Effects of Dietary Selenium Deficiency or Excess on Selenoprotein Gene Expression in the Spleen Tissue of Pigs
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Pigs and Diets
2.3. Quantification of Selenoproteins mRNA
2.4. Statistical Analysis
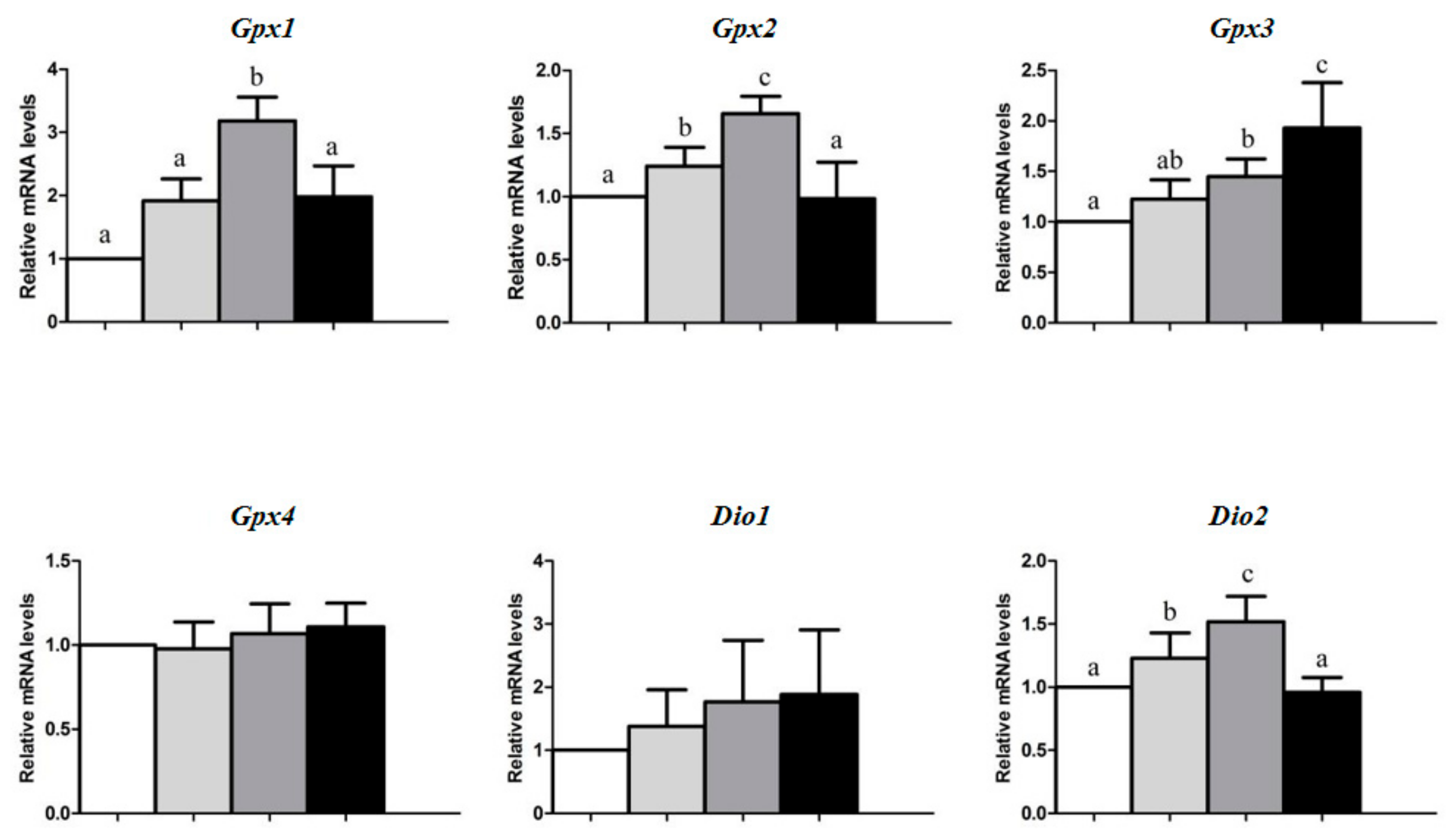
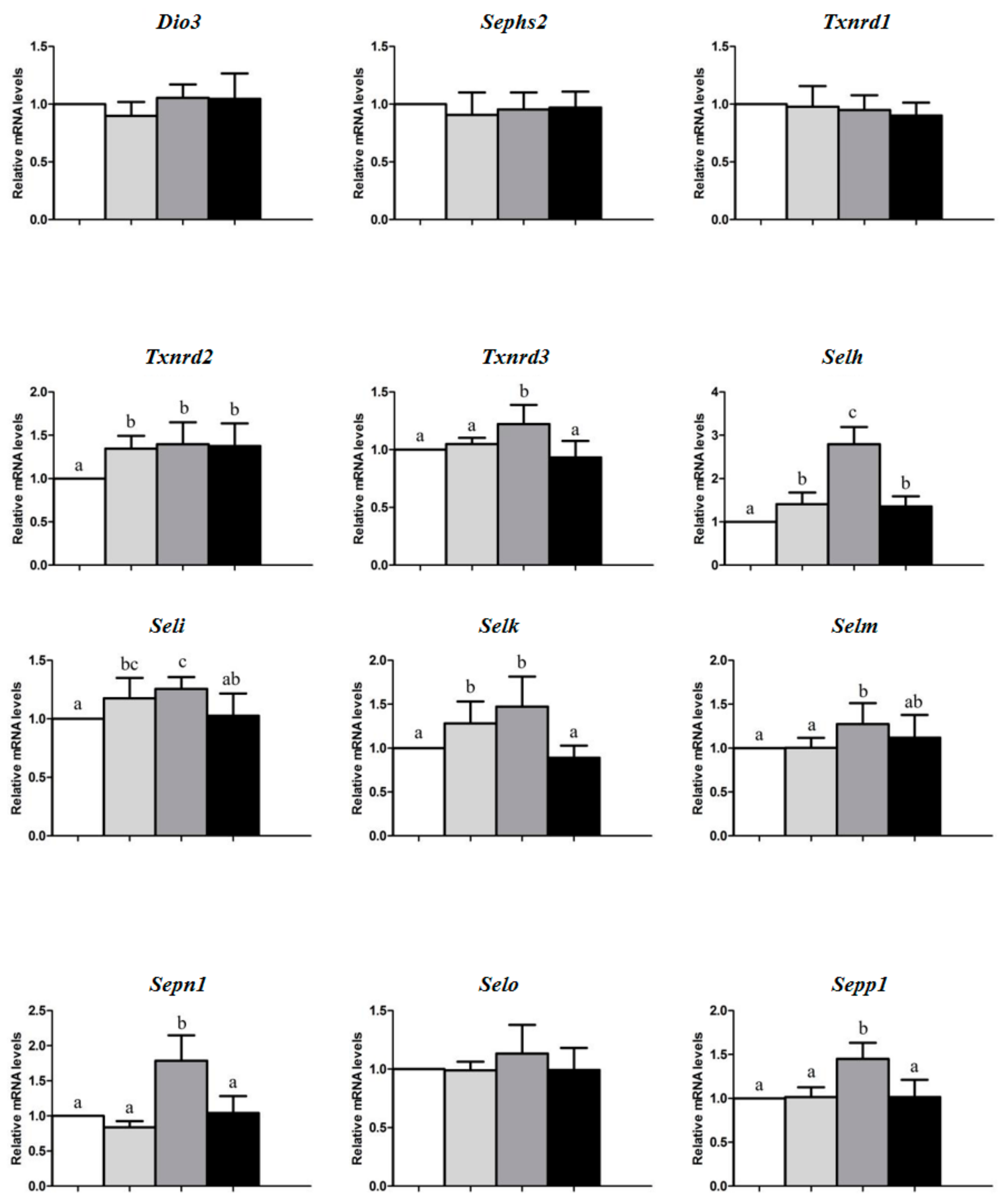
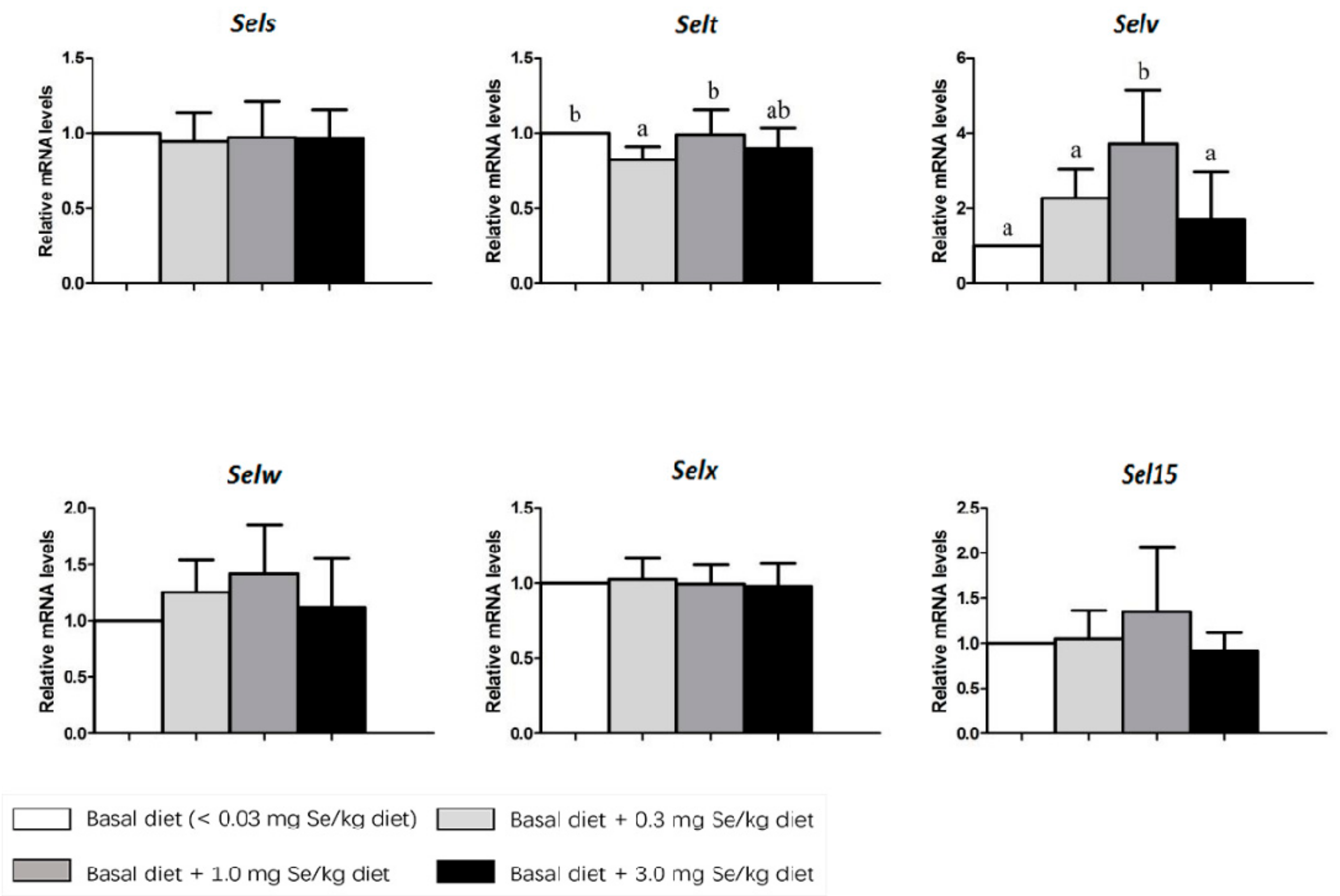
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Loflin, J.; Lopez, N.; Whanger, P.D.; Kioussi, C. Selenoprotein W during development and oxidative stress. J. Inorg. Biochem. 2006, 100, 1679–1684. [Google Scholar] [CrossRef] [PubMed]
- Li, J.L.; Gao, R.; Li, S.; Wang, J.T.; Tang, Z.X.; Xu, S.W. Testicular toxicity induced by dietary cadmium in cocks and ameliorative effect by selenium. Biometals 2010, 23, 695–705. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F., Jr.; Clark, L.C.; Turnbull, B.W. An analysis of cancer prevention by selenium. Biofactors 2001, 14, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Chariot, P.; Bignani, O. Skeletal muscle disorders associated with selenium deficiency in humans. Muscle Nerve. 2003, 27, 662–668. [Google Scholar] [CrossRef]
- Terry, E.N.; Michal, J.J.; Hostetler, C.E.; Kincaid, R.L. Levels of mRNA for three selenoproteins in skeletal muscle of fetal and newborn pigs. Livest. Sci. 2009, 124, 21–25. [Google Scholar] [CrossRef]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Schweizer, U.; Schomburg, L.; Savaskan, N.E. The neurobiology of selenium: Lessons from transgenic mice. J. Nutr. 2004, 134, 707–710. [Google Scholar] [CrossRef]
- Martin-Romero, F.J.; Kryukov, G.V.; Lobanov, A.V.; Carlson, B.A.; Lee, B.J.; Gladyshev, V.N.; Hatfield, D.L. Selenium metabolism in Drosophila: Selenoproteins, selenoprotein mRNA expression, fertility, and mortality. J. Biol. Chem. 2001, 276, 29798–29804. [Google Scholar] [CrossRef]
- Kaur, P.; Bansal, M.P. Effect of selenium-induced oxidative stress on the cell kinetics in testis and reproductive ability of male mice. Nutrition 2005, 21, 351–357. [Google Scholar] [CrossRef]
