Investigating Foraging Niches for Tenrecs, Bats, and Rodents at Betampona Réserve Naturelle Intégrale (Central Eastern Madagascar) Using Stable Carbon and Nitrogen Isotopes in Fur and Bone
Simple Summary
Abstract
1. Introduction
1.1. Site Description
1.2. Mammal Species Overview
1.3. Background on Carbon and Nitrogen Isotopes
2. Methods
2.1. Sample Collection
2.2. Sample Preparation
2.3. Sample Analysis
2.4. Data Analysis
3. Results
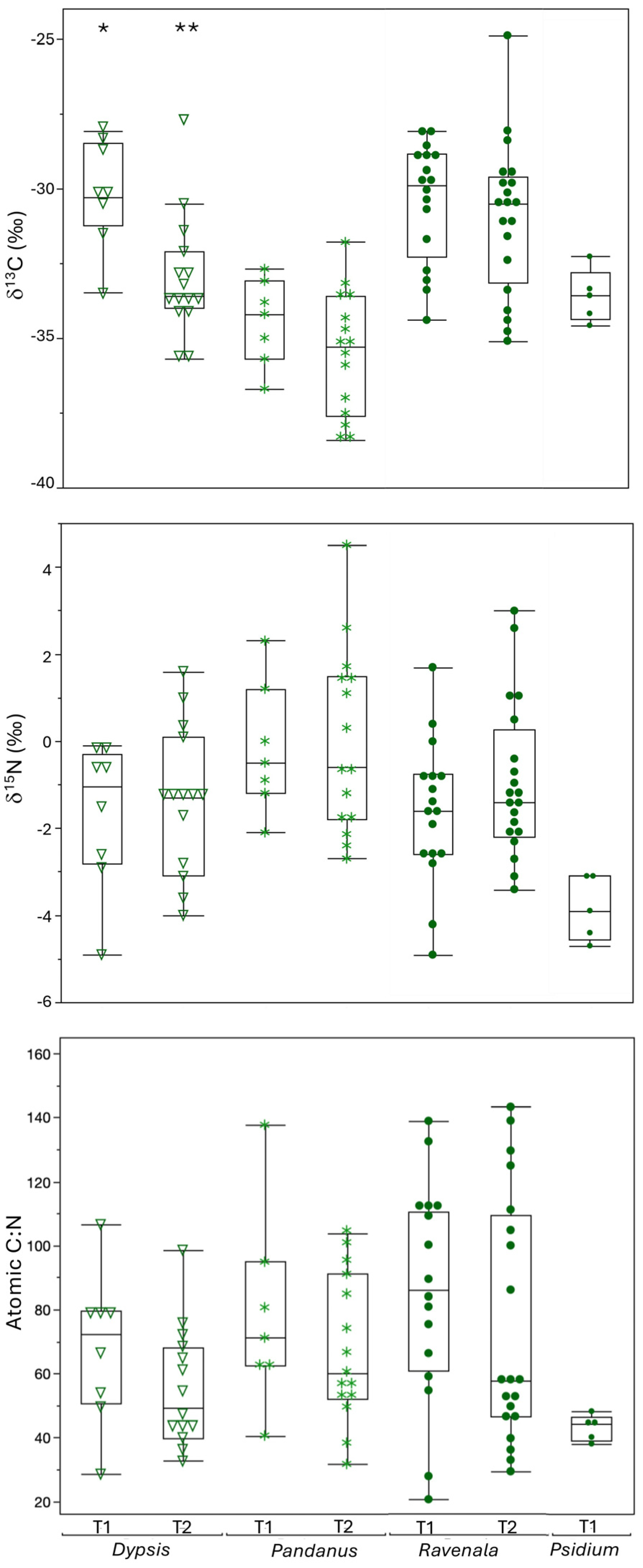
3.1. Trends for Vegetation
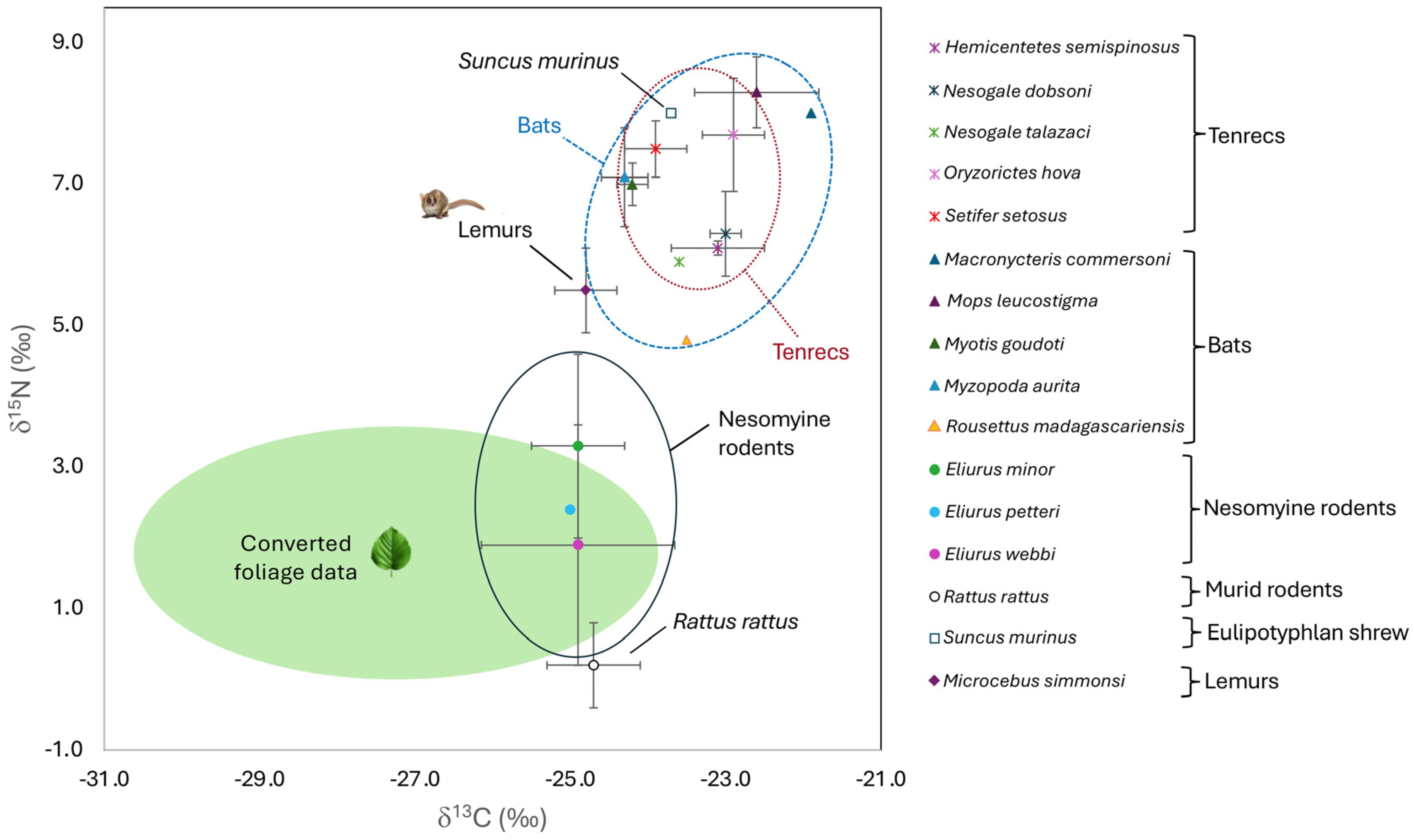
3.2. Small Mammals
4. Discussion
4.1. Plants
4.2. Small Mammals
4.2.1. Tenrecs
4.2.2. Bats
4.2.3. Endemic Rodents
4.2.4. Introduced Taxa
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goodman, S.M. The New Natural History of Madagascar; Princeton University Press: Princeton, NJ, USA, 2022. [Google Scholar]
- Crowley, B.E. A refined chronology of prehistoric Madagascar and the demise of the megafauna. Quat. Sci. Rev. 2010, 29, 2591–2603. [Google Scholar] [CrossRef]
- Ralimanana, H.; Perrigo, A.L.; Smith, R.J.; Borrell, J.S.; Faurby, S.; Rajaonah, M.T.; Randriamboavonjy, T.; Vorontsova, M.S.; Cooke, R.S.C.; Phelps, L.N.; et al. Madagascar’s extraordinary biodiversity: Threats and opportunities. Science 2022, 378, eadf1466. [Google Scholar] [CrossRef] [PubMed]
- Kremen, C.; Cameron, A.; Moilanen, A.; Phillips, J.; Thomas, C.D.; Beentje, H.; Dransfield, J.; Fisher, B.L.; Glaw, F.; Good, T.C.; et al. Aligning conservation priorities across taxa in Madagascar with high-resolution planning tools. Science 2008, 320, 222–226. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Neugarten, R.A.; Honzák, M.; Carret, P.; Koenig, K.; Andriamaro, L.; Cano, C.A.; Grantham, H.S.; Hole, D.; Juhn, D.; McKinnon, M.; et al. Rapid assessment of ecosystem service co-benefits of biodiversity priority areas in Madagascar. PLoS ONE 2016, 11, e0168575. [Google Scholar] [CrossRef]
- Carrasco, J.; Price, V.; Tulloch, V.; Mills, M. Selecting priority areas for the conservation of endemic trees species and their ecosystems in Madagascar considering both conservation value and vulnerability to human pressure. Biodivers. Conserv. 2020, 29, 1841–1854. [Google Scholar] [CrossRef]
- Rogers, H.M.; Glew, L.; Honzák, M.; Hudson, M.D. Prioritizing key biodiversity areas in Madagascar by including data on human pressure and ecosystem services. Landsc. Urban Plan. 2010, 96, 48–56. [Google Scholar] [CrossRef]
- Razafindratsima, O.H.; Ramananjato, V.; Randimbiarison, F.; Andriantsaralaza, S.; Rafaharetana, A.R.; Rabarijaonina, T.H.; Raoelinjanakolona, N.N.; Razafimandimby, D.N.; Nantenaina, R.H.; Raharinoro, N.A.; et al. 120 years of “Lemurology”: A qualitative review of what we have learned. Biotropica 2025, 57, e70047. [Google Scholar] [CrossRef]
- Mittermeier, R.A.; Reuter, K.E.; Rylands, A.B.; Louis, E.E.; Ratsimbazafy, J.; Rene de Roland, L.-A.; Langrand, O.; Schwitzer, C.; Johnson, S.E.; Godfrey, L.R.; et al. Lemurs of Madagascar, 5th ed.; Lynx Nature Books: Barcelona, Spain, 2023. [Google Scholar]
- Ben-David, M.; Flaherty, E.A. Stable isotopes in mammalian research: A beginner’s guide. J. Mammal. 2012, 93, 312–328. [Google Scholar] [CrossRef]
- Hixon, S.W.; Fernandes, R.; Andriamahaihavana, A.; Baden, A.L.; Blanco, M.B.; Caulier, G.; Dammhahn, M.; Eeckhaut, I.; Eppley, T.M.; Frédérich, B.; et al. Introducing IsoMad, a compilation of isotopic datasets for Madagascar. Sci. Data 2024, 11, 857. [Google Scholar] [CrossRef]
- Gautier, L.; Tahinarivony, J.A.; Ranirison, P.; Wohlhauser, S. Végétation/Vegetation. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Goodman, S.M., Raherilalao, M.J., Wohlhauser, S., Eds.; Association Vahatra: Antananarivo, Madagascar, 2018; pp. 207–242. [Google Scholar]
- Critical Ecosystem Partnership Fund. Ecosystem Profile. Madagascar and the Indian Ocean Islands Biodiversity Hotspot: 2022 Update; Arlington, VA, USA, 2022; p. 345. Available online: www.cepf.net/resources/ecosystem-profile-documents/madioprofile2022en (accessed on 20 May 2025).
