Fecal Microbiota, Bile Acids, Sterols, and Fatty Acids in Dogs with Chronic Enteropathy Fed a Home-Cooked Diet Supplemented with Coconut Oil
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Diets
2.3. Study Design, Clinical Evaluations, and Blood and Fecal Sample Collection
2.4. Chemical Analyses
2.5. Microbial Analyses
2.6. Statistical Analysis
3. Results
3.1. Animals
3.2. Fecal Chemical Parameters
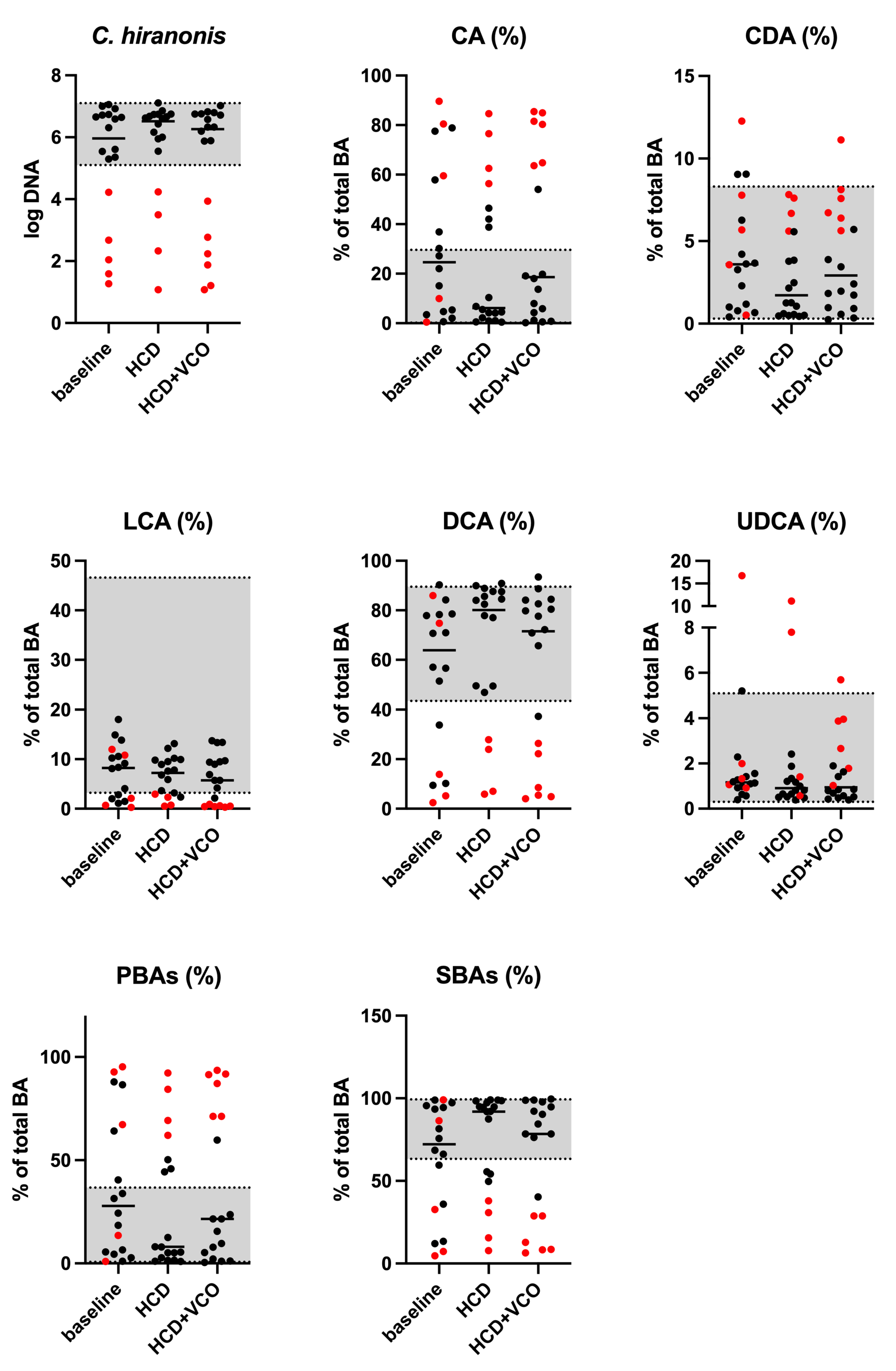
3.3. Fecal Microbial Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Makielski, K.; Cullen, J.; O’Connor, A.; Jergens, A.E. Narrative Review of Therapies for Chronic Enteropathies in Dogs and Cats. J. Vet. Intern. Med. 2019, 33, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Kathrani, A. Dietary and Nutritional Approaches to the Management of Chronic Enteropathy in Dogs and Cats. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Rudinsky, A.J.; Rowe, J.C.; Parker, V.J. Nutritional Management of Chronic Enteropathies in Dogs and Cats. J. Am. Vet. Med. Assoc. 2018, 253, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Odle, J. New Insights into the Utilization of Medium-Chain Triglycerides by the Neonate: Observations from a Piglet Model. J. Nutr. 1997, 127, 1061–1067. [Google Scholar] [CrossRef]
- Marten, B.; Pfeuffer, M.; Schrezenmeir, J. Medium-Chain Triglycerides. Int. Dairy J. 2006, 16, 1374–1382. [Google Scholar] [CrossRef]
- Chwen, L.T.; Foo, H.L.; Thanh, N.T.; Choe, D.W. Growth Performance, Plasma Fatty Acids, Villous Height and Crypt Depth of Preweaning Piglets Fed with Medium Chain Triacylglycerol. Asian-Australasian J. Anim. Sci. 2013, 26, 700–704. [Google Scholar] [CrossRef]
- Rial, S.A.; Karelis, A.D.; Bergeron, K.F.; Mounier, C. Gut Microbiota and Metabolic Health: The Potential Beneficial Effects of a Medium Chain Triglyceride Diet in Obese Individuals. Nutrients 2016, 8, 281. [Google Scholar] [CrossRef]
- Desai, A.P.; Guvenc, B.H.; Carachi, R. Evidence for Medium Chain Triglycerides in the Treatment of Primary Intestinal Lymphangiectasia. Eur. J. Pediatr. Surg. 2009, 19, 241–245. [Google Scholar] [CrossRef]
- Vignes, S.; Bellanger, J. Primary Intestinal Lymphangiectasia (Waldmann’s Disease). Orphanet J. Rare Dis. 2008, 3, 5. [Google Scholar] [CrossRef]
- Willard, M.D.; Helman, G.; Fradkin, J.M.; Becker, T.; Brown, R.M.; Lewis, B.C.; Weeks, B.R. Intestinal Crypt Lesions Associated with Protein-Losing Enteropathy in the Dog. J. Vet. Intern. Med. 2000, 14, 298–307. [Google Scholar] [CrossRef]
- Olson, N.; Zimmer, J. Protein-Losing Enteropathy Secondary to Intestinal Lymphangiectasia in a Dog. J. Am. Vet. Med. Assoc. 1978, 173, 271–274. [Google Scholar]
- Zhang, J.Y.; Baek, D.H.; Kim, I.H. Effect of Dietary Supplemental Medium Chain Fatty Acids Instead of Antibiotics on the Growth Performance, Digestibility and Blood Profiles in Growing Pigs. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1946–1951. [Google Scholar] [CrossRef]
- Hanczakowska, E. The Use of Medium-Chain Fatty Acids in Piglet Feeding-A Review. Ann. Anim. Sci. 2017, 17, 967–977. [Google Scholar] [CrossRef]
- Lauridsen, C. Effects of Dietary Fatty Acids on Gut Health and Function of Pigs Pre-And Post-Weaning. J. Anim. Sci. 2020, 98, skaa086. [Google Scholar] [CrossRef]
