Effect of Two Transport Options on the Welfare of Two Genetic Lines of Organic Free Range Pullets in Switzerland
Simple Summary
Abstract
1. Introduction
2. Animals, Materials and Methods
2.1. Experimental Design
2.2. Corticosterone Monitoring
2.3. Hen-Human Relationship: Touch Test
2.4. Body Condition
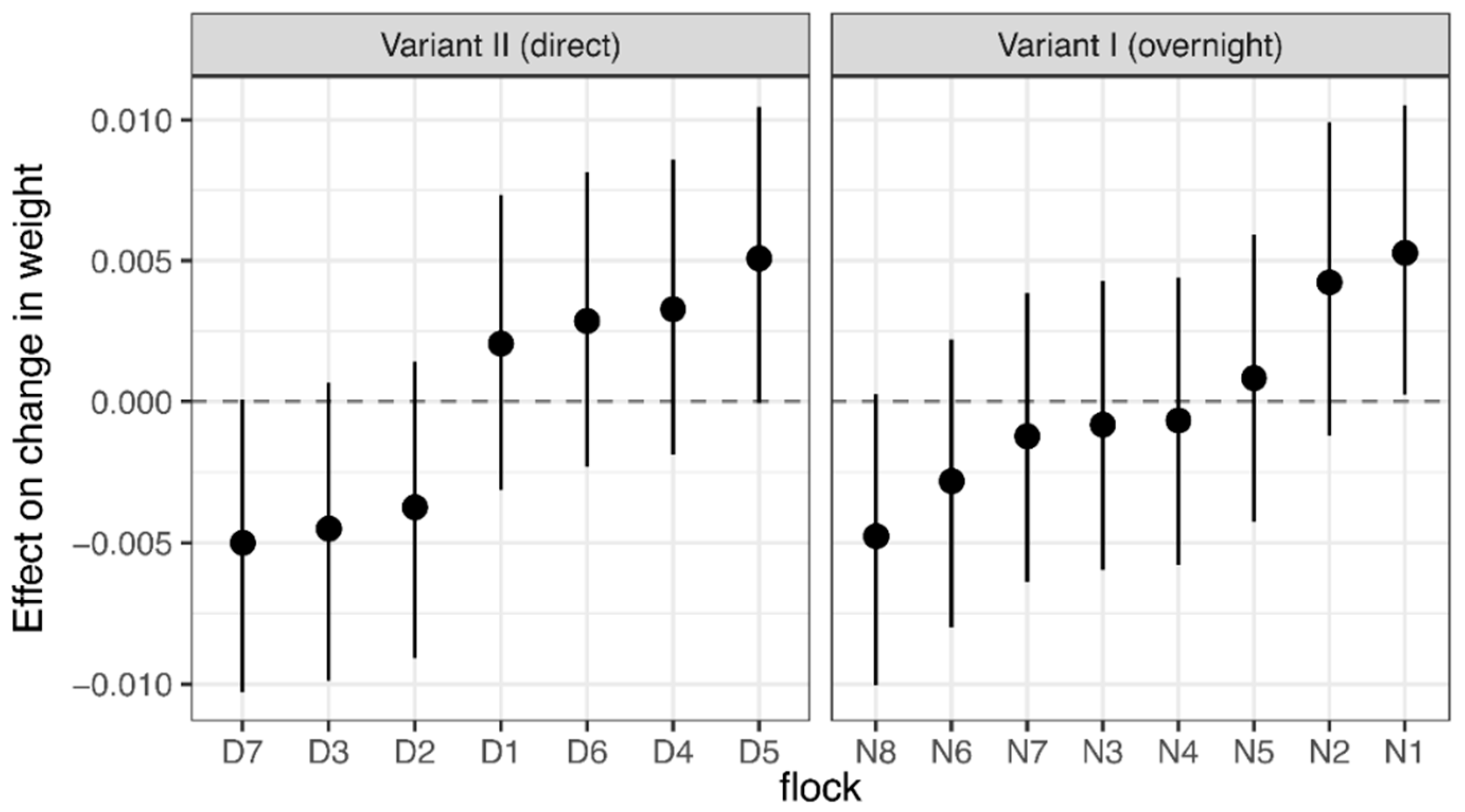
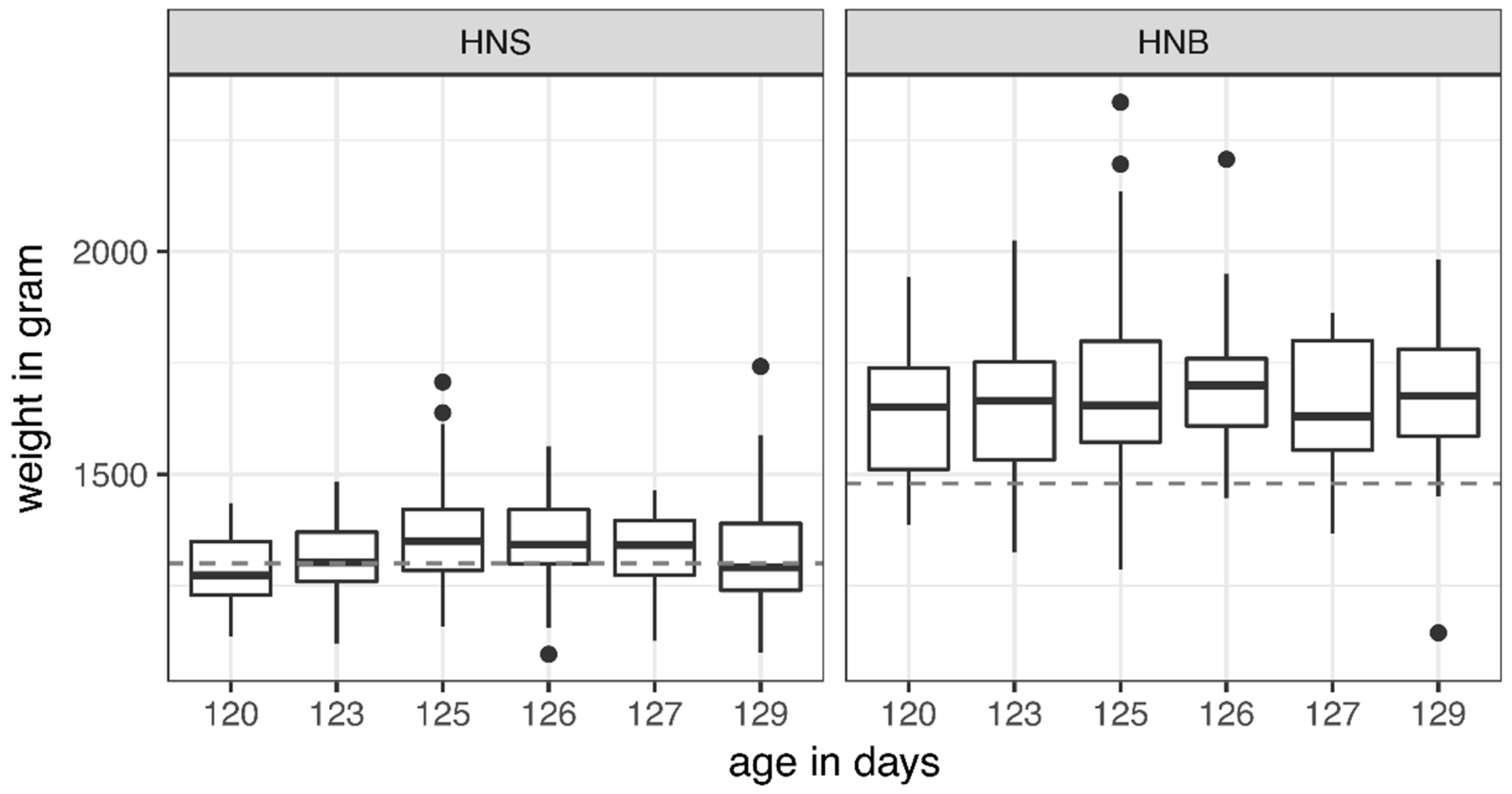
2.5. Live Weight
2.6. Statistical Analysis
