Neutering Effects on Social Behaviour of Urban Unowned Free-Roaming Domestic Cats
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Description of the Cat Group
2.3. Experimental Procedure
2.3.1. Data Collection
2.3.2. Data Analyses
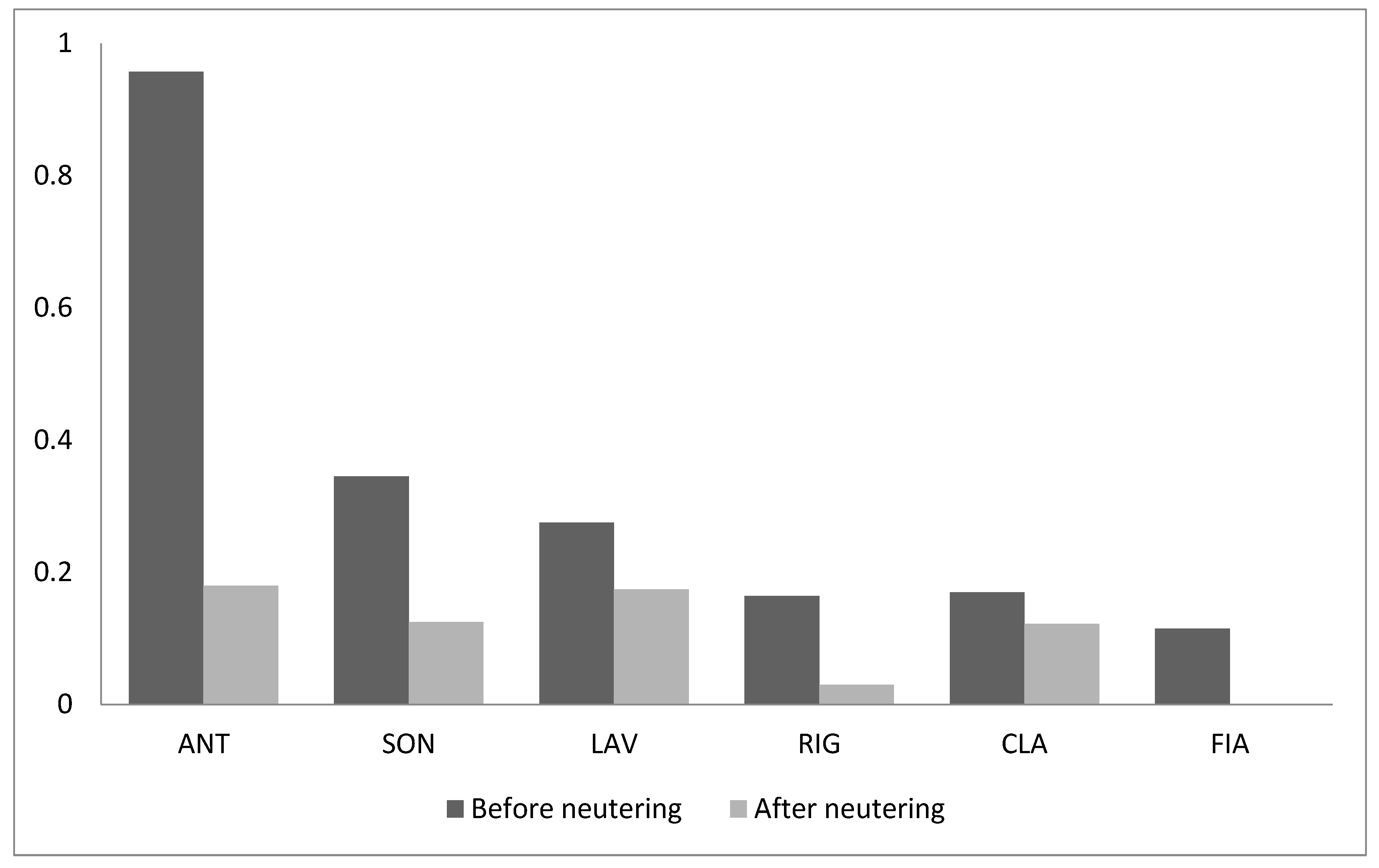
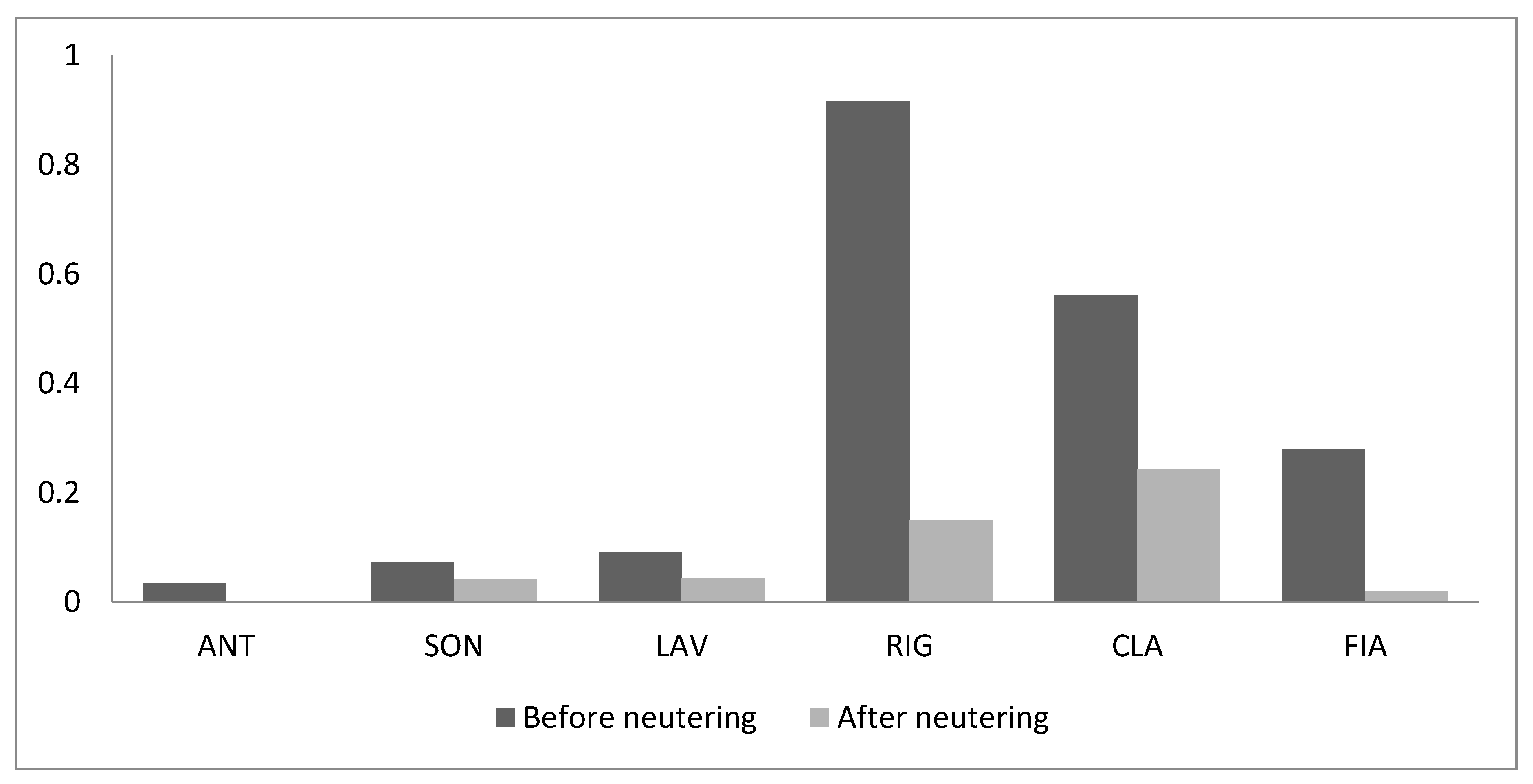
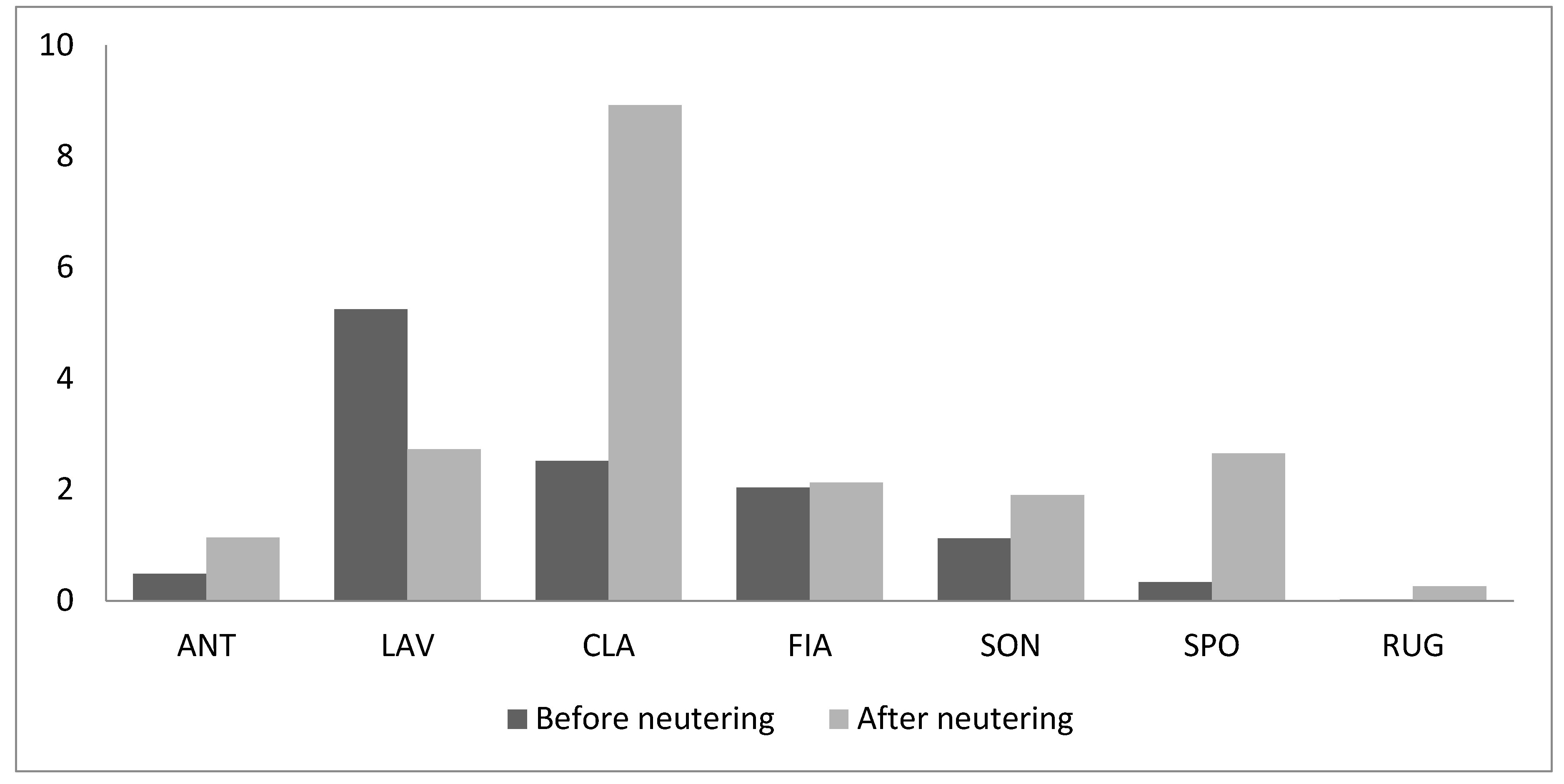
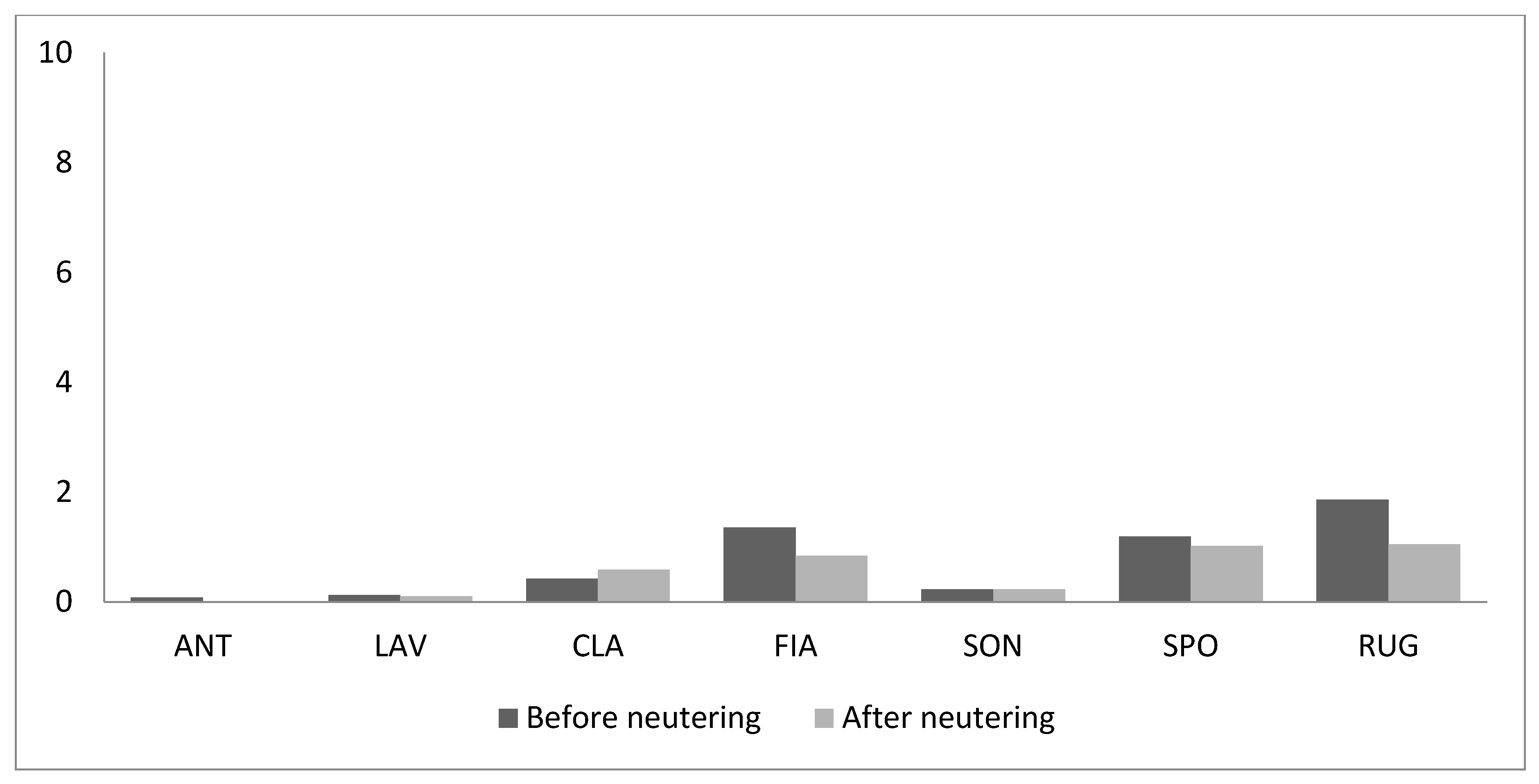
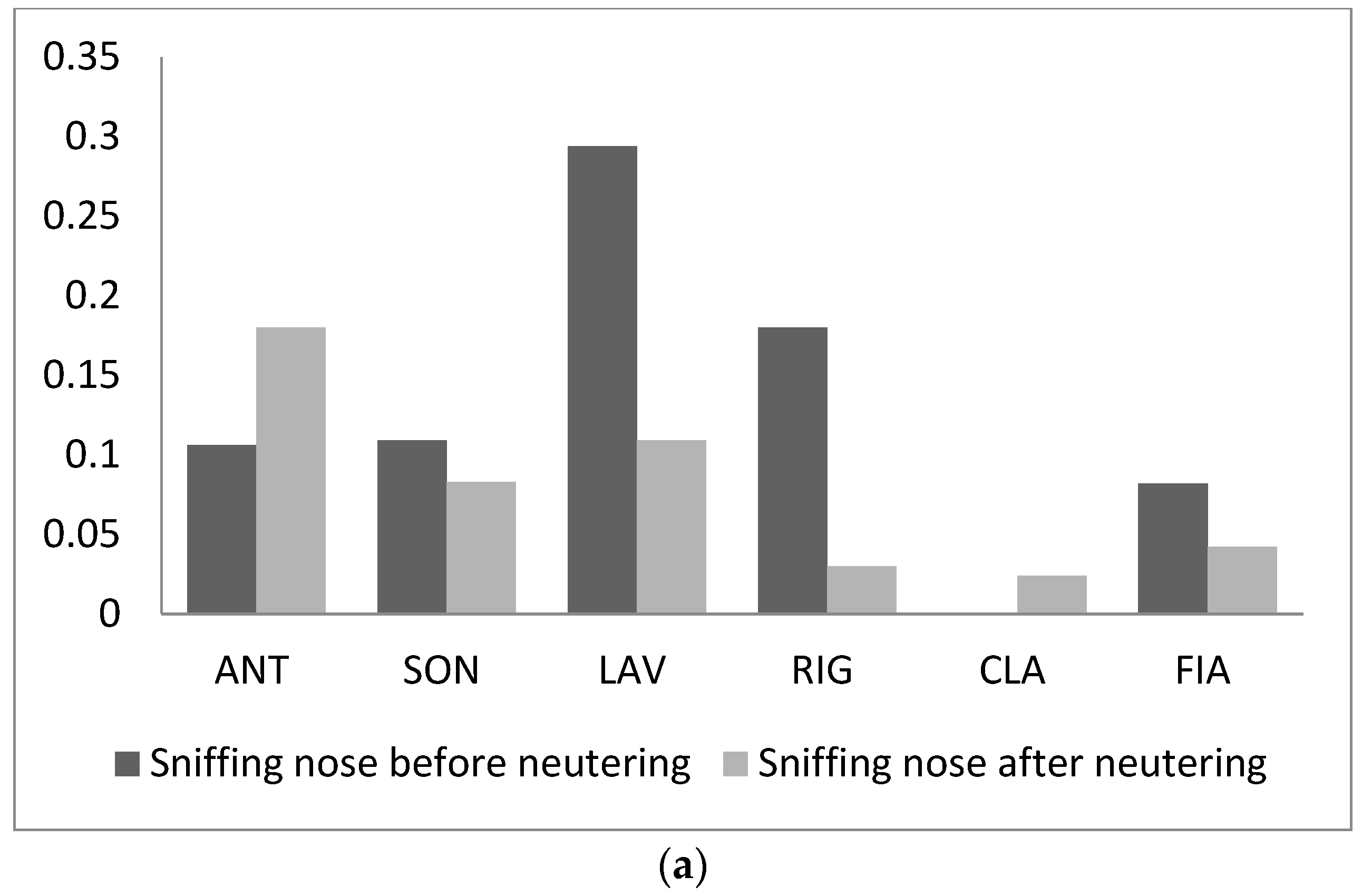
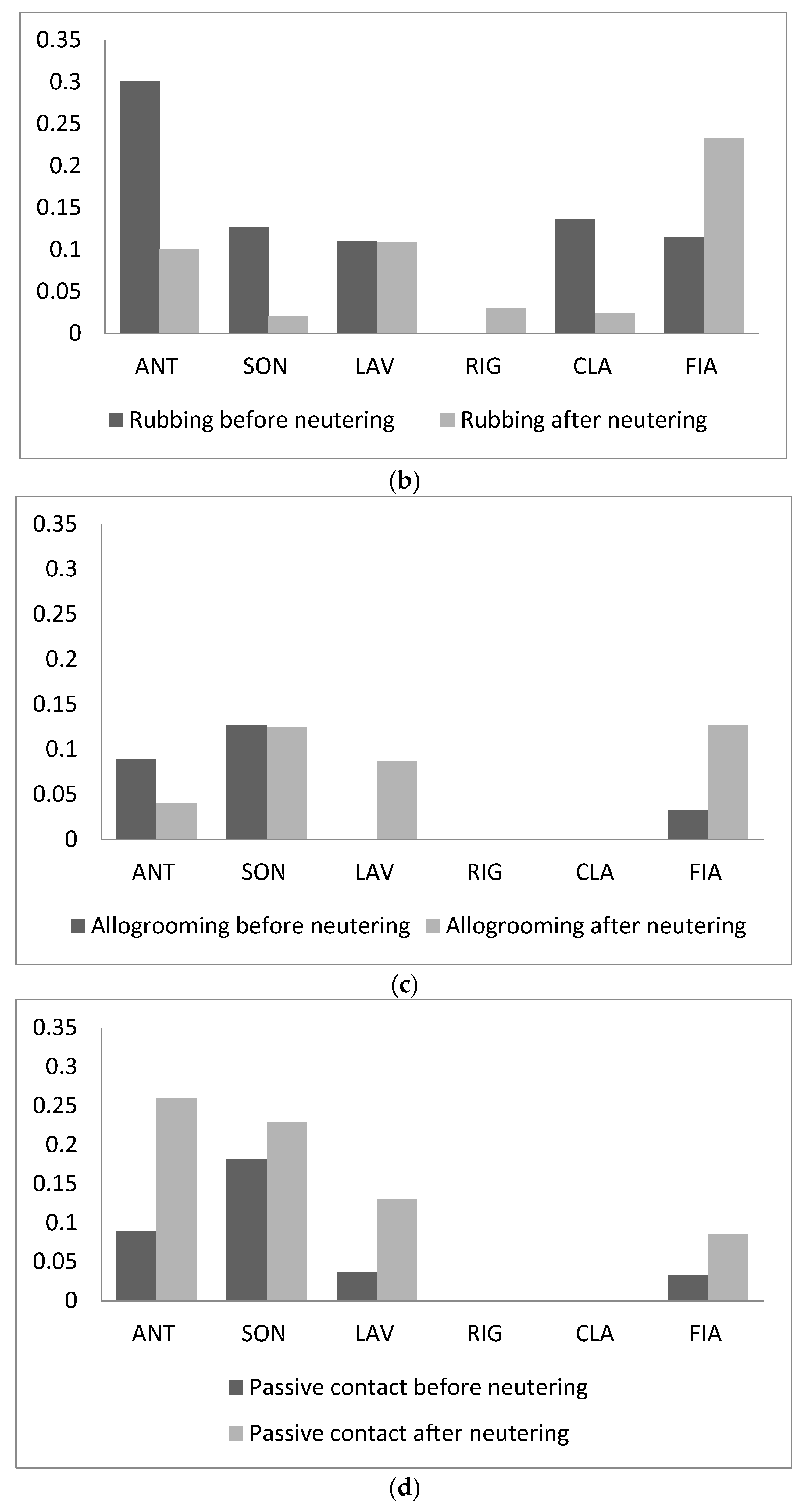
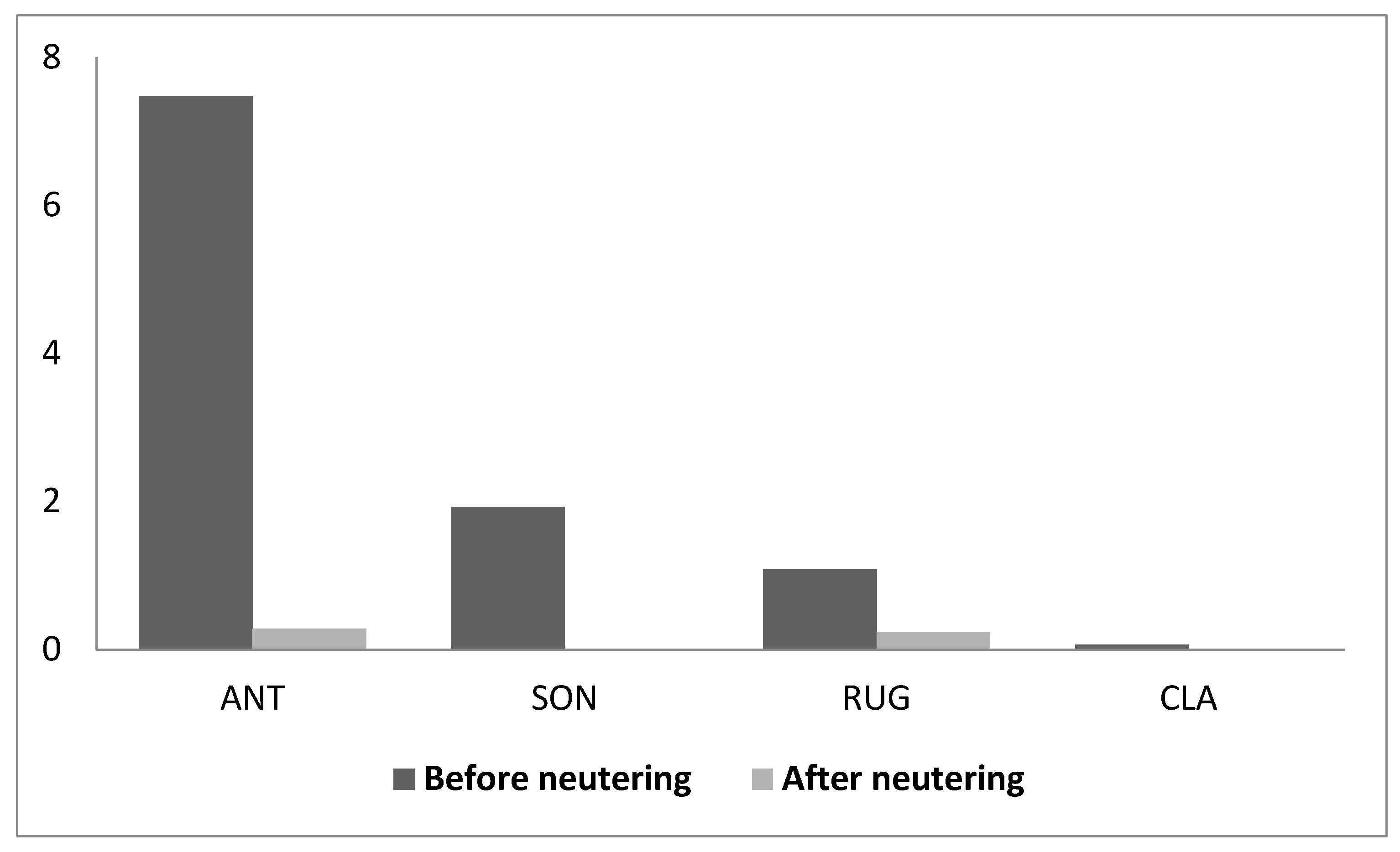
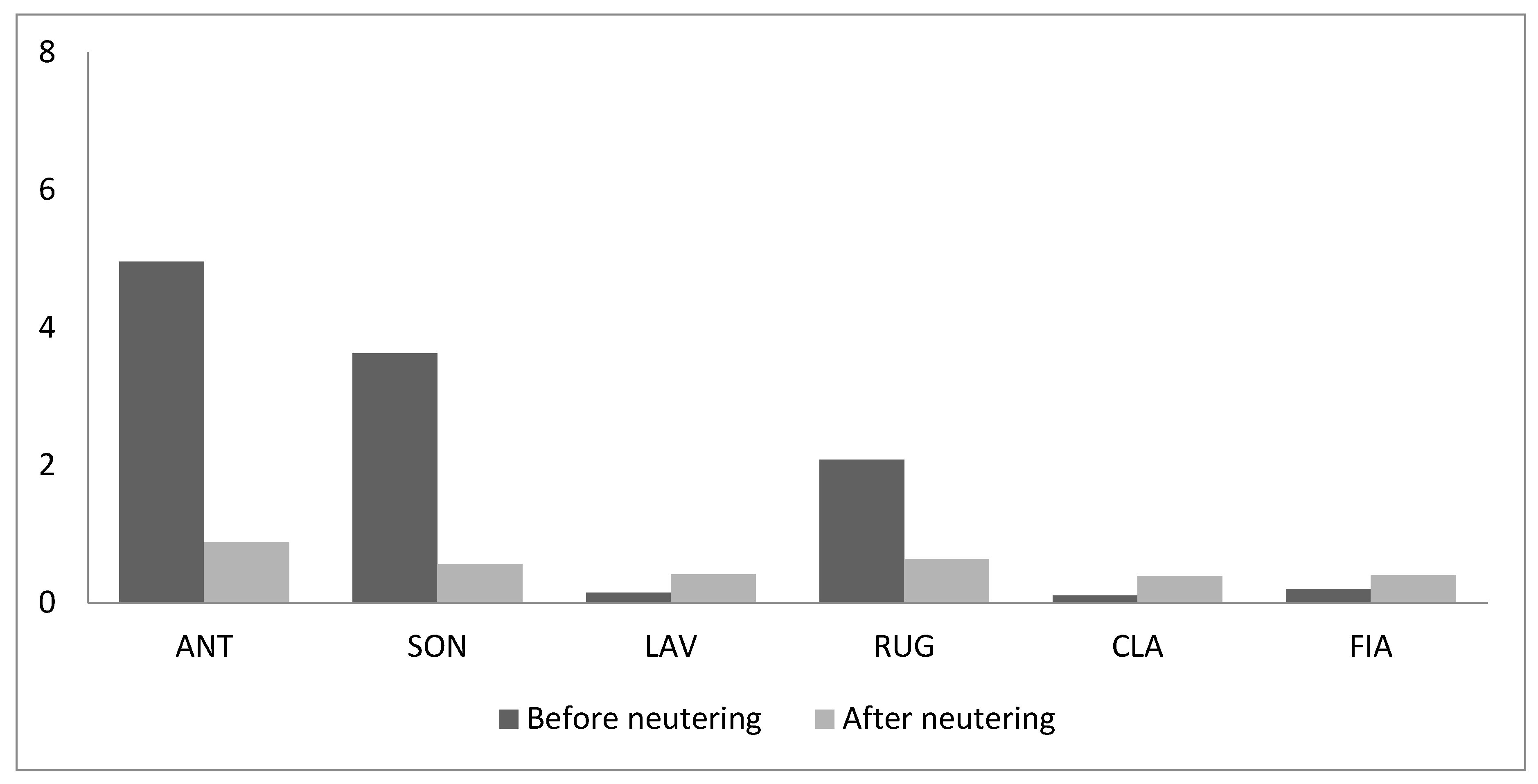
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Natoli, E.; Ziegler, N.; Dufau, A.; Pinto Teixeira, M. Unowned free-roaming domestic cats: Reflection of animal welfare and ethical aspects in animal laws in six European Countries. J. Appl. Anim. Ethics Res. 2019, 1–19. [Google Scholar] [CrossRef]
- Natoli, E.; Malandrucco, L.; Minati, L.; Verzichi, S.; Perino, R.; Longo, L.; Pontecorvo, F.; Faini, A. Evaluation of Unowned Domestic Cat Management in the Urban Environment of Rome After 30 Years of Implementation of the No-Kill Policy (National and Regional Laws). Front. Vet. Sci. 2019, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Natoli, E.; Maragliano, L.; Cariola, G.; Faini, A.; Bonanni, R.; Cafazzo, S.; Fantini, C. Management of feral domestic cats in the urban environment of Rome (Italy). Prev. Vet. Med. 2006, 77, 180–185. [Google Scholar] [CrossRef]
