Confirmation of the Prognostic Value of Foxp3+ Cells in Canine Mammary Tumors
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Tissue Samples
2.2. Histopathological Investigation
2.3. Immunohistochemistry
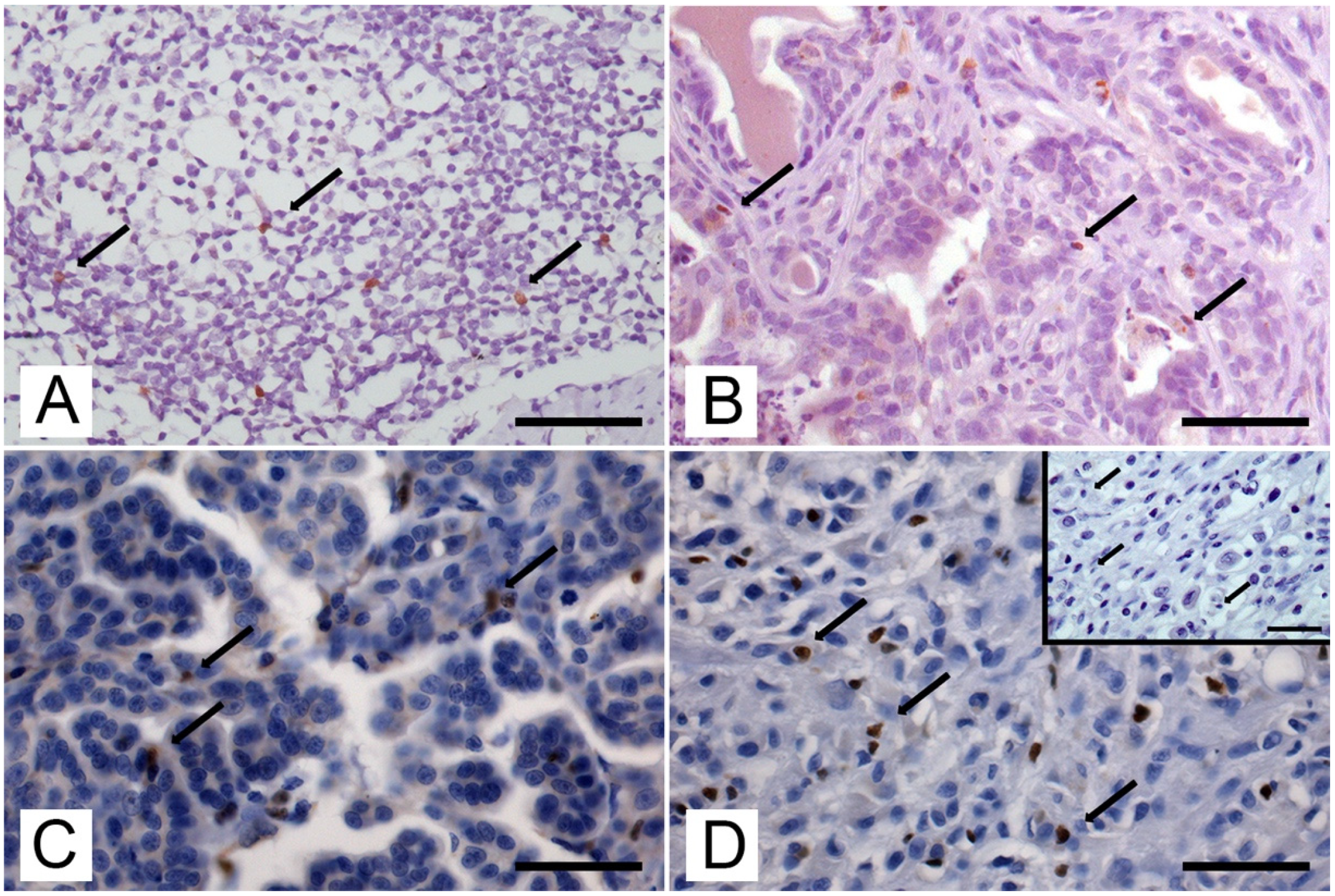
2.4. Quantification of Immunolabelling
2.5. Statistical Analysis
3. Results
3.1. Clinicopathological Data
3.2. Expression and Number of Foxp3
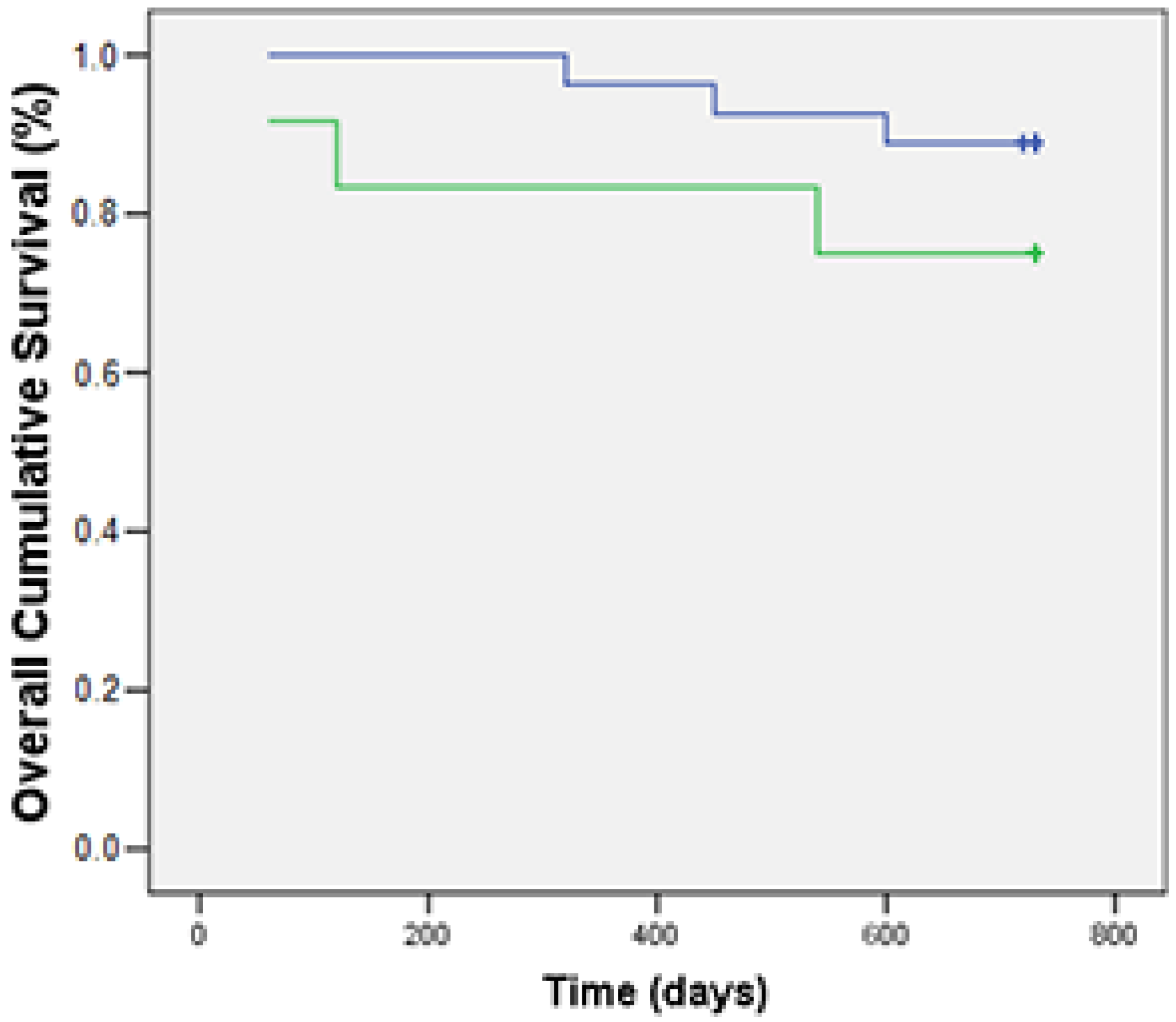
3.3. Associations of Foxp3 with Clinicopathological and Immunohistochemical Features and OS
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- LeBleu, V. Imaging the tumor microenvironment. Cancer J. 2015, 21, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Arneth, B. Tumor Microenvironment. Medicina 2019, 56, 15. [Google Scholar] [CrossRef] [PubMed]
- Korneev, K.V.; Atretkhany, K.N.; Drutskaya, M.S.; Grivennikov, S.I.; Kuprash, D.V.; Nedospasov, S.A. TLR-signaling and proinflammatory cytokines as drivers of tumorigenesis. Cytokine 2017, 89, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.S.; Lee, H.M.; Lio, C.W. Selection of regulatory T cells in the thymus. Nat. Rev. Immunol. 2012, 12, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Whiteside, T.L. FOXP3+ Treg as a therapeutic target for promoting anti-tumor immunity. Expert Opin. Ther. Targets 2018, 22, 353–363. [Google Scholar] [CrossRef]
- Bates, G.J.; Fox, S.B.; Han, C.; Leek, R.D.; Garcia, J.F.; Harris, A.L.; Banham, A.H. Quantification of regulatory T cells enables the identification of high-risk breast cancer patients and those at risk of late relapse. J. Clin. Oncol. 2006, 24, 5373–5380. [Google Scholar] [CrossRef]
- Gao, Y.; Tang, J.; Chen, W.; Li, Q.; Nie, J.; Lin, F.; Wu, Q.; Chen, Z.; Gao, Z.; Fan, H.; et al. Inflammation negatively regulates FOXP3 and regulatory T-cell function via DBC1. Proc. Natl. Acad. Sci. USA 2015, 112, E3246–E3254. [Google Scholar] [CrossRef]