- McKenzie, R.C.; Rafferty, T.S.; Beckett, G.J. Selenium: An essential element for immune function. Immunol. Today 1998, 19, 342–345. [Google Scholar] [CrossRef]
- Hoffmann, P.R.; Berry, M.J. The influence of selenium on immune responses. Mol. Nutr. Food Res. 2008, 52, 1273–1280. [Google Scholar] [CrossRef] [PubMed]
- Oda, S.S.; El-Maddawy, Z.K. Protective effect of vitamin E and selenium combination on deltamethrin-induced reproductive toxicity in male rats. Exp. Toxicol. Pathol. 2012, 64, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.C.; Polk, B.F.; Morris, J.S.; Stampfer, M.J.; Pressel, S.; Rosner, B.; Taylor, J.O.; Schneider, K.; Hames, C.G. Prediagnostic serum selenium and risk of cancer. Lancet 1983, 322, 130–134. [Google Scholar] [CrossRef]
- Green, D.E.; Albers, P.H. Diagnostic criteria for selenium toxicosis in aquatic birds: Histologic lesions. J. Wildl. Dis. 1997, 33, 385–404. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Cui, Y.; Cui, W.; Deng, J.; Cui, H. The decrease of relative weight, lesions, and apoptosis of bursa of Fabricius induced by excess dietary selenium in chickens. Biol. Trace Elem. Res. 2009, 131, 33–42. [Google Scholar] [CrossRef]
- Vega, L.; Rodríguez-Sosa, M.; García-Montalvo, E.A.; Del Razo, L.M.; Elizondo, G. Non-optimal levels of dietary selenomethionine alter splenocyte response and modify oxidative stress markers in female mice. Food Chem. Toxicol. 2007, 45, 1147–1153. [Google Scholar] [CrossRef]
- Ueno, H.; Hasegawa, G.; Ido, R.; Okuno, T.; Nakamuro, K. Effect of selenium status and supplementary selenoi-chemical sources on mouse T-cell mitogenesis. J. Trace Elem. Med. Biol. 2008, 22, 9–16. [Google Scholar] [CrossRef]
- Peng, X.; Cui, H.; Deng, J.; Zuo, Z.; Lai, W. Histological lesion of spleen and inhibition of splenocyte proliferation in broilers fed on diets excess in selenium. Biol. Trace Elem. Res. 2011, 140, 66–72. [Google Scholar] [CrossRef]
