Optimal Salinity for Head-Starting Northern River Terrapins (Batagur baska Gray, 1831)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Survival, Growth Performance, and Feed Utilization
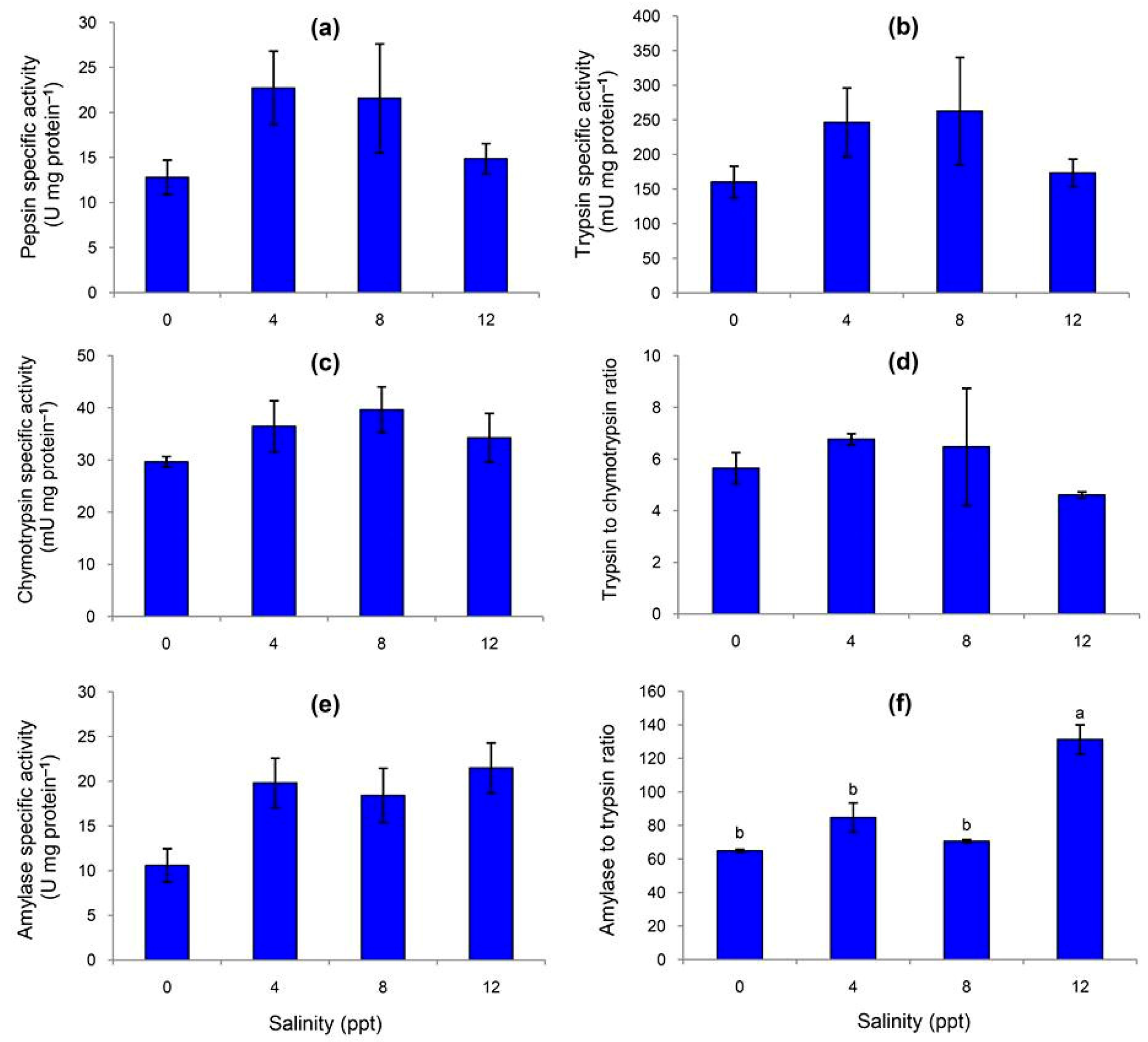
3.2. Specific Activities of Fecal Digestive Enzymes
3.3. Thermal Properties of Feces
3.4. Elemental Composition in Carapace
3.5. Hematological Parameters
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moll, E.O.; Platt, K.; Platt, S.G.; Praschag, P.; van Dijk, P.P. Batagur baska (Gray, 1831)–Northern river terrapin. In Conservation Biology of Freshwater Turtles: A Compilation Project of the IUCN/SSC Tortoise and Freshwater Turtle Specialist Group; Rhodin, A.G.J., Pritchard, P.C.H., Eds.; Chelonian Research Foundation: Lunenburg, MA, USA, 2009; pp. 037.1–037.10. [Google Scholar]
- Chutirode, Y.; Ninwat, S.; Petchrit, N.; Jualaong, S.; Khwanmaung, S.; Kanghae, H.; Thongprajukaew, K. Optimal Tank Color for Head-Starting Mangrove Terrapin (Batagur baska Gray, 1831). In Proceedings of the 6th Marine Science Conference, Chonburi, Thailand, 18–20 June 2018. [Google Scholar]
- Platt, M.; Thorbjarnarson, J.; Thirakhupt, K. An overview of the current population and conservation status of the critically endangered river terrapin, Batagur baska (Gray, 1831) in Myanmar, Thailand and Malaysia. Nat. Hist. J. Chulalongkorn Univ. 2007, 7, 51–65. [Google Scholar]
- Ernst, C.H.; Altenburg, R.G.M.; Barbour, R.W. Turtles of the World; Expert Center for Taxonomic Identification: Amsterdam, The Netherlands, 2000; pp. 203–209. [Google Scholar]
- Davenport, J.; Wong, T.M. Observations on the water economy of the estuarine turtles Batagur baska (Gray) and Callagur borneoensis (Schlegel and Muller). Comp. Biochem. Physiol. 1986, 84, 703–707. [Google Scholar] [CrossRef]
- Rasmussen, A.R.; Murphy, J.C.; Ompi, M.; Gibbons, J.W.; Uetz, P. Marine reptiles. PLoS ONE 2011, 6, e27373. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.S.K.; Tisen, O.B. Freshwater turtle and tortoise utilization and conservation status in Malaysia. Chelonian Res. Monogr. 2000, 2, 120–128. [Google Scholar]
- Dunson, W.A.; Moll, E.O. Osmoregulation in sea water of hatchling emydid turtles, Callagur borneoensis, from a Malaysian sea beach. J. Herpetol. 1980, 14, 31–36. [Google Scholar] [CrossRef]