- Zhu, S.; Lin, J.; Qiao, G.; Xu, Y.; Zou, H. Differential regulation and function of tumor-infiltrating T cells in different stages of breast cancer patients. Tumour. Biol. 2015, 36, 7907–7913. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, Y.; Hao, Q.; Wang, S.; Li, H.; Li, J.; Gao, Y.; Li, M.; Li, W.; Xue, X.; et al. FOXP3 suppresses breast cancer metastasis through downregulation of CD44. Int. J. Cancer 2015, 137, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Hur, J.H.; Lee, S.M.; Im, K.S.; Kim, N.H.; Sur, J.H. Correlation of Foxp3 positive regulatory T cells with prognostic factors in canine mammary carcinomas. Vet. J. 2012, 193, 222–227. [Google Scholar] [CrossRef]
- Carvalho, M.I.; Pires, I.; Prada, J.; Gregório, H.; Lobo, L.; Queiroga, F.L. Intratumoral FoxP3 expression is associated with angiogenesis and prognosis in malignant canine mammary tumors. Vet. Immunol. Immunopathol. 2016, 178, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Maeda, S.; Yamada, Y.; Chambers, J.K.; Uchida, K.; Nakayama, H.; Yonezawa, T.; Matsuki, N. Association of tumour-infiltrating regulatory T cells with adverse outcomes in dogs with malignant tumours. Vet. Comp. Oncol. 2018, 16, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Fontenot, J.D.; Rasmussen, J.P.; Williams, L.M.; Dooley, J.L.; Farr, A.G.; Rudensky, A.Y. Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity 2005, 22, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Sorenmo, K.U.; Rasotto, R.; Zappulli, V.; Goldschmidt, M.H. Development, anatomy, histology, lymphatic drainage, clinical features, and cell differentiation markers of canine mammary gland neoplasms. Vet. Pathol. 2011, 48, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Goldschmidt, M.; Penã, L.; Rasotto, R.; Zappulli, V. Classification and grading of canine mammary tumors. Vet. Pathol. 2011, 48, 117–131. [Google Scholar] [CrossRef]
- Peña, L.; De Andres, P.J.; Clemente, M.; Cuesta, P.; Perez-Alenza, M.D. Prognostic value of histological grading in noninflammatory canine mammary carcinomas in a prospective study with two-year follow-up: Relationship with clinical and histological characteristics. Vet. Pathol. 2013, 50, 94–105. [Google Scholar] [CrossRef]
- Mahmoud, S.M.; Paish, E.C.; Powe, D.G.; Macmillan, R.D.; Lee, A.H.; Ellis, I.O.; Green, A.R. An evaluation of the clinical significance of FOXP3(+) infiltrating cells in human breast cancer. Breast Cancer Res. Treat. 2011, 127, 99–108. [Google Scholar] [CrossRef]
