The Effects of Commercially-Relevant Disturbances on Sleep Behaviour in Laying Hens
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Housing
2.3. Surgical Implantation of EEG
2.4. Post-Surgical Recovery and Monitoring
2.5. EEG Devices
2.6. Data Recording
2.7. Data Collection and Processing
2.8. Statistical Analysis
3. Results
3.1. Lights-Off
3.2. Lights-On
4. Discussion
4.1. Lights-Off
4.2. Lights-On
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morton, D.B.; Hau, J. Welfare Assessment and Humane Endpoints. In Handbook of Laboratory Animal Science; CRC Press Inc.: Boca Raton, FL, USA, 2002; pp. 457–486. [Google Scholar]
- Baumans, V. Science-Based Assessment of Animal Welfare: Laboratory Animals. OIE Rev. Sci. Tech. 2005, 24, 503–514. [Google Scholar] [CrossRef]
- Mench, J.A. Assessing Animal Welfare at the Farm and Group Level: A United States Perspective. Anim. Welf. 2003, 12, 493–503. [Google Scholar] [CrossRef]
- Main, D.C.J.; Whay, H.R.; Leeb, C.; Webster, A.J.F. Formal Animal-Based Welfare Assessment in UK Certification Schemes. Anim. Welf. 2007, 16, 233–236. [Google Scholar] [CrossRef]
- Bassett, L.; Buchanan-Smith, H.M. Effects of Predictability on the Welfare of Captive Animals. Appl. Anim. Behav. Sci. 2007, 102, 223–245. [Google Scholar] [CrossRef]
- Sandilands, V.; Tolkamp, B.J.; Savory, C.J.; Kyriazakis, I. Behaviour and Welfare of Broiler Breeders Fed Qualitatively Restricted Diets during Rearing: Are There Viable Alternatives to Quantitative Restriction? Appl. Anim. Behav. Sci. 2006, 96, 53–67. [Google Scholar] [CrossRef]
- Castelhano-Carlos, M.J.; Baumans, V. The Impact of Light, Noise, Cage Cleaning and in-House Transport on Welfare and Stress of Laboratory Rats. Lab. Anim. 2009, 43, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Dawkins, M.S.; Donnelly, C.A.; Jones, T.A. Chicken Welfare Is Influenced More by Housing Conditions than by Stocking Density. Nature 2004, 427, 342–344. [Google Scholar] [CrossRef]
- Appleby, M.C.C.; Mench, J.A.A.; Hughes, B.O.O. Poultry Behaviour and Welfare; CABI Publishing: Wallingford, UK, 2004. [Google Scholar]
- Forslind, S.; Blokhuis, H.J.; Riber, A.B. Disturbance of Resting Behaviour of Broilers under Different Environmental Conditions. Appl. Anim. Behav. Sci. 2021, 242, 105425. [Google Scholar] [CrossRef]
- Blokhuis, H.J. The Relevance of Sleep in Poultry. Worlds. Poult. Sci. J. 1983, 39, 33–37. [Google Scholar] [CrossRef]
- Fraser, D. Animal Behaviour, Animal Welfare and the Scientific Study of Affect. Appl. Anim. Behav. Sci. 2009, 118, 108–117. [Google Scholar] [CrossRef]
- Schwean-Lardner, K.; Fancher, B.I.; Classen, H.L. Impact of Daylength on Behavioural Output in Commercial Broilers. Appl. Anim. Behav. Sci. 2012, 137, 43–52. [Google Scholar] [CrossRef]
- Yngvesson, J.; Wedin, M.; Gunnarsson, S.; Jonsson, L.; Blokhuis, H.; Wallenbeck, A. Let Me Sleep! Welfare of Broilers (Gallus Gallus Domesticus) with Disrupted Resting Behaviour. Acta Agric. Scand. A Anim. Sci. 2017, 67, 123–133. [Google Scholar] [CrossRef]
- Rial, R.V.; Nicolau, M.C.; Gamundi, A.; Akaarir, M.; Aparicio, S.; Garau, C.; Tejada, S.; Roca, C.; Gene, L.; Moranta, D.; et al. The Trivial Function of Sleep. Sleep Med. Rev. 2007, 11, 311–325. [Google Scholar] [CrossRef]
- Assefa, D.Z.; Diaz-Abad, M.; Wickwire, E.M.; Scharf, S.M. The Functions of Sleep. AIMS Neurosci. 2015, 2, 155–171. [Google Scholar] [CrossRef]
- Ookawa, T. Avian Wakefulness and Sleep on the Basis of Recent Electroencephalographic Observations. Poult. Sci. 1972, 51, 1565–1574. [Google Scholar] [CrossRef]
- Flanigan, W.F., Jr.; Wilcox, R.H.; Rechtschaffen, A. The EEG and Behavioral Continuum of the Crocodilian, Caiman Sclerops. Electroencephalogr. Clin. Neurophysiol. 1973, 34, 521–538. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.S.; Tobler, I. Animal Sleep: A Review of Sleep Duration across Phylogeny. Neurosci. Biobehav. Rev. 1984, 8, 269–300. [Google Scholar] [CrossRef]
- Ruckebusch, Y. The Relevance of Drowsiness in the Circadian Cycle of Farm Animals. Anim. Behav. 1972, 20, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Ruckebusch, Y. The Hypnogram as an Index of Adaptation of Farm Animals to Changes in Their Environment. Appl. Anim. Ethol. 1975, 2, 3–18. [Google Scholar] [CrossRef]
- Dallaire, A.; Ruckebusch, Y. Sleep and Wakefulness in the Housed Pony under Different Dietary Conditions. Can. J. Comp. Med. 1974, 38, 65–71. [Google Scholar] [PubMed]
- Tobler, I. Behavioral Sleep in the Asian Elephant in Captivity. Sleep 1992, 15, 1–12. [Google Scholar]
