Getting to the Meat of It: The Effects of a Captive Diet upon the Skull Morphology of the Lion and Tiger
Simple Summary
Abstract
1. Introduction
2. Approach and Methods
Morphometric Methods
3. Results
3.1. All Data
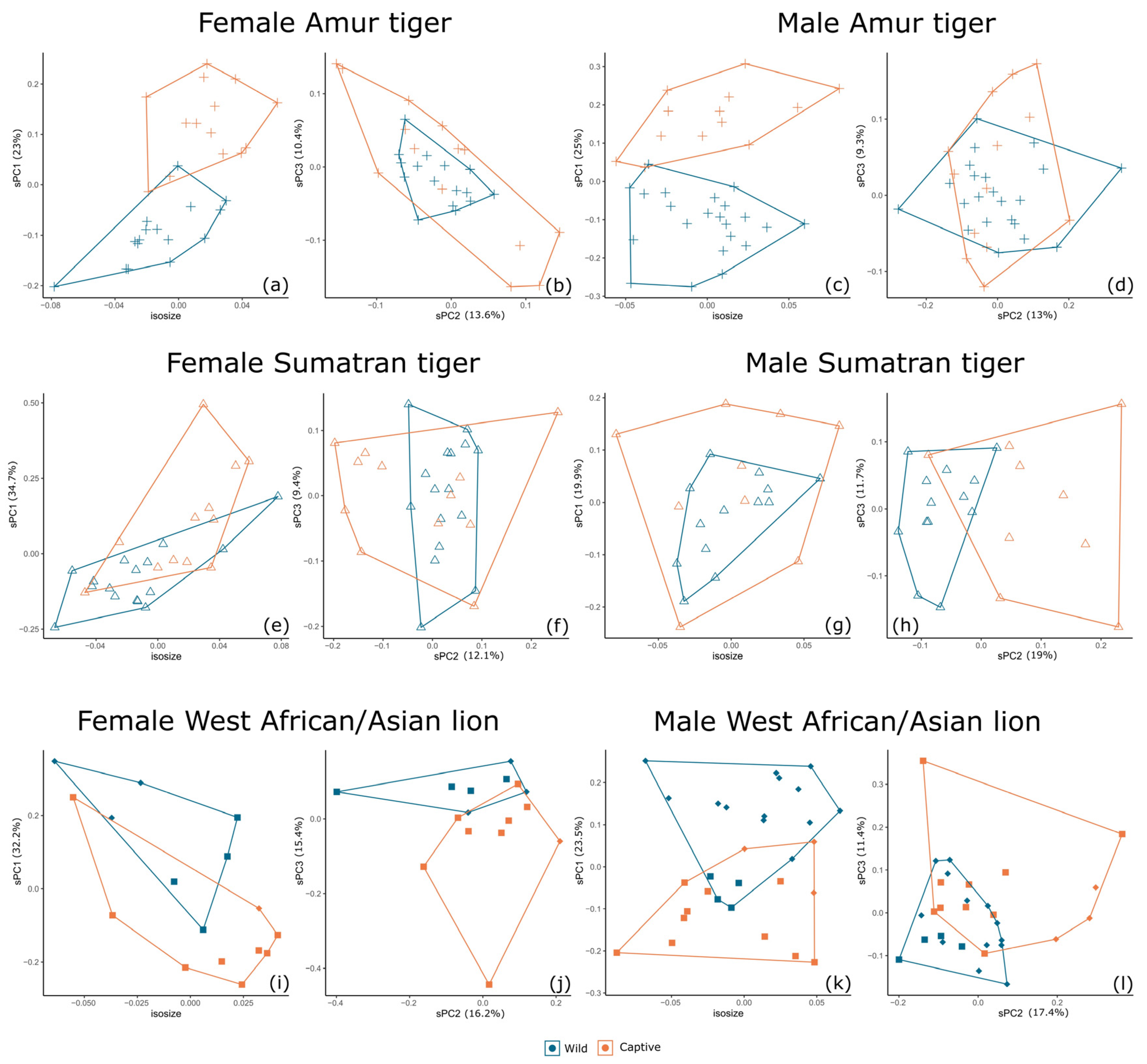
3.2. Separation by Sex and Species
3.3. Separation by Geographical Origin
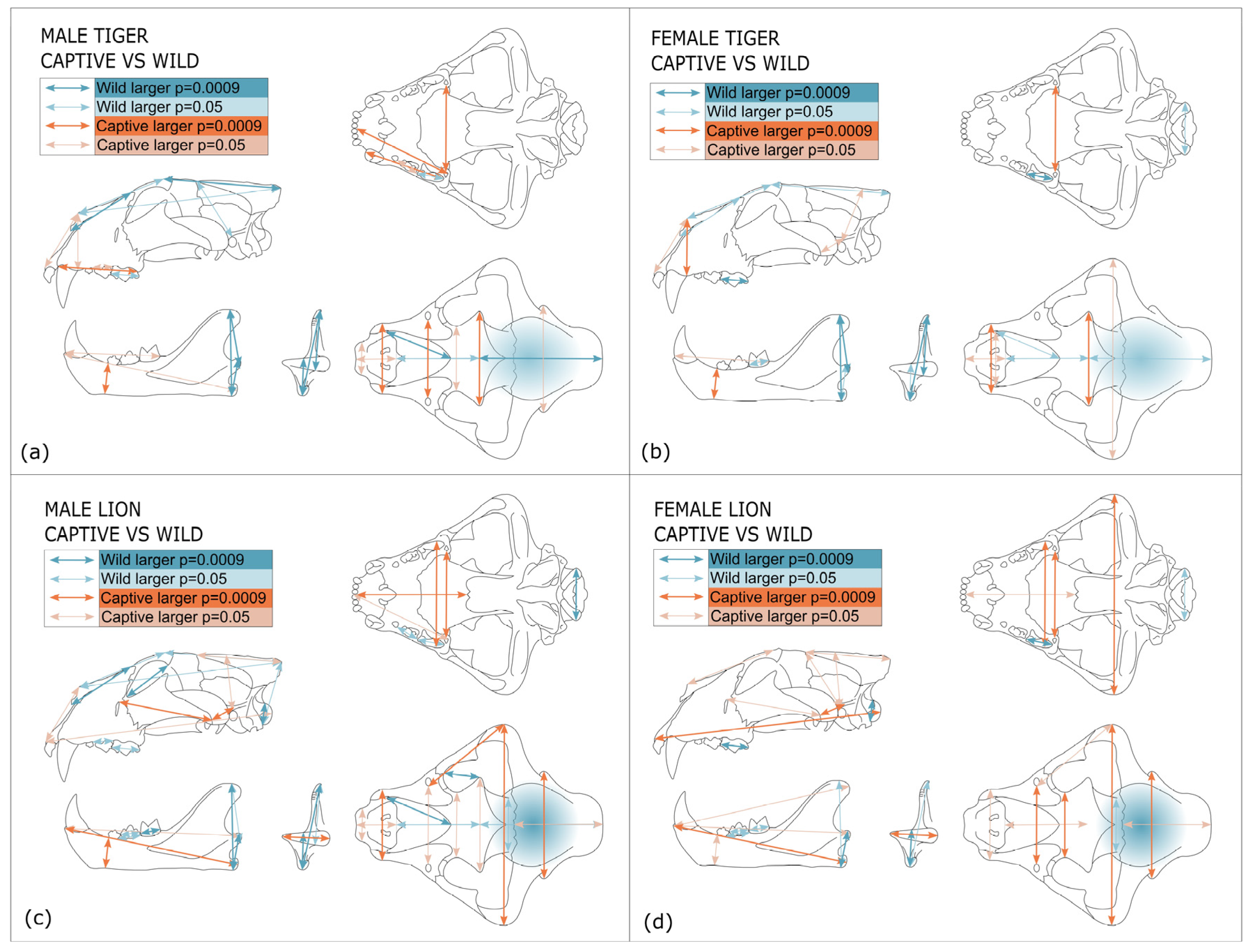
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Smuts, G.L.; Anderson, J.L.; Austin, J.C. Age Determination of the African Lion (Panthera leo). J. Zool. 1978, 185, 115–146. [Google Scholar] [CrossRef]
- O’Regan, H.J.; Kitchener, A.C. The Effects of Captivity on the Morphology of Captive, Domesticated and Feral Mammals. Mammal. Rev. 2005, 35, 215–230. [Google Scholar] [CrossRef]
- Hollister, N. Some Effects of Environment and Habit on Captive Lions. Proc. U. S. Natl. Mus. 1917, 53, 177–193. [Google Scholar] [CrossRef][Green Version]
- Duckler, G.L. An Unusual Osteological Formation in the Posterior Skulls of Captive Tigers (Panthera tigris). Zoo. Biol. 1998, 17, 135–142. [Google Scholar] [CrossRef]
- Hartstone-Rose, A.; Selvey, H.; Villari, J.R.; Atwell, M.; Schmidt, T. The Three-Dimensional Morphological Effects of Captivity. PLoS ONE 2014, 9, e113437. [Google Scholar] [CrossRef] [PubMed]
- Currey, J.D. The Many Adaptations of Bone. J. Biomech. 2003, 36, 1487–1495. [Google Scholar] [CrossRef]
- Ruff, C.; Holt, B.; Trinkaus, E. Who’s Afraid of the Big Bad Wolff?: “Wolff’s Law” and Bone Functional Adaptation. Am. J. Phys. Anthropol. 2006, 129, 484–498. [Google Scholar] [CrossRef]
- IUCN/SSC. Guidelines on the Use of Ex. Situ Management for Species Conservation; IUCN Species Survival Commission: Gland, Switzerland, 2014. [Google Scholar]
- McGowan, P.J.K.; Traylor-Holzer, K.; Leus, K. IUCN Guidelines for Determining When and How Ex Situ Management Should Be Used in Species Conservation: IUCN Ex Situ Management Guidelines. Conserv. Lett. 2017, 10, 361–366. [Google Scholar] [CrossRef]
- Reeves, J.; Smith, C.; Dierenfeld, E.S.; Whitehouse-Tedd, K. Captivity-Induced Metabolic Programming in an Endangered Felid: Implications for Species Conservation. Sci. Rep. 2020, 10, 3630. [Google Scholar] [CrossRef]
- Wilting, A.; Courtiol, A.; Christiansen, P.; Niedballa, J.; Scharf, A.K.; Orlando, L.; Balkenhol, N.; Hofer, H.; Kramer-Schadt, S.; Fickel, J.; et al. Planning Tiger Recovery: Understanding Intraspecific Variation for Effective Conservation. Sci. Adv. 2015, 1, 1–13. [Google Scholar] [CrossRef]
