Duetting Patterns of Titi Monkeys (Primates, Pitheciidae: Callicebinae) and Relationships with Phylogeny
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Video and Sound Recordings
2.1.1. Field Recordings in Bolivia
2.1.2. Field Recordings in Ecuador
2.1.3. Field Recordings in Peru
2.1.4. Macaulay Library Sound Archive
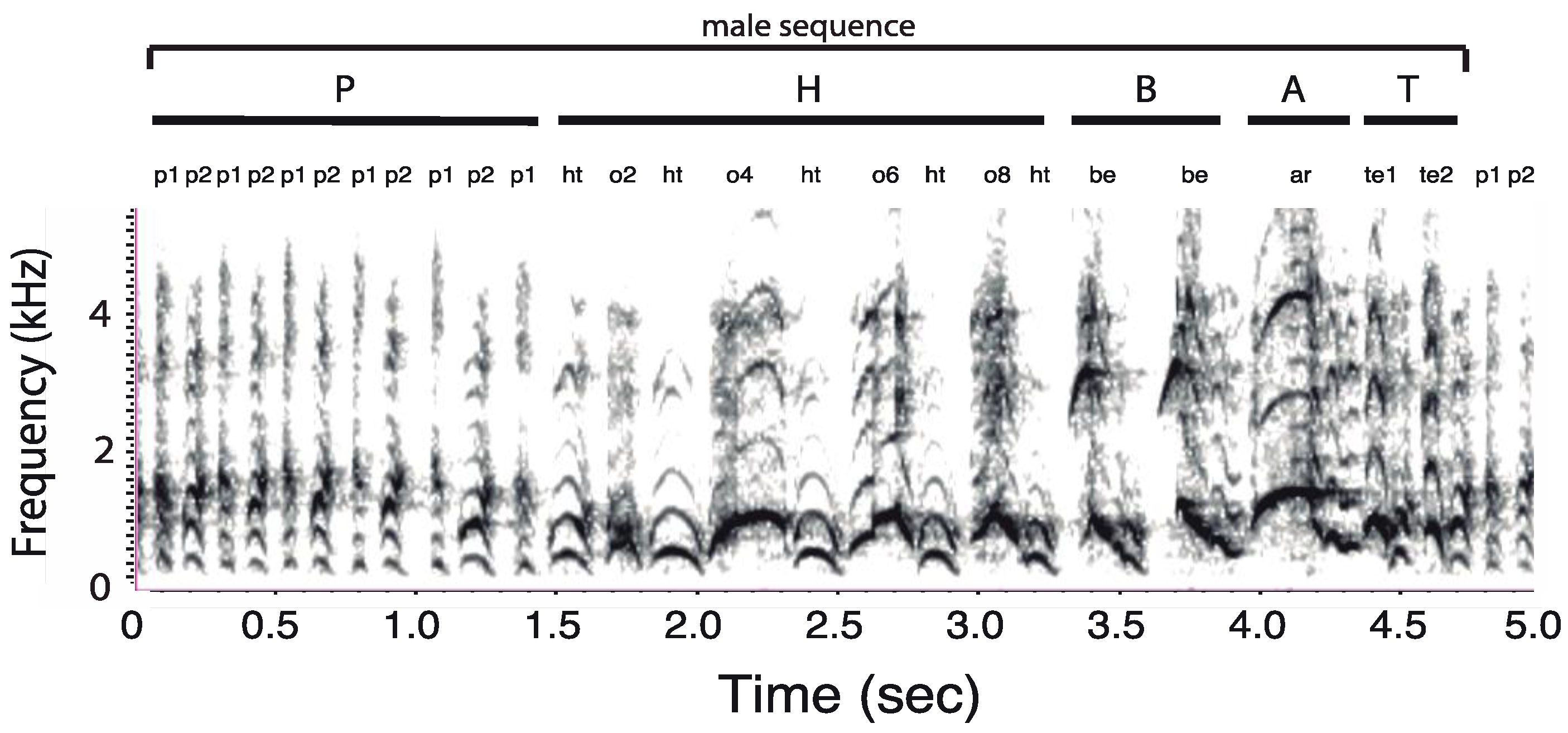
2.2. Acoustic Terminology
2.3. Acoustic Analysis
2.3.1. Power Spectrum of the Joint Signal
2.3.2. Dominant Frequency of the Joint Signal
2.3.3. Sequence Duration
2.3.4. Call Rate
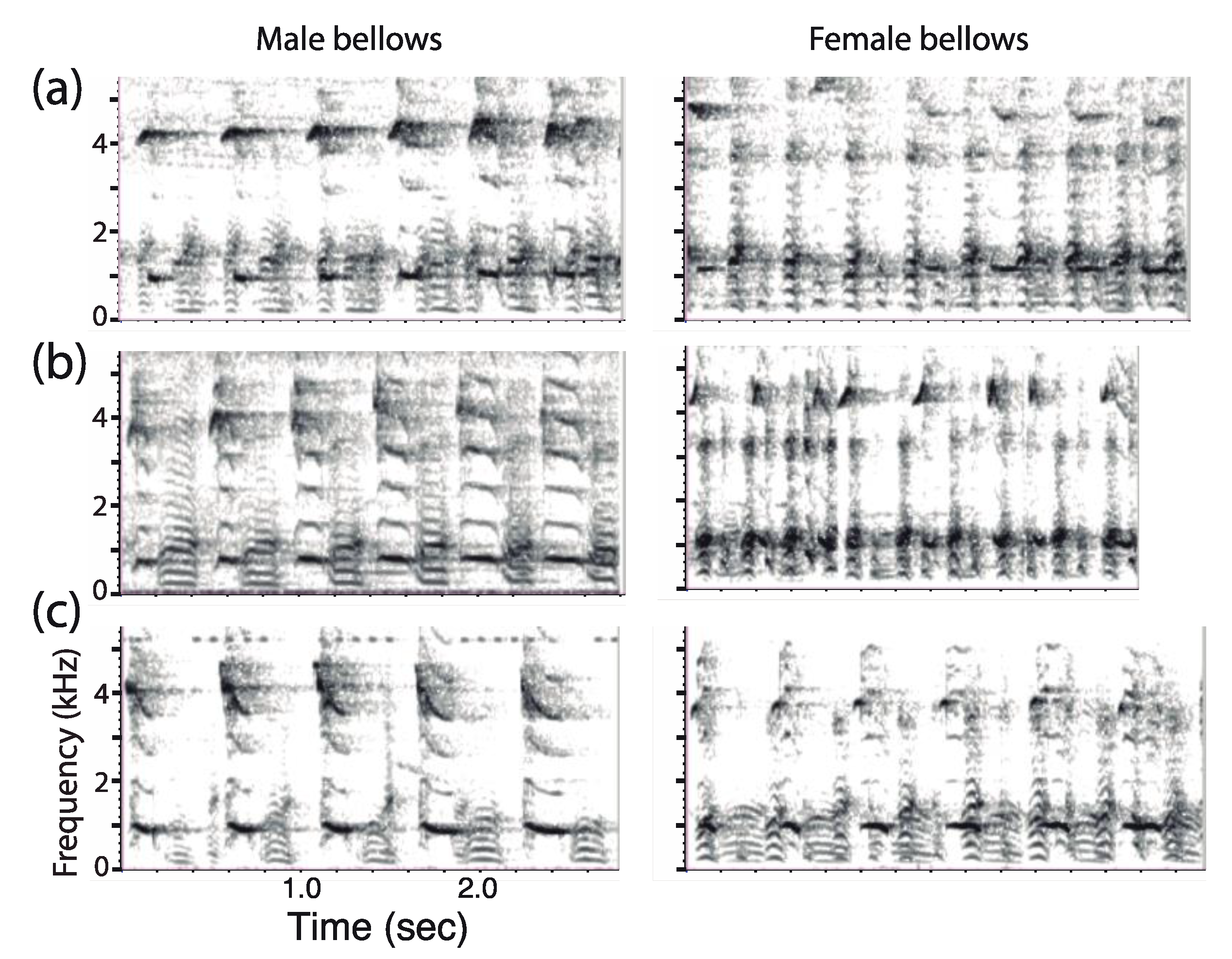
2.3.5. Sex Difference in Call Structure
2.4. Statistics
3. Results
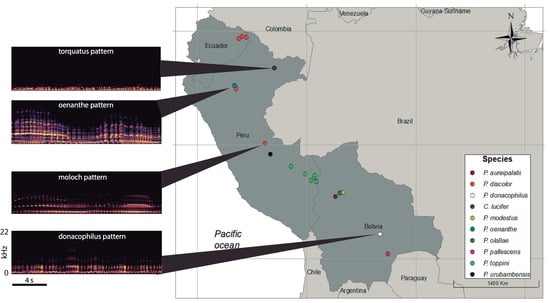
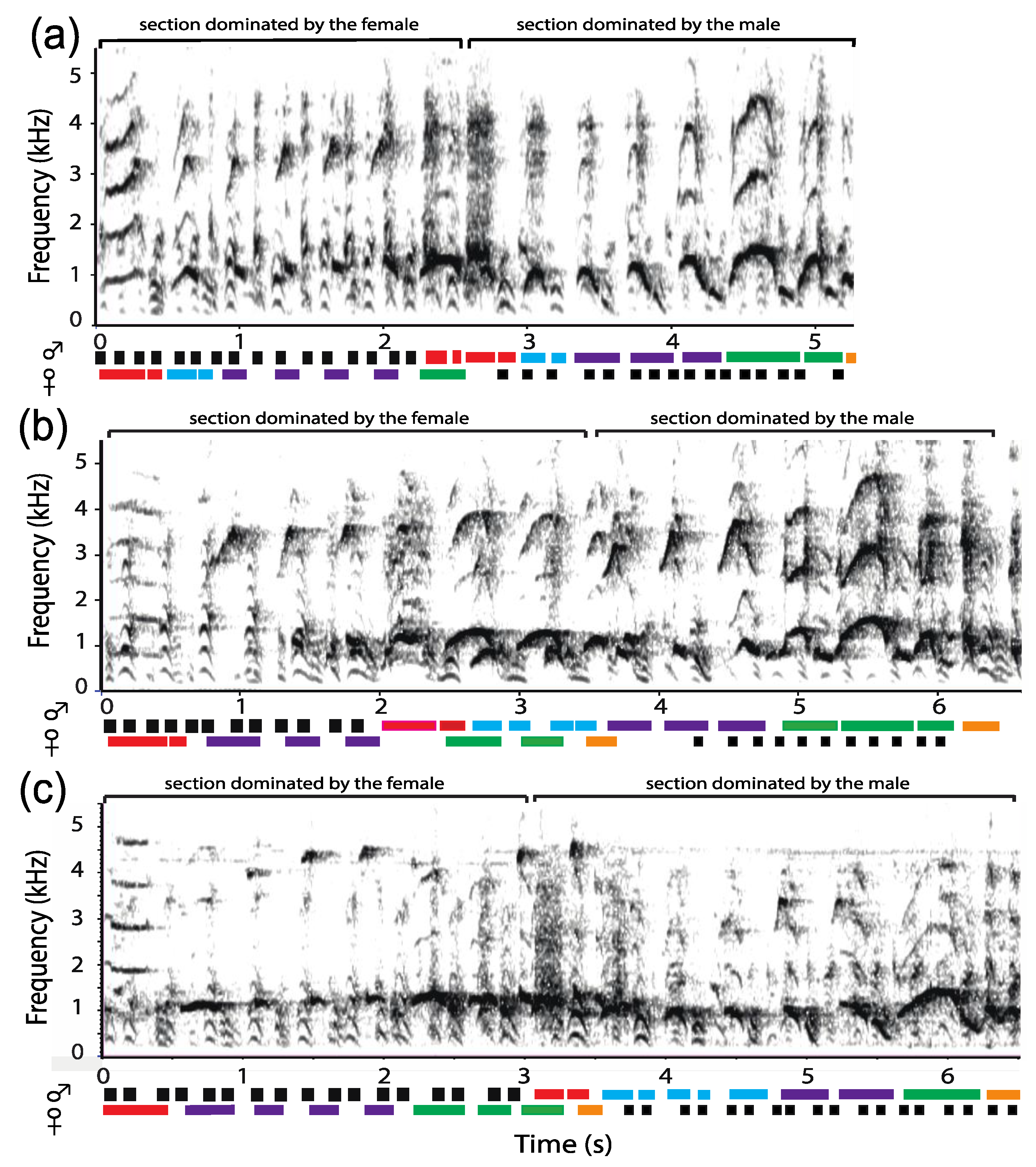
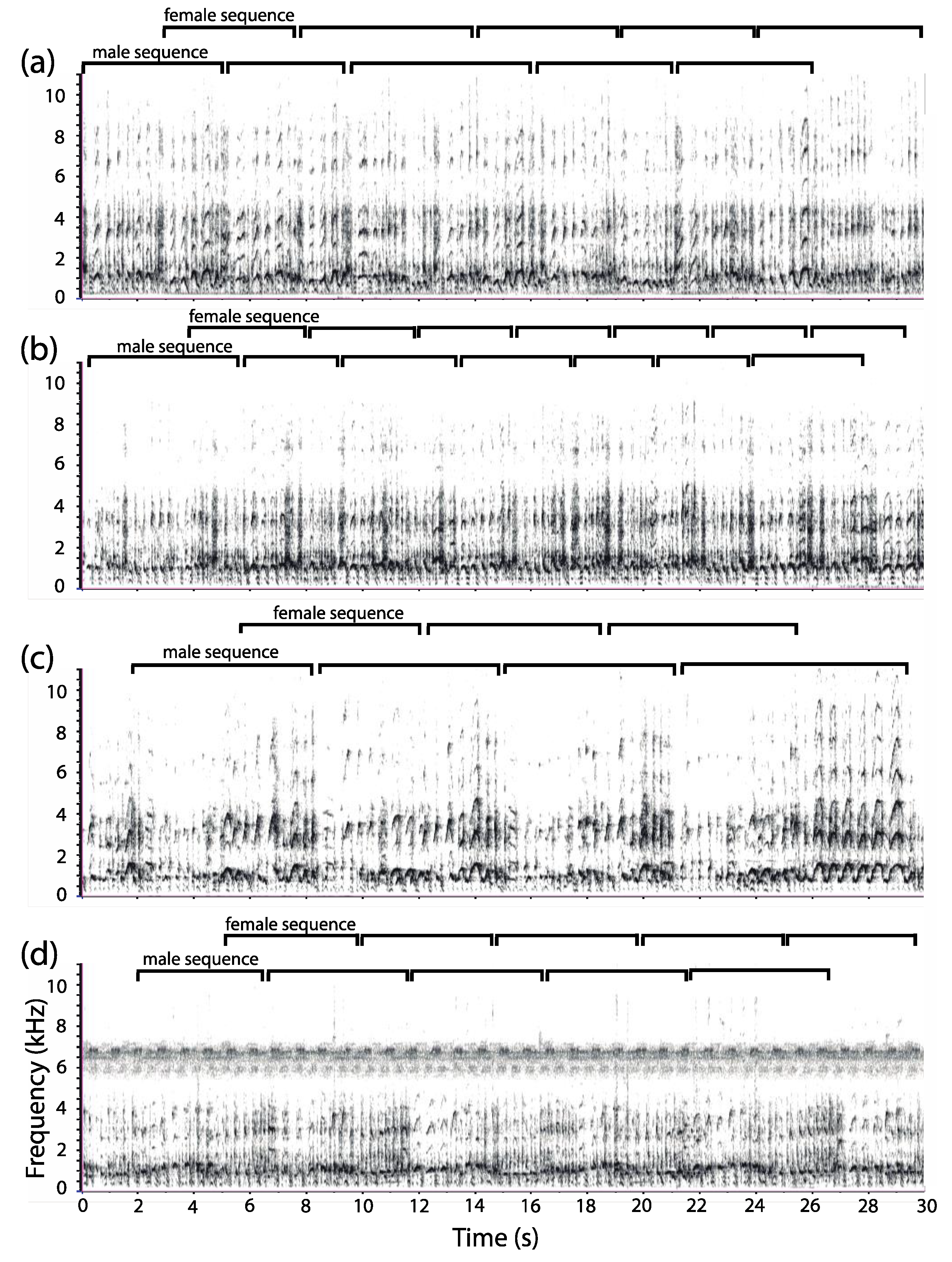
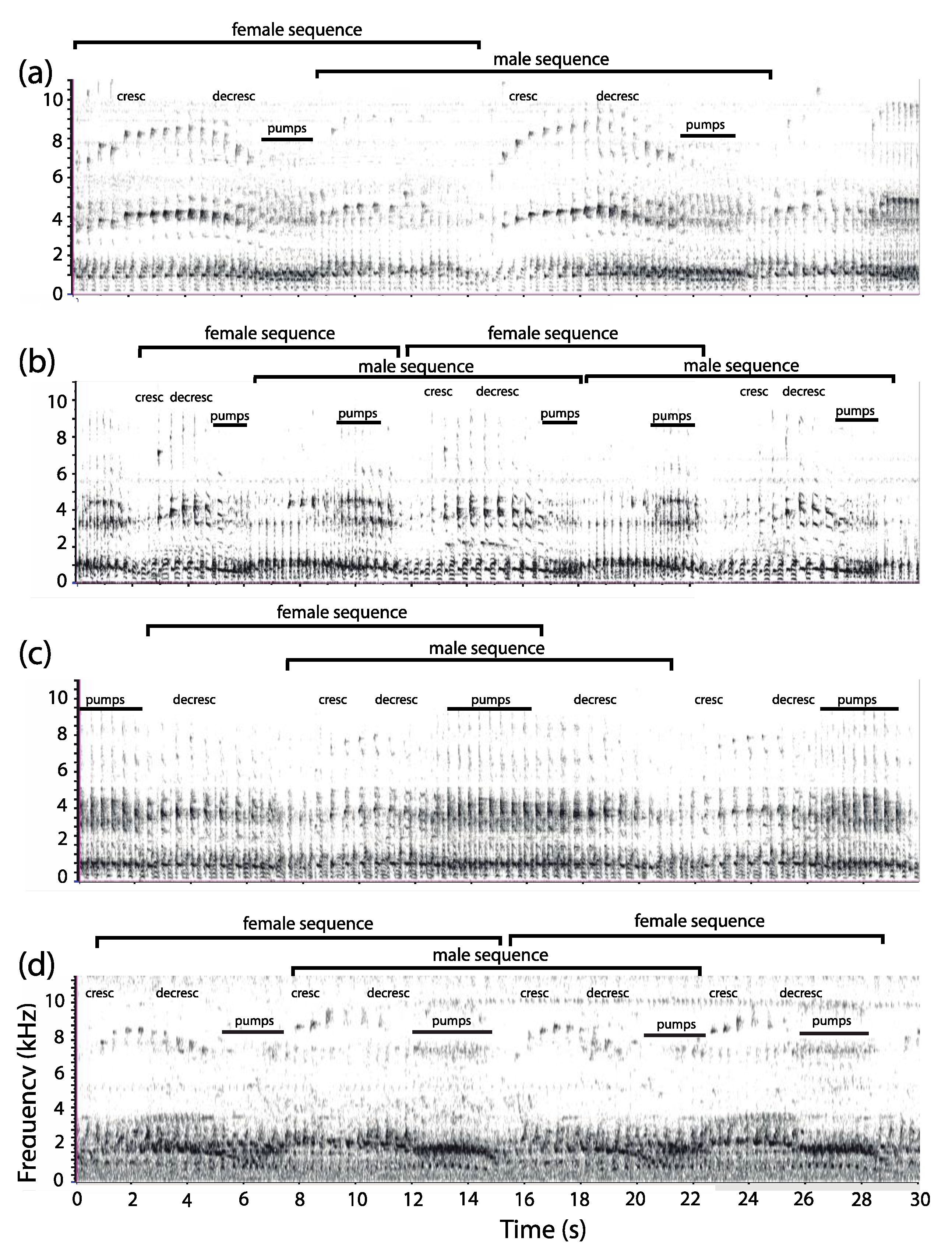
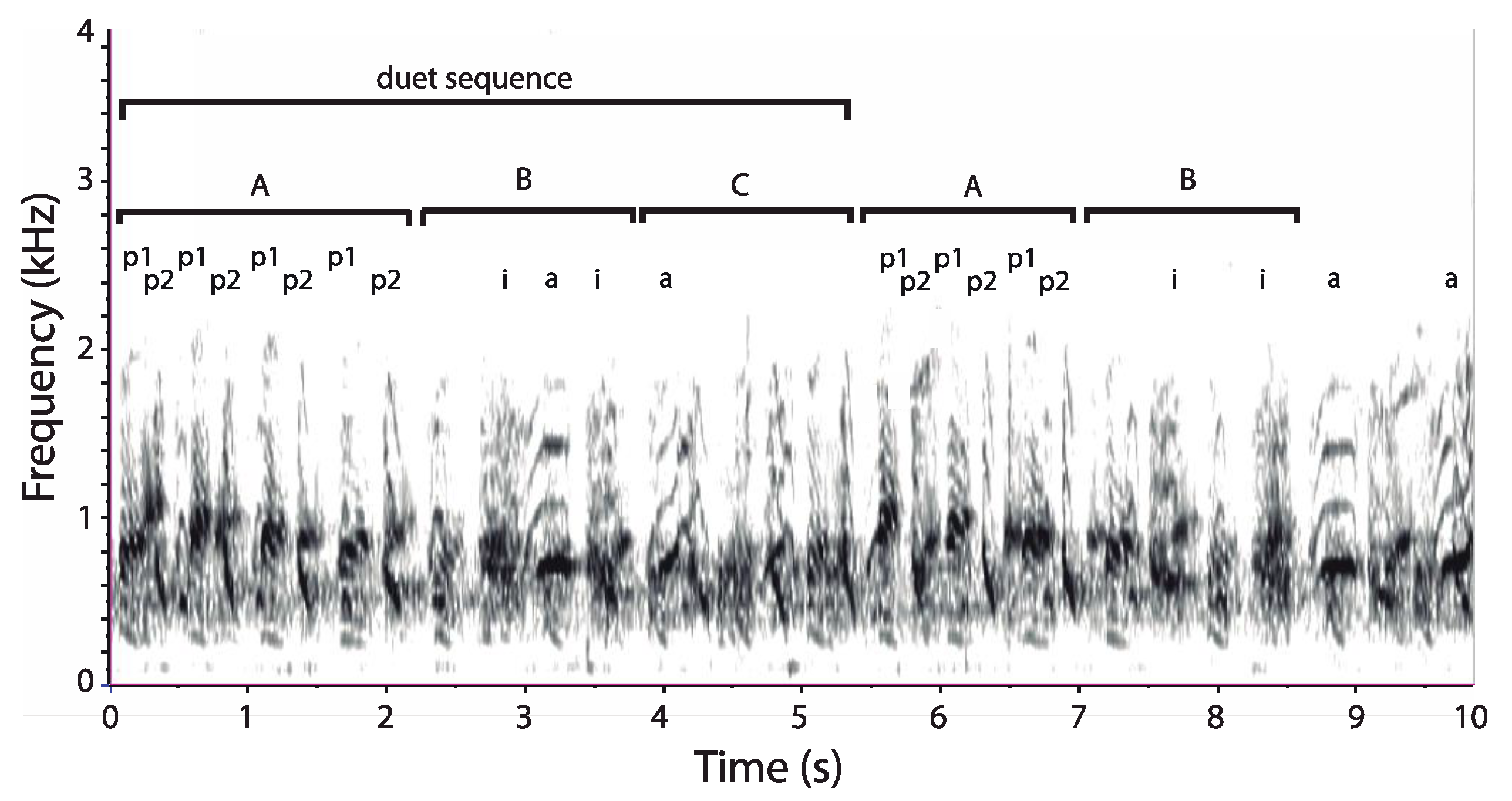
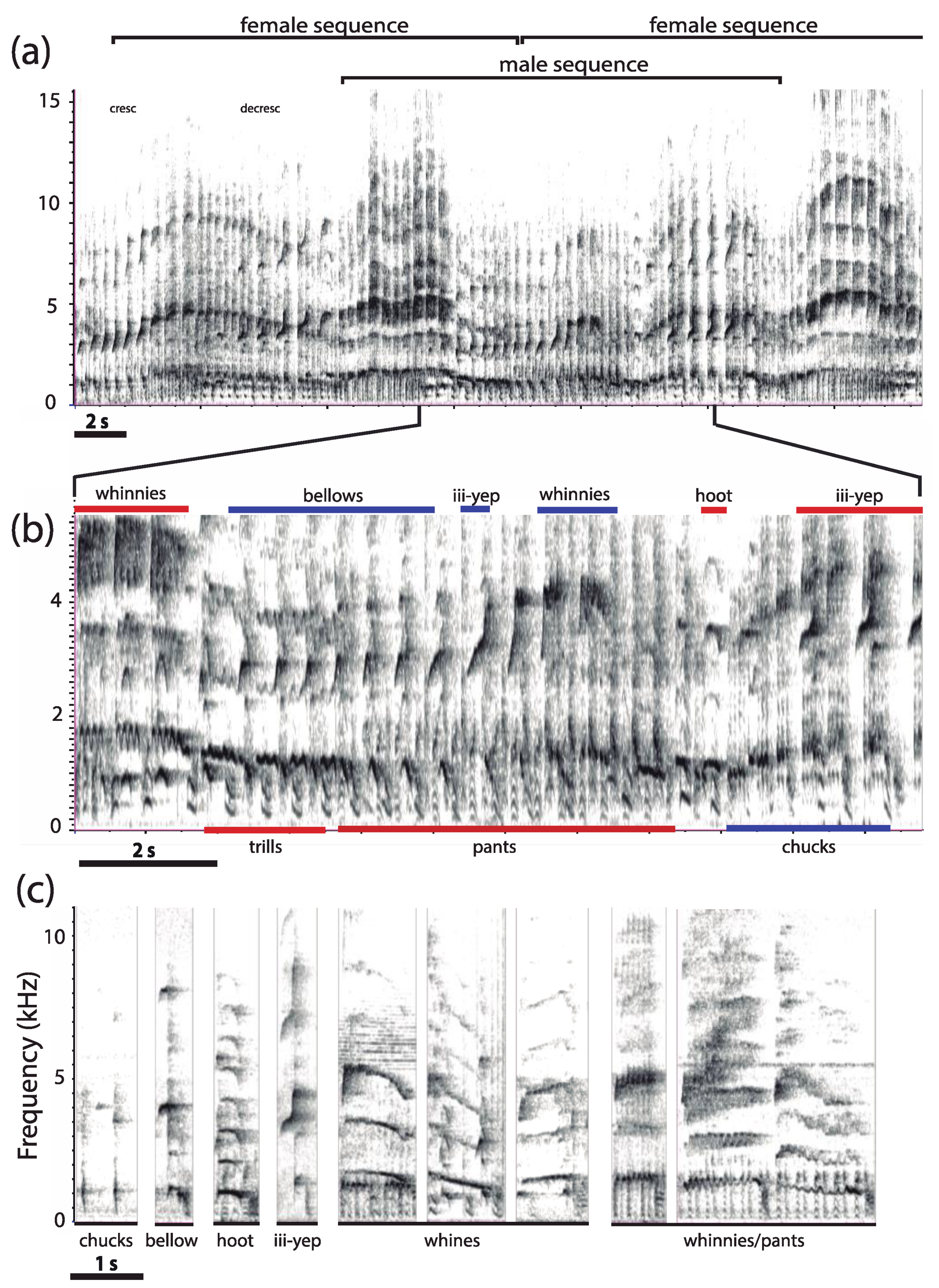
3.1. Organization of Duets in the Donacophilus Group
3.2. Organization of Duets in the Moloch Group
3.3. Organization of Duets in the Torquatus Group
3.4. Organization of Duets in the San Martin Titi Monkey
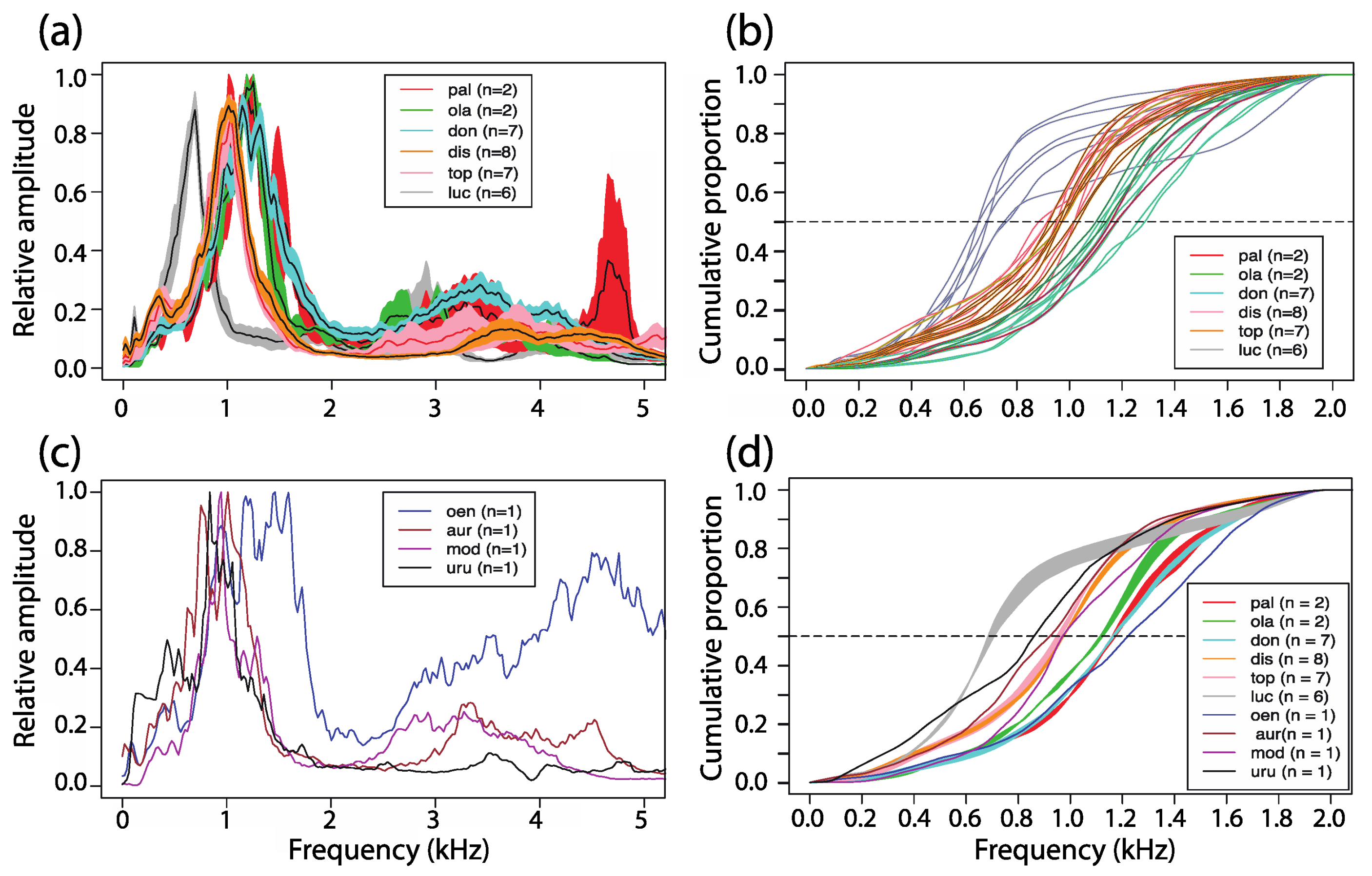
3.5. Power Spectral Density
3.6. Acoustic Parameters
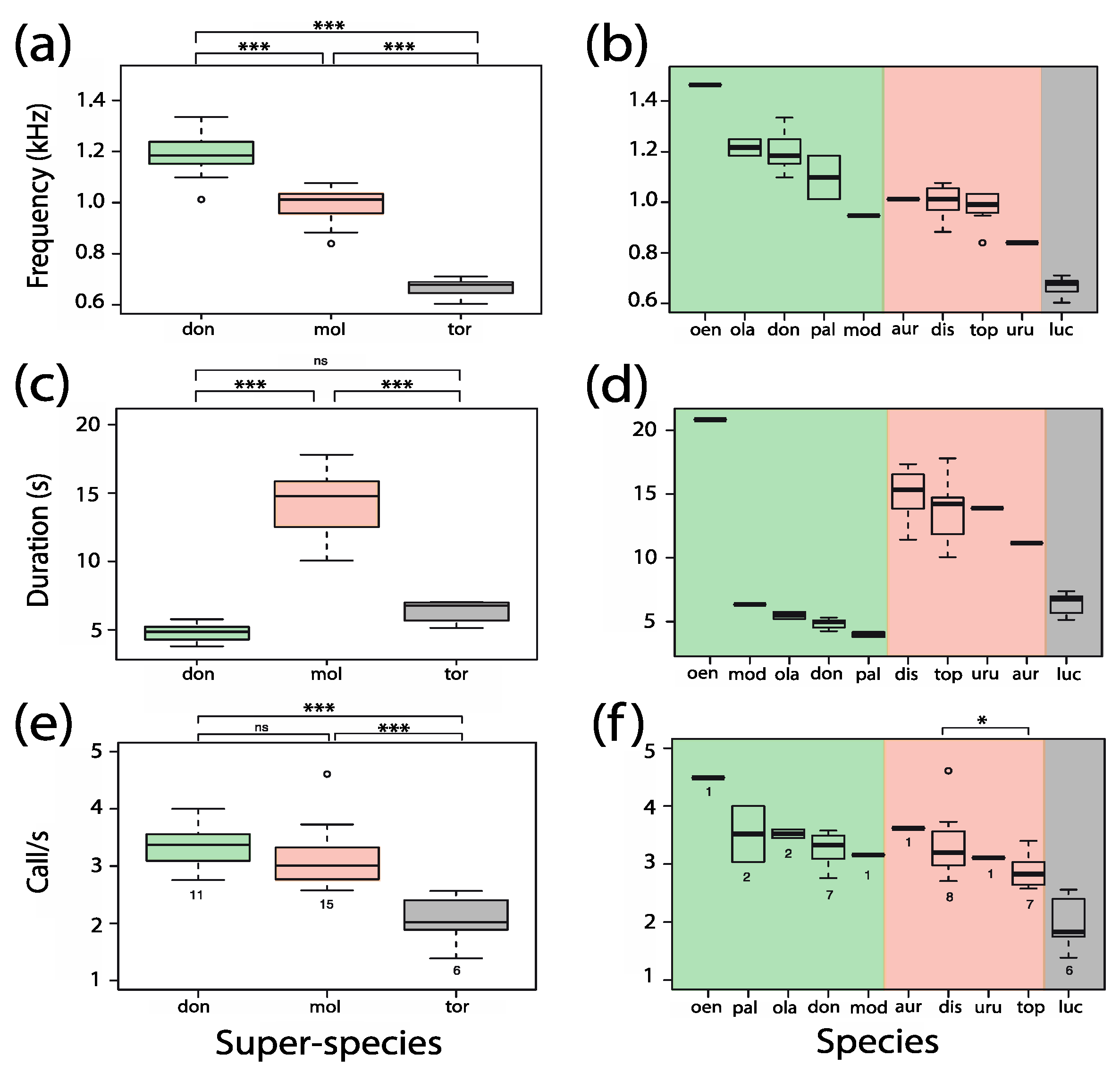
3.6.1. Dominant Frequency
3.6.2. Sequence Duration
3.6.3. Pant Call Rate
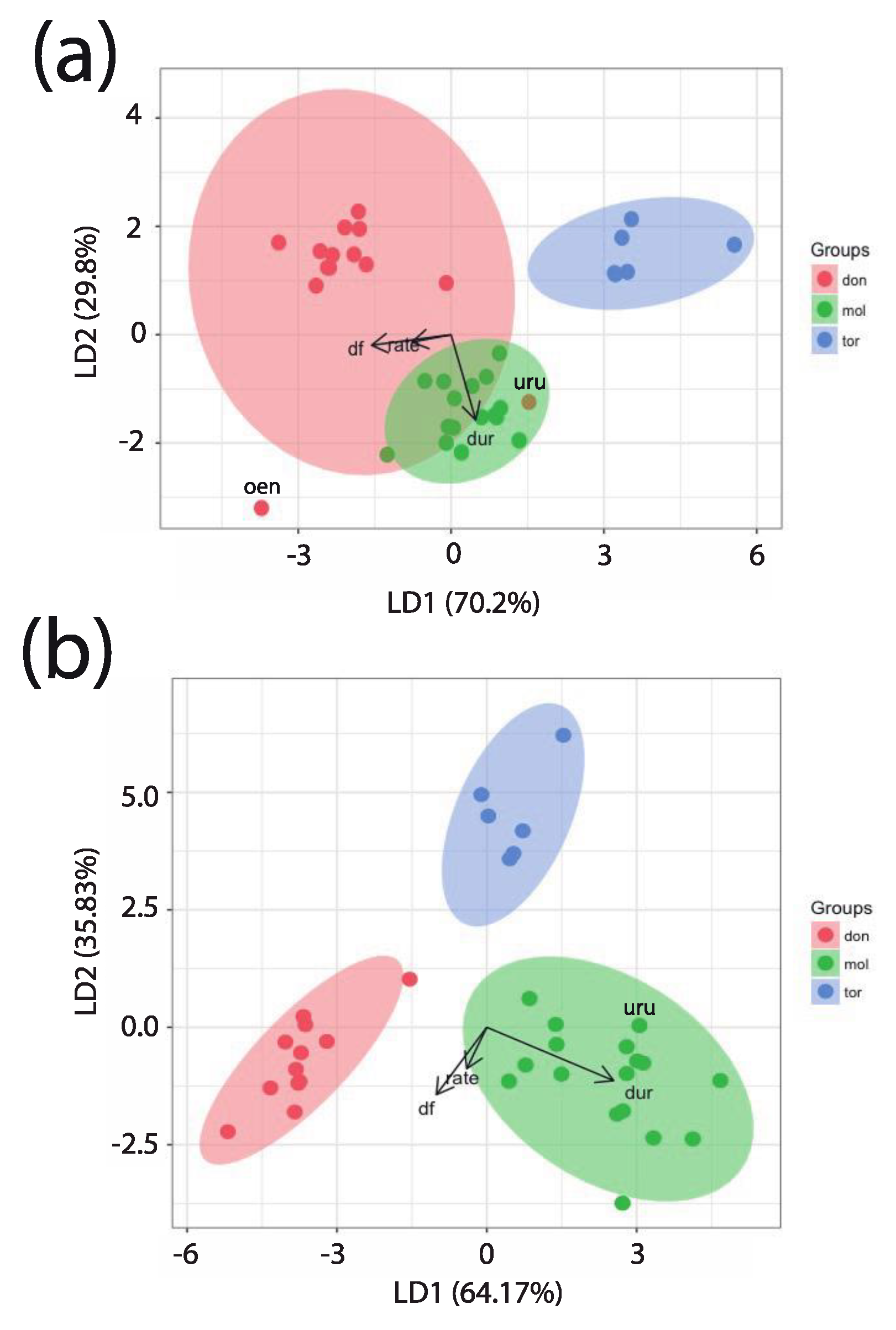
3.7. Discriminant Function Analysis
4. Discussion
4.1. Form and Structure of Titi Monkey Duets
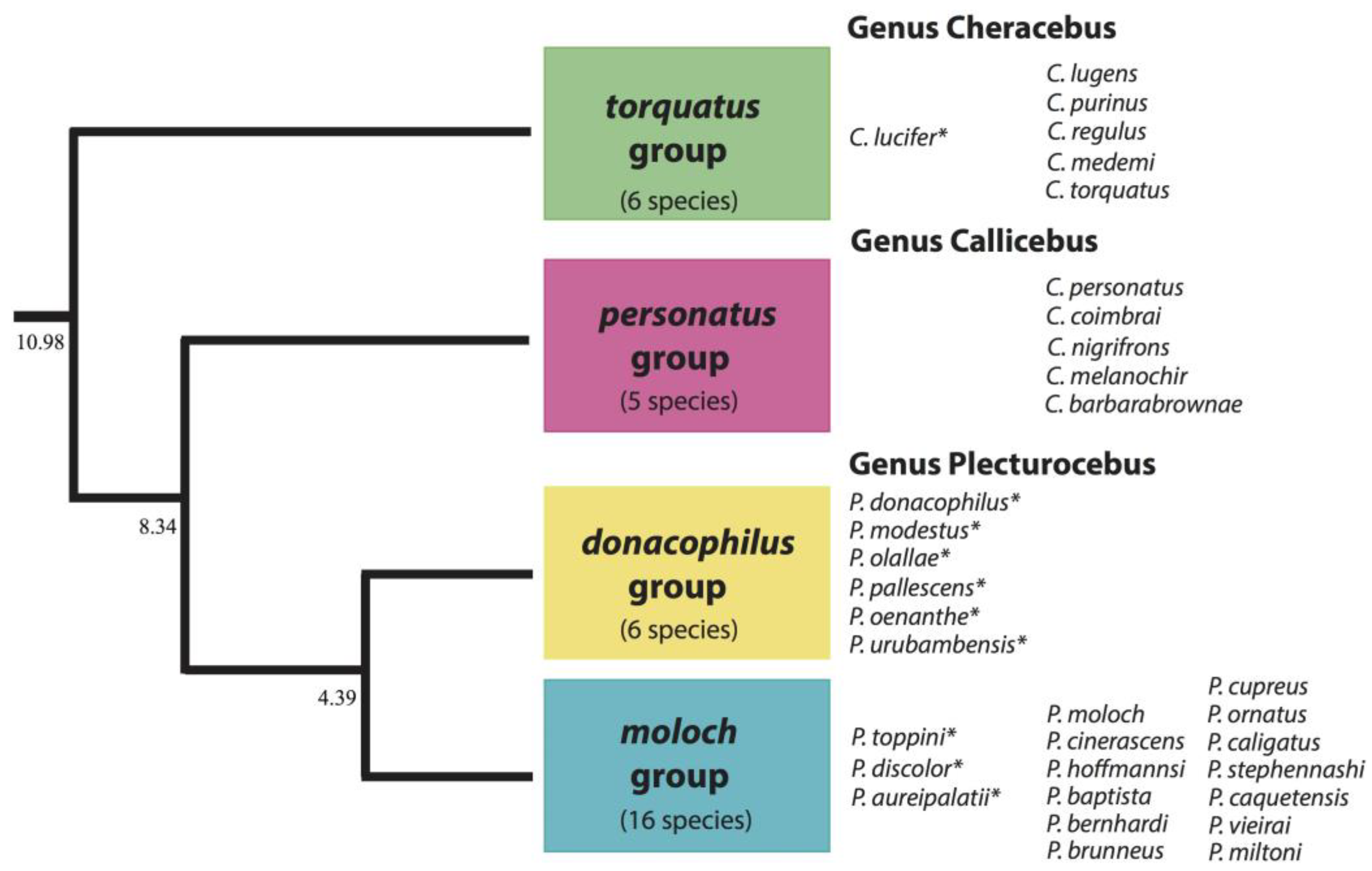
4.2. Titi Monkey Duets and the Taxonomy of the Callicebinae
4.3. Titi Monkey Duets and Environmental Influences
4.4. Implications for Conservation Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix
References
- Bradbury, J.W.; Vehrencamp, S.L. Principles of Animal Communication, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2011. [Google Scholar]
- Stegmann, U.E. Animal Communication Theory: Information and Influence; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Snowdon, C.T.; Brown, C.H.; Petersen, M.R. Primate Communication; Cambridge University Press: Cambridge, UK, 1982. [Google Scholar]
- Rogers, L.J.; Kaplan, G. Songs, Roars, and Rituals: Communication in Birds, Mammals, and Other Animals, 2nd ed.; Harvard University Press: Cambridge, MA, USA, 2002. [Google Scholar]
- Liebal, K.; Waller, B.M.; Burrows, A.M.; Slocombe, K.E. Primate Communication: A Multimodal Approach; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Uetz, G.W.; Clark, D.L.; Roberts, J.A. Multimodal communication in wolf spiders (Lycosidae)—An emerging model for study. Adv. Study Behav. 2016, 48, 117–159. [Google Scholar]
- LaRue, K.M.; Clemens, J.; Berman, G.J.; Murthy, M. Acoustic duetting in Drosophila viridis relies on the integration of auditory and tactile signals. eLife 2015, 4, e07277. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, L.S.; Pessoa, D.M.A.; Mountford, J.K.; Davies, W.I.L.; Hunt, D.M. The genetic and evolutionary drives behind primate color vision. Front. Ecol. Evol. 2017, 5, 34. [Google Scholar] [CrossRef]
- Goulart, V.D.L.R.; Boubli, J.P.; Young, R.J. Medium/long wavelength sensitive opsin diversity in Pitheciidae. Sci. Rep. 2017, 7, 7737. [Google Scholar] [CrossRef] [PubMed]
- Waser, P.M.; Waser, M.S. Experimental studies of primate vocalization: Specializations for long-distance propagation. Z. Tierpsychol. 1977, 43, 239–263. [Google Scholar] [CrossRef]
- Waser, P.M.; Brown, C.H. Habitat acoustics and primate communication. Am. J. Primatol. 1986, 10, 135–154. [Google Scholar] [CrossRef]
- Henwood, K.; Fabrick, A. A quantitative analysis of the dawn chorus: Temporal selection for communicatory optimization. Am. Nat. 1979, 114, 260–274. [Google Scholar] [CrossRef]
- Marten, K.; Marler, P. Sound transmission and its significance for animal vocalization. Behav. Ecol. Sociobiol. 2004, 2, 291–302. [Google Scholar] [CrossRef]
- Horwich, R.H.; Gebhard, K. Roaring rhythms in black howler monkeys (Alouatta pigra) of Belize. Primates 1983, 24, 290–296. [Google Scholar] [CrossRef]