- Rattenborg, N.C.; Amlaner, C.J.; Lima, S.L. Behavioral, Neurophysiological and Evolutionary Perspectives on Unihemispheric Sleep. Neurosci. Biobehav. Rev. 2000, 24, 817–842. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, M.S.; Lesku, J.A.; Libourel, P.-A.; Schmidt, M.H.; Rattenborg, N.C. What Is REM Sleep? Curr. Biol. 2020, 30, R38–R49. [Google Scholar] [CrossRef]
- Amlaner, C.J.; Ball, N.J. Avian Sleep. In Principles and Practice of Sleep Medicine; Saunders Press: London, UK, 1994; pp. 81–94. [Google Scholar]
- Lesku, J.A.; Meyer, L.C.R.; Fuller, A.; Maloney, S.K.; Dell’Omo, G.; Vyssotski, A.L.; Rattenborg, N.C. Ostriches Sleep like Platypuses. PLoS ONE 2011, 6, e23203. [Google Scholar] [CrossRef]
- Rattenborg, N.C.; Van Der Meij, J.; Beckers, G.J.L.; Lesku, J.A. Local Aspects of Avian Non-REM and REM Sleep. Front. Neurosci. 2019, 13, 567. [Google Scholar] [CrossRef] [PubMed]
- Mukhametov, L.M.; Supin, A.Y.; Polyakova, I.G. Interhemispheric Asymmetry of the Electroencephalographic Sleep Patterns in Dolphins. Brain Res. 1977, 134, 581–584. [Google Scholar] [CrossRef]
- Szymczak, J.T.; Kaiser, W.; Helb, H.W.; Beszczyńska, B. A Study of Sleep in the European Blackbird. Physiol. Behav. 1996, 60, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Lyamin, O.I.; Mukhametov, L.M.; Siegel, J.M. Relationship between Sleep and Eye State in Cetaceans and Pinnipeds. Arch. Ital. Biol. 2004, 142, 557–568. [Google Scholar]
- Lyamin, O.I.; Manger, P.R.; Ridgway, S.H.; Mukhametov, L.M.; Siegel, J.M. Cetacean Sleep: An Unusual Form of Mammalian Sleep. Neurosci. Biobehav. Rev. 2008, 32, 1451–1484. [Google Scholar] [CrossRef]
- Fuchs, T.; Maury, D.; Moore, F.R.; Bingman, V.P. Daytime Micro-Naps in a Nocturnal Migrant: An EEG Analysis. Biol. Lett. 2009, 5, 77–80. [Google Scholar] [CrossRef]
- Putyora, E.; Brocklehurst, S.; Tuyttens, F.; Sandilands, V. The Effects of Mild Disturbances on Sleep Behaviour in Laying Hens. Animals 2023, 13, 1251. [Google Scholar] [CrossRef]
- Gjorgovska, N.; Filev, K.; Levkov, V.; Kostadinov, T.; Jusufi, E. Egg Production during the Artificial Moulting and a New Laying Cycle of Aged Hens. Arch. Zootech. 2012, 15, 53–60. [Google Scholar]
- Santos, I.C.L.; Teixeira, R.S.C.; Lopes, E.S.; Albuquerque, A.H.; Rocha e Silva, R.C.; Salles, R.P.R.; Machado, D.N.; Horn, R.V.; Bezerra, W.G.A.; Buxade, C.C.; et al. Experimental Infection of Salmonella Enteritidis in Quails Submitted to Forced Molting by Feed Fasting. Braz. J. Poult. Sci. 2015, 17, 31–36. [Google Scholar] [CrossRef]
- Benington, J.H.; Heller, C.H. Restoration of Brain Energy Metabolism as the Function of Sleep. Prog. Neurobiol. 1995, 45, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Borbély, A.A. Sleep in the Rat during Food Deprivation and Subsequent Restitution of Food. Brain Res. 1977, 124, 457–471. [Google Scholar] [CrossRef]
- Jacobs, B.L.; McGinty, D.J. Effects of Food Deprivation on Sleep and Wakefulness in the Rat. Exp. Neurol. 1971, 30, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Obal JR, F. Thermoregulation during Sleep. Exp. Brain Res. 1984, 8, 157–172. [Google Scholar]
- Cerri, M.; Luppi, M.; Tupone, D.; Zamboni, G.; Amici, R. REM Sleep and Endothermy: Potential Sites and Mechanism of a Reciprocal Interference. Front. Physiol. 2017, 8, 624. [Google Scholar] [CrossRef]
- Horne, J.A. REM Sleep—By Default? Neurosci. Biobehav. Rev. 2000, 24, 777–797. [Google Scholar] [CrossRef]
- Wang, G.; Ekstrand, C.; Svedberg, J. Wet Litter and Perches as Risk Factors for the Development of Foot Pad Dermatitis in Floor-Housed Hens. Br. Poult. Sci. 1998, 39, 191–197. [Google Scholar] [CrossRef]
- Lay, D.C.; Fulton, R.M.; Hester, P.Y.; Karcher, D.M.; Kjaer, J.B.; Mench, J.A.; Mullens, B.A.; Newberry, R.C.; Nicol, C.J.; O’Sullivan, N.P.; et al. Hen Welfare in Different Housing Systems. Poult. Sci. 2011, 90, 278–294. [Google Scholar] [CrossRef]
- Blatchford, R.A.; Fulton, R.M.; Mench, J.A. The Utilization of the Welfare Quality® Assessment for Determining Laying Hen Condition across Three Housing Systems. Poult. Sci. 2016, 95, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Palma, B.D.; Suchecki, D.; Tufik, S. Differential Effects of Acute Cold and Footshock on the Sleep of Rats. Brain Res. 2000, 861, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Finan, P.H.; Goodin, B.R.; Smith, M.T. The Association of Sleep and Pain: An Update and a Path Forward. J. Pain 2013, 14, 1539–1552. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing. 2022. Available online: https://www.r-project.org/ (accessed on 10 August 2023).