- Robertson, S. A review of feral cat control. J. Fel. Med. Surg. 2008, 10, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Guttilla, D.A.; Stapp, P. Effects of sterilization on movements of feral cats at a wildland—Urban interface. J. Mammal. 2010, 91, 482–489. [Google Scholar] [CrossRef]
- Scott, K.C.; Levy, J.K.; Gorman, S.P.; Newell, S.M. Body condition of feral cats and the effect of neutering. J. Appl. Anim. Welf. Sci. 2002, 5, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Natoli, E. Urban feral cats (Felis catus L.): Perspectives for a demographic control respecting the psycho-biological welfare of the species. Ann. Ist. Sup. San. 1994, 30, 223–227. [Google Scholar]
- Root Kustritz, M.V. Effects of Surgical Sterilization on Canine and Feline Health and on Society. Reprod. Domest. Anim. 2012, 47 (Suppl. S4), 214–222. [Google Scholar] [CrossRef]
- Howe, L.M. Current perspectives on the optimal age to spay/castrate dogs and cats. Vet. Med. Res. Rep. 2015, 6, 171–180. [Google Scholar] [CrossRef]
- Neville, P.F. Feral cats: Management of urban populations and pest problems by neutering. In Mammals as Pests; Putman, R.J., Ed.; Chapman and Hall: London, UK; New York, NY, USA, 1989; pp. 261–267. [Google Scholar]