3. Results
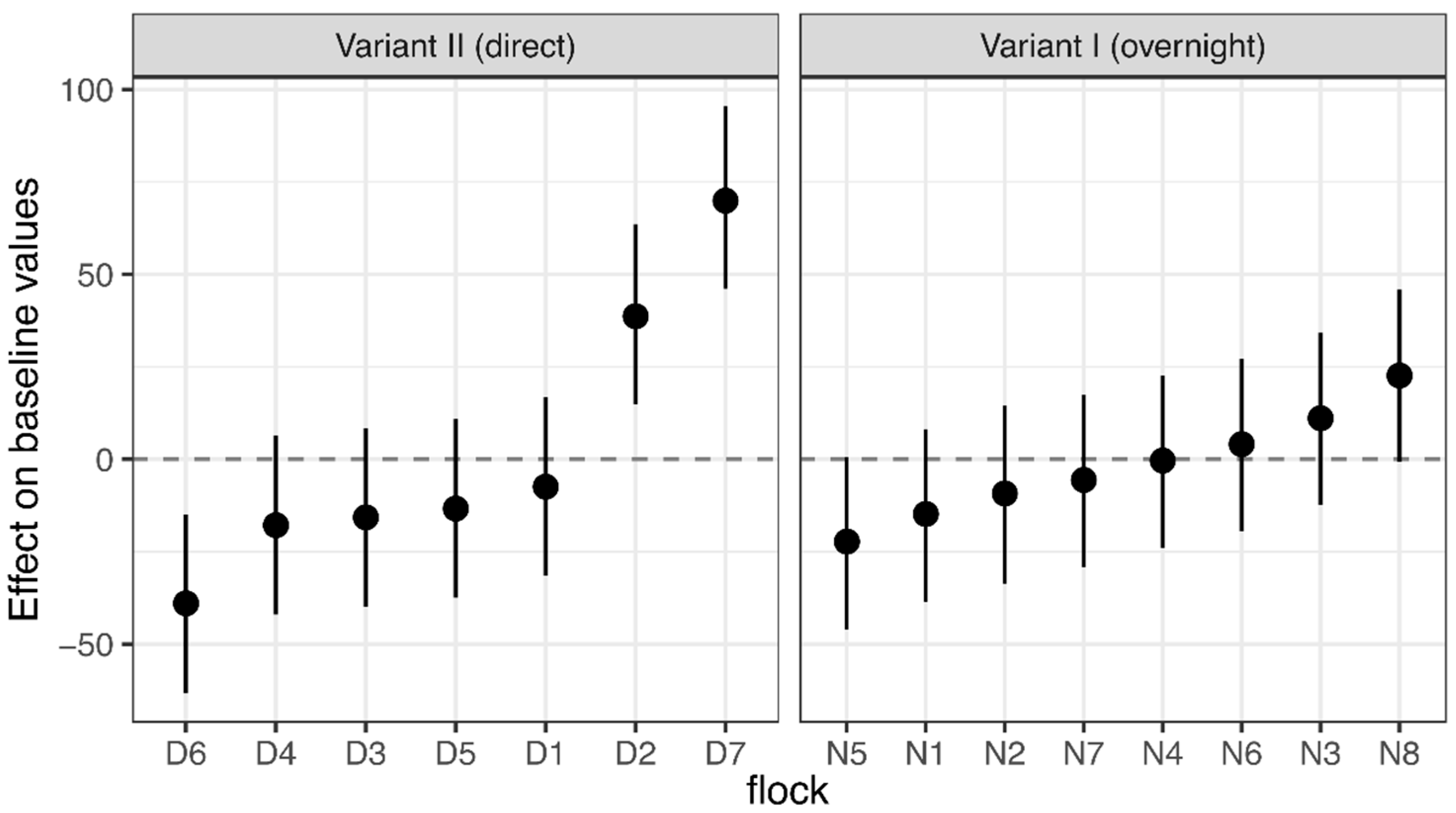
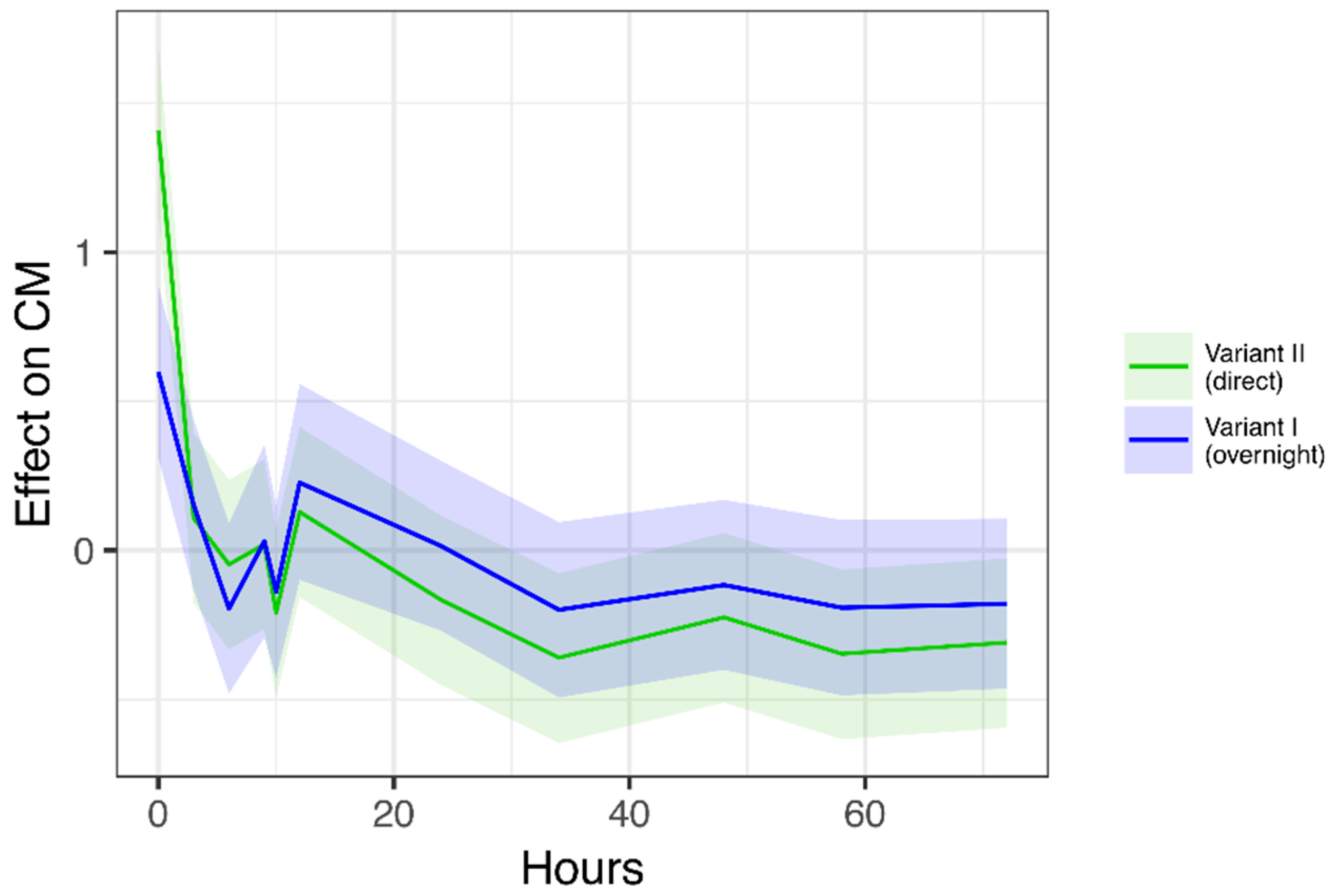
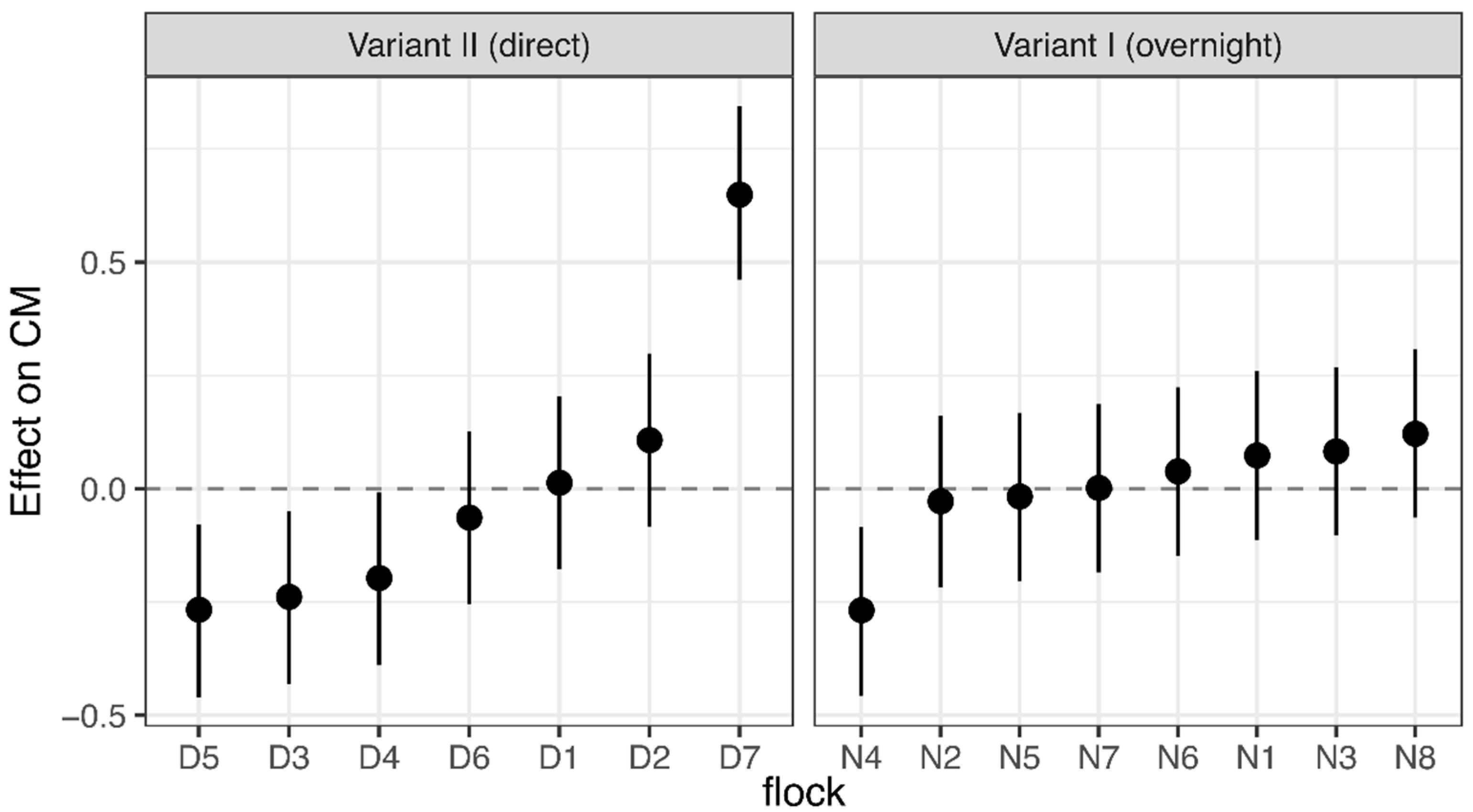
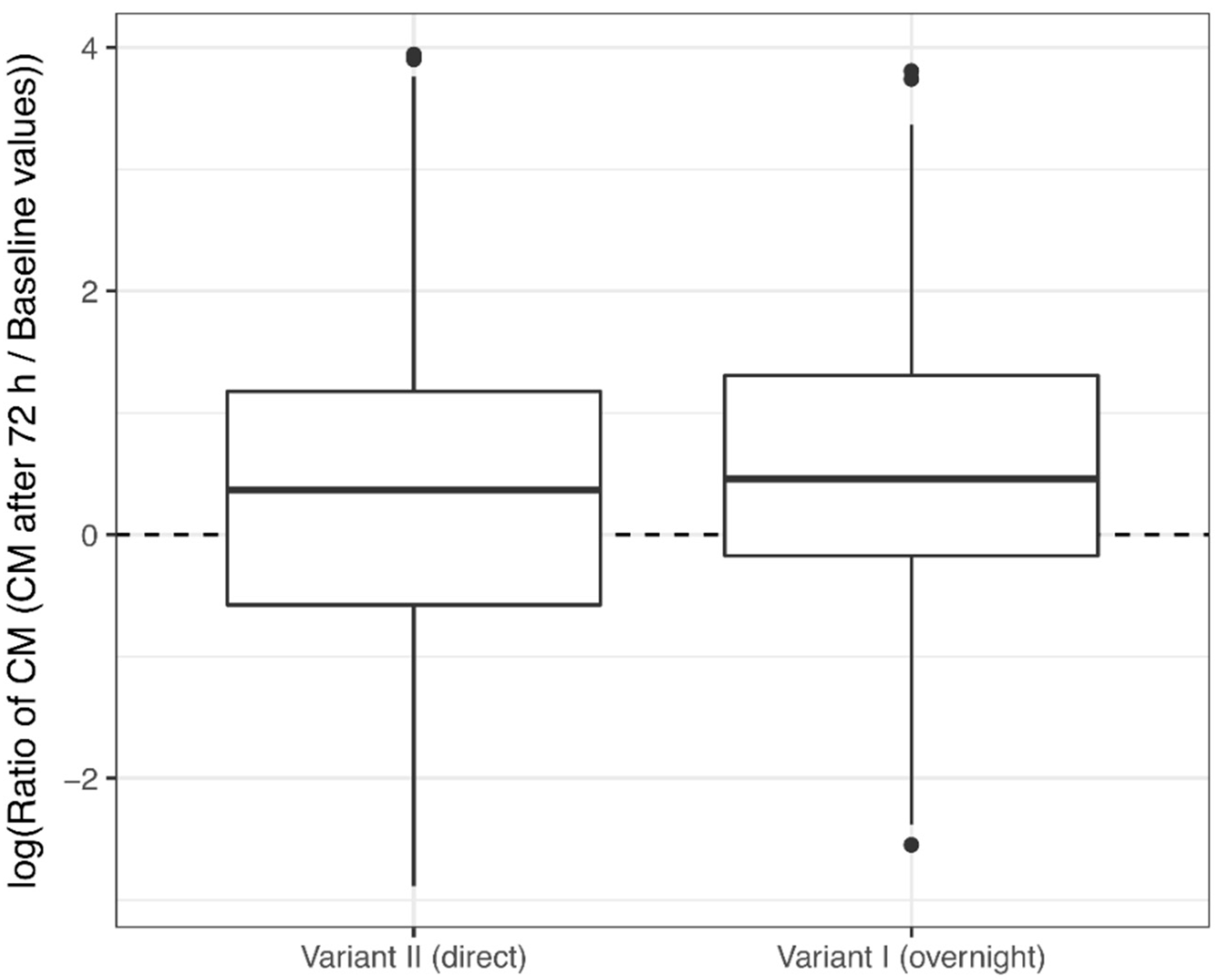
3.1. Corticosterone Monitoring