- Lenth, R. Emmeans: Estimated Marginal Means, Aka Least-Squares Means. 2023. Available online: chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://cran.r-project.org/web/packages/emmeans/emmeans.pdf (accessed on 10 August 2023).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using Lme4; R Core Team: Vienna, Austria, 2015. [Google Scholar]
- Bates, D.; Maechler, M.; Jagan, M. Matrix: Sparse and Dense Matrix Classes and Methods; R Core Team: Vienna, Austria, 2023. [Google Scholar]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. LmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Lüdecke, D.; Ben-Shachar, M.; Patil, I.; Makowski, D. Extracting, Computing and Exploring the Parameters of Statistical Models Using R. J. Open Source Softw. 2020, 5, 2445. [Google Scholar] [CrossRef]
- Lüdecke, D.; Ben-Shachar, M.; Patil, I.; Waggoner, P.; Makowski, D. Performance: An R Package for Assessment, Comparison and Testing of Statistical Models. J. Open Source Softw. 2021, 6, 3139. [Google Scholar] [CrossRef]
- Jones, S.G.; Vyazovskiy, V.V.; Cirelli, C.; Tononi, G.; Benca, R.M. Homeostatic Regulation of Sleep in the White-Crowned Sparrow (Zonotrichia leucophrys gambelii). BMC Neurosci. 2008, 9, 47. [Google Scholar] [CrossRef]
- Berger, R.J.; Phillips, N.H. Constant Light Suppresses Sleep and Circadian Rhythms in Pigeons without Consequent Sleep Rebound in Darkness. Am. J. Physiol.—Regul. Integr. Comp. Physiol. 1994, 267, R945–R952. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, D.; Lesku, J.A.; Rattenborg, N.C. Increased EEG Spectral Power Density during Sleep Following Short-Term Sleep Deprivation in Pigeons (Columba Livia): Evidence for Avian Sleep Homeostasis. J. Sleep Res. 2008, 17, 140–153. [Google Scholar] [CrossRef]
- Rattenborg, N.C.; Mandt, B.H.; Obermeyer, W.H.; Winsauer, P.J.; Huber, R.; Wikelski, M.; Benca, R.M. Migratory Sleeplessness in the White-Crowned Sparrow (Zonotrichia leucophrys gambelii). PLoS Biol. 2004, 2, e212. [Google Scholar] [CrossRef] [PubMed]
- Borbély, A.A.; Achermann, P. Sleep Homeostasis and Models of Sleep Regulation. In Principles and Practice of Sleep Medicine; Elsevier: Philadelphia, PA, USA, 2005; pp. 405–417. [Google Scholar]