- Puyalto, M.; Sol, C.; Mallo, J.J. Coconut oil: A new option for controlling pig pathogens. Pig Prog. 2016, 32, 35–36. [Google Scholar]
- Zentek, J.; Buchheit-Renko, S.; Ferrara, F.; Vahjen, W.; Van Kessel, A.G.; Pieper, R. Nutritional and Physiological Role of Medium-Chain Triglycerides and Medium-Chain Fatty Acids in Piglets. Anim. Health Res. Rev. 2011, 12, 83–93. [Google Scholar] [CrossRef]
- Deen, A.; Visvanathan, R.; Wickramarachchi, D.; Marikkar, N.; Nammi, S.; Jayawardana, B.C.; Liyanage, R. Chemical Composition and Health Benefits of Coconut Oil: An Overview. J. Sci. Food Agric. 2021, 101, 2182–2193. [Google Scholar] [CrossRef]
- Dubois, V.; Breton, S.; Linder, M.; Fanni, J.; Parmentier, M. Fatty Acid Profiles of 80 Vegetable Oils with Regard to Their Nutritional Potential. Eur. J. Lipid Sci. Technol. 2007, 109, 710–732. [Google Scholar] [CrossRef]
- Kappally, S.; Shirwaikar, A.; Shirwaikar, A. COCONUT OIL a Review of Potential Applications. Hygeia. J. Drugs Med. 2015, 7, 34–41. [Google Scholar] [CrossRef]
- Wallace, T.C. Health Effects of Coconut Oil—A Narrative Review of Current Evidence. J. Am. Coll. Nutr. 2019, 38, 97–107. [Google Scholar] [CrossRef]
- Rolinec, M.; Medo, J.; Gábor, M.; Miluchová, M.; Bíro, D.; Šimko, M.; Juráček, M.; Hanušovský, O.; Schubertová, Z.; Gálik, B. The Effect of Coconut Oil Addition to Feed of Pigs on Rectal Microbial Diversity and Bacterial Abundance. Animals 2020, 10, 1764. [Google Scholar] [CrossRef] [PubMed]
- Braundmeier-Fleming, A.G.; Skenandore, C.S.; Gil, L.; Jacobsen, V.; Cregger, M.; Badger, T.; Karr, M.; Wu, G.; Smith, S.B.; Newell-Fugate, A.E. Dietary Substitution of Soybean Oil with Coconut Oil in the Absence of Dietary Antibiotics Supports Growth Performance and Immune Function in Nursery and Grower Pigs. J. Anim. Sci. Biotechnol. 2020, 11, 27. [Google Scholar] [CrossRef] [PubMed]
- Beynen, A.C.; Kappert, H.J.; Lemmens, A.G.; Van Dongen, A.M. Plasma Lipid Concentrations, Macronutrient Digestibility and Mineral Absorption in Dogs Fed a Dry Food Containing Medium-Chain Triglycerides. J. Anim. Physiol. Anim. Nutr. 2002, 86, 306–312. [Google Scholar] [CrossRef]
- Rutz, G.M.; Steiner, J.M.; Bauer, J.E.; Williams, D.A. Effects of Exchange of Dietary Medium Chain Triglycerides for Long-Chain Triglycerides on Serum Biochemical Variables and Subjectively Assessed Well-Being of Dogs with Exocrine Pancreatic Insufficiency. Am. J. Vet. Res. 2004, 65, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Remillard, R.L.; Thatcher, C.D. Dietary and Nutritional Management of Gastrointestinal Diseases. Vet. Clin. N. Am. Small Anim. Pract. 1989, 19, 797–816. [Google Scholar] [CrossRef]
- Van Dongen, A.M.; Stokhof, A.A.; Geelen, M.J.H.; Beynen, A.C. An Observation: The High Intake of Medium-Chain Triglycerides Elevates Plasma Cholesterol in Dogs. Folia Vet. 2000, 44, 173–174. [Google Scholar]
- Allenspach, K.; Wieland, B.; Gröne, A.; Gaschen, F. Chronic Enteropathies in Dogs: Evaluation of Risk Factors for Negative Outcome. J. Vet. Intern. Med. 2007, 21, 700–708. [Google Scholar] [CrossRef]
- FEDIAF Scientific Advisory Board Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs. 2021, pp. 1–98. Available online: https://europeanpetfood.org/wp-content/uploads/2022/03/Updated-Nutritional-Guidelines.pdf (accessed on 11 November 2022).
