Mixing Sows into Alternative Lactation Housing Affects Sow Aggression at Mixing, Future Reproduction and Piglet Injury, with Marked Differences between Multisuckle and Sow Separation Systems
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
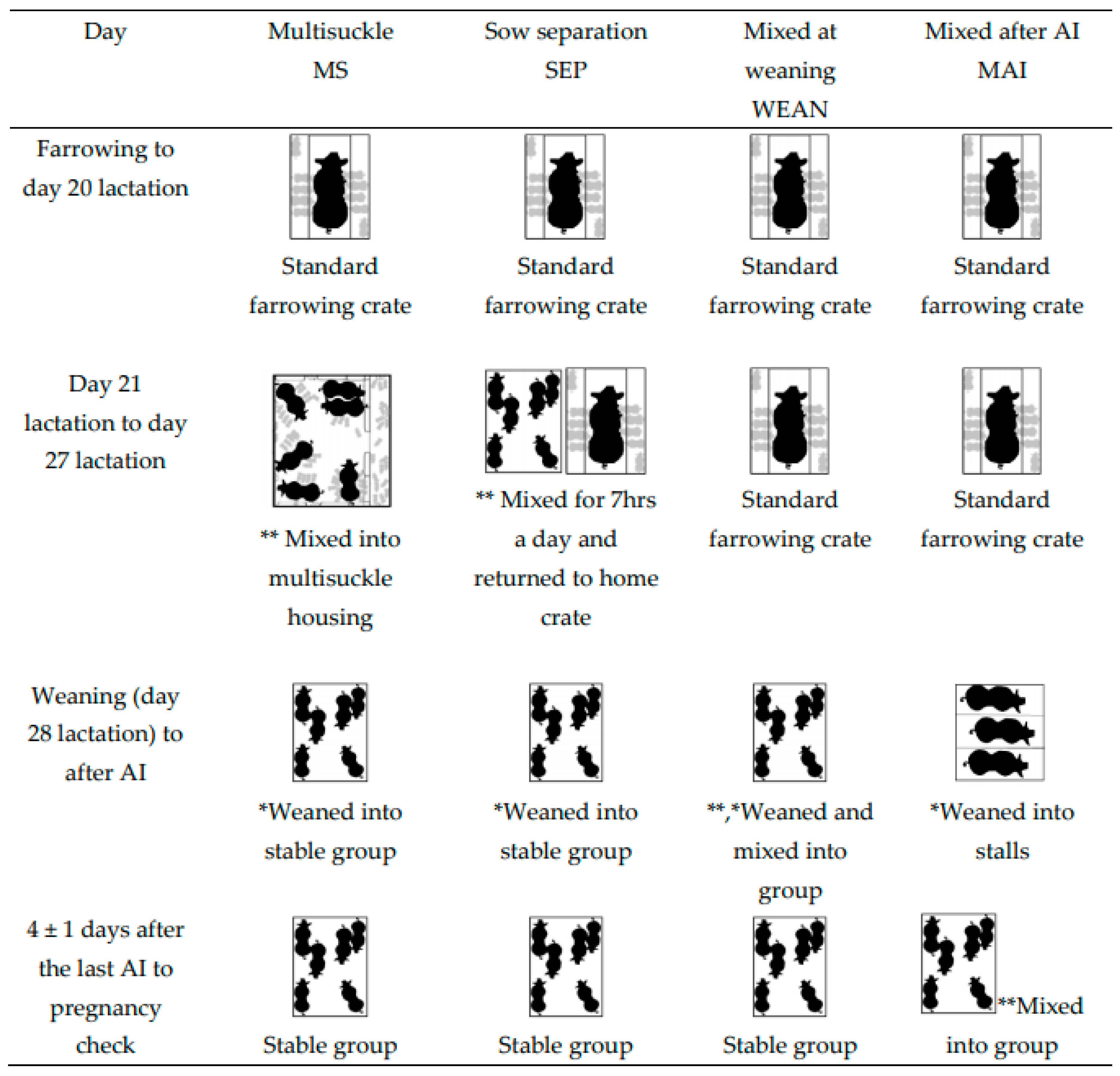
2.1. Animals and Treatments
- Group-housed sows and litters (multisuckle) from Day 21 lactation (MS).MS sows (n = 30) were mixed with their litters into groups of 6 (allowing 4.7 m2 of sow accessible space plus 1.1 m2 of creep per litter, or 5.8 m2/sow and litter total area, Figure 2) beginning on Day 21 post-farrowing (which was calculated from the piggery’s set weaning day, which was an average day 28 of lactation). The sows and litter remained in the same housing until weaning. Piglets were then weaned from the sows on day 28 of lactation into the same stable groups and sows were inseminated in the same groups (2 m2/sow after weaning).