- Canavan, S.V.; Margoliash, D. Budgerigars Have Complex Sleep Structure Similar to That of Mammals. PLoS Biol. 2020, 18, e3000929. [Google Scholar] [CrossRef] [PubMed]
- Connelly, F.; Johnsson, R.D.; Aulsebrook, A.E.; Mulder, R.A.; Hall, M.L.; Vyssotski, A.L.; Lesku, J.A. Urban Noise Restricts, Fragments, and Lightens Sleep in Australian Magpies. Environ. Pollut. 2020, 267, 115484. [Google Scholar] [CrossRef] [PubMed]
- Johnsson, R.D.; Connelly, F.; Vyssotski, A.L.; Roth II, T.C.; Lesku, J.A. Homeostatic Regulation of NREM Sleep, but Not REM Sleep, in Australian Magpies. Sleep 2021, 45, zsab218. [Google Scholar] [CrossRef] [PubMed]
- Low, P.S.; Shank, S.S.; Sejnowski, T.J.; Margoliash, D. Mammalian-like Features of Sleep Structure in Zebra Finches. Proc. Natl. Acad. Sci. USA 2008, 105, 9081–9086. [Google Scholar] [CrossRef]
- Ephron, H.S.; Carrington, P. Rapid Eye Movement Sleep and Cortical Homeostasis. Physiol. Rev. 1966, 73, 500–526. [Google Scholar] [CrossRef]
- Aulsebrook, A.E.; Connelly, F.; Johnsson, R.D.; Jones, T.M.; Mulder, R.A.; Hall, M.L.; Vyssotski, A.L.; Lesku, J.A. White and Amber Light at Night Disrupt Sleep Physiology in Birds. Curr. Biol. 2020, 30, 3657–3663. [Google Scholar] [CrossRef]
- Foote, S.L. Compensatory Changes in REM Sleep Time of Cats during Ad Libitum Sleep and Following Brief REM Sleep Deprivation. Brain Res. 1973, 54, 261–276. [Google Scholar] [CrossRef]
- Lesku, J.A.; Bark, R.J.; Martinez-Gonzalez, D.; Rattenborg, N.C.; Amlaner, C.J.; Lima, S.L. Predator-Induced Plasticity in Sleep Architecture in Wild-Caught Norway Rats (Rattus Norvegicus). Behav. Brain Res. 2008, 189, 298–305. [Google Scholar] [CrossRef]
- Borbely, A.A.; Tobler, I. Endogenous Sleep-Promoting Substances and Sleep Regulation. Physiol. Rev. 1989, 69, 605–670. [Google Scholar] [CrossRef]
- Parmeggiani, P.L.; Rabini, C. Sleep and Environmental Temperature. Arch. Ital. Biol. 1970, 108, 369–381. [Google Scholar] [PubMed]
