The Importance of Animal Models in Biomedical Research: Current Insights and Applications
Abstract
Simple Summary
Abstract
1. Introduction
2. Search Methodology
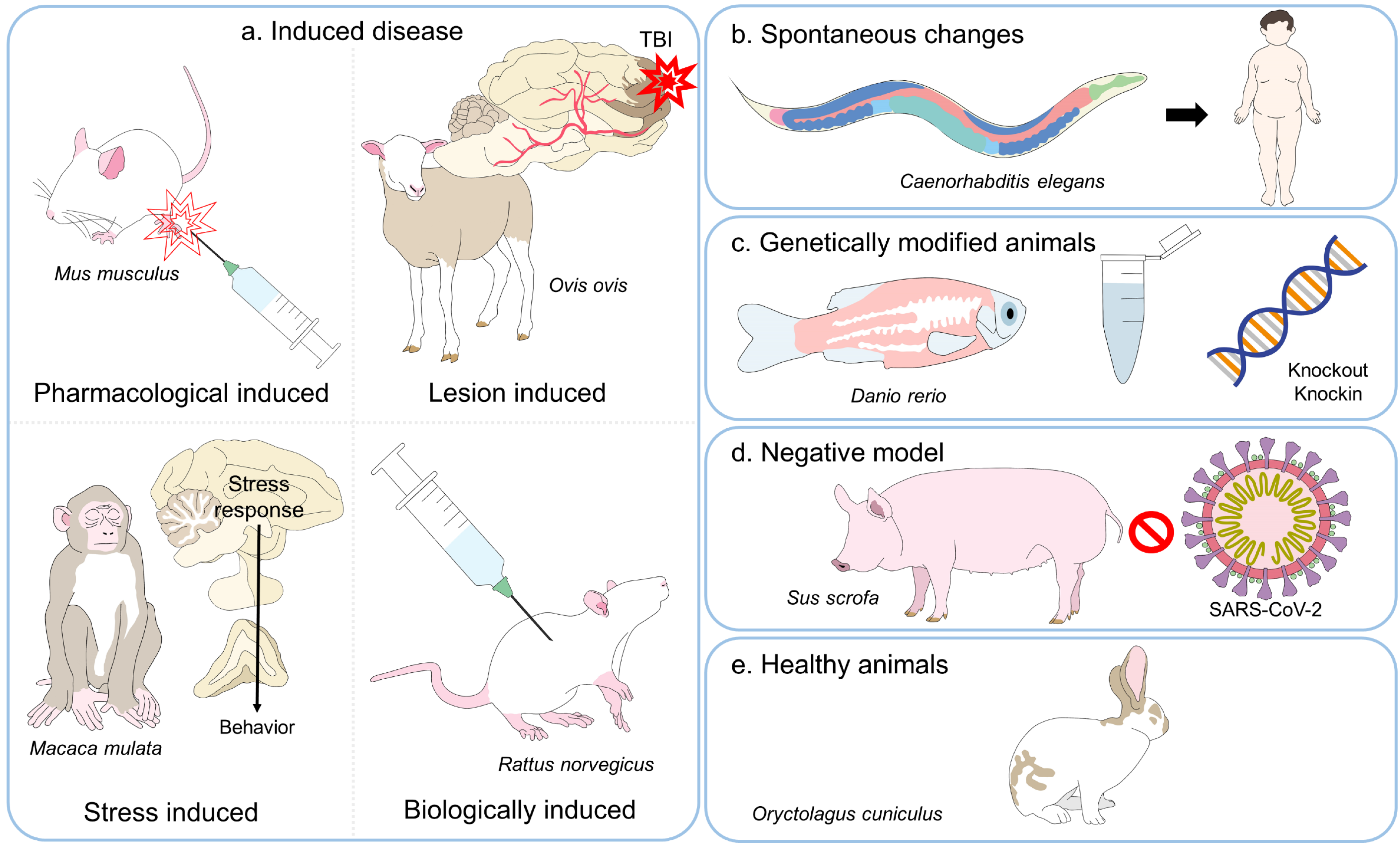
3. A Review of Animal Experimentation
4. Animal Models and Their Application in Distinct Fields of Current Biomedical Science
4.1. Emerging Infectious Diseases
4.2. Endocrinology and Metabolic Pathologies
4.3. Cancer in Biomedicine
4.4. Pharmacology and Therapeutics
4.5. Experimental Surgical Tecniques
4.6. Neurosciences
4.7. Physiotherapy and Rehabilitation
5. New Models and Strategies Applied in Animal Research
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Davies, G.; Gorman, R.; Greenhough, B.; Hobson-West, P.; Kirk, R.G.W.; Message, R.; Myelnikov, D.; Palmer, A.; Roe, E.; Ashall, V.; et al. Animal research nexus: A new approach to the connections between science, health and animal welfare. Med. Humanit. 2020, 46, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Hobson-West, P.; Davies, A. Societal Sentience: Constructions of the public in animal research policy and practice. Sci. Technol. Hum. Values 2018, 43, 671–693. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.B.; Krieger, K.; Khan, F.M.; Huffman, W.; Chang, M.; Naik, A.; Yongle, R.; Hameed, I.; Krieger, K.; Girardi, L.N.; et al. The current state of animal models in research: A review. Int. J. Surg. 2019, 72, 9–13. [Google Scholar] [CrossRef]
- Lee, K.H.; Lee, D.W.; Kang, B.C. The ‘R’ principles in laboratory animal experiments. Lab. Anim. Res. 2020, 36, 45. [Google Scholar] [CrossRef]
- Andersen, M.L.; Winter, L.M.F. Animal models in biological and biomedical research—Experimental and ethical concerns. An. Acad. Bras. Cienc. 2019, 91, e20170238. [Google Scholar] [CrossRef]
- Juárez-Portilla, C.; Zepeda-Hernández, R.C.; Sánchez-Salcedo, J.A.; Flores-Muñoz, M.; López-Franco, Ó.; Cortés-Sol, A. El uso de los animales en la investigación y en la enseñanza: Lineamientos y directrices para su manejo. Rev. Eduscientia. Divulg. Cienc. Educ. 2019, 2, 4–19. [Google Scholar]
- Jota Baptista, C.V.; Faustino-Rocha, A.I.; Oliveira, P.A. Animal models in pharmacology: A brief history awarding the nobel prizes for physiology or medicine. Pharmacology 2021, 106, 356–368. [Google Scholar] [CrossRef] [PubMed]
- Swearengen, J.R. Choosing the right animal model for infectious disease research. Anim. Model. Exp. Med. 2018, 1, 100–108. [Google Scholar] [CrossRef]
- Makowska, I.J.; Weary, D.M. A good life for laboratory rodents? ILAR J. 2020, 60, 373–388. [Google Scholar] [CrossRef]
- Carbone, L. Estimating mouse and rat use in American laboratories by extrapolation from Animal Welfare Act-regulated species. Sci. Rep. 2021, 11, 493. [Google Scholar] [CrossRef]
- Smith, J.R.; Bolton, E.R.; Dwinell, M.R. The rat: A model used in biomedical research. In Rat Genomics; Hayman, T., Smith, J.R., Dwinell, M.R., Shimoyama, M., Eds.; Springer: Hertfordshire, UK, 2019; pp. 1–41. [Google Scholar]
- Joyce, C.; Scallan, C.D.; Mateo, R.; Belshe, R.B.; Tucker, S.N.; Moore, A.C. Orally administered adenoviral-based vaccine induces respiratory mucosal memory and protection against RSV infection in cotton rats. Vaccine 2018, 36, 4265–4277. [Google Scholar] [CrossRef] [PubMed]
- Munster, V.J.; Feldmann, F.; Williamson, B.N.; van Doremalen, N.; Pérez-Pérez, L.; Schulz, J.; Meade-White, K.; Okumura, A.; Callison, J.; Brumbaugh, B.; et al. Respiratory disease in rhesus macaques inoculated with SARS-CoV-2. Nature 2020, 585, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Mariscal, A.; Caldarone, L.; Tikkanen, J.; Nakajima, D.; Chen, M.; Yeung, J.; Cypel, M.; Liu, M.; Keshavjee, S. Pig lung transplant survival model. Nat. Protoc. 2018, 13, 1814–1828. [Google Scholar] [CrossRef] [PubMed]
- Lasko, P.; Lüthy, K. Investigating rare and ultrarare epilepsy syndromes with Drosophila models. Fac. Rev. 2021, 10, 10. [Google Scholar] [CrossRef]
- Babac, B.D.; Milton Dulay, R.R.; Arwen Calpito, R.S.; Domingo, M.A.; Grace Macamos, M.M.; Mangabat, A.R.; Zhyra Zoberiaga, N.L. In-vitro activity of ethanolic extract of Lentinus strigosus mycelia in N2 wild strain Caenorhabditis elegans-An animal model for obesity and its chemical composition. J. Appl. Biol. Biotech. 2021, 9, 41–46. [Google Scholar] [CrossRef]
- Zang, L.; Maddison, L.A.; Chen, W. Zebrafish as a Model for Obesity and Diabetes. Front. Cell Dev. Biol. 2018, 6, 91. [Google Scholar] [CrossRef]
- Fernandes, M.R.; Pedroso, A.R. Animal experimentation: A look into ethics, welfare and alternative methods. Rev. Assoc. Med. Bras. 2017, 63, 923–928. [Google Scholar] [CrossRef]
- Adir, O.; Poley, M.; Chen, G.; Froim, S.; Krinsky, N.; Shklover, J.; Shainsky-Roitman, J.; Lammers, T.; Schroeder, A. Integrating artificial intelligence and nanotechnology for precision cancer medicine. Adv. Mater. 2020, 32, 1901989. [Google Scholar] [CrossRef]