- Behne, D.; Kyriakopoulos, A. Mammalian seleniumcontaining proteins. Annu. Rev. Nutr. 2001, 21, 453–473. [Google Scholar] [CrossRef]
- Stadtman, T.C. Selenium biochemistry. Mammalian selenoenzymes. Ann. N. Y. Acad. Sci. 2000, 899, 399–402. [Google Scholar] [CrossRef]
- Zhou, J.C.; Zhao, H.; Li, J.G.; Xia, X.J.; Wang, K.N.; Zhang, Y.J.; Liu, Y.; Zhao, Y.; Lei, X.G. Selenoprotein gene expression in thyroid and pituitary of young pigs is not affected by dietary selenium deficiency or excess. J. Nutr. 2009, 139, 1061–1066. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Q.; Li, D.L.; Zhao, H.; Sun, L.H.; Xia, X.J.; Wang, K.N.; Luo, X.; Lei, X.G. The selenium deficiency disease exudative diathesis in chicks is associated with downregulation of seven common selenoprotein genes in liver and muscle. J. Nutr. 2011, 141, 1605–1610. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhao, H.; Zhang, Q.; Tang, J.; Li, K.; Xia, X.J.; Wang, K.N.; Li, K.; Lei, X.G. Prolonged dietary selenium deficiency or excess does not globally affect selenoprotein gene expression and/or protein production in various tissues of pigs. J. Nutr. 2012, 142, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Shrimali, R.K.; Irons, R.D.; Carlson, B.A.; Sano, Y.; Gladyshev, V.N.; Park, J.M.; Hatfield, D.L. Selenoproteins mediate T cell immunity through an antioxidant mechanism. J. Biol. Chem. 2008, 283, 20181–20185. [Google Scholar] [CrossRef]
- Miller, E.R.; Ullrey, D.E. The pig as a model for human nutrition. Annu. Rev. Nutr. 1987, 7, 361–382. [Google Scholar] [CrossRef]
- Rowan, A.M.; Moughan, P.J.; Wilson, M.N.; Maher, K.; Tasman-Jones, C. Comparison of the ileal and faecal digestibility of dietary amino acids in adult humans and evaluation of the pig as a model animal for digestion studies in man. Br. J. Nutr. 1994, 71, 29–42. [Google Scholar] [CrossRef]
