Genotype by Environment Interactions (G*E) of Chickens Tested in Ethiopia Using Body Weight as a Performance Trait
Abstract
Simple Summary
Abstract
1. Introduction
- (1)
- Is S*E present, i.e., do different strains react differently to the various Ethiopian environments;
- (2)
- Which strain performs best in which environment in terms of predicted BW.
2. Materials and Methods
2.1. Study Design and Data Collection
2.2. Strain Descriptions
2.2.1. Horro
2.2.2. Koekoek
2.2.3. Kuroiler
2.2.4. S-RIR and Sasso
2.3. Area Descriptions
2.4. Data Cleaning
2.5. Statistical Analysis
3. Results
3.1. Simple S*E Model
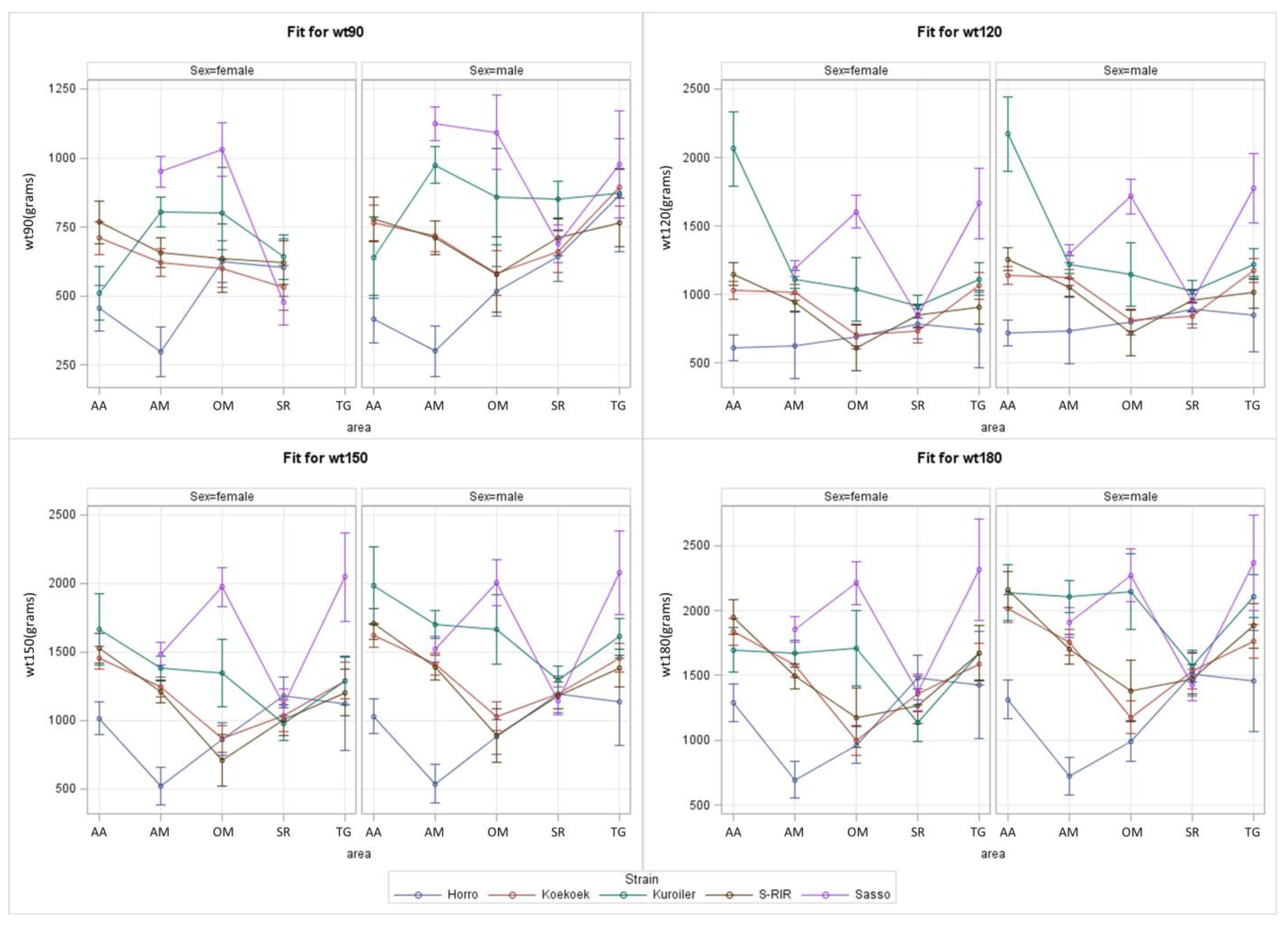
3.2. Complex S*E Model for Ethiopian Regions
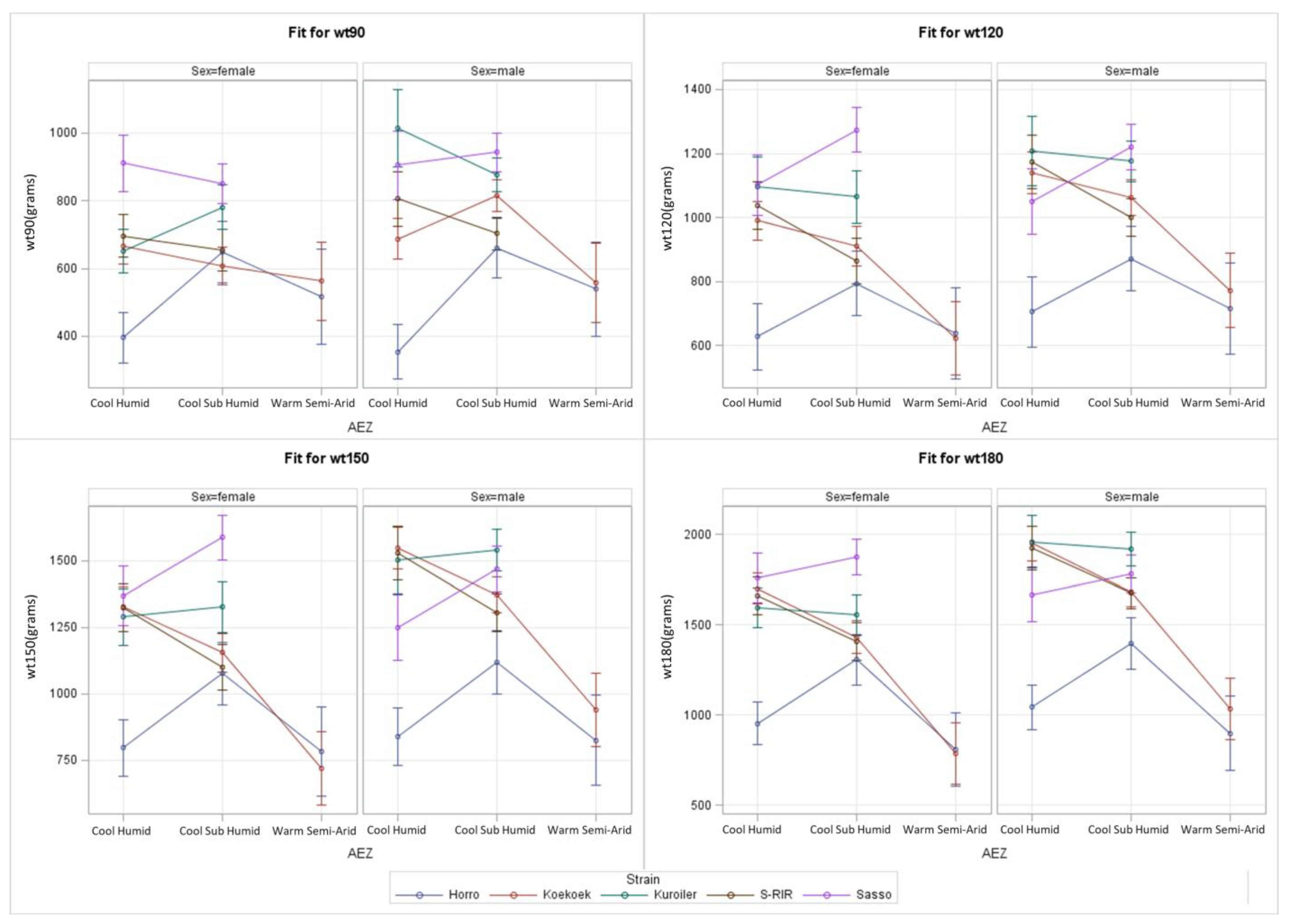
3.3. Complex S*E Model for AEZ
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. Decision Tools for Family Poultry Development. In FAO Animal Production and Health Guidelines; Food and Agriculture Organization of the United Nationsin in Collaboration with the International Fund for Agricultural Development and KYEEMA Foundation: Rome, Italy, 2014; ISBN 978-92-5-108087-0. [Google Scholar]
- Vernooij, A.; Masaki, M.N.; Meijer-Willems, D. Regionalisation in Poultry Developmen in Eastern Africa; Wageningen Livestock Research Rapport; Wageningen Livestock Research: Wageningen, The Netherlands, 2018; p. 47. [Google Scholar]
- Goromela, E.H.; Sonaiya, E.B.; Assefa, G.; Mbaga, S.H.; Adebambo, O.; Abegaz, S.; Bamidele, O.; Esatu, W.; Alemayehu, T.; Bruno, J.; et al. Understanding the Entry Points for Improving the Smallholder Chicken Production in Sub-Saharan Africa. In Proceedings of the 7th All Africa Conference on Animal Agriculture, Accra, Ghana, 29 July–2 August 2019. [Google Scholar]