- Bale, T.L.; Abel, T.; Akil, H.; Carlezon, W.A.; Moghaddam, B.; Nestler, E.J.; Ressler, K.J.; Thompson, S.M. The critical importance of basic animal research for neuropsychiatric disorders. Neuropsychopharmacology 2019, 44, 1349–1353. [Google Scholar] [CrossRef]
- Zeggini, E.; Baumann, M.; Götz, M.; Herzig, S.; Hrabe de Angelis, M.; Tschöp, M.H. Biomedical research goes viral: Dangers and opportunities. Cell 2020, 181, 1189–1193. [Google Scholar] [CrossRef]
- Okechukwu, I.B. Introductory Chapter: Animal models for human diseases, a major contributor to modern medicine. In Experimental Animal Models of Human Diseases—An Effective Therapeutic Strategy; Batholomew, I., Ed.; InTech: London, UK, 2018; pp. 3–10. [Google Scholar]
- Kleinert, M.; Clemmensen, C.; Hofmann, S.M.; Moore, M.C.; Renner, S.; Woods, S.C.; Huypens, P.; Beckers, J.; de Angelis, M.H.; Schürmann, A.; et al. Animal models of obesity and diabetes mellitus. Nat. Rev. Endocrinol. 2018, 14, 140–162. [Google Scholar] [CrossRef]
- Wright, J.R. Frederick Banting’s actual great idea: The role of fetal bovine islets in the discovery of insulin. Islets 2021, 13, 121–133. [Google Scholar] [CrossRef]
- Understanding Animal Research. EU-Wide Animal Research Statistics. Available online: https://www.understandinganimalresearch.org.uk/news/eu-wide-animal-research-statistics-2019 (accessed on 9 March 2023).
- Canadian Council on Animal Care. CCAC Animal Data Report 2020. Available online: https://speakingofresearch.files.wordpress.com/2021/11/canada-2020.pdf (accessed on 9 March 2020).
- Speaking of Research. UK Animal Research Statistics. Available online: https://speakingofresearch.com/facts/uk-statistics/ (accessed on 9 March 2023).
- Speaking of Research. US animal Research Statistics. Available online: https://speakingofresearch.com/facts/statistics/ (accessed on 9 March 2023).
- Speaking of Research. Rise in Animal Research in South Korea in 2017. Available online: https://speakingofresearch.com/2018/04/12/rise-in-animal-research-in-south-korea-in-2017/ (accessed on 9 March 2023).
- Statista. Annual Number of Animals Used in Research and Testing in Selected Countries Worldwide as of 2020. Available online: https://www.statista.com/statistics/639954/animals-used-in-research-experiments-worldwide/ (accessed on 9 March 2023).
- Speaking of Research. Worldwide Animal Research Statistics. Available online: https://speakingofresearch.com/facts/animal-research-statistics/ (accessed on 9 March 2023).
- Gil, G.; Rodríguez, X. Experimentos con Animales en México: 20 Años de Caos y Riesgo. Available online: https://aristeguinoticias.com/2405/mexico/__trashed-25/ (accessed on 9 March 2023).
- Hubrecht, R.C. The 3Rs and humane experimental technique: Implementing change. Animals 2019, 9, 754. [Google Scholar] [CrossRef] [PubMed]
- Marquardt, N.; Feja, M.; Hünigen, H.; Plendl, J.; Menken, L.; Fink, H.; Bert, B. Euthanasia of laboratory mice: Are isoflurane and sevoflurane real alternatives to carbon dioxide? PLoS ONE 2018, 13, e0203793. [Google Scholar] [CrossRef] [PubMed]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. J. Cereb. Blood Flow Metab. 2020, 40, 1769–1777. [Google Scholar] [CrossRef]
- Smith, A.J.; Clutton, R.E.; Lilley, E.; Hansen, K.E.A.; Brattelid, T. PREPARE: Guidelines for planning animal research and testing. Lab. Anim. 2018, 52, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Bert, B.; Heinl, C.; Chmielewska, J.; Schwarz, F.; Grune, B.; Hensel, A.; Greiner, M.; Schönfelder, G. Refining animal research: The Animal Study Registry. PLoS Biol. 2019, 17, e3000463. [Google Scholar] [CrossRef]
- Singh, A.; Gupta, U. Animal models of tuberculosis: Lesson learnt. Indian J. Med. Res. 2018, 147, 456. [Google Scholar] [CrossRef]
- Kim, T.-W.; Che, J.-H.; Yun, J.-W. Use of stem cells as alternative methods to animal experimentation in predictive toxicology. Regul. Toxicol. Pharmacol. 2019, 105, 15–29. [Google Scholar] [CrossRef]
- Yang, H.; Wu, Z. Genome editing of pigs for agriculture and biomedicine. Front. Genet. 2018, 9, 360. [Google Scholar] [CrossRef]
- Muñoz-Fontela, C.; Dowling, W.E.; Funnell, S.G.P.; Gsell, P.-S.; Riveros-Balta, A.X.; Albrecht, R.A.; Andersen, H.; Baric, R.S.; Carroll, M.W.; Cavaleri, M.; et al. Animal models for COVID-19. Nature 2020, 586, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Dissanayake, H.A.; de Silva, N.L.; Sumanatilleke, M.; de Silva, S.D.N.; Gamage, K.K.K.; Dematapitiya, C.; Kuruppu, D.C.; Ranasinghe, P.; Pathmanathan, S.; Katulanda, P. Prognostic and therapeutic role of vitamin D in COVID-19: Systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2022, 107, 1484–1502. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 9 June 2022).