- Davenport, J.; Ward, J.F. The effects of salinity and temperature on appetite in the diamondback terrapin Malaclemys terrapin (Latreille). Herpetol. J. 1993, 3, 95–98. [Google Scholar]
- Bower, D.S.; Scheltinga, D.M.; Clulow, S.; Clulow, J.; Franklin, C.E.; Georges, A. Salinity tolerances of two Australian freshwater turtles, Chelodina expansa and Emydura macquarii (Testudinata: Chelidae). Conserv. Physiol. 2016, 4, 1–9. [Google Scholar] [CrossRef]
- Hamed, S.S.; Jiddawi, N.S.; Poj, B. Effect of salinity levels on growth, feed utilization, body composition and digestive enzymes activities of juvenile silver pompano Trachinotus blochii. Int. J. Fish. Aquat. Stud. 2016, 4, 279–283. [Google Scholar]
- Ke, Z.; Ling, H.M.; Tao, S.H.; Qin, C.X.; Meng, J.M.; Li, L.D. Effects of salinity on Na+/K+-ATPase and digestive enzyme activities of turtle Trachemys scripta elegans. Fish. Sci. 2014, 33, 520–524. [Google Scholar]
- Sutthinon, P.; Thongprajukaew, K.; Saekhow, S.; Ketmanee, R. Juvenile hybrid grouper (Epinephelus coioides × E. lanceolatus) are euryhaline and can grow in a wide range of salinities. Aquac. Int. 2015, 23, 671–682. [Google Scholar] [CrossRef]
- Kanghae, H.; Thongprajukaew, K.; Phromkunthong, W.; Plangsri, S.; Jatupornpitukchat, S.; Kittiwattanawong, K. Pre-soaking of the feed pellets: A trick for successful feed utilization in juvenile green turtles (Chelonia mydas Linnaeus, 1758). J. Anim. Physiol. Anim. Nutr. 2017, 101, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Kanghae, H.; Thongprajukaew, K.; Yeetam, P.; Jarit-ngam, T.; Hwan-air, W.; Rueangjeen, S.; Kittiwattanawong, K. Optimal feeding frequency of captive head-started green turtles (Chelonia mydas). J. Anim. Physiol. Anim. Nutr. 2017, 101, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Thongprajukaew, K.; Kanghae, H.; Kittiwattanawong, K. Faecal characteristics as markers of Chelonia mydas feeding. ScienceAsia 2016, 42, 237–246. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosenbrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Worthington, V. Worthington Enzyme Manual: Enzymes and Related Biochemicals; Worthington Chemical: Lakewood, NJ, USA, 1993; p. 399. [Google Scholar]