3.2. Hen-Human Relationship: Touch Test
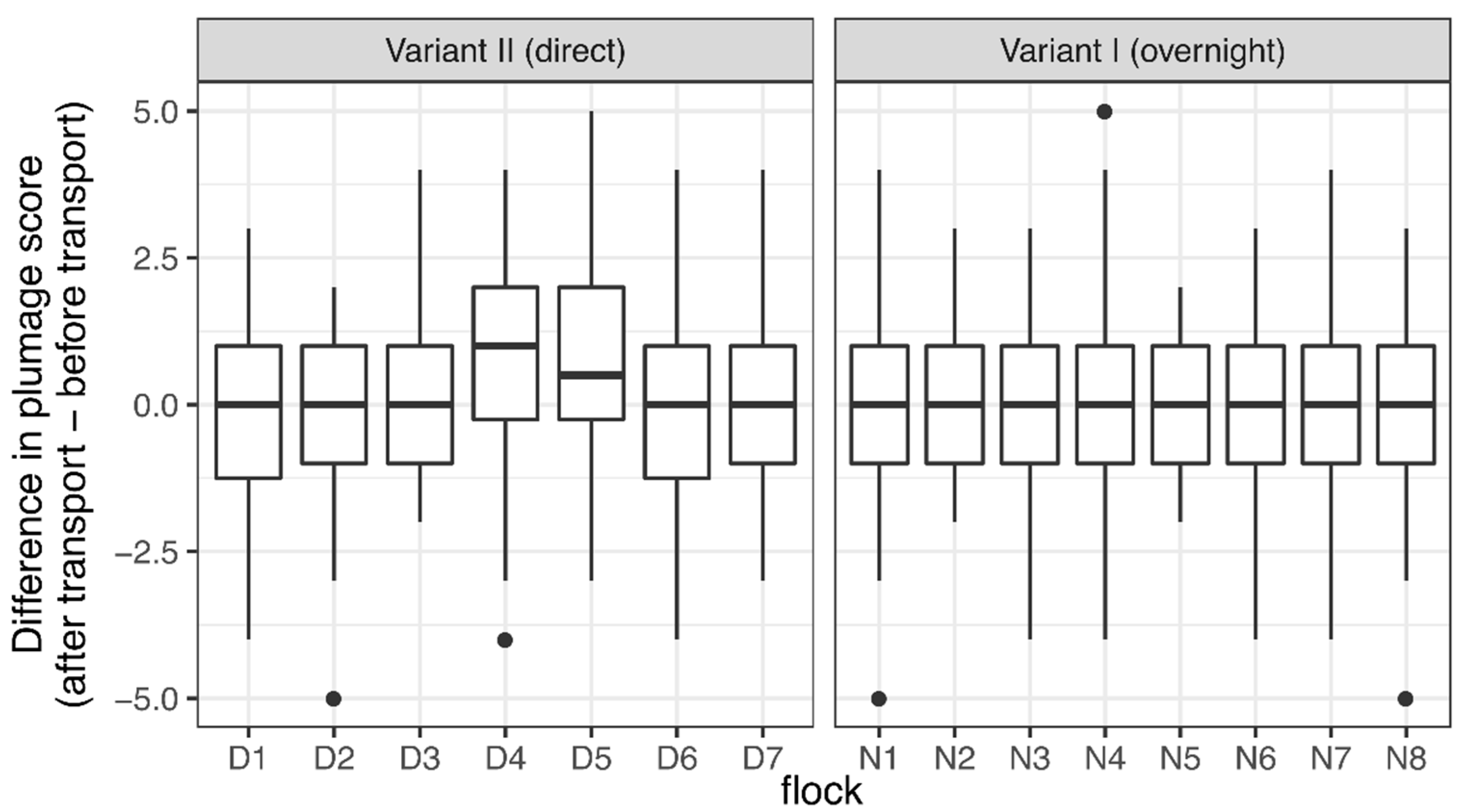
3.3. Body Condition
4. Discussion
4.1. Corticosterone Monitoring
4.2. Hen-Human Relationship: Touch Test
4.3. Body Condition
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Organisation for Animal Health. Terrestrial Animal Health Code. Available online: http://www.oie.int/international-standard-setting/terrestrial-code/ (accessed on 19 October 2017).