- Alexandratos, N.; Bruinsma, J.; Boedeker, G.; Schmidhuber, J.; Broca, S.; Shetty, P.; Ottaviani, M. World Agriculture: Towards 2030/2050; Interim Report; Prospects for Food, Nutrition, Agriculture and Major Commodity Groups; Global Perspective Studies Unit. Food and Agriculture Organization of the United Nations: Rome, Italy, 2006; Available online: http://www.fao.org/fileadmin/user_upload/esag/docs/Interim_report_AT2050web.pdf (accessed on 26 August 2021).
- Alexandratos, N.; Bruinsma, J. World Agriculture towards 2030/2050: The 2012 Revision; ESA Working Paper No. 12-03; Agricultural Development Economics Division Food and Agriculture Organization of the United Nations: Roma, Italy, 2012; p. 154. Available online: http://www.fao.org/3/ap106e/ap106e.pdf (accessed on 26 August 2021).
- Oonincx, D.G.A.B.; de Boer, I.J.M. Environmental Impact of the Production of Mealworms as a Protein Source for Humans—A Life Cycle Assessment. PLoS ONE 2012, 7, 51145. [Google Scholar] [CrossRef] [PubMed]
- Bettridge, J.M.; Psifidi, A.; Terfa, Z.G.; Desta, T.T.; Lozano-Jaramillo, M.; Dessie, T.; Kaiser, P.; Wigley, P.; Hanotte, O.; Christley, R.M. The Role of Local Adaptation in Sustainable Village Chicken Production. Nat. Sustain. 2018, 1, 574–582. [Google Scholar] [CrossRef]
- African Chicken Genetic Gains (ACGG). Available online: https://www.ilri.org/research/projects/african-chicken-genetic-gains-acgg (accessed on 27 August 2021).
- Tropical Poultry Genetic Solutions. Available online: https://africacgg.net/ (accessed on 27 August 2021).
- Lozano-Jaramillo, M. Predicting Breed by Environment Interaction Using Ecological Modelling. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2019. [Google Scholar]
- Lozano-Jaramillo, M.; Alemu, S.W.; Dessie, T.; Komen, H.; Bastiaansen, J.W.M. Using Phenotypic Distribution Models to Predict Livestock Performance. Sci. Rep. 2019, 9, 15371. [Google Scholar] [CrossRef]