- Rungruangsak-Torrissen, K.; Moss, R.; Andresen, L.H.; Berg, A.; Waagbo, R. Different expressions of trypsin and chymotrypsin in relation to growth in Atlantic salmon (Salmo salar L.). Fish. Physiol. Biochem. 2006, 32, 7–23. [Google Scholar] [CrossRef]
- Bernfeld, P. Enzymes of starch degradation and synthesis. Adv. Enzymol. 1951, 12, 379–428. [Google Scholar]
- Blaxhall, P.C.; Daisley, K.W. Routine haematological methods for use with fish blood. J. Fish. Biol. 1973, 5, 771–781. [Google Scholar] [CrossRef]
- Larsen, H.N.; Snieszko, S.F. Comparison of various methods of determination of haemoglobin in trout blood. Prog. Fish. Cult. 1961, 23, 8–17. [Google Scholar] [CrossRef]
- Dacie, J.V.; Lewis, S.M. Practical Haematology. In Practical Haematology, 9th ed.; Lewis, S.M., Bain, B.J., Bates, I., Eds.; Churchill Livingstone: London, UK, 2001; pp. 444–451. [Google Scholar]
- Rowe, C.L. Maximum standard metabolic rate corresponds with the salinity of maximum growth in hatchlings of the estuarine northern diamondback terrapin (Malaclemys terrapin terrapin): Implications for habitat conservation. Acta Oecol. 2018, 86, 79–83. [Google Scholar] [CrossRef]
- Dunson, W.A. Effect of water salinity and food salt content on growth and sodium efflux of hatchling diamondback terrapins (Malaclemys). Physiol. Zool. 1985, 58, 736–747. [Google Scholar] [CrossRef]
- Holliday, D.K.; Elskus, A.A.; Roosenburg, W.M. Impacts of multiple stressors on growth and metabolic rate of Malaclemys terrapin. Environ. Toxicol. Chem. 2009, 28, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Agha, M.; Ennen, J.R.; Bower, D.S.; Nowakowski, A.J.; Sweat, S.C.; Todd, B.D. Salinity tolerances and use of saline environments by freshwater turtles: Implications of sea level rise. Biol. Rev. 2018, 93, 1634–1648. [Google Scholar] [CrossRef] [PubMed]
- Moutou, K.A.; Panagiotaki, P.; Mamuris, Z. Effects of salinity on digestive protease activity in the euryhaline sparid Sparus aurata L.: A preliminary study. Aquac. Res. 2004, 35, 912–914. [Google Scholar]
- Hofer, R.; Schiemer, F. Proteolytic activity in the digestive tract of several species of fish with different feeding habits. Oecologia 1981, 48, 342–345. [Google Scholar] [CrossRef]
- Thongprajukaew, K.; Kovitvadhi, U.; Kovitvadhi, S.; Somsueb, P.; Rungruangsak-Torrissen, K. Effects of different modified diets on growth, digestive enzyme activities and muscle compositions in juvenile Siamese fighting fish (Betta splendens Regan, 1910). Aquaculture 2011, 322–323, 1–9. [Google Scholar] [CrossRef]