- Parmeggiani, P.L. Influence of the Temperature Signal on Sleep in Mammals. Biol. Signals Recept. 2000, 9, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Nicol, S.C.; Andersen, N.A.; Phillips, N.H.; Berger, R.J. The Echidna Manifests Typical Characteristics of Rapid Eye Movement Sleep. Neurosci. Lett. 2000, 283, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Downs, C.T.; Awuah, A.; Jordaan, M.; Magagula, L.; Mkhize, T.; Paine, C.; Raymond-Bourret, E.; Hart, L.A. Too Hot to Sleep? Sleep Behaviour and Surface Body Temperature of Wahlberg’s Epauletted Fruit Bat. PLoS ONE 2015, 10, e0119419. [Google Scholar] [CrossRef] [PubMed]
- Opp, M.R.; Ball, N.J.; Miller, D.E.; Amlaner, C.J. Thermoregulation and sleep: Effects of thermal stress on sleep patterns of glaucous-winged gulls (Larus glaucescens). J. Therm. Biol. 1987, 12, 199–202. [Google Scholar] [CrossRef]
- Stuber, E.F.; Dingemanse, N.J.; Mueller, J.C. Temperature Affects Frequency but Not Rhythmicity of Nocturnal Awakenings in Free-Living Great Tits, Parus Major. Anim. Behav. 2017, 128, 135–141. [Google Scholar] [CrossRef]
- Sakaguchi, S.; Glotzbach, S.F.; Heller, H.C. Influence of Hypothalamic and Ambient Temperatures on Sleep in Kangaroo Rats. Am. J. Physiol. 1979, 237, 80–88. [Google Scholar] [CrossRef]
- Vitali, A.; Segnalini, M.; Bertocchi, L.; Bernabucci, U.; Nordone, A.; Lacetera, N. Seasonal Pattern of Mortality and Relationships between Mortality and Temperature-Humidity Index in Dairy Cows. J. Dairy Sci. 2009, 92, 3781–3790. [Google Scholar] [CrossRef]
- Mitchell, M.A.; Kettlewell, P.J. Welfare of Poultry during Transport—A Review. Poult. Welf. Symp. 2009, 1, 90–100. [Google Scholar]
- Najafi, P.; Zulkifli, I.; Soleimani, A.F.; Goh, Y.M. Acute Phase Proteins Response to Feed Deprivation in Broiler Chickens. Poult. Sci. 2016, 95, 760–763. [Google Scholar] [CrossRef]
- Astheimer, L.B.; Buttemer, W.A.; Wingfield, J.C. Interactions of Corticosterone with Feeding, Activity and Metabolism in Passerine Birds. Ornis Scand. 1992, 23, 355–365. [Google Scholar] [CrossRef]
