Biogeochemical Response of the Water Column of Concepción Bay, Chile, to a New Regime of Atmospheric and Oceanographic Variability
Abstract
1. Introduction
2. Materials and Methods
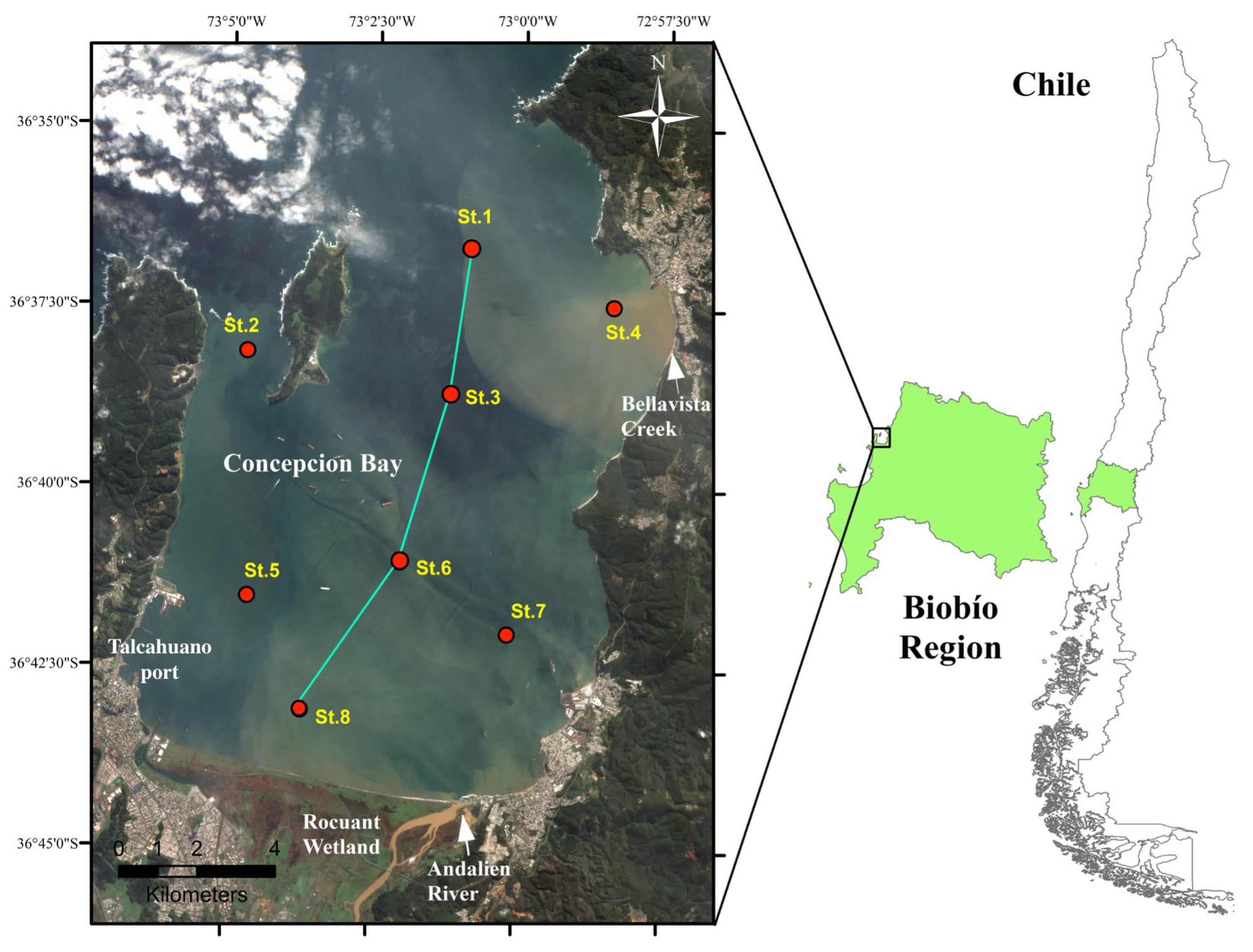
2.1. Study Site and Sample Collection
2.2. Atmospheric Analyses
2.3. Hydrographic Analyses
2.4. Statistical Analyses
3. Results
3.1. Meteorological and Hydrological Physical Variability
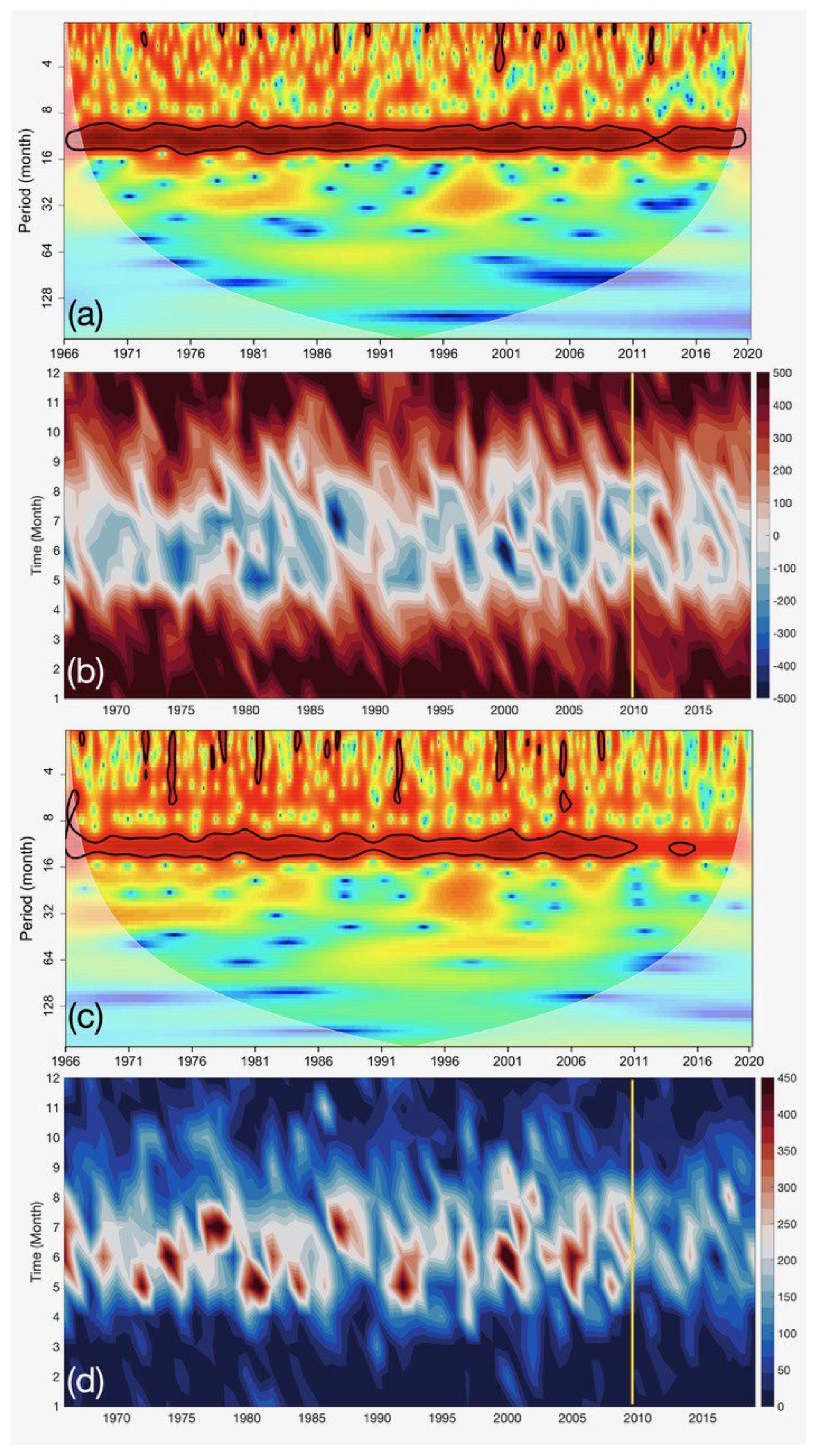
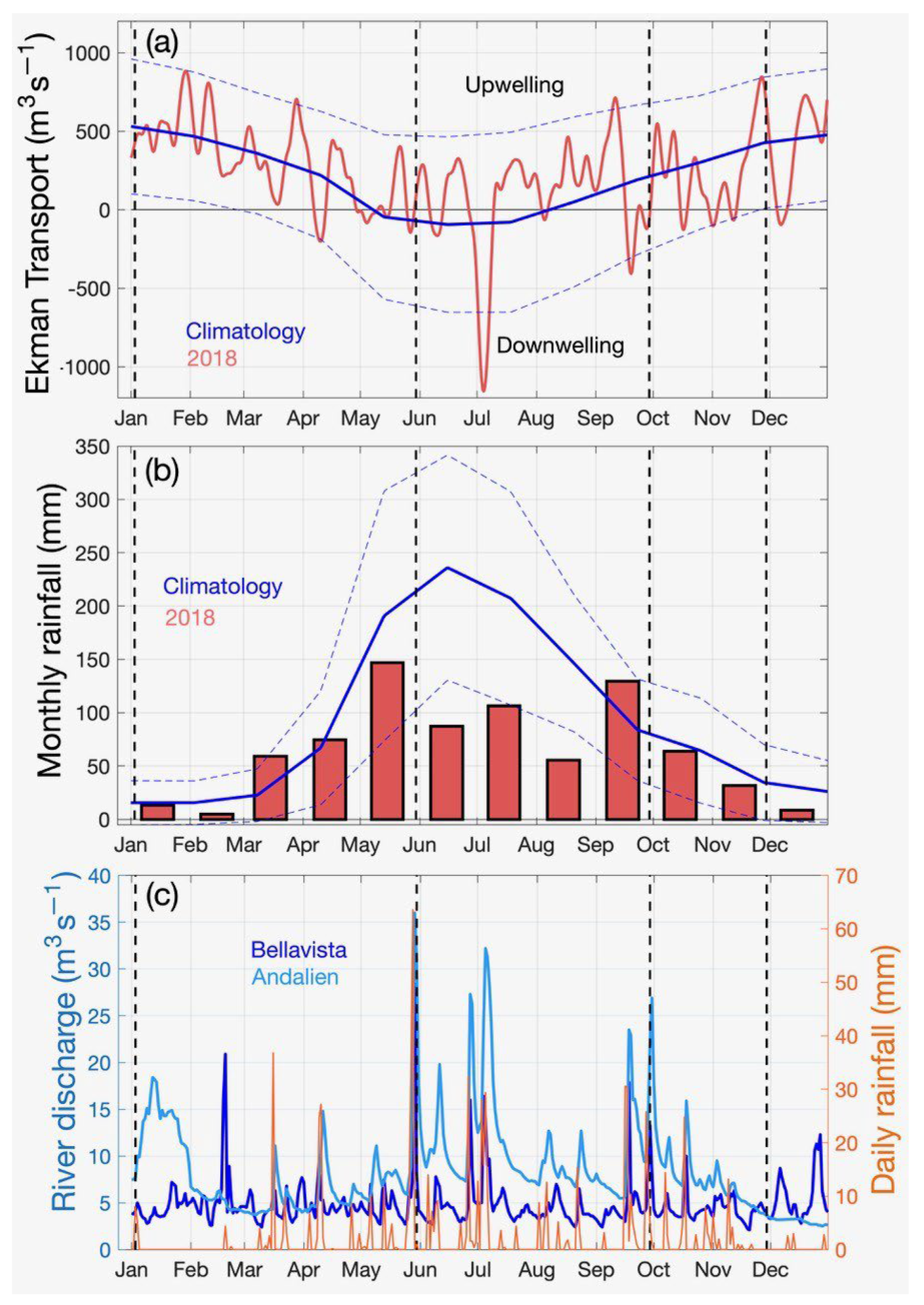
3.1.1. Ekman Transport
3.1.2. Precipitation and River Streamflow
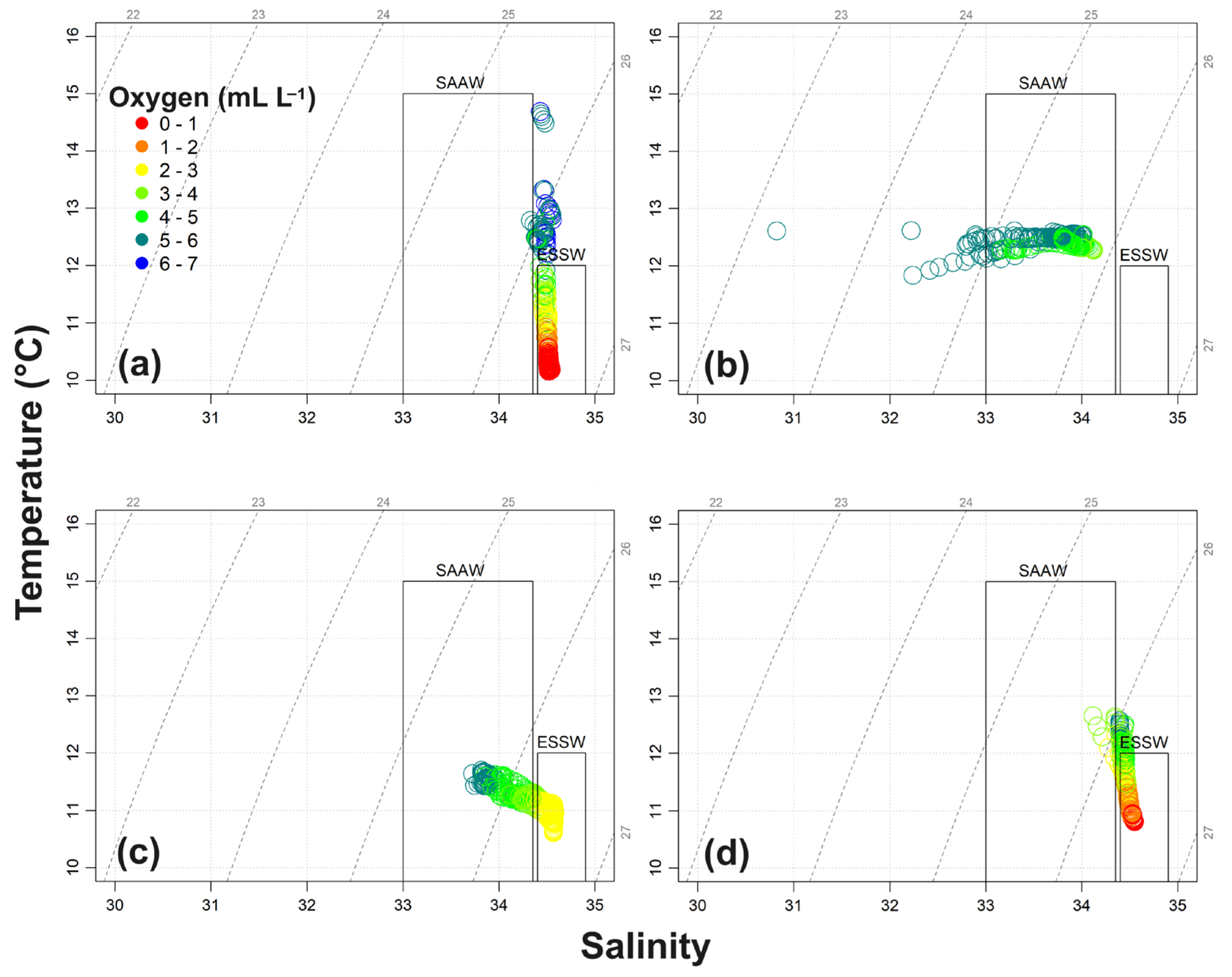
3.1.3. Hydrographic Variability and T/S Diagram
3.2. Biogeochemical Variability
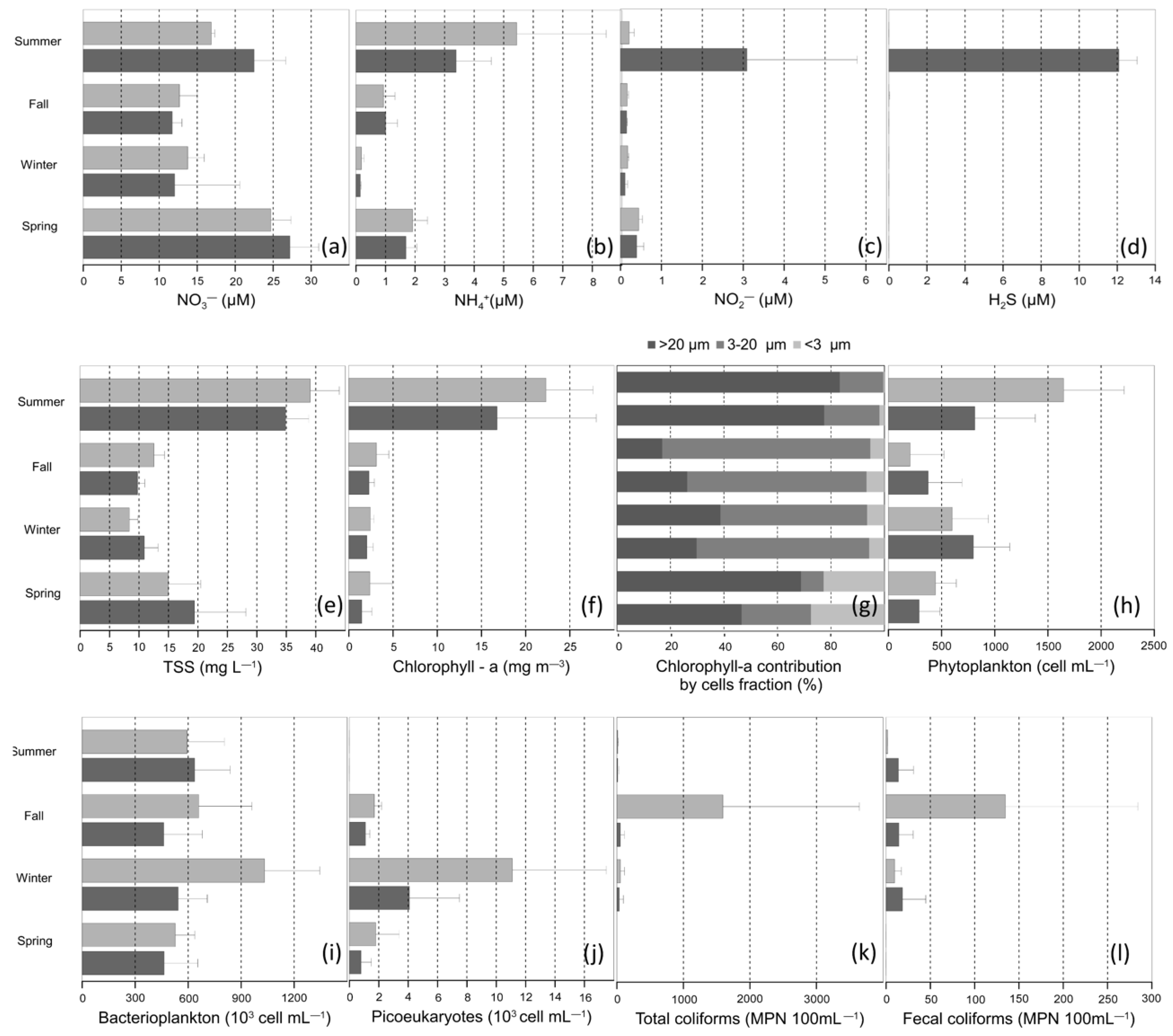
3.2.1. Nutrients and Hydrogen Sulfide (H2S)
3.2.2. Total Suspended Solids and Chlorophyll-a
3.2.3. Microbial Community Abundances
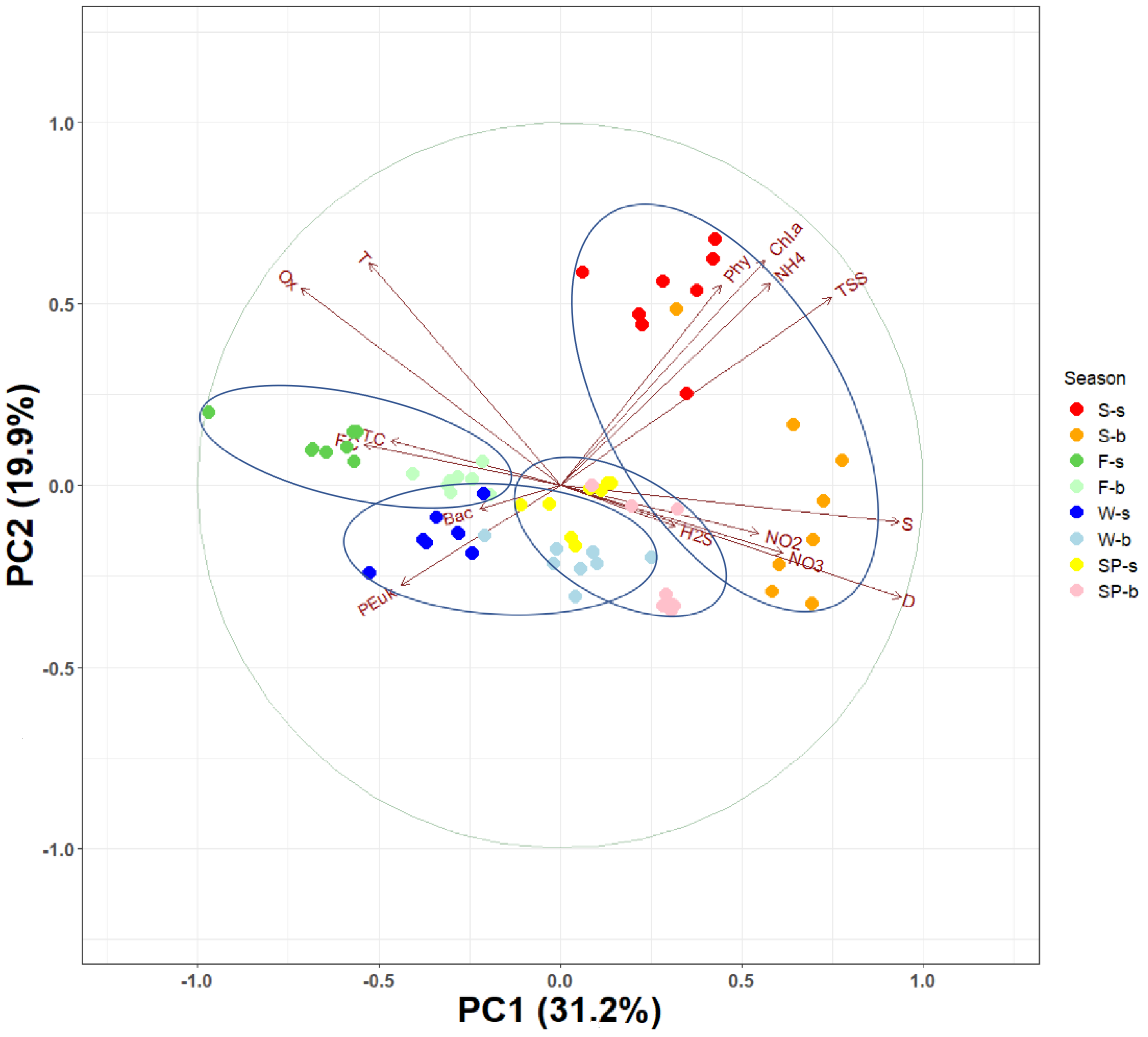
3.3. Principal Component Analysis
4. Discussion
4.1. Meteorological and Hydrological Temporal Variability
4.2. Seasonal and Vertical Biogeochemical Variability
4.3. Seasonal and Vertical Variability of Biological Communities
5. Final Considerations
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahumada, R. Producción y destino de la biomasa fitoplanctónica en un sistema de bahías en Chile central: Una hipótesis. Biol. Pesq. 1989, 18, 53–66. [Google Scholar]
- Djurfeldt, L. Circulation and mixing in a coastal upwelling embayment; Gulf of Arauco, Chile. Cont. Shelf Res. 1989, 9, 1003–1016. [Google Scholar] [CrossRef]
- Cubillos, L.; Arancibia, H. Sobre el crecimiento estacional de la sardina común (Strangomera bentincki) y anchoa (Anillo de Engraulis) off Talcahuano, Chile. Rev. Biol. Mar. 1993, 28, 43–49. [Google Scholar]