- Beha, G.; Brunetti, B.; Asproni, P.; Muscatello, L.V.; Millanta, F.; Poli, A.; Sarli, G.; Benazzi, C. Molecular portrait-based correlation between primary canine mammary tumor and its lymph node metastasis: Possible prognostic-predictive models and/or stronghold for specific treatments? BMC Vet. Res. 2012, 8, 219. [Google Scholar] [CrossRef]
- Millanta, F.; Calandrella, M.; Bari, G.; Niccolini, M.; Vannozzi, I.; Poli, A. Comparison of steroid receptor expression in normal, dysplastic, and neoplastic canine and feline mammary tissues. Res. Vet. Sci. 2005, 79, 225–232. [Google Scholar] [CrossRef]
- Balkwill, F.R.; Capasso, M.; Hagemann, T. The tumor microenvironment at a glance. J. Cell Sci. 2012, 125, 5591–5596. [Google Scholar] [CrossRef]
- Hanahan, D.; Coussens, L. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef]
- Fozza, C.; Longinotti, M. T-cell traffic jam in Hodgkin’s lymphoma: Pathogenetic and therapeutic implications. Adv. Hematol. 2011, 2011, 501659. [Google Scholar] [CrossRef]
- Guillerey, C.; Huntington, N.; Smyth, M. Targeting natural killer cells in cancer immunotherapy. Nat. Immunol. 2016, 17, 1025–1036. [Google Scholar] [CrossRef]
- De Oliveira, L.O.; De Oliveira, R.T.; Loretti, A.P.; Rodrigues, R.; Driemeier, D. Aspectos epidemiológicos da neoplasia mamária canina. Acta. Sci. Vet. 2003, 31, 105–110. [Google Scholar] [CrossRef]
- Sorenmo, K. Canine mammary gland tumors. Vet. Clin. N. Am. Small Anim. Pract. 2003, 33, 573–596. [Google Scholar] [CrossRef]
- Monteiro, L.N.; Dos Reis, D.C.; Salgado, B.S.; Cassali, G.D. Clinical significance and prognostic role of tumor-associated macrophages infiltration according to histologic location in canine mammary carcinomas. Res. Vet. Sci. 2021, 135, 329–334. [Google Scholar] [CrossRef]
- Parisi, F.; Tesi, M.; Millanta, F.; Gnocchi, M.; Poli, A. M1 and M2 tumour-associated macrophages subsets in canine malignant mammary tumours: An immunohistochemical study. Res. Vet. Sci. 2021, 136, 32–38. [Google Scholar] [CrossRef]
