Butter from Different Species: Composition and Quality Parameters of Products Commercialized in the South of Spain
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Material
2.2. Physicochemical Analysis
2.3. Color Analyses
2.4. Textural Analyses
2.5. Fatty Acid Composition Analysis
2.6. Volatile Compound Composition Analysis
2.7. Statistical Analysis
3. Results
3.1. Physicochemical Analysis
3.2. Color Parameters
3.3. Texture Parameters
3.4. Fatty Acid Composition Analysis
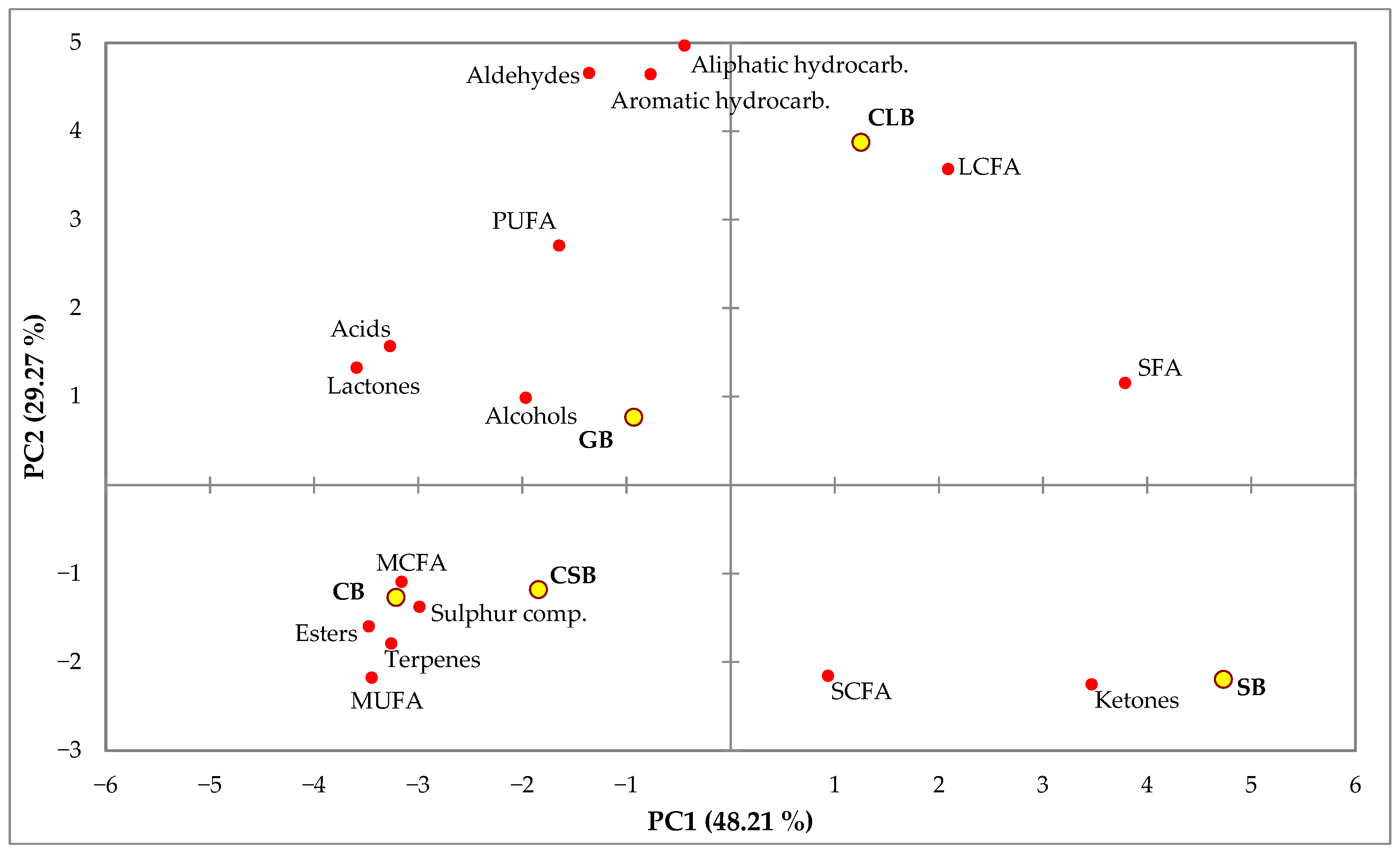
3.5. Volatile Compound Composition Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Parliament and the Council Regulation (EU) No 1308/2013: Establishing a Common Organisation of the Markets in Agricultural Products and Repealing Council Regulations (EEC) No 922/72, (EEC) No 234/79, (EC) No 1037/2001 and (EC) No 1234/2007; European Parliament: Strasbourg, France; Council of the European Union: Brussels, Belgium, 2013.
- Cakmakci, S.; Tahmas Kahyaoğlu, D. A Comparative Study on Some Properties and Oxidation Stability during Storage of Butter Produced from Different Animals’ Milk. GIDA J. Food 2018, 43, 283–293. [Google Scholar] [CrossRef]
- Staniewski, B.; Ogrodowska, D.; Staniewska, K.; Kowalik, J. The Effect of Triacylglycerol and Fatty Acid Composition on the Rheological Properties of Butter. Int. Dairy J. 2021, 114, 104913. [Google Scholar] [CrossRef]
- Adewumi, O.O.; Adegbite, J.A.; Akanmu, O.O. Physicochemical, Microbial and Sensory Properties of Milk, Butter and Garlic Butter. Niger. J. Anim. Sci. 2017, 19, 33–38. [Google Scholar]
- Hayaloglu, A.A.; Yuceer, Y. Utilization and Characterization of Small Ruminants’ Milk and Milk Products in Turkey: Current Status and New Perspectives. Small Rumin. Res. 2011, 101, 73–83. [Google Scholar] [CrossRef]
- Marangoni, A.; Duynhoven, J.; Acevedo, N.; Nicholson, R.; Patel, A. Advances in Our Understanding of the Structure and Functionality of Edible Fats and Fat Mimetics. Soft Matter 2020, 16, 289–306. [Google Scholar] [CrossRef]
- Chamberlain, M.B.; Veltri, B.C.; Taylor, S.J.; Pareas, J.W.; Jimenez-Flores, R.; Juchem, S.O.; Getachew, G.; DePeters, E.J. Feeding Lactating Holstein Cows a Lipid Source High in Palmitic Acid Changes the Fatty Acid Composition and Thermal Properties of Lipids in Milk and Butter. Prof. Anim. Sci. 2016, 32, 672–680. [Google Scholar] [CrossRef]
- Krause, A.J.; Lopetcharat, K.; Drake, M.A. Identification of the Characteristics That Drive Consumer Liking of Butter. J. Dairy Sci. 2007, 90, 2091–2102. [Google Scholar] [CrossRef]
- Ni, Q.; Khomenko, I.; Gallo, L.; Biasioli, F.; Bittante, G. Rapid Profiling of the Volatilome of Cooked Meat by PTR-ToF-MS: Characterization of Chicken, Turkey, Pork, Veal and Beef Meat. Foods 2020, 9, 1776. [Google Scholar] [CrossRef]
- Lawless, H.T. Sensory Evaluation of Food: Principles and Practices; Chapman & Hall/International Thomson Publishing: New York, NY, USA, 1998. [Google Scholar]
- FAO Faostat. Statistical Databases; Food and Agriculture Organization of the United Nations: Rome, Italy, 2023. [Google Scholar]
- MERCASA Alimentacion en España 2022 Producción, Industria, Distribución, Consumo; Mercasa: Barcelona, Spain, 2023.