- Bär, M. Wer warum bio kauft. Bioaktuell 2011, 1, 1–2. [Google Scholar]
- European Council. Council Regulation (EC) No 1/2005 of 22 December 2004 on the protection of animals during transport and related operations and amending Directives 64/432/EEC and 93/119/EC and regulation (EC) No 1255/97. Off. J. Eur. Union 2004, 3, 1–44. [Google Scholar]
- Animal Welfare Regulation of 23 April 2008 (Status as of 1 May 2017), AS 2008 2985. Switzerland. 2008. Available online: https://www.admin.ch/opc/de/classified-compilation/20080796/201705010000/455.1.pdf (accessed on 21 November 2017).
- Animal Transportation Regulation of 11 February 2009 (BGBL. I S. 375), Amended by Article 9 (14) of the Regulation of 3 December 2015 (BGBL. I S. 2178). Germany. 2015. Available online: https://www.gesetze-im-internet.de/tierschtrv_2009/BJNR037500009.html (accessed on 5 December 2017).
- Demeter Standards of 1 October 2017. Germany. 2017. Available online: https://www.demeter.de/sites/default/files/richtlinien/richtlinien_gesamt.pdf (accessed on 5 December 2017).
- Bioland Standards of 28 November 2017. Germany. 2017. Available online: https://www.bioland.de/fileadmin/dateien/HP_Dokumente/Richtlinien/Bioland_Richtlinien_28_Nov_2017.pdf (accessed on 5 December 2017).
- Naturland Standards of May 2017. Germany. 2017. Available online: https://www.naturland.de/images/Naturland/Richtlinien/Naturland-Richtlinien_Erzeugung.pdf (accessed on 5 December 2017).
- Bio Suisse. Richtlinien für die Erzeugung, Verarbeitung und den Handel von Knospe-Produkten. Available online: https://www.bio-suisse.ch/media/VundH/Regelwerk/2018/DE/rl_2018_1.1_d_gesamt_11.12.2017.pdf (accessed on 17 October 2018).
- KAGfreiland-Livestock-Standards 2017. Switzerland. 2017. Available online: https://www.kagfreiland.ch/images/BAUERN_PRODUKTE/Label/Infos_KAG-Bauern/KAG_RL2017.pdf (accessed on 5 December 2017).
- Schweizer Tierschutz. Transport von Gross- und Kleinvieh: Richtlinie für die Überwachung durch den Kontrolldienst des Schweizer Tierschutz STS vom 1. Available online: https://www.viehhandel-schweiz.ch/fileadmin/files/Haendlerinfos-Weiteres/Richtlinien_Tiertransporte_GrossKleinvieh_2018.pdf (accessed on 17 October 2018).
- Freeman, B.M. The Stress Syndrome. Worlds Poult. Sci. J. 1987, 43, 15–19. [Google Scholar] [CrossRef]
- Mitchell, M.A.; Kettlewell, P.J. Physiological stress and welfare of broiler chickens in transit: Solutions not problems! Poult. Sci. 1998, 77, 1803–1814. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, M.A.; Kettlewell, P.J. Catching and transport of broiler chickens. Proc. Fourth Eur. Symp. Poult. Welf. 1993, 219–229. [Google Scholar]
- Mitchell, M.A.; Kettlewell, P.J. Road transportation of broiler chickens: Induction of physiological stress. Worlds Poult. Sci. J. 1994, 50, 57–59. [Google Scholar] [CrossRef]
- Scott, G.B. Poultry handling: A review of mechanical devices and their effect on bird welfare. Worlds Poult. Sci. J. 1993, 49, 44–57. [Google Scholar] [CrossRef]
- Beuving, G.; Vonder, G.M.A. Comparison of the adrenal sensitivity to ACTH of laying hens with immobilization and plasma baseline levels of corticosterone. Gen. Comp. Endocrinol. 1986, 62, 353–358. [Google Scholar] [CrossRef]
- Animal transportation law of 31 July 2007 (status as of 16 January 2016). Austria. 2017. Available online: https://www.ris.bka.gv.at/GeltendeFassung.wxe?Abfrage=Bundesnormen&Gesetzesnummer=20005398 (accessed on 8 April 2018).
- Bio Austria standards of May 2017. Austria. 2017. Available online: https://www.bio-austria.at/app/uploads/BIO-AUSTRIA_Produktionsrichtlinien_201803.pdf (accessed on 8 April 2018).