- Shu, C.H.; Zhang, K.; Hong, M.L.; Xie, D.; Liu, Q.C.; Shi, H.T. Effect of salinity stress on Trachemys scripta elegans growth and blood biochemical ingredient. Sichuan J. Zool. 2012, 31, 912–916. [Google Scholar]
- Hong, M.; Zhang, K.; Shu, C.; Xie, D.; Shi, H. Effect of salinity on the survival, ions and urea modulation in red-eared slider (Trachemys scripta elegans). Asian Herpetol. Res. 2014, 5, 128–136. [Google Scholar]
- Kanghae, H.; Thongprajukaew, K.; Jatupornpitukchat, S.; Kittiwattanawong, K. Optimal rearing density for head-starting green turtles (Chelonia mydas Linnaeus, 1758). Zoo Biol. 2016, 35, 454–461. [Google Scholar] [CrossRef]
- Bao, J.; Corke, H. Pasting properties of gamma-irradiated rice starches as affected by pH. J. Agric. Food Chem. 2002, 50, 336–341. [Google Scholar] [CrossRef]
- Espinoza, E.O.; Baker, B.W. The analysis of sea turtle and bovid keratin artefacts using drift spectroscopy and discriminant analysis. Archaeometry 2007, 49, 685–698. [Google Scholar] [CrossRef]
- Rhee, H.; Horstemeyer, M.F.; Hwang, Y.; Lim, H.; El Kadiri, H.; Trim, W. A study on the structure and mechanical behavior of the Terrapene carolina carapace: A pathway to design bio-inspired synthetic composites. Mat. Sci. Eng. 2009, 29, 2333–2339. [Google Scholar] [CrossRef]
- Infante, C.; Ponce, M.; Asensio, E.; Zerolo, R.; Manchado, M. Molecular characterization of a novel type II keratin gene (sseKer3) in the Senegalese sole (Solea senegalensis): Differential expression of keratin genes by salinity. Comp. Biochem. Physiol. 2011, 160, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Magnadottir, B. Immunological control of fish diseases. Mar. Biotechnol. 2010, 12, 361–379. [Google Scholar] [CrossRef] [PubMed]
- Birrer, S.C.; Reusch, T.B.; Roth, O. Salinity change impairs pipefish immune defense. Fish. Shellfish Immunol. 2012, 33, 1238–1248. [Google Scholar] [CrossRef]
- Gilles-Baillien, M. Urea and osmoregulation in the diamondback terrapin Malaclemys centrata centrata (Latreille). J. Exp. Biol. 1970, 52, 691–697. [Google Scholar]