- ISO 3727-1:2001; Butter—Determination of Moisture, Non-Fat Solids and Fat Contents—Part 1: Determination of Moisture Content. International Organization for Standardization: Geneva, Switzerland, 2001.
- ISO 3727-3:2003; Butter—Determination of Moisture, Non-Fat Solids and Fat Contents—Part 3: Calculation of Fat Content. International Organization for Standardization: Geneva, Switzerland, 2003.
- ISO 3727-2:2001; Butter—Determination of Moisture, Non-Fat Solids and Fat Contents—Part 2: Determination of Non-Fat Solids Content. International Organization for Standardization: Geneva, Switzerland, 2001.
- ISO 1738:2004; Butter—Determination of Salt Content. International Organization for Standardization: Geneva, Switzerland, 2004.
- ISO 1740:2004; Milkfat Products and Butter—Determination of Fat Acidity. International Organization for Standardization: Geneva, Switzerland, 2004.
- ISO 18787:2017; Foodstuffs—Determination of Water Activity. International Organization for Standardization: Geneva, Switzerland, 2017.
- Sukhija, P.S.; Palmquist, D.L. Rapid Method for Determination of Total Fatty Acid Content and Composition of Feedstuffs and Feces. J. Agric. Food Chem. 1988, 36, 1202–1206. [Google Scholar] [CrossRef]
- Juárez, M.; Polvillo, O.; Contò, M.; Ficco, A.; Ballico, S.; Failla, S. Comparison of Four Extraction/Methylation Analytical Methods to Measure Fatty Acid Composition by Gas Chromatography in Meat. J. Chromatogr. A 2008, 1190, 327–332. [Google Scholar] [CrossRef]
- Povolo, M.; Contarini, G. Comparison of Solid-Phase Microextraction and Purge-and-Trap Methods for the Analysis of the Volatile Fraction of Butter. J. Chromatogr. A 2003, 985, 117–125. [Google Scholar] [CrossRef]
- Lapčíková, B.; Lapčík, L.; Valenta, T.; Kučerová, T. Functional and Quality Profile Evaluation of Butters, Spreadable Fats, and Shortenings Available from Czech Market. Foods 2022, 11, 3437. [Google Scholar] [CrossRef]
- Rønholt, S.; Kirkensgaard, J.J.K.; Mortensen, K.; Knudsen, J.C. Effect of Cream Cooling Rate and Water Content on Butter Microstructure during Four Weeks of Storage. Food Hydrocoll. 2014, 34, 169–176. [Google Scholar] [CrossRef]
- Vereecken, J.; Foubert, I.; Meeussen, W.; Lesaffer, A.; Dewettinck, K. Fat Structuring with Partial Acylglycerols: Effect on Solid Fat Profiles. Eur. J. Lipid Sci. Technol. 2009, 111, 259–272. [Google Scholar] [CrossRef]
- Rousseau, D.; Ghosh, S.; Park, H. Comparison of the Dispersed Phase Coalescence Mechanisms in Different Tablespreads. J. Food Sci. 2009, 74, E1–E7. [Google Scholar] [CrossRef]
- López, A.L.; Barriga, D. La Leche, Composición y Características; Ganadería, Fomación; Instituto de Investigación y Formación Agraria y Pesquera, Consejería de Agricultura, Pesca y Desarrollo Rural, Junta de Andalucía: Sevilla, Spain, 2016. [Google Scholar]
- Dias, R.S.; Balthazar, C.F.; Cavalcanti, R.N.; Sobral, L.A.; Rodrigues, J.F.; Neto, R.P.C.; Tavares, M.I.B.; Ribeiro, A.P.B.; Grimaldi, R.; Sant’Anna, C.; et al. Nutritional, Rheological and Sensory Properties of Butter Processed with Different Mixtures of Cow and Sheep Milk Cream. Food Biosci. 2022, 46, 101564. [Google Scholar] [CrossRef]
- Silanikove, N.; Leitner, G.; Merin, U.; Prosser, C.G. Recent Advances in Exploiting Goat’s Milk: Quality, Safety and Production Aspects. Small Rumin. Res. 2010, 89, 110–124. [Google Scholar] [CrossRef]
- Sakkas, L.; Spiliopoulos, M.; Moatsou, G. Partial Substitution of Sheep and Goat Milks of Various Fat Contents by the Respective Sweet Buttermilks: Effect of Cream Heat Treatment. LWT 2020, 133, 109926. [Google Scholar] [CrossRef]
- Salvador, A.; Igual, M.; Contreras, C.; Martínez-Navarrete, N.; del Mar Camacho, M. Effect of the Inclusion of Citrus Pulp in the Diet of Goats on Cheeses Characteristics. Small Rumin. Res. 2014, 121, 361–367. [Google Scholar] [CrossRef]
- Queirós, M.S.; Grimaldi, R.; Gigante, M.L. Addition of Olein from Milk Fat Positively Affects the Firmness of Butter. Food Res. Int. 2016, 84, 69–75. [Google Scholar] [CrossRef]