- Wondmeneh, E.; van der Waaij, E.H.; Dessie, T.; Okeyo Mwai, A.; van Arendonk, J.A.M. A Running Breeding Program for Indigenous Chickens in Ethiopia: Evaluation of Success. In Proceedings of the 10th World Congress of Genetics Applied to Livestock Production, Vancouver, BC, Canada, 17–22 August 2014; American Society of Animal Science: Vancouver, BC, Canada, 2014. [Google Scholar]
- Falconer, D.S. The Problem of Environment and Selection. Am. Nat. 1952, 86, 293–298. [Google Scholar] [CrossRef]
- Falconer, D.S.; Mackay, T.F.C. Introduction to Quantitative Genetics, 4th ed.; Prentice Hall: Harlow, UK, 1996; ISBN 978-0-582-24302-6. [Google Scholar]
- Yakubu, A.; Salako, A.E.; Ige, A.O. Effects of Genotype and Housing System on the Laying Performance of Chickens in Different Seasons in the Semi-Humid Tropics. Int. J. Poult. Sci. 2007, 6, 434–439. [Google Scholar] [CrossRef]
- Mulder, H.A.; Bijma, P. Effects of Genotype × Environment Interaction on Genetic Gain in Breeding Programs. J. Anim. Sci. 2005, 83, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Ali, K.O.; Katule, A.M.; Syrstad, O. Genotype × Environment Interaction in Growing Chickens: Comparison of Four Genetic Groups on Two Rearing Systems under Tropical Conditions. Acta Agric. Scand. Sect. A—Anim. Sci. 2000, 50, 65–71. [Google Scholar] [CrossRef]
- Bekele, F.; Gjøen, H.M.; Kathle, J.; Adnøy, T.; Abebe, G. Genotype X Environment Interaction in Two Breeds of Chickens Kept under Two Management Systems in Southern Ethiopia. Trop. Anim. Health Prod. 2009, 41, 1101–1114. [Google Scholar] [CrossRef]
- Lwelamira, J. Genotype-Environmental (G × E) Interaction for Body Weights for Kuchi Chicken Ecotype of Tanzania Reared On-Station and On-Farm. Int. J. Poult. Sci. 2012, 11, 96–102. [Google Scholar] [CrossRef][Green Version]
- Home|ARRIVE Guidelines. Available online: https://arriveguidelines.org/ (accessed on 21 September 2023).