- De Jong, I.C.; Van Voorst, S.; Ehlhardt, D.A.; Blokhuis, H.J. Effects of Restricted Feeding on Physiological Stress Parameters in Growing Broiler Breeders. Br. Poult. Sci. 2002, 43, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Heerkens, J.L.T.; Delezie, E.; Ampe, B.; Rodenburg, T.B.; Tuyttens, F.A.M. Ramps and Hybrid Effects on Keel Bone and Foot Pad Disorders in Modified Aviaries for Laying Hens. Poult. Sci. 2016, 95, 2479–2488. [Google Scholar] [CrossRef] [PubMed]
- Mendl, M.; Burman, O.H.P.; Paul, E.S. An Integrative and Functional Framework for the Study of Animal Emotion and Mood. Proc. R. Soc. B Biol. Sci. 2010, 277, 2895–2904. [Google Scholar] [CrossRef] [PubMed]
- Sneddon, L.U.; Elwood, R.W.; Adamo, S.A.; Leach, M.C. Defining and Assessing Animal Pain. Anim. Behav. 2014, 97, 201–212. [Google Scholar] [CrossRef]
- Blokhuis, H.J. Rest in Poultry. Appl. Anim. Behav. Sci. 1984, 12, 289–303. [Google Scholar] [CrossRef]
- Alvino, G.M.; Archer, G.S.; Mench, J.A. Behavioural Time Budgets of Broiler Chickens Reared in Varying Light Intensities. Appl. Anim. Behav. Sci. 2009, 118, 54–61. [Google Scholar] [CrossRef]
- Karmanova, I.G.; Churnosov, E.V. Electrophysiology Studies on Natural Sleep and Wakefulness in Turtles and Hens. Zh. Evol. Biokhim. Fiziol. 1972, 8, 59–66. [Google Scholar]
- Bäckman, J.; Andersson, A.; Pedersen, L.; Sjöberg, S.; Tøttrup, A.P.; Alerstam, T. Actogram Analysis of Free-Flying Migratory Birds: New Perspectives Based on Acceleration Logging. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 2017, 203, 543–564. [Google Scholar] [CrossRef]
- Bryant, P.A.; Trinder, J.; Curtis, N. Sick and Tired: Does Sleep Have a Vital Role in the Immune System? Nat. Rev. Immunol. 2004, 4, 457–467. [Google Scholar] [CrossRef]
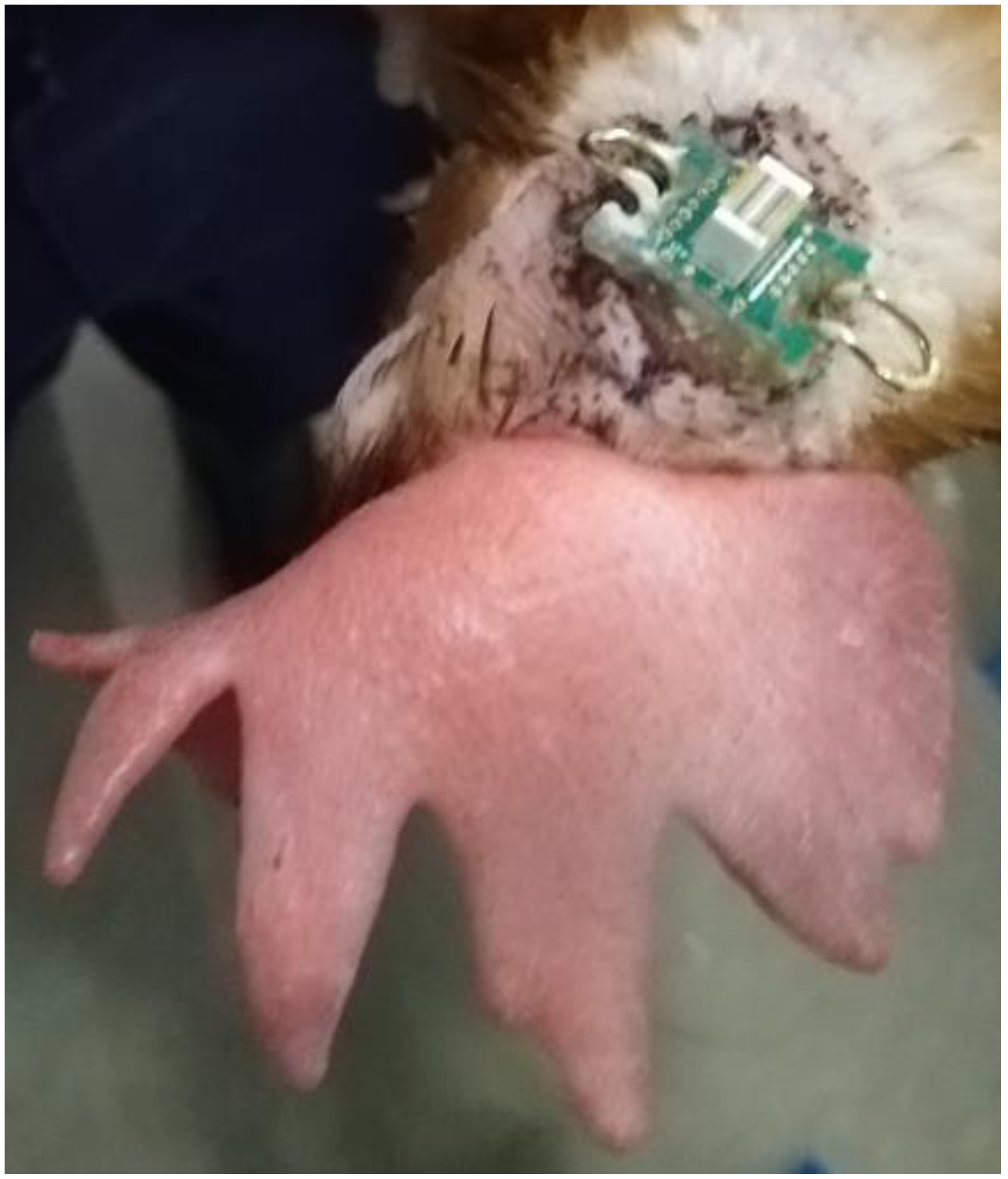

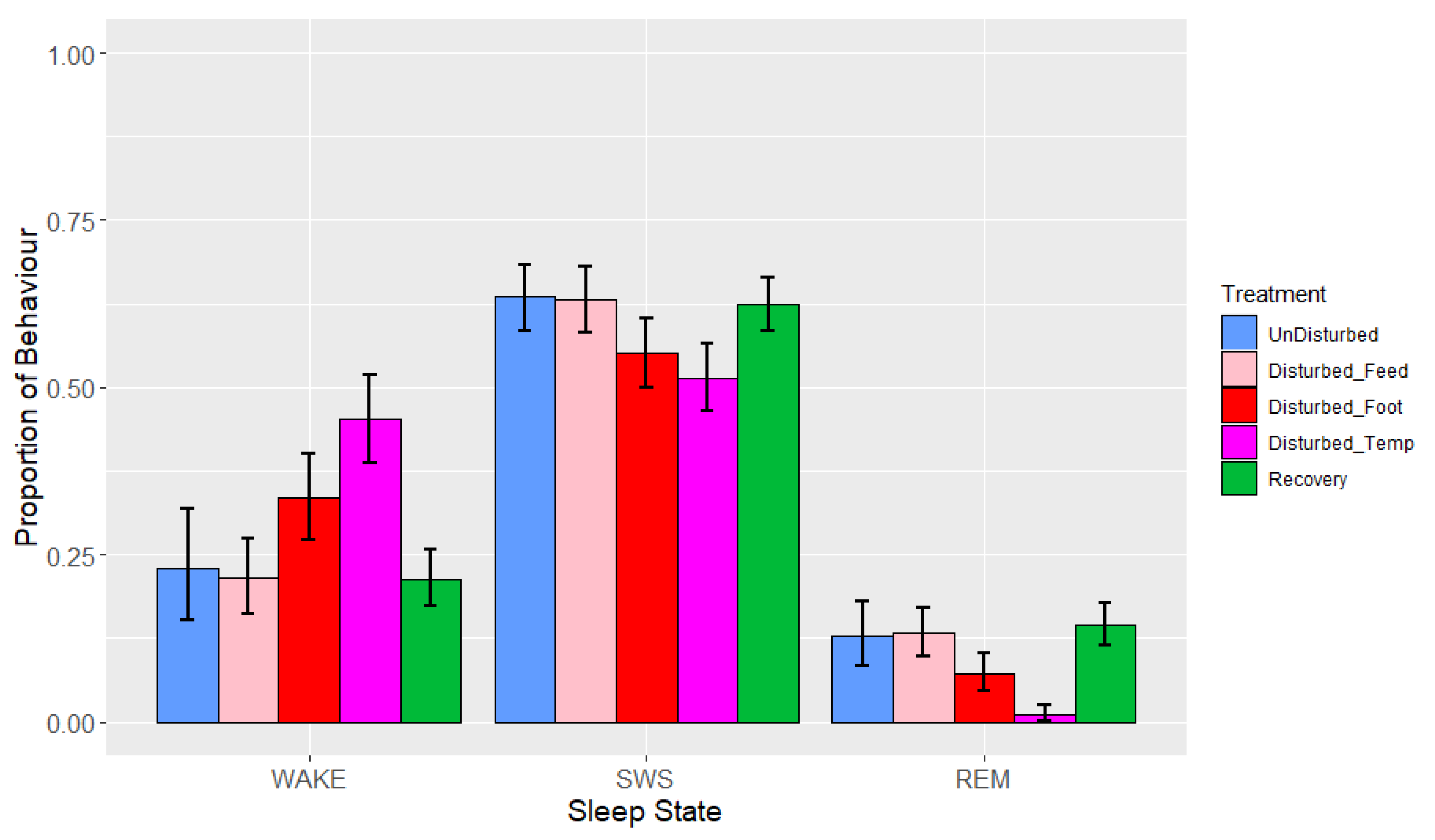
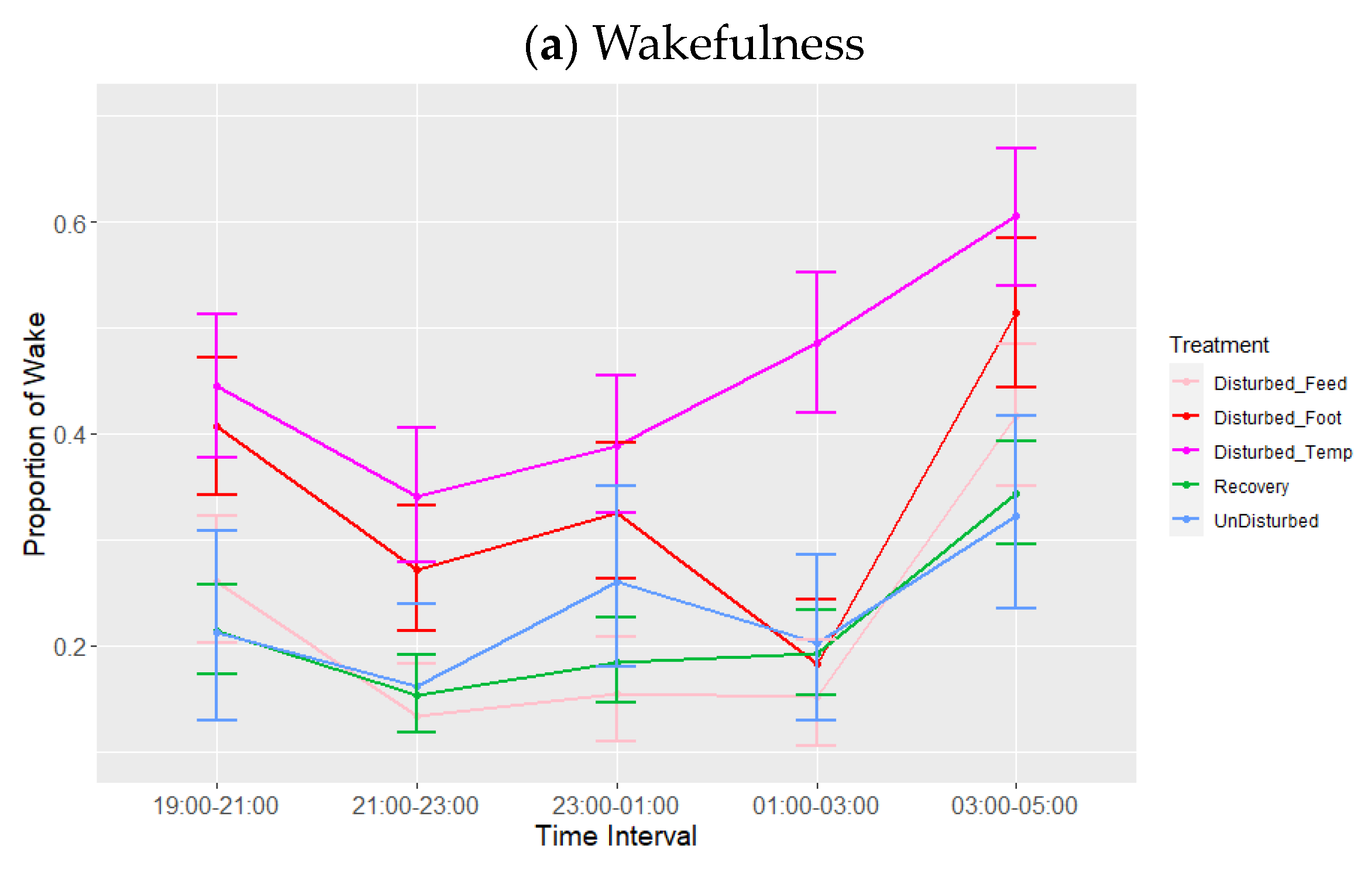
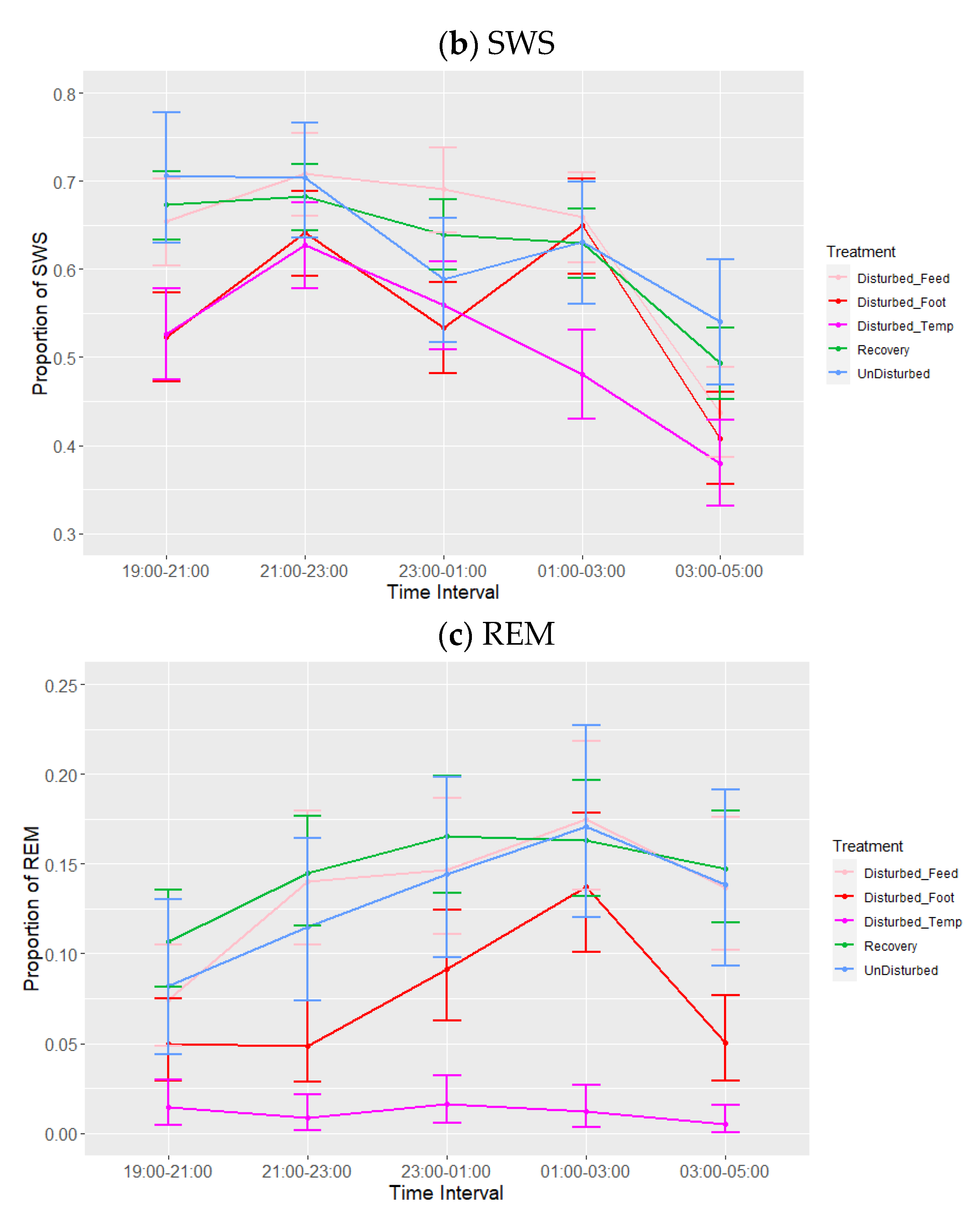



| Batch | Companion Bird ID Numbers | Experimental Bird ID Numbers |
|---|---|---|
| 1 | C1, C2 | E1, E2, E3 |
| 2 | C3, C4 | E4, |
| 3 | C3, C4 | E7, E8, E9 |
| 4 | C5, C6 | E10, E11, E12 |
| Day | Batch 1 and 4 | Batch 2 and 3 |
|---|---|---|
| Day −21 | Surgery | Surgery |
| Day −20–0 | Study 1 | Study 1 |
| Day 1 | Undisturbed sleep recording | Disturbed sleep recording (feed deprivation) |
| Day 2 | Disturbed sleep recording (increased ambient temperature) | Recovery sleep recording |