- Arcos, D. Variabilidad de Pequena Escala En La Zona Neritica Del Sistema de Surgencia de Talcahuano (Chile Central): Identificacion y Dinamica de Aeas de Retencion Larval. Gayana. Ocean. 1996, 4, 21–58. [Google Scholar]
- Castro, L.; Quiñones, R.; Arancibia, H.; Figueroa, D.; Roa, R.; Sobarzo, M.; Retamal, M. Áreas de Desove de Anchoveta y Sardina Común en la Zona Central; Informe Final Proyecto Fondo de Investigación Pesquera No. 96-11; Universidad de Concepción: Concepción, Chile, 1997; 115p. [Google Scholar]
- Ahumada, R.; Rudolph, A.; Martinez, V. Circulation and fertility of waters in Concepcion Bay. Estuar. Coast. Shelf Sci. 1983, 16, 95–105. [Google Scholar]
- Pantoja, S.; González, H.; Bernal, P.A. Size-fractioned autotrophic production in a shallow bay. Biol. Pesq. 1987, 16, 99–105. [Google Scholar]
- Ahumada, R.; Matra, P.; Silva, N. Phytoplankton biomass distribution and relationship to nutrient enrichment during an upwelling event off Concepcion Bay Chile. Boletín Soc. Biol. Concepción 1991, 62, 7–19. [Google Scholar]
- Brandhorst, W. Condiciones oceanográficas estivales frente a la Costa de Chile. Rev. Biol. Mar. 1971, 14, 45–84. [Google Scholar]
- Arcos, D.F.; Wilson, R.E. Upwelling and the distribution of chlorophyll a within the Bay of Concepción, Chile. Estuar. Coast. Shelf Sci. 1984, 18, 25–35. [Google Scholar] [CrossRef]
- Gómez, V.; Pozo, K.; Nuñez, D.; Přibylová, P.; Audy, O.; Baini, M.; Fossi, M.C.; Klánová, J. Marine plastic debris in Central Chile: Characterization and abundance of macroplastics and burden of persistent organic pollutants (POPs). Mar. Pollut. Bull. 2020, 152, 110881. [Google Scholar] [CrossRef]
- Pozo, K.; Gómez, V.; Přibylová, P.; Lammel, G.; Klánová, J.; Rudolph, A.; Ahumada, R. Multicompartmental analysis of POPs and PAHs in Concepciόn Bay, central Chile: Part I—Levels and patterns after the 2010 tsunami. Mar. Pollut. Bull. 2022, 174, 113144. [Google Scholar] [CrossRef] [PubMed]