- Sows separated from crates and litters for seven hours each day (by mixing the sows alone into a group pen, and later returning to their home crates and litters) (SEP). Beginning on Day 21 post-farrowing, SEP sows (n = 30) were removed from their standard lactation crate and mixed away from their piglets, into groups of six, for seven hours daily (from 06:50 to 13:50). Piglets remained in the home crate and sows were returned to the piglets after the days mixing. Sows in the SEP treatment group had their piglets weaned (lactation day 28) from them and were then mixed into, and inseminated in, these same stable groups (2 m2/sow after weaning).
- Sows mixed together after weaning at Day 28 lactation (WEAN). WEAN sows (n = 30) were weaned from standard farrowing crates directly into pens of 6, providing 2 m2/sow, and inseminated in the same stable groups.
- Sows placed into stalls and then mixed 4 ± 1 days after the last insemination (MAI). MAI sows (n = 30) were weaned into stalls on Day 28 lactation. After their last AI (4 ± 1 days), the sows were mixed in groups of six (2 m2/sow).
2.2. Feeding
2.3. Piglets
2.4. Sow Behavioural Observations
2.5. Sow Injury Counts and Locomotion
2.6. Sow Saliva Sample Collection and Analysis
2.7. Sow Reproduction
2.8. Piglet Measures
2.9. Statistical Analyses
3. Results
3.1. Injury
3.2. Aggressive Behaviour
3.3. Other Behaviours
3.4. Salivary Cortisol Concentration
3.5. Reproduction
3.6. Piglet Results
3.6.1. Piglet Weights
3.6.2. Piglet Total Circulating Cortisol Concentrations
3.6.3. Piglet Injury Scores
3.6.4. Piglet Mortality
4. Discussion
4.1. Mixing Day
4.2. Sow Multisuckle Housing in Lactation
4.3. Sow Socialisation and Separation from Piglets in Lactation
4.4. Mixing Sows After Weaning
4.5. The Piglets
4.5.1. Mortality in Alternative Lactation Systems
4.5.2. Piglet Growth and Final Weights
4.5.3. Piglet Stress and Injury
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barnett, J.L.; Hemsworth, P.H.; Cronin, G.M.; Jongman, E.C.; Hutson, G.D. A review of the welfare issues for sows and piglets in relation to housing. Aust. J. Agric. Res. 2001, 52, 1–28. [Google Scholar] [CrossRef]
- Arey, D.S.; Sancha, E.S. Behaviour and productivity of sows and piglets in a family system and in farrowing crates. Appl. Anim. Behav. Sci. 1996, 50, 135–145. [Google Scholar] [CrossRef]
- D’Eath, R.B. Socialising piglets before weaning improves social hierarchy formation when pigs are mixed post-weaning. Appl. Anim. Behav. Sci. 2005, 93, 199–211. [Google Scholar] [CrossRef]
- Pedersen, L.J.; Berg, P.; Jørgensen, G.; Andersen, I.L. Neonatal traits of importance for survival in crates and indoor pens. J. Anim. Sci. 2011, 89, 1207–1218. [Google Scholar] [CrossRef]
- Van Nieuwamerongen, S.E.; Bolhuis, J.E.; van der Peet-Schwering, C.M.; Soede, N.M. A review of sow and piglet behaviour and performance in group housing systems for lactating sows. Animal 2014, 8, 448–460. [Google Scholar] [CrossRef]
- Campbell, J.M.; Crenshaw, J.D.; Polo, J. The biological stress of early weaned piglets. J. Anim. Sci. Biotechnol. 2013, 4, 1–4. [Google Scholar] [CrossRef]
- Greenwood, E.C.; Plush, K.J.; van Wettere, W.H.; Hughes, P.E. Hierarchy formation in newly mixed, group housed sows and management strategies aimed at reducing its impact. Appl. Anim. Behav. Sci. 2014, 160, 1–11. [Google Scholar] [CrossRef]
- Verdon, M.; Hansen, C.F.; Rault, J.L.; Jongman, E.; Hansen, L.U.; Plush, K.; Hemsworth, P. Effects of group housing on sow welfare: A review. J. Anim. Sci. 2015, 93, 1999–2017. [Google Scholar] [CrossRef]
- Greenwood, E.C.; Plush, K.J.; van Wettere, W.H.; Hughes, P.E. Group and individual sow behavior is altered in early gestation by space allowance in the days immediately following grouping. J. Anim. Sci. 2016, 94, 385–393. [Google Scholar] [CrossRef]
- Arey, D.S.; Edwards, S.A. Factors influencing aggression between sows after mixing and the consequences for welfare and production. Livest. Prod. Sci. 1998, 56, 61–70. [Google Scholar] [CrossRef]
- Kirkwood, R.; Zanella, A.J. Influence of gestation housing on sow welfare and productivity. In National Pork Board Final Report; Michigan State University: East Lansing, MI, USA, 2005; Available online: https://www.researchgate.net/publication/242544635_Title_Influence_of_Gestation_Housing_on_Sow_Welfare_and_Productivity_-_NPB_03-055 (accessed on 7 March 2019).