- Goodman, S.M.; Raherilalao, M.J.; Wohlhauser, S. Site 35. Betampona. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Association Vahatra: Antananarivo, Madagascar, 2018; pp. 904–917. [Google Scholar]
- Goodman, S.M. A description of the ground burrow of Eliurus webbi (Nesomyinae) and case of cohabitation with an endemic bird (Brachypteraciidae, Brachypteracias). Mammalia 1994, 58, 670–672. [Google Scholar] [CrossRef]
- Goodman, S.M.; Raherilalao, M.J.; Ratsirarson, J.; Langrand, O. Atlas of Selected Land Vertebrates of Madagascar; Association Vahatra: Antananarivo, Madagascar, 2014. [Google Scholar]
- Goodman, S.M. Les Chauves-souris de Madagascar; Association Vahatra: Antananarivo, Madagascar, 2011; p. 129. [Google Scholar]
- Goodman, S.M.; Carleton, M.D. The rodents of the Réserve Naturelle Intégrale d’Andringitra, Madagascar. In A Floral and Faunal Inventory of the Réserve Naturelle Intégrale d’Andringitra, Madagascar: With Reference to Elevational Variation; Goodman, S.M., Ed.; Field Museum of Natural History: Chicago, IL, USA, 1996; Volume 85, pp. 257–283. [Google Scholar]
- Goodman, S.M.; Jenkins, P.D. The insectivores of the Réserve Spéciale d’Anjanaharibe-Sud, Madagascar. In A Floral and Faunal Inventory of the Eastern Slopes of the Réserve Spéciale d’Anjanaharibe-Sud, Madagascar: With Reference to Elevational Variation; Goodman, S.M., Ed.; Field Museum of Natural History: Chicago, IL, USA, 1998; Volume 90, pp. 139–161. [Google Scholar]
- Soarimalala, V.; Goodman, S.M. Les Petits Mammifères de Madagascar; Association Vahatra: Antananarivo, Madagascar, 2011. [Google Scholar]
- Soarimalala, V.; Goodman, S.M. The food habits of Lipotyphla. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; The University of Chicago Press: Chicago, IL, USA, 2003; pp. 1203–1205. [Google Scholar]
- Goodman, S.M. Oryzorictes, mole tenrec or rice tenrec. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 1278–1281. [Google Scholar]
- Stephenson, P.J. Hemicentetes; Streaked Tenrecs, Sora, Tsora. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 1281–1283. [Google Scholar]
- Monadjem, A.; Andriafidison, D.; Cardiff, S.G.; Goodman, S.; Hutson, A.M.; Jenkins, R.K.B.; Kofoky, A.; Racey, P.A.; Ranivo, J.; Ratrimomanarivo, F.H.; et al. Macronycteris commersoni. IUCN Red List of Threatened Species 2019; IUCN: Gland, Switzerland, 2019; p. e.T10120A22092860. [Google Scholar] [CrossRef]
- MacKinnon, J.L.; Hawkins, C.E.; Racey, P.A. Pteropodidae, Fruit Bats, Fanihy, Angavo. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 1299–1302. [Google Scholar]
- Ratrimomanarivo, F.H.; Goodman, S.M.; Hoosen, N.; Taylor, P.J.; Lamb, J.A. Morphological and molecular variation in Mops leucostigma (Chiroptera: Molossidae) of Madagascar and the Comoros: Phylogeny, phylogeography, and geographic variation. Mitteilungen Aus Dem Hambgurgischen Zool. Mus. 2008, 105, 57–101. [Google Scholar]
- Carleton, M.D. Eliurus, tufted-tail rats. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; The University of Chicago Press: Chicago, IL, USA, 2003; pp. 1373–1380. [Google Scholar]
- Eger, J.L.; Mitchell, L. Bats, Chiroptera. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 1287–1298. [Google Scholar]
- Garbutt, N. Mammals of Madagascar: A Complete Guide; Yale University Press: New Haven, CT, USA, 2007. [Google Scholar]
- Schliemann, H.; Goodman, S.M. Myzopoda aurita, Old World sucker-foot bat. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 1303–1306. [Google Scholar]
- Everson, K.M.; Soarimalala, V.; Goodman, S.M.; Olson, L.E. Multiple loci and complete taxonomic sampling resolve the phylogeny and biogeographic history of tenrecs (Mammalia: Tenrecidae) and reveal higher speciation rates in Madagascar’s humid forests. Syst. Biol. 2016, 65, 890–909. [Google Scholar] [CrossRef]
- Foley, N.M.; Goodman, S.M.; Whelan, C.V.; Puechmaille, S.J.; Teeling, E. Towards navigating the minotaur’s labyrinth: Cryptic diversity and taxonomic revision within the speciose genus Hipposideros (Hipposideridae). Acta Chiropterol. 2017, 19, 1–18. [Google Scholar] [CrossRef]
- Wohlhauser, S.; Armstrong, A.H. Site 35. Betampona, vegetation. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Goodman, S.M., Raherilalao, M.J., Wohlhauser, S., Eds.; Association Vahatra: Antananarivo, Madagascar, 2018; pp. 907–909. [Google Scholar]
- Mertz, E. The effects of horizontal and vertical environmental heterogeneity on lemur community structure: A landscape ecology approach. In The New Natural History of Madagascar; Goodman, S.M., Ed.; Princeton University Press: Princeton, NJ, USA, 2022; pp. 134–142. [Google Scholar] [CrossRef]
- Poudyal, M.; Ramamonjisoa, B.S.; Hockley, N.; Rakotonarivo, O.S.; Gibbons, J.M.; Mandimbiniaina, R.; Rasoamanana, A.; Jones, J.P. Can REDD+ social safeguards reach the ‘right’people? Lessons from Madagascar. Glob. Environ. Change 2016, 37, 31–42. [Google Scholar] [CrossRef]
- Andreone, F.; Rosa, G.M.; Noël, J.; Crottini, A.; Vences, M.; Raxworthy, C.J. Living within fallen palm leaves: The discovery of an unknown Blommersia (Mantellidae: Anura) reveals a new reproductive strategy in the amphibians of Madagascar. Naturwissenschaften 2010, 97, 525–543. [Google Scholar] [CrossRef]
- Pabijan, M.; Gehring, P.S.; Koehler, J.; Glaw, F.; Vences, M. A new microendemic frog species of the genus Blommerisia (Anura: Mantellidae) from the east coast of Madagascar. Zootaxa 2011, 2978, 34–50. [Google Scholar] [CrossRef]
- Piccoli, C.; Paoletti, A.; Andreone, F.; Freeman, K.; Harris, G.D.J.; Lava, N.; Melo, M.; Nöel, J.; Ramamonjisoa, D.L.; Randriamamialisoa; et al. The amphibians of Betampona: An updated account of the remarkable diversity of Madagascar’s central-eastern Strict Nature Reserve. Syst. Biodivers. 2025, 23, 2505180. [Google Scholar] [CrossRef]
- Ranivo Rakotoson, J.; Razafimahatratra, P.M. Legal, judicial, and financial aspects of Madagascar’s protected areas: Past, present, and future. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Goodman, S.M., Raherilalao, M.J., Wohlhauser, S., Eds.; Association Vahatra: Antananarivo, Madagascar, 2018; pp. 105–165. [Google Scholar]
- Fenton, M.B.; Bogdanowicz, W. Relationship between external morphology and foraging behaviour: Bats in the genus Myotis. Can. J. Zool. 2002, 80, 1004–1013. [Google Scholar] [CrossRef]
- Rakotoarivelo, A.A.; Ranaivoson, N.; Ramilijaona, O.R.; Kofoky, A.F.; Racey, P.A.; Jenkins, R.K. Seasonal food habits of five sympatric forest microchiropterans in western Madagascar. J. Mammal. 2007, 88, 959–966. [Google Scholar] [CrossRef]
- Ramasindrazana, B.; Rajemison, B.; Goodman, S.M. The diet of the endemic bat Myzopoda aurita (Myzopodidae) based on fecal analysis. Afr. J. Ecol. 2009, 45, 645–660. [Google Scholar]