- Pozo, K.; Gómez, V.; Tucca, F.; Galbán-Malagón, C.; Ahumada, R.; Rudolph, A.; Klánová, J.; Lammel, G. Multicompartmental analysis of POPs and PAHs in Concepciόn Bay, central Chile: Part II—Air-sea exchange during Austral summer. Mar. Pollut. Bull. 2022, 177, 113518. [Google Scholar] [CrossRef] [PubMed]
- Valiela, I. Marine ecological processes. J. Mar. Biol. Assoc. 1995, 76, 551. [Google Scholar] [CrossRef]
- Farias, L. Remineralization and accumulation of organic carbon and nitrogen in marine sediments of eutrophic bays: The case of the Bay of Concepcion, Chile. Estuar. Coast. Shelf Sci. 2003, 57, 829–841. [Google Scholar] [CrossRef]
- Campos, C.J.A.; Avant, J.; Gustar, N.; Lowther, J.; Powell, A.; Stockley, L.; Lees, D.N. Fate of Human Noroviruses in Shellfish and Water Impacted by Frequent Sewage Pollution Events. Environ. Sci. Technol. 2015, 49, 8377–8385. [Google Scholar] [CrossRef] [PubMed]
- González-Saldía, R.R.; Pino-Maureira, N.L.; Muñoz, C.; Soto, L.; Durán, E.; Barra, M.J.; Gutiérrez, S.; Díaz, V.; Saavedra, A. Fecal pollution source tracking and thalassogenic diseases: The temporal-spatial concordance between maximum concentrations of human mitochondrial DNA in seawater and Hepatitis A outbreaks among a coastal population. Sci. Total Environ. 2019, 686, 158–170. [Google Scholar] [CrossRef] [PubMed]
- Sanhueza, G.; Cachicas, V. Detección y cuantificación de virus de hepatitis A en moluscos en las bahías de Concepción y Arauco, Chile. Rev. Inst. Salud Pública 2020, 4, 10–19. [Google Scholar] [CrossRef]
- EPA 506-6-90-003; Contaminated Sediments. Relevant Statutes and EPA Program Activities. U.S Environmental Protection Agency, Sediment Oversight Technical Committee: Washington, DC, USA, 1990; p. 158.