- Lee, S.M.L.; Wong, W.P.; Hiong, K.C.; Loong, A.I.M.; Chew, S.F.; Ip, Y.K. Nitrogen metabolism and excretion in the aquatic Chinese soft-shelled turtle, Pelodiscus sinensis, exposed to a progressive increase in ambient salinity. J. Exp. Zool. 2006, 305, 995–1009. [Google Scholar] [CrossRef]
- Liu, S.; Jiang, X.; Hu, X.; Gong, J.; Hwang, H.; Mai, K. Effects of temperature on non-specific immune parameters in two scallop species: Argopecten irradians (Lamarck 1819) and Chlamys farreri (Jones & Preston 1904). Aquac. Res. 2004, 35, 678–682. [Google Scholar]
- Fang, Z.H.; Tian, X.L.; Dong, S.L.; Dai, C.; Wang, G.D. Effects of salinity on the activity of non-specific immune enzymes of juvenile tongue sole cultured in various salinities. J. Ocean. Univ. China 2014, 44, 46–53. [Google Scholar]
- Sui, Y.; Huang, X.; Kong, H.; Lu, W.; Wang, Y. Physiological responses to salinity increase in blood parrotfish (Cichlasoma synspilum♀ × Cichlasoma citrinellum♂). SpringerPlus 2016, 5, 1246. [Google Scholar] [CrossRef]
- Feng, J.; Xu, L.W.; Lin, H.Z.; Guo, Z.X.; Guo, G.X. Effects of salinity on growth and several immune parameters of juvenile cobia, Rachycentron canadum. J. Fish. Sci. China 2007, 14, 120–125. [Google Scholar]
- Fazio, E.; Medica, P.; Bruschetta, G.; Ferlazzo, A. Do handling and transport stress adrenocortical response in the tortoises (Testudo hermanni)? ISRN Vet. Sci. 2014. [CrossRef] [PubMed]
- Zhou, X.; Xie, M.; Niu, C.; Sun, R. The effects of dietary vitamin C on growth, liver vitamin C and serum cortisol in stressed and unstressed juvenile soft-shelled turtles (Pelodiscus sinensis). Comp. Biochem. Physiol. 2003, 135, 263–270. [Google Scholar] [CrossRef]
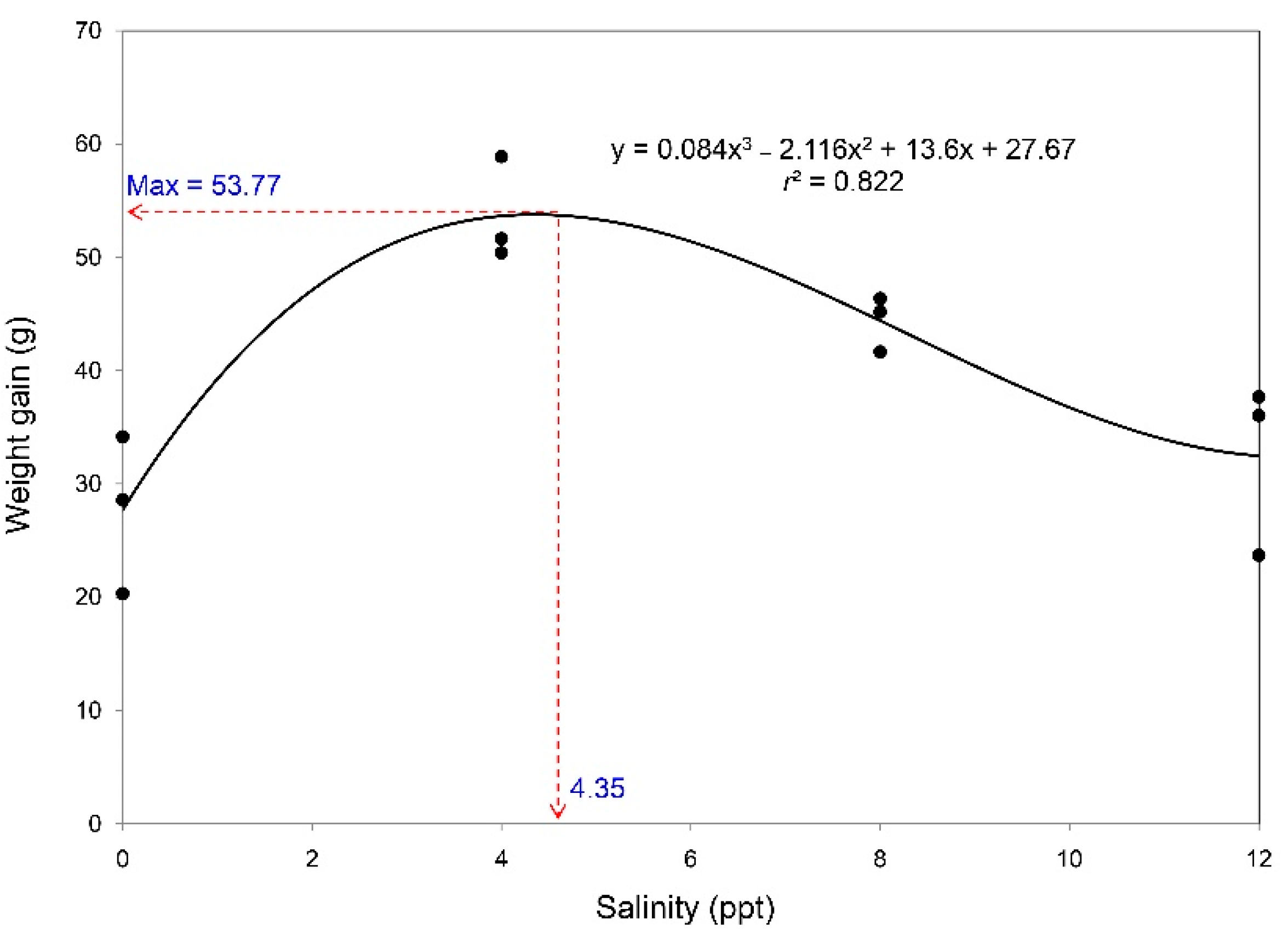
| Parameter | Salinity (ppt) | p-Value | |||
|---|---|---|---|---|---|
| 0 | 4 | 8 | 12 | ||
| Survival (%) | 100 | 100 | 100 | 100 | – |
| Initial body weight (g) | 54.03 ± 0.48 | 54.73 ± 0.26 | 55.20 ± 0.12 | 54.59 ± 0.22 | 0.121 |
| Final body weight (g) | 81.71 ± 3.71b | 108.37 ± 2.70a | 99.62 ± 1.51a | 87.05 ± 4.30b | 0.002 |
| WG (g) | 27.68 ± 4.02b | 53.64 ± 2.65a | 44.40 ± 1.42a | 32.46 ± 4.41b | 0.002 |
| SCL (cm) | 8.02 ± 0.21 | 8.67 ± 0.15 | 8.27 ± 0.08 | 7.99 ± 0.20 | 0.073 |
| SCW (cm) | 8.17 ± 0.17 | 8.66 ± 0.12 | 8.34 ± 0.07 | 8.16 ± 0.22 | 0.165 |
| SCL/SCW | 0.98 ± 0.01 | 1.00 ± 0.01 | 0.99 ± 0.01 | 0.98 ± 0.01 | 0.062 |
| BCI (kg cm−3) | 1.59 ± 0.06 | 1.66 ± 0.05 | 1.76 ± 0.02 | 1.71 ± 0.05 | 0.135 |