- Sullivan, T.P.; Eaglstein, W.H.; Davis, S.C.; Mertz, P. The pig as a model for human wound healing. Wound Repair Regen. 2001, 9, 66–76. [Google Scholar] [CrossRef]
- Wassen, F.W.; Klootwijk, W.; Kaptein, E.; Duncker, D.J.; Visser, T.J.; Kuiper, G.G. Characteristics and thyroid state-dependent regulation of iodothyronine deiodinases in pigs. Endocrinology 2004, 145, 4251–4263. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Marsh, J.A.; Combs, G.F., Jr.; Whitacre, M.E.; Dietert, R.R. Effect of selenium and vitamin E dietary deficiencies on chick lymphoid organ development. Proc. Soc. Exp. Biol. Med. 1986, 182, 425–436. [Google Scholar] [CrossRef]
- Reeves, M.A.; Hoffmann, P.R. The human selenoproteome: Recent insights into functions and regulation. Cell. Mol. Life Sci. 2009, 66, 2457–2478. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.G.; Dann, H.M.; Ross, D.A.; Cheng, W.H.; Combs, G.F.; Roneker, K.R. Dietary selenium supplementation is required to support full expression of three selenium-dependent glutathione peroxidases in various tissues of weanling pigs. J. Nutr. 1998, 128, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Hrdina, J.; Banning, A.; Kipp, A.; Loh, G.; Blaut, M.; Brigelius-Flohé, R. The gastrointestinal microbiota affects the selenium status and selenoprotein expression in mice. J. Nutr. Biochem. 2009, 20, 638–648. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Guo, F.C.; Zhang, L.Y.; Dong, B.; Gong, L.M. Effects of dietary Selenomethionine supplementation on growth performance, antioxidant status, plasma selenium concentration, and immune function in weaning pigs. J. Anim. Sci. Biotechnol. 2014, 5, 46. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.C. Cracking the code for thyroid hormone signaling. Trans. Am. Clin. Climatol. Assoc. 2012, 124, 26. [Google Scholar]
- Lin, S.L.; Wang, C.W.; Tan, S.R.; Liang, Y.; Yao, H.D.; Zhang, Z.W.; Xu, S.W. Selenium deficiency inhibits the conversion of thyroidal thyroxine (T4) to triiodothyronine (T3) in chicken thyroids. Biol. Trace Elem. Res. 2014, 161, 263–271. [Google Scholar] [CrossRef]