- European Council. Council Regulation (EC) No 889/2008 of 5 September 2008 laying down detailed rules for the implementation of Council Regulation (EC) No 834/2007 on organic production and labelling of organic products with regard to organic production, labelling and control. Off. J. Eur. Union. 2008, 250, 1–84. [Google Scholar]
- Rettenbacher, S.; Möstl, E.; Hackl, R.; Ghareeb, K.; Palme, R. Measurement of corticosterone metabolites in chicken droppings. Br. Poult. Sci. 2004, 45, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Palme, R.; Touma, C.; Arias, N.; Dominchin, M.F.; Lepschy, M. Steroid extraction: Get the best out of faecal samples. Wien. Tierärztliche Monatsschr. 2013, 100, 238–246. [Google Scholar]
- Rettenbacher, S.; Palme, R. Biological validation of a non-invasive method for stress assessment in chickens. Berl. Munch. Tierarztl. Wochenschr. 2009, 122, 8–12. [Google Scholar] [PubMed]
- Jones, R.B. Regular handling and the domestic chick’s fear of human beings: Generalisation of response. Appl. Anim. Behav. Sci. 1994, 42, 129–143. [Google Scholar] [CrossRef]
- Barnett, J.L.; Hemsworth, P.H.; Hennessy, D.P.; McCallum, T.H.; Newman, E.A. The effects of modifying the amount of human contact on behavioural, physiological and production responses of laying hens. Appl. Anim. Behav. Sci. 1994, 41, 87–100. [Google Scholar] [CrossRef]
- Hemsworth, P.H. Human–animal interactions in livestock production. Appl. Anim. Behav. Sci. 2003, 81, 185–198. [Google Scholar] [CrossRef]
- Raubek, J.; Niebuhr, K.; Waiblinger, S. Development of on-farm methods to assess the animal-human relationship in laying hens kept in non-cage systems. Anim. Welf. 2007, 16, 173–175. [Google Scholar]
- Graml, C.; Waiblinger, S.; Niebuhr, K. Validation of tests for on-farm assessment of the hen–human relationship in non-cage systems. Appl. Anim. Behav. Sci. 2008, 111, 301–310. [Google Scholar] [CrossRef]
- Tauson, R.; Elwinger, K.; Holm, K.-E.; Wall, H. Welfare Implications of Changes in Production Systems for Laying Hens, deliverables d.3.2–d.3.3., work Package 3, Health. Available online: http://www.laywel.eu/web/pdf/deliverables%2031-33%20health.pdf (accessed on 17 August 2018).
- Schwarzer, A.; Louton, H.; Erhard, M.H. Vorstellung Junghennen. In Tagung der DVG-Fachgruppen Umwelt-und Tierhygiene im Rahmen des DVG-Vet-Congresses am 12; Estrel Convention Center Berlin: Berlin, Germany, 2015; Volume 74. [Google Scholar]
- R Core Team: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: http://www.R-project.org/ (accessed on 10 April 2017).
- Rue, H.; Martino, S.; Chopin, N. Approximate Bayesian inference for latent Gaussian models by using integrated nested Laplace approximations. J. R. Stat. Soc. Ser. B 2009, 71, 319–392. [Google Scholar] [CrossRef]
- Fraisse, F.; Cockrem, J.F. Corticosterone and fear behaviour in white and brown caged laying hens. Br. Poult. Sci. 2006, 47, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Gallup, G.G.J.; Ledbetter, D.H.; Maser, J.D. Strain differences among chickens in tonic immobility: Evidence for an emotionality component. J. Comp. Physiol. Psychol. 1976, 90. [Google Scholar] [CrossRef]
- Jones, R.B.; Faure, J.M. Sex and strain comparisons of tonic immobility (“righting time”) in the domestic fowl and the effects of various methods of induction. Behav. Process. 1981, 6, 47–55. [Google Scholar] [CrossRef]
- Albentosa, M.J.; Kjær, J.B.; Nicol, C.J. Strain and age differences in behaviour, fear response and pecking tendency in laying hens. Br. Poult. Sci. 2003, 44, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Mahboub, H.D.H.; Müller, J.; Von Borell, E. Outdoor use, tonic immobility, heterophil/lymphocyte ratio and feather condition in free-range laying hens of different genotype. Br. Poult. Sci. 2004, 45, 738–744. [Google Scholar] [CrossRef] [PubMed]
- De Haas, E.N.; Kemp, B.; Bolhuis, J.E.; Groothuis, T.; Rodenburg, T.B. Fear, stress, and feather pecking in commercial white and brown laying hen parent-stock flocks and their relationships with production parameters. Poult. Sci. 2013, 92, 2259–2269. [Google Scholar] [CrossRef] [PubMed]
- Uitdehaag, K.A.; Rodenburg, T.B.; Komen, H.; Kemp, B.; Van Arendonk, J.A.M. The association of response to a novel object with subsequent performance and feather damage in adult, cage-housed, pure-bred Rhode Island Red laying hens. Poult. Sci. 2008, 87, 2486–2492. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.W.; Jefferson, L. Different behavioral and physiological responses in two genetic lines of laying hens after transportation. Poult. Sci. 2008, 87, 885–892. [Google Scholar] [CrossRef] [PubMed]
- de Jong, I.C.; van Voorst, A.S.; Erkens, J.H.F.; Ehlhardt, D.A.; Blokhuis, H.J. Determination of the circadian rhythm in plasma corticosterone and catecholamine concentrations in growing broiler breeders using intravenous cannulation. Physiol. Behav. 2001, 74, 299–304. [Google Scholar] [CrossRef]
- Graml, C.; Niebuhr, K.; Waiblinger, S. Reaction of laying hens to humans in the home or a novel environment. Appl. Anim. Behav. Sci. 2008, 113, 98–109. [Google Scholar] [CrossRef]
- Tauson, R.; Kjaer, J.; Maria, G.A.; Cepero, R.; Holm, K.E. The Creation of A Common Scoring System for the Integument and Health of Laying Hens. Applied Scoring Of Integument And Health In Laying Hens. Available online: http://www.laywel.eu (accessed on 17 August 2017).