- Laflamme, D.P. Development and Validation of a Body Condition Score System for Dogs. Canine Pract. 1997, 22, 10–15. [Google Scholar]
- AOAC. Official Methods of Analysis of AOAC International, 17th ed.; Association of Analytical Communities: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Folch, J.; Lees, M.; Stanley, G. A Simple Method for the Isolation and Purification of Total Lipids from Animal Tissue. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Campidonico, L.; Toral, P.G.; Priolo, A.; Luciano, G.; Valenti, B.; Hervás, G.; Frutos, P.; Copani, G.; Ginane, C.; Niderkorn, V. Fatty Acid Composition of Ruminal Digesta and Longissimus Muscle from Lambs Fed Silage Mixtures Including Red Clover, Sainfoin, and Timothy12. J. Anim. Sci. 2016, 94, 1550–1560. [Google Scholar] [CrossRef]
- Galler, A.I.; Suchodolski, J.S.; Steiner, J.M.; Sung, C.H.; Hittmair, K.M.; Richter, B.; Burgener, I.A. Microbial Dysbiosis and Fecal Metabolomic Perturbations in Yorkshire Terriers with Chronic Enteropathy. Sci. Rep. 2022, 12, 12977. [Google Scholar] [CrossRef]
- AlShawaqfeh, M.K.; Wajid, B.; Minamoto, Y.; Markel, M.; Lidbury, J.A.; Steiner, J.M.; Serpedin, E.; Suchodolski, J.S. A Dysbiosis Index to Assess Microbial Changes in Fecal Samples of Dogs with Chronic Inflammatory Enteropathy. FEMS Microbiol. Ecol. 2017, 93, fix136. [Google Scholar] [CrossRef]
- Toresson, L.; Steiner, J.M.; Spodsberg, E.; Olmedal, G.; Suchodolski, J.S.; Lidbury, J.A.; Spillmann, T. Effects of Oral versus Parenteral Cobalamin Supplementation on Methylmalonic Acid and Homocysteine Concentrations in Dogs with Chronic Enteropathies and Low Cobalamin Concentrations. Vet. J. 2019, 243, 8–14. [Google Scholar] [CrossRef]
- Berk, B.A.; Packer, R.M.A.; Fritz, J.; Volk, H.A. Oral Palatability Testing of a Medium-Chain Triglyceride Oil Supplement in a Cohort of Healthy Dogs in a Non-Clinical Setting Oral Palatability of Medium-Chain Triglyceride Oil in Healthy Dogs. Animals 2022, 12, 1639. [Google Scholar] [CrossRef]
- Allenspach, K.; Culverwell, C.; Chan, D. Long-Term Outcome in Dogs with Chronic Enteropathies: 203 Cases. Vet. Rec. 2016, 178, 368. [Google Scholar] [CrossRef]
- Berghoff, N.; Suchodolski, J.S.; Steiner, J.M. Association between Serum Cobalamin and Methylmalonic Acid Concentrations in Dogs. Vet. J. 2012, 191, 306–311. [Google Scholar] [CrossRef]
- Kather, S.; Grützner, N.; Kook, P.H.; Dengler, F.; Heilmann, R.M. Review of Cobalamin Status and Disorders of Cobalamin Metabolism in Dogs. J. Vet. Intern. Med. 2020, 34, 13–28. [Google Scholar] [CrossRef]
- Jergens, A.E.; Heilmann, R.M. Canine Chronic Enteropathy—Current State-of-the-Art and Emerging Concepts. Front. Vet. Sci. 2022, 9, 923013. [Google Scholar] [CrossRef]
- Wenzl, H.H.; Fine, K.D.; Schiller, L.R.; Fordtran, J.S. Determinants of Decreased Fecal Consistency in Patients with Diarrhea. Gastroenterology 1995, 108, 1729–1738. [Google Scholar] [CrossRef]
- Eherer, A.J.; Ana, C.A.S.; Porter, J.; Fordtran, J.S. Effect of Psyllium, Calcium Polycarbophil, and Wheat Bran on Secretory Diarrhea Induced by Phenolphthalein. Gastroenterology 1993, 104, 1007–1012. [Google Scholar] [CrossRef]
- Heller, S.N.; Hackler, L.R. Water-Holding Capacity of Various Sources of Plant Fiber. J. Food Sci. 1977, 42, 1137. [Google Scholar] [CrossRef]