- Sun, X.; Liu, Y.-C.; Tiunov, M.P.; Gimranov, D.O.; Zhuang, Y.; Han, Y.; Driscoll, C.A.; Pang, Y.; Li, C.; Pan, Y.; et al. Ancient DNA Reveals Genetic Admixture in China during Tiger Evolution. Nat. Ecol. Evol. 2023, 7, 1914–1929. [Google Scholar] [CrossRef] [PubMed]
- Frankham, R.; Bradshaw, C.J.A.; Brook, B.W. Genetics in Conservation Management: Revised Recommendations for the 50/500 Rules, Red List Criteria and Population Viability Analyses. Biol. Conserv. 2014, 170, 56–63. [Google Scholar] [CrossRef]
- Kitchener, A.C.; Breitenmoser-Wuersten, C.; Eizirik, E.; Gentry, A.; Werdelin, L.; Wilting, L.; Wilting, A.; Yamaguchi, N.; Abramov, A.V.; Christiansen, P.; et al. A Revised Taxonomy of the Felidae. The Final Report of the Cat Classification Task Force of the IUCN/SSC Cat Specialist Group; Species Survival Commission: Gland, Switzerland, 2017. [Google Scholar]
- Arenson, J.L.; Simons, E.A.; Anderson, M.; Eller, A.R.; White, F.J.; Frost, S.R. Comparison of Captive and Wild Fascicularis-group Macaques (Primates, Cercopithecidae) Provides Insight into Cranial Form Changes in Response to Rapid Environmental Changes. Am. J. Biol. Anthropol. 2022, 178, 417–436. [Google Scholar] [CrossRef]
- Cooper, D.M.; Yamaguchi, N.; Macdonald, D.W.; Nanova, O.G.; Yudin, V.G.; Dugmore, A.J.; Kitchener, A.C. Phenotypic Plasticity Determines Differences between the Skulls of Tigers from Mainland Asia. R. Soc. Open. Sci. 2022, 9, 220697. [Google Scholar] [CrossRef]
- Zuccarelli, M.D. Comparative Morphometric Analysis of Captive vs. Wild African Lion (Panthera leo) Skulls. Bios 2004, 75, 131–138. [Google Scholar] [CrossRef]
- Siciliano-Martina, L.; Light, J.E.; Lawing, A.M. Cranial Morphology of Captive Mammals: A Meta-Analysis. Front. Zool. 2021, 18, 1–13. [Google Scholar] [CrossRef]
- Curry, B.A.; Drane, A.L.; Atencia, R.; Feltrer, Y.; Howatson, G.; Calvi, T.; Palmer, C.; Moittie, S.; Unwin, S.; Tremblay, J.C.; et al. Body Mass and Growth Rates in Captive Chimpanzees (Pan Troglodytes ) Cared for in African Wildlife Sanctuaries, Zoological Institutions, and Research Facilities. Zoo. Biol. 2023, 42, 98–106. [Google Scholar] [CrossRef]
- Howell, A.B. Pathologic Skulls of Captive Lions. J. Mammal. 1917, 6, 163–168. [Google Scholar] [CrossRef]