- IPCC. Global Warming of 1.5 °C. An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; Masson-Delmotte, V., Zhai, H.-O.P., Pörtner, D., Roberts, J., Skea, P.R., Shukla, A., Pirani, W., Moufouma-Okia, C., Péan, R., Pidcock, S., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2018; pp. 3–24. [Google Scholar] [CrossRef]
- Seager, R.; Ting, M.; Held, I.; Kushnir, Y.; Lu, J.; Vecchi, G.; Huang, H.-P.; Harnik, N.; Leetmaa, A.; Lau, N.-C.; et al. Model Projections of an Imminent Transition to a More Arid Climate in Southwestern North America. Science 2007, 316, 1181–1184. [Google Scholar] [CrossRef] [PubMed]
- Ummenhofer, C.C.; Meehl, G.A. Extreme weather and climate events with ecological relevance: A review. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20160135. [Google Scholar] [CrossRef]
- Andrys, J.; Kala, J.; Lyons, T.J. Regional climate projections of mean and extreme climate for the southwest of Western Australia (1970–1999 compared to 2030–2059). Clim. Dyn. 2017, 48, 1723–1747. [Google Scholar] [CrossRef]
- Szwed, M. Variability of precipitation in Poland under climate change. Theor. Appl. Climatol. 2019, 135, 1003–1015. [Google Scholar] [CrossRef]
- Berg, P.; Moseley, C.; Haerter, J.O. Strong increase in convective precipitation in response to higher temperatures. Nat. Geosci. 2013, 6, 181–185. [Google Scholar] [CrossRef]
- Boucher, O.; Randall, D.; Artaxo, P.; Bretherton, C.; Feingold, G.; Forster, P.; Stocker, T.F.; Qin, D.; Plattner, G.-K.; Zhang, X.Y. Clouds and aerosols. In Climate Change 2013: The Physical Science Basis; Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2013; pp. 571–657. [Google Scholar]
- Kharin, V.V.; Zwiers, F.W.; Zhang, X.; Wehner, M. Changes in temperature and precipitation extremes in the CMIP5 ensemble. Clim. Chang. 2013, 119, 345–357. [Google Scholar] [CrossRef]
- Sillmann, J.; Kharin, V.V.; Zwiers, F.W.; Zhang, X.; Bronaugh, D. Climate extremes indices in the CMIP5 multimodel ensemble: Part 2. Future climate projections. J. Geophys. Res. Atmos. 2013, 118, 2473–2493. [Google Scholar] [CrossRef]
- Fischer, E.M.; Knutti, R. Observed heavy precipitation increase confirms theory and early models. Nat. Clim. Chang. 2016, 6, 986–991. [Google Scholar] [CrossRef]
- Du, H.; Alexander, L.V.; Donat, M.G.; Lippmann, T.; Srivastava, A.; Salinger, J.; Kruger, A.; Choi, G.; He, H.S.; Fujibe, F.; et al. Precipitation from Persistent Extremes is Increasing in Most Regions and Globally. Geophys. Res. Lett. 2019, 46, 6041–6049. [Google Scholar] [CrossRef]
- Myhre, G.; Alterskjær, K.; Stjern, C.W.; Hodnebrog, Ø.; Marelle, L.; Samset, B.H.; Sillmann, J.; Schaller, N.; Fischer, E.; Schulz, M.; et al. Frequency of extreme precipitation increases extensively with event rareness under global warming. Sci. Rep. 2019, 9, 16063. [Google Scholar] [CrossRef] [PubMed]
- Stramma, L.; Johnson, G.C.; Sprintall, J.; Mohrholz, V. Expanding Oxygen-Minimum Zones in the Tropical Oceans. Science 2008, 320, 655–658. [Google Scholar] [CrossRef]
- Li, G.; Cheng, L.; Zhu, J.; Trenberth, K.E.; Mann, M.E.; Abraham, J.P. Increasing ocean stratification over the past half-century. Nat. Clim. Chang. 2020, 10, 1116–1123. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Spreading Dead Zones and Consequences for Marine Ecosystems. Science 2008, 321, 926–929. [Google Scholar] [CrossRef]
- Bakun, A. Global Climate Change and Intensification of Coastal Ocean Upwelling. Science 1990, 247, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Sydeman, W.J.; García-Reyes, M.; Schoeman, D.S.; Rykaczewski, R.R.; Thompson, S.A.; Black, B.A.; Bograd, S.J. Climate change and wind intensification in coastal upwelling ecosystems. Science 2014, 345, 77–80. [Google Scholar] [CrossRef]
- Sobarzo, M.B.; Figueroa, D.; Arcos, D.R. The Influence of Winds and Tides in the Formation of Circulation Layers in a Bay, a Case Study: Concepción Bay, Chile. Estuar. Coast. Shelf Sci. 1997, 45, 729–736. [Google Scholar] [CrossRef]
- Wong, Z.; Saldías, G.S.; Largier, J.L.; Strub, P.T.; Sobarzo, M. Surface thermal structure and variability of upwelling shadows in the Gulf of Arauco, Chile. J. Geophys. Res. Ocean. 2021, 126, e2020JC016194. [Google Scholar] [CrossRef]
- Large, W.G.; Pond, S. Open Ocean Momentum Flux Measurements in Moderate to Strong Winds. J. Phys. Oceanogr. 1981, 11, 324–336. [Google Scholar] [CrossRef]
- Smith, R.L. Upwelling. Oceanogr. Mar. Biol. Annu. Rev. 1968, 6, 11–46. [Google Scholar]
- Torrence, C.; Compo, G.P. A practical guide to wavelet analysis. Bull. Am. Meteorol. Soc. 1998, 79, 61–78. [Google Scholar] [CrossRef]
- Cazelles, B.; Chavez, M.; Berteaux, D.; Ménard, F.; Vik, J.O.; Jenouvrier, S.; Stenseth, N.C. Wavelet analysis of ecological time series. Oecologia 2008, 156, 287–304. [Google Scholar] [CrossRef]
- Garreaud, R.D.; Alvarez-Garreton, C.; Barichivich, J.; Boisier, J.P.; Christie, D.; Galleguillos, M.; LeQuesne, C.; McPhee, J.; Zambrano-Bigiarini, M. The 2010–2015 megadrought in central Chile: Impacts on regional hydroclimate and vegetation. Hydrol. Earth Syst. Sci. 2017, 21, 6307–6327. [Google Scholar] [CrossRef]
- Garreaud, R.D.; Boisier, J.P.; Rondanelli, R.; Montecinos, A.; Sepúlveda, H.H.; Veloso-Aguila, D. The Central Chile Mega Drought (2010–2018): A climate dynamics perspective. Int. J. Climatol. 2020, 40, 421–439. [Google Scholar] [CrossRef]
- Saldías, G.S.; Largier, J.L.; Mendes, R.; Pérez-Santos, I.; Vargas, C.A.; Sobarzo, M. Satellite-measured interanual variability of turbid river plumes off central-southern Chile: Spatial patterns and the influence of climate variability. Prog. Oceanogr. 2016, 146, 212–222. [Google Scholar] [CrossRef]
- Schneider, W.; Donoso, D.; Garcés-Vargas, J.; Escribano, R. Water-column cooling and sea surface salinity increase in the upwelling region off central-south Chile driven by a poleward displacement of the South Pacific High. Prog. Oceanogr. 2017, 151, 38–48. [Google Scholar] [CrossRef]
- Donat, M.G.; Peterson, T.C.; Brunet, M.; King, A.D.; Almazroui, M.; Kolli, R.K.; Boucherf, D.; Al-Mulla, A.Y.; Nour, A.Y.; Aly, A.A.; et al. Changes in extreme temperature and precipitation in the Arab region: Long-term trends and variability related to ENSO and NAO. Int. J. Climatol. 2014, 34, 581–592. [Google Scholar] [CrossRef]
- Rao, K.K.; Patwardhan, S.K.; Kulkarni, A.; Kamala, K.; Sabade, S.S.; Kumar, K.K. Projected changes in mean and extreme precipitation indices over India using PRECIS. Glob. Planet. Chang. 2014, 113, 77–90. [Google Scholar] [CrossRef]
- Ren, Z.; Zhang, M.; Wang, S.; Qiang, F.; Zhu, X.; Dong, L. Changes in daily extreme precipitation events in South China from 1961 to 2011. J. Geogr. Sci. 2015, 25, 58–68. [Google Scholar] [CrossRef]
- de los Skansi, M.M.; Brunet, M.; Sigró, J.; Aguilar, E.; Arevalo-Groening, J.A.; Bentancur, O.J.; Geier, Y.R.C.; Amaya, R.L.C.; Jácome, H.; Ramos, A.M.; et al. Warming and wetting signals emerging from analysis of changes in climate extreme indices over South America. Glob. Planet. Chang. 2013, 100, 295–307. [Google Scholar] [CrossRef]
- Carritt, D.E.; Carpenter, J.H. Comparison and Evaluation of Currently Employed Modifications of the Winkler Method for Determining Dissolved Oxygen in Seawater; a NASCO Report. J. Mar. Res. 1966, 24, 286–318. [Google Scholar]
- Parsons, T.; Maita, C.; Lally, C. A Manual of Chemical and Biological Methods of Seawater Analysis; Pergamon Press: Oxford, UK, 1984; 173p. [Google Scholar]
- APHA-AWWA-WPCF Standard Methods (9010) for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association/American Water Works Association/Water Environment Federation: Washington, DC, USA, 2017.