- Hart, B.; Barrett, R.E. Effects of castration on fighting, roaming, and urine spraying in adult male cats. JAVMA 1973, 163, 290–292. [Google Scholar]
- Pascal, M.; Castanet, J. Methode de determination de l’age chez le chat haret des ˆıles Kergulen. La terre et la Vie 1978, 4, 529–555. [Google Scholar]
- Natoli, E. Spacing pattern of a colony of urban stray cats (Felis catus L.) in the centre of Rome. Appl. Anim. Behav. Sci. 1985, 14, 289–304. [Google Scholar] [CrossRef]
- Bonanni, R.; Cafazzo, S.; Fantini, C.; Pontier, D.; Natoli, E. Feeding-order in an urban feral domestic cat colony: Relationship to dominance rank, sex and age. Anim. Behav. 2007, 74, 1369–1379. [Google Scholar] [CrossRef]
- Altmann, J. Observational study of behaviour: Sampling methods. Behaviour 1974, 49, 227–267. [Google Scholar] [CrossRef] [PubMed]
- Hinde, R.A. Animal Behaviour: A Synthesis of Ethology and Comparative Psychology; McGraw-Hill: New York, NY, USA, 1970. [Google Scholar]
- Natoli, E.; De Vito, E. The mating system of feral cats living in a group. In The Domestic Cat: The Biology of Its Behaviour; Dennis, C.T., Patrick, P.B., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 1988; pp. 99–108. [Google Scholar]
- De Vries, H. An improved test of linearity in dominance hierarchies containing unknown or tied relationships. Anim. Behav. 1995, 50, 1375–1389. [Google Scholar] [CrossRef]