- Woolsey, C.; Borisevich, V.; Prasad, A.N.; Agans, K.N.; Deer, D.J.; Dobias, N.S.; Heymann, J.C.; Foster, S.L.; Levine, C.B.; Medina, L.; et al. Establishment of an African green monkey model for COVID-19 and protection against re-infection. Nat. Immunol. 2021, 22, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Cleary, S.J.; Pitchford, S.C.; Amison, R.T.; Carrington, R.; Robaina Cabrera, C.L.; Magnen, M.; Looney, M.R.; Gray, E.; Page, C.P. Animal models of mechanisms of SARS-CoV-2 infection and COVID-19 pathology. Br. J. Pharmacol. 2020, 177, 4851–4865. [Google Scholar] [CrossRef]
- Da Costa, C.B.P.; Cruz, A.C.D.M.; Penha, J.C.Q.; Castro, H.C.; Da Cunha, L.E.R.; Ratcliffe, N.A.; Cisne, R.; Martins, F.J. Using in vivo animal models for studying SARS-CoV-2. Expert Opin. Drug Discov. 2022, 17, 121–137. [Google Scholar] [CrossRef]
- Younes, S.; Younes, N.; Shurrab, F.; Nasrallah, G.K. Severe acute respiratory syndrome coronavirus-2 natural animal reservoirs and experimental models: Systematic review. Rev. Med. Virol. 2021, 31, e2196. [Google Scholar] [CrossRef]
- Sun, S.-H.; Chen, Q.; Gu, H.-J.; Yang, G.; Wang, Y.-X.; Huang, X.-Y.; Liu, S.-S.; Zhang, N.-N.; Li, X.-F.; Xiong, R.; et al. A mouse model of SARS-CoV-2 infection and pathogenesis. Cell Host Microbe 2020, 28, 124–133.e4. [Google Scholar] [CrossRef]
- Mueller, A.L.; McNamara, M.S.; Sinclair, D.A. Why does COVID-19 disproportionately affect older people? Aging 2020, 12, 9959–9981. [Google Scholar] [CrossRef]
- Devaux, C.A.; Lagier, J.-C.; Raoult, D. New insights into the physiopathology of COVID-19: SARS-CoV-2-associated gastrointestinal illness. Front. Med. 2021, 8, 640073. [Google Scholar] [CrossRef]
- Bi, Z.; Hong, W.; Yang, J.; Lu, S.; Peng, X. Animal models for SARS-CoV-2 infection and pathology. MedComm 2021, 2, 548–568. [Google Scholar] [CrossRef]
- Urano, E.; Okamura, T.; Ono, C.; Ueno, S.; Nagata, S.; Kamada, H.; Higuchi, M.; Furukawa, M.; Kamitani, W.; Matsuura, Y.; et al. COVID-19 cynomolgus macaque model reflecting human COVID-19 pathological conditions. Proc. Natl. Acad. Sci. USA 2021, 118, e2104847118. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Qi, F.; Xu, Y.; Li, F.; Liu, P.; Liu, J.; Bao, L.; Deng, W.; Gao, H.; Xiang, Z.; et al. Age-related rhesus macaque models of COVID-19. Anim. Model. Exp. Med. 2020, 3, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Csiszar, A.; Jakab, F.; Valencak, T.G.; Lanszki, Z.; Tóth, G.E.; Kemenesi, G.; Tarantini, S.; Fazekas-Pongor, V.; Ungvari, Z. Companion animals likely do not spread COVID-19 but may get infected themselves. GeroScience 2020, 42, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Harilal, S.; Al-Sehemi, A.G.; Pannipara, M.; Behl, T.; Mathew, G.E.; Mathew, B. COVID-19 and domestic animals: Exploring the species barrier crossing, zoonotic and reverse zoonotic transmission of SARS-CoV-2. Curr. Pharm. Des. 2021, 27, 1194–1201. [Google Scholar] [CrossRef]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z.; et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS–coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef]
- Hosie, M.J.; Hofmann-Lehmann, R.; Hartmann, K.; Egberink, H.; Truyen, U.; Addie, D.D.; Belák, S.; Boucraut-Baralon, C.; Frymus, T.; Lloret, A.; et al. Anthropogenic infection of cats during the 2020 COVID-19 pandemic. Viruses 2021, 13, 185. [Google Scholar] [CrossRef]
- Pramod, R.K.; Nair, A.V.; Tambare, P.K.; Chauhan, K.; Kumar, T.V.; Rajan, R.A.; Mani, B.M.; Asaf, M.; Pandey, A.K. Reverse zoonosis of coronavirus disease-19: Present status and the control by one health approach. Vet. World 2021, 14, 2817–2826. [Google Scholar] [CrossRef]
- Kang, K.; Chen, Q.; Gao, Y.; Yu, K. Detection of SARS-CoV-2 B.1.617.2 (Delta) variant in three cats owned by a confirmed COVID-19 patient in Harbin, China. Vet. Med. Sci. 2022, 8, 945–946. [Google Scholar] [CrossRef]
- da Costa Rodrigues, K.C.; Pereira, R.M.; de Campos, T.D.P.; de Moura, R.F.; da Silva, A.S.R.; Cintra, D.E.; Ropelle, E.R.; Pauli, J.R.; de Araújo, M.B.; de Moura, L.P. The role of physical exercise to improve the browning of white adipose tissue via POMC neurons. Front. Cell. Neurosci. 2018, 12, 88. [Google Scholar] [CrossRef]
- Rios, J.L.; Bomhof, M.R.; Reimer, R.A.; Hart, D.A.; Collins, K.H.; Herzog, W. Protective effect of prebiotic and exercise intervention on knee health in a rat model of diet-induced obesity. Sci. Rep. 2019, 9, 3893. [Google Scholar] [CrossRef]
- Burhans, M.S.; Hagman, D.K.; Kuzma, J.N.; Schmidt, K.A.; Kratz, M. Contribution of adipose tissue inflammation to the development of type 2 diabetes mellitus. Compr. Physiol. 2018, 9, 1–58. [Google Scholar] [CrossRef] [PubMed]
- Khalafi, M.; Mohebbi, H.; Symonds, M.E.; Karimi, P.; Akbari, A.; Tabari, E.; Faridnia, M.; Moghaddami, K. The impact of moderate-intensity continuous or high-intensity interval training on adipogenesis and browning of subcutaneous adipose tissue in obese male rats. Nutrients 2020, 12, 925. [Google Scholar] [CrossRef]
- Lee, H.E.; Yang, G.; Han, S.-H.; Lee, J.-H.; An, T.-J.; Jang, J.-K.; Lee, J.Y. Anti-obesity potential of Glycyrrhiza uralensis and licochalcone A through induction of adipocyte browning. Biochem. Biophys. Res. Commun. 2018, 503, 2117–2123. [Google Scholar] [CrossRef]
- Wang, P.; Li, D.; Ke, W.; Liang, D.; Hu, X.; Chen, F. Resveratrol-induced gut microbiota reduces obesity in high-fat diet-fed mice. Int. J. Obes. 2020, 44, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Lasker, S.; Rahman, M.M.; Parvez, F.; Zamila, M.; Miah, P.; Nahar, K.; Kabir, F.; Sharmin, S.B.; Subhan, N.; Ahsan, G.U.; et al. High-fat diet-induced metabolic syndrome and oxidative stress in obese rats are ameliorated by yogurt supplementation. Sci. Rep. 2019, 9, 20026. [Google Scholar] [CrossRef] [PubMed]
- Bouyanfif, A.; Jayarathne, S.; Koboziev, I.; Moustaid-Moussa, N. The nematode Caenorhabditis elegans as a model organism to study metabolic effects of ω-3 polyunsaturated fatty acids in obesity. Adv. Nutr. 2019, 10, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Ke, W.; Reed, J.N.; Yang, C.; Higgason, N.; Rayyan, L.; Wählby, C.; Carpenter, A.E.; Civelek, M.; O’Rourke, E.J. Genes in human obesity loci are causal obesity genes in C. elegans. PLoS Genet. 2021, 17, e1009736. [Google Scholar] [CrossRef]