- Cline, J.D. Spectrophotometric determination of hydrogen sulfide in natural waters. Limnol. Oceanogr. 1969, 14, 454–458. [Google Scholar] [CrossRef]
- Marie, D.; Partensky, F.; Simon, N.; Guillou, L.; Vaulot, D. Protocols in flow cytometry and cell sorting. In Living Colors; Diamond, R.A., DeMaggio, S., Eds.; Springer: New York, NY, USA, 2000; pp. 421–454. [Google Scholar]
- Utermöhl, H. Zur vervollkommnung der quantitativen phytoplankton-methodik: Mit 1 Tabelle und 15 abbildungen im Text und auf 1 Tafel. Int. Ver. Theor. Angew. Limnol. Mitteilungen 1958, 9, 1–38. [Google Scholar] [CrossRef]
- Mishra, A. Assessment of water quality using principal component analysis: A case study of the river Ganges. J. Water Chem. Technol. 2010, 32, 227–234. [Google Scholar] [CrossRef]
- Anderson, M.J. Permutational Multivariate Analysis of Variance (PERMANOVA); Balakrishnan, N., Colton, T., Everitt, B., Piegorsch, W., Ruggeri, F., Teugels, J.L., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 1–15. [Google Scholar] [CrossRef]
- Oksanen, J.; Guillaume, F.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Szoecs, E.; Solymos, P.; Stevens, M.H.H.; Wagner, H.H.; et al. Vegan: Community Ecology Package: Ordination, Diversity and Dissimilarities. R Package Version 2.3-0. 2015. Available online: https://www.researchgate.net/publication/311493644_Vegan_commity_ecology_package_R_package_version_23-0 (accessed on 18 April 2019).
- R Core Team. R: A Language and Environment for Statistical Computing [Computer Software]. R Foundation for Statistical Computing. 2019. Available online: https://www.scirp.org/reference/ReferencesPapers?ReferenceID=2631126 (accessed on 21 January 2019).
- Strub, P.T.; Mesias, J.M.; Montecino, V.; Rutllant, J.; Salinas, S. Coastal ocean circulation off western South America, In The Sea; Robinson, A.R. , Brink, K.H., Eds.; John Wiley: New York, NY, USA, 1998; Volume 11, pp. 273–313. [Google Scholar]
- Sobarzo, M.; Bravo, L.; Donoso, D.; Garcés-Vargas, J.; Schneider, W. Coastal upwelling and seasonal cycles that influence the water column over the continental shelf off central Chile. Prog. Oceanogr. 2007, 75, 363–382. [Google Scholar] [CrossRef]
- Morales, C.E.; Hormazabal, S.; Andrade, I.; Correa-Ramirez, M.A. Time-Space Variability of Chlorophyll-a and Associated Physical Variables within the Region off Central-Southern Chile. Remote Sens. 2013, 5, 5550–5571. [Google Scholar] [CrossRef]
- Daneri, G.; Dellarossa, V.; Quiñones, R.; Jacob, B.; Montero, P.; Ulloa, O. Primary production and community respiration in the Humboldt Current System off Chile and associated oceanic areas. Mar. Ecol. Prog. Ser. 2000, 197, 41–49. [Google Scholar] [CrossRef]
- Farías, L.; Fernández, C.; Faúndez, J.; Cornejo, M.; Alcaman, M.E. Chemolithoautotrophic production mediating the cycling of the greenhouse gases N2O and CH4 in an upwelling ecosystem. Biogeosciences 2009, 6, 3053–3069. [Google Scholar] [CrossRef]
- Montecino, V.; Paredes, M.A.; Paolini, P.; Rutllant, J. Revisiting chlorophyll data along the coast in north-central Chile, considering multiscale environmental variability. Rev. Chil. Hist. Nat. 2006, 79, 213–223. [Google Scholar] [CrossRef][Green Version]
- Ahumada, R.; Chuecas, L. Algunas características hidrogramas de la Bahía de Concepción (36′40′S; 73”02′W) y áreas adyacentes Chile. Gayana Myscelánea 1979, 8, 1–56. [Google Scholar]
- Sobarzo, M. Caracterización de la Circulación de Bahía Concepción, Chile: Un análisis temporal en el dominio de la frecuencia. Ph.D. Thesis, Tesis de Grado de Magister en Ciencias mención Oceanografía, Universidad de Concepción, Departamento de Oceanografía, Concepción, Chile, 1993. [Google Scholar]
- Sobarzo, M. Oceanografía Física entre Punta Nugurne (35°57′S; 72°47′W) y Punta Manuel (38°30′S; 73°31′W), Chile: Una revisión histórica (1936–1990). Gayana Oceanol. 1994, 2, 5–17. [Google Scholar]
- Arcos, D.; Núñez, S.; Urrutia, A.; Chuecas, L. Shelf-embayment water exchange and residence times within Concepcion Bay, Chile. Gayana Oceanol. 1995, 3, 75–87. [Google Scholar]
- Montencino, V.; Lange, C. The Humboldt Current System: Ecosystem components and processes, fisheries, and sediment studies. Prog. Oceanogr. 2009, 83, 65–79. [Google Scholar] [CrossRef]
- Belmadani, A.; Echevin, V.; Codron, F.; Takahashi, K.; Junquas, C. What dynamics drive future wind scenarios for coastal upwelling off Peru and Chile? Clim. Dyn. 2014, 43, 1893–1914. [Google Scholar] [CrossRef]
- Wang, D.; Gouhier, T.C.; Menge, B.A.; Ganguly, A.R. Intensification and spatial homogenization of coastal upwelling under climate change. Nature 2015, 518, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Anabalón, V.; Morales, C.E.; González, H.E.; Menschel, E.; Schneider, W.; Hormazabal, S.; Valencia, L.; Escribano, R. Micro-phytoplankton community structure in the coastal upwelling zone off Concepción (central Chile): Annual and inter-annual fluctuations in a highly dynamic environment. Prog. Oceanogr. 2016, 149, 174–188. [Google Scholar] [CrossRef]
- Medellín-Mora, J.; Escribano, R.; Schneider, W. Community response of zooplankton to oceanographic changes (2002–2012) in the central/southern upwelling system of Chile. Prog. Oceanogr. 2016, 142, 17–29. [Google Scholar] [CrossRef]
- Castro, L.R.; Claramunt, G.; González, H.E.; Krautz, M.C.; Llanos-Rivera, A.; Méndez, J.; Schneider, W.; Soto, S. Fatty acids in eggs of anchoveta Engraulis ringens during two contrasting winter spawning seasons. Mar. Ecol. Prog. Ser. 2010, 420, 193–205. [Google Scholar] [CrossRef]
- Castro, L.R.; Salinas, G.R.; Hernández, E.H. Environmental influences on winter spawning of the anchoveta Engraulis ringens off central Chile. Mar. Ecol. Prog. Ser. 2000, 197, 247–258. [Google Scholar] [CrossRef]
- Castro, L.R. Environmental conditions and larval survival during the winter spawning season of the southernmost anchoveta stock off Chile. Globec. Newsl. 2001, 7, 15–17. [Google Scholar]
- Gilbert, P.M. Margalef revisited: A new phytoplankton mandala incorporating twelve dimensions, including nutritional physiology. Harmful Algae 2016, 55, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Saldías, G.S.; Sobarzo, M.; Largier, J.; Moffat, C.; Letelier, R. Seasonal variability of turbid river plumes off central Chile based on high-resolution MODIS imagery. Remote Sens. Environ. 2012, 123, 220–233. [Google Scholar] [CrossRef]
- Garreaud, R.; Rutllant, J.; Fuenzalida, H. Coastal Lows along the Subtropical West Coast of South America: Mean Structure and Evolution. Mon. Weather Rev. 2002, 130, 75–88. [Google Scholar] [CrossRef]
- Garreaud, R.D. The Andes climate and weather. Adv. Geosci. 2009, 22, 3–11. [Google Scholar] [CrossRef]
- Montecinos, A.; Aceituno, P. Seasonality of the ENSO-Related Rainfall Variability in Central Chile and Associated Circulation Anomalies. J. Clim. 2003, 16, 281–296. [Google Scholar] [CrossRef]