- Méndez-Cid, F.J.; Centeno, J.A.; Martínez, S.; Carballo, J. Changes in the Chemical and Physical Characteristics of Cow’s Milk Butter during Storage: Effects of Temperature and Addition of Salt. J. Food Compos. Anal. 2017, 63, 121–132. [Google Scholar] [CrossRef]
- Kaya, A. Properties and Stability of Butter Oil Obtained from Milk and Yoghurt. Nahrung 2000, 44, 126–129. [Google Scholar] [CrossRef]
- Bodart, M.; de Peñaranda, R.; Deneyer, A.; Flamant, G. Photometry and Colorimetry Characterisation of Materials in Daylighting Evaluation Tools. Build. Environ. 2008, 43, 2046–2058. [Google Scholar] [CrossRef]
- Zimbru, R.-O.; Pădureţ, S.; Amariei, S. Physicochemical and Color Evaluation of Confectionery Mousses. Food Environ. Saf. 2020, 19, 228–236. [Google Scholar]
- Quintanilla, P.; Beltrán, M.C.; Molina, A.; Escriche, I.; Molina, M.P. Characteristics of Ripened Tronchón Cheese from Raw Goat Milk Containing Legally Admissible Amounts of Antibiotics. J. Dairy Sci. 2019, 102, 2941–2953. [Google Scholar] [CrossRef]
- Adekunte, A.O.; Tiwari, B.K.; Cullen, P.J.; Scannell, A.G.M.; O’Donnell, C.P. Effect of Sonication on Colour, Ascorbic Acid and Yeast Inactivation in Tomato Juice. Food Chem. 2010, 122, 500–507. [Google Scholar] [CrossRef]
- Oeffner, S.P.; Qu, Y.; Just, J.; Quezada, N.; Ramsing, E.; Keller, M.; Cherian, G.; Goddick, L.; Bobe, G. Effect of Flaxseed Supplementation Rate and Processing on the Production, Fatty Acid Profile, and Texture of Milk, Butter, and Cheese. J. Dairy Sci. 2013, 96, 1177–1188. [Google Scholar] [CrossRef]
- Pădureţ, S. The Effect of Fat Content and Fatty Acids Composition on Color and Textural Properties of Butter. Molecules 2021, 26, 4565. [Google Scholar] [CrossRef]
- Mills, S.; Ross, R.P.; Hill, C.; Fitzgerald, G.F.; Stanton, C. Milk Intelligence: Mining Milk for Bioactive Substances Associated with Human Health. Int. Dairy J. 2011, 21, 377–401. [Google Scholar] [CrossRef]
- Burns-Whitmore, B.; Froyen, E.; Heskey, C.; Parker; Pablo, S. Alpha-Linolenic and Linoleic Fatty Acids in the Vegan Diet: Do They Require Dietary Reference Intake/Adequate Intake Special Consideration? Nutrients 2019, 11, 2365. [Google Scholar] [CrossRef]
- Jensen, R.G. The Composition of Bovine Milk Lipids: January 1995 to December 2000. J. Dairy Sci. 2002, 85, 295–350. [Google Scholar] [CrossRef]
- Collomb, M.; Bühler, T. Analyse de la Composition en Acides Gras de la Graisse de Lait: I. Optimisation et Validation d’une Méthode Générale à Haute Résolution; Information FAM; Station Fédérale de Recherches Laitières Liebefeld: Bern, Switzerland, 2000. [Google Scholar]
- Teng, F.; Wang, P.; Yang, L.; Ma, Y.; Day, L. Quantification of Fatty Acids in Human, Cow, Buffalo, Goat, Yak, and Camel Milk Using an Improved One-Step GC-FID Method. Food Anal. Methods 2017, 10, 2881–2891. [Google Scholar] [CrossRef]
- Månsson, H. Fatty Acids in Bovine Milk Fat. Food Nutr. Res. 2008, 52. [Google Scholar] [CrossRef]
- Ozcan, T.; Akpınar, A.; Yilmaz-Ersan, L.; Cetin, K.; Delikanli Kiyak, B. Evaluation of Fatty Acid Profile of Trabzon Butter. Int. J. Chem. Eng. Appl. 2016, 7, 190–194. [Google Scholar] [CrossRef]
- Gómez-Cortés, P.; Juárez, M.; de la Fuente, M.A. Milk Fatty Acids and Potential Health Benefits: An Updated Vision. Trends Food Sci. Technol. 2018, 81, 1–9. [Google Scholar] [CrossRef]
- Balthazar, C.; Pimentel, T.C.; Ferrão, L.L.; Almada, C.N.; Santillo, A.; Albenzio, M.; Mollakhalili, N.; Mortazavian, A.M.; Nascimento, J.S.; Silva, M.C.; et al. Sheep Milk: Physicochemical Characteristics and Relevance for Functional Food Development. Compr. Rev. Food Sci. Food Saf. 2017, 16, 247–262. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Krauss, R.M. Public Health Guidelines Should Recommend Reducing Saturated Fat Consumption as Much as Possible: YES. Am. J. Clin. Nutr. 2020, 112, 13–18. [Google Scholar] [CrossRef]