- Farias-Pereira, R.; Zhang, Z.; Park, C.; Kim, D.; Kim, K.; Park, Y. Butein inhibits lipogenesis in Caenorhabditis elegans. BioFactors 2020, 46, 777–787. [Google Scholar] [CrossRef]
- Yusela Kris, A.; Rich Milton, D.; Sofronio, K. Effect of Lentinus strigosus extract on the food intake and locomotion of N2 wild strain Caenorhabditis elegans as model for obesity. J. Appl. Pharm. Sci. 2020, 10, 23–28. [Google Scholar] [CrossRef]
- Benchoula, K.; Khatib, A.; Jaffar, A.; Ahmed, Q.U.; Sulaiman, W.M.A.W.; Wahab, R.A.; El-Seedi, H.R. The promise of zebrafish as a model of metabolic syndrome. Exp. Anim. 2019, 68, 407–416. [Google Scholar] [CrossRef]
- Rodríguez, R.R.; González-Bulnes, A.; Garcia-Contreras, C.; Elena Rodriguez-Rodriguez, A.; Astiz, S.; Vazquez-Gomez, M.; Luis Pesantez, J.; Isabel, B.; Salido-Ruiz, E.; González, J.; et al. The Iberian pig fed with high-fat diet: A model of renal disease in obesity and metabolic syndrome. Int. J. Obes. 2020, 44, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Chagnac, A.; Zingerman, B.; Rozen-Zvi, B.; Herman-Edelstein, M. Consequences of glomerular hyperfiltration: The role of physical forces in the pathogenesis of chronic kidney disease in diabetes and obesity. Nephron 2019, 143, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Rodríguez, A.E.; Donate-Correa, J.; Luis-Lima, S.; Díaz-Martín, L.; Rodríguez-González, C.; Pérez-Pérez, J.A.; Acosta-González, N.G.; Fumero, C.; Navarro-Díaz, M.; López-Álvarez, D.; et al. Obesity and metabolic syndrome induce hyperfiltration, glomerulomegaly, and albuminuria in obese ovariectomized female mice and obese male mice. Menopause 2021, 28, 1296–1306. [Google Scholar] [CrossRef]
- Hoffman, J.M.; Creevy, K.E.; Franks, A.; O’Neill, D.G.; Promislow, D.E.L. The companion dog as a model for human aging and mortality. Aging Cell 2018, 17, e12737. [Google Scholar] [CrossRef]
- Suarez, L.; Bautista-Castaño, I.; Peña Romera, C.; Montoya-Alonso, J.A.; Corbera, J.A. Is dog owner obesity a risk factor for canine obesity? a “one-health” study on human–animal interaction in a region with a high prevalence of obesity. Vet. Sci. 2022, 9, 243. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 9 March 2023).
- National Cancer Institute. Common Cancer Types. Available online: https://www.cancer.gov/types/common-cancers (accessed on 9 March 2023).
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Berish, R.B.; Ali, A.N.; Telmer, P.G.; Ronald, J.A.; Leong, H.S. Translational models of prostate cancer bone metastasis. Nat. Rev. Urol. 2018, 15, 403–421. [Google Scholar] [CrossRef]
- Onaciu, A.; Munteanu, R.; Munteanu, V.C.; Gulei, D.; Raduly, L.; Feder, R.-I.; Pirlog, R.; Atanasov, A.G.; Korban, S.S.; Irimie, A.; et al. Spontaneous and induced animal models for cancer research. Diagnostics 2020, 10, 660. [Google Scholar] [CrossRef]
- Valcourt, D.M.; Kapadia, C.H.; Scully, M.A.; Dang, M.N.; Day, E.S. Best practices for preclinical in vivo testing of cancer nanomedicines. Adv. Healthc. Mater. 2020, 9, 2000110. [Google Scholar] [CrossRef]
- Santana-Krímskaya, S.E.; Kawas, J.R.; Zarate-Triviño, D.G.; Ramos-Zayas, Y.; Rodríguez-Padilla, C.; Franco-Molina, M.A. Orthotopic and heterotopic triple negative breast cancer preclinical murine models: A tumor microenvironment comparative. Res. Vet. Sci. 2022, 152, 364–371. [Google Scholar] [CrossRef]
- Madonna, M.C.; Duer, J.E.; Lee, J.V.; Williams, J.; Avsaroglu, B.; Zhu, C.; Deutsch, R.; Wang, R.; Crouch, B.T.; Hirschey, M.D.; et al. In vivo optical metabolic imaging of long-chain fatty acid uptake in orthotopic models of triple-negative breast cancer. Cancers 2021, 13, 148. [Google Scholar] [CrossRef] [PubMed]
- Koosha, S.; Mohamed, Z.; Sinniah, A.; Alshawsh, M.A. Evaluation of anti-tumorigenic effects of diosmetin against human colon cancer xenografts in athymic nude mice. Molecules 2019, 24, 2522. [Google Scholar] [CrossRef]
- Sliepen, S.H.J.; Diaz-del Castillo, M.; Korioth, J.; Olsen, R.B.; Appel, C.K.; Christoph, T.; Heegaard, A.-M.; Rutten, K. Cancer-induced bone pain impairs burrowing behaviour in mouse and rat. In Vivo 2019, 33, 1125–1132. [Google Scholar] [CrossRef] [PubMed]
- Overgaard, N.H.; Fan, T.M.; Schachtschneider, K.M.; Principe, D.R.; Schook, L.B.; Jungersen, G. Of Mice, Dogs, Pigs, and Men: Choosing the Appropriate Model for Immuno-Oncology Research. ILAR J. 2018, 59, 247–262. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, A.; Hiyama, T.; Fujimura, A.; Yoshikawa, S. Sympathetic and parasympathetic innervation in cancer: Therapeutic implications. Clin. Auton. Res. 2021, 31, 165–178. [Google Scholar] [CrossRef]
- Sadighparvar, S.; Darband, S.G.; Ghaderi-Pakdel, F.; Mihanfar, A.; Majidinia, M. Parasympathetic, but not sympathetic denervation, suppressed colorectal cancer progression. Eur. J. Pharmacol. 2021, 913, 174626. [Google Scholar] [CrossRef]
- Kamiya, A.; Hayama, Y.; Kato, S.; Shimomura, A.; Shimomura, T.; Irie, K.; Kaneko, R.; Yanagawa, Y.; Kobayashi, K.; Ochiya, T. Genetic manipulation of autonomic nerve fiber innervation and activity and its effect on breast cancer progression. Nat. Neurosci. 2019, 22, 1289–1305. [Google Scholar] [CrossRef]
- Grelet, S.; Fréreux, C.; Obellianne, C.; Noguchi, K.; Howley, B.V.; Dalton, A.C.; Howe, P.H. TGFβ-induced expression of long noncoding lincRNA Platr18 controls breast cancer axonogenesis. Life Sci. Alliance 2022, 5, e202101261. [Google Scholar] [CrossRef]
- Kirkpatrick, J.D.; Warren, A.D.; Soleimany, A.P.; Westcott, P.M.K.; Voog, J.C.; Martin-Alonso, C.; Fleming, H.E.; Tammela, T.; Jacks, T.; Bhatia, S.N. Urinary detection of lung cancer in mice via noninvasive pulmonary protease profiling. Sci. Transl. Med. 2020, 12, eaaw0262. [Google Scholar] [CrossRef]
- Xu, X.; An, H.; Zhang, D.; Tao, H.; Dou, Y.; Li, X.; Huang, J.; Zhang, J. A self-illuminating nanoparticle for inflammation imaging and cancer therapy. Sci. Adv. 2019, 5, eaat2953. [Google Scholar] [CrossRef]