- Clutton-Brock, T.H.; Guinness, F.E.; Albon, S.D. Red Deer. In Behaviour and Ecology of Two Sexes; The University of Chicago Press: Chicago, IL, USA; Edinburgh University Press: Edinburgh, UK, 1982. [Google Scholar]
- De Vries, H. The rowwise correlation between two proximity matrices and the partial rowwise correlation. Psychometrika 1993, 58, 53–69. [Google Scholar] [CrossRef]
- Sapolsky, R.M. The influence of Social Hierarchy on Primate Health. Rev. Sci. 2005, 308, 648–652. [Google Scholar] [CrossRef]
- Keeney, A.; Jessop, D.S.; Harbuz, M.S.; Marsden, C.A.; Hogg, S.; Blackburn, R.E. Differential Effects of Acute and Chronic Social Defeat Stress on Hypothalamic-Pituitary-Adrenal Axis Function and Hippocampal Serotonin Release in Mice. J. Neuroendocrinol. 2006, 18, 330–338. [Google Scholar] [CrossRef]
- Tarttelin, M.F.; Hendriks, W.H.; Moughan, P.J. Relationship between plasma testosterone and urinary felinine in the growing kitten. Physiol. Behav. 1998, 65, 83–87. [Google Scholar] [CrossRef]
- Gunther, I.; Finkler, H.; Terkel, J. Demographic differences between urban feeding groups of neutered and sexually intact free-roaming cats following a trap-neuter-return procedure. JAVMA 2011, 238, 1134–1140. [Google Scholar] [CrossRef]