- O’Leary, M.H. Carbon isotopes in photosynthesis. BioScience 1988, 38, 328–336. [Google Scholar] [CrossRef]
- von Caemmerer, S.; Ghannoum, O.; Pengelly, J.J.; Cousins, A.B. Carbon isotope discrimination as a tool to explore C4 photosynthesis. J. Exp. Bot. 2014, 65, 3459–3470. [Google Scholar] [CrossRef] [PubMed]
- Kohn, M.J. Carbon isotope compositions of terrestrial C3 plants as indicators of (paleo)ecology and (paleo)climate. Proc. Natl. Acad. Sci. USA 2010, 107, 19691–19695. [Google Scholar] [CrossRef] [PubMed]
- Cerling, T.E.; Hart, J.A.; Hart, T.B. Isotope ecology in the Ituri forest. Oecologia 2004, 138, 5–12. [Google Scholar] [CrossRef]
- Kapos, V.; Ganade, G.; Matsui, E.; Victoria, R.L. δ13C as an indicator of edge effects in tropical rainforest reserves. J. Ecol. 1993, 81, 425–432. [Google Scholar] [CrossRef]
- Heaton, T.H.E. Spatial, species, and temporal variations in the 13C/12C ratios of C3 plants: Implications for palaeodiet studies. J. Archaeol. Sci. 1999, 26, 637–649. [Google Scholar] [CrossRef]
- Handley, L.L.; Austin, A.T.; Robinson, D.; Scrimgeour, C.M.; Raven, J.A.; Heaton, T.H.E.; Schmidt, S.; Stewart, G.R. The 15N natural abundance of δ15N of ecosystem samples reflects measures of water availability. Aust. J. Plant Physiol. 1999, 26, 185–199. [Google Scholar] [CrossRef]
- Evans, R.D. Physiological mechanisms influencing plant nitrogen isotope composition. Trends Plant Sci. 2001, 6, 121–126. [Google Scholar] [CrossRef]
- Craine, J.M.; Elmore, A.J.; Aider, M.P.M.; Bustamente, M.; Dawson, T.E.; Hobbie, E.A.; Kahmen, A.; Mack, M.C.; McLauchlan, K.K.; Michelsen, A.; et al. Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol. 2009, 183, 980–992. [Google Scholar] [CrossRef]
- Lowry, B.E.; Wittig, R.M.; Pittermann, J.; Oelze, V.M. Stratigraphy of stable isotope ratios and leaf structure within an African rainforest canopy with implications for primate isotope ecology. Sci. Rep. 2021, 11, 14222. [Google Scholar] [CrossRef] [PubMed]
- Guerrieri, R.; Templer, P.; Magnani, F. Canopy exchange and modification of nitrogen fluxes in forest ecosystems. Curr. For. Rep. 2021, 7, 115–137. [Google Scholar] [CrossRef]
- Crowley, B.E.; McGoogan, K.C.; Lehman, S.M. Edge effects on foliar stable isotope values in a Madagascan tropical dry forest. PLoS ONE 2012, 7, e44538. [Google Scholar] [CrossRef] [PubMed]
- Remy, E.; Wuyts, K.; Boeckx, P.; Gundersen, P.; Verheyen, K. Edge effects in temperate forests subjected to high nitrogen deposition. Proc. Natl. Acad. Sci. USA 2017, 114, E7032. [Google Scholar] [CrossRef]
- Remy, E.; Wuyts, K.; Boeckx, P.; Ginzburg, S.; Gundersen, P.; Demey, A.; Van Den Bulcke, J.; Van Acker, J.; Verheyen, K. Strong gradients in nitrogen and carbon stocks at temperate forest edges. For. Ecol. Manag. 2016, 376, 45–58. [Google Scholar] [CrossRef]
- Weintraub, S.R.; Taylor, P.G.; Porder, S.; Cleveland, C.C.; Asner, G.P.; Townsend, A.R. Topographic controls on soil nitrogen availability in a lowland tropical forest. Ecology 2015, 96, 1561–1574. [Google Scholar] [CrossRef]
- Reinmann, A.B.; Hutyra, L.R. Reply to Remy et al.: Local and global limitations to forest productivity as mediators of biogeochemical response to forest edge effects. Proc. Natl. Acad. Sci. USA 2017, 114, E7033–E7034. [Google Scholar] [CrossRef]
- Weathers, K.C.; Cadenasso, M.L.; Pickett, S.T. Forest edges as nutrient and pollutant concentrators: Potential synergisms between fragmentation, forest canopies, and the atmosphere. Conserv. Biol. 2001, 15, 1506–1514. [Google Scholar] [CrossRef]
- Vanneste, T.; Depauw, L.; De Lombaerde, E.; Meeussen, C.; Govaert, S.; De Pauw, K.; Sanczuk, P.; Bollmann, K.; Brunet, J.; Calders, K.; et al. Trade-offs in biodiversity and ecosystem services between edges and interiors in European forests. Nat. Ecol. Evol. 2024, 8, 880–887. [Google Scholar] [CrossRef]
- Codron, J.; Codron, D.; Lee-Thorp, J.A.; Sponheimer, M.; Bond, W.J.; de Ruiter, D.; Grant, R. Taxonomic, anatomical, and spatio-temporal variations in the stable carbon and nitrogen isotopic compositions of plants from an African savanna. J. Archaeol. Sci. 2005, 32, 1757–1772. [Google Scholar] [CrossRef]
- Crowley, B.E.; Blanco, M.B.; Arrigo-Nelson, S.J.; Irwin, M.T. Stable isotopes document resource partitioning and differential response to forest disturbance in sympatric cheirogaleid lemurs. Naturwissenschaften 2013, 100, 943–956. [Google Scholar] [CrossRef]
- Carlson, B.A.; Crowley, B.E. Variation in carbon isotope values among chimpanzee foods at Ngogo, Kibale National Park and Bwindi Impenetrable National Park, Uganda. Am. J. Primatol. 2016, 78, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Wessling, E.G.; Oelze, V.M.; Eshuis, H.; Pruetz, J.D.; Kühl, H.S. Stable isotope variation in savanna chimpanzees (Pan troglodytes verus) indicate avoidance of energetic challenges through dietary compensation at the limits of the range. Am. J. Phys. Anthropol. 2019, 168, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Cernusak, L.A.; Tcherkez, G.; Keitel, C.; Cornwell, W.K.; Santiago, L.S.; Knohl, A.; Barbour, M.M.; Williams, D.G.; ReichI, P.B.; Ellsworth, D.S.; et al. Why are non-photosynthetic tissues generally 13C enriched compared with leaves in C3 plants? Review and synthesis of current hypotheses. Funct. Plant Biol. 2009, 36, 199–213. [Google Scholar] [CrossRef]
- Vanderklift, M.A.; Ponsard, S. Sources of variation in consumer-diet δ15N enrichment: A meta-analysis. Oecologia 2003, 136, 169–182. [Google Scholar] [CrossRef]
- Ambrose, S.H.; Butler, B.M.; Hanson, D.B.; Hunter-Anderson, R.L.; Krueger, H.W. Stable isotopic analysis of human diet in the Marianas Archipelago, Western Pacific. Am. J. Phys. Anthropol. 1997, 104, 343–361. [Google Scholar] [CrossRef]
- Ambrose, S.H. Effects of diet, climate and physiology on nitrogen isotope abundances in terrestrial foodwebs. J. Archaeol. Sci. 1991, 18, 293–317. [Google Scholar] [CrossRef]
- Crowley, B.E. Stable isotope techniques and applications for primatologists. Int. J. Primatol. 2012, 33, 673–701. [Google Scholar] [CrossRef]
- Stephens, R.B.; Shipley, O.N.; Moll, R.J. Meta-analysis and critical review of trophic discrimination factors (Δ13C and Δ15N): Importance of tissue, trophic level and diet source. Funct. Ecol. 2023, 37, 2535–2548. [Google Scholar] [CrossRef]
- Caut, S.; Angulo, E.; Courchamp, F. Variation in discrimination factors (Δ15N and Δ13C): The effect of diet isotopic values and applications for diet reconstruction. J. Appl. Ecol. 2009, 46, 443–453. [Google Scholar] [CrossRef]
- Crowley, B.E.; Carter, M.L.; Karpanty, S.M.; Zihlman, A.L.; Koch, P.L.; Dominy, N.J. Stable carbon and nitrogen isotope enrichment in primate tissues. Oecologia 2010, 164, 611–626. [Google Scholar] [CrossRef]