- Dana, N.; van der Waaij, E.H.; van Arendonk, J.A.M. Genetic and Phenotypic Parameter Estimates for Body Weights and Egg Production in Horro Chicken of Ethiopia. Trop. Anim. Health Prod. 2011, 43, 21–28. [Google Scholar] [CrossRef]
- Dessie, T.; Esatu, W.; Waaij, L.V.; Zegeye, F.; Gizaw, S.; Okeyo Mwai, A.; van Arendonk, J.A.M. Village Chicken Production in the Central and Western Highlands of Ethiopia: Characteristics and Strategies for Improvement; International Livestock Research Institute (ILRI): Nairobi, Kenya, 2013. [Google Scholar]
- Getachew, F.; Wondmeneh, E.; Dessie, T. Preliminary Information on Chicken Strains to Be Tested in Ethiopia. Afr. Chick. Genet. Gains 2016, 2, 1–3. Available online: https://core.ac.uk/download/pdf/132683011.pdf (accessed on 8 March 2022).
- Lozano-Jaramillo, M.; Bastiaansen, J.W.M.; Dessie, T.; Komen, H. Use of Geographic Information System Tools to Predict Animal Breed Suitability for Different Agro-Ecological Zones. Animal 2019, 13, 1536–1543. [Google Scholar] [CrossRef] [PubMed]
- Getiso, A. Management Practices and Productive Performances of Sasso Chickens Breed under Village Production System in SNNPR, Ethiopia. J. Biol. 2017, 7, 120–135. Available online: https://www.iiste.org/Journals/index.php/JBAH/article/view/36482 (accessed on 26 August 2021).
- Federal Democratic Republic of Ethiopia Regional States. Available online: http://www.ethiopia.gov.et/regional-states1 (accessed on 23 April 2020).
- Alemu, M.M.; Bawoke, G.T. Analysis of Spatial Variability and Temporal Trends of Rainfall in Amhara Region, Ethiopia. J. Water Clim. Change 2019, 11, 1505–1520. [Google Scholar] [CrossRef]
- Osei-Amponsah, R.; Kayang, B.B.; Naazie, A.; Barchia, I.M.; Arthur, P.F. Evaluation of Models to Describe Temporal Growth in Local Chickens of Ghana. Iran. J. Appl. Anim. Sci. 2014, 4, 855–861. Available online: https://ijas.rasht.iau.ir/article_513494.html (accessed on 9 May 2022).