- Levy, J.K.; Gale, D.W.; Gale, L.A. Evaluation of the effect of a long-term trap-neuter-return and adoption program on a free-roaming cat population. JAVMA 2003, 222, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Natoli, E.; Say, L.; Cafazzo, S.; Bonanni, R.; Schmid, M.; Pontier, D. Bold attitude makes male urban feral domestic cats more vulnerable to Feline Immunodeficiency Virus. Neurosci. Biobehav. Rev. 2005, 29, 151–157. [Google Scholar] [CrossRef] [PubMed]

| Name | Sex | Periods of Study | |
|---|---|---|---|
| Before Neutering (Sep. 2001–Jun. 2002) | After Neutering (Oct. 2002–Jun. 2003) | ||
| Leoncino (LEO) | Male | Present, adult | Dispersed, adult |
| Antonio (ANT) *° | Male | Present, adult | Present, adult |
| Sonny (SON) *° | Male | Present, adult | Present, adult |
| Pallino (PAL) | Male | Present, adult | Died, adult |
| Pelliccione (PEL) | Male | Present, adult | Dispersed |
| Righetto (RIG) * | Male | Present, adult | Present, adult |
| Gnocco (GNO) | Male | Present, adult | Died |
| Red (RED) | Male | Present, adult | Died |
| Lavinia (LAV) *° | Female | Present, adult | Present, adult |
| Piccoletta # (PIC) * | Female | Present, adult | Present, adult |
| Claudia (CLA) *° | Female | Present, adult | Present, adult |
| Fiaschetta (FIA) *° | Female | Present, adult | Present, adult |