- Momcilovic, M.; Jones, A.; Bailey, S.T.; Waldmann, C.M.; Li, R.; Lee, J.T.; Abdelhady, G.; Gomez, A.; Holloway, T.; Schmid, E.; et al. In vivo imaging of mitochondrial membrane potential in non-small-cell lung cancer. Nature 2019, 575, 380–384. [Google Scholar] [CrossRef]
- Nicolson, F.; Andreiuk, B.; Andreou, C.; Hsu, H.-T.; Rudder, S.; Kircher, M.F. Non-invasive in vivo imaging of cancer using surface-enhanced spatially offset Raman Spectroscopy (SESORS). Theranostics 2019, 9, 5899–5913. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zuo, W.; Xiao, Z.; Jin, Q.; Liu, J.; Wu, L.; Liu, N.; Zhu, X. A carrier-free metal-coordinated dual-photosensitizers nanotheranostic with glutathione-depletion for fluorescence/photoacoustic imaging-guided tumor phototherapy. J. Colloid Interface Sci. 2021, 600, 243–255. [Google Scholar] [CrossRef]
- Hernández-Avalos, I.; Mota-Rojas, D.; Mendoza-Flores, J.E.; Casas-Alvarado, A.; Flores-Padilla, K.; Miranda-Cortes, A.E.; Torres-Bernal, F.; Gómez-Prado, J.; Mora-Medina, P. Nociceptive pain and anxiety in equines: Physiological and behavioral alterations. Vet. World 2021, 14, 2984–2995. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, A.; Mittal, G. Ketamine-polymer based drug delivery system for prolonged analgesia: Recent advances, challenges and future prospects. Expert Opin. Drug Deliv. 2021, 18, 1117–1130. [Google Scholar] [CrossRef] [PubMed]
- Parra, S.; Thanawala, V.J.; Rege, A.; Giles, H. A novel excisional wound pain model for evaluation of analgesics in rats. Korean J. Pain 2021, 34, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Millecam, J.; van Bergen, T.; Schauvliege, S.; Antonissen, G.; Martens, A.; Chiers, K.; Gehring, R.; Gasthuys, E.; Vande Walle, J.; Croubels, S.; et al. Developmental pharmacokinetics and safety of ibuprofen and its enantiomers in the conventional pig as potential pediatric animal model. Front. Pharmacol. 2019, 10, 505. [Google Scholar] [CrossRef]
- Domínguez-Oliva, A.; Casas-Alvarado, A.; Miranda-Cortés, A.E.; Hernández-Avalos, I. Clinical pharmacology of tramadol and tapentadol, and their therapeutic efficacy in different models of acute and chronic pain in dogs and cats. J. Adv. Vet. Anim. Res. 2021, 8, 404. [Google Scholar] [CrossRef]
- Mulier, J.P. Is opioid-free general anesthesia for breast and gynecological surgery a viable option? Curr. Opin. Anaesthesiol. 2019, 32, 257–262. [Google Scholar] [CrossRef]
- Cicirelli, V.; Burgio, M.; Lacalandra, G.M.; Aiudi, G.G. Local and regional anaesthetic techniques in canine ovariectomy: A review of the literature and technique description. Animals 2022, 12, 1920. [Google Scholar] [CrossRef]
- Miranda-Cortés, A.E.; Ruiz-García, A.G.; Olivera-Ayub, A.E.; Garza-Malacara, G.; Ruiz-Cervantes, J.G.; Toscano-Zapien, J.A.; Hernández-Avalos, I. Cardiorespiratory effects of epidurally administered ketamine or lidocaine in dogs undergoing ovariohysterectomy surgery: A comparative study. Iran. J. Vet. Res. 2020, 21, 92–96. [Google Scholar] [PubMed]
- Komatsu, A.; Miyano, K.; Nakayama, D.; Mizobuchi, Y.; Uezono, E.; Ohshima, K.; Karasawa, Y.; Kuroda, Y.; Nonaka, M.; Yamaguchi, K.; et al. Novel opioid analgesics for the development of transdermal opioid patches that possess morphine-like pharmacological profiles rather than fentanyl: Possible opioid switching alternatives among patch formula. Anesth. Analg. 2022, 134, 1082–1093. [Google Scholar] [CrossRef]
- Godfrey, L.; Iannitelli, A.; Garrett, N.L.; Moger, J.; Imbert, I.; King, T.; Porreca, F.; Soundararajan, R.; Lalatsa, A.; Schätzlein, A.G.; et al. Nanoparticulate peptide delivery exclusively to the brain produces tolerance free analgesia. J. Control. Release 2018, 270, 135–144. [Google Scholar] [CrossRef]
- Wang, B.; Wang, S.; Zhang, Q.; Deng, Y.; Li, X.; Peng, L.; Zuo, X.; Piao, M.; Kuang, X.; Sheng, S.; et al. Recent advances in polymer-based drug delivery systems for local anesthetics. Acta Biomater. 2019, 96, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Gan, J.; Li, R.; Duan, J.; Zhou, J.; Lv, M.; Qi, R. Controlled delivery of ketamine from reduced graphene oxide hydrogel for neuropathic pain: In vitro and in vivo studies. J. Drug Deliv. Sci. Technol. 2020, 60, 101964. [Google Scholar] [CrossRef]
- Kania, K.; Chang, D.K.; Abu-Ghname, A.; Reece, E.M.; Chu, C.K.; Maricevich, M.; Buchanan, E.P.; Winocour, S. Microsurgery training in plastic surgery. Plast. Reconstr. Surg.—Glob. Open 2020, 8, e2898. [Google Scholar] [CrossRef]
- Ozols, D.; Zarins, J.; Petersons, A. Novel technique for toe-to-hand transplantation: The fourth-toe as an alternative option for toe-to-hand transplantation for pediatric patients. Tech. Hand Up. Extrem. Surg. 2019, 23, 74–80. [Google Scholar] [CrossRef]
- Dvořák, Z.; Stupka, I. Atypical replantation and reconstruction of frozen ear. Medicine 2020, 99, e20068. [Google Scholar] [CrossRef]
- Khachatryan, A.; Tevosyan, A.; Novoselskiy, D.; Arakelyan, G.; Yushkevich, A.; Nazaretovich Nazarian, D. Experimental reimplantation models. In Microsurgery Manual for Medical Students and Residents; Springer International Publishing: Cham, Switzerland, 2021; pp. 139–144. [Google Scholar]
- Goutard, M.; Randolph, M.A.; Taveau, C.B.; Lupon, E.; Lantieri, L.; Uygun, K.; Cetrulo, C.L.; Lellouch, A.G. Partial heterotopic hindlimb transplantation model in rats. J. Vis. Exp. 2021, 172, e62586. [Google Scholar] [CrossRef]
- Vernon, R.; Wang, J.; Song, M.; Wilson, N.; Moris, D.; Cendales, L. Vascularized Composite Allotransplantation: A Functional Hind Limb Model in Mice. J. Surg. Res. 2020, 250, 119–124. [Google Scholar] [CrossRef]
- Tee, R.; Morrison, W.A.; Dilley, R.J. A novel microsurgical rodent model for the transplantation of engineered cardiac muscle flap. Microsurgery 2018, 38, 544–552. [Google Scholar] [CrossRef]
- Scaglioni, M.; Giovanoli, P.; Scaglioni, M.F.; Yang, J.C. Microsurgical head and neck reconstruction in patients with coronary artery disease: A perioperative assessment algorithm. Microsurgery 2019, 39, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Kotz, D. UM Medicine Performs Historic Xenotransplantation. Available online: https://www.umaryland.edu/news/archived-news/january-2022/um-medicine-performs-historic-xenotransplantation.php (accessed on 7 June 2022).