- Robertson, J.A.; Eastwood, M.A. An Examination of Factors Which May Affect the Water Holding Capacity of Dietary Fibre. Br. J. Nutr. 1981, 45, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Panasevich, M.R.; Rossoni Serao, M.C.; de Godoy, M.R.C.; Swanson, K.S.; Guérin-Deremaux, L.; Lynch, G.L.; Wils, D.; Fahey, G.C.; Dilger, R.N. Potato Fiber as a Dietary Fiber Source in Dog Foods. J. Anim. Sci. 2013, 91, 5344–5352. [Google Scholar] [CrossRef] [PubMed]
- Meyer, H.; Zentek, J.; Habernoll, H.; Maskell, I. Digestibility and Compatibility of Mixed Diets and Faecal Consistency in Different Breeds of Dog. J. Vet. Med. Ser. A 1999, 46, 155–165. [Google Scholar] [CrossRef]
- Deschamps, C.; Humbert, D.; Zentek, J.; Denis, S.; Priymenko, N.; Apper, E.; Blanquet-Diot, S. From Chihuahua to Saint-Bernard: How Did Digestion and Microbiota Evolve with Dog Sizes. Int. J. Biol. Sci. 2022, 18, 5086–5102. [Google Scholar] [CrossRef]
- Weber, M. Influence of Size on the Dog’s Digestive Function. Bull. Acad. Vet. Fr. 2006, 159, 327–332. [Google Scholar] [CrossRef]
- Hill, R.C.; Burrows, C.F.; Ellison, G.W.; Finke, M.D.; Huntington, J.L.; Bauer, J.E. Water Content of Faeces Is Higher in the Afternoon than in the Morning in Morning-Fed Dogs Fed Diets Containing Texturised Vegetable Protein from Soya. Br. J. Nutr. 2011, 106 (Suppl. S1), 202–205. [Google Scholar] [CrossRef]
- Rolfe, V.E.; Adams, C.A.; Butterwick, R.F.; Batt, R.M. Relationship between Faecal Character and Intestinal Transit Time in Normal Dogs and Diet-Sensitive Dogs. J. Small Anim. Pract. 2002, 43, 290–294. [Google Scholar] [CrossRef]
- Martineau, B.; Laflamme, D.P. Effect of Diet on Markers of Intestinal Health in Dogs. Res. Vet. Sci. 2002, 72, 223–227. [Google Scholar] [CrossRef]
- Rolfe, V. Colonic Fluid and Electrolyte Transport in Health and Disease. Vet. Clin. N. Am. Small Anim. Pract. 1999, 29, 577–588. [Google Scholar] [CrossRef]
- Rutgers, H.; Batt, R.; Proud, F.; Sørensen, S.; Elwood, C.; Petrie, G.; Matthewman, L.; Forster-van Hijfte, M.; Boswood, A.; Entwistle, M.; et al. Intestinal Permeability and Function in Dogs with Small Intestinal Bacterial Overgrowth. J. Small Anim. Pract. 1996, 37, 428–434. [Google Scholar] [CrossRef]
- Badiani, A.; Nanni, N.; Gatta, P.P.; Tolomelli, B.; Manfredini, M. Nutrient Profile of Horsemeat. J. Food Compos. Anal. 1997, 10, 254–269. [Google Scholar] [CrossRef]
- Marino, R.; della Malva, A.; Maggiolino, A.; De Palo, P.; D’angelo, F.; Lorenzo, J.M.; Sevi, A.; Albenzio, M. Nutritional Profile of Donkey and Horse Meat: Effect of Muscle and Aging Time. Animals 2022, 12, 746. [Google Scholar] [CrossRef]
- Ramírez, M.; Amate, L.; Gil, A. Absorption and Distribution of Dietary Fatty Acids from Different Sources. Early Hum. Dev. 2001, 65, 95–101. [Google Scholar] [CrossRef]
- Jensen, G.L.; McGarvey, N.; Taraszewski, R.; Wixson, S.K.; Seidner, D.L.; Pai, T.; Yeh, Y.Y.; Lee, T.W.; Demichele, S.J. Lymphatic Absorption of Enterally Fed Structured Triacylglycerol vs Physical Mix in a Canine Model. Am. J. Clin. Nutr. 1994, 60, 518–524. [Google Scholar] [CrossRef]
- McCarty, M.F.; DiNicolantonio, J.J. Lauric Acid-Rich Medium-Chain Triglycerides Can Substitute for Other Oils in Cooking Applications and May Have Limited Pathogenicity. Open Heart 2016, 3, e000467. [Google Scholar] [CrossRef]
- Tolbert, M.K.; Murphy, M.; Gaylord, L.; Witzel-Rollins, A. Dietary Management of Chronic Enteropathy in Dogs. J. Small Anim. Pract. 2022, 63, 425–434. [Google Scholar] [CrossRef]
- Heersma, J.R.; Annegers, J.H. Effect of Bile Diversion on Fecal Fat and Nitrogen Excretion. Am. J. Physiol. Content 1948, 153, 143–147. [Google Scholar] [CrossRef]