- Youssao, I.a.K.; Alkoiret, I.T.; Dahouda, M.; Assogba, M.N.; Idrissou, N.-D.; Kayang, B.B.; Yapi-Gnaoré, V.; Assogba, H.M.; Houinsou, A.S.; Ahounou, S.G.; et al. Comparison of Growth Performance, Carcass Characteristics and Meat Quality of Benin Indigenous Chickens and Label Rouge (T55 × SA51). Afr. J. Biotechnol. 2012, 11, 15569–15579. [Google Scholar] [CrossRef]
- SAS Institute Inc. Base SAS© 9.4 (Version 9.4) Procedures Guide, 7th ed.; SAS Institute Inc.: Cary, NC, USA, 2017. [Google Scholar]
- Abegaz, S.; Esatu, W.; Assefa, G.; Goromela, E.H.; Sonaiya, E.B.; Mbaga, S.H.; Adebambo, O.; Bamidele, O.; Teressa, A.; Bruno, J.; et al. On-Farm Performance Testing of Tropically Adaptable Chicken Strains under Small Holder Management in Three Countries of Sub-Saharan Africa. In Proceedings of the 7th All Africa Conference on Animal Agriculture, Accra, Ghana, 29 July–2 August 2019. [Google Scholar]
- Wondmeneh, E.; Van der Waaij, E.H.; Udo, H.M.J.; Tadelle, D.; Van Arendonk, J.A.M. Comparison of Different Poultry Breeds under Station and On-Farm Conditions in Ethiopia. Livest. Sci. 2016, 183, 72–77. [Google Scholar] [CrossRef]
- Bamidele, O.; Sonaiya, E.B.; Adebambo, O.A.; Dessie, T. On-Station Performance Evaluation of Improved Tropically Adapted Chicken Breeds for Smallholder Poultry Production Systems in Nigeria. Trop. Anim. Health Prod. 2020, 52, 1541–1548. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.; Xie, J.; Boggess, M.; Galukande, E.; Semambo, D.; Sharma, S. Higher Weight Gain by Kuroiler Chickens than Indigenous Chickens Raised under Scavenging Conditions by Rural Households in Uganda. Livest. Res. Rural. Dev. 2015, 27, 1–4. Available online: http://www.lrrd.org/lrrd27/9/shar27178.html (accessed on 27 August 2021).
- Alemu, S.W.; Hanotte, O.; Kebede, F.G.; Esatu, W.; Abegaz, S.; Bruno, J.E.; Abrar, B.; Alemayehu, T.; Mrode, R.; Dessie, T. Evaluation of Live-Body Weight and the Number of Eggs Produced for Introduced and Local Chickens in Ethiopia. Acta Agric. Scand. Sect. A Anim. Sci. 2021, 70, 71–77. [Google Scholar] [CrossRef]
- De Kinderen, M.A.J.; Sölkner, J.; Mészáros, G.; Alemu, S.W.; Esatu, W.; Bastiaansen, J.W.M.; Komen, H.; Dessie, T. Genotype by Environment Interactions for Egg Number and Egg Weight of Five Dual-Purpose Chicken Breeds in Different Zones of Oromia Region in Ethiopia. Acta Fytotech. Zootech. 2020, 23, 205–213. [Google Scholar] [CrossRef]