- Symes, C.T.; Wilson, J.W.; Woodborne, S.M.; Shaikh, Z.S.; Scantlebury, M. Resource partitioning of sympatric small mammals in an African forest-grassland vegetation mosaic. Austral Ecol. 2013, 38, 721–729. [Google Scholar] [CrossRef]
- Crowley, B.E.; Castro, I.; Soarimalala, V.; Goodman, S.M. Isotopic evidence for niche partitioning and the influence of anthropogenic disturbance on endemic and introduced rodents in central Madagascar. Sci. Nat. 2018, 105, 44. [Google Scholar] [CrossRef] [PubMed]
- Krigbaum, J.; Berger, M.H.; Daegling, D.J.; McGraw, W.S. Stable isotope canopy effects for sympatric monkeys at Taï Forest, Côte d’Ivoire. Biol. Lett. 2013, 9, 20130466. [Google Scholar] [CrossRef] [PubMed]
- Balčiauskas, L.; Skipitytė, R.; Balčiauskienė, L.; Jasiulionis, M. Resource partitioning confirmed by isotopic signatures allows small mammals to share seasonally flooded meadows. Ecol. Evol. 2019, 9, 5479–5489. [Google Scholar] [CrossRef] [PubMed]
- Dransfield, J.; Beentje, H.J. The Palms of Madagascar; International Palms Society and The Royal Botanic Gardens: Kew London, UK, 1995; p. 475. [Google Scholar]
- Callmander, M.W.; Laivo, M.O.; Buerki, S. Pandanaceae, screw-pines, vakoa, hofa, frandra. In The New Natural History of Madagascar; Goodman, S.M., Ed.; Princeton University Press: Princeton, NJ, USA, 2022; pp. 551–559. [Google Scholar]
- Haevermans, T.; Hladik, A.; Hladik, C.M.; Rzanatsoa, J.; Haevermans, A.; Jeannoda, V.; Blanc, P. Description of five new species of the Madagascan flagship plant genus Ravenala (Strelitziaceae). Sci. Rep. 2021, 11, 21965. [Google Scholar] [CrossRef]
- Soarimalala, V.; Rasolobera, F.; Goodman, S.M. Diversité et distribution des petits mammifères terrestres dans le Parc National de Marojejy, Nord-est de Madagascar: Gradient altitudinal et variation temporelle. In: Goodman, S.M., Raherilalao, M.R., editors. A floral and faunal inventory of the Parc National de Marojejy: Altitudinal gradient and temporal variation. Malagasy Nat. 2023, 17, 211–228. [Google Scholar]
- Steadman, D.W.; DiAntonio, L.L.; Wilson, J.J.; Sheridan, K.E.; Tammariello, S.P. The effects of chemical and heat maceration techniques on the recovery of nuclear and mitochondrial DNA from bone. J. Forensic Sci. 2006, 51, 11–17. [Google Scholar] [CrossRef]
- Skrzypek, G. Normalization procedures and reference material selection in stable HCNOS isotope analyses: An overview. Anal. Bioanal. Chem. 2013, 405, 2815–2823. [Google Scholar] [CrossRef]
- Gannes, L.Z.; Martinez del Rio, C.; Koch, P.L. Natural abundance variations in stable isotopes and their potential uses in animal physiological ecology. Comp. Biochem. Physiol. 1998, 119A, 725–757. [Google Scholar] [CrossRef]
- Racey, P.A.; Stephenson, P.J. Reproductive and energetic differentiation of the Tenrecidae of Madagascar. In Biogeography de Madagascar; Lourenço, W.R., Ed.; ORSTOM Editions: Paris, France, 1996; pp. 307–319. [Google Scholar]
- Andrianaivoarivelo, A.R.; Ranaivoson, N.; Racey, P.A.; Jenkins, R.K. The diet of three synanthropic bats (Chiroptera: Molossidae) from eastern Madagascar. Acta Chiropterol. 2006, 82, 439–444. [Google Scholar] [CrossRef]
- Olson, L.E. Tenrecs. Curr. Biol. 2013, 23, R5–R8. [Google Scholar] [CrossRef] [PubMed]
- Reher, S.; Rabarison, H.; Schoroth, M.; Dausmann, K. Seasonal movements of insectivorous bat species in southwestern Madagascar. Malagasy Nat. 2019, 13, 117–124. [Google Scholar]
- Reher, S.; Rabarison, H.; Montero, B.K.; Turner, J.M.; Dausmann, K.H. Disparate roost sites drive intraspecific physiological variation in a Malagasy bat. Oecologia 2022, 198, 35–52. [Google Scholar] [CrossRef]
- Vololona, J.; Ramavovololona, P.; Noroalintseheno Lalarivoniaina, O.S.; Goodman, S.M. Fleurs visiteés par Rousettus madagascariensis. G. Grandidier, 1928 (Chiroptera: Pteropodidae) dans la Réserve Spéciale d’Ankarana, Madagascar. Bull. Société Zool. Fr. 2020, 145, 49–67. [Google Scholar]
- Kjeldgaard, M.K.; Hewlett, J.A.; Eubanks, M.D. Widespread variation in stable isotope trophic position estimates: Patterns, causes, and potential consequences. Ecol. Monogr. 2021, 91, e01451. [Google Scholar] [CrossRef]
- Tieszen, L.L.; Fagre, T. Effect of diet quality and composition on the isotopic composition of respiratory CO2, bone collagen, bioapatite, and soft tissues. In Prehistoric Human Bone: Prehistoric Human Bone: Archaeology at the Molecular Level; Lambert, J.B., Grupe, G., Eds.; Springer: Berlin/Heidelberg, Germany, 1993; pp. 121–155. [Google Scholar] [CrossRef]
- Crowley, B.E.; Thorén, S.; Rasoazanabary, E.; Vogel, E.R.; Barrett, M.A.; Zohdy, S.; Blanco, M.B.; McGoogan, K.C.; Arrigo-Nelson, S.J.; Irwin, M.T.; et al. Explaining geographical variation in the isotope composition of mouse lemurs (Microcebus). J. Biogeogr. 2011, 38, 2106–2121. [Google Scholar] [CrossRef]
- Stone, B.C.; Huynh, K.; Poppendieck, H. Pandanaceae. In Flowering Plants Monocotyledons. The Families and Genera of Vascular Plants, vol 3; Kubitzki, K., Ed.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 397–404. [Google Scholar] [CrossRef]
- Andersson, L. Strelitziaceae. In Flowering Plants Monocotyledons. The Families and Genera of Vascular Plants, vol 4; Kubitzki, K., Ed.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 451–455. [Google Scholar] [CrossRef]
- Seubert, E. Root anatomy of palms IV. Arecoideae, part 1, general remarks and descriptions on the roots. Feddes Repert. 1998, 109, 89–127. [Google Scholar] [CrossRef]
- Camargo, J.L.; Kapos, V. Complex edge effects on soil moisture and microclimate in central Amazonian forest. J. Trop. Ecol. 1995, 11, 205–211. [Google Scholar] [CrossRef]
- Armstrong, A.H.; McGroddy, M. Site 35, Betampoma, soils. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Goodman, S.M., Raherilalao, M.J., Wohlhauser, S., Eds.; Association Vahatra: Antananarivo, Madagascar, 2018; p. 906. [Google Scholar]
- Crowley, B.E.; Sparks, J.M. Site 35, Betampona, geology. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Goodman, S.M., Raherilalao, M.J., Wohlhauser, S., Eds.; Association Vahatra: Antananarivo, Madagascar, 2018; p. 906. [Google Scholar]
- Austin, A.T.; Vitousek, P.M. Nutrient dynamics on a precipitation gradient in Hawai’i. Oecologia 1998, 113, 519–529. [Google Scholar] [CrossRef]
- Ometto, J.P.H.B.; Ehleringer, J.R.; Domingues, T.F.; Berry, J.A.; Ishida, F.Y.; Mazzi, E.; Higuchi, N.; Flanagan, L.B.; Nardoto, G.B.; Martinelli, L.A. The stable carbon and nitrogen isotopic composition of vegetation in tropical forests of the Amazon Basin, Brazil. Biogeochemistry 2006, 79, 251–274. [Google Scholar] [CrossRef]