- De Souza, T.A.; de Campos, C.B.; De Biasi Bassani Gonçalves, A.; Nunes, F.C.; Monteiro, L.N.; de Oliveira Vasconcelos, R.; Cassali, G.D. Relationship between the inflammatory tumor microenvironment and different histologic types of canine mammary tumors. Res. Vet. Sci. 2018, 119, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Estrela-Lima, A.; Araújo, M.S.; Costa-Neto, J.M.; Teixeira-Carvalho, A.; Barrouin-Melo, S.M.; Cardoso, S.V.; Martins-Filho, O.A.; Serakides, R.; Cassali, G.D. Immunophenotypic features of tumor infiltrating lymphocytes from mammary carcinomas in female dogs associated with prognostic factors and survival rates. BMC Cancer 2010, 10, 256. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Chon, S.K.; Im, K.S.; Kim, N.H.; Sur, J.H. Correlation of tumor-infiltrating lymphocytes to histopathological features and molecular phenotypes in canine mammary carcinoma: A morphologic and immunohistochemical morphometric study. Can. J. Vet. Res. 2013, 77, 142–149. [Google Scholar] [PubMed]
- Serr, I.; Kral, M.; Scherm, M.G.; Daniel, C. Advances in Human Immune System Mouse Models for Personalized Treg-Based Immunotherapies. Front. Immunol. 2021, 12, 643544. [Google Scholar] [CrossRef]
- Franzoni, M.S.; Brandi, A.; de Oliveira Matos Prado, J.K.; Elias, F.; Dalmolin, F.; de Faria Lainetti, P.; Prado, M.C.M.; Leis-Filho, A.F.; Fonseca-Alves, C.E. Tumor-infiltrating CD4+ and CD8+ lymphocytes and macrophages are associated with prognostic factors in triple-negative canine mammary complex type carcinoma. Res. Vet. Sci. 2019, 126, 29–36. [Google Scholar] [CrossRef]
- Pinard, C.J.; International Immuno-Oncology Biomarker Working Group; Lagree, A.; Lu, F.I.; Klein, J.; Oblak, M.L.; Salgado, R.; Cardenas, J.C.P.; Brunetti, B.; Muscatello, L.V.; et al. Comparative Evaluation of Tumor-Infiltrating Lymphocytes in Companion Animals: Immuno-Oncology as a Relevant Translational Model for Cancer Therapy. Cancers 2022, 14, 5008. [Google Scholar] [CrossRef]
- Muscatello, L.V.; Avallone, G.; Brunetti, B.; Bacci, B.; Foschini, M.P.; Sarli, G. Standardized approach for evaluating tumor infiltrating lymphocytes in canine mammary carcinoma: Spatial distribution and score as relevant features of tumor malignancy. Vet. J. 2022, 283–284, 105833. [Google Scholar] [CrossRef]