- Oliveira, D.A.G.; Ades, C. Long-distance calls in Neotropical primates. Ann. Acad. Brazileira de Ciências 2004, 76, 393–398. [Google Scholar] [CrossRef]
- Mendes, F.D.C.; Ades, C. Vocal sequential exchanges and intragroup spacing in the Northern muriqui Brachyteles arachnoides hypoxanthus. Ann. Acad. Brazileira de Ciências 2004, 76, 399–404. [Google Scholar] [CrossRef]
- Teixeira da Cunha, R.G.; Jalles-Filho, E. The roaring of Brown howler monkeys (Alouatta guariba clamitans) as a mechanism of active defence of borders. Folia Primatol. 2006, 78, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Mann, N.I.; Dingess, K.A.; Slater, P.J.B. Antiphonal four-part synchronized chorusing in a Neotropical wren. Biol. Lett. 2006, 2, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Baker-Médard, M.S.A.; Baker, M.C.; Logue, D.M. Chorus song of the Indri (Indri indri: Primates, Lemuridae): Group differences and analysis of vocal interactions. Int. J. Comp. Psychol. 2013, 26, 241–255. [Google Scholar]
- Fedurek, P.; Machanda, Z.P.; Schel, A.M.; Slocombe, K.E. Pant hoot chorusing and social bonds in male chimpanzees. Anim. Behav. 2013, 86, 189–196. [Google Scholar] [CrossRef]
- Marshall, J.T.; Sugardjito, J. Gibbon systematics. In Comparative Primate Biology, Vol. 1: Systematics, Evolution, and Anatomy; Swindler, D.R., Erwin, J., Eds.; A.R. Liss: New York, NY, USA, 1986; pp. 137–185. [Google Scholar]
- Catchpole, C.K.; Slater, P.J.B. Bird Song: Themes and Variations, 2nd ed.; Cambridge University Press: Cambridge, UK, 2008. [Google Scholar]
- Heller, K.G.; von Helversen, D. Acoustic communication in phaneropterid bushcrickets: Species-specific delay of female stridulatory response and matching male sensory time window. Behav. Ecol. Sociobiol. 1986, 18, 189–198. [Google Scholar] [CrossRef]
- Hall, M.L.; Magrath, R.D. Temporal coordination signals coalition quality in a duetting songbird. Curr. Biol. 2007, 17, R406–R407. [Google Scholar] [CrossRef] [PubMed]
- Brumm, H.; Slater, P. Animal communication: Timing counts. Curr. Biol. 2007, 17, R521–R523. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Cáceres, K.D. Plain wrens Cantorchilus modestus zeledoni adjust their singing tempo based on self and partner’s cues to perform precisely coordinated duets. J. Avian Biol. 2015, 46, 361–368. [Google Scholar] [CrossRef]
- Wickler, W.; Seibt, U. Vocal dueting and the pair bond. II. Unisono dueting in the African forest weaver, Symplectes bicolor. Z. Tierpsychol. 1980, 52, 217–226. [Google Scholar] [CrossRef]
- Fortune, E.; Rodrígues, C.; Li, D.; Ball, G.M.; Coleman, M.J. Neural mechanisms for the coordination of duet singing in wrens. Science 2011, 334, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Farabaugh, S.M. The ecological and social significance of duetting. In Acoustic Communication in Birds; Kroodsma, D.E., Miller, E.H., Ouellet, H., Eds.; Academic Press: New York, NY, USA, 1982; pp. 85–124. [Google Scholar]
- Hall, M. A review of vocal duetting in birds. Adv. Stud. Behav. 2009, 40, 66–121. [Google Scholar]
- Mann, N.I.; Dingess, K.A.; Barker, F.K.; Graves, J.A.; Slater, P.J.B. A comparative study of song form and duetting in neotropical Thryothorus wrens. Behaviour 2009, 146, 1–43. [Google Scholar] [CrossRef]
- Logue, D.M.; Krupp, D.B. Duetting as a collective behavior. Front. Ecol. Evol. 2016, 4, 7. [Google Scholar] [CrossRef]
- Rivera-Cáceres, K.D.; Quirós-Gerrero, E.; Araya-Salas, M.; Templeton, C.N.; Searcy, W.A. Early development of vocal interaction rules in a duetting songbird. R. Soc. Open Sci. 2018, 5, 171791. [Google Scholar] [CrossRef] [PubMed]
- Wickler, W. Vocal dueting and the pair bond. I. Coyness and partner commitment. A hypothesis. Z. Tierpsychol. 1980, 52, 201–209. [Google Scholar] [CrossRef]
- Hall, M.L. A review of hypotheses for the functions of avian duetting. Behav. Ecol. Sociobiol. 2004, 55, 415–430. [Google Scholar] [CrossRef]
- Tilson, R.L.; Norton, P.M. Alarm duetting and pursuit deterrence in an African antelope. Am. Nat. 1981, 118, 455–462. [Google Scholar] [CrossRef]
