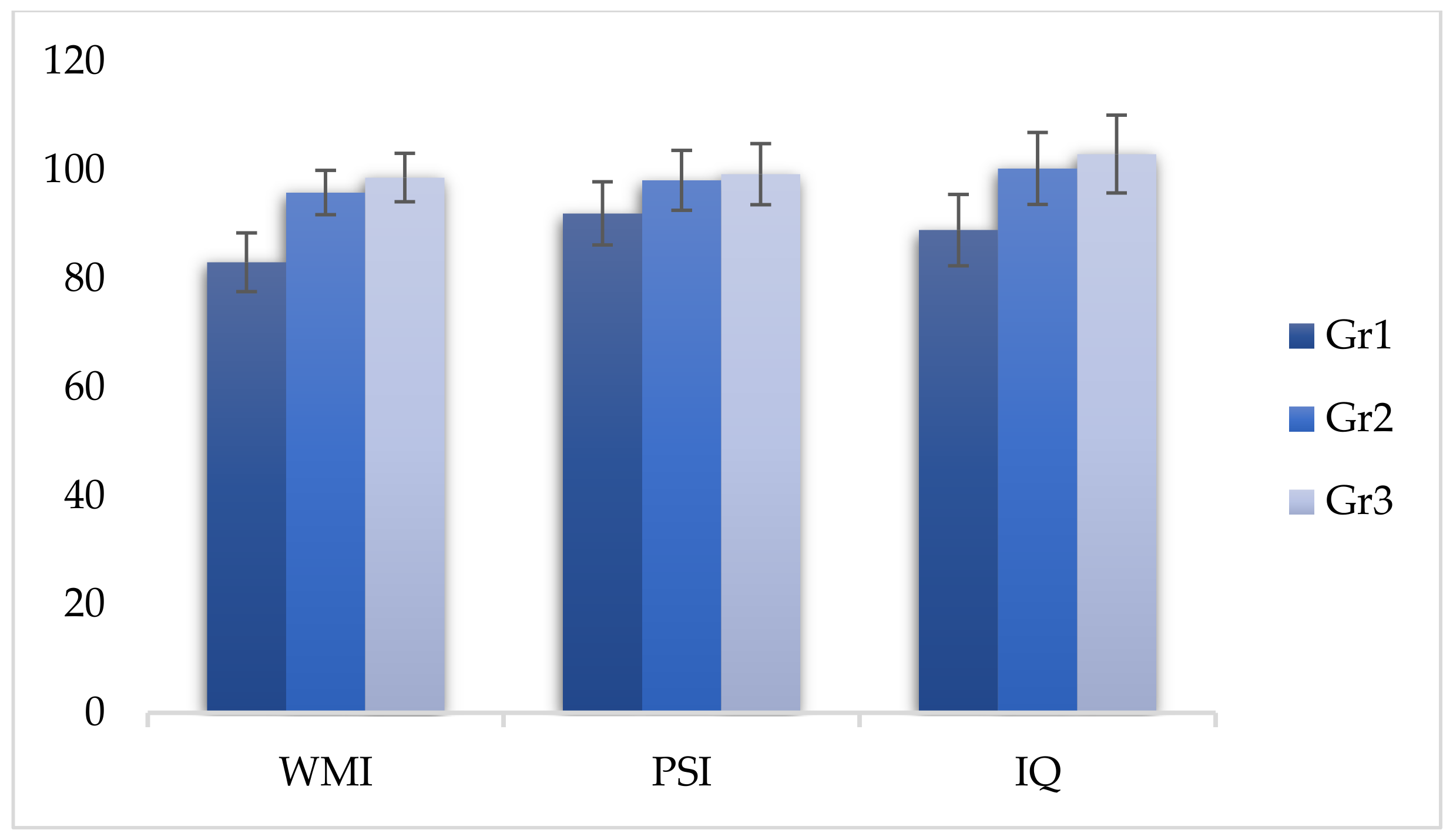
| WMI | 83.02 | 5.41 | 95.83 | 4.08 | 98.59 | 4.46 | 314.113 | <0.05 * |
| PSI | 91.99 | 5.81 | 98.11 | 5.53 | 99.25 | 5.63 | 47.539 | <0.05 * |
| IQ | 88.94 | 6.57 | 100.28 | 6.64 | 102.92 | 7.16 | 119.343 | <0.05 * |
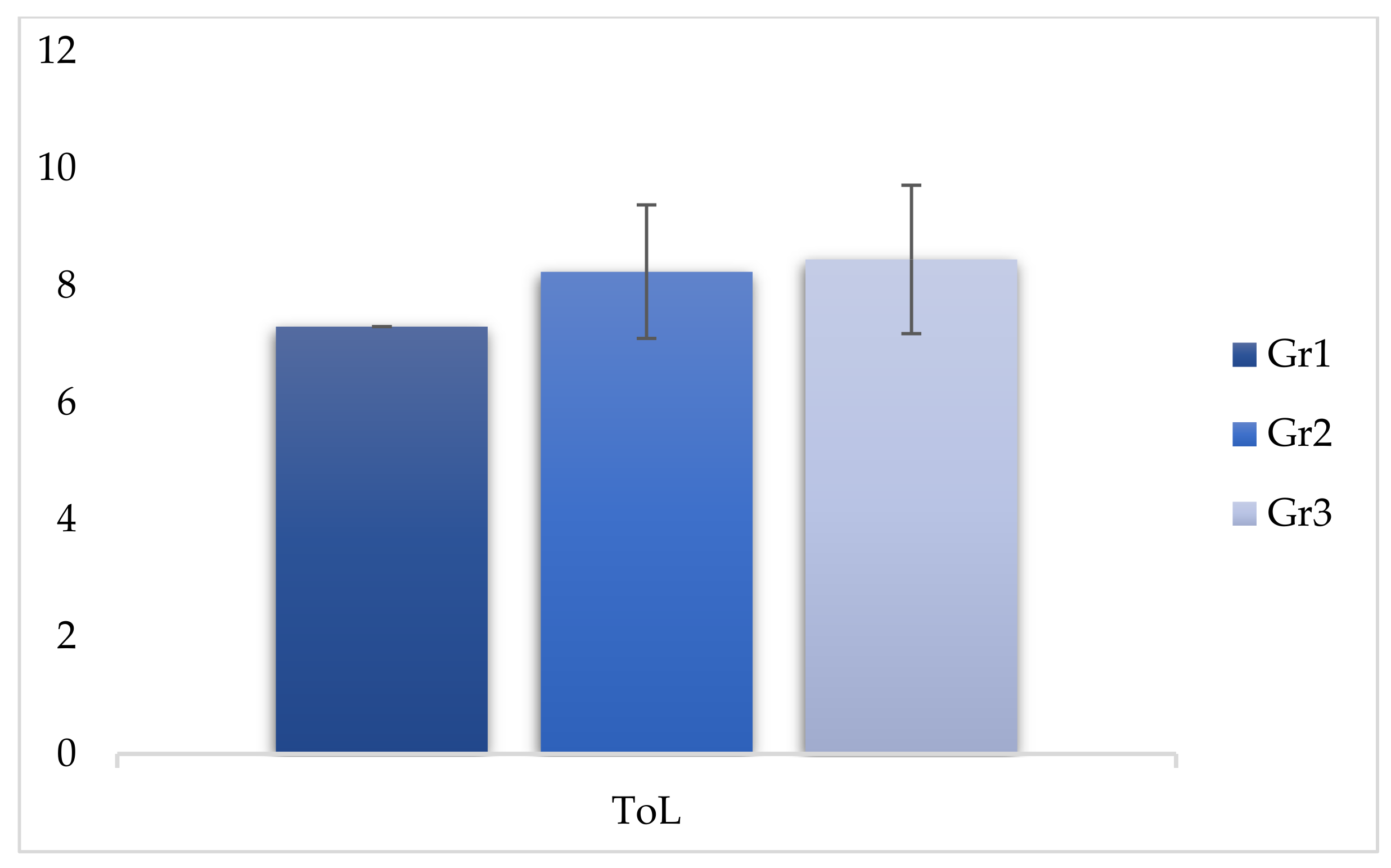
| ToL | 7.30 | 0.969 | 8.24 | 1.14 | 8.45 | 1.27 | 29.007 | <0.05 * |
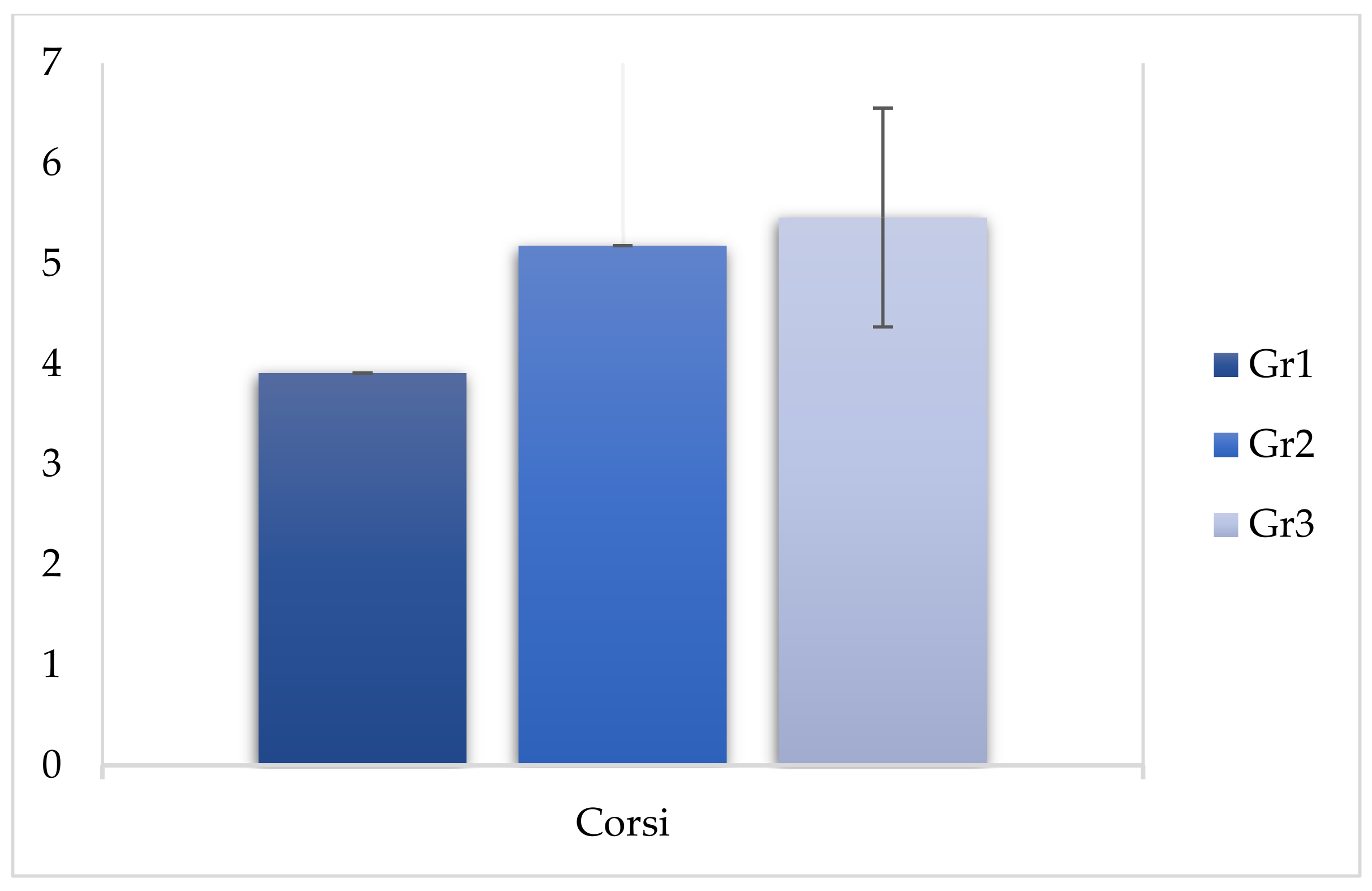
| Corsi | 3.91 | 0.740 | 5.18 | 0.914 | 5.46 | 1.09 | 79.222 | <0.05 * |
| Indices | Group 1 | Group 3 | Difference in Means | p | ||
|---|---|---|---|---|---|---|
| Means | SD | Means | SD | |||
| VCI | 98.76 | 4.57 | 101.78 | 5.33 | −3.020 | <0.05 * |
| PRI | 99.12 | 6.61 | 102.89 | 4.88 | −3.770 | <0.05 * |
| WMI | 83.02 | 5.41 | 98.59 | 4.46 | −15.570 | <0.05 * |