- Elkord, E.; Alcantar-Orozco, E.M.; Dovedi, S.J.; Tran, D.Q.; Hawkins, R.E.; Gilham, D.E. T regulatory cells in cancer: Recent advances and therapeutic potential. Expert Opin. Biol. Ther. 2010, 10, 1573–1586. [Google Scholar] [CrossRef]
- Nishikawa, H.; Sakaguchi, S. Regulatory T cells in cancer immunotherapy. Curr. Opin. Immunol. 2014, 27, 1–7. [Google Scholar] [CrossRef]
- Whiteside, T.L. Induced regulatory T cells in inhibitory microenvironments created by cancer. Expert Opin. Biol. Ther. 2014, 14, 1411–1425. [Google Scholar] [CrossRef]
- Merlo, A.; Casalini, P.; Carcangiu, M.L.; Malventano, C.; Triulzi, T.; Menard, S.; Tagliabue, E.; Balsari, A. FOXP3 expression and overall survival in breast cancer. J. Clin. Oncol. 2009, 27, 1746–1752. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Sakaguchi, S. Regulatory T cells in immune surveillance and treatment of cancer. Semin Cancer Biol. 2006, 16, 115–123. [Google Scholar] [CrossRef]
- Zou, W. Regulatory T cells, tumour immunity and immunotherapy. Nat. Rev. Immunol. 2006, 6, 4. [Google Scholar]
- Oh, S.Y.; Ryu, H.H.; Yoo, D.Y.; Hwang, I.K.; Kweon, O.K.; Kim, W.H. Evaluation of FOXP3 expression in canine mammary gland tumours. Vet. Comp. Oncol. 2014, 12, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Tarighati, E.; Keivan, H.; Mahani, H. A review of prognostic and predictive biomarkers in breast cancer. Clin. Exp. Med. 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Bertozzi, S.; Londero, A.P.; Seriau, L.; Di Vora, R.; Cedolini, C.; Mariuzzi, L. Biomarkers in Breast Cancer. In Bio-Marker Indicator of Abnormal Physiological Process; Begum, G., Ed.; IntechOpen: London, UK, 2018. [Google Scholar]
- Queiroga, F.L.; Raposo, T.; Carvalho, M.I.; Prada, J.; Pires, I. Canine mammary tumours as a model to study human breast cancer: Most recent findings. In Vivo 2011, 25, 455–465. [Google Scholar] [PubMed]
- Nguyen, F.; Peña, L.; Ibisch, C.; Loussouarn, D.; Gama, A.; Rieder, N.; Belousov, A.; Campone, M.; Abadie, J. Canine invasive mammary carcinomas as models of human breast cancer. Part 1: Natural history and prognostic factors. Breast Cancer Res. Treat. 2018, 167, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Raposo, T.P.; Arias-Pulido, H.; Chaher, N.; Fiering, S.N.; Argyle, D.J.; Prada, J.; Pires, I.; Queiroga, F.L. Comparative aspects of canine and human inflammatory breast cancer. Semin. Oncol. 2017, 44, 288–300. [Google Scholar] [CrossRef]