- Moynihan, M. Communication in the titi monkey, Callicebus. J. Zool. 1966, 150, 77–127. [Google Scholar] [CrossRef]
- Robinson, J.G. An analysis of the organization of vocal communication in the titi monkey, Callicebus moloch. Z. Tierpsychol. 1979, 49, 381–403. [Google Scholar] [CrossRef] [PubMed]
- Deputte, B.L. Duetting in male and female songs of the white-cheeked gibbon (Hylobates concolor leucogenys). In Primate Communication; Snowdon, C.T., Brown, C.H., Petersen, M.R., Eds.; Cambridge University Press: Cambridge, UK, 1982; pp. 67–93. [Google Scholar]
- Haimoff, E.H. Convergence in the duetting of monogamous Old World primates. J. Hum. Evol. 1986, 15, 51–59. [Google Scholar] [CrossRef]
- Pollock, J.I. The song of the indris (Indri indri; Primates; Lemuroidea): Natural history, form, and function. Int. J. Primatol. 1986, 7, 225–264. [Google Scholar] [CrossRef]
- Nietsch, A. Duet vocalizations among different populations of Sulawesi tarsiers. Int. J. Primatol. 1999, 20, 567–583. [Google Scholar] [CrossRef]
- Geissmann, T. Duet-splitting and the evolution of gibbon songs. Biol. Rev. 2002, 77, 55–76. [Google Scholar] [CrossRef]
- Müller, A.E.; Anzenberger, G. Duetting in the titi monkey Callicebus cupreus: Structure, pair specificity and development of duets. Folia Primatol. 2002, 73, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Cárdenas, M.G.; Zimmermann, E. Duetting—A mechanism to strengthen pair bonds in a dispersed pair-living primate (Lepilemur edwardsi)? Am. J. Phys. Anthropol. 2009, 139, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Burton, J.A.; Nietsch, A. Geographical variation in duet songs of Sulawesi tarsiers: Evidence for new cryptic species in South and Southeast Sulawesi. Int. J. Primatol. 2010, 31, 1123–1146. [Google Scholar] [CrossRef]
- Petter, J.J.; Charles-Dominique, P. Vocal communication in prosimians. In The Study of Prosimian Behaviour; Doyle, G.A., Martin, R.D., Eds.; New York Academic Press: New York, NY, USA, 1979; pp. 272–282. [Google Scholar]
- Caselli, C.B.; Mennill, D.J.; Bicca-Marques, J.C.; Setz, E.Z.F. Vocal behavior of black-fronted titi monkeys (Callicebus nigrifrons): Acoustic properties and behavioral contexts of loud calls. Am. J. Primatol. 2014, 76, 788–800. [Google Scholar] [CrossRef] [PubMed]
- Snowdon, C.T. Vocal communication in family-living and pair-bonded primates. In Primate Hearing and Communication; Quam, R., Ramsier, M., Fay, R., Popper, A., Eds.; Springer Handbook of Auditory Research; Springer: Cham, Switzerland, 2017; Volume 63, pp. 141–174. [Google Scholar]
- Wright, P.C. Callicebus in Manu National Park: Territory, resources, scent marking and vocalizations. In Evolutionary Biology and Conservation of Titis, Sakis and Uacaris; Barnett, A.A., Veiga, L.M., Ferrari, S.F., Norconk, M.A., Eds.; Cambridge University Press: Cambridge, UK, 2013; pp. 232–239. [Google Scholar]
- Caselli, C.B.; Mennill, D.J.; Gestich, C.C.; Setz, E.Z.F.; Bicca-Marques, J.C. Playback responses of socially monogamous black-fronted titi monkeys to simulated solitary and paired intruders. Am. J. Primatol. 2015, 77, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Mason, W.A. Social organization of the South American monkey Callicebus moloch: A preliminary report. Tulane Stud. Zool. 1966, 13, 23–28. [Google Scholar]
- Anzenberger, G. Monogamous social systems and paternity in primates. In Paternity in Primates: Genetic Tests and Theories; Martin, R.D., Dixson, A.F., Wickings, E.J., Eds.; Karger: Basel, Switzerland, 1992; pp. 203–224. [Google Scholar]
- Fernandez-Duque, E.; Di Fiore, A.; de Luna, A.G. Pair-mate relationships and parenting in equatorial saki monkeys (Pithecia aequatorialis) and red titi monkeys (Callicebus discolor) of Ecuador. In Evolutionary Biology and Conservation of Titis, Sakis and Uacaris; Veiga, L.M., Barnett, A.A., Ferrari, S.F., Norconk, M.A., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 295–302. [Google Scholar]