- Von Borell, E.; Dobson, H.; Prunier, A. Stress, behaviour and reproductive performance in female cattle and pigs. Horm. Behav. 2007, 52, 130–138. [Google Scholar] [CrossRef]
- Turner, A.I.; Hemsworth, P.H.; Tilbrook, A.J. Susceptibility of reproduction in female pigs to impairment by stress and the role of the hypothalamo-pituitary-adrenal axis. Reprod. Fertil. Dev. 2002, 14, 377–391. [Google Scholar] [CrossRef]
- Einarsson, S.; Brandt, Y.; Rodriguez-Martinez, H.; Madej, A. Conference lecture: Influence of stress on estrus, gametes and early embryo development in the sow. Theriogenology 2008, 70, 1197–1201. [Google Scholar] [CrossRef]
- Van Wettere, W.H.; Pain, S.J.; Stott, P.G.; Hughes, P.E. Mixing gilts in early pregnancy does not affect embryo survival. Anim. Reprod. Sci. 2008, 104, 382–388. [Google Scholar] [CrossRef]
- Spoolder, H.A.M.; Geudeke, M.J.; van der Peet-Schwering, C.M.C.; Soede, N.M. Group housing of sows in early pregnancy: A review of success and risk factors. Livest. Sci. 2009, 125, 1–14. [Google Scholar] [CrossRef]
- Soede, N.; Roelofs, J.; Verheijen, R.; Schouten, W.; Hazeleger, W.; Kemp, B. Effect of repeated stress treatments during the follicular phase and early pregnancy on reproductive performance of gilts. Reprod. Domest. Anim. 2007, 42, 135–142. [Google Scholar] [CrossRef]
- Tilbrook, A.J.; Clarke, I.J. Neuroendocrine mechanisms of innate states of attenuated responsiveness of the hypothalamo-pituitary adrenal axis to stress. Front. Neuroendocrinol. 2006, 27, 285–307. [Google Scholar] [CrossRef]
- Windle, R.J.; Brady, M.M.; Kunanandam, T.; Da Costa, A.P.; Wilson, B.C.; Harbuz, M.; Lightman, S.L.; Ingram, C.D. Reduced response of the hypothalamo-pituitary-adrenal axis to alpha1-agonist stimulation during lactation. Endocrinology 1997, 138, 3741–3748. [Google Scholar] [CrossRef]
- Hasiec, M.; Misztal, T. Adaptive Modifications of Maternal Hypothalamic-Pituitary-Adrenal Axis Activity during Lactation and Salsolinol as a New Player in this Phenomenon. Int. J. Endocrinol. 2018, 2018, 3786038. [Google Scholar] [CrossRef]
- NHMRC, Australian Code of Practice for the Care and Use of Animals for Scientific Purposes. 2013. Available online: http://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/ea28_code_care_use_animals_131209.pdf (accessed on 7 March 2019).
- Karlen, G.A.M.; Hemsworth, P.H.; Gonyou, H.W.; Fabrega, E.; Strom, A.D.; Smits, R.J. The welfare of gestating sows in conventional stalls and large groups on deep litter. Appl. Anim. Behav. Sci. 2007, 105, 87–101. [Google Scholar] [CrossRef]
- Widowski, T.M.; Cottrell, T.; Dewey, C.E.; Friendship, R.M. Observations of piglet-directed behavior patterns and skin lesions in eleven commercial swine herds. JSHAP 2003, 11, 181–185. [Google Scholar]