- Quintana, J.M.; Aceituno, P. Changes in the rainfall regime along the extratropical west coast of South America (Chile): 30–43° S. Atmósfera 2012, 25, 1–22. [Google Scholar]
- Viale, M.; Valenzuela, R.; Garreaud, R.; Ralph, F.M. Impacts of atmospheric rivers on precipitacion in southern South America. J. Hydrometeorol. 2018, 19, 1671–1687. [Google Scholar] [CrossRef]
- Daneri, G.; Lizárraga, L.; Montero, P.; González, H.E.; Tapia, F.J. Wind forcing and short-term variability of phytoplankton and heterotrophic bacterioplankton in the coastal zone of the Concepción upwelling system (Central Chile). Prog. Oceanogr. 2012, 92, 92–96. [Google Scholar] [CrossRef]
- Galán, A.; Zirbel, M.J.; Saldías, G.S.; Chan, F.; Letelier, R. The role of upwelling intermittence in the development of hypoxia and nitrogen loss over the Oregon shelf. J. Mar. Syst. 2020, 207, 103342. [Google Scholar] [CrossRef]
- Galán, A.; Faúndez, J.; Thamdrup, B.; Santibáñez, J.F.; Farías, L. Temporal dynamics of nitrogen loss in the coastal upwelling ecosystem off central Chile: Evidence of autotrophic denitrification through sulfide oxidation. Limnol. Oceanogr. 2014, 59, 1865–1878. [Google Scholar] [CrossRef]
- Devol, A.H. Bacterial oxygen uptake kinetics as related to biological processes in oxygen deficient zones of the oceans. Deep Sea Res. 1978, 25, 137–146. [Google Scholar] [CrossRef]
- Devol, A.H. Solution to a marine mystery. Nature 2003, 422, 575–576. [Google Scholar] [CrossRef]
- Zehr, J.P.; Kudela, R.M. Nitrogen Cycle of the Open Ocean: From Genes to Ecosystems. Annu. Rev. Mar. Sci. 2011, 3, 197–225. [Google Scholar] [CrossRef]
- Lam, P.; Kuypers, M.M.M. Microbial Nitrogen Cycling Processes in Oxygen Minimum Zones. Annu. Rev. Mar. Sci. 2011, 3, 317–345. [Google Scholar] [CrossRef]
- Cornejo, M.; Farías, L.; Gallegos, M. Seasonal cycle of N2O vertical distribution and air–sea fluxes over the continental shelf waters off central Chile (~36°S). Prog. Oceanogr. 2007, 75, 383–395. [Google Scholar] [CrossRef]
- Stein, L.Y.; Yung, Y.L. Production, isotopic composition, and atmospheric fate of biologically produced nitrous oxide. Annu. Rev. Earth Planet. Sci. 2003, 31, 329–356. [Google Scholar] [CrossRef]
- Ulloa, O.; Canfield, D.E.; DeLong, E.F.; Letelier, R.M.; Stewart, F.J. Microbial oceanography of anoxic oxygen minimum zones. Proc. Natl. Acad. Sci. USA 2012, 109, 15996–16003. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, B.B.; Postgate, J.R.; Postgate, J.R.; Kelly, D.P. Ecology of the bacteria of the sulphur cycle with special reference to anoxic—Oxic interface environments. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1982, 298, 543–561. [Google Scholar] [CrossRef] [PubMed]
- Graco, M.; Farías, L.; Molina, V.; Gutiérrez, D.; Nielsen, L.P. Massive developments of microbial mats following phytoplankton blooms in a naturally eutrophic bay: Implications for nitrogen cycling. Limnol. Oceanogr. 2001, 46, 821–832. [Google Scholar] [CrossRef]
- Graco, M.; Gutiérrez, D.; Farías, L. Inter-annual variability of the Pelagic-Benthic coupling in the upwelling system off central Chile. Adv. Geosci. 2006, 6, 127–132. [Google Scholar] [CrossRef][Green Version]
- Brüchert, V.; Currie, B.; Peard, K.R. Hydrogen sulphide and methane emissions on the central Namibian shelf. Prog. Oceanogr. 2009, 83, 169–179. [Google Scholar] [CrossRef]
- Dale, A.W.; Sommer, S.; Lomnitz, U.; Bourbonnais, A.; Wallmann, K. Biological nitrate transport in sediments on the Peruvian margin mitigates benthic sulfide emissions and drives pelagic N loss during stagnation events. Deep Sea Res. Part I Oceanogr. Res. Pap. 2016, 112, 123–136. [Google Scholar] [CrossRef]
- Lavik, G.; Stührmann, T.; Brüchert, V.; Van der Plas, A.; Mohrholz, V.; Lam, P.; Mußmann, M.; Fuchs, B.M.; Amann, R.; Lass, U.; et al. Detoxification of sulphidic African shelf waters by blooming chemolithotrophs. Nature 2009, 457, 581–584. [Google Scholar] [CrossRef]
- Schunck, H.; Lavik, G.; Desai, D.K.; Großkopf, T.; Kalvelage, T.; Löscher, C.R.; Paulmier, A.; Contreras, S.; Siegel, H.; Holtappels, M.; et al. Giant Hydrogen Sulfide Plume in the Oxygen Minimum Zone off Peru Supports Chemolithoautotrophy. PLoS ONE 2013, 8, e68661. [Google Scholar] [CrossRef]
- Weeks, S.J.; Currie, B.; Bakun, A. Massive emissions of toxic gas in the Atlantic. Nature 2002, 415, 493–494. [Google Scholar] [CrossRef] [PubMed]
- Ohde, T.; Siegel, H.; Reißmann, J.; Gerth, M. Identification and investigation of sulphur plumes along the Namibian coast using the MERIS sensor. Cont. Shelf Res. 2007, 27, 744–756. [Google Scholar] [CrossRef]
- Copenhagen, W.J. The periodic mortality of fish in the Walvis region. S. Afr. J. Sci. 1953, 49, 330. [Google Scholar]
- Hart, T.J.; Currie, R.I. The Benguela Current. In Discovery Report 31; Cambridge University Press: Cambridge, UK, 1960; pp. 123–298. [Google Scholar]
- Reiffenstein, R.J.; Hulbert, W.C.; Roth, S.H. Toxicology of hydrogen sulfide. Annu. Rev. Pharmacol. Toxicol. 1992, 32, 109–134. [Google Scholar] [CrossRef]
- Hamukuaya, H.; O’Toole, M.J.; Woodhead, P.M.J. Observations of severe hypoxia and offshore displacement of Cape hake over the Namibian shelf in 1994. S. Afr. J. Mar. Sci. 1998, 19, 57–59. [Google Scholar] [CrossRef]
- Gallardo, V.A.; Espinoza, C. The evolution of ocean color. Instrum. Methods Mission. Astrobiol. XI 2008, 7097, 128–134. [Google Scholar] [CrossRef]
- Murillo, A.A.; Ramírez-Flandes, S.; DeLong, E.F.; Ulloa, O. Enhanced metabolic versatility of planktonic sulfur-oxidizing γ-proteobacteria in an oxygen-deficient coastal ecosystem. Front. Mar. Sci. 2014, 1, 18. [Google Scholar] [CrossRef]
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P. Primary Production of the Biosphere: Integrating Terrestrial and Oceanic Components. Science 1998, 281, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.B.; Galvão, H.M.; Mendes, P.A.; Álvarez-Salgado, X.A.; Figueiras, F.G.; Joint, I. Short-term variability of heterotrophic bacterioplankton during upwelling off the NW Iberian margin. Prog. Oceanogr. 2001, 51, 339–359. [Google Scholar] [CrossRef]
- Troncoso, V.A.; Daneri, G.; Cuevas, L.A.; Jacob, B.; Montero, P. Bacterial carbon flow in the Humboldt Current System off Chile. Mar. Ecol. Prog. Ser. 2003, 250, 1–12. [Google Scholar] [CrossRef][Green Version]
- Cuevas, L.A.; Daneri, G.; Jacob, B.; Montero, P. Microbial abundance and activity in the seasonal upwelling area off Concepción (~36°S), central Chile: A comparison of upwelling and non-upwelling conditions. Deep Sea Res. Part II Top. Stud. Oceanogr. 2004, 51, 2427–2440. [Google Scholar] [CrossRef]
- Cubillos, L.; Núñez, S.; Arcos, D. Primary production required to sustain the Chilean pelagic fisheries. Investig. Mar. 1998, 26, 83–96. [Google Scholar]
- FAO. FAO Yearbooks. Fishery and Aquaculture Statistics. Fisheries and Aquaculture Information and Statistics Service. Fishery and Aquaculture Economics and Policy Division. 2070–6057. 2006. Available online: https://www.fao.org/fishery/fr/publications/51136 (accessed on 9 October 2022).