- Sunde, R.A.; Raines, A.M. Selenium regulation of the selenoprotein and nonselenoprotein transcriptomes in rodents. Adv. Nutr. 2011, 2, 138–150. [Google Scholar] [CrossRef]
- Arthur, J.R.; Nicol, F.; Beckett, G.J.; Trayhurn, P. Impairment of iodothyronine 5′-deiodinase activity in brown adipose tissue and its acute stimulation by cold in selenium deficiency. Can. J. Physiol. Pharmacol. 1991, 69, 782–785. [Google Scholar] [CrossRef]
- Wang, R.; Sun, B.; Zhang, Z.; Li, S.; Xu, S. Dietary selenium influences pancreatic tissue levels of selenoprotein W in chickens. J. Inorg. Biochem. 2011, 105, 1156–1160. [Google Scholar] [CrossRef]
- Zhou, J.C.; Zhao, H.; Tang, J.Y.; Li, J.G.; Liu, X.L.; Zhu, Y.M. Molecular cloning, chromosomal localization and expression profiling of porcine selenoprotein M gene. Genes Genom. 2011, 33, 529–534. [Google Scholar] [CrossRef]
- Sun, Q.A.; Wu, Y.; Zappacosta, F.; Jeang, K.T.; Lee, B.J.; Hatfield, D.L.; Gladyshev, V.N. Redox regulation of cell signaling by selenocysteine in mammalian thioredoxin reductases. J. Biol. Chem. 1999, 274, 24522–24530. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Holmgren, A. Selenoproteins. J. Biol. Chem. 2009, 284, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.P.; Fu, J.; Lin, S.L.; Wang, X.S.; Li, S. Effects of dietary selenium deficiency on mrna levels of twenty-one selenoprotein genes in the liver of layer chicken. Biol. Trace Elem. Res. 2014, 159, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, S.V.; Kryukov, G.V.; Xu, X.M.; Carlson, B.A.; Hatfield, D.L.; Gladyshev, V.N. Selenoprotein H is a nucleolar thioredoxin-like protein with a unique expression pattern. J. Biol. Chem. 2007, 282, 11960–11968. [Google Scholar] [CrossRef] [PubMed]
- Du, S.; Zhou, J.; Jia, Y.; Huang, K. SelK is a novel ER stress-regulated protein and protects HepG2 cells from ER stress agent-induced apoptosis. Arch. Biochem. Biophys. 2010, 502, 137–143. [Google Scholar] [CrossRef]