- Cui, S.; Chen, D.; Li, J.; Li, X.; Yin, G.; Bao, J. A comparison of postural changes and maternal responsiveness during early lactation in Landrace and Minipig sows. Appl. Anim. Behav. Sci. 2011, 131, 40–47. [Google Scholar] [CrossRef]
- Nowicki, J.; Klocek, C.; Schwarz, T. Factors affecting maternal behaviour and responsiveness in sows during periparturient and lactation periods. Ann. Anim. Sci. 2012, 12, 455–469. [Google Scholar] [CrossRef]
- Uvnäs-Moberg, K.; Johansson, B.; Lupoli, B.; Svennersten-Sjaunja, K. Oxytocin facilitates behavioural, metabolic and physiological adaptations during lactation. Appl. Anim. Behav. Sci. 2001, 72, 225–234. [Google Scholar] [CrossRef]
- Langendijk, P.; Soede, N.M.; Kemp, B. Effects of boar contact and housing conditions on oestrus expression in weaned sows. J. Anim. Sci. 2000, 78, 871–878. [Google Scholar] [CrossRef]
- Terry, R.; Kind, K.L.; Hughes, P.E.; Kennaway, D.J.; Herde, P.J.; van Wettere, W.H.E.J. Split weaning increases the incidence of lactation oestrus in boar-exposed sows. Anim. Reprod. Sci. 2013, 142, 48–55. [Google Scholar] [CrossRef]
- Thomsson, O.; Sjunnesson, Y.; Magnusson, U.; Eliasson-Selling, L.; Wallenbeck, A.; Bergqvist, A.S. Consequences for Piglet Performance of Group Housing Lactating Sows at One, Two, or Three Weeks Post-Farrowing. PLoS ONE 2016, 11, e0156581. [Google Scholar] [CrossRef]
- Weary, D.M.; Pajor, E.A.; Bonenfant, M.; Fraser, D.; Kramer, D.L. Alternative housing for sows and litters: Part 4. Effects of sow-controlled housing combined with a communal piglet area on pre- and post-weaning behaviour and performance. Appl. Anim. Behav. Sci. 2002, 76, 279–290. [Google Scholar] [CrossRef]
- Rault, J.; Morrison, R.; Hansen, C.; Hansen, L.; Hemsworth, P. Effects of group housing after weaning on sow welfare and sexual behaviour. J. Anim. Sci. 2014, 9, 5683–5692. [Google Scholar] [CrossRef]
- Kemp, B.; Soede, N.M.; Langendijk, P. Effects of boar contact and housing condition on oestrus expression in sows. Theriogenology 2005, 63, 643–656. [Google Scholar] [CrossRef]
- Hultén, F.; Lundeheim, N.; Dalin, A.M.; Einarsson, S. Pre- and post-weaning piglet performance, sow food intake and change in backfat thickness in a group housing system for lactating sows. Acta Vet. Scand. 1997, 38, 119–133. [Google Scholar]
- Rantzer, D.; Svendsen, J.; Weström, B. Weaning of pigs in group housing and in conventional housing systems for lactating sows. Swed. J. Agric. Res. 1997, 27, 23–31. [Google Scholar]
- Wattanakul, W.; Sinclair, A.G.; Stewart, A.H.; Edwards, S.A.; English, P.R. Performance and behaviour of lactating sows and piglets in crate and multisuckling systems: A study involving European White and Manor Meishan genotypes. Anim. Sci. 1997, 64, 339–349. [Google Scholar] [CrossRef]