- Cabiddu, A.; Addis, M.; Pinna, G.; Spada, S.; Fiori, M.; Sitzia, M.; Pirisi, A.; Piredda, G.; Molle, G. The Inclusion of a Daisy Plant (Chrysanthemum coronarium) in Dairy Sheep Diet. 1: Effect on Milk and Cheese Fatty Acid Composition with Particular Reference to C18:2 Cis-9, Trans-11. Livest. Sci. 2006, 101, 57–67. [Google Scholar] [CrossRef]
- Dhiman, T.R.; Satter, L.D.; Pariza, M.W.; Galli, M.P.; Albright, K.; Tolosa, M.X. Conjugated Linoleic Acid (CLA) Content of Milk from Cows Offered Diets Rich in Linoleic and Linolenic Acid. J. Dairy Sci. 2000, 83, 1016–1027. [Google Scholar] [CrossRef]
- Ulbricht, T.L.V.; Southgate, D.A.T. Coronary Heart Disease: Seven Dietary Factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef]
- Poppitt, S.; Keogh, G.; Mulvey, T.; McArdle, B.; MacGibbon, A.; Cooper, G. Lipid-Lowering Effects of a Modified Butter-Fat: A Controlled Intervention Trial in Healthy Men. Eur. J. Clin. Nutr. 2002, 56, 64–71. [Google Scholar] [CrossRef]
- Yurchenko, S.; Sats, A.; Tatar, V.; Kaart, T.; Mootse, H.; Jõudu, I. Fatty Acid Profile of Milk from Saanen and Swedish Landrace Goats. Food Chem. 2018, 254, 326–332. [Google Scholar] [CrossRef]
- Aguilar, C.; Toro-Mujica, P.; Vargas-Bello-Pérez, E.; Vera, R.; Ugalde, C.; Rodríguez, S.; Briones, I. A Comparative Study of the Fatty Acid Profiles in Commercial Sheep Cheeses. Grasas Aceites 2014, 65, e048. [Google Scholar] [CrossRef][Green Version]
- Tahmas-Kahyaoğlu, D.; Cakmakci, S.; Hayaloglu, A.A. Changes during Storage in Volatile Compounds of Butter Produced Using Cow, Sheep or Goat’s Milk. Small Rumin. Res. 2022, 211, 106691. [Google Scholar] [CrossRef]
- Contarini, G.; Povolo, M.; Leardi, R.; Toppino, P.M. Influence of Heat Treatment on the Volatile Compounds of Milk. J. Agric. Food Chem. 1997, 45, 3171–3177. [Google Scholar] [CrossRef]
- Condurso, C.; Verzera, A.; Romeo, V.; Ziino, M.; Conte, F. Solid-Phase Microextraction and Gas Chromatography Mass Spectrometry Analysis of Dairy Product Volatiles for the Determination of Shelf-Life. Int. Dairy J. 2008, 18, 819–825. [Google Scholar] [CrossRef]
- Vialloninsta, C.; Martin, B.; Verdier-Metz, I.; Pradel, P.; Garel, J.-P.; Coulon, J.-B.; Berdagué, J.-L. Transfer of Monoterpenes and Sesquiterpenes from Forages into Milk Fat. Lait 2000, 80, 635–641. [Google Scholar] [CrossRef]
- Curioni, P.M.G.; Bosset, J.O. Key Odorants in Various Cheese Types as Determined by Gas Chromatography-Olfactometry. Int. Dairy J. 2002, 12, 959–984. [Google Scholar] [CrossRef]
- Dadalı, C.; Elmacı, Y. Influence of Fat and Emulsifier Content on Volatile Release of Butter Aroma Used in Water Phase and Physical Attributes of Model Margarines. Eur. J. Lipid Sci. Technol. 2020, 122, 2000036. [Google Scholar] [CrossRef]
- Kondyli, E.; Katsiari, M.C.; Masouras, T.; Voutsinas, L.P. Free Fatty Acids and Volatile Compounds of Low-Fat Feta-Type Cheese Made with a Commercial Adjunct Culture. Food Chem. 2002, 79, 199–205. [Google Scholar] [CrossRef]
- Gatzias, I.S.; Karabagias, I.K.; Kontominas, M.G.; Badeka, A.V. Geographical Differentiation of Feta Cheese from Northern Greece Based on Physicochemical Parameters, Volatile Compounds and Fatty Acids. LWT 2020, 131, 109615. [Google Scholar] [CrossRef]
- Kondyli, E.; Pappa, E.C.; Vlachou, A.M. Effect of Package Type on the Composition and Volatile Compounds of Feta Cheese. Small Rumin. Res. 2012, 108, 95–101. [Google Scholar] [CrossRef]
- Cheng, Z.; O’Sullivan, M.G.; Miao, S.; Kerry, J.P.; Kilcawley, K.N. Sensorial, Cultural and Volatile Properties of Milk, Dairy Powders, Yoghurt and Butter: A Review. Int. J. Dairy Technol. 2022, 75, 761–790. [Google Scholar] [CrossRef]
- Clarke, H.J.; Fitzpatrick, E.; Hennessy, D.; O’Sullivan, M.G.; Kerry, J.P.; Kilcawley, K.N. The Influence of Pasture and Non-Pasture-Based Feeding Systems on the Aroma of Raw Bovine Milk. Front. Nutr. 2022, 9, 841454. [Google Scholar] [CrossRef] [PubMed]