- Chavez, F.P.; Bertrand, A.; Guevara-Carrasco, R.; Soler, P.; Csirke, J. The northern Humboldt Current System: Brief history, present status and a view towards the future. Prog. Oceanogr. 2008, 79, 95–105. [Google Scholar] [CrossRef]
- Anabalón, V.; Morales, C.E.; Escribano, R.; Varas, M. The contribution of nano- and micro-planktonic assemblages in the surface layer (0–30 m) under different hydrographic conditions in the upwelling area off Concepción, central Chile. Prog. Oceanogr. 2007, 75, 396–414. [Google Scholar] [CrossRef]
- González, H.E.; Menschel, E.; Aparicio, C.; Barría, C. Spatial and temporal variability of microplankton and detritus, and their export to the shelf sediments in the upwelling area off Concepción, Chile (~36°S), during 2002–2005. Prog. Oceanogr. 2007, 75, 435–451. [Google Scholar] [CrossRef]
- Morales, C.E.; González, H.E.; Hormazabal, S.E.; Yuras, G.; Letelier, J.; Castro, L.R. The distribution of chlorophyll-a and dominant planktonic components in the coastal transition zone off Concepción, central Chile, during different oceanographic conditions. Prog. Oceanogr. 2007, 75, 452–469. [Google Scholar] [CrossRef]
- Sánchez, G.E.; Lange, C.B.; González, H.E.; Vargas, G.; Muñoz, P.; Cisternas, C.; Pantoja, S. Siliceous microorganisms in the upwelling center off Concepción, Chile (36°S): Preservation in surface sediments and downcore fluctuations during the past ~150 years. Prog. Oceanogr. 2012, 92–95, 50–65. [Google Scholar] [CrossRef]
- Böttjer, D.; Morales, C.E. Nanoplanktonic assemblages in the upwelling area off Concepción (~36°S), central Chile: Abundance, biomass, and grazing potential during the annual cycle. Prog. Oceanogr. 2007, 75, 415–434. [Google Scholar] [CrossRef]
- Peterson, W.T.; Arcos, D.F.; McManus, G.H.; Dam, H.; Bellantoni, D.; Johnson, T.; Tiselius, P. The nearshore zone during coastal upwelling: Daily variability and coupling between primary and secondary production off central Chile. Prog. Oceanogr. 1998, 20, 1–40. [Google Scholar] [CrossRef]
- Sobarzo, M.; Djurfeldt, L. Coastal upwelling process on a continental shelf limited by submarine canyons, Concepción, central Chile. J. Geophys. Res. Ocean. 2004, 109, C12012. [Google Scholar] [CrossRef]
- Margalef, R. Phytoplankton communities in upwelling areas. The example of NW Africa. Oecologia Aquat. 1978, 3, 97–132. [Google Scholar]
- Margalef, R. The organization of space. Oikos 1979, 33, 152–159. [Google Scholar] [CrossRef]
- Bec, B.; Husseini-Ratrema, J.; Collos, Y.; Souchu, P.; Vaquer, A. Phytoplankton seasonal dynamics in a Mediterranean coastal lagoon: Emphasis on the picoeukaryote community. J. Plankton Res. 2005, 27, 881–894. [Google Scholar] [CrossRef]
- Worden, A.Z.; Nolan, J.K.; Palenik, B. Assessing the dynamics and ecology of marine picophytoplankton: The importance of the eukaryotic component. Limnol. Oceanogr. 2004, 49, 168–179. [Google Scholar] [CrossRef]
- Collado-Fabbri, S.; Vaulot, D.; Ulloa, O. Structure and seasonal dynamics of the eukaryotic picophytoplankton community in a wind-driven coastal upwelling ecosystem. Limnol. Oceanogr. 2011, 56, 2334–2346. [Google Scholar] [CrossRef]
- Vargas, C.A.; Martínez, R.A.; Cuevas, L.A.; Pavez, M.A.; Cartes, C.; González, H.E.; Escribano, R.; Daneri, G. The relative importance of microbial and classical food webs in a highly productive coastal upwelling area. Limnol. Oceanogr. 2007, 52, 1495–1510. [Google Scholar] [CrossRef]
- Azam, F.; Fenchel, T.; Field, J.G.; Gray, J.S.; Meyer-Reil, L.A.; Thingstad, F. The Ecological Role of Water-Column Microbes in the Sea. Mar. Ecol. Prog. Ser. 1983, 10, 257–263. [Google Scholar] [CrossRef]
- Santo Domingo, J.; Edge, T. Identification of primary sources of fecal pollution. In Safe Management of Shellfish and Harvest Waters; Rees, G., Pond, K., Kay, D., Bartram, J., Santo Domingo, J., Eds.; IWA Publishing: London, UK, 2010; pp. 51–90. [Google Scholar]
- Rodríguez, R.A.; Gundy, P.M.; Rijal, G.K.; Gerba, C.P. The Impact of Combined Sewage Overflows on the Viral Contamination of Receiving Waters. Food Environ. Virol. 2012, 4, 34–40. [Google Scholar] [CrossRef]
- WHO. World Health Organization position paper on hepatitis A vaccines—June 2012. Wkly. Epidemiol. Rec. Relev. Épidémiologique Hebd. 2012, 87, 261–276. [Google Scholar]
- Hata, A.; Katayama, H.; Kojima, K.; Sano, S.; Kasuga, I.; Kitajima, M.; Furumai, H. Effects of rainfall events on the occurrence and detection efficiency of viruses in river water impacted by combined sewer overflows. Sci. Total Environ. 2014, 468–469, 757–763. [Google Scholar] [CrossRef]
- Suffredini, E.; Proroga, Y.T.R.; Di Pasquale, S.; Di Maro, O.; Losardo, M.; Cozzi, L.; Capuano, F.; De Medici, D. Occurrence and Trend of Hepatitis A Virus in Bivalve Molluscs Production Areas Following a Contamination Event. Food Environ. Virol. 2017, 9, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Tall, J.A.; Gatton, M.L. Flooding and Arboviral Disease: Predicting Ross River Virus Disease Outbreaks across Inland Regions of South-Eastern Australia. J. Med. Entomol. 2020, 57, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.W.; Donaldson, K.A.; Paul, J.H.; Rose, J.B. Pathogenic Human Viruses in Coastal Waters. Clin. Microbiol. Rev. 2003, 16, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Grodzki, M.; Ollivier, J.; Le Saux, J.C.; Piquet, J.C.; Noyer, M.; Le Guyader, F.S. Impact of Xynthia tempest on Viral Contamination of Shellfish. Appl. Environ. Microbiol. 2012, 78, 3508–3511. [Google Scholar] [CrossRef] [PubMed]
- Campos, C.J.A.; Lees, D.N. Environmental Transmission of Human Noroviruses in Shellfish Waters. Appl. Environ. Microbiol. 2014, 80, 3552–3561. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud de Chile. Boletín Epidemiológico Trimestral, Hepatitis A SE 1–52, 2019. Departamento de Epidemiología. Available online: http://epi.minsal.cl/wp-content/uploads/2020/01/BET_HEPATITIS_2019.pdf (accessed on 10 March 2022).