- Moore, W.J. Masticatory Function and Skull Growth. Proc. Zool. Soc. Lond. 1965, 146, 123–131. [Google Scholar] [CrossRef]
- Yamamoto, S. The Effects of Food Consistency on Maxillary Growth in Rats. Eur. J. Orthod. 1996, 18, 601–615. [Google Scholar] [CrossRef]
- Katsaros, C.; Berg, R.; Kiliaridis, S. Influence of Masticatory Muscle Function on Transverse Skull Dimensions in the Growing Rat. J. Orofac. Orthop./Fortschr. Kieferorthopädie 2002, 63, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.S.; Renaud, S.; Rayfield, E.J. Adaptive Plasticity in the Mouse Mandible. BMC Evol. Biol. 2014, 14, 85. [Google Scholar] [CrossRef] [PubMed]
- Fujita, Y.; Goto, S.; Ichikawa, M.; Hamaguchi, A.; Maki, K. Effect of Dietary Calcium Deficiency and Altered Diet Hardness on the Jawbone Growth: A Micro-CT and Bone Histomorphometric Study in Rats. Arch. Oral. Biol. 2016, 72, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Hampson, M.C.; Schwitzer, C. Effects of Hand-Rearing on Reproductive Success in Captive Large Cats Panthera tigris altaica, Uncia uncia, Acinonyx jubatus and Neofelis nebulosa. PLoS ONE 2016, 11, e0155992. [Google Scholar] [CrossRef] [PubMed]
- Segura, V.; Cassini, G.H.; Prevosti, F.J. Three-Dimensional Cranial Ontogeny in Pantherines ( Panthera leo, P. onca, P. pardus, P. tigris; Carnivora: Felidae). Biol. J. Linn. Soc. 2017, 120, 210–227. [Google Scholar] [CrossRef]
- Dierenfield, E.S.; Bush, M.; Phillips, L.; Montali, R. Nutrition, Food Preparation and Feeding. In Management and Conservation of Captive Tigers, Panthera tigris; Tilson, R., Brady, G., Traylor-Holzer, K., Armstrong, D., Eds.; Minnesota Zoo: Apple Valley, MN, USA, 1994. [Google Scholar]
- Ashton, D.G.; Jones, D.M. Veterinary Aspects of the Management of Non-Domestic Cats. In Proceedings of the Symposium of the Association of British Wild Animal Keepers, London, UK, 31 March 1979; pp. 12–22. [Google Scholar]
- Bond, J.C.; Lindburg, D.G. Carcass Feeding of Captive Cheetahs (Acinonyx jubatus): The Effects of a Naturalistic Feeding Program on Oral Health and Psychological Well-Being. Appl. Anim. Behav. Sci. 1990, 26, 373–382. [Google Scholar] [CrossRef]
- Kleinlugtenbelt, C.L.M.; Burkevica, A.; Clauss, M. Large Carnivore Feeding in European Zoos. Zool. Gart. 2023, 91, 9–39. [Google Scholar]
- DeSantis, L.R.G.; Patterson, B.D. Dietary Behaviour of Man-Eating Lions as Revealed by Dental Microwear Textures. Sci. Rep. 2017, 7, 904. [Google Scholar] [CrossRef]