- Dana, N.; van der Waaij, L.H.; Dessie, T.; van Arendonk, J.A.M. Production Objectives and Trait Preferences of Village Poultry Producers of Ethiopia: Implications for Designing Breeding Schemes Utilizing Indigenous Chicken Genetic Resources. Trop. Anim. Health Prod. 2010, 42, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Woldegiorgiss, W.; Waaij, E.; Udo, H.M.J.; Dessie, T.; Arendonk, J. Village Poultry Production System: Perception of Farmers and Simulation of Impacts of Interventions. Afr. J. Agric. Res. 2016, 11, 2075–2081. [Google Scholar] [CrossRef]
- Yitayih, M.; Esatu, W.; Geremew, K.; Yemane, T.; Dessie, T. Economic Contribution of Tropically Adapted and More Productive Breeds Based Village Chicken Production in Sub-Saharan Africa: ACGG Model. In Proceedings of the Seventh All Africa Conference on Animal Agriculture, Accra, Ghana, 2 August 2019. [Google Scholar]
- Esatu, W. Highlights of the Results of the ACGG Baseline Survey in Ethiopia. In Proceedings of the ACGG Ethiopia Second National Innovation Platform Meeting, Debre Zeit, Ethiopia, 22–23 March 2016; Ethiopian Institute of Agricultural Research: Addis Ababa, Ethiopia, 2016. [Google Scholar]
- Bamidele, O.; Sonaiya, E.B.; Adebambo, O.; Assefa, G.; Abegaz, S.; Esatu, W.; Goromela, E.H.; Mbaga, S.H.; Dessie, T. On-Station Performance Evaluation of Improved Tropically Adapted Chicken Strains for Smallholder Poultry Production Systems in Sub-Saharan Africa. In Proceedings of the Seventh All Africa Conference on Animal Agriculture, Accra, Ghana, 2 August 2019. [Google Scholar]
- Ibrahim, D.; Goshu, G.; Esatu, W.; Cahaner, A. Dual-Purpose Production of Genetically Different Chicken Crossbreeds in Ethiopia. 1. Parent Stocks’ Feed Intake, Body Weight, and Reproductive Performance. Poult. Sci. 2019, 98, 3119–3129. [Google Scholar] [CrossRef]
- Besbes, B. Genotype Evaluation and Breeding of Poultry for Performance under Sub-Optimal Village Conditions. World’s Poult. Sci. J. 2009, 65, 260–271. [Google Scholar] [CrossRef]
- Getachew, F.; Worku, S.; Esatu, W.; Dessie, T. Unleashing the Power of Data in Transforming Livestock Agriculture in Ethiopia; International Livestock Research Institute: Nairobi, Kenya; EIAR: Addis Ababa, Ethiopia, 2019. [Google Scholar]
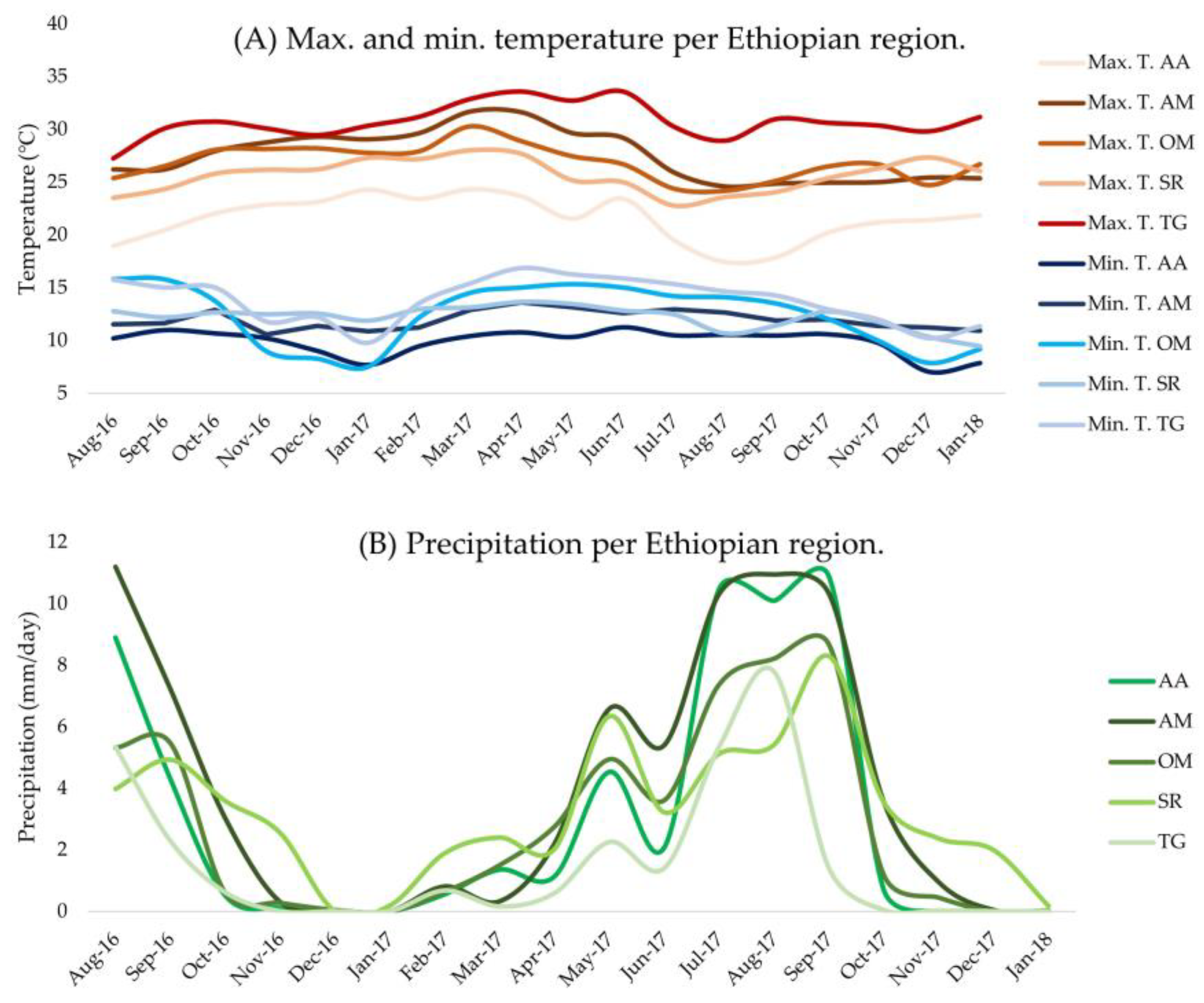
| Region | AA | AM | OM | SR | TG |
|---|---|---|---|---|---|
| Climate description | Mild, Afro-Alpine temp-, warm temperate | 25% cool to cold, 44% warm to cool, 31% warm to hot | Big diversity; dry, tropical rainy climate, temperate rainy | 56% hottest lowland, 44% temperate | 39% semi-arid, 49% warm temp-, 12% temperate |