- Nardoto, G.B.; Ometto, J.P.H.B.; Ehleringer, J.R.; Higuchi, N.; Bustamante, M.M.C.; Martinelli, L.A. Understanding the influences of spatial patterns on N availability within the Brazilian Amazon forest. Ecosystems 2008, 11, 1234–1246. [Google Scholar] [CrossRef]
- Treseder, K.K. Nitrogen additions and microbial biomass: A meta-analysis of ecosystem studies. Ecol. Lett. 2008, 11, 1111–1120. [Google Scholar] [CrossRef]
- Bejarano-Castillo, M.; Campo, J.; Roa-Fuentes, L.L. Effects of increased nitrogen availability on C and N cycles in tropical forests: A meta-analysis. PLoS ONE 2015, 10, e0144253. [Google Scholar] [CrossRef]
- Martinelli, L.A.; Piccolo, M.C.; Townsend, A.R.; Vitousek, P.M.; Cuevas, E.; McDowell, W.; Robertson, G.P.; Santos, O.C.; Treseder, K. Nitrogen stable isotopic composition of leaves and soil: Tropical versus temperate forests. Biogeochemistry 1999, 46, 45–65. [Google Scholar] [CrossRef]
- Hedin, L.O.; Brookshire, E.J.; Menge, D.N.; Barron, A.R. The nitrogen paradox in tropical forest ecosystems. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 613–635. [Google Scholar] [CrossRef]
- Morreale, L.L.; Thompson, J.R.; Tang, X.; Reinmann, A.B.; Hutyra, L.R. Elevated growth and biomass along temperate forest edges. Nat. Commun. 2021, 12, 7181. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, J.F.; Gould, E. The tenrecs: A study in mammalian behavior and evolution. Smithson. Contrib. Zool. 1970, 27, 1–137. [Google Scholar] [CrossRef]
- Jenkins, P.D.; Olson, L.E.; Everson, K.M.; Soarimalala, V.; Goodman, S.M. Tenrecidae: Microgale and Nesogale, shrew tenrecs. In The New Natural History of Madagascar; Goodman, S.M., Ed.; Princeton University Press: Princeton, NJ, USA, 2022; pp. 1880–1997. [Google Scholar]
- Hyodo, F.; Matsumoto, T.; Takematsu, Y.; Kamoi, T.; Fukuda, D.; Nakagawa, M.; Itioka, T. The structure of a food web in a tropical rain forest in Malaysia based on carbon and nitrogen stable isotope ratios. J. Trop. Ecol. 2010, 26, 205–214. [Google Scholar] [CrossRef]
- Fox-Dobbs, K.; Bump, J.; Peterson, R.; Fox, D.; Koch, P. Carnivore-specific stable isotope variables and variation in the foraging ecology of modern and ancient wolf populations: Case studies from Isle Royale, Minnesota, and La Brea. Can. J. Zool. 2007, 85, 458–471. [Google Scholar] [CrossRef]
- Hyodo, F.; Kohzu, A.; Tayasu, I. Linking aboveground and belowground food webs through carbon and nitrogen stable isotope analyses. Ecol. Res. 2010, 25, 745–756. [Google Scholar] [CrossRef]
- Briones, M.J.I.; Bol, R.; Sleep, D.; Sampedro, L.; Allen, D. A dynamic study of earthworm feeding ecology using stable isotopes. Rapid Comm. Mass Spectrom. 1999, 13, 1300–1304. [Google Scholar] [CrossRef]
- Hsu, G.C.; Szlavecz, K.; Csuzdi, C.; Bernard, M.; Chang, C.H. Ecological groups and isotopic niches of earthworms. Appl. Soil Ecol. 2023, 181, 104655. [Google Scholar] [CrossRef]
- Jenkins, S.G.; Partridge, T.R.; Farley, S.D.; Robbins, C.T. Nitrogen and carbon isotope fractionation between mothers, neonates, and nursing offspring. Oecologia 2001, 129, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.N.; Richter, M.M.; Williams, C.T.; Tøien, Ø.; Barnes, B.M.; O’Brien, D.M.; Buck, C.L. Stable isotope analysis of CO2 in breath indicates metabolic fuel shifts in torpid arctic ground squirrels. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2017, 209, 10–15. [Google Scholar] [CrossRef]
- Lee, T.N.; Buck, C.L.; Barnes, B.M.; O’Brien, D.M. A test of alternative models for increased tissue nitrogen isotope ratios during fasting in hibernating arctic ground squirrels. J. Exp. Biol. 2012, 215, 3354–3361. [Google Scholar] [CrossRef]
- Schmid, J.; Stephenson, P.J. Physiological adaptations in Malagasy mammals: Lemurs and tenrecs compared. In The Natural History of Madagascar; Goodman, S.M., Benstead, J.P., Eds.; The University of Chicago Press: Chicago, IL, USA, 2003; pp. 1198–1203. [Google Scholar]
- Levesque, D.L.; Lovasoa, O.M.A.; Rakotoharimalala, S.N.; Lovegrove, B.G. High mortality and annual fecundity in a free-ranging basal placental mammal, Setifer setosus (Tenrecidae: Afrosoricida). J. Zool. 2013, 291, 205–212. [Google Scholar] [CrossRef]
- Dammhahn, M.; Soarimalala, V.; Goodman, S.M. Trophic niche differentiation and microhabitat utilization in a species-rich montane forest small mammal community of eastern Madagascar. Biotropica 2013, 45, 111–118. [Google Scholar] [CrossRef]
- Goodman, S.M.; Louis, E.E., Jr.; Raselimanana, A.M. Site 35, Betampona, fauna. In Les Aires Protégées Terrestres de Madagascar: Leur Histoire, Description et Biote/The Terrestrial Protected Areas of Madagascar: Their History, Description, and Biota; Goodman, S.M., Raherilalao, M.J., Wohlhauser, S., Eds.; Association Vahatra: Antananarivo, Madagascar, 2018; pp. 911–913. [Google Scholar]
- Dammhahn, M.; Rakotondramanana, C.F.; Goodman, S.M. Coexistence of morphologically similar bats (Vespertilionidae) on Madagascar: Stable isotopes reveal fine-grained niche differentiation among cryptic species. J. Trop. Ecol. 2015, 31, 153–164. [Google Scholar] [CrossRef]
- Andrianaivoarivelo, R.A.; Jenkins, R.K.; Petit, E.J.; Ramilijaona, O.; Razafindrakoto, N.; Racey, P.A. Rousettus madagascariensis (Chiroptera: Pteropodidae) shows a preference for native and commercially unimportant fruits. Endanger. Species Res. 2012, 19, 19–27. [Google Scholar] [CrossRef]
- Vololona, J.; Ramavovololona, P.; Goodman, S.M. Régime alimentaire de Rousettus madagascariensis (Chiroptera: Pteropodidae) dans la Réserve Spéciale d’Ankarana, nord de Madagascar. Bull. Société Zool. Fr. 2020, 145, 365–381. [Google Scholar]
- Ralisata, M.; Andriamboavonjy, F.R.; Rakotondravony, D.; Ravoahangimalala, O.R.; Randrianandrianina, F.H.; Racey, P.A. Monastic Myzopoda: The foraging and roosting ecology of a sexually segregated Malagasy endemic bat. J. Zool. 2010, 282, 130–139. [Google Scholar] [CrossRef]
- Jokimäki, J.; Huhta, E.; Itämies, J.; Rahko, P. Distribution of arthropods in relation to forest patch size, edge, and stand characteristics. Can. J. For. Res. 1998, 28, 1068–1072. [Google Scholar] [CrossRef]
- Adamski, Z.; Bufo, S.A.; Chowański, S.; Falabella, P.; Lubawy, J.; Marciniak, P.; Pacholska-Bogalska, J.; Salvia, R.; Scrano, L.; Słocińska, M.; et al. Beetles as model organisms in physiological, biomedical and environmental studies-–A review. Front. Physiol. 2019, 10, 319. [Google Scholar] [CrossRef]
- Růžičková, J.; Elek, Z. Beetles on the move: Not-just-a-technical review of beetles’ radio-tracking. Entomol. Exp. Appl. 2023, 171, 82–93. [Google Scholar] [CrossRef]
- Brack, V., Jr.; LaVal, R.K. Diet of the gray myotis (Myotis grisescens): Variability and consistency, opportunism, and selectivity. J. Mammal. 2006, 87, 7–18. [Google Scholar] [CrossRef]