- Mallia, S.; Escher, F.; Schlichtherle-Cerny, H. Aroma-Active Compounds of Butter: A Review. Eur. Food Res. Technol. 2008, 226, 315–325. [Google Scholar] [CrossRef]
- Christensen, T.C.; Hølmer, G.K. GC/MS Analysis of Volatile Aroma Components in Butter during Storage in Different Catering Packaging. Milchwissenschaft 1996, 53, 134–139. [Google Scholar]
- McSweeney, P.L.H.; Sousa, M.J. Biochemical Pathways for the Production of Flavour Compounds in Cheeses during Ripening: A Review. Lait 2000, 80, 293–324. [Google Scholar] [CrossRef]
- Guzmán, J.L.; Delgado Pertíñez, M.; Galán Soldevilla, H.; Ruiz Pérez-Cacho, P.; Polvillo Polo, O.; Zarazaga, L.Á.; Avilés Ramírez, C. Effect of Citrus By-Product on Physicochemical Parameters, Sensory Analysis and Volatile Composition of Different Kinds of Cheese from Raw Goat Milk. Foods 2020, 9, 1420. [Google Scholar] [CrossRef]
- Demirkol, A.; Guneser, O.; Yuceer, Y. Volatile Compounds, Chemical and Sensory Properties of Butters Sold in Canakkale. Tarim Bilim. Derg. 2016, 22, 99–108. [Google Scholar]
- Villeneuve, M.-P.; Lebeuf, Y.; Gervais, R.; Tremblay, G.F.; Vuillemard, J.C.; Fortin, J.; Chouinard, P.Y. Milk Volatile Organic Compounds and Fatty Acid Profile in Cows Fed Timothy as Hay, Pasture, or Silage. J. Dairy Sci. 2013, 96, 7181–7194. [Google Scholar] [CrossRef]
- Lozano, P.R.; Miracle, E.R.; Krause, A.J.; Drake, M.; Cadwallader, K.R. Effect of Cold Storage and Packaging Material on the Major Aroma Components of Sweet Cream Butter. J. Agric. Food Chem. 2007, 55, 7840–7846. [Google Scholar] [CrossRef]
- O’Callaghan, T.F.; Faulkner, H.; McAuliffe, S.; O’Sullivan, M.G.; Hennessy, D.; Dillon, P.; Kilcawley, K.N.; Stanton, C.; Ross, R.P. Quality Characteristics, Chemical Composition, and Sensory Properties of Butter from Cows on Pasture versus Indoor Feeding Systems. J. Dairy Sci. 2016, 99, 9441–9460. [Google Scholar] [CrossRef]
- Buchin, S.; Delague, V.; Duboz, G.; Berdague, J.L.; Beuvier, E.; Pochet, S.; Grappin, R. Influence of Pasteurization and Fat Composition of Milk on the Volatile Compounds and Flavor Characteristics of a Semi-Hard Cheese. J. Dairy Sci. 1998, 81, 3097–3108. [Google Scholar] [CrossRef]
| SB | GB | CB | CSB | CLB | p-Value | |
|---|---|---|---|---|---|---|
| Moisture (%) | 15.89 b ± 0.73 | 13.55 c ± 0.10 | 15.63 bc ± 0.25 | 14.89 bc ± 0.60 | 58.89 a ± 0.29 | *** |
| SFC (%) | 81.95 c ± 0.41 | 85.61 a ± 0.12 | 83.73 b ± 0.06 | 84.35 ab ± 0.38 | 37.21 d ± 0.63 | *** |
| Non-fat solids (%) | 2.16 b ± 0.33 | 0.84 c ± 0.02 | 0.88 c ± 0.23 | 0.75 c ± 0.23 | 3.90 a ± 0.34 | *** |
| Chlorides (%) | 1.03 b ± 0.05 | 1.17 ab ± 0.01 | 0.04 c ± 0.00 | 1.29 a ± 0.05 | 1.22 ab ± 0.11 | *** |
| Tritable acidity (%) | 0.241 a ± 0.005 | 0.062 b ± 0.004 | 0.077 b ± 0.003 | 0.052 b ± 0.018 | 0.074 b ± 0.001 | *** |
| aW | 0.915 b ± 0.001 | 0.909 b ± 0.007 | 0.928 b ± 0.010 | 0.902 b ± 0.012 | 0.986 a ± 0.002 | *** |
| SB | GB | CB | CSB | CLB | p-Value | |
|---|---|---|---|---|---|---|
| L* | 89.27 ab ± 0.93 | 82.97 d ± 0.57 | 86.87 bc ± 0.94 | 85.70 cd ± 1.84 | 90.21 a ± 2.16 | *** |
| a* | −3.17 c ± 0.19 | −2.81 c ± 0.12 | 4.25 a ± 0.05 | 3.36 b ± 0.32 | 3.17 b ± 0.47 | *** |
| b* | 11.48 d ± 0.63 | 6.50 d ± 0.14 | 31.10 a ± 0.73 | 27.46 b ± 1.01 | 22.29 c ± 0.96 | *** |
| C* | 11.91 d ± 0.65 | 7.08 e ± 0.11 | 31.39 a ± 0.72 | 27.66 b ± 1.02 | 22.52 c ± 1.01 | *** |
| h0 | 82.22 b ± 0.13 | 83.04 b ± 0.57 | 81.94 b ± 0.94 | 105.45 a ± 0.63 | 113.41 a ± 1.21 | *** |
| WI | 65.96 e ± 0.30 | 68.81 d ± 0.42 | 75.36 c ± 0.39 | 83.94 a ± 0.37 | 81.56 b ± 0.54 | *** |
| YI | 51.14 a ± 0.66 | 45.76 a ± 0.96 | 35.29 b ± 0.86 | 18.36 c ± 0.88 | 11.19 d ± 0.29 | *** |
| ∆E* | 4.53 ± 1.79 | 9.80 ± 0.89 |
| SB | GB | CB | CSB | CLB | Temp | BT | Temp × BT | |
|---|---|---|---|---|---|---|---|---|
| Hardness (cutting force) (g) | ||||||||
| 4 °C | 233.7 a,yz ± 2.0 | 399.2 a,wx ± 29.6 | 335.8 a,xy ± 16.0 | 467.7 a,w ± 90.2 | 124.7 a,z ± 8.1 | *** | *** | *** |
| 10 °C | 102.6 b,y ± 9.9 | 255.6 b,v ± 12.8 | 189.4 b,x ± 6.0 | 231.2 b,w ± 2.1 | 58.2 b,z ± 1.0 | |||