- Peña, L.; Gama, A.; Goldschmidt, M.H.; Abadie, J.; Benazzi, C.; Castagnaro, M.; Díez, L.; Gärtner, F.; Hellmén, E.; Kiupel, M.; et al. Canine mammary tumors: A review and consensus of standard guidelines on epithelial and myoepithelial phenotype markers, HER2, and hormone receptor assessment using immunohistochemistry. Vet. Path. 2014, 51, 127–145. [Google Scholar] [CrossRef]
- Lamb, C.A.; Vanzulli, S.I.; Lanari, C. Hormone receptors in breast cancer: More than estrogen receptors. Receptores hormonales en cáncer de mama: Receptores de estrógenos y algo más. Medicina 2019, 79, 540–545. [Google Scholar]
- Payne, S.J.; Bowen, R.L.; Jones, J.L.; Wells, C.A. Predictive markers in breast cancer--the present. Histopathology 2008, 52, 82–90. [Google Scholar] [CrossRef]
- Cao, W.; Zhang, B.; Liu, Y.; Li, H.; Zhang, S.; Fu, L.; Niu, Y.; Ning, L.; Cao, X.; Liu, Z.; et al. High-level SLP-2 expression and HER-2/neu protein expression are associated with decreased breast cancer patient survival. Am. J. Clin. Pathol. 2007, 128, 430–436. [Google Scholar] [CrossRef]
- Reix, N.; Malina, C.; Chenard, M.P.; Bellocq, J.P.; Delpous, S.; Molière, S.; Sevrin, A.; Neuberger, K.; Tomasetto, C.; Mathelin, C. A prospective study to assess the clinical utility of serum HER2 extracellular domain in breast cancer with HER2 overexpression. Breast Cancer Res. Treat. 2016, 160, 249–259. [Google Scholar] [CrossRef]
| Foxp3 intra | p | Foxp3 adj | p | Foxp3 dist | p | |
|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | ||||
| Diagnosis | p < 0.05 | NS | ||||
| Adenoma (n = 7) | 4.8 ± 7.3 | p < 0.0005 | 3.1 ± 5.1 | 6.6 ± 0.97 | ||
| Carcinoma (n = 39) | 67.2 ± 27.8 | 12.0 ± 15.9 | 10.98 ± 1.23 | |||
| Histotype CMCs | ||||||
| CC (n = 14) | 50.6 ± 17.3 a | (a) p < 0.0005 (b) p > 0.05 | 5.0 ± 17.3 c | (c) p < 0.0005 | 1.3 ± 2.4 d | (d),(e) p < 0.0005 |
| STC (n = 5) | 46.2 ± 29.5 b | 0.0 | 0.0 e | |||