- Gidna, A.; Yravedra, J.; Domínguez-Rodrigo, M. A Cautionary Note on the Use of Captive Carnivores to Model Wild Predator Behavior: A Comparison of Bone Modification Patterns on Long Bones by Captive and Wild Lions. J. Archaeol. Sci. 2013, 40, 1903–1910. [Google Scholar] [CrossRef]
- Mora, R.; Aramendi, J.; Courtenay, L.A.; González-Aguilera, D.; Yravedra, J.; Maté-González, M.Á.; Prieto-Herráez, D.; Vázquez-Rodríguez, J.M.; Barja, I. Ikhnos: A Novel Software to Register and Analyze Bone Surface Modifications Based on Three-Dimensional Documentation. Animals 2022, 12, 2861. [Google Scholar] [CrossRef]
- Kapoor, V.; Antonelli, T.; Parkinson, J.A.; Hartstone-Rose, A. Oral Health Correlates of Captivity. Res. Vet. Sci. 2016, 107, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Fitch, H.M.; Fagan, D.A. Focal Palatine Erosion Associated with Dental Malocclusion in Captive Cheetahs. Zoo. Biol. 1982, 1, 295–310. [Google Scholar] [CrossRef]
- Zordan, M.; Deem, S.L.; Sanchez, C.R. Focal Palatine Erosion in Captive and Free-Living Cheetahs (Acinonyx jubatus) and Other Felid Species. Zoo. Biol. 2012, 31, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Steenkamp, G.; Boy, S.C.; van Staden, P.J.; Bester, M.N. Focal Palatitis (Previously Focal Palatine Erosions) in Captive Cheetahs (Acinonyx jubatus). Front. Vet. Sci. 2021, 8, 682150. [Google Scholar] [CrossRef]
- Doige, C.E.; Owen, B.D.; Mills, J.H.L. Influence of Calcium and Phosphorus on Growth and Skeletal Development of Growing Swine. Can. J. Anim. Sci. 1975, 55, 147–164. [Google Scholar] [CrossRef]
- Luke, D.A.; Tonge, C.H.; Reid, D.J. Metrical Analysis of Growth Changes in the Jaws and Teeth of Normal, Protein Deficient and Calorie Deficient Pigs. J. Anat. 1979, 129, 449–457. [Google Scholar]
- Miller, S.C.; Halloran, B.P.; DeLuca, H.F.; Jee, W.S.S. Studies on the Role of Vitamin D in Early Skeletal Development, Mineralization, and Growth in Rats. Calcif. Tissue Int. 1983, 35, 455–460. [Google Scholar] [CrossRef]
- Mehta, G.; Roach, H.I.; Langley-Evans, S.; Taylor, P.; Reading, I.; Oreffo, R.O.C.; Aihie-Sayer, A.; Clarke, N.M.P.; Cooper, C. Intrauterine Exposure to a Maternal Low Protein Diet Reduces Adult Bone Mass and Alters Growth Plate Morphology in Rats. Calcif. Tissue Int. 2002, 71, 493–498. [Google Scholar] [CrossRef]