| Silvestrina (SIL) | Female | Present, adult | Died |
| Calicò a (CAL) | Female | Present, kitten | Died |
| Ruggine a (RUG) *° | Female | Present, kitten | Present, adult |
| Spotted a (SPO) *° | Male | Present, kitten | Present, adult |
| Nero a (NER) | Male | Present, kitten | Died |
| Tartaruga a (TAR) ° | Female | Newborn | Present, juveniles |
| Macchiola a (MAC) ° | Female | Newborn | Present, juveniles |
| Cicì b (CIC) ° | Male | Newborn | Present, juveniles |
| Stefy b (STE) ° | Male | Newborn | Present, juveniles |
| Fly b (FLY) *° | Male | Newborn | Present, juveniles |
| White b (WHI) ° | Male | Newborn | Present, juveniles |
| Nanà c (NAN) ° | Female | Newborn | Present, juveniles |
| Total | 17 | 16 | |
| ANT | LAV | SON | PIC | RIG | SPO | CLA | FIA | RUG | FLY | Tot | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ANT | 0 | ||||||||||
| LAV | 4 | 2 | |||||||||
| SON | 2 | 4 | |||||||||
| PIC | 2 | 1 | 3 | ||||||||
| RIG | 8 | 1 | 1 | 10 | |||||||
| SPO | 5 | 2 | 1 | 1 | 9 | ||||||
| CLA | 4 | 2 | 2 | 1 | 7 | ||||||
| FIA | 1 | 3 | 3 | 9 | |||||||
| RUG | 1 | 2 | 1 | 2 | 6 | ||||||
| FLY | 1 | 1 | 1 | 1 | 1 | 4 | 1 | 10 | |||
| Tot | 27 | 5 | 10 | 3 | 3 | 3 | 4 | 3 | 2 | 0 | 60 |
| ANT | LAV | CLA | FIA | SON | SPO | TAR | FLY | CIC | RUG | MAC | STE | WHI | NAN | Tot | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ANT | 0 | ||||||||||||||