- Gerritsen, R.; Soede, N.M.; Langedijk, P.; Hazeleger, W.; Kemp, B. The intermittent suckling regimen in pigs: Consequences for reproductive performance of sows. Reprod. Domes. Anim. 2008, 43, 29–35. [Google Scholar] [CrossRef]
- Verdon, M.; Morrison, R.; Rault, J.L. 1A113 Welfare Implications of Group Lactation at Various Ages. Report Prepared for the Co-Operative Research Centre for High Integrity Australian Pork 2017. Available online: http://porkcrc.com.au/wp-content/uploads/2017/09/1A-113-Final-report.pdf (accessed on 7 March 2019).
- Lambertz, C.; Petig, M.; Elkmann, A.; Gauly, M. Confinement of sows for different periods during lactation: Effects on behaviour and lesions of sows and performance of piglets. Animal 2015, 9, 1373–1378. [Google Scholar] [CrossRef]
- Berkeveld, M.; Langendijk, P.; Soede, N.M.; Kemp, B.; Taverne, M.A.M.; Verheijden, J.H.M.; Kuijken, N.; Koets, A.P. Improving adaptation to weaning: Effect of intermittent suckling regimens on piglet feed intake, growth, and gut characteristics. J. Anim. Sci. 2009, 87, 3156–3166. [Google Scholar] [CrossRef]
- Hessel, E.F.; Reiners, K.; Van den Weghe, H.F.A. Socializing piglets before weaning: Effects on behaviour of lactating sows, pre- and postweaning behaviour, and performance of piglets. J. Anim. Sci. 2006, 84, 2847–2855. [Google Scholar] [CrossRef]
- Kutzer, T.; Bünger, B.; Kjaer, J.B.; Schrader, L. Effects of early contact between non-littermate piglets and of the complexity of farrowing conditions on social behaviour and weight gain. Appl. Anim. Behav. Sci. 2009, 121, 16–24. [Google Scholar] [CrossRef]
- Kuller, W.I.; Soede, N.M.; van Beers-Schreurs, H.M.G.; Langendijk, P.; Taverne, M.A.M.; Kemp, B. Intermittent suckling: Effects on piglet and sow performance before and after weaning. Anim. Sci. 2004, 82, 405–413. [Google Scholar] [CrossRef]
- Thymann, T.; Gudbergsen, C.; Bresson, S.; Kristensen, N.B.; Hansen, C.F. Intermittent suckling improves post-weaning feed uptake but does not change functional gut characteristics of piglets. Livest. Sci. 2007, 108, 132–136. [Google Scholar] [CrossRef]
- Reimert, I.; Bolhuis, J.E.; Kemp, B.; Rodenburg, T.B. Indicators of positive and negative emotions and emotional contagion in pigs. Physiol. Behav. 2013, 109, 42–50. [Google Scholar] [CrossRef]
- Petersen, H.V.; Vestergaard, K.; Jensen, P. Integration of piglets into social groups of free-ranging domestic pigs. Appl. Anim. Behav. Sci. 1989, 23, 223–236. [Google Scholar] [CrossRef]
- Hotzel, M.J.; de Souza, G.P.P.; Dalla Costa, O.A.; Filho, L.C.P. Disentangling the effects of weaning stressors on piglets’ behaviour and feed intake: Changing the housing and social environment. Appl. Anim. Behav. Sci. 2011, 135, 44–50. [Google Scholar] [CrossRef]