- Silva, M.J.; Eekhoff, J.D.; Patel, T.; Kenney-Hunt, J.P.; Brodt, M.D.; Steger-May, K.; Scheller, E.L.; Cheverud, J.M. Effects of High-Fat Diet and Body Mass on Bone Morphology and Mechanical Properties in 1100 Advanced Intercross Mice. J. Bone Min. Res. 2019, 34, 711–725. [Google Scholar] [CrossRef]
- Kerr, K.R.; Morris, C.L.; Burke, S.L.; Swanson, K.S. Apparent Total Tract Macronutrient and Energy Digestibility of 1- to- 3-Day-Old Whole Chicks, Adult Ground Chicken, and Extruded and Canned Chicken-Based Diets in African Wildcats (Felis silvestris Lybica). Zoo. Biol. 2013, 32, 510–517. [Google Scholar] [CrossRef]
- Bennett, C.L.; Booth-Binczik, S.D.; Steele, S.R.E. Nutritional Composition and Digestibility by Ocelots (Leopardus Pardalis) of Whole Animals and a Commercial Diet. Zoo. Biol. 2010, 29, 753–759. [Google Scholar] [CrossRef] [PubMed]
- Barnett, R.; Yamaguchi, N.; Shapiro, B.; Sabin, R. Ancient DNA Analysis Indicates the First English Lions Originated from North Africa. Contrib. Zool. 2008, 77, 7–16. [Google Scholar] [CrossRef]
- van Buuren, S. Flexible Imputation of Missing Data; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]
- Baur, H.; Leuenberger, C. Analysis of Ratios in Multivariate Morphometry. Syst. Biol. 2011, 60, 813–825. [Google Scholar] [CrossRef] [PubMed]
- Baur, H.; Kranz-Baltensperger, Y.; Cruaud, A.; Rasplus, J.Y.; Timokhov, A.V.; Gokhman, V.E. Morphometric Analysis and Taxonomic Revision of Anisopteromalus Ruschka (Hymenoptera: Chalcidoidea: Pteromalidae)—An Integrative Approach. Syst. Entomol. 2014, 39, 691–709. [Google Scholar] [CrossRef] [PubMed]
- Cardini, A.; Jansson, A.-U.; Elton, S. A Geometric Morphometric Approach to the Study of Ecogeographical and Clinal Variation in Vervet Monkeys. J. Biogeogr. 2007, 34, 1663–1678. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Driscoll, C.A.; Werdelin, L.; Abramov, A.V.; Cuisin, J.; Fernholm, B.; Hiermeier, M.; Hills, D.; Hunter, L.; Johansson, U.S.; et al. Locating Specimens of Extinct Tiger (Panthera tigris) Subspecies: Javan Tiger (P.T. Sondaica), Balinese Tiger (P.T. Balica), and Caspian Tiger (P.T. Virgata), Including Previously Unpublished Specimens. Mammal. Soc. Jpn. 2013, 38, 187–198. [Google Scholar] [CrossRef]
- Kitchener, A.C. Tiger Distribution, Phenotypic Variation and Conservation Issues. In Riding the Tiger. Tiger Conservation in Human-Dominated Landscapes; Seidensticker, J., Christie, J., Jackson, P., Eds.; Cambridge University Press: Cambridge, UK, 1999; pp. 19–39. [Google Scholar]