| LAV | 2 | 3 | 5 | ||||||||||||
| CLA | 7 | 8 | 4 | 19 | |||||||||||
| FIA | 4 | 4 | 11 | 12 | 2 | 33 | |||||||||
| SON | 1 | 6 | 2 | 1 | 10 | ||||||||||
| SPO | 3 | 8 | 15 | 10 | 20 | 56 | |||||||||
| TAR | 2 | 5 | 68 | ||||||||||||
| FLY | 31 | 33 | 19 | 11 | 45 | 8 | 2 | 2 | 1 | 18 | |||||
| CIC | 1 | 11 | 5 | 5 | 5 | 4 | 3 | 17 | |||||||
| RUG | 3 | 11 | 14 | 5 | 16 | 10 | 8 | 1 | 48 | ||||||
| MAC | 2 | 2 | 1 | 8 | 3 | 1 | 1 | 152 | |||||||
| STE | 10 | 1 | 3 | 2 | 1 | 34 | |||||||||
| WHI | 4 | 13 | 9 | 1 | 12 | 6 | 2 | 1 | 7 | ||||||
| NAN | 2 | 3 | 1 | 3 | 1 | 4 | 2 | 3 | 2 | 2 | 23 | ||||
| Tot | 20 | 71 | 123 | 53 | 53 | 107 | 17 | 11 | 5 | 3 | 24 | 2 | 1 | 0 | 490 |
| Rank Orders in the Absence of Any Sources of Competition | |
|---|---|
| Before Neutering | After Neutering |
| LEO | Dispersed before the 2nd period |
| ANT | ANT |
| SON | LAV |
| LAV | SON |
| PIC | PIC |
| PAL | Died before the 2nd period |
| PEL | Dispersed before the 2nd period |
| RIG | RIG |
| GNO | Died before the 2nd period |
| RED | Died before the 2nd period |
| CLA | CLA |
| FIA | FIA |
| SIL | Died before the 2nd period |
| kitten | RUG |
| newborn | FLY |
| Rank Orders in the Presence of Food | |
|---|---|
| Before Neutering | After Neutering |
| ANT | ANT |