| Day 3 | Recovery sleep recording | Disturbed sleep recording (increased ambient temperature) |
| Day 4 | Disturbed sleep recording (feed deprivation) | Recovery sleep recording |
| Day 5 | Recovery sleep recording | Disturbed sleep recording (footpad pain) |
| Day 6 | Disturbed sleep recording (footpad pain) | Recovery sleep recording |
| Day 7 | Equipment switched off | Equipment switched off |
| Video | EEG | Definition |
|---|---|---|
| Awake | Waking EEG (low-amplitude high-frequency waves) | Bird is clearly awake and engaged in activity, including walking, preening, nest building, laying, feeding, drinking, foraging, panting and standing on one leg. |
| Resting | Waking EEG (low-amplitude high-frequency waves) | Minor and infrequent head movements while in a stereotypic sleep posture (sitting with wings folded or head retracted into the breast), and not engaged in any active behaviours. One or both eyes may be closed with occasional opening. EEG is the same as awake EEG. |
| Sleep | Slow-wave sleep (SWS) EEG (high-amplitude low-frequency waves) | Bird is in a stereotypic sleep posture (sitting or perching with wings folded and head retracted) with one or both eyes closed. EEG has transitioned from waking/resting to SWS. |
| Sleep | Rapid eye movement (REM) sleep EEG (low-amplitude high-frequency waves—must be preceded by SWS) | Bird is in a stereotypic REM sleep posture (sitting or perching with wings relaxed and head hanging downwards) with both eyes closed. EEG has transitioned from SWS to REM sleep. |
| Wakefulness | SWS | REM | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | Period | Treatment | Time × Period | Time × Treatment | Time | Period | Treatment | Time × Period | Time × Treatment | Time | Period | Treatment | Time × Period | Time × Treatment | |
| ndf,ddf | 4,210 | 2,19 | 2,17 | 8,209 | 8,209 | 4,212 | 2,18 | 2,16 | 8,211 | 8,211 | 4,209 | 2,17 | 2,15 | 8,208 | 8,208 |
| F value | 23.2 | 7.6 | 9.2 | 0.6 | 1.6 | 28.8 | 4.1 | 5.3 | 1.0 | 1.5 | 7.2 | 16.7 | 20.5 | 0.4 | 1.8 |
| p value | <0.001 | 0.004 | 0.002 | 0.768 | 0.127 | <0.001 | 0.035 | 0.017 | 0.475 | 0.177 | <0.001 | <0.001 | <0.001 | 0.915 | 0.070 |