- Mazák, J.H.; Groves, C.P. A Taxonomic Revision of the Tigers (Panthera tigris) of Southeast Asia. Mamm. Biol. 2006, 71, 268–287. [Google Scholar] [CrossRef]
- Mazák, J.H. Geographical Variation and Phylogenetics of Modern Lions Based on Craniometric Data. J. Zool. 2010, 281, 194–209. [Google Scholar] [CrossRef]
- Mazák, J.H. Craniometric Variation in the Tiger (Panthera tigris): Implications for Patterns of Diversity, Taxonomy and Conservation. Mamm. Biol.-Z. Für Säugetierkunde 2010, 75, 45–68. [Google Scholar] [CrossRef]
- Saragusty, J.; Shavit-Meyrav, A.; Yamaguchi, N.; Nadler, R.; Bdolah-Abram, T.; Gibeon, L.; Hildebrandt, T.B.; Shamir, M.H. Comparative Skull Analysis Suggests Species-Specific Captivity-Related Malformation in Lions (Panthera leo). PLoS ONE 2014, 9, e94527. [Google Scholar] [CrossRef]
- Tidière, M.; Gaillard, J.-M.; Berger, V.; Müller, D.W.H.; Bingaman Lackey, L.; Gimenez, O.; Clauss, M.; Lemaître, J.-F. Comparative Analyses of Longevity and Senescence Reveal Variable Survival Benefits of Living in Zoos across Mammals. Sci. Rep. 2016, 6, 36361. [Google Scholar] [CrossRef] [PubMed]
- Kitchener, A.C. The Longevity Legacy: The Challenges of Old Animals in Zoos. In Optimal Wellbeing of Ageing Wild Animals in Human Care; Brando, S., Chapman, S., Eds.; Springer Nature: Cham, Switzerland, 2023. [Google Scholar]
- McPhee, M.E. Intact Carcasses as Enrichment for Large Felids: Effects on on- and off-Exhibit Behaviors. Zoo. Biol. 2002, 21, 37–47. [Google Scholar] [CrossRef]
- Cracraft, J.; Feinstein, J.; Vaughn, J.; Helm-Bychowski, K. Sorting out Tigers (Panthera tigris): Mitochondrial Sequences, Nuclear Inserts, Systematics, and Conservation Genetics. Anim. Conserv. 1998, 1, 139–150. [Google Scholar] [CrossRef]
- Henry, P.; Miquelle, D.; Sugimoto, T.; McCullough, D.R.; Caccone, A.; Russello, M.A. In Situ Population Structure and Ex Situ Representation of the Endangered Amur Tiger. Mol. Ecol. 2009, 18, 3173–3184. [Google Scholar] [CrossRef]
- Skibiel, A.L.; Trevino, H.S.; Naugher, K. Comparison of Several Types of Enrichment for Captive Felids. Zoo. Biol. 2007, 26, 371–381. [Google Scholar] [CrossRef]
- Berthaume, M.A. Food Mechanical Properties and Dietary Ecology. Am. J. Phys. Anthropol. 2016, 159, 79–104. [Google Scholar] [CrossRef] [PubMed]