| PSI | 91.99 | 5.81 | 99.25 | 5.63 | −7.260 | <0.05 * |
| IQ | 88.94 | 6.57 | 102.92 | 7.16 | −13.980 | <0.05 * |
| ToL | 7.30 | 0.969 | 8.45 | 1.27 | −1.150 | <0.05 * |
| CORSI | 3.91 | 0.740 | 5.46 | 1.09 | −1.550 | <0.05 * |
| Indices | Group 1 | Group 2 | Difference in Means | P | ||
|---|---|---|---|---|---|---|
| Means | SD | Means | SD | |||
| VCI | 98.76 | 4.57 | 101.70 | 6.30 | −2.940 | <0.05 * |
| PRI | 99.12 | 6.61 | 102.50 | 5.88 | −3.380 | <0.05 * |
| WMI | 83.02 | 5.41 | 95.83 | 4.08 | −12.810 | <0.05 * |
| PSI | 91.99 | 5.81 | 98.11 | 5.53 | −6.120 | <0.05 * |
| IQ | 88.94 | 6.57 | 100.28 | 6.64 | −11.340 | <0.05 * |
| ToL | 7.30 | 0.969 | 8.24 | 1.14 | −0.940 | <0.05 * |
| CORSI | 3.91 | 0.740 | 5.18 | 0.914 | −1.270 | <0.05 * |