- Vesterinen, E.J.; Ruokolainen, L.; Wahlberg, N.; Peña, C.; Roslin, T.; Laine, V.N.; Vasko, V.; Sääksjärvi, I.E.; Norrdahl, K.; Lilley, T.M. What you need is what you eat? Prey selection by the bat Myotis daubentonii. Mol. Ecol. 2016, 25, 1581–1594. [Google Scholar] [CrossRef]
- Gamboa Alurralde, S.; Díaz, M.M. Feeding habits of four species of Myotis (Mammalia, Chiroptera) from Argentina. Mammal. Res. 2019, 64, 511–518. [Google Scholar] [CrossRef]
- Todilahy, L.; Rajemison, B.; Goodman, S.M. Régime alimentaire de Mops leucostigma (Chiroptera: Molossidae) dans la Station Forestière d’Ivoloina, Centre-est de Madagascar. Malagasy Nat. 2022, 16, 127–133. [Google Scholar]
- Reuter, K.E.; Wills, A.R.; Lee, R.W.; Cordes, E.E.; Sewall, B.J. Using stable isotopes to infer the impacts of habitat change on the diets and vertical stratification of frugivorous bats in Madagascar. PLoS ONE 2016, 11, e0153192. [Google Scholar] [CrossRef]
- Dammhahn, M.; Goodman, S.M. Trophic niche differentiation and microhabitat utilization revealed by stable isotope analyses in a dry-forest bat assemblage at Ankarana, northern Madagascar. J. Trop. Ecol. 2014, 30, 97–109. [Google Scholar] [CrossRef]
- Carleton, M.D. Systematic studies of Madagascar’s endemic rodents (Muroidea: Nesomyinae): Revision of the genus Eliurus. Am. Mus. Novit. 1994, 3087, 1–55. [Google Scholar]
- Nakagawa, M.; Hyodo, F.; Nakashizuka, T. Effect of forest use on trophic levels of small mammals: An analysis using stable isotopes. Can. J. Zool. 2007, 85, 472–478. [Google Scholar] [CrossRef]
- Goodman, S.M. Rattus on Madagascar: The dilemma of protecting the endemic rodent fauna. Conserv. Biol. 1995, 9, 450–453. [Google Scholar] [CrossRef]
- Miljutin, A.; Lehtonen, J.T. Probability of competition between introduced and native rodents in Madagascar: An estimation based on morphological traits. Est. J. Ecol. 2008, 57, 133–152. [Google Scholar] [CrossRef]
- Heim de Balsac, H. Insectivores. In Biogeography and Ecology in Madagascar; Battisitini, R., Richard-Vindard, G., Eds.; Dr. W. Junk, B.V.: The Hague, The Netherlands, 1972; pp. 629–660. [Google Scholar]
- Dammhahn, M.; Randriamoria, T.M.; Goodman, S.M. Broad and flexible stable isotope niches in invasive non-native Rattus spp. in anthropogenic and natural habitats of central eastern Madagascar. BMC Ecol. 2017, 17, 16. [Google Scholar] [CrossRef]



| Order | Family/ Subfamily | Genus and Species | Mass (in Grams) Average (Range) No. of Individuals | Details | Habit | Habitat | Other Notes |
|---|---|---|---|---|---|---|---|
| Afrosoricida | Tenrecinae | Hemicentetes semispinosus (E) | 133.9 (103–184) N = 14 | Entomophage *** Soft invertebrates in leaf litter, primarily annelid worms. | Terrestrial (fossorial) | Primary and degraded moist forest, agricultural areas, and villages in eastern Madagascar from sea level up to 1550 masl. | Excavate burrows up to 15 cm below ground level. Social, sometimes living in family groups > 20 individuals. |
| Tenrecinae | Setifer setosus (E) | 247.0 (230–280) N = 5 | Omnivore Annelid worms, Orthoptera, ants, fruits, carrion, mollusks. | Primarily terrestrial but also scansorial | Widespread in all forest types, including degraded forest and open habitats from sea level up to 2250 masl. | Tends to be solitary. Mostly forages on ground; sleeps underground in short tunnels it digs. | |
| Oryzorictinae | Nesogale dobsoni * (E) | 25.5 (30.5–30.0) N = 7 | Carnivore Invertebrates (Orthoptera and occasionally annelids), frogs, and sometimes smaller Microgale species. | Terrestrial to scansorial | Intact and disturbed moist evergreen forest, as well as agricultural areas and other non-forested areas from sea level up to 2500 masl. | Captive observations similar to Nesogale talazaci. Mostly thought to forage in leaf litter but scansorial behavior supported by a few individuals that were trapped 1–2.5 m above the ground. Thought to be solitary but nest in male/female pairs in captivity. Unique among tenrecs in ability to seasonally store fat reserves in the tail. | |
| Oryzorictinae | Nesogale talazaci * (E) | 37.4 (25.0–45.5) N = 18 | Carnivore Small vertebrates like frogs as well as arthropods. | Primarily terrestrial | Low altitude to montane moist evergreen intact and disturbed forest (from ca. 800 to 2300 masl). | Primarily forages in leaf litter, under fallen branches, and among roots in captivity. Thought to be solitary but nest in male/female pairs in captivity. Can be spatially clumped within forests. | |
| Oryzorictinae | Oryzorictes hova (E) | 42.5 (30.0–15.5) N = 35 | Entomophage *** Grasshoppers and soil invertebrates, especially annelid worms. | Terrestrial (fossorial) | Moist and sclerophyllous forest, swamps, forest, secondary vegetation and rice paddies near forests from sea level to 1990 masl. | Virtually nothing known about diet. What we do know is based on stomach contents and feeding in captivity. | |
| Chiroptera | Hipposideridae | Macronycteris commersoni ** (E) | Males 73.2 (51.5–98.0) N = 13 Females: 45.1 (39.5–50.0) N = 5 | Entomophage *** Large variety of insects but predominantly Coleoptera. Suspected to eat small frogs in southeastern Madagascar, but this has not been directly verified. | Volant | Widespread in both intact and degraded forest across Madagascar. Found in the ecotone between forest and agricultural areas. | This is the largest insectivorous species on Madagascar. |
| Vespertillonidae | Myotis goudoti (E) | 6.0 (4.2–9.2) N = 37 | Entomophage *** Varies among studies; Coleoptera, Isoptera, Lepidoptera, Hymenoptera, and Araneae all noted, and in western Madagascar, relative importance of different groups may shift seasonally. | Volant | Widespread in a variety of habitats from sea level to 1600 masl, including intact and degraded low elevation and montane forest, open grassy areas, and agricultural regions. | May roost with other bats (particularly Minopterus). Tends to be active earlier in the evening than other bats. | |
| Pteropodidae | Rousettus madagascariensis (E) | Males: 61.1 (49.0–87.0) N = 36 Females: 52.0 (30.5–77.0) N = 32 | Herbivore (frugivore) Known to forage on the fruits of native and introduced trees; an important seed disperser. | Volant | Broadly associated with forests and caves. Widespread in multiple forest types as well as agricultural areas, but absent from vast treeless areas. | Likely an important pollinator for Ravenala. | |
| Myzopodidae | Myzopoda aurita (E) | 8.3 (6.7–10.5) N = 22 | Entomophage *** Primarily Lepidoptera as well as some Coleoptera and cockroaches. | Volant | Widespread in a variety of habitats ranging from intact to degraded and fragmented forest, marsh, agricultural areas, and rice paddies at low to mid elevations in eastern Madagascar. | Has unique “horseshoe-shaped” adhesive disks on hands and feet. Appears closely tied to places where Ravenala grows. | |