| LAV | LAV |
| LEO | Dispersed before the 2nd period |
| PIC | Adopted before the 2nd period |
| CLA | CLA |
| PEL | Dispersed before the 2nd period |
| FIA | FIA |
| SON | SON |
| RIG | Dispersed before the 2nd period |
| RED | Died before the 2nd period |
| CAL | Died before the 2nd period |
| NER | Died before the 2nd period |
| Kitten | SPO |
| Newborn | TAR |
| Newborn | FLY |
| Newborn | CIC |
| Kitten | RUG |
| Newborn | MAC |
| Newborn | STE |
| Newborn | WHI |
| Newborn | NAN |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cafazzo, S.; Bonanni, R.; Natoli, E. Neutering Effects on Social Behaviour of Urban Unowned Free-Roaming Domestic Cats. Animals 2019, 9, 1105. https://doi.org/10.3390/ani9121105
Cafazzo S, Bonanni R, Natoli E. Neutering Effects on Social Behaviour of Urban Unowned Free-Roaming Domestic Cats. Animals. 2019; 9(12):1105. https://doi.org/10.3390/ani9121105
Chicago/Turabian StyleCafazzo, Simona, Roberto Bonanni, and Eugenia Natoli. 2019. "Neutering Effects on Social Behaviour of Urban Unowned Free-Roaming Domestic Cats" Animals 9, no. 12: 1105. https://doi.org/10.3390/ani9121105
APA StyleCafazzo, S., Bonanni, R., & Natoli, E. (2019). Neutering Effects on Social Behaviour of Urban Unowned Free-Roaming Domestic Cats. Animals, 9(12), 1105. https://doi.org/10.3390/ani9121105