| FR (% BW day−1) | 0.67 ± 0.02a | 0.69 ± 0.01a | 0.59 ± 0.01b | 0.54 ± 0.03b | 0.002 |
| FCR (g feed g gain−1) | 0.90 ± 0.10a | 0.55 ± 0.01b | 0.54 ± 0.01b | 0.64 ± 0.04b | 0.004 |
| PER (g gain g protein−1) | 3.30 ± 0.33b | 5.24 ± 0.13a | 5.32 ± 0.03a | 4.58 ± 0.26a | 0.001 |
| Thermal Parameter | Salinity (ppt) | |||
|---|---|---|---|---|
| 0 | 4 | 8 | 12 | |
| Peak 1 | ||||
| To (°C) | - | 43.67 | 44.29 | 47.97 |
| Tp (°C) | - | 64.61 | 67.45 | 63.44 |
| Tc (°C) | - | 81.17 | 102.25 | 79.64 |
| Tc–To (°C) | - | 37.50 | 57.96 | 31.67 |
| ΔH1 (J g−1) | - | 18.57 | 27.06 | 21.86 |
| Peak 2 | ||||
| To (°C) | 88.11 | 91.63 | 91.82 | 91.56 |
| Tp (°C) | 95.61 | 99.45 | 123.16 | 99.53 |
| Tc (°C) | 101.99 | 105.64 | 133.19 | 107.93 |
| Tc – To (°C) | 13.88 | 14.01 | 41.37 | 16.37 |
| ΔH2 (J g−1) | 2.81 | 2.91 | 3.01 | 2.07 |
| Peak 3 | ||||
| To (°C) | 177.23 | - | - | - |
| Tp (°C) | 186.10 | - | - | - |
| Tc (°C) | 191.06 | - | - | - |
| Tc – To (°C) | 13.83 | - | - | - |
| ΔH3 (J g−1) | 4.93 | - | - | - |
| Peak 4 | ||||
| To (°C) | 353.13 | 323.38 | 315.15 | 334.68 |
| Tp (°C) | 368.33 | 351.39 | 349.00 | 352.72 |
| Tc (°C) | 388.50 | 378.59 | 375.95 | 373.80 |
| Tc – To (°C) | 35.37 | 55.21 | 60.80 | 39.12 |
| ΔH4 (J g−1) | 23.32 | 86.11 | 28.21 | 20.20 |
| ΣΔH (J g−1) | 31.06 | 108.19 | 58.29 | 44.13 |
| Element (% of dry weight) | Salinity (ppt) | p-Value | |||
|---|---|---|---|---|---|
| 0 | 4 | 8 | 12 | ||
| C | 46.77 ± 0.80 | 48.38 ± 0.44 | 52.00 ± 1.99 | 47.16 ± 0.99 | 0.052 |
| O | 32.91 ± 0.58a | 29.88 ± 0.48ab | 27.63 ± 1.23b | 30.56 ± 1.13ab | 0.023 |
| N | 19.50 ± 0.28 | 21.19 ± 0.39 | 19.48 ± 1.35 | 21.91 ± 0.24 | 0.109 |
| S | 0.20 ± 0.03b | 0.30 ± 0.01b | 0.67 ± 0.17a | 0.20 ± 0.02b | 0.041 |
| Al | 0.23 ± 0.02 | 0.17 ± 0.05 | 0.13 ± 0.02 | 0.10 ± 0.02 | 0.085 |
| P | 0.10 ± 0.01 | 0.10 ± 0.01 | 0.10 ± 0.01 | 0.10 ± 0.01 | 1.000 |
| Na | 0.20 ± 0.06 | 0.10 ± 0.01 | 0.10 ± 0.02 | 0.17 ± 0.02 | 0.160 |
| Cl | 0.12 ± 0.02 | 0.12 ± 0.01 | 0.13 ± 0.03 | 0.10 ± 0.01 | 0.745 |
| Mg | 0.10 ± 0.01 | 0.10 ± 0.01 | 0.10 ± 0.01 | 0.10 ± 0.01 | 1.000 |
| Hematological Parameter | Salinity (ppt) | p-Value | |||
|---|---|---|---|---|---|
| 0 | 4 | 8 | 12 | ||
| RBC (×106 cells μL−1) | 0.42 ± 0.04 | 0.43 ± 0.03 | 0.39 ± 0.04 | 0.37 ± 0.02 | 0.500 |