| Molossidae | Mops leucostigma | Males: 22.5 (17.0–28.0) N = 275 Females: 20.1 (16.0–24.0) N = 102 | Entomophage *** Coleoptera, Hemiptera, Lepidoptera, and Diptera. | Volant | Occurs on Madagascar and neighboring islands. Widespread in intact and degraded forest, ecotones, agricultural, and even urban areas that are near forest with the exceptions of the extreme south, and elevations > 1200 m in central Madagascar. | ||
| Eulipotyphla | Soricidae | Suncus murinus | 29.3 (17.0–45.5) N = 19 | Omnivore Small mammals, arthropods, plant material. | Terrestrial and fossorial | Introduced and widespread in nearly every environment, including anthropogenic settings. | |
| Rodentia | Nesomyidae | Eliurus minor (E) | 35.1 (21.5–49.5) N = 25 | Frugivore and granivore Diet presumed based on behavior for other Eliurus taxa. | Primarily arboreal, but also terrestrial | Moist evergreen forest from sea level up to 1875 masl. Potentially tolerant of some disturbance. | |
| Eliurus petteri (E) | 74.0 N = 1 | Frugivore, insectivore, and granivore Diet presumed. Weak incisors suggest less reliance on hard seeds than other Eliurus species. | Primarily terrestrial and perhaps partially arboreal | Restricted to moist forest in central Eastern Madagascar from ca. 400 to 1000 masl. | Not thought to live in sympatry with other Eliurus species, but other species present in localities near those where E. petteri found. | ||
| Eliurus webbi (E) | 70.1 (54.0–90.0) N = 19 | Granivore (and some frugivory and insectivory) Gnaws holes in shells to extract endocarp. Frugivory and insectivory inferred based on behavior for other Eliurus taxa. | Primarily arboreal, but also terrestrial | Intact and degraded low elevation to montane evergreen moist forest in eastern Madagascar, mostly from sea level to 800 masl, but occasionally up to ca. 1500 masl. | Observed to store seeds in its burrows. Granivory inferred based on gnaw holes in seeds stored in burrows. Frugivory and insectivory presumed, based on behavior for other Eliurus species. | ||
| Muridae | Rattus rattus | 105.7 (86.0–134) N = 10 **** | Omnivore and granivore Eats a wide variety of foods. | Scansorial | Widespread globally. Introduced to Madagascar and broadly distributed, from sea level up to ca. 2500 masl in both anthropogenic and natural settings. |
| Transect | Taxon | δ13C (‰) | δ15N (‰) | Atomic C:N | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Mean ± 1σ | Median | Min | Max | N | Mean ± 1σ | Median | Min | Max | N | Mean ± 1σ | Median | Min | Max | |||
| Transect 1 | Dypsis | Total | 8 | −30.2 ± 1.8 | −30.3 ab | −33.5 | −28.1 | 8 | −1.7 ± 1.7 | −1.1 ab | −4.9 | −0.1 | 8 | 67.8 ± 23.7 | 72.4 a | 28.6 | 106.6 |
| Edge (<900 m) | 2 | −30.1 ± 2.0 | −30.1 | −31.5 | −28.7 | 2 | −1.5 ± 2.0 | −1.5 | −2.9 | −0.1 | 53.5 ± 35.1 | 53.5 | 28.6 | 78.3 | |||
| Interior (>900 m) | 6 | −30.2 ± 1.9 | −30.3 | −33.5 | −28.1 | 6 | −1.7 ± 1.8 | −1.1 | −4.9 | −0.2 | 72.6 ± 20.8 | 72.9 | 49.6 | 106.6 | |||
| Edge vs. Interior comparison | χ2 = 0.11, df = 1, p = 0.74 | χ2 = 0.11, df = 1, p = 0.74 | χ2 = 1.0, df = 1, p = 0.32 | ||||||||||||||
| Pandanus | Total | 7 | −34.5 ± 1.4 | −34.2 c | −36.7 | −32.7 | 7 | −0.2 ± 1.5 | −0.5 a | −2.1 | 2.3 | 7 | 78.7 ± 31.0 | 71.2 a | 40.6 | 137.7 | |
| Interior (>900 m) | 7 | −34.5 ± 1.4 | −34.2 | −36.7 | −32.7 | 7 | −0.2 ± 1.5 | −0.5 | −2.1 | 2.3 | 7 | 78.7 ± 31.0 | 71.2 | 40.6 | 137.7 | ||
| Ravenala | Total | 16 | −30.2 ± 1.8 | −30.3 b | −33.5 | −28.1 | 16 | −1.8 ± 1.6 | −1.6 a | −4.9 | 1.7 | 16 | 85.6 ± 34.1 | 85.8 a | 20.5 | 138.8 | |
| Edge (<900 m) | 5 | −29.3 ± 0.7 | −29.0 | −30.3 | −32.3 | 5 | −2.8 ± 1.8 | −2.6 | −4.9 | −0.7 | 5 | 103.9 ± 24.2 | 110.4 | 66.2 | 132.4 | ||
| Interior (>900 m) | 11 | −31.2 ± 2.1 | −30.7 | −34.4 | −28.1 | 11 | −1.3 ± 1.3 | −1.5 | −2.8 | 1.7 | 11 | 77.2 ± 35.6 | 80.8 | 20.5 | 138.8 | ||
| Edge vs. Interior comparison | χ2 = 2.70, df = 1, p = 0.10 | χ2 = 1.86, df = 3, p = 0.17 | χ2 = 2.0, df = 1, p = 0.16 | ||||||||||||||
| Psidium | Total | 5 | −33.6 ± 0.9 | −33.6 abc | −34.6 | −32.3 | 5 | −3.8 ± 0.7 | −3.9 b | −4.7 | −3.1 | 5 | 43.2 ± 4.0 | 44.5 a | 38.1 | 48.2 | |
| Edge (<900 m) | 5 | −33.6 ± 0.9 | −33.6 | −34.6 | −32.3 | 5 | −3.8 ± 0.7 | −3.9 | −4.7 | −3.1 | 5 | 43.2 ± 4.0 | 44.5 | 38.1 | 48.2 | ||
| T1 Genus comparison | χ2 = 17.50, df = 3, p = 0.0005 | χ2 = 12.55, df = 3, p = 0.0057 | χ2 = 9.18, df = 3, p = 0.027 | ||||||||||||||
| Levene p = 0.27 | Levene p = 0.53 | Levene p = 0.068 | |||||||||||||||
| Transect 2 | Dypsis | Total | 15 | −33.0 ± 2.0 | −33.6 a | −35.7 | −27.7 | 14 * | −1.3 ± 1.7 | −1.25 | −4.0 | 1.6 | 14 * | 54.8 ± 18.8 | 49.2 a | 32.7 | 98.6 |
| Edge (<900 m) | 4 | −34.3 ± 1.0 | −34.0 | −35.7 | −33.6 | 4 | −1.8 ± 1.3 | −1.5 | −2.8 | 1.7 | 4 | 48 ± 15.7 | 43.0 | 35.2 | 70.8 | ||
| Interior (>900 m) | 11 | −32.5 ± 2.1 | −33.0 | −35.5 | −27.7 | 10 * | −1.1 ± 1.9 | −1.3 | −4.0 | 1.6 | 10 * | 57.5 ± 20.0 | 57.9 | 32.7 | 98.6 | ||
| Edge vs. Interior comparison | χ2 = 4.1, df = 1, p = 0.043 | χ2 = 0.18, df = 1, p = 0.67 | χ2 = 0.32, df = 1, p = 0.57 | ||||||||||||||
| Pandanus | Total | 15 | −35.5 ± 2.0 | −35.3 b | −38.4 | −31.8 | 15 | −0.01 ± 2.1 | −0.6 | −2.7 | 4.5 | 15 | 67.6 ± 22.9 | 60.2 a | 31.7 | 103.7 | |
| Edge (<900 m) | 3 | −36.8 ± 1.5 | −37.0 | −38.2 | −35.3 | 3 | −1.1 ± 2.0 | −1.8 | −2.7 | 1.1 | 3 | 59.5 ± 6.2 | 56.1 | 55.7 | 66.7 | ||
| Interior (>900 m) | 12 | −35.1 ± 2.0 | −34.9 | −38.4 | −31.8 | 12 | 0.3 ± 2.1 | −0.2 | −2.4 | 4.5 | 12 | 69.6 ± 25.3 | 67.2 | 31.7 | 103.7 | ||
| Edge vs. Interior comparison | χ2 = 1.69, df = 1, p = 0.19 | χ2 = 1.69, df = 1, p = 0.19 | χ2 = 0.083, df = 1, p = 0.77 | ||||||||||||||
| Ravenala | Total | 20 | −31.0 ± 2.5 | −30.5 a | −35.1 | −24.9 | 20 | −0.9 ± 1.8 | −1.4 | −3.4 | 3.0 | 20 | 74.8 ± 38.4 | 57.7 a | 29.3 | 143.2 | |
| Edge (<900 m) | 7 | −29.6 ± 2.5 | −29.9 | −32.4 | −24.9 | 7 | −1.5 ± 1.0 | −1.5 | −2.7 | 0.5 | 7 | 94.2 ± 45.7 | 111.1 | 36.1 | 143.2 | ||
| Interior (>900 m) | 13 | −31.8 ± 2.3 | −31.0 | −35.1 | −28.4 | 13 | −0.6 ± 2.0 | −1.0 | −3.4 | 3.0 | 13 | 64.3 ± 30.8 | 53.0 | 29.3 | 129.5 | ||
| Edge vs. Interior comparison | χ2 = 2.77, df = 1, p = 0.096 | χ2 = 0.98, df = 1, p = 0.32 | χ2 = 2.39, df = 1, p = 0.12 | ||||||||||||||
| T2 Genus Comparison | χ2 = 20.75, df = 2, p < 0.0001 | χ2 = 2.8, df = 2, p = 0.25 | χ2 = 2.26, df = 2, p = 0.32 | ||||||||||||||
| Levene p = 0.64 | Levene p = 0.39 | Levene p = 0.0003 | |||||||||||||||
| Order | Species | δ13C (‰) | δ15N (‰) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Mean | ± 1σ | Median | Min | Max | Mean | ± 1σ | Median | Min | Max | ||