| Indices | Group 2 | Group 3 | Difference in Means | p | ||
|---|---|---|---|---|---|---|
| Means | SD | Means | SD | |||
| VCI | 101.70 | 6.30 | 101.78 | 5.33 | −0.80 | 1.000 |
| PRI | 102.50 | 5.89 | 102.89 | 4.88 | −0.390 | 1.000 |
| WMI | 95.83 | 4.08 | 98.59 | 4.46 | −2.760 | <0.05 * |
| PSI | 98.11 | 5.53 | 99.25 | 5.63 | −1.140 | 0.467 |
| IQ | 100.28 | 6.64 | 102.92 | 7.16 | −2.640 | 0.019 |
| ToL | 8.24 | 1.14 | 8.45 | 1.27 | −0.210 | 0.578 |
| CORSI | 5.18 | 0.914 | 5.46 | 1.09 | −0.280 | 0.101 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frolli, A.; Ricci, M.C.; Cavallaro, A.; Lombardi, A.; Bosco, A.; Di Carmine, F.; Operto, F.F.; Franzese, L. Cognitive Development and Cannabis Use in Adolescents. Behav. Sci. 2021, 11, 37. https://doi.org/10.3390/bs11030037
Frolli A, Ricci MC, Cavallaro A, Lombardi A, Bosco A, Di Carmine F, Operto FF, Franzese L. Cognitive Development and Cannabis Use in Adolescents. Behavioral Sciences. 2021; 11(3):37. https://doi.org/10.3390/bs11030037
Chicago/Turabian StyleFrolli, Alessandro, Maria Carla Ricci, Antonella Cavallaro, Agnese Lombardi, Antonia Bosco, Francesca Di Carmine, Francesca Felicia Operto, and Luisa Franzese. 2021. "Cognitive Development and Cannabis Use in Adolescents" Behavioral Sciences 11, no. 3: 37. https://doi.org/10.3390/bs11030037
APA StyleFrolli, A., Ricci, M. C., Cavallaro, A., Lombardi, A., Bosco, A., Di Carmine, F., Operto, F. F., & Franzese, L. (2021). Cognitive Development and Cannabis Use in Adolescents. Behavioral Sciences, 11(3), 37. https://doi.org/10.3390/bs11030037