- Wang, W.; He, W.; Ruan, Y.; Geng, Q. First pig-to-human heart transplantation. Innovation 2022, 3, 100223. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, J.S.; Chee, H.K.; Yun, I.J.; Park, K.S.; Yang, H.S.; Park, J.H. Seven years of experiences of preclinical experiments of xeno-heart transplantation of pig to non-human primate (Cynomolgus Monkey). Transplant. Proc. 2018, 50, 1167–1171. [Google Scholar] [CrossRef]
- Cui, Y.; Yamamoto, T.; Raza, S.S.; Morsi, M.; Nguyen, H.Q.; Ayares, D.; Cooper, D.K.C.; Hara, H. Evidence for GTKO/β4GalNT2KO pigs as the preferred organ-source for old world nonhuman primates as a preclinical model of xenotransplantation. Transplant. Direct. 2020, 6, e590. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, A.; Zaman, M.; Wahab Amjad, M.; Adnan, S.; Abdul Ghafoor Raja, M.; Haider Rizvi, S.F.; Mustafa, M.W.; Farooq, U.; Abbas, G.; Shah, S. Solid lipid nanoparticles of mycophenolate mofetil: An attempt to control the release of an immunosuppressant. Int. J. Nanomed. 2020, 15, 5603–5612. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Liang, J.; He, Y.; Qin, J.; He, H.; Lee, S.; Pang, Z.; Wang, J. Sustained release of immunosuppressant by nanoparticle-anchoring hydrogel scaffold improved the survival of transplanted stem cells and tissue regeneration. Theranostics 2018, 8, 878–893. [Google Scholar] [CrossRef]
- Xie, H.; Zhu, H.; Zhou, K.; Wan, J.; Zhang, L.; Yang, Z.; Zhou, L.; Chen, X.; Xu, X.; Zheng, S.; et al. Target-oriented delivery of self-assembled immunosuppressant cocktails prolongs allogeneic orthotopic liver transplant survival. J. Control. Release 2020, 328, 237–250. [Google Scholar] [CrossRef]
- Natoli, S.; Oliveira, V.; Calabresi, P.; Maia, L.F.; Pisani, A. Does SARS-Cov-2 invade the brain? Translational lessons from animal models. Eur. J. Neurol. 2020, 27, 1764–1773. [Google Scholar] [CrossRef]
- Badaut, J.; Adami, A.; Huang, L.; Obenaus, A. Noninvasive magnetic resonance imaging stratifies injury severity in a rodent model of male juvenile traumatic brain injury. J. Neurosci. Res. 2019, 98, 129–140. [Google Scholar] [CrossRef]
- Vink, R. Large animal models of traumatic brain injury. J. Neurosci. Res. 2018, 96, 527–535. [Google Scholar] [CrossRef]
- Ackermans, N.L.; Varghese, M.; Wicinski, B.; Torres, J.; De Gasperi, R.; Pryor, D.; Elder, G.A.; Gama Sosa, M.A.; Reidenberg, J.S.; Williams, T.M.; et al. Unconventional animal models for traumatic brain injury and chronic traumatic encephalopathy. J. Neurosci. Res. 2021, 99, 2463–2477. [Google Scholar] [CrossRef] [PubMed]
- Grovola, M.R.; Paleologos, N.; Brown, D.P.; Tran, N.; Wofford, K.L.; Harris, J.P.; Browne, K.D.; Shewokis, P.A.; Wolf, J.A.; Cullen, D.K.; et al. Diverse changes in microglia morphology and axonal pathology during the course of 1 year after mild traumatic brain injury in pigs. Brain Pathol. 2021, 31, e12953. [Google Scholar] [CrossRef]
- Filipp, M.; Travis, B.; Henry, S.; Idzikowski, E.; Magnuson, S.; Loh, M.; Hellenbrand, D.; Hanna, A. Differences in neuroplasticity after spinal cord injury in varying animal models and humans. Neural Regen. Res. 2019, 14, 7. [Google Scholar] [CrossRef]
- Perucca, P.; Bahlo, M.; Berkovic, S.F. The genetics of epilepsy. Annu. Rev. Genom. Hum. Genet. 2020, 21, 205–230. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, S.L.; Law, T.H.; Volk, H.A.; Younis, C.; Casey, R.A.; Packer, R.M.A. Impact of canine epilepsy on judgement and attention biases. Sci. Rep. 2020, 10, 17719. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.A.; Sehgal, A. Anandamide metabolites protect against seizures through the TRP channel water witch in Drosophila melanogaster. Cell Rep. 2020, 31, 107710. [Google Scholar] [CrossRef]
- Dissel, S. Drosophila as a model to study the relationship between sleep, plasticity, and memory. Front. Physiol. 2020, 11, 533. [Google Scholar] [CrossRef]
- Toda, H.; Williams, J.A.; Gulledge, M.; Sehgal, A. A sleep-inducing gene, nemuri, links sleep and immune function in Drosophila. Science 2019, 363, 509–515. [Google Scholar] [CrossRef]
- Lei, Z.; Henderson, K.; Keleman, K. A neural circuit linking learning and sleep in Drosophila long-term memory. Nat. Commun. 2022, 13, 609. [Google Scholar] [CrossRef]
- Shi, L.; Su, B. A transgenic monkey model for the study of human brain evolution. Zool. Res. 2019, 40, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Hoffe, B.; Holahan, M.R. The use of pigs as a translational model for studying neurodegenerative diseases. Front. Physiol. 2019, 10, 838. [Google Scholar] [CrossRef]
- Calvo-Rodriguez, M.; Hou, S.S.; Snyder, A.C.; Kharitonova, E.K.; Russ, A.N.; Das, S.; Fan, Z.; Muzikansky, A.; Garcia-Alloza, M.; Serrano-Pozo, A.; et al. Increased mitochondrial calcium levels associated with neuronal death in a mouse model of Alzheimer’s disease. Nat. Commun. 2020, 11, 2146. [Google Scholar] [CrossRef]
- Kin, K.; Yasuhara, T.; Kameda, M.; Date, I. Animal models for parkinson’s disease research: Trends in the 2000s. Int. J. Mol. Sci. 2019, 20, 5402. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.L.; Lones, M.A.; Bedder, M.; Currie, P.D.; Smith, S.L.; Pownall, M.E. Machine learning discriminates a movement disorder in a zebrafish model of Parkinson’s disease. Dis. Model. Mech. 2020, 13, dmm045815. [Google Scholar] [CrossRef] [PubMed]
- Barnhill, L.M.; Murata, H.; Bronstein, J.M. Studying the pathophysiology of Parkinson’s disease using zebrafish. Biomedicines 2020, 8, 197. [Google Scholar] [CrossRef]
- Kalyn, M.; Hua, K.; Mohd Noor, S.; Wong, C.E.D.; Ekker, M. Comprehensive analysis of neurotoxin-induced ablation of dopaminergic neurons in zebrafish larvae. Biomedicines 2019, 8, 1. [Google Scholar] [CrossRef]
- Vaz, R.L.; Outeiro, T.F.; Ferreira, J.J. Zebrafish as an animal model for drug discovery in parkinson’s disease and other movement disorders: A systematic review. Front. Neurol. 2018, 9, 347. [Google Scholar] [CrossRef]