- Van Belle, S.; Fernandez-Duque, E.; Di Fiore, A. Demography and life history of wild Red titi monkeys (Callicebus discolor) and Equatorial sakis (Pithecia aequatorialis) in Amazonian Ecuador: A 12-year study. Am. J. Primatol. 2016, 78, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Hershkovitz, P.A. Systematic and zoogeographic account of the monkeys of the genus Callicebus (Cebidae) of the Amazonas and Orinoco River basins. Mammalia 1963, 27, 1–80. [Google Scholar] [CrossRef]
- Hershkovitz, P. Titis, New World monkeys of the genus Callicebus (Cebidae, Platyrrhini): A preliminary taxonomic review. Fieldiana Zool. 1990, 55, 1–105. [Google Scholar]
- Kobayashi, S.A. A phylogenetic study of titi monkeys, genus Callicebus, based on cranial measurements: I. Phyletic groups of Callicebus. Primates 1995, 36, 101–120. [Google Scholar] [CrossRef]
- Van Roosmalen, M.G.M.; Van Roosmalen, T.; Mittermeier, R.A.A. Taxonomic review of the Titi monkeys genus Callicebus Thomas, 1903, with the description of two new species, Callicebus bernhardi and Callicebus Stephennashi from Brazilian Amazonia. Neotrop. Primates 2002, 10, 1–52. [Google Scholar]
- Auricchio, P.A. Morphological analysis of some species of Callicebus, Thomas, 1903 (Pitheciidae, Callicebinae). Neotrop. Primates 2010, 17, 47–58. [Google Scholar] [CrossRef]
- Serrano-Villavicencio, J.E.; Vendramel, R.L.; Siniciato Terra Garbino, G. Species, subspecies, or color morphs? Reconsidering the taxonomy of Callicebus Thomas, 1903 in the Purus-Madeira interfluvium. Primates 2016, 58, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Byrne, H.; Rylands, A.B.; Carneiro, J.C.; Lynch Alfaro, J.W.; Bertuol, F.; da Silva, M.N.F.; Messias, M.; Groves, C.P.; Mittermeier, R.A.; Farias, I.; et al. Phylogenetic relationships of the New World titi monkeys (Callicebus): First appraisal of taxonomy based on molecular evidence. Front Zool. 2016, 13, 10. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, J.; De Sousa e Silva, J., Jr.; Sampaio, I.; Pissinatti, A.; Hrbek, T.; Rezende Messias, M.; Röhe, F.; Farias, I.; Boubli, J.; Schneider, H. Phylogeny of the titi monkeys of the Callicebus moloch group (Pitheciidae, Primates). Am. J. Primatol. 2016, 78, 904–913. [Google Scholar] [CrossRef] [PubMed]
- Hoyos, M.; Bloor, P.; Defler, T.; Vermeer, J.; Röhe, F.; Farias, I. Phylogenetic relationships within the Callicebus cupreus species group (Pitheciidae; Primates): Biogeographic and taxonomic implications. Mol. Phyl. Evol. 2016, 102, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Green, A. The Phylogeography of Cheracebus Species Located in the Rio Negro Basin and Its Implications for Our Understanding of the Historical Biogeography of Amazonia. MSc Thesis, University of Salford, Salford, UK, 2017; 70p. [Google Scholar]
- Carneiro, J.; Sampaio, I.; De Sousa e Silva, J., Jr.; Farias, I.; Hrbek, T.; Pissinatti, A.; Silva, R.; Martins-Junior, A.; Boubli, J.; Ferrari, S.F.; et al. Phylogeny, molecular dating and zoogeographic history of the titi monkeys (Callicebus, Pitheciidae) of Eastern Brazil. Mol. Phyl. Gen. Evol. 2018, 124, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Langguth, A.A. New species of titi monkey, Callicebus Thomas, from North-Eastern Brazil (Primates, Cebidae). Revista Brasileira de Zoologia 1999, 16, 531–551. [Google Scholar] [CrossRef]
- Wallace, R.B.; Gómez, H.; Felton, A.; Felton, A.M. On a new species of Titi monkey, genus Callicebus Thomas (Primates, Pitheciidae), from Western Bolivia with preliminary notes on distribution and abundance. Primate Conserv. 2006, 20, 29–39. [Google Scholar] [CrossRef]
- Defler, T.R.; Bueno, M.L.; García, J. Callicebus caquetensis. A new and critically endangered Titi monkey from Southern Caquetá, Colombia. Primate Conserv. 2010, 25, 1–9. [Google Scholar] [CrossRef]
- Gualda-Barros, J.; Oliveira do Nacimento, F.; Kerr do Amaral, M.A. New species of Callicebus, Thomas, 1903 (Primates, Pitheciidae) from the states of Mato Grosso and Pará, Brazil. Pap. Avulsos Zool. (São Paulo) 2012, 52, 261–279. [Google Scholar] [CrossRef]