| Temp. (°C) | 9.9–24.6 | 15–21 | 18–39 | 15–30 | - |
| Rain (mm) | - | 80% Ethiopia 1111 | 410–2000 | 500–2200 | 450–980 |
| Altitude (m) | 2200–2500 | High > 1500, Low 500–1500 | >500–4607 | 376–4207 56% < 1500 | 600–2700 |
| AEZ | Cool humid | Cool humid, cool sub-humid | Cool sub-humid, warm semi-arid | Cool humid, cool sub-humid | Cool sub-humid |
| Ethiopian Region | Elevation (m) | Max. T. (°C) | Min. T. (°C) | Precipitation (mm/day) | Relative Humidity (%) |
|---|---|---|---|---|---|
| AA | 2548 | 21.5 | 9.9 | 3.1 | 56 |
| AM | 1936 | 27.5 | 12.0 | 4.1 | - |
| OM | 1707 | 26.9 | 12.4 | 2.8 | - |
| SR | 2032 | 25.7 | 12.2 | 3.2 | 67 |
| TG | 1717 | 30.8 | 13.8 | 1.5 | - |
| Number of weather stations data are obtained from | |||||
| AA | 3 | 3 | 3 | 3 | 2 |
| AM | 9 | 5 | 5 | 10 | - |
| OM | 7 | 5 | 5 | 7 | - |
| SR | 7 | 5 | 5 | 7 | 1 |
| TG | 4 | 4 | 4 | 5 | - |
| Female | Male | ||||
|---|---|---|---|---|---|
| Villages | ER | AEZ | Villages | ER | AEZ |
| 6 | AA | cool humid | 6 | AA | cool humid |
| 7 | AM | cool sub-humid | 7 | AM | cool sub-humid |
| 7 | cool humid | 7 | cool humid | ||
| 10 | OM | cool sub-humid | 6 | OM | cool sub-humid |
| 3 | warm semi-arid | 3 | warm semi-arid | ||
| 5 | SR | cool sub-humid | 1 | SR | cool humid |
| 11 | cool sub-humid | ||||
| 8 | TG | cool sub-humid | |||
| 38 | Tot | 49 | Tot | ||
| N | Birds per hh | N | Birds per hh | ||
| Horro | 157 | 8.1 | Horro | 139 | 6.8 |
| Koekoek | 288 | 9.8 | Koekoek | 303 | 8.2 |
| Kuroiler | 185 | 5.9 | Kuroiler | 178 | 5.5 |
| S-RIR | 196 | 10.4 | S-RIR | 222 | 6.9 |
| Sasso | 173 | 6.7 | Sasso | 147 | 7.8 |
| Tot | 999 | 8.4 | Tot | 989 | 7.2 |
| Age BW (Days) | Min (g) | Max (g) |
|---|---|---|
| 90 | 200 | 2500 |
| 120 | 300 | 3000 |
| 150 | 400 | 3500 |
| 180 | 500 | 4000 |
| Region | AA | AM | OM | SR | TG | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | F | M | F | M | F | M | F | M | F | M | ||||||
| Horro | 37–39 | 32 | 7–41 | 3–43 | 44–47 | 26–29 | 17–23 | 28–30 | 0 | 8 | ||||||
| Koekoek | 82 | 61–63 | 105–108 | 81–85 | 61–62 | 35–37 | 33–35 | 36–39 | 0 | 73 | ||||||
| Kuroiler | 7–40 | 0–2 | 96–100 | 59–62 | 7–8 | 4–7 | 31–35 | 59–63 | 0 | 44 | ||||||
| S-RIR | 43 | 39–40 | 95–103 | 63–65 | 13–16 | 8 | 32–33 | 63–65 | 0 | 41–43 | ||||||
| Sasso | 0 | 0 | 95–98 | 71–72 | 31–40 | 4–5 | 32–35 | 58–61 | 0 | 9 | ||||||