- Verdon, M. Rearing Piglets in Multi-litter Group Lactation Systems: Effects on Piglet Aggression and Injuries Post-weaning. Appl. Anim. Behav. Sci. 2016, 183, 35–41. [Google Scholar] [CrossRef]


| Active, resting, eating and drinking | Sows were classed as eating or drinking if they were obviously doing either of the two. If the sow’s behaviour was unclear, she was coded as active. If a sow was dog sitting, standing or walking she was considered active. Sows were considered resting if flat to the floor. | Continuous events |
| Exploring | Actively manipulating and exploring the surrounding environment, such as rooting, moving drinkers and chewing fences. | Continuous event |
| Displacement | Movement of one sow by another, from a valued resource such as food, drinker or lying space (if multiple knocks or bites are required, this is a fight). | Point event |
| Fighting | Aggression lasting >3 s, including two or more knocks or bites. Aggression can be reciprocal or non-reciprocal. | Continuous event |
| Knock | One sow knocks another sow using her head and neck, contacting any part of the receiving sow. | Point event |
| Bite | One single bite delivered from one sow to any part of another. | Point event |
| Lunge | Sow lunges at another but does not make physical contact | Point event |
| Flee | Sow moves herself quickly and as far away as she can get from another sow, in response to an aggressive action. | Point event |
| Mounting | One sow mounts another, with her front legs both over the back of the other animal. | Point event |
| Suckling | Sows in multisuckle housing could be coded as suckling: Suckling began when sows began to actively grunt on their side and piglets rushed to the udder. Suckling finished when sows rolled onto their udder, blocking access to the piglets. | Continuous |
| Interaction with piglets (non-aggressive or aggressive) | Exclusive to sows in multisuckle housing. Investigation of piglets, including nose-to-nose contact and moving the piglets around. The modifier aggressive or non-aggressive was used. Interaction was considered as aggressive if piglets were pushed with force so that they fell, were thrown, or were bitten. | Continuous |
| Boar exposure | Not coded for MAI. Boar exposure was coded from the start of exposure, to when the boar was no longer present or the sows returned to their housing. There were no other behaviours coded while boar exposure was coded for, leaving a blank section of video when exposure was taking place. | Continuous |
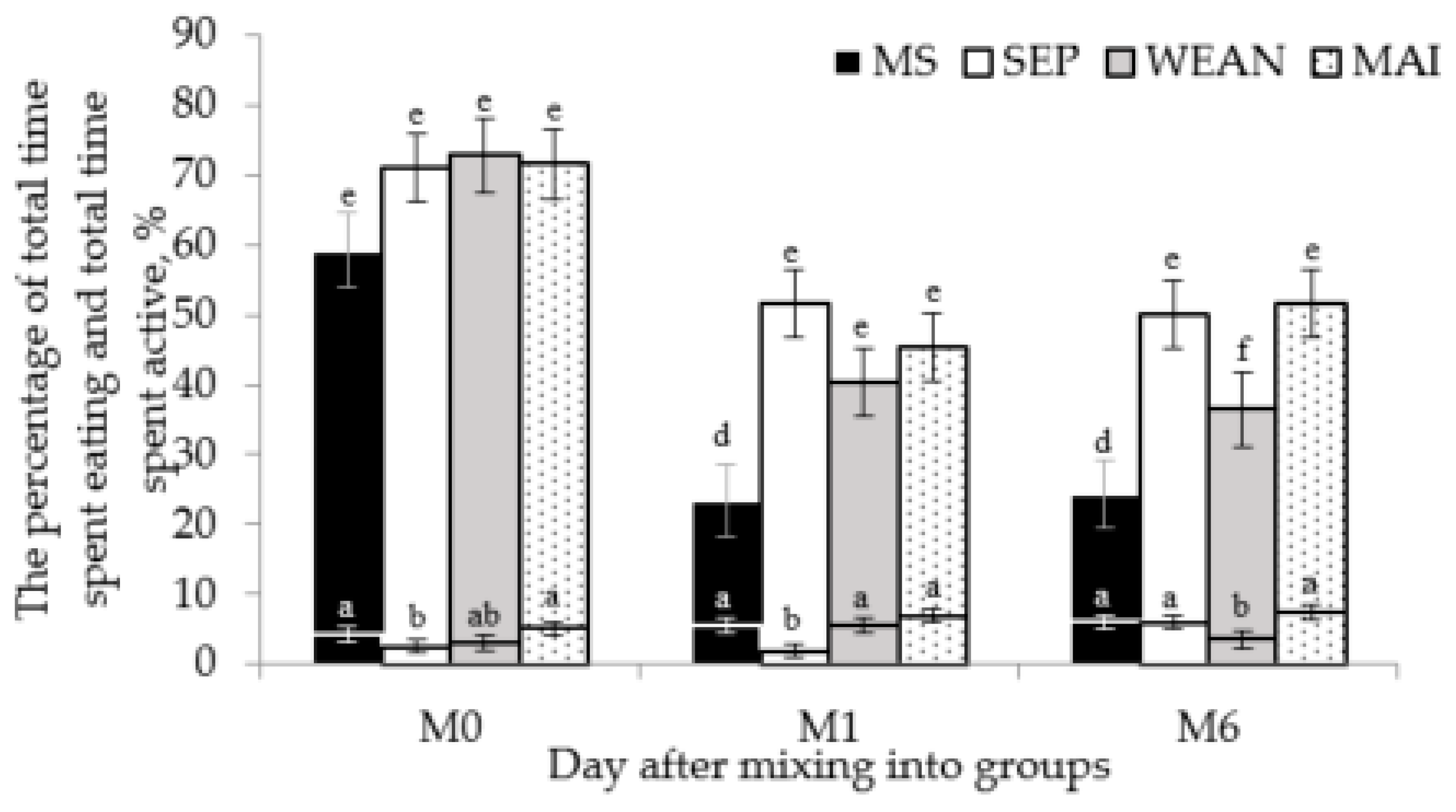
| Total time active | All continuous behaviour codes considered as a sow being active were summed, allowing for differences in behaviour between treatments, such as suckling in the MS treatment group (all behaviour except time spent resting). | Calculated after |