- Piccione, G.; Fazio, F.; Giudice, E.; Grasso, F.; Caola, G. Blood Lipids, Fecal Fat and Chymotrypsin Excretion in the Dog: Influence of Age, Body Weight and Sex. J. Vet. Med. Sci. 2004, 66, 59–62. [Google Scholar] [CrossRef]
- Marx, F.R.; Trevizan, L.; Saad, F.M.O.B.; Lisenko, K.G.; Reis, J.S.; Kessler, A.M. Endogenous Fat Loss and True Total Tract Digestibility of Poultry Fat in Adult Dogs. J. Anim. Sci. 2017, 95, 2928–2935. [Google Scholar] [CrossRef]
- Bo-Linn, G.W.; Fordtran, J.S. Fecal Fat Concentration in Patients with Steatorrhea. Gastroenterology 1984, 87, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Fine, K.D.; Fordtran, J.S. The Effect of Diarrhea on Fecal Fat Excretion. Gastroenterology 1992, 102, 1936–1939. [Google Scholar] [CrossRef] [PubMed]
- Sabchuk, T.T.; Lima, D.C.; Bastos, T.S.; Oliveira, S.G.; Félix, A.P.; Maiorka, A. Crude Corn Oil as Dietary Fat Source for Dogs. Anim. Feed Sci. Technol. 2019, 247, 173–182. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Z.; Ding, J.; Ming, D.; Wang, W.; Jiang, Z.; Liu, L.; Wang, F. Effects of Dietary Fiber Content and Different Fiber-Rich Ingredients on Endogenous Loss of Fat and Fatty Acids in Growing Pigs. J. Anim. Sci. Biotechnol. 2019, 10, 42. [Google Scholar] [CrossRef]
- Sabchuk, T.T.; Risolia, L.W.; Souza, C.M.M.; Félix, A.P.; Maiorka, A.; Oliveira, S.G. Endogenous Fat Losses and True and Apparent Fat Digestibility in Adult and Growing Dogs Fed Diets Containing Poultry Offal Fat. J. Anim. Physiol. Anim. Nutr. 2020, 104, 1927–1937. [Google Scholar] [CrossRef]
- Clement, J. Nature and Importance of Endogenous Fatty Acids during Intestinal Absorption of Fats. World Rev. Nutr. Diet. 1975, 21, 281–307. [Google Scholar] [CrossRef]
- Da Silva, S.A.; Sampaio, G.R.; da Silva Torres, E.A.F. Phytosterols Content in Vegetable Oils of Brazil: Coconut, Safflower, Linseed and Evening Primrose. Braz. Arch. Biol. Technol. 2020, 63, 1–8. [Google Scholar] [CrossRef]
- Honneffer, J.B. Microbiota and Metabolomic Changes Across Various Canine Gastrointestinal Diseases. Ph.D. Thesis, Texas A & M University, College Station, TX, USA, 2017; pp. 1–239. [Google Scholar]
- Cuevas-Tena, M.; Alegría, A.; Lagarda, M.J. Relationship between Dietary Sterols and Gut Microbiota: A Review. Eur. J. Lipid Sci. Technol. 2018, 120, 1800054. [Google Scholar] [CrossRef]
- Blake, A.B.; Guard, B.C.; Honneffer, J.B.; Lidbury, J.A.; Steiner, J.M.; Suchodolski, J.S. Altered Microbiota, Fecal Lactate, and Fecal Bile Acids in Dogs with Gastrointestinal Disease. PLoS ONE 2019, 14, e0224454. [Google Scholar] [CrossRef]
- Honneffer, J.; Guard, B.; Steiner, J.M.; Suchodolski, J.S. Mo1805 Untargeted Metabolomics Reveals Disruption within Bile Acid, Cholesterol, and Tryptophan Metabolic Pathways in Dogs with Idiopathic Inflammatory Bowel Disease. Gastroenterology 2015, 148, S-715. [Google Scholar] [CrossRef]
- Pezzali, J.G.; Shoveller, A.K.; Ellis, J. Examining the Effects of Diet Composition, Soluble Fiber, and Species on Total Fecal Excretion of Bile Acids: A Meta-Analysis. Front. Vet. Sci. 2021, 8, 1162. [Google Scholar] [CrossRef]
- Ziese, A.L.; Suchodolski, J.S. Impact of Changes in Gastrointestinal Microbiota in Canine and Feline Digestive Diseases. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 155–169. [Google Scholar] [CrossRef]