| Hematocrit (%) | 35.33 ± 1.76 | 38.00 ± 1.15 | 38.33 ± 2.33 | 36.33 ± 2.19 | 0.665 |
| MCV (fL) | 887.63 ± 59.75 | 879.68 ± 85.44 | 995.19 ± 40.32 | 995.55 ± 15.98 | 0.424 |
| WBC (×103 cells μL−1) | 5.28 ± 0.11 | 5.83 ± 0.32 | 5.43 ± 0.62 | 3.36 ± 0.50 | 0.050 |
| Lymphocyte (%) | 27.33 ± 8.11b | 31.00 ± 4.36b | 56.00 ± 3.00a | 57.67 ± 5.24a | 0.015 |
| Azurophil (%) | 45.00 ± 9.00 | 53.00 ± 3.00 | 31.00 ± 2.00 | 35.67 ± 4.10 | 0.054 |
| Heterophil (%) | 4.00 ± 0.00b | 7.00 ± 1.00a | 1.50 ± 0.50c | 2.00 ± 1.00bc | 0.007 |
| Monocyte (%) | 15.33 ± 1.76a | 9.33 ± 1.76ab | 13.33 ± 2.33a | 3.50 ± 0.50b | 0.020 |
| Thrombocyte (×103) | 2.06 ± 0.20a | 1.45 ± 0.34ab | 0.94 ± 0.06bc | 0.56 ± 0.06c | 0.023 |
| Plasma protein (g %) | 2.40 ± 0.12 | 2.80 ± 0.12 | 2.53 ± 0.33 | 2.60 ± 0.31 | 0.705 |
| Total protein (g L−1) | 17.63 ± 1.14 | 15.80 ± 1.25 | 13.43 ± 1.49 | 13.80 ± 0.70 | 0.115 |
| BUN (mg dL−1) | 22.87 ± 1.76c | 32.94 ± 1.51c | 57.85 ± 0.71b | 159.03 ± 8.93a | <0.001 |
| ALP (U L−1) | 110.30 ± 0.90a | 88.63 ± 0.52b | 72.95 ± 3.95c | 61.30 ± 4.60d | <0.001 |
| AST (U L−1) | 118.23 ± 5.69 | 110.45 ± 4.55 | 114.00 ± 14.90 | 118.60 ± 14.50 | 0.927 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jualaong, S.; Songnui, A.; Thongprajukaew, K.; Ninwat, S.; Khwanmaung, S.; Hahor, W.; Khunsaeng, P.; Kanghae, H. Optimal Salinity for Head-Starting Northern River Terrapins (Batagur baska Gray, 1831). Animals 2019, 9, 855. https://doi.org/10.3390/ani9110855
Jualaong S, Songnui A, Thongprajukaew K, Ninwat S, Khwanmaung S, Hahor W, Khunsaeng P, Kanghae H. Optimal Salinity for Head-Starting Northern River Terrapins (Batagur baska Gray, 1831). Animals. 2019; 9(11):855. https://doi.org/10.3390/ani9110855
Chicago/Turabian StyleJualaong, Suthep, Anida Songnui, Karun Thongprajukaew, Santi Ninwat, Suwandee Khwanmaung, Waraporn Hahor, Pairat Khunsaeng, and Hirun Kanghae. 2019. "Optimal Salinity for Head-Starting Northern River Terrapins (Batagur baska Gray, 1831)" Animals 9, no. 11: 855. https://doi.org/10.3390/ani9110855
APA StyleJualaong, S., Songnui, A., Thongprajukaew, K., Ninwat, S., Khwanmaung, S., Hahor, W., Khunsaeng, P., & Kanghae, H. (2019). Optimal Salinity for Head-Starting Northern River Terrapins (Batagur baska Gray, 1831). Animals, 9(11), 855. https://doi.org/10.3390/ani9110855