- King, J.R. Adaptive periodic fat storage by birds. Proc. Int. Ornithol. Congr. 1972, 15, 200–217. [Google Scholar]
- Clark, G.A. Body weights of birds: A review. Condor 1979, 81, 193–202. [Google Scholar] [CrossRef]
- Haftorn, S. Seasonal and diurnal body weight variations in titmice, based on analyses of individual birds. Wilson Bull. 1989, 101, 217–235. [Google Scholar]
- Lehikoinen, E. Seasonality of the daily weight cycle in wintering passerines and its consequences. Ornis Scand. 1987, 18, 216–226. [Google Scholar] [CrossRef]
- Scholtyssek, S.; Ehinger, F. Transporteinflüsse auf Broiler und deren Schlachtkörper. Arch. Geflügelk. 1976, 40, 27–35. [Google Scholar]
| Regulation | Country level | |||||||
|---|---|---|---|---|---|---|---|---|
| Switzerland | Germany | Austria | EU | |||||
| Federal | Label: Bio Suisse | Label: KAGfreiland | Federal | Label: Demeter, Bioland, Naturland | Federal | Label: Bio Austria | ||
| Maximum Stocking Density Per Pen | ||||||||
| Pullets | No specification [4] | 4000 [9] | 4000 [10] | No specification [5] | 3150 [6] | 4800 [18] | 4800 [19] | 4800 [20] |
| Layers | 18,000 [4] | 2000 [9] | 2000 [10] | 6000 [5] | 3000 [6,7,8] | 3000 [18] | 3000 [19] | 3000 [20] |
| Transport | ||||||||
| Minimum Space in Transport Cage | 160 cm2/kg for <3.0 kg [4] | According to Transport Guideline of STS [9] | According to Transport Guideline of STS [10] | 200 cm2/kg for <1.0 kg 190 cm2/kg for <1.3 kg 180 cm2/kg for <1.6 kg 170 cm2/kg for <2.0 kg [5] | No specification [6,7,8] | 180–200 cm2/kg for <1.6 kg 160 cm2/kg for <3.0 kg [18] | 180–200 cm2/kg for <1.6 kg 160 cm2/kg for <3.0 kg [19] | 180–200 cm2/kg for <1.6 kg 160 cm2/kg for <3.0 kg [20] |
| Maximum Transport Duration | 8 h [4] | According to Transport Guideline of STS [9] | No specification for pullets [10] | No specification for pullets [5] | 4 h [6,7,8] | No specification for pullets [18] | 6 h [19] | 12 h [3] |
| Variant | Line | Baseline Value (ng/g) | SEM (ng/g) | Baseline Value Total (ng/g) | SEM (ng/g) |
|---|---|---|---|---|---|
| I | HNS | 57 | 3 | 43 | 2 |
| HNB | 30 | 2 | |||
| II | HNS | 93 | 8 | 66 | 4 |
| HNB | 40 | 3 |
| Variant | Hours after Transportation | Min CM (ng/g) | Max CM (ng/g) | Mean CM (ng/g) | SEM (ng/g) |
|---|---|---|---|---|---|
| I | 0 | 4 | 1337 | 173 | 22 |
| 3 | 4 | 570 | 111 | 7 | |
| 6 | 3 | 440 | 61 | 7 | |
| 9 | 5 | 568 | 96 | 5 | |
| 10 | 4 | 459 | 69 | 6 | |
| 12 | 11 | 315 | 128 | 3 | |
| 24 | 4 | 786 | 92 | 4 | |
| 48 | 3 | 402 | 73 | 3 | |
| 72 | 2 | 245 | 64 | 3 | |
| II | 0 | 4 | 2215 | 323 | 11 |
| 3 | 4 | 967 | 134 | 5 | |
| 6 | 3 | 992 | 112 | 3 | |
| 10 | 4 | 590 | 89 | 11 | |
| 24 | 5 | 632 | 95 | 4 | |
| 34 | 4 | 307 | 67 | 9 | |
| 48 | 5 | 462 | 86 | 5 | |
| 58 | 4 | 330 | 69 | 3 | |
| 72 | 4 | 456 | 74 | 2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sprafke, H.; Palme, R.; Schmidt, P.; Erhard, M.; Bergmann, S. Effect of Two Transport Options on the Welfare of Two Genetic Lines of Organic Free Range Pullets in Switzerland. Animals 2018, 8, 183. https://doi.org/10.3390/ani8100183
Sprafke H, Palme R, Schmidt P, Erhard M, Bergmann S. Effect of Two Transport Options on the Welfare of Two Genetic Lines of Organic Free Range Pullets in Switzerland. Animals. 2018; 8(10):183. https://doi.org/10.3390/ani8100183
Chicago/Turabian StyleSprafke, Helena, Rupert Palme, Paul Schmidt, Michael Erhard, and Shana Bergmann. 2018. "Effect of Two Transport Options on the Welfare of Two Genetic Lines of Organic Free Range Pullets in Switzerland" Animals 8, no. 10: 183. https://doi.org/10.3390/ani8100183
APA StyleSprafke, H., Palme, R., Schmidt, P., Erhard, M., & Bergmann, S. (2018). Effect of Two Transport Options on the Welfare of Two Genetic Lines of Organic Free Range Pullets in Switzerland. Animals, 8(10), 183. https://doi.org/10.3390/ani8100183