- Giaretta, P.R.; Rech, R.R.; Guard, B.C.; Blake, A.B.; Blick, A.K.; Steiner, J.M.; Lidbury, J.A.; Cook, A.K.; Hanifeh, M.; Spillmann, T.; et al. Comparison of Intestinal Expression of the Apical Sodium-Dependent Bile Acid Transporter between Dogs with and without Chronic Inflammatory Enteropathy. J. Vet. Intern. Med. 2018, 32, 1918–1926. [Google Scholar] [CrossRef]
- Pilla, R.; Suchodolski, J.S. The Gut Microbiome of Dogs and Cats, and the Influence of Diet. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 605–621. [Google Scholar] [CrossRef]
- Minamoto, Y.; Minamoto, T.; Isaiah, A.; Sattasathuchana, P.; Buono, A.; Rangachari, V.R.; McNeely, I.H.; Lidbury, J.; Steiner, J.M.; Suchodolski, J.S. Fecal Short-Chain Fatty Acid Concentrations and Dysbiosis in Dogs with Chronic Enteropathy. J. Vet. Intern. Med. 2019, 33, 1608–1618. [Google Scholar] [CrossRef]
- Schmidt, M.; Unterer, S.; Suchodolski, J.S.; Honneffer, J.B.; Guard, B.C.; Lidbury, J.A.; Rg, J.; Steiner, M.; Fritz, J.; Kö Lle, P. The Fecal Microbiome and Metabolome Differs between Dogs Fed Bones and Raw Food (BARF) Diets and Dogs Fed Commercial Diets. PLoS ONE 2018, 13, e0201279. [Google Scholar] [CrossRef]
- Bermingham, E.N.; Maclean, P.; Thomas, D.G.; Cave, N.J.; Young, W. Key Bacterial Families (Clostridiaceae, Erysipelotrichaceae and Bacteroidaceae) Are Related to the Digestion of Protein and Energy in Dogs. PeerJ 2017, 2017, e3019. [Google Scholar] [CrossRef]
- Dingeo, G.; Brito, A.; Samouda, H.; Iddir, M.; La Frano, M.R.; Bohn, T. Phytochemicals as Modifiers of Gut Microbial Communities. Food Funct. 2020, 11, 8444–8471. [Google Scholar] [CrossRef]
- Żółkiewicz, J.; Marzec, A.; Ruszczyński, M.; Feleszko, W. Postbiotics—A Step Beyond Pre- and Probiotics. Nutrients 2020, 12, 2189. [Google Scholar] [CrossRef]
- Pilla, R.; Gaschen, F.P.; Barr, J.W.; Olson, E.; Honneffer, J.; Guard, B.C.; Blake, A.B.; Villanueva, D.; Khattab, M.R.; AlShawaqfeh, M.K.; et al. Effects of Metronidazole on the Fecal Microbiome and Metabolome in Healthy Dogs. J. Vet. Intern. Med. 2020, 34, 1853–1866. [Google Scholar] [CrossRef]
- Wernimont, S.M.; Radosevich, J.; Jackson, M.I.; Ephraim, E.; Badri, D.V.; MacLeay, J.M.; Jewell, D.E.; Suchodolski, J.S. The Effects of Nutrition on the Gastrointestinal Microbiome of Cats and Dogs: Impact on Health and Disease. Front. Microbiol. 2020, 11, 1266. [Google Scholar] [CrossRef] [PubMed]

| Ingredients (g/100 g) | HCD | HCD + VCO |
|---|---|---|
| Potatoes | 59.5 | 59.5 |
| Horse meat | 29.7 | 29.7 |
| Zucchini | 8.5 | 8.5 |
| Sunflower oil | 1.5 | 0.5 |
| Linseed oil | 0.2 | 0.2 |
| VCO | / | 1.0 * |
| Minerals and vitamins supplement | 0.6 | 0.6 |
| Chemical composition (%) | ||
| Moisture | 80.47 | 80.42 |
| Dry matter (DM) | 19.53 | 19.58 |
| % on DM basis | ||
| Crude protein | 39.2 | 39.0 |
| Crude fat | 7.9 | 8.1 |
| Starch | 17.97 | 18.12 |
| Crude fiber | 1.30 | 1.47 |
| Ash | 6.29 | 6.27 |
| Fatty Acid, % | HCD | HCD + VCO | VCO |
|---|---|---|---|
| C6:0 | 1.30 | 0.42 | 0.67 |
| C8:0 | 0.43 | 6.21 | 10.37 |
| C10:0 | 0.14 | 3.72 | 6.11 |
| C12:0 | 0.16 | 26.42 | 44.36 |
| C14:0 | 2.87 | 14.16 | 21.24 |
| C16:0 | 31.91 | 17.06 | 7.86 |
| C18:0 | 8.16 | 4.65 | 3.03 |
| C20:0 | 0.42 | 0.16 | 0.08 |
| C22:0 | 1.19 | 0.24 | n.d. |
| C24:0 | 0.42 | 0.12 | n.d. |
| Total MCFAs | 2.03 | 36.77 | 61.51 |
| Total SFAs | 47.00 | 73.16 | 93.72 |
| C16:1cis-9 | 4.60 | 2.76 | n.d. |
| C18:1cis-9 | 44.46 | 21.99 | 5.28 |
| Total MUFAs | 49.06 | 24.75 | 5.28 |