- Zhao, Y.; Han, Y.; Wang, Z.; Chen, T.; Qian, H.; He, J.; Li, J.; Han, B.; Wang, T. Rosmarinic acid protects against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced dopaminergic neurotoxicity in zebrafish embryos. Toxicol. Vitr. 2020, 65, 104823. [Google Scholar] [CrossRef]
- Dongjie, S.; Rajendran, R.S.; Xia, Q.; She, G.; Tu, P.; Zhang, Y.; Liu, K. Neuroprotective effects of Tongtian oral liquid, a Traditional Chinese Medicine in the Parkinson’s disease-induced zebrafish model. Biomed. Pharmacother. 2022, 148, 112706. [Google Scholar] [CrossRef]
- Kodera, K.; Matsui, H. Zebrafish, medaka and turquoise killifish for understanding human neurodegenerative/neurodevelopmental disorders. Int. J. Mol. Sci. 2022, 23, 1399. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Lei, L.; Tian, L.; Hou, F.; Roper, C.; Ge, X.; Zhao, Y.; Chen, Y.; Dong, Q.; Tanguay, R.L.; et al. Developmental and behavioral alterations in zebrafish embryonically exposed to valproic acid (VPA): An aquatic model for autism. Neurotoxicol. Teratol. 2018, 66, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Arafat, E.A.; Shabaan, D.A. The possible neuroprotective role of grape seed extract on the histopathological changes of the cerebellar cortex of rats prenatally exposed to Valproic Acid: Animal model of autism. Acta Histochem. 2019, 121, 841–851. [Google Scholar] [CrossRef]
- Baek, H.; Sariev, A.; Lee, S.; Dong, S.-Y.; Royer, S.; Kim, H. Deep cerebellar low-intensity focused ultrasound stimulation restores interhemispheric balance after ischemic stroke in mice. IEEE Trans. Neural Syst. Rehabil. Eng. 2020, 28, 2073–2079. [Google Scholar] [CrossRef]
- Chen, C.-D.; Kim, S.-K.; HwangBo, G. Effect of Acute Phase Pain Control Using TENS on Pain Relief in Knee Osteoarthritis in a Rat Model. J. Korean Soc. Phys. Med. 2021, 16, 15–20. [Google Scholar] [CrossRef]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Carvalho, C.; Silva, C.; Coelho, T.; Gamboa, Ó.; Ferreira, A. Functional neurorehabilitation in dogs with an incomplete recovery 3 months following intervertebral disc surgery: A case series. Animals 2021, 11, 2442. [Google Scholar] [CrossRef] [PubMed]
- Iijima, H.; Eguchi, R.; Shimoura, K.; Yamada, K.; Aoyama, T.; Takahashi, M. Transcutaneous electrical nerve stimulation improves stair climbing capacity in people with knee osteoarthritis. Sci. Rep. 2020, 10, 7294. [Google Scholar] [CrossRef]
- Lee, J.E.; Anderson, C.M.; Perkhounkova, Y.; Sleeuwenhoek, B.M.; Louison, R.R. Transcutaneous electrical nerve stimulation reduces resting pain in head and neck cancer patients. Cancer Nurs. 2019, 42, 218–228. [Google Scholar] [CrossRef]
- Hsiao, I.-H.; Liao, H.-Y.; Lin, Y. Optogenetic modulation of electroacupuncture analgesia in a mouse inflammatory pain model. Sci. Rep. 2022, 12, 9067. [Google Scholar] [CrossRef]
- Truong, B.T.; Artinger, K.B. The power of zebrafish models for understanding the co-occurrence of craniofacial and limb disorders. Genesis 2021, 59, e23407. [Google Scholar] [CrossRef]
- Bergen, D.J.M.; Tong, Q.; Shukla, A.; Newham, E.; Zethof, J.; Lundberg, M.; Ryan, R.; Youlten, S.E.; Frysz, M.; Croucher, P.I.; et al. Regenerating zebrafish scales express a subset of evolutionary conserved genes involved in human skeletal disease. BMC Biol. 2022, 20, 21. [Google Scholar] [CrossRef]
- Yonekura, M.; Kondoh, N.; Han, C.; Toyama, Y.; Ohba, T.; Ono, K.; Itagaki, S.; Tomita, H.; Murakami, M. Medaka as a model for ECG analysis and the effect of verapamil. J. Pharmacol. Sci. 2018, 137, 55–60. [Google Scholar] [CrossRef]
- Marzęda, P.; Drozd, M.; Tchórz, M.; Kisiel, K. Importance of early intervention in verapamil overdose—Case Report and antidotes review. J. Pre-Clin. Clin. Res. 2021, 15, 142–147. [Google Scholar] [CrossRef]
- Ruetten, H.; Vezina, C.M. Relevance of dog as an animal model for urologic diseases. In Progress in Molecular Biology and Translational Science; Tao, Y.-X., Ed.; Academic Press: Oxford, UK, 2022; pp. 35–65. [Google Scholar]
- Gajski, G.; Žegura, B.; Ladeira, C.; Pourrut, B.; Del Bo’, C.; Novak, M.; Sramkova, M.; Milić, M.; Gutzkow, K.B.; Costa, S.; et al. The comet assay in animal models: From bugs to whales—(Part 1 Invertebrates). Mutat. Res. Mutat. Res. 2019, 779, 82–113. [Google Scholar] [CrossRef]
- Bouriga, N.; Mili, S.; Bahri, W.R.; Jammeli, B.; Ben-Attia, M.; Quignard, J.-P.; Trabelsi, M. Skin Wound Healing Potential and Antioxidant Effect of Hyaluronic Acid Extracted from Mytilus galloprovincialis and Crassostrea gigas. Pharm. Chem. J. 2022, 56, 381–386. [Google Scholar] [CrossRef]
- Imperadore, P.; Uckermann, O.; Galli, R.; Steiner, G.; Kirsch, M.; Fiorito, G. Nerve regeneration in the cephalopod mollusc Octopus vulgaris: Label-free multiphoton microscopy as a tool for investigation. J. R. Soc. Interface 2018, 15, 20170889. [Google Scholar] [CrossRef] [PubMed]
- NIH When Are Alternatives to Animals Used in Research? Available online: https://grants.nih.gov/grants/policy/air/alternatives (accessed on 10 March 2023).
- Conroy, G. These Are the 10 Best Countries for Life Sciences Research. Available online: https://www.nature.com/nature-index/news-blog/ten-best-countries-life-sciences-research-rankings (accessed on 10 March 2023).
- Van Norman, G.A. Limitations of animal studies for predicting toxicity in clinical trials. JACC Basic Transl. Sci. 2019, 4, 845–854. [Google Scholar] [CrossRef]
- Hernandez, J. The FDA No Longer Requires All Drugs to Be Tested on Animals before Human Trials. Available online: https://www.npr.org/2023/01/12/1148529799/fda-animal-testing-pharmaceuticals-drug-development (accessed on 10 March 2023).
- Canadian Council on Animal Care. CCAC Animal Data Report 2019. Available online: https://speakingofresearch.files.wordpress.com/2021/08/canada-2019.pdf (accessed on 10 March 2023).