- Salkina, G.P. The Tiger and It’s Relations with Other Species in South. Sikhote-Alin: Materials on Snow Tracking and Observations in Snowless Period; Lambert Academic Publishing: Saarbrücken, Germany, 2011; ISBN 978-3-8433-9063-7. [Google Scholar]
- Sicuro, F.L.; Oliveira, L.F.B. Skull Morphology and Functionality of Extant Felidae (Mammalia: Carnivora): A Phylogenetic and Evolutionary Perspective. Zool. J. Linn. Soc. 2011, 161, 414–462. [Google Scholar] [CrossRef]
- Hartstone-Rose, A.; Perry, J.M.G.; Morrow, C.J. Bite Force Estimation and the Fiber Architecture of Felid Masticatory Muscles. Anat. Rec. 2012, 295, 1336–1351. [Google Scholar] [CrossRef]
- Turnbull, W.D. Mammalian Masticatory Apparatus. Fieldiana: Geol. 1970, 18, 149–356. [Google Scholar]
- Washburn, S.L. The Relation of the Temporal Muscle to the Form of the Skull. Anat. Rec. 1947, 99, 239–248. [Google Scholar] [CrossRef]
- He, T.; Kiliaridis, S. Effects of Masticatory Muscle Function on Craniofacial Morphology in Growing Ferrets (Mustela putorius Furo). Eur. J. Oral. Sci. 2003, 111, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Kitchener, A.C.; van Valkenburgh, B.; Yamaguchi, N. Felid Form and Function. In Biology and Conservation of Wild Felids; Macdonald, D.W., Loveridge, A.J., Eds.; Oxford University Press: Oxford, UK, 2010; pp. 83–106. ISBN 978-0-19-923445-5. [Google Scholar]
- Santana, S.E. Quantifying the Effect of Gape and Morphology on Bite Force: Biomechanical Modelling and in Vivo Measurements in Bats. Funct. Ecol. 2016, 30, 557–565. [Google Scholar] [CrossRef]
- Vester, B.M.; Beloshapka, A.N.; Middelbos, I.S.; Burke, S.L.; Dikeman, C.L.; Simmons, L.G.; Swanson, K.S. Evaluation of Nutrient Digestibility and Fecal Characteristics of Exotic Felids Fed Horse- or Beef-Based Diets: Use of the Domestic Cat as a Model for Exotic Felids. Zoo. Biol. 2009, 29, 432–448. [Google Scholar] [CrossRef] [PubMed]
- Wroe, S. Cranial Mechanics Compared in Extinct Marsupial and Extant African Lions Using a Finite-element Approach. J. Zool. 2007, 274, 332–339. [Google Scholar] [CrossRef]
- Bouvier, M.; Hylander, W.L. Effect of Bone Strain on Cortical Bone Structure in Macaques (Macaca mulatta). J. Morphol. 1981, 167, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hart, N.H.; Nimphius, S.; Rantalainen, T.; Ireland, A.; Siafarikas, A.; Newton, R.U. Mechanical Basis of Bone Strength: Influence of Bone Material, Bone Structure and Muscle Action. J. Musculoskelet. Neuronal Interact. 2017, 17, 114–139. [Google Scholar] [PubMed]