- Kelly, E.; Greene, C.M.; Carroll, T.P.; McElvaney, N.G.; O’Neill, S.J. Selenoprotein S/SEPS1 modifies endoplasmic reticulum stress in Z variant alpha1-antitrypsin deficiency. J. Biol. Chem. 2009, 284, 16891–16897. [Google Scholar] [CrossRef]
- Yu, D.; Li, J.L.; Zhang, J.L.; Gao, X.J.; Xu, S. Effects of dietary selenium on selenoprotein W gene expression in the chicken immune organs. Biol. Trace Elem. Res. 2011, 144, 678–687. [Google Scholar] [CrossRef]
| Gene | Accession Number | Primer Sequence (5′→3′) |
|---|---|---|
| Glutathione peroxidase 1 (Gpx1) | AF532927 | GATGCCACTGCCCTCATGA |
| TCGAAGTTCCATGCGATGTC | ||
| Glutathione peroxidase 2 (Gpx2) | DQ898282 | AGAATGTGGCCTCGCTCTGA |
| GGCATTGCAGCTCGTTGAG | ||
| Glutathione peroxidase 3 (Gpx3) | AY368622 | TGCACTGCAGGAAGAGTTTGAA |
| CCGGTTCCTGTTTTCCAAATT | ||
| Glutathione peroxidase 4 (Gpx4) | NM_214407 | TGAGGCAAGACGGAGGTAAACT |
| TCCGTAAACCACACTCAGCATATC | ||
| Deiodinase type I (Dio1) | AY533206 | CATGGCCAAGAACCCTCACT |
| CCAGAAATACTGGGCACTGAAGA | ||
| Deiodinase type II (Dio2) | NM_001001626 | GCCACTTGACCTCCTTTCACT |
| CTGGTTCTGGTGCTTCTTCAC | ||
| Deiodinase type III (Dio3) | AY533208 | TGAAGTGGAGCTCAACAGTGATG |
| TGTCGTCAGACACGCAGATAGG | ||
| Selenophosphate synthetase 2 (Sephs2) | EF033624 | TGGCTTGATGCACACGTTTAA |
| TGCGAGTGTCCCAGAATGC | ||
| Thioredoxin reductase 1 (Txnrd1) | AF537300 | GATTTAACAAGCGGGTCATGGT |
| CAACCTACATTCACACACGTTCCT | ||
| Thioredoxin reductase 2 (Txnrd2) | NM_001168702 | CAATGCTACGACCTCCTGGT |
| GGCGAAGGGCTCACATAGTC | ||
| Thioredoxin reductase 3 (Txnrd3) | XM_005669829 | CACTTTCAGCATCCACCACAT |
| TAAATCCATCCCTTTCCTCGT | ||
| Selenoprotein H (Selh) | NM_001184948 | GAAGGGAATGGAGGAGGCTA |
| TTCACCCTCACTGGAAGCTC | ||
| Selenoprotein I (Seli) | NM_001244662 | GATGGTGTGGATGGAAAGCAA |
| GCCATGGTCAAAGAGTTCTCCTA | ||
| Selenoprotein K (Selk) | DQ372075 | CAGGAAACCCCCCTAGAAGAA |
| CTCATCCACCGGCCATTG | ||
| Selenoprotein M (Selm) | FJ968780 | CAGCTGAATCGCCTCAAAGAG |