- Xinaris, C. Organoids for replacement therapy: Expectations, limitations and reality. Curr. Opin. Organ Transplant. 2019, 24, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Driehuis, E.; Kretzschmar, K.; Clevers, H. Establishment of patient-derived cancer organoids for drug-screening applications. Nat. Protoc. 2020, 15, 3380–3409. [Google Scholar] [CrossRef]
- Matsui, T.K.; Tsuru, Y.; Hasegawa, K.; Kuwako, K. Vascularization of human brain organoids. Stem Cells 2021, 39, 1017–1024. [Google Scholar] [CrossRef]
- Homan, K.A.; Gupta, N.; Kroll, K.T.; Kolesky, D.B.; Skylar-Scott, M.; Miyoshi, T.; Mau, D.; Valerius, M.T.; Ferrante, T.; Bonventre, J.V.; et al. Flow-enhanced vascularization and maturation of kidney organoids in vitro. Nat. Methods 2019, 16, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Wörsdörfer, P.; Dalda, N.; Kern, A.; Krüger, S.; Wagner, N.; Kwok, C.K.; Henke, E.; Ergün, S. Generation of complex human organoid models including vascular networks by incorporation of mesodermal progenitor cells. Sci. Rep. 2019, 9, 15663. [Google Scholar] [CrossRef] [PubMed]
- Rawal, P.; Tripathi, D.M.; Ramakrishna, S.; Kaur, S. Prospects for 3D bioprinting of organoids. Bio-Des. Manuf. 2021, 4, 627–640. [Google Scholar] [CrossRef]
- Briceño, J. Artificial intelligence and organ transplantation: Challenges and expectations. Curr. Opin. Organ Transpl. 2020, 25, 393–398. [Google Scholar] [CrossRef]
- Fattahi Sani, M.; Ascione, R.; Dogramadzi, S. Mapping surgeons hand/finger movements to surgical tool motion during conventional microsurgery using machine learning. J. Med. Robot. Res. 2021, 6, 2150004. [Google Scholar] [CrossRef]
- Azer, S.A. Deep learning with convolutional neural networks for identification of liver masses and hepatocellular carcinoma: A systematic review. World J. Gastrointest Oncol. 2019, 11, 1218–1230. [Google Scholar] [CrossRef]
- Wang, S.; Kang, B.; Ma, J.; Zeng, X.; Xiao, M.; Guo, J.; Cai, M.; Yang, J.; Li, Y.; Meng, X.; et al. A deep learning algorithm using CT images to screen for Corona virus disease (COVID-19). Eur. Radiol. 2021, 31, 6096–6104. [Google Scholar] [CrossRef]
- Gurovich, Y.; Hanani, Y.; Bar, O.; Nadav, G.; Fleischer, N.; Gelbman, D.; Basel-Salmon, L.; Krawitz, P.M.; Kamphausen, S.B.; Zenker, M.; et al. Identifying facial phenotypes of genetic disorders using deep learning. Nat. Med. 2019, 25, 60–64. [Google Scholar] [CrossRef]
- Feighelstein, M.; Shimshoni, I.; Finka, L.R.; Luna, S.P.L.; Mills, D.S.; Zamansky, A. Automated recognition of pain in cats. Sci. Rep. 2022, 12, 9575. [Google Scholar] [CrossRef]
- Herrmann, K. Beyond the 3Rs: Expanding the use of human-relevant replacement methods in biomedical research. ALTEX 2019, 36, 343–352. [Google Scholar] [CrossRef]
- Pound, P.; Ritskes-Hoitinga, M. Is it possible to overcome issues of external validity in preclinical animal research? Why most animal models are bound to fail. J. Transl. Med. 2018, 16, 304. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, C.; Gaspar, A.; Knight, A.; Vicente, L. Ethical and scientific pitfalls concerning laboratory research with non-human primates, and possible solutions. Animals 2018, 9, 12. [Google Scholar] [CrossRef] [PubMed]
| Country | Year | Specie | Percentage (%) | Reference |
|---|---|---|---|---|
| European Union | 2019 | Rodents | 61.9 | [25] |
| Fish | 24.6 | |||
| Birds | 6.2 | |||
| Amphibians | 0.5 | |||
| Cephalopods | 0.2 | |||
| Dogs | 0.1 | |||
| Non-human primates | 0.07 | |||
| Other mammals | 6.5 | |||
| Canada | 2020 | Birds | 50.0 | [26] |
| Rodents | 24.5 | |||
| Fish | 11.7 | |||
| Cattle | 11.3 | |||
| Amphibians | 1.1 | |||
| Pigs | 0.4 | |||
| Dogs | 0.2 | |||
| Non-human primates | 0.1 | |||
| Reptiles | 0.1 | |||
| Other animals | 0.5 | |||
| United Kingdom 1 | 2021 | Mice | 68.2 | [27] |
| Fish | 12.9 | |||
| Rats | 6.5 | |||
| Birds | 8 | |||
| Dogs | 0.14 | |||
| Non-human primates | 0.09 | |||
| Cats | 0.01 | |||
| Other animals | 3.3 | |||
| United States 2 | 2019 | Guinea pigs | 23 | [28] |
| Rabbits | 18 | |||
| Hamsters | 12 | |||
| Non-human primates | 9 | |||
| Dogs | 7 | |||
| Pigs | 6 | |||
| Cats | 2 | |||
| Sheep | 2 | |||
| Other species | 21 | |||
| South Korea | 2017 | Rodents | 91.8 | [29] |
| Fish | 3.3 | |||
| Birds | 2.3 | |||
| Rabbits | 1 | |||
| Non-human primates | 0.08 | |||
| Amphibians | 0.07 | |||
| Other species | 1.21 | |||
| Total | 3,085,259 |
| Country | Number of Animals | References |
|---|---|---|
| United States | 20,000,000–24,000,000 | [30,31,32] |
| China | 16,000,000 | |
| Japan | 11,000,000 | |
| European Union | 9,400,000 | |
| Australia | 6,700,000 | |
| Canada | 5,067,778 | |
| South Korea | 4,141,433 | |
| United Kingdom | 3,300,000 | |
| Norway | 2,282,710 | |
| Germany | 2,151,805 | |
| France | 1,865,403 | |
| Spain | 761,012 | |
| Mexico | 685,315 | |
| Switzerland | 556,107 | |
| Belgium | 437,275 | |
| New Zealand | 240,000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domínguez-Oliva, A.; Hernández-Ávalos, I.; Martínez-Burnes, J.; Olmos-Hernández, A.; Verduzco-Mendoza, A.; Mota-Rojas, D. The Importance of Animal Models in Biomedical Research: Current Insights and Applications. Animals 2023, 13, 1223. https://doi.org/10.3390/ani13071223
Domínguez-Oliva A, Hernández-Ávalos I, Martínez-Burnes J, Olmos-Hernández A, Verduzco-Mendoza A, Mota-Rojas D. The Importance of Animal Models in Biomedical Research: Current Insights and Applications. Animals. 2023; 13(7):1223. https://doi.org/10.3390/ani13071223
Chicago/Turabian StyleDomínguez-Oliva, Adriana, Ismael Hernández-Ávalos, Julio Martínez-Burnes, Adriana Olmos-Hernández, Antonio Verduzco-Mendoza, and Daniel Mota-Rojas. 2023. "The Importance of Animal Models in Biomedical Research: Current Insights and Applications" Animals 13, no. 7: 1223. https://doi.org/10.3390/ani13071223
APA StyleDomínguez-Oliva, A., Hernández-Ávalos, I., Martínez-Burnes, J., Olmos-Hernández, A., Verduzco-Mendoza, A., & Mota-Rojas, D. (2023). The Importance of Animal Models in Biomedical Research: Current Insights and Applications. Animals, 13(7), 1223. https://doi.org/10.3390/ani13071223