- Dalponte, J.C.; Silva, F.E.; de Sousa e Silva Júnior, J. New species of titi monkey, genus Callicebus Thomas, 1903 (Primates, Pitheciidae), from Southern Amazonia, Brazil. Pap. Avulsos Zool. (São Paulo) 2014, 54, 32. [Google Scholar]
- Vermeer, J.; Tello-Alvarado, J.C. The distribution and taxonomy of titi monkeys (Callicebus) in central and southern Peru, with the description of a new species. Primate Conserv. 2015, 29, 9–29. [Google Scholar] [CrossRef]
- Barros, R.M.S.; Pieczarka, J.C.; Do Carmo, O.; Obrigido, M.; Muniz, J.A.P.C.; Rodriguez, L.R.R.; Nagamachi, C.Y. A new karyotype in Callicebus torquatus (Cebidae, Primates). Hereditas 2000, 133, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Bonvicino, C.R.; Penna-Firme, V.; Nascimento, F.F.; Lemos, B.; Stanyon, R.; Seuánez, H.N. The lowest diploid number (2n = 16) yet found in any primate: Callicebus lugens (Humboldt, 1811). Folia Primatol. 2003, 74, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.R.R.; Cardozo De Souza, M.; Pieczarka, J.C.; Nagamachi, C.Y. Karyotypic study of Callicebus coimbrai: A rare and threatened primate species from Brazil. Caryologia 2006, 59, 248–252. [Google Scholar] [CrossRef]
- Bueno, M.L.; Defler, T.R. Aportes citogeneticos en el esclarecimiento de la taxonomia del genero Callicebus. Orinoquia 2010, 14 (Suppl. S1), 139–152. [Google Scholar]
- Snowdon, C.T.; Hodun, A.; Rosenberger, A.; Coimbra-Filho, A.F. Long call structure and its relation to taxonomy in lion tamarin. Am. J. Primatol. 1986, 11, 253–261. [Google Scholar] [CrossRef]
- Thinh, V.N.; Hallam, C.; Roos, C.; Hammerschmidt, K. Concordance between vocal and genetic diversity in crested gibbons. BMC Evol. Biol. 2011, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Garbino, G.S.T.; Martins-Junior, A.M.G. Phenotypic evolution in marmoset and tamarin monkeys (Cebidae, Callitrichinae) and a revised genus-level classification. Mol. Phyl. Evol. 2018, 118, 156–171. [Google Scholar] [CrossRef] [PubMed]
- Aldrich, B.C.; Shanee, S. Preliminary observations on the San Martin titi monkey, Plecturocebus oenanthe Thomas, 1924 (Mammalia: Primates: Pitheciidae) vocalizations at Tarangue, Peru. Neotrop. Primates 2016, 23, 21–26. [Google Scholar]
- Robinson, J.G. Vocal regulation of use of space by groups of titi monkeys Callicebus moloch. Behav. Ecol. Sociobiol. 1979, 5, 1–15. [Google Scholar] [CrossRef]
- Robinson, J.G. Vocal regulation of inter- and intragroup spacing during boundary encounters in the titi monkey, Callicebus moloch. Primates 1981, 22, 161–172. [Google Scholar] [CrossRef]
- Kinzey, W.G.; Rosenberger, A.L.; Heisler, P.S.; Prowse, D.L.; Trilling, J.S. A preliminary field investigation of the Yellow handed titi monkey, Callicebus torquatus torquatus, in Northern Peru. Primates 1977, 18, 159–181. [Google Scholar] [CrossRef]
- Kinzey, W.G.; Robinson, J.G. Intergroup loud calls, range size, and spacing in Callicebus torquatus. Am. J. Phys. Anthropol. 1983, 60, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Haimoff, E.H. The organization of song in the Hainan black gibbon (Hylobates concolor hainanus). Primates 1984, 25, 225–235. [Google Scholar] [CrossRef]
- Adret, P.; Cochran, J.S.; Suarez Roda, M. Airborne vs. radio-transmitted vocalizations in two primates: A technical report. Bioacoustics 2018, 27, 273–294. [Google Scholar] [CrossRef]
- Bezerra, B.; Cäsar, C.; Jerusalinsky, L.; Barnett, A.; Bastos, M.; Souto, A.; Jones, G. Pitheciid vocal communication: What can we say about what they are saying? Ethnobiol. Conserv. 2017, 6, 1–18. [Google Scholar] [CrossRef]
- Rowe, N.; Martinez, W. Callicebus sightings in Bolivia, Peru and Ecuador. Neotrop. Primates 2003, 11, 32–35. [Google Scholar]
- Felton, A.; Felton, A.M.; Wallace, R.B.; Gómez, H. Identification, behavioral observations, and notes on the distribution of the titi monkeys Callicebus modestus Lönnberg 1939 and Callicebus olallae Lönnberg 1939. Primate Conserv. 2006, 20, 41–46. [Google Scholar] [CrossRef]