| C18:2n−6 | 3.30 | 2.05 | 1.00 |
| C18:3n−3 | 0.50 | n.d. | n.d. |
| C20:4n−6 | 0.14 | 0.04 | n.d. |
| Total PUFAs | 3.94 | 2.09 | 1.00 |
| Baseline | HCD + VCO | p-Value | |
|---|---|---|---|
| Clinical parameters | |||
| BW (kg) | 15.8 | 15.6 | 0.711 |
| 5.3–40.0 | 4.6–39.7 | ||
| BCS (9 points scale) | 4.5 | 4.5 | 0.717 |
| 2–6 | 3–5 | ||
| CCECAI | 3 | 1 | 0.001 |
| 1–10 | 0–6 | ||
| Selected serum parameters | |||
| Cobalamin (ng/L) | 286 | / | |
| RI [250–730] | 150–940 | ||
| Cholesterol (mg/dL) | 199 | 176 | 0.396 |
| RI [123–345] | 120–278 | 119–273 | |
| Triglycerides (mg/dL) | 43 | 38 | 0.807 |
| RI [30–120] | 18–88 | 27–175 | |
| Albumin (g/dL) | 3.14 | 3.10 | 0.659 |
| RI [2.75–3.85] | 1.85–3.53 | 2.1–3.7 | |
| Total protein (g/dL) | 6.42 | 6.53 | 0.632 |
| RI [5.60–7.30] | 4.34–7.1 | 5.52–6.90 |
| Baseline | HCD | HCD + VCO | p-Value | |
|---|---|---|---|---|
| Water content (%) | 71.5 a | 77.5 b | 76.7 b | 0.001 |
| 57.8–80.4 | 73.7–85.4 | 72–81.7 | ||
| Fat content (%, on a DM basis) | 3.50 a | 2.60 b | 2.75 ab | 0.034 |
| 1.3–8.9 | 1.1–6.6 | 1.3–4.8 | ||
| Fatty acids (% of total FA) | ||||
| C6:0 | 0.14 a | 0.25 b | 0.22 ab | 0.006 |
| 0.06–0.84 | 0.11–0.60 | 0.11–1.92 | ||
| C8:0 | 0.13 | 0.33 | 0.21 | 0.065 |
| 0.02–0.52 | 0.04–1.52 | 0.06–2.32 | ||
| C10:0 | 0.06 a | 0.12 ab | 0.17 b | 0.015 |
| 0.009–0.33 | 0.016–1.07 | 0.05–1.93 | ||
| C12:0 | 0.09 a | 0.23 a | 0.63 b | 0.001 |
| 0.016–2.4 | 0.031–3.97 | 0.081–8.21 | ||
| C14:0 | 1.35 a | 2.36 b | 3.27 b | <0.001 |
| 0.64–5.1 | 0.39–5.8 | 1.27–15.3 | ||
| C16:0 | 28 a | 31 a | 34 b | <0.001 |
| 14–35.2 | 16.3–37.9 | 23.9–47.9 | ||
| C18:0 | 22.0 | 23.0 | 23.8 | 0.513 |
| 4.64–41.8 | 14.3–33.1 | 13.5–43.9 | ||
| C20:0 | 0.94 a | 1.34 b | 1.23 ab | 0.012 |
| 0.43–2.41 | 0.71–2.22 | 0.66–1.90 | ||
| C22:0 | 1.01 a | 1.60 b | 1.34 b | 0.006 |
| 0.4–21.9 | 0.54–7.30 | 0.62–4.95 | ||
| C24:0 | 1.30 a | 1.91 b | 1.65 ab | 0.005 |
| 0.64–7.04 | 0.75–3.08 | 0.69–3.06 | ||
| Total MCFAs | 0.50 a | 1.14 ab | 1.31 b | 0.012 |
| 0.11–3.14 | 0.30–5.44 | 0.80–9.17 | ||
| Total SFAs | 55.2 a | 65.5 ab | 70.8 b | 0.014 |
| 24–79 | 39–75 | 49–80 | ||
| C16:1cis-9 | 1.51 | 2.33 | 2.37 | 0.060 |
| 0.21–4.52 | 0.23–3.76 | 0.66–3.33 | ||
| C18:1cis-9 | 23.3 a | 16.6 b | 14.5 b | 0.002 |
| 8.35–33.4 | 3.61–46.5 | 8.29–27.5 | ||
| Total MUFAs | 24.6 a | 19.0 b | 17.5 b | 0.002 |
| 9.87–35.3 | 7.37–48.7 | 9.39–29.7 | ||
| C18:2n−6 | 17.0 a | 15.5 ab | 12.9 b | 0.030 |
| 5.92–52.1 | 6.05–31.0 | 8.42–23.3 | ||
| C18:3n−3 | 0.36 a | 0.74 b | 0.51 ab | 0.009 |
| 0.049–4.43 | 0.042–6.30 | 0.045–3.82 | ||
| C20:4n−6 | 0.41 a | 0.64 b | 0.60 ab | 0.018 |
| 0.075–1.19 | 0.30–1.56 | 0.28–1.43 | ||
| Total PUFAs | 19.8 a | 17.1 ab | 14.6 b | 0.024 |
| 8.62–52.3 | 6.91–32.0 | 9.09–24.3 |
| Sterols | Baseline | HCD | HCD + VCO | p-Value |
|---|---|---|---|---|
| Coprostanol | 0.056 a | 0.041 b | 0.041 b | 0.007 |
| RI [0.04–0.19] | 0.031–0.12 | 0.031–0.058 | 0.035–0.055 | |
| Cholesterol | 5.79 | 6.26 | 6.48 | 0.837 |
| RI [0.81–8.83] | 1.83–14.6 | 2.57–14.4 | 3.05–14.3 | |
| Cholestanol | 0.19 | 0.21 | 0.24 | 0.137 |
| RI [0.05–0.42] | 0.048–0.32 | 0.14–0.43 | 0.12–0.58 | |
| Lathosterol | 0.034 | 0.039 | 0.045 | 0.211 |
| RI [0.02–0.06] | 0.024–0.07 | 0.024–0.22 | 0.025–0.11 | |
| Total Zoosterols | 3.77 | 5.54 | 5.06 | 0.179 |
| RI [0.93–9.24] | 1.94–14.9 | 2.79–14.8 | 3.28–14.6 | |
| Brassicasterol | 0.055 | 0.047 | 0.046 | 0.056 |