- Carpenter, R.D.; Carter, D.R. The Mechanobiological Effects of Periosteal Surface Loads. Biomech. Model. Mechanobiol. 2008, 7, 227–242. [Google Scholar] [CrossRef]
- Van Valkenburgh, B. Feeding Behavior in Free-Ranging, Large African Carnivores. J. Mammal. 1996, 77, 240–254. [Google Scholar] [CrossRef]
- Patterson, B.D.; Neiburger, E.J.; Kasiki, S.M. Tooth Breakage and Dental Disease as Causes of Carnivore-Human Conflicts. J. Mammal. 2003, 84, 190–196. [Google Scholar] [CrossRef]
- Siciliano-Martina, L.; Michaud, M.; Tanis, B.P.; Scicluna, E.L.; Lawing, A.M. Endocranial Volume Increases across Captive Generations in the Endangered Mexican Wolf. Sci. Rep. 2022, 12, 8147. [Google Scholar] [CrossRef]
- Yamaguchi, N.; Kitchener, A.C.; Gilissen, E.; Macdonald, D.W. Brain Size of the Lion (Panthera leo) and the Tiger (P. tigris): Implications for Intrageneric Phylogeny, Intraspecific Differences and the Effects of Captivity. Biol. J. Linn. Soc. 2009, 98, 85–93. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cooper, D.M.; Yamaguchi, N.; Macdonald, D.W.; Patterson, B.D.; Salkina, G.P.; Yudin, V.G.; Dugmore, A.J.; Kitchener, A.C. Getting to the Meat of It: The Effects of a Captive Diet upon the Skull Morphology of the Lion and Tiger. Animals 2023, 13, 3616. https://doi.org/10.3390/ani13233616
Cooper DM, Yamaguchi N, Macdonald DW, Patterson BD, Salkina GP, Yudin VG, Dugmore AJ, Kitchener AC. Getting to the Meat of It: The Effects of a Captive Diet upon the Skull Morphology of the Lion and Tiger. Animals. 2023; 13(23):3616. https://doi.org/10.3390/ani13233616
Chicago/Turabian StyleCooper, David M., Nobuyuki Yamaguchi, David W. Macdonald, Bruce D. Patterson, Galina P. Salkina, Viktor G. Yudin, Andrew J. Dugmore, and Andrew C. Kitchener. 2023. "Getting to the Meat of It: The Effects of a Captive Diet upon the Skull Morphology of the Lion and Tiger" Animals 13, no. 23: 3616. https://doi.org/10.3390/ani13233616
APA StyleCooper, D. M., Yamaguchi, N., Macdonald, D. W., Patterson, B. D., Salkina, G. P., Yudin, V. G., Dugmore, A. J., & Kitchener, A. C. (2023). Getting to the Meat of It: The Effects of a Captive Diet upon the Skull Morphology of the Lion and Tiger. Animals, 13(23), 3616. https://doi.org/10.3390/ani13233616