- Martínez, J.; Wallace, R.B. Pitheciidae. In Distribución y Ecología de los Mamíferos Medianos y Grandes de Bolivia; Wallace, R.B., Gómez, H., Porcel, Z.R., Rumiz, D.I., Eds.; Centro de Ecología Difusión, Fundación Simón I. Patiño: La Paz, Bolivia, 2010; pp. 307–330. [Google Scholar]
- Martínez, J.; Wallace, R.B. Further notes on the distribution of endemic Bolivian titi monkeys, Callicebus modestus and Callicebus olallae. Neotrop. Primates 2007, 14, 47–54. [Google Scholar] [CrossRef]
- Martínez, J.; Wallace, R.B. New information about the distribution of Callicebus (Pithecidae, Primates) in northern Beni Department, Bolivia. Ecología en Bolivia 2013, 48, 57–62. [Google Scholar]
- Martínez, J.; Wallace, R.B. Ecological and Behavioural factors influencing territorial call rates for the Bolivian titi monkeys, Plecturocebus modestus and Plecturocebus olallae. Folia Primatol. 2017, 87, 279–290. [Google Scholar] [CrossRef] [PubMed]
- López-Strauss, H.E.; Wallace, R.B. Estimación de dos primates endémicos bolivianos, Callicebus olallae y Callicebus modestus. Mastozool. Neotrop. 2015, 22, 23–34. [Google Scholar]
- Rumíz, D.I. Distribution, habitat and status of the White-coated titi monkey (Callicebus pallescens) in the Chaco–Chiquitano forests of Santa Cruz, Bolivia. Neotrop. Primates 2013, 19, 8–15. [Google Scholar] [CrossRef]
- Rumíz, D.I. Los primates de Bolivia. Bolivia Ecológica 2013, 71, 1–36. [Google Scholar]
- Cäsar, C.; Byrne, R.W.; Young, R.J.; Zuberbühler, K. The alarm call system of wild black-fronted titi monkeys, Callicebus nigrifrons. Behav. Ecol. Sociobiol. 2012, 66, 653–667. [Google Scholar] [CrossRef]
- Yu, A.C.; Margoliash, D. Temporal hierarchichal control of singing in birds. Science 1996, 273, 1871–1875. [Google Scholar] [CrossRef] [PubMed]
- Sueur, J.; Aubin, T.; Simonis, C. Equipment review: Seewave, a free modular tool for sound analysis and synthesis. Bioacoustics 2008, 18, 213–226. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Fitch, T.W. Rhythmic cognition in humans and animals: Distinguishing meter and pulse perception. Front. Syst. Neurosci. 2013, 7, 68. [Google Scholar] [CrossRef] [PubMed]
- Adret, P. A Voice of Their Own: Titi Monkeys. 2012. Available online: https://www.youtube.com/watch?v=CkUCCyhv5Xc (accessed on 24 June 2012).
- Beck, M.W. ggord: Ordination Plots with ggplot2, R Package version 1.1.0; CRAN: Zurich, Switzerland, 2017. [Google Scholar]
- Verboom, W.C. Bird vocalizations: Three Great tit (Parus major) vocalizations. JunoBioacoustics 2018, 5, 1–6. [Google Scholar]
- Wallace, R.B.; Martinez, J.; López-Strauss, H.; Barreta, J.; Reinaga, A.; López, L. Conservation challenges facing two threatened endemic titi monkeys in a naturally fragmented Bolivian forest. In Primates in Fragments: Complexity and Resilience; Marsh, L.K., Chapman, C.A., Eds.; Springer Science: New York, NY, USA, 2013; pp. 493–501. [Google Scholar]
- Gautier, J.P. A redrawn phylogeny of guenons based upon their calls—Biogeographical implications. Bioacoustics 1989, 2, 11–21. [Google Scholar] [CrossRef]
- Brown, W.L.; Wilson, E.O. Character displacement. Syst. Zool. 1956, 5, 49–64. [Google Scholar] [CrossRef]
- Groves, C.P. Order Primates. In Mammal Species of the World: A Taxonomic and Geographic Reference; Wilson, D.E., Reeder, D.M., Eds.; Johns Hopkins University Press: Baltimore, MD, USA, 2005; Volume 1, pp. 111–184. [Google Scholar]
- Janik, V.M.; Slater, P.J.B. The different roles of social learning in vocal communication. Anim. Behav. 2000, 60, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Vermeer, J.; Tello-Alvarado, J.C.; Moreno-Moreno, S.; Guerra-Vásquez, F. Extension of the geographical range of white-browed titi monkeys (Callilcebus discolor) and evidence for sympatry with San Martin titi monkeys (Callicebus oenanthe). Int. J. Primatol. 2011, 32, 924–930. [Google Scholar] [CrossRef]