| Afrosoricida | 18; ♂ = 13; ♀ = 5 | −23.2 | 0.5 | −23.1 1 | −24.2 | −22.4 | 7.1 | 0.9 | 7.1 1 | 5.8 | 8.5 | |
| Hemicentetes semispinosus | 3; ♂ = 3 | −23.1 | 0.6 | −23.1 a | −23.6 | −22.5 | 6.1 | 0.1 | 6.1 a | 6.0 | 6.2 | |
| Nesogale dobsoni | 2; ♂ = 2 | −23.0 | 0.2 | −23 a | −23.1 | −22.8 | 6.3 | 0.6 | 6.3 a | 5.8 | 6.7 | |
| Nesogale talazaci | 1; ♀ = 1 | −23.6 | −23.6 a | −23.6 | −23.6 | 5.9 | 5.9 a | 5.9 | 5.9 | |||
| Oryzorictes hova | 9; ♂ = 5; ♀ = 4 | −22.9 | 0.4 | −22.8 a | −23.7 | −22.4 | 0.7 | 0.8 | 8.0 a | 6.6 | 8.5 | |
| Setifer setosus | 3; ♂ = 3 | −23.9 | 0.4 | −24.1 a | −24.2 | −23.5 | 7.5 | 0.4 | 7.5 a | 7.2 | 7.9 | |
| Species comparison: | χ2 = 7.16, df = 4, p = 0.13 | χ2 = 10.45, df = 4, p = 0.033 | ||||||||||
| Levene p = 0.42 | Levene p = 0.13 | |||||||||||
| Excluding Neogale talazaci: | χ2 = 5.21, df = 3, p = 0.16 | χ2 = 5.21, df = 3, p = 0.030 | ||||||||||
| Levene p = 0.74 | Levene p = 0.049 | |||||||||||
| Chiroptera | 27; ♂ = 19; ♀ = 8 | −23.8 | 0.9 | −24.2 2 | −24.7 | −21.0 | 7.2 | 0.9 | 7.2 1 | 4.8 | 8.8 | |
| Macronycteris commersoni | 1; ♀ = 1 | −21.9 | −21.9 ab | −21.9 | −21.9 | 8.0 | 8.0 ab | 8.0 | 8.0 | |||
| Mops leucostigma | 6; ♂ = 3; ♀ = 3 | −22.6 | 0.8 | −22.9 a | −23.3 | −21.0 | 8.3 | 0.5 | 8.5 a | 7.5 | 8.8 | |
| Myotis goudoti | 10; ♂ = 6; ♀ = 4 | −24.2 | 0.2 | −24.2 b | −24.5 | −23.9 | 7.0 | 0.3 | 7.0 b | 6.4 | 7.3 | |
| Myzopoda aurita | 9; ♂ = 9 | −24.3 | 0.3 | −24.4 b | −24.7 | −23.6 | 7.1 | 0.7 | 7.1 ab | 6.2 | 8.1 | |
| Rousettus madagascariensis | 1; ♂ = 1 | −23.5 | −23.5 ab | −23.5 | −23.5 | 4.8 | 4.8 ab | 4.8 | 4.8 | |||
| Species comparison: | χ2 = 18.05, df = 4, p = 0.0012 | χ2 = 14.67, df = 4, p = 0.0054 | ||||||||||
| Levene p = 0.020 | Levene p = 0.043 | |||||||||||
| Excluding Macronycteris commersoni and Rousettus madagascariensis: | χ2 = 14.84, df = 2, p = 0.0006 | χ2 = 11.48, df = 2, p = 0.0032 | ||||||||||
| Levene p = 0.020 | Levene p = 0.043 | |||||||||||
| Excluding Mops leucostigma: | χ2 = 7.50, df = 3, p = 0.058 | χ2 = 4.77, df =3, p = 0.19 | ||||||||||
| Levene p = 0.68 | Levene p = 0.016 | |||||||||||
| Eulipotyphla | Suncus murinus | 1; ♂ = 1 | −23.7 | −23.7 123 | −23.7 | −23.7 | 8.0 | 8.0 12 | 8.0 | 8.0 | ||
| Rodentia | 14; ♂ = 6; ♀ = 8 | −24.9 | 0.4 | −24.9 3 | −25.7 | −24.1 | 2.1 | 1.7 | 2.0 2 | −0.5 | 4.6 | |
| Eliurus minor | 5; ♂ = 2; ♀ = 3 | −24.9 | 0.6 | −24.7 a | −25.7 | −24.3 | 3.3 | 1.3 | 3.5 a | 1.9 | 4.6 | |
| Eliurus petteri | 1; ♀ = 1 | −25.0 | −25.0 a | −25.0 | −25.0 | 2.4 | 2.4 a | 2.4 | 2.4 | |||
| Eliurus webbi | 5; ♂ = 2; ♀ = 3 | −24.9 | 0.3 | −2.05 a | −25.3 | −24.5 | 1.9 | 1.7 | 1.4 a | −0.2 | 4.3 | |
| Rattus rattus | 3; ♂ = 2; ♀ = 1 | −24.7 | 0.6 | −24.7 a | −25.3 | −24.1 | 0.2 | 0.6 | 0.5 a | −0.5 | 0.5 | |
| Species comparison: | χ2 = 0.40, df = 3, p = 0.94 | χ2 = 6.98, df = 3, p = 0.073 | ||||||||||
| Levene p = 0.53 | Levene p = 0.25 | |||||||||||
| Excluding Eliurus petteri: | χ2 = 0,32 df = 2, p = 0.85 | χ2 = 4.77, df =3, p = 0.034 | ||||||||||
| Levene p = 0.53 | Levene p = 0.25 | |||||||||||
| Order comparisons: | χ2 = 32.83, df = 3, p < 0.0001 | χ2 = 32.37, df = 3, p < 0.0001 | ||||||||||
| Levene p = 0.034 | Levene p = 0.0042 | |||||||||||
| Excluding Suncus murinus: | χ2 = 32.76, df = 2, p < 0.001 | χ2 = 4.77, df =3, p < 0.0001 | ||||||||||
| Levene p = 0.022 | Levene p = 0.0042 | |||||||||||
| Excluding Mops leucostigma: | χ2 = 36.60, df = 3, p < 0.0001 | χ2 = 31.46, df = 3, p < 0.0001 | ||||||||||
| Levene p = 0.82 | Levene p = 0.0006 | |||||||||||
| Order | δ13C (‰) | δ15N (‰) | |||||
|---|---|---|---|---|---|---|---|
| N | Mean ± 1σ | Median | N | Mean ± 1σ | Median | ||
| Region comparisons | |||||||
| Afrosoricida | Forest interior | 18 | −23.1 ± 0.5 | −23.1 | 18 | 7.2 ± 0.9 | 7.1 |
| Chiroptera | Forest interior | 4 | −24.3 ± 0.3 | −24.3 b | 4 | 6.9 ± 0.3 | 7.1 b |
| Forest edge | 17 | −24.1 ± 0.7 | −24.2 b | 17 | 6.9 ± 0.8 | 6.9 b | |
| Outside of forest | 6 | −24.3 ± 0.3 | −22.9 a | 6 | 8.3 ± 0.5 | 8.5 a | |
| χ2 = 11.67, df = 2, p = 0.0029 * | χ2 = 11.16, df = 2, p = 0.0038 * | ||||||
| Levene p = 0.38 | Levene p = 0.35 | ||||||
| Eulipotyphla | Forest interior | 1 | −23.7 | −23.7 | 1 | 8.0 | 8.0 |
| Rodentia | Forest interior | 11 | −25.0 ± 0.4 | −25.1 | 11 | 2.3 ± 1.6 | 2.0 |
| Forest edge | 3 | −24.5 ± 0.3 | −24.7 | 3 | 1.2 ± 2.1 | 0.5 | |
| χ2 = 2.23, df = 1, p = 0.14 | χ2 = 1.19, df = 1, p = 0.27 | ||||||
| Levene p = 0.66 | Levene p = 0.63 | ||||||
| Habitat comparisons | |||||||
| Afrosoricida | Undisturbed moist forest | 10 | −23.2 ± 0.5 | −23.1 | 10 | 7.2 ± 1.0 | 7.5 |
| Slightly degraded moist forest | 8 | −23.0 ± 0.5 | −23.0 | 8 | 7.1 ± 1.0 | 6.9 | |
| χ2 = 0.80, df = 1, p = 0.37 | χ2 = 0.071, df = 1, p = 0.79 | ||||||
| Levene p = 0.97 | Levene p = 0.76 | ||||||
| Chiroptera | Slightly degraded moist forest | 4 | −24.3 ± 0.3 | −24.3 b | 4 | 6.9 ± 0.3 | 7.1 b |
| Second-growth forest | 17 | −24.1 ± 0.7 | −24.2 b | 17 | 6.9 ± 0.8 | 6.9 b | |
| Anthropogenic habitat | 6 | −24.3 ± 0.3 | −22.9 a | 6 | 8.3 ± 0.5 | 8.5 a | |
| χ2 = 11.67, df = 2, p = 0.0029 * | χ2 = 11.16, df = 2, p = 0.0038 * | ||||||
| Levene p = 0.38 | Levene p = 0.35 | ||||||
| Eulipotyphla | Slightly degraded moist forest | 1 | −23.7 | −23.7 | 1 | 8.0 | 8.0 |
| Rodentia | Undisturbed moist forest | 5 | −25.1 ± 0.4 | −25.1 | 5 | 3.5 ± 1.4 | 4.3 |
| Slightly degraded moist forest | 9 | −24.7 ± 0.4 | −24.7 | 9 | 1.3 ± 1.3 | 1.4 | |
| χ2 = 2.61, df = 1, p = 0.11 | χ2 = 5.16, df = 1, p = 0.023 | ||||||
| Levene p = 0.50 | Levene p = 0.85 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crowley, B.E.; Goodman, S.M. Investigating Foraging Niches for Tenrecs, Bats, and Rodents at Betampona Réserve Naturelle Intégrale (Central Eastern Madagascar) Using Stable Carbon and Nitrogen Isotopes in Fur and Bone. Animals 2025, 15, 2423. https://doi.org/10.3390/ani15162423
Crowley BE, Goodman SM. Investigating Foraging Niches for Tenrecs, Bats, and Rodents at Betampona Réserve Naturelle Intégrale (Central Eastern Madagascar) Using Stable Carbon and Nitrogen Isotopes in Fur and Bone. Animals. 2025; 15(16):2423. https://doi.org/10.3390/ani15162423
Chicago/Turabian StyleCrowley, Brooke Erin, and Steven Michael Goodman. 2025. "Investigating Foraging Niches for Tenrecs, Bats, and Rodents at Betampona Réserve Naturelle Intégrale (Central Eastern Madagascar) Using Stable Carbon and Nitrogen Isotopes in Fur and Bone" Animals 15, no. 16: 2423. https://doi.org/10.3390/ani15162423
APA StyleCrowley, B. E., & Goodman, S. M. (2025). Investigating Foraging Niches for Tenrecs, Bats, and Rodents at Betampona Réserve Naturelle Intégrale (Central Eastern Madagascar) Using Stable Carbon and Nitrogen Isotopes in Fur and Bone. Animals, 15(16), 2423. https://doi.org/10.3390/ani15162423