| 20 °C | 9.9 c,z ± 0.2 | 20.7 c,y ± 2.0 | 43.4 c,w ± 4.6 | 36.3 c,x ± 0.6 | 9.9 c,z ± 0.7 | |||
| Hardness (penetration force) (g) | ||||||||
| 4 °C | 300.1 a,y ± 20.9 | 517.1 a,x ± 43.8 | 569.7 a,wx ± 36.3 | 655.7 a,w ± 60.5 | 136.5 a,z ± 29.7 | *** | *** | *** |
| 10 °C | 155.3 b,y ± 8.4 | 295.6 b,x ± 42.2 | 308.8 b,x ± 9.2 | 333.4 b,x ± 6.9 | 80.2 b,z ± 3.9 | |||
| 20 °C | 10.2 c,z ± 0.04 | 38.4 c,y ± 2.7 | 86.1 c,x ± 3.3 | 87.7 c,x ± 5.9 | 18.3 c,z ± 0.6 | |||
| Adhesiveness (g) | ||||||||
| 4 °C | 48.9 ± 23.5 | 14.3 b ± 11.2 | 39.8 b ± 15.3 | 32.6 b ± 21.4 | 56.1 a ± 22.4 | *** | ** | *** |
| 10 °C | 44.9 z ± 28.6 | 22.4 b,z ± 1.0 | 252.9 a,y ± 73.4 | 168.3 a,yz ± 140.7 | 55.1 a,z ± 0.11 | |||
| 20 °C | 14.3 z ± 3.1 | 60.2 a,z ± 7.2 | 226.4 a,y ± 48.9 | 228.4 a,y ± 11.2 | 14.3 b,z ± 0.04 | |||
| Fatty Acids and Indexes | SB | GB | CB | CSB | CLB | p-Value |
|---|---|---|---|---|---|---|
| C4:0 | 4.18 a ± 0.04 | 4.11 a ± 0.08 | 2.77 b ± 0.05 | 2.39 c ± 0.09 | 1.72 d ± 0.00 | *** |
| C6:0 | 3.84 a ± 0.05 | 3.77 a ± 0.10 | 2.53 b ± 0.13 | 2.18 c ± 0.04 | 1.45 d ± 0.02 | *** |
| C8:0 | 3.43 a ± 0.07 | 3.46 a ± 0.02 | 2.31 b ± 0.02 | 2.07 c ± 0.00 | 1.52 d ± 0.02 | *** |
| C10:0 | 6.53 a ± 0.13 | 5.16 b ± 0.13 | 3.06 c ± 0.04 | 2.51 d ± 0.02 | 2.34 d ± 0.04 | *** |
| C10:1 | 0.23 a ± 0.02 | 0.13 b ± 0.00 | 0.30 a ± 0.02 | 0.23 a ± 0.01 | 0.25 a ± 0.03 | ** |
| C12:0 | 4.75 a ± 0.04 | 4.16 bc ± 0.12 | 3.81 c ± 0.14 | 3.51 cd ± 0.12 | 2.81 d ± 0.07 | *** |
| C12:1 | 0.13 cd ± 0.01 | 0.19 b ± 0.00 | 0.11 d ± 0.01 | 0.18 bc ± 0.01 | 0.32 a ± 0.03 | *** |
| C14:0 | 10.32 c ± 0.04 | 9.20 d ± 0.05 | 12.29 a ± 0.10 | 11.77 b ± 0.13 | 7.02 e ± 0.03 | *** |
| iC15:0 | 0.17 b ± 0.00 | 0.18 b ± 0.01 | 0.25 a ± 0.03 | 0.18 b ± 0.01 | 0.24 a ± 0.00 | ** |
| C14:1 | 0.45 d ± 0.00 | 0.27 e ± 0.03 | 1.40 a ± 0.06 | 1.22 b ± 0.04 | 1.03 c ± 0.05 | *** |
| aC15:0 | 0.11 c ± 0.01 | 0.23 b ± 0.01 | 0.00 d ± 0.00 | 0.08 c ± 0.01 | 0.32 a ± 0.01 | *** |
| C15:0 | 0.78 c ± 0.02 | 0.55 d ± 0.04 | 1.19 a ± 0.01 | 1.10 b ± 0.00 | 0.84 c ± 0.01 | *** |
| iC16:0 | 0.11 ab ± 0.01 | 0.13 a ± 0.01 | 0.00 c ± 0.00 | 0.07 b ± 0.02 | 0.11 ab ± 0.00 | *** |
| C15:1 | 0.13 d ± 0.01 | 0.14 cd ± 0.02 | 0.25 ab ± 0.01 | 0.19 bc ± 0.02 | 0.29 a ± 0.02 | *** |
| C16:0 | 27.97 d ± 0.19 | 31.05 c ± 0.05 | 32.83 b ± 0.40 | 33.64 b ± 0.66 | 36.48 a ± 0.08 | *** |
| C16:1 | 1.02 b ± 0.08 | 0.77 c ± 0.01 | 1.66 a ± 0.09 | 1.52 a ± 0.03 | 0.95 bc ± 0.02 | *** |
| iC17:0 | 0.23 b ± 0.04 | 0.19 b ± 0.00 | 0.28 ab ± 0.00 | 0.33 a ± 0.04 | 0.27 ab ± 0.01 | * |
| C17:0 | 0.43 a ± 0.04 | 0.31 b ± 0.01 | 0.45 a ± 0.00 | 0.43 a ± 0.03 | 0.26 b ± 0.00 | ** |
| C17:1 | 0.20 ± 0.02 | 0.22 ± 0.01 | 0.14 ± 0.01 | 0.22 ± 0.01 | 0.11 ± 0.15 | ns |
| C18:0 | 11.52 d ± 0.34 | 15.86 b ± 0.27 | 10.93 d ± 0.13 | 12.90 c ± 0.03 | 21.81 a ± 0.17 | *** |
| C18:1n9t | 1.45 a ± 0.07 | 1.17 b ± 0.04 | 0.82 c ± 0.04 | 0.32 d ± 0.06 | 0.83 c ± 0.04 | *** |
| C18:1n9c | 16.62 b ± 0.28 | 15.37 c ± 0.29 | 19.09 a ± 0.02 | 18.85 a ± 0.31 | 14.95 c ± 0.07 | *** |
| C18:2n6t | 0.17 b ± 0.00 | 0.21 ab ± 0.02 | 0.14 b ± 0.01 | 0.27 a ± 0.03 | 0.15 b ± 0.03 | * |
| C18:2n6c | 2.93 b ± 0.01 | 2.01 d ± 0.05 | 2.49 c ± 0.10 | 2.80 b ± 0.02 | 3.17 a ± 0.01 | *** |
| C18:3n6g | 0.08 ± 0.01 | 0.07 ± 0.00 | 0.05 ± 0.01 | 0.06 ± 0.01 | 0.07 ± 0.03 | ns |
| C20:0 | 0.17 a ± 0.01 | 0.16 a ± 0.01 | 0.09 b ± 0.01 | 0.14 ab ± 0.03 | 0.11 ab ± 0.01 | * |
| C18:3n3a | 1.52 a ± 0.09 | 0.21 b ± 0.02 | 0.29 b ± 0.02 | 0.33 b ± 0.04 | 0.16 b ± 0.01 | *** |
| C20:1n9 | 0.47 b ± 0.00 | 0.69 a ± 0.00 | 0.35 bc ± 0.05 | 0.37 bc ± 0.09 | 0.26 c ± 0.01 | ** |
| C20:3n6 | 0.05 b ± 0.00 | 0.06 b ± 0.00 | 0.12 a ± 0.01 | 0.15 a ± 0.02 | 0.17 a ± 0.02 | ** |
| SFA | 74.55 c ± 0.16 | 78.51 a ± 0.21 | 72.79 d ± 0.18 | 73.31 d ± 0.35 | 77.28 b ± 0.15 | *** |
| MUFA | 20.70 c ± 0.22 | 18.95 d ± 0.25 | 24.11 a ± 0.07 | 23.09 b ± 0.38 | 19.00 d ± 0.11 | *** |