| STPC (n = 8) | 69.6 ± 6.4 | 7.5 ± 5.6 | 2.7 ± 3.3 | |||
| SSC + AC (n = 12) | 95.0 ± 24.0 | 28.8 ± 20.0 | 12 ± 6.9 | |||
| Grading | ||||||
| Grade I (n = 30) | 58.6 ± 21.5 f | (f) p < 0.0005 (g) p < 0.05 | 7.5 ± 8.5 h | (h),(i) p < 0.0005 | 2.8 ± 5.0 l | (l) p < 0.05 |
| Grade II (n = 6) | 80.5 ± 7.5 g | 20.1 ± 13.1 i | 11.8 ± 6.0 | |||
| Grade III (n = 3) | 131.7 ± 9.0 | 41.5 ± 39.1 | 10.0 ± 10.4 | |||
| Lymphatic invasion | ||||||
| Negative (n = 30) | 59.2 ± 22 | p < 0.0005 | 9.6 ± 10.9 | NS | 4.1 ± 6.2 | NS |
| Positive (n = 9) | 95.7 ± 27.9 | 20.2 ± 26.0 | 6.9 ± 7.6 | |||
| ER | ||||||
| Positive (n = 24) | 54.6 ± 20.9 | p < 0.0005 | 6.7 ± 7.1 | p < 0.01 | 1.5 ± 2.5 | p < 0.0005 |
| Negative (n = 15) | 88.5 ± 25.3 | 20.6 ± 21.9 | 9.7 ± 7.8 | |||
| PR | ||||||
| Positive (n = 12) | 57.7 ± 23.0 | NS | 8.8 ± 8.3 | NS | 1.9 ± 2.8 | NS |
| Negative (n = 27) | 72.0 ± 29.0 | 13.5 ± 18.2 | 5.9 ± 7.4 | |||
| C-erBb-2 | ||||||
| Positive (n = 8) | 67.6 ± 23.2 | NS | 13.9 ± 27.4 | NS | 4.7 ± 7.2 | NS |
| Negative (n = 31) | 67.6 ± 29.5 | 11.5 ± 11.1 | 4.7 ± 6.4 | |||
| Cancer-related death | ||||||
| Alive (n = 30) | 63.1 ± 25.9 | p < 0.05 | 10.1 ± 11 | NS | 4.3 ± 6.3 | NS |
| Death (n = 9) | 92.2 ± 27.7 | 22.5 ± 31.5 | 6.8 ± 7.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parisi, F.; Millanta, F.; Nicastro, M.; Vannozzi, I.; Poli, A. Confirmation of the Prognostic Value of Foxp3+ Cells in Canine Mammary Tumors. Animals 2023, 13, 505. https://doi.org/10.3390/ani13030505
Parisi F, Millanta F, Nicastro M, Vannozzi I, Poli A. Confirmation of the Prognostic Value of Foxp3+ Cells in Canine Mammary Tumors. Animals. 2023; 13(3):505. https://doi.org/10.3390/ani13030505
Chicago/Turabian StyleParisi, Francesca, Francesca Millanta, Marika Nicastro, Iacopo Vannozzi, and Alessandro Poli. 2023. "Confirmation of the Prognostic Value of Foxp3+ Cells in Canine Mammary Tumors" Animals 13, no. 3: 505. https://doi.org/10.3390/ani13030505
APA StyleParisi, F., Millanta, F., Nicastro, M., Vannozzi, I., & Poli, A. (2023). Confirmation of the Prognostic Value of Foxp3+ Cells in Canine Mammary Tumors. Animals, 13(3), 505. https://doi.org/10.3390/ani13030505