- Van Kuijk, S.M.; Garcia-Suikkanen, C.; Tello-Alvarado, J.C.; Vermeer, J.; Hill, C.M. Estimating population density of the San Martin titi monkey (Callicebus oenanthe) in Peru using vocalisations. Folia Primatol. 2015, 86, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Bóveda-Penalba, A.J.; Vermeer, J.; Rodrigo, F.; Geurra-Vásquez, F. Preliminary report on the distribution of Callicebus oenanthe on the eastern feet of the Andes. Int. J. Primatol. 2009, 30, 467–480. [Google Scholar] [CrossRef]
- Busnel, R.-G.; Classe, A. Whistled Languages; Springer Verlag: Berlin, Germany, 1976. [Google Scholar]
- Meyer, J. Bioacoustics of human whistled languages: An alternative approach to the cognitive processes of language. Ann. Acad. Brasileira de Ciências 2004, 76, 405–412. [Google Scholar] [CrossRef]
- De La Torre, S.; Snowdon, C.T. Dialects in pygmy marmosets? Population variation in call tructure. Am. J. Primatol. 2009, 71, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Snowdon, C.T. Cognitive components of vocal communication: A case study. Animals 2018, 8, 126. [Google Scholar] [CrossRef] [PubMed]
- De La Torre, S.; Snowdon, C.T. Environmental correlates of vocal communication of wild pygmy marmosets, Cebuella pygmaea. Anim. Behav. 2002, 63, 847–856. [Google Scholar] [CrossRef]
- Souza-Alves, J.P.; Ferrari, S.F. Responses of wild titi monkeys, Callicebus coimbrai (Primates: Platyrrhini: Pitheciidae), to the habituation process. Zoologia 2010, 27, 861–866. [Google Scholar] [CrossRef]
- Aldrich, B.C.; Molleson, L.; Nekaris, K.A.I. Vocalizations as a conservation tool: An auditory survey of the Andean titi monkey Callicebus oenanthe Thomas, 1924 (Mammalia: Primates: Pitheciidae) at Tarangue, Northern Peru. Contrib. Zool. 2008, 77, 1–6. [Google Scholar]
- Bezerra, B.M.; Souto, A.S.; Jones, G. Responses of golden-backed uakaris, Cacajao melanocephalus, to call playback: Implications for surveys in the flooded Igapó forest. Primates 2010, 51, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Gestich, C.C.; Caselli, C.B.; Nagy-Reis, M.B.; Setz, E.; Da Cunha, R.G.T. Estimating primate population densities: The systematic use of playbacks along transects in population surveys. Am. J. Primatol. 2017, 79, e22586. [Google Scholar] [CrossRef] [PubMed]
- Dacier, A.; de Luna, A.G.; Fernandez-Duque, E.; Di Fiore, A. Estimating population density of Amazonian titi monkeys (Callicebus discolor) via playback point counts. Biotropica 2011, 43, 135–140. [Google Scholar] [CrossRef]
- Lorenz, K. The Foundations of Ethology; Springer: New York, NY, USA, 1981. [Google Scholar]
- Tinbergen, N. On aims and methods of Ethology. Z. Tierpsychol. 1963, 20, 410–433. [Google Scholar] [CrossRef]
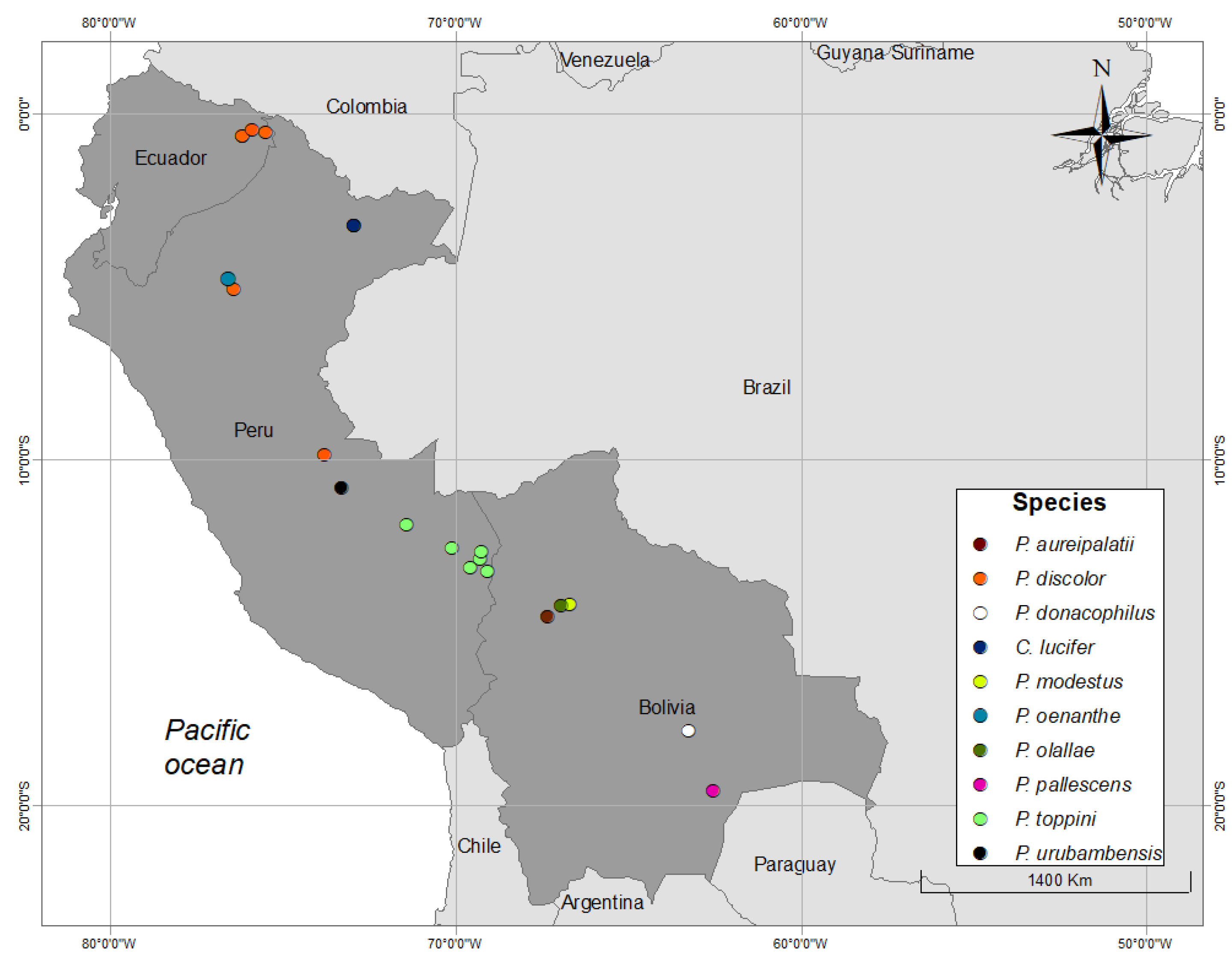
| ID | Species | Location | Country | Context | Recordist | Genus | Species * |
|---|---|---|---|---|---|---|---|
| 24308 | moloch | Madre de Dios | Peru | advertising call | T.A. Parker, III | P. | toppini |
| 30000 | moloch | Madre de Dios | Peru | response to playback | T.A. Parker, III | P. | toppini |
| 30709 | moloch | Loreto | Peru | advertising call | T.A. Parker, III | C. | lucifer |
| 39016 | moloch | Madre de Dios | Peru | advertising call | T.A. Parker, III | P. | toppini |
| 39551 | moloch | Madre de Dios | Peru | - | M.L. Isler | P. | toppini |
| 53419 | cupreus | Napo | Ecuador | - | L.F. Kibler | P. | discolor |
| 75934 | moloch | Madre de Dios | Peru | advertising call | C.A. Marantz | P. | toppini |
| 77771 | cupreus | Napo | Ecuador | advertising call | M.B. Robbins | P. | discolor |
| 126317 | moloch | Madre de Dios | Peru | advertising call | A.B. van den Berg | P. | toppini |
| 131447 | moloch | Madre de Dios | Peru | advertising call | J.H. Barry | P. | toppini |
| 148573 | cupreus | Orellana | Ecuador | - | G.F. Seeholzer | P. | discolor |
| 187992 | moloch | Loreto | Peru | advertising call | P.K. Donahue | C. | lucifer |
| 188515 | moloch | Loreto | Peru | troop chorus+ | P.K. Donahue | C. | lucifer |
| 188546 | moloch | Loreto | Peru | troop chorus++ | P.K. Donahue | C. | lucifer |
| 188610 | torquatus | Loreto | Peru | - | P.K. Donahue | C. | lucifer |