| Mixing Day | Treatment | Total Injury, Number per Sow | Front Total Injury, Number | Fights per Hour, Number |
|---|---|---|---|---|
| M-1 | MS | 5.6 ± 4.2 a | 2.0 ± 2.1 a | |
| SEP | 6.7 ± 4.2 a | 2.5 ± 2.1 a | ||
| WEAN | 4.4 ± 3.8 a | 1.8 ± 1.9 a | ||
| MAI | 4.4 ± 3.8 a | 2.1 ± 1.9 a | ||
| M0 | MS | 14.6 ± 3.8 a | 6.7 ± 1.9 a | 0.27 ± 0.07 |
| SEP | 32.3 ± 3.8 b | 15.1 ± 1.9 b | 0.48 ± 0.07 | |
| WEAN | 16.3 ± 3.8 a | 7.2 ± 1.9 a | 0.27 ± 0.07 | |
| MAI | 19.7 ± 3.8 a | 11.2 ± 1.9 b | 0.20 ± 0.07 | |
| M1 | MS | 11.4 ± 4.2 a | 5.1 ± 2.1 a | 0.00 + 0.06 |
| SEP | 34.4 ± 3.8 b | 15.9 ± 1.9 b | 0.10 ± 0.06 | |
| WEAN | 20.7 ± 3.8 c | 10.8 ± 1.9 a | 0.06 ± 0.06 | |
| MAI | 21.6 ± 3.8 c | 14.3 ± 1.9 b | 0.06 ± 0.06 | |
| M6 | MS | 9.5 ± 3.8 a | 4.1 ± 1.9 a | 0.00 ± 0.06 |
| SEP | 36.1 ± 3.8 b | 18.2 ± 1.9 b | 0.08 ± 0.06 | |
| WEAN | 39.1 ± 3.8 b | 20.3 ± 1.9 b | 0.13 ± 0.07 | |
| MAI | 40.9 ± 3.8 b | 24.0 ± 1.9 b | 0.06 ± 0.06 | |
| Original Transformation | Sqrt | Sqrt | Lg10(1 + x) | |
| p value | <0.0001 | <0.0001 | >0.05 | |
| Behaviour | M0 | M1 | M6 | Trans. | p Value |
|---|---|---|---|---|---|
| fights per hour | 0.3 ± 0.03 a | 0.05 ± 0.03 b | 0.07 ± 0.03 b | Lg10(1 + x) | <0.0001 |
| fight duration, second | 34.1 ± 6.2 a | 3.1 ± 6.0 b | 3.9 ± 6.0 b | Lg10(1 + x) | <0.0001 |
| Time spent fighting, % | 0.3 ± 0.08 | 0.004 ± 0.08 | 0.08 ± 0.08 | NA | <0.05 |
| bites per sow per hr | 2.4 ± 0.2 a | 0.3 ± 0.2 b | 0.6 ± 0.02 b | Lg10(1 + x) | 0.001 |
| knocks per sow per hr | 1.2 ± 0.3 a | 0.4 ± 0.2 b | 1.2 ± 0.2 a | Lg10(1 + x) | < 0.001 |
| displacements, per sow per hr | 0.4 ± 0.04 a | 0.1 ± 0.04 b | 0.2 ± 0.04 b | Log | <0.05 |
| spent exploring, % | 2.3 ± 0.4 a | 1.4 ± 0.4 b | 2.2 ± 0.1 ab | Sqrt | <0.05 |
| Behaviour | MS | SEP | WEAN | MAI | Trans. | p Value |
|---|---|---|---|---|---|---|
| fights per hour, per sow | 0.08 ± 0.04 a | 0.20 ± 0.03 b | 0.15 ± 0.04 ab | 0.11 ± 0.03 ab | Lg10(1 + x) | <0.05 |
| fight duration, seconds | 3.0 ± 7.5 a | 15.6 ± 7.0 b | 16.9 ± 7.5 b | 16.5 ± 7.0 b | Lg10(1 + x) | <0.05 |
| Bites per hour, per sow | 0.4 ± 0.2 a | 2.1 ± 0.2 b | 1.0 ± 0.2 c | 0.8 ± 0.2 c | Lg10(1 + x) | <0.0001 |
| Knocks per hour, per sow | 0.4 ± 0.3 a | 1.5 ± 0.3 b | 1.1 ± 0.3 ab | 0.7 ± 0.3 ab | Lg10 | <0.05 |
| Behaviour | MS | SEP | WEAN | MAI | Trans. | p Value |
|---|---|---|---|---|---|---|
| Sows which failed to show oestrus, % | 6.7 ± 4.9 | 16.7 ± 4.9 | 10.0 ± 4.9 | 0.0 ± 4.9 | NA | >0.1 |
| Pregnant, % sows mated | 93.3 ± 7.0 | 90.0 ± 7.0 | 91.7 ± 7.0 | 80.0 ± 7.0 | NA | >0.05 |
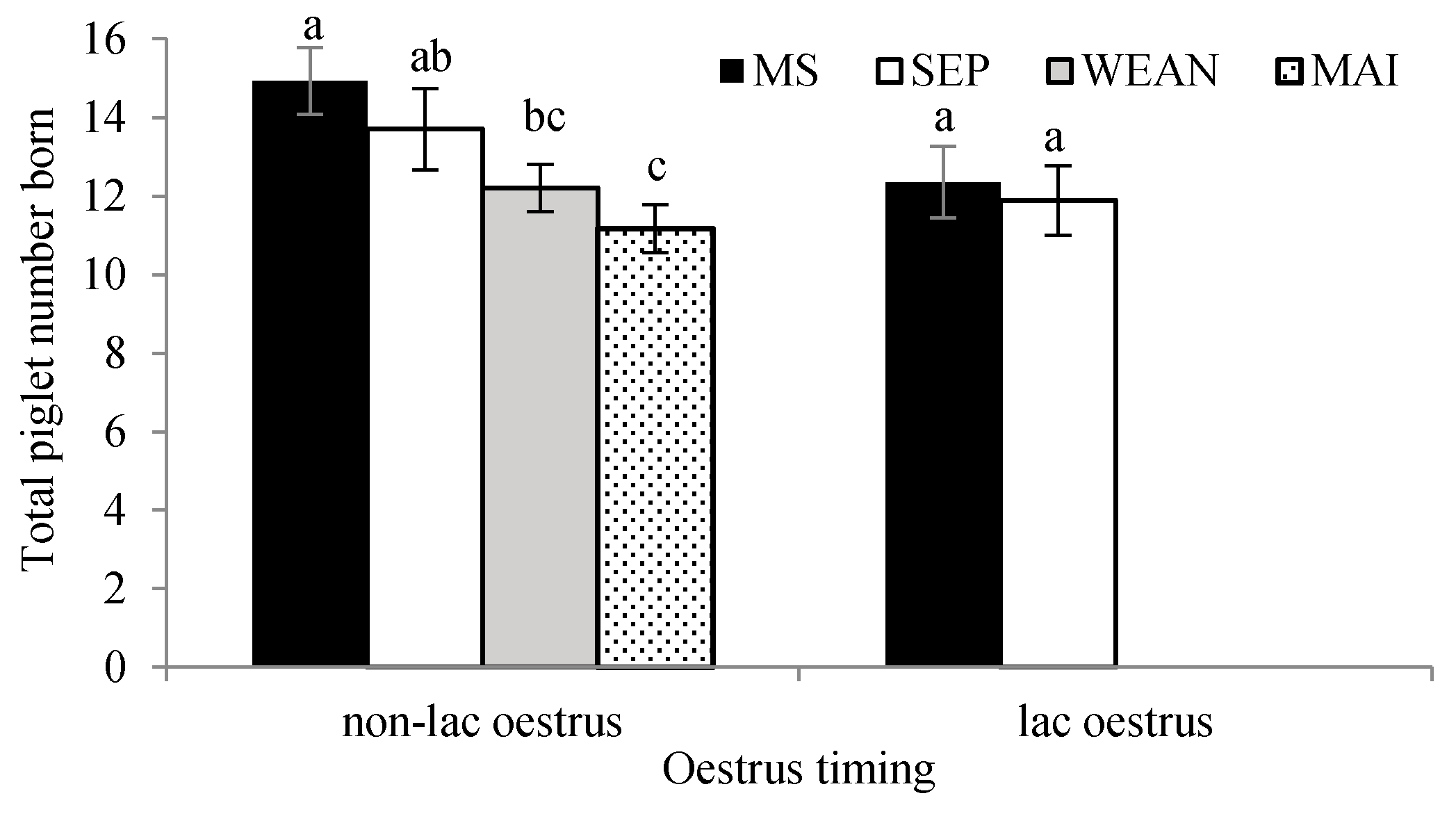
| Total born, piglets | 13.8 ± 0.5 a | 12.2 ± 0.5 a,b | 12.3 ± 0.5 a,b | 11.0 ± 0.5 b | Sqrt | <0.05 |
| Number of days from weaning to oestrus | 1.0 ± 0.7 a | 1.0 ± 0.7 a | 4.3 ± 0.7 b | 4.9 ± 0.7 b | NA | <0.005 |