| PUFA | 4.75 a ± 0.07 | 2.54 d ± 0.04 | 3.09 c ± 0.11 | 3.60 b ± 0.02 | 3.72 b ± 0.04 | *** |
| SCFA | 17.97 a ± 0.19 | 16.49 b ± 0.08 | 10.67 c ± 0.23 | 9.15 d ± 0.15 | 7.02 e ± 0.01 | *** |
| MCFA | 44.25 d ± 0.27 | 45.39 c ± 0.13 | 50.48 a ± 0.02 | 50.41 a ± 0.36 | 47.61 b ± 0.32 | *** |
| LCFA | 11.90 d ± 0.62 | 16.17 b ± 0.05 | 11.24 d ± 0.19 | 13.26 c ± 0.33 | 22.05 a ± 0.00 | *** |
| n-3 | 3.23 b ± 0.02 | 2.34 d ± 0.06 | 2.80 c ± 0.09 | 3.28 b ± 0.02 | 3.56 a ± 0.05 | *** |
| n-6 | 1.52 a ± 0.09 | 0.21 b ± 0.02 | 0.29 b ± 0.02 | 0.33 b ± 0.04 | 0.16 b ± 0.01 | *** |
| n-9 | 16.62 b ± 0.28 | 15.37 c ± 0.29 | 19.09 a ± 0.02 | 18.85 a ± 0.31 | 14.95 c ± 0.07 | *** |
| Atherogenic index | 3.58 b ± 0.04 | 3.80 a ± 0.06 | 3.56 b ± 0.02 | 3.65 ab ± 0.06 | 3.54 b ± 0.01 | ** |
| Thrombogenic index | 4.01 d ± 0.01 | 5.23 b ± 0.09 | 4.13 cd ± 0.01 | 4.39 c ± 0.10 | 5.77 a ± 0.06 | *** |
| Spreadability index | 1.68 c ± 0.04 | 2.02 b ± 0.04 | 1.72 c ± 0.06 | 1.79 c ± 0.06 | 2.44 a ± 0.02 | *** |
| Volatile Compounds | LRI | SB | GB | CB | CSB | CLB | p-Value |
|---|---|---|---|---|---|---|---|
| Ketones | |||||||
| Acetone | 859 | 1.32 c ± 1.32 | 4.72 bc ± 1.15 | 18.61 ab ± 4.58 | 5.32 bc ± 1.74 | 20.06 a ± 10.93 | ** |
| 2-Pentanone | 990 | 5.23 ± 5.00 | 0.96 ± 0.50 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 | ns |
| Heptan-2-one | 1186 | 30.97 a ± 0.15 | 2.75 c ± 0.51 | 2.75 c ± 0.39 | 2.00 c ± 0.55 | 12.55 b ± 0.14 | *** |
| 3-Hydroxy 2-butanone (acetoin) | 1299 | 0.65 c ± 0.25 | 25.4 a ± 2.36 | 15.69 b ± 2.96 | 31.94 a ± 3.77 | 6.28 c ± 1.37 | *** |
| 2-Nonanone | 1396 | 47.57 a ± 4.08 | 8.48 b ± 0.47 | 2.04 b ± 0.02 | 2.01 b ± 0.05 | 5.14 b ± 4.32 | *** |
| 8-Nonen-2-one | 1453 | 2.92 a ± 0.10 | 0.70 b ± 0.01 | 0.98 b ± 0.21 | 0.72 b ± 0.02 | 0.00 c ± 0.00 | *** |
| 2-Undecanone | 1613 | 2.70 a ± 0.24 | 1.67 b ± 0.10 | 0.26 c ± 0.03 | 0.11 c ± 0.02 | 0.00 c ± 0.00 | *** |
| Acetophenone | 1672 | 0.27 ± 0.08 | 0.39 ± 0.17 | 0.48 ± 0.14 | 0.30 ± 0.17 | 0.43 ± 0.42 | ns |
| Total ketones (8) | 91.63 | 45.07 | 40.81 | 42.40 | 44.46 | ||
| Terpenes | |||||||
| α-Pinene | 1024 | 0.00 b ± 0.00 | 7.19 a ± 2.12 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | *** |
| p-Menth-3-ene | 1129 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | 1.40 a ± 0.16 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | *** |
| Limonene | 1202 | 0.00 b ± 0.00 | 0.66 b ± 0.08 | 7.16 a ± 1.89 | 9.95 a ± 1.95 | 3.25 b ± 0.67 | *** |
| p-Cymene | 1277 | 0.00 d ± 0.00 | 0.00 d ± 0.00 | 11.96 b ± 0.64 | 15.11 a ± 0.64 | 1.26 c ± 0.42 | *** |
| 4-Carene | 1289 | 1.13 a ± 0.05 | 0.00 b ± 0.00 | 1.42 a ± 0.22 | 1.19 a ± 0.34 | 0.00 b ± 0.00 | *** |
| Total terpenes (5) | 1.13 | 7.85 | 21.94 | 26.25 | 4.51 | ||
| Acids | |||||||
| Acetic acid | 1477 | 0.13 c ± 0.01 | 7.09 ab ± 0.11 | 10.16 a ± 1.72 | 5.92 ab ± 1.40 | 4.09 bc ± 3.93 | *** |
| Butanoic acid | 1654 | 2.85 ± 0.17 | 4.39 ± 1.50 | 2.74 ± 0.13 | 1.60 ± 0.47 | 2.28 ± 1.78 | ns |
| 2-Methylbutanoic acid | 1695 | 0.24 b ± 0.03 | 0.27 ab ± 0.09 | 0.38 a ± 0.05 | 0.10 c ± 0.01 | 0.00 c ± 0.00 | *** |
| Hexanoic acid | 1873 | 0.30 b ± 0.08 | 3.00 a ± 1.19 | 1.22 b ± 0.38 | 1.04 b ± 0.22 | 3.26 a ± 0.05 | *** |
| Octanoic acid | 2088 | 0.11 c ± 0.01 | 0.00 c ± 0.00 | 0.80 ab ± 0.42 | 0.50 bc ± 0.06 | 1.07 a ± 0.03 | *** |
| Total acids (5) | 3.63 | 14.75 | 15.30 | 9.16 | 10.70 | ||
| Aliphatic hydrocarbons | |||||||
| Nonane | 1001 | 0.00 c ± 0.00 | 8.70 b ± 1.81 | 3.25 bc ± 2.89 | 3.81 bc ± 1.77 | 17.59 a ± 5.00 | *** |
| Undecane | 1183 | 0.00 ± 0.00 | 1.55 ± 1.50 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.88 ± 0.30 | ns |
| Tetradecane | 1504 | 0.58 bc ± 0.20 | 0.34 c ± 0.19 | 1.34 a ± 0.08 | 0.35 c ± 0.31 | 1.19 ab ± 0.33 | *** |
| Hexadecane | 1608 | 0.04 c ± 0.01 | 0.51 bc ± 0.12 | 1.23 ab ± 0.58 | 0.50 bc ± 0.21 | 1.82 a ± 0.36 | *** |
| Heptadecane | 1709 | 0.06 c ± 0.01 | 0.55 bc ± 0.07 | 0.88 ab ± 0.19 | 0.49 bc ± 0.10 | 1.14 a ± 0.35 | *** |