| RI [0.04–0.23] | 0.04–0.16 | 0.04–0.06 | 0.038–0.16 | |
| Campesterol | 0.95 a | 0.58 ab | 0.42 b | 0.011 |
| RI [0.27–2.27] | 0.32–2.95 | 0.15–1.41 | 0.13–1.24 | |
| Stigmasterol | 0.52 | 0.39 | 0.39 | 0.329 |
| RI [0.08–1.06] | 0.15–1.28 | 0.14–1.01 | 0.12–0.65 | |
| Fusosterol | 0.08 | 0.11 | 0.10 | 0.301 |
| RI [0.03–0.22] | 0.03–0.32 | 0.06–0.16 | 0.06–0.26 | |
| β-sitosterol | 1.84 | 1.75 | 1.28 | 0.411 |
| RI [0.37–4.28] | 0.91–7.29 | 0.40–6.64 | 0.41–3.81 | |
| Sitostanol | 0.072 a | 0.01 b | 0.01 b | <0.001 |
| RI [0.01–0.74] | 0.031–0.41 | 0.01–0.12 | 0.10–0.11 | |
| Total Phytosterols | 3.67 | 2.88 | 2.19 | 0.179 |
| RI [0.82–7.71] | 1.67–12.1 | 0.79–9.24 | 0.80–5.99 | |
| Total Phyto- to Zoosterols | 0.93 a | 0.42 ab | 0.36 b | 0.006 |
| RI [0.20–3.54] | 0.15–2.69 | 0.07–1.70 | 0.08–1.13 | |
| Total sterols | 8.11 | 9.15 | 8.51 | 0.946 |
| RI [2.31–20] | 4.18–27 | 3.62–18 | 4.31–16 |
| Bacterial Populations (Log DNA) | Baseline | HCD | HCD + VCO | p-Value |
|---|---|---|---|---|
| Universal bacteria | 11.1 a | 11.2 b | 11.3 b | 0.005 |
| 10.7–11.4 | 10.9–11.6 | 11.0–11.6 | ||
| Faecalibacterium spp. | 5.58 | 4.77 | 4.83 | 0.556 |
| RI [3.4–8.0] | 3.45–6.83 | 2.76–7.26 | 2.82–7.25 | |
| Turicibacter spp. | 7.37 | 7.35 | 7.0 | 0.854 |
| RI [4.6–8.1] | 5.74–8.12 | 6.07–8.24 | 5.79–8.67 | |
| Streptococcus spp. | 5.07 | 5.54 | 4.71 | 0.198 |
| RI [1.9–8.0] | 3.04–8.93 | 3.33–8.87 | 2.75–7.33 | |
| E. coli | 7.26 | 6.91 | 7.24 | 0.945 |
| RI [0.9–8.0] | 4.42–8.84 | 3.60–8.47 | 4.22–7.89 | |
| Blautia spp. | 10.7 | 10.7 | 10.6 | 0.385 |
| RI [9.5–11] | 7.07–11.1 | 9.73–11.1 | 10.0–11.3 | |
| Fusobacterium spp. | 9.27 a | 9.80 b | 9.85 b | 0.028 |
| RI [7.0–10.3] | 7.37–10.0 | 7.80–10.5 | 7.50–10.4 | |
| C. hiranonis | 5.96 | 6.52 | 6.26 | 0.379 |
| RI [5.1–7.1] | 1.27–7.06 | 1.08–7.11 | 1.08–7.02 | |
| Dysbiosis Index | −2.08 | −1.83 | −2.18 | 0.680 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vecchiato, C.G.; Pinna, C.; Sung, C.-H.; Borrelli De Andreis, F.; Suchodolski, J.S.; Pilla, R.; Delsante, C.; Sportelli, F.; Mammi, L.M.E.; Pietra, M.; et al. Fecal Microbiota, Bile Acids, Sterols, and Fatty Acids in Dogs with Chronic Enteropathy Fed a Home-Cooked Diet Supplemented with Coconut Oil. Animals 2023, 13, 502. https://doi.org/10.3390/ani13030502
Vecchiato CG, Pinna C, Sung C-H, Borrelli De Andreis F, Suchodolski JS, Pilla R, Delsante C, Sportelli F, Mammi LME, Pietra M, et al. Fecal Microbiota, Bile Acids, Sterols, and Fatty Acids in Dogs with Chronic Enteropathy Fed a Home-Cooked Diet Supplemented with Coconut Oil. Animals. 2023; 13(3):502. https://doi.org/10.3390/ani13030502
Chicago/Turabian StyleVecchiato, Carla Giuditta, Carlo Pinna, Chi-Hsuan Sung, Francesca Borrelli De Andreis, Jan S. Suchodolski, Rachel Pilla, Costanza Delsante, Federica Sportelli, Ludovica Maria Eugenia Mammi, Marco Pietra, and et al. 2023. "Fecal Microbiota, Bile Acids, Sterols, and Fatty Acids in Dogs with Chronic Enteropathy Fed a Home-Cooked Diet Supplemented with Coconut Oil" Animals 13, no. 3: 502. https://doi.org/10.3390/ani13030502
APA StyleVecchiato, C. G., Pinna, C., Sung, C.-H., Borrelli De Andreis, F., Suchodolski, J. S., Pilla, R., Delsante, C., Sportelli, F., Mammi, L. M. E., Pietra, M., & Biagi, G. (2023). Fecal Microbiota, Bile Acids, Sterols, and Fatty Acids in Dogs with Chronic Enteropathy Fed a Home-Cooked Diet Supplemented with Coconut Oil. Animals, 13(3), 502. https://doi.org/10.3390/ani13030502