| Lactation oestrus, % | 50.7 ± 10.1 a | 61.7 ± 10.1 a | 0.0 ± 10.1 b | 0.0 ± 10.1 b | Sqrt | <0.0001 |
| Heading | Baseline (Lac 19/20) | Lac 24 1 | Lac 28/W 1 | W1 1 | W2 1 | Tran. | p Value |
|---|---|---|---|---|---|---|---|
| Weight, kg | * 6.1 ± 0.2 a | 7.2 ± 0.2 b | 8.1 ± 0.2 c | 8.3 ± 0.2 c | 8.3 ± 0.2 c | NA | <0.001 |
| Injury score | * 0.43 ± 0.04 a | 0.39 ± 0.04 a | 1.06 ± 0.04 b | 1.25 ± 0.04 b | Sqrt | <0.001 | |
| Cortisol, ng/ml | # 31.8 ± 4.6 a | 29.1 ± 4.4 a | 50.4 ± 4.6 b | Lg10 | <0.005 |
| Mixing Day 2 | Treatment 1 | Weight Change, kg | Injury Score Change | Mixing Day 2 | Cortisol Change, ng/ml |
|---|---|---|---|---|---|
| Lac 20 to Lac 24 | C | 1.22 ± 0.26 | −0.1 ± 0.08 a | Lac 19 to Lac 24 | −2.06 ± 10.27 |
| MS | 0.8 ± 0.27 | 0.12 ± 0.09 b | 9.59 ± 14.25 | ||
| SEP | 1.06 ± 0.27 | −0.09 ± 0.09 a | 4.53 ± 11.77 | ||
| Lac 24 to Lac 28/W | C | 0.97 ± 0.26 | 0.77 ± 0.08 a | Lac 24 to W1 | 29.34 ± 11.04 |
| MS | 1.19 ± 0.27 | 0.31 ± 0.08 b | −6.3 ± 12.95 | ||
| SEP | 1.01 ± 0.27 | 0.87 ± 0.08 a | 21.95 ± 11.77 | ||
| Lac 28/W to W1 | C | 0.03 ± 0.26 | 0.01 ± 0.08 a | ||
| MS | 0.09 ± 0.27 | 0.19 ± 0.09 ab | |||
| SEP | 0.39 ± 0.27 | 0.26 ± 0.09 b | |||
| W1 to W2 | C | 0.09 ± 0.26 | |||
| MS | 0.16 ± 0.27 | ||||
| SEP | -0.08 ± 0.27 | ||||
| p value | >0.05 | <0.0005 | >0.05 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greenwood, E.C.; van Dissel, J.; Rayner, J.; Hughes, P.E.; van Wettere, W.H.E.J. Mixing Sows into Alternative Lactation Housing Affects Sow Aggression at Mixing, Future Reproduction and Piglet Injury, with Marked Differences between Multisuckle and Sow Separation Systems. Animals 2019, 9, 658. https://doi.org/10.3390/ani9090658
Greenwood EC, van Dissel J, Rayner J, Hughes PE, van Wettere WHEJ. Mixing Sows into Alternative Lactation Housing Affects Sow Aggression at Mixing, Future Reproduction and Piglet Injury, with Marked Differences between Multisuckle and Sow Separation Systems. Animals. 2019; 9(9):658. https://doi.org/10.3390/ani9090658
Chicago/Turabian StyleGreenwood, Emma C., Jonathon van Dissel, Jessica Rayner, Paul E. Hughes, and William H. E. J. van Wettere. 2019. "Mixing Sows into Alternative Lactation Housing Affects Sow Aggression at Mixing, Future Reproduction and Piglet Injury, with Marked Differences between Multisuckle and Sow Separation Systems" Animals 9, no. 9: 658. https://doi.org/10.3390/ani9090658
APA StyleGreenwood, E. C., van Dissel, J., Rayner, J., Hughes, P. E., & van Wettere, W. H. E. J. (2019). Mixing Sows into Alternative Lactation Housing Affects Sow Aggression at Mixing, Future Reproduction and Piglet Injury, with Marked Differences between Multisuckle and Sow Separation Systems. Animals, 9(9), 658. https://doi.org/10.3390/ani9090658