| GAGATGTTTCATGACCAGGTTGTG | ||
| Selenoprotein N, 1 (Sepn1) | EF113595 | ACCTGGTCCCTGGTGAAAGAG |
| AGGCCAGCCAGCTTCTTGT | ||
| Selenoprotein O (Selo) | AK236851 | CTTCCGACCCCAGATGGAT |
| GGTTCGACTGTGCCAGCAT | ||
| Selenoprotein P1 (Sepp1) | EF113596 | AACCAGAAGCGCCAGACACT |
| TGCTGGCATATCTCAGTTCTCAGA | ||
| Selenoprotein S (Sels) | GU983865 | ACAGGAGGCTTTAGCAGCAG |
| CGCTGTCCCATCTTTCAATC | ||
| Selenoprotein T (Selt) | NM_001163408 | CGCTGCTCAAATTCCAGATA |
| CTCTCCTTCAATGCGGATGT | ||
| Selenoprotein V (Selv) | GQ478346 | CACTGGTCGCCAATGGATTC |
| AGTGGCCAACGGAGAAAGC | ||
| Selenoprotein W (Selw) | NM_213977 | CACCCCTGTCTCCCTGCAT |
| GAGCAGGATCACCCCAAACA | ||
| Selenoprotein X (Selx_) | EF113597 | ATCCCTAAAGGCCAAGAATCATC |
| GGCCACCAAGCAGTGTTCA | ||
| Selenoprotein 15 (Sep15) | EF178474 | ACAGCCCTGCCAAGCAGAT |
| AACAGGGAGGCTGGGTAACAC | ||
| β-actin | AY550069 | CCCAAAGCCAACCGTGAGAA |
| CCACGTACATGGCTGGGGTG |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, Z.; Wang, P.; Teng, T.; Shi, B.; Shan, A.; Lei, X.G. Effects of Dietary Selenium Deficiency or Excess on Selenoprotein Gene Expression in the Spleen Tissue of Pigs. Animals 2019, 9, 1122. https://doi.org/10.3390/ani9121122
Lu Z, Wang P, Teng T, Shi B, Shan A, Lei XG. Effects of Dietary Selenium Deficiency or Excess on Selenoprotein Gene Expression in the Spleen Tissue of Pigs. Animals. 2019; 9(12):1122. https://doi.org/10.3390/ani9121122
Chicago/Turabian StyleLu, Zhuang, Pengzu Wang, Teng Teng, Baoming Shi, Anshan Shan, and Xin Gen Lei. 2019. "Effects of Dietary Selenium Deficiency or Excess on Selenoprotein Gene Expression in the Spleen Tissue of Pigs" Animals 9, no. 12: 1122. https://doi.org/10.3390/ani9121122
APA StyleLu, Z., Wang, P., Teng, T., Shi, B., Shan, A., & Lei, X. G. (2019). Effects of Dietary Selenium Deficiency or Excess on Selenoprotein Gene Expression in the Spleen Tissue of Pigs. Animals, 9(12), 1122. https://doi.org/10.3390/ani9121122