- Diaz, R.J. Overview of Hypoxia around the World. J. Environ. Qual. 2001, 30, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.J.; Konwar, K.M.; Hallam, S.J. Microbial ecology of expanding oxygen minimum zones. Nat. Rev. Microbiol. 2012, 10, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Schmidtko, S.; Stramma, L.; Visbeck, M. Decline in global oceanic oxygen content during the past five decades. Nature 2017, 542, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Breitburg, D.; Levin, L.A.; Oschlies, A.; Grégoire, M.; Chavez, F.P.; Conley, D.J.; Garçon, V.; Gilbert, D.; Gutiérrez, D.; Isensee, K.; et al. Declining oxygen in the global ocean and coastal waters. Science 2018, 359, eaam7240. [Google Scholar] [CrossRef]
- De La Maza, L.; Farías, L. The intensification of coastal hypoxia off central Chile: Long term and high frequency variability. Front. Earth Sci. 2023, 10, 929271. [Google Scholar] [CrossRef]
- Bakun, A.; Black, B.A.; Bograd, S.J.; García-Reyes, M.; Miller, A.J.; Rykaczewski, R.R.; Sydeman, W.J. Anticipated effects of climate change on coastal upwelling ecosystems. Curr. Clim. Chang. Rep. 2015, 1, 85–93. [Google Scholar] [CrossRef]
- Capone, D.G.; Hutchins, D.A. Microbial biogeochemistry of coastal upwelling regimes in a changing ocean. Nat. Geosci. 2013, 6, 711–717. [Google Scholar] [CrossRef]
- Valle-Levinson, A.; Schneider, W.; Sobarzo, M.; Bello, M.; Bravo, L.; Castillo, M.; Duarte, L.; Fuenzalida, R.; Gallegos, J.M.; Garcés, J.; et al. Wind-induced exchange at the entrance to Concepción Bay, an equatorward facing embayment in central Chile. Deep Sea Res. Part II Top. Stud. Oceanogr. 2004, 51, 2371–2388. [Google Scholar] [CrossRef]

| General Information | ||||||
|---|---|---|---|---|---|---|
| Campaign | Dates | Season | ||||
| CB 1 | 3 January 2018 | Summer | ||||
| CB 2 | 30 May 2018 | Fall | ||||
| CB 3 | 29 August 2018 | Winter | ||||
| CB 4 | 29 November 2018 | Spring | ||||
| Measurements per campaign | ||||||
| Parameters | Method | Parameters | Method | |||
| Physicochemical: | Temperature (°C) | SeaBird SBE−19 plus CTD | Nitrite − NO2− (µM) | Standard methods | ||
| Salinity (psu) | SeaBird SBE−19 plus CTD | Nitrate − NO3− (µM) | Standard methods | |||
| Density (sigma-t) | SeaBird SBE−19 plus CTD | Ammonium − NH4+ (µM) | Standard methods | |||
| Oxygen (mL L−1) | SeaBird SBE−19 plus CTD | Hydrogen Sulfide − H2S (µM) | Spectrophotometry | |||
| Total Suspended Solid − TSS (mg L−1) | Standard methods | |||||
| Chlorophyll-a (mg m−3) | Fluorometry | |||||
| Biological: | Total coliforms (MPN 100 mL−1) | Standard methods | Bacterioplankton (103 cell mL−1) | Cytometry | ||
| Fecal coliforms (MPN 100 mL−1) | Standard methods | Picoeukaryotes (103 cell mL−1) | Cytometry | |||
| Phytoplankton (cell mL−1) | Utermöhl | |||||
| Stations | Latitude | Lengitude | Maximum Depth | Sampling Depth (m) | |
|---|---|---|---|---|---|
| (South) | (West) | (m) | Surface | Bottom | |
| St. 1 | 36°36,68′ | 73°0,89′ | 34 | 2 | 30 |
| St. 2 | 36°38,13′ | 73°4,71′ | 18 | 2 | 15 |
| St. 3 | 36°38,69′ | 73°1,22′ | 29 | 2 | 25 |
| St. 4 | 36°37,46′ | 72°58,43′ | 22 | 2 | 20 |
| St. 5 | 36°41,52′ | 73°4,65′ | 8 | 2 | 13 |
| St. 6 | 36°41,03′ | 73°2,03′ | 22 | 2 | 18 |
| St. 7 | 36°42,01′ | 73°0,17′ | 19 | 2 | 15 |
| St. 8 | 36°43,09′ | 73°3,70′ | 8 | 2 | 6 |
| Measurements | Summer | Fall | Winter | Spring | ||||
|---|---|---|---|---|---|---|---|---|
| CB 1 | CB 2 | CB 3 | CB 4 | |||||
| Surface | Bottom | Surface | Bottom | Surface | Bottom | Surface | Bottom | |
| Temperature (°C) | 12.80 ± 0.78 | 10.60 ± 0.79 | 12.40 ± 0.15 | 12.42 ± 0.06 | 11.55 ± 0.10 | 11.06 ± 0.20 | 12.22 ± 0.14 | 11.21 ± 0.47 |
| Salinity (psu) | 34.47 ± 0.04 | 34.51 ± 0.04 | 33.22 ± 0.33 | 33.91 ± 0.08 | 33.93 ± 0.11 | 34.45 ± 0.21 | 34.39 ± 0.05 | 34.50 ± 0.05 |
| Density (Sigma-t) | 26.01 ± 0.16 | 26.46 ± 0.17 | 25.13 ± 0.24 | 25.66 ± 0.07 | 25.84 ± 0.10 | 26.34 ± 0.19 | 26.08 ± 0.05 | 26.35 ± 0.12 |
| Oxygen (mL L−1) | 5.46 ± 0.88 | 1.61 ± 1.61 | 5.51 ± 0.25 | 4.70 ± 0.44 | 5.13 ± 0.36 | 2.72 ± 0.84 | 4.01 ± 0.95 | 1.84 ± 1.43 |
| Chlorophyll−a (mg m−3) | 22.32 ± 5.32 | 16.77 ± 11.22 | 3.12 ± 1.41 | 2.29 ± 0.60 | 2.44 ± 0.42 | 2.03 ± 0.75 | 2.39 ± 2.51 | 1.47 ± 1.14 |
| Total coliforms (MPN 100 mL−1) | 12.38 ± 9.88 | 13.62 ± 18.61 | 1595.0 ± 2050.32 | 52.83 ± 61.54 | 54.20 ± 61.34 | 37.28 ± 61.83 | BDL | BDL |
| Fecal coliforms (MPN 100 mL−1) | 2.00 ± 0 | 14.0 ± 16.97 | 134.75 ± 149.73 | 14.61 ± 15.96 | 9.76 ± 7.58 | 18.50 ± 26.44 | BDL | BDL |
| NO2− (µM) | 0.23 ± 0.13 | 3.33 ± 2.90 | 0.18 ± 0.03 | 0.16 ± 0.02 | 0.19 ± 0.03 | 0.12 ± 0.07 | 0.48 ± 0.10 | 0.43 ± 0.19 |
| NO3− (µM) | 16.87 ± 0.45 | 22.5 ± 4.16 | 12.68 ± 2.34 | 11.72 ± 1.28 | 13.76 ± 2.18 | 12.04 ± 8.57 | 24.7 ± 2.66 | 27.22 ± 3.79 |
| NH4+ (µM) | 5.45 ± 3.03 | 3.39 ± 1.19 | 0.94 ± 0.39 | 1.01 ± 0.40 | 0.19 ± 0.09 | 0.15 ± 0.02 | 1.92 ± 0.50 | 1.70 ± 0.37 |
| H2S (µM) | BDL | 12.09 ± 0.96 | BDL | BDL | BDL | BDL | BDL | BDL |
| TSS (mg L−1) | 39.08 ± 4.93 | 34.91 ± 3.94 | 12.55 ± 1.79 | 9.76 ± 1.22 | 8.37 ± 1.44 | 10.9 ± 2.34 | 15.01 ± 5.48 | 19.4 ± 8.76 |
| Phytoplankton (cell mL−1) | 1649.00 ± 533.90 | 813.90 ± 568.00 | 204.70 ± 96.60 | 373.50± 318.20 | 600.20 ± 391.10 | 799.60 ± 341.30 | 443.60 ± 594.70 | 289.60 ± 195.10 |
| Bacterioplankton (103 cell mL−1) | 594.30 ± 210.80 | 637.10 ± 200.90 | 660.40 ± 301.00 | 462.40 ± 218.50 | 1033.00 ± 312.30 | 544.20 ± 164.40 | 527.70 ± 110.70 | 464.10 ± 190.70 |
| Picoeukaryotes (103 cell mL−1) | BDL | BDL | 1.70 ± 0.50 | 1.10 ± 0.30 | 11.10 ± 6.40 | 4.10 ± 3.40 | 1.80 ± 1.60 | 0.80 ± 0.70 |
| Df | Sums of Squares | Mean Squares | F. Model | R2 | Pr (>F) | ||
|---|---|---|---|---|---|---|---|
| Seasonality | 3 | 17,413,563 | 5,804,521 | 8.5158 | 0.23688 | 0.001 | *** |
| Stratus | 1 | 3,358,419 | 3,358,419 | 4.9271 | 0.04569 | 0.001 | *** |
| Sampling Stations | 1 | 908,203 | 908,203 | 1.3324 | 0.01235 | 0.286 | |
| Seasonality:Stratus | 3 | 10,656,583 | 3,552,194 | 5.2114 | 0.14496 | 0.001 | *** |
| Seasonality:Stations | 3 | 5,459,898 | 1,819,966 | 2.6701 | 0.07427 | 0.032 | * |
| Stratus:Stations | 1 | 952,764 | 952,764 | 1.3978 | 0.01296 | 0.269 | |
| Residuals | 51 | 34,762,614 | 681,620 | 0.47288 | |||
| Total | 63 | 73,512,045 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bustos-Espinoza, L.; Torres-Ramírez, P.; Figueroa, S.; González, P.S.; Pavez, M.A.; Jerez, R.; Saldías, G.S.; Espinoza, C.; Galán, A. Biogeochemical Response of the Water Column of Concepción Bay, Chile, to a New Regime of Atmospheric and Oceanographic Variability. Geosciences 2024, 14, 125. https://doi.org/10.3390/geosciences14050125
Bustos-Espinoza L, Torres-Ramírez P, Figueroa S, González PS, Pavez MA, Jerez R, Saldías GS, Espinoza C, Galán A. Biogeochemical Response of the Water Column of Concepción Bay, Chile, to a New Regime of Atmospheric and Oceanographic Variability. Geosciences. 2024; 14(5):125. https://doi.org/10.3390/geosciences14050125
Chicago/Turabian StyleBustos-Espinoza, Luis, Patricio Torres-Ramírez, Sergio Figueroa, Pablo S. González, Marcelo A. Pavez, Rodolfo Jerez, Gonzalo S. Saldías, Claudio Espinoza, and Alexander Galán. 2024. "Biogeochemical Response of the Water Column of Concepción Bay, Chile, to a New Regime of Atmospheric and Oceanographic Variability" Geosciences 14, no. 5: 125. https://doi.org/10.3390/geosciences14050125
APA StyleBustos-Espinoza, L., Torres-Ramírez, P., Figueroa, S., González, P. S., Pavez, M. A., Jerez, R., Saldías, G. S., Espinoza, C., & Galán, A. (2024). Biogeochemical Response of the Water Column of Concepción Bay, Chile, to a New Regime of Atmospheric and Oceanographic Variability. Geosciences, 14(5), 125. https://doi.org/10.3390/geosciences14050125