| 188624 | torquatus | Loreto | Peru | - | P.K. Donahue | C. | lucifer |
| Category | Call | Donacophilus | Moloch | Personatus | Torquatus | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| don10,11 | pal11 | ola11 | mod11 | oen7,11 | uru11 | dis1 | orn2,3 | cup1,4 | aur11 | top9,11 | nig5,6 | luc8,11 | ||
| R | pant | + | + | + | + | + | + | + | + | + | + | + | + | + |
| R | honk | + | + | + | + | + | + | + | + | + | + | + | + | |
| R | bellow | + | + | + | + | + | + | + | + | + | + | + | + | |
| P | pump | − | − | − | − | − | + | + | + | + | + | + | + | + |
| hoot | + | + | + | + | + | + | ||||||||
| arch * | + | + | + | + | + | |||||||||
| te * | + | + | + | + | ||||||||||
| rhythmic * | + | + | + | + | ||||||||||
| chirrup | + | + | + | + | + | + | + | + | + | + | ||||
| high-pitched | + | + | + | + | + | + | + | + | ||||||
| moan | + | + | + | + | + | + | ||||||||
| warble | + | |||||||||||||
| whistle | + | + | + | + | + | + | ||||||||
| squeak | + | + | + | |||||||||||
| chirp | + | + | ||||||||||||
| trill | + | + | + | + | ||||||||||
| cheep | + | + | ||||||||||||
| chuck | + | + | + | + | ||||||||||
| scream | + | + | + | + | ||||||||||
| grunt | + | + | ||||||||||||
| iii-yep * | − | − | − | − | + | − | − | − | − | − | − | − | − | |
| whine | − | − | − | − | + | − | − | − | − | − | − | − | − | |
| whinny | − | − | − | − | + | − | − | − | − | − | − | − | − | |
| pant-hoot | − | − | − | − | + | − | − | − | − | − | − | − | − | |
| SS | gobble | + | + | + | + | + | + | + | + | + | + | + | ||
| SS | CP | + | + | + | + | + | + | + | + | + | + | + | + | |
| SS | CR | + | ||||||||||||
| SS | CRP | + | ||||||||||||
| LS | hoot song | + | ||||||||||||
| LS | m. calling | + | + | + | + | + | + | + | + | + | + | + | + | + |
| LS | f. calling | + | ||||||||||||
| LS | duet | + | + | + | + | + | + | + | + | + | + | + | + | + |
| LS | chorus | + | + | + | + | + | + | + | + | + | + | + | + | + |
| Genus | Lineage | Species | #Groups | #Duets | #Choruses | Dur (s) | Media |
|---|---|---|---|---|---|---|---|
| P. | donacophilus | donacophilus ++ | 7 | 6 | 1 | 798 | v |
| pallescens | 2 | 2 | - | 283 | v | ||
| olallae | 2 | 2 | - | 230 | v | ||
| modestus | 1 | 1 | - | 173 | v | ||
| oenanthe * | 1 | 1 | - | 189 | a | ||
| uubambensis * | 1 | 1 | - | 75 | v | ||
| moloch | 14 | 13 | 1 | 1748 | |||
| toppini | 7 | 6 | 1 | 810 | a | ||
| discolor ++ | 8 | 6 | 2 | 801 | a/v | ||
| aureipalatii | 1 | 1 | - | 127 | a | ||
| 16 | 13 | 3 | 1738 | ||||
| C. | torquatus | lucifer ++ | 6 | 5 | 1 | 618 | a |
| Total | 36 | 31 | 5 | 4104 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adret, P.; Dingess, K.A.; Caselli, C.B.; Vermeer, J.; Martínez, J.; Luna Amancio, J.C.; Van Kuijk, S.M.; Hernani Lineros, L.M.; Wallace, R.B.; Fernandez-Duque, E.; et al. Duetting Patterns of Titi Monkeys (Primates, Pitheciidae: Callicebinae) and Relationships with Phylogeny. Animals 2018, 8, 178. https://doi.org/10.3390/ani8100178
Adret P, Dingess KA, Caselli CB, Vermeer J, Martínez J, Luna Amancio JC, Van Kuijk SM, Hernani Lineros LM, Wallace RB, Fernandez-Duque E, et al. Duetting Patterns of Titi Monkeys (Primates, Pitheciidae: Callicebinae) and Relationships with Phylogeny. Animals. 2018; 8(10):178. https://doi.org/10.3390/ani8100178
Chicago/Turabian StyleAdret, Patrice, Kimberly A. Dingess, Christini B. Caselli, Jan Vermeer, Jesus Martínez, Jossy C. Luna Amancio, Silvy M. Van Kuijk, Lucero M. Hernani Lineros, Robert B. Wallace, Eduardo Fernandez-Duque, and et al. 2018. "Duetting Patterns of Titi Monkeys (Primates, Pitheciidae: Callicebinae) and Relationships with Phylogeny" Animals 8, no. 10: 178. https://doi.org/10.3390/ani8100178
APA StyleAdret, P., Dingess, K. A., Caselli, C. B., Vermeer, J., Martínez, J., Luna Amancio, J. C., Van Kuijk, S. M., Hernani Lineros, L. M., Wallace, R. B., Fernandez-Duque, E., & Di Fiore, A. (2018). Duetting Patterns of Titi Monkeys (Primates, Pitheciidae: Callicebinae) and Relationships with Phylogeny. Animals, 8(10), 178. https://doi.org/10.3390/ani8100178