| AEZ | cool humid | cool sub-humid | warm semi-arid | |||||||||||||
| Sex | F | M | F | M | F | M | ||||||||||
| Horro | 44–79 | 35–66 | 45–48 | 44–48 | 19 | 18–19 | ||||||||||
| Koekoek | 135–138 | 100–102 | 112–122 | 164–167 | 26–27 | 26–27 | ||||||||||
| Kuroiler | 60–96 | 28–30 | 81–86 | 142–146 | 0 | 0 | ||||||||||
| S-RIR | 87–94 | 57–58 | 96–101 | 159–163 | 0 | 0 | ||||||||||
| Sasso | 52 | 33–36 | 106–121 | 109–111 | 0 | 0 | ||||||||||
| Effect: | Region | Strain | Sex | R*Sn | R*Sx | Sn*Sx | R*Sn*Sx |
|---|---|---|---|---|---|---|---|
| 90 days | <0.001 | <0.001 | <0.001 | <0.001 | 0.042 | 0.007 | - |
| 120 days | <0.001 | <0.001 | <0.001 | <0.001 | - | - | - |
| 150 days | <0.001 | <0.001 | <0.001 | <0.001 | - | 0.001 | - |
| 180 days | <0.001 | <0.001 | <0.001 | <0.001 | - | <0.001 | - |
| Effect | AEZ | Strain | Sex | A*Sn | A*Sx | Sn*Sx | A*Sn*Sx |
|---|---|---|---|---|---|---|---|
| 90 days | 0.006 | <0.001 | <0.001 | <0.001 | 0.842 | 0.002 | <0.001 |
| 120 days | <0.001 | <0.001 | <0.001 | <0.001 | - | 0.009 | - |
| 150 days | <0.001 | <0.001 | <0.001 | <0.001 | - | <0.001 | - |
| 180 days | <0.001 | <0.001 | <0.001 | <0.001 | - | <0.001 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Kinderen, M.A.J.; Sölkner, J.; Mészáros, G.; Alemu, S.W.; Esatu, W.; Bastiaansen, J.W.M.; Komen, H.; Dessie, T. Genotype by Environment Interactions (G*E) of Chickens Tested in Ethiopia Using Body Weight as a Performance Trait. Animals 2023, 13, 3121. https://doi.org/10.3390/ani13193121
de Kinderen MAJ, Sölkner J, Mészáros G, Alemu SW, Esatu W, Bastiaansen JWM, Komen H, Dessie T. Genotype by Environment Interactions (G*E) of Chickens Tested in Ethiopia Using Body Weight as a Performance Trait. Animals. 2023; 13(19):3121. https://doi.org/10.3390/ani13193121
Chicago/Turabian Stylede Kinderen, Maud A. J., Johann Sölkner, Gábor Mészáros, Setegn W. Alemu, Wondmeneh Esatu, John W. M. Bastiaansen, Hans Komen, and Tadelle Dessie. 2023. "Genotype by Environment Interactions (G*E) of Chickens Tested in Ethiopia Using Body Weight as a Performance Trait" Animals 13, no. 19: 3121. https://doi.org/10.3390/ani13193121
APA Stylede Kinderen, M. A. J., Sölkner, J., Mészáros, G., Alemu, S. W., Esatu, W., Bastiaansen, J. W. M., Komen, H., & Dessie, T. (2023). Genotype by Environment Interactions (G*E) of Chickens Tested in Ethiopia Using Body Weight as a Performance Trait. Animals, 13(19), 3121. https://doi.org/10.3390/ani13193121