| Nonadecane | 1912 | 0.04 b ± 0.01 | 0.23 b ± 0.08 | 0.26 b ± 0.24 | 0.20 b ± 0.04 | 0.69 a ± 0.03 | *** |
| Total alip. hydrocarb. (6) | 0.72 | 11.88 | 6.96 | 5.35 | 23.31 | ||
| Alcohols | |||||||
| Ethanol | 941 | 0.30 b ± 0.14 | 7.77 a ± 4.90 | 0.00 b ± 0.00 | 4.73 ab ± 1.25 | 1.94 ab ± 0.91 | ** |
| 1-Pentanol | 1263 | 0.14 c ± 0.10 | 0.53 b ± 0.12 | 0.00 c ± 0.00 | 1.23 a ± 0.13 | 0.70 b ± 0.14 | *** |
| 1-Nonanol | 1497 | 0.06 b ± 0.02 | 0.59 ab ± 0.30 | 0.66 ab ± 0.42 | 0.56 ab ± 0.21 | 0.88 a ± 0.06 | * |
| Octan1-ol | 1569 | 0.34 a ± 0.14 | 0.25 ab ± 0.16 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | 0.22 ab ± 0.08 | ** |
| 2,3- Butanediol | 1593 | 0.46 ± 0.09 | 0.69 ± 0.02 | 2.48 ± 2.29 | 1.99 ± 0.40 | 0.49 ± 0.12 | ns |
| Total alcohols (5) | 1.30 | 9.83 | 3.14 | 8.51 | 4.23 | ||
| Aldehydes | |||||||
| Hexanal | 1086 | 0.50 c ± 0.04 | 1.84 b ± 0.04 | 0.00 d ± 0.00 | 1.49 b ± 0.01 | 2.53 a ± 0.39 | *** |
| Benzaldehyde | 1543 | 0.28 c ± 0.21 | 0.92 bc ± 0.23 | 0.98 b ± 0.35 | 0.72 bc ± 0.17 | 1.67 a ± 0.21 | *** |
| Tetradecanal | 1727 | 0.14 b ± 0.00 | 1.42 a ± 0.38 | 2.02 a ± 0.33 | 1.28 a ± 0.20 | 2.06 a ± 0.56 | *** |
| Octadecanal | 1942 | 0.10 ± 0.03 | 0.37 ± 0.09 | 0.47 ± 0.16 | 0.37 ± 0.14 | 0.62 ± 0.48 | ns |
| Total aldehydes (4) | 1.02 | 4.55 | 3.47 | 3.86 | 6.88 | ||
| Lactones | |||||||
| δ-Caprolactone | 1820 | 0.04 c ± 0.01 | 1.27 b ± 0.15 | 1.99 a ± 0.38 | 1.28 b ± 0.28 | 1.00 b ± 0.05 | *** |
| δ-Octalactone | 1996 | 0.05 b ± 0.01 | 0.53 ab ± 0.21 | 0.97 a ± 0.05 | 0.52 ab ± 0.13 | 0.90 a ± 0.41 | ** |
| Total lactones (2) | 0.09 | 1.80 | 2.96 | 1.80 | 1.90 | ||
| Aromatic hydrocarbons | |||||||
| Toluene | 1047 | 0.00 b ± 0.00 | 1.84 ab ± 1.25 | 1.05 ab ± 0.01 | 0.00 b ± 0.00 | 2.78 a ± 1.14 | ** |
| p-Xylene | 1138 | 0.23 a ± 0.01 | 0.24 a ± 0.14 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | *** |
| Phenol | 2039 | 0.04 ± 0.01 | 0.32 ± 0.11 | 0.71 ± 0.35 | 0.46 ± 0.19 | 0.79 ± 0.46 | ns |
| Total arom. hydrocarb. (3) | 0.24 | 2.40 | 1.76 | 0.46 | 3.57 | ||
| Esters | |||||||
| Ethyl butanoate | 1037 | 0.00 c ± 0.00 | 0.00 c ± 0.00 | 1.89 a ± 0.33 | 1.33 b ± 0.29 | 0.00 c ± 0.00 | *** |
| Butyl hexanoate | 1412 | 0.06 b ± 0.01 | 0.30 a ± 0.10 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | 0.00 b ± 0.00 | *** |
| 2-Phenylethyl acetate | 1862 | 0.00 ± 0.00 | 0.30 ± 0.20 | 0.32 ± 0.25 | 0.14 ± 0.03 | 0.00 ± 0.00 | ns |
| Methyl pentadecanoate | 2028 | 0.02 b ± 0.01 | 0.17 ab ± 0.10 | 0.33 ab ± 0.20 | 0.21 ab ± 0.04 | 0.43 a ± 0.16 | * |
| Total esters (4) | 0.08 | 0.77 | 2.54 | 1.68 | 0.43 | ||
| Suphur compounds | |||||||
| Dimethylsulfone | 1934 | 0.11 b ± 0.02 | 1.12 a ± 0.43 | 1.12 a ± 0.13 | 0.54 b ± 0.04 | 0.00 b ± 0.00 | *** |
| Total sulphur comp. (1) | 0.11 | 1.12 | 1.12 | 0.54 | 0.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vioque-Amor, M.; Gómez-Díaz, R.; Del Río-Celestino, M.; Avilés-Ramírez, C. Butter from Different Species: Composition and Quality Parameters of Products Commercialized in the South of Spain. Animals 2023, 13, 3559. https://doi.org/10.3390/ani13223559
Vioque-Amor M, Gómez-Díaz R, Del Río-Celestino M, Avilés-Ramírez C. Butter from Different Species: Composition and Quality Parameters of Products Commercialized in the South of Spain. Animals. 2023; 13(22):3559. https://doi.org/10.3390/ani13223559
Chicago/Turabian StyleVioque-Amor, Montserrat, Rafael Gómez-Díaz, Mercedes Del Río-Celestino, and Carmen Avilés-Ramírez. 2023. "Butter from Different Species: Composition and Quality Parameters of Products Commercialized in the South of Spain" Animals 13, no. 22: 3559. https://doi.org/10.3390/ani13223559
APA StyleVioque-Amor, M., Gómez-Díaz, R., Del Río-Celestino, M., & Avilés-Ramírez, C. (2023). Butter from Different Species: Composition and Quality Parameters of Products Commercialized in the South of Spain. Animals, 13(22), 3559. https://doi.org/10.3390/ani13223559