| Wakefulness | SWS | REM | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Proportion | Lower Bound | Upper Bound | Proportion | Lower Bound | Upper Bound | Proportion | Lower Bound | Upper Bound | |
| 19:00–21:00 | 0.26 b | 0.22 | 0.31 | 0.65 a | 0.61 | 0.69 | 0.08 b | 0.05 | 0.10 |
| 21:00–23:00 | 0.18 b | 0.15 | 0.22 | 0.68 a | 0.64 | 0.72 | 0.10 b | 0.08 | 0.13 |
| 23:00–01:00 | 0.24 b | 0.20 | 0.28 | 0.61 a | 0.57 | 0.64 | 0.12 a | 0.10 | 0.15 |
| 01:00–03:00 | 0.22 b | 0.18 | 0.26 | 0.62 a | 0.58 | 0.66 | 0.14 a | 0.11 | 0.17 |
| 03:00–05:00 | 0.39 a | 0.35 | 0.44 | 0.48 b | 0.44 | 0.52 | 0.11 b | 0.08 | 0.13 |
| Wakefulness | SWS | Resting | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | Period | Treatment | Time × Period | Time × Treatment | Time | Period | Treatment | Time × Period | Time × Treatment | Time | Period | Treatment | Time × Period | Time × Treatment | |
| ndf,ddf | 2,42 | 1,42 | 2,42 | 2,42 | 4,42 | 2,31 | 1,42 | 2,38 | 2,27 | 4,38 | 2,39 | 1,40 | 2,39 | 2,40 | 4,40 |
| F value | 5.7 | 0.8 | 9.6 | 0.1 | 2.3 | 7.1 | 0.1 | 13.7 | 0.6 | 0.7 | 3.9 | 0.9 | 5.5 | 0.3 | 2.3 |
| p value | 0.007 | 0.385 | <0.001 | 0.929 | 0.071 | 0.003 | 0.786 | <0.001 | 0.540 | 0.586 | 0.028 | 0.336 | 0.008 | 0.777 | 0.075 |
| Wakefulness | SWS | Resting | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Proportion | Lower Bound | Upper Bound | Proportion | Lower Bound | Upper Bound | Proportion | Lower Bound | Upper Bound | |
| 12:00–15:30 | 0.83 a | 0.81 | 0.86 | 0.03 b | 0.02 | 0.04 | 0.13 a | 0.11 | 0.15 |
| 15:30–19:00 | 0.89 a | 0.86 | 0.91 | 0.01 a | 0.00 | 0.01 | 0.10 a | 0.08 | 0.14 |
| 05:00–08:30 | 0.83 a | 0.81 | 0.85 | 0.01 a | 0.01 | 0.02 | 0.14 a | 0.12 | 0.17 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Putyora, E.; Brocklehurst, S.; Sandilands, V. The Effects of Commercially-Relevant Disturbances on Sleep Behaviour in Laying Hens. Animals 2023, 13, 3105. https://doi.org/10.3390/ani13193105
Putyora E, Brocklehurst S, Sandilands V. The Effects of Commercially-Relevant Disturbances on Sleep Behaviour in Laying Hens. Animals. 2023; 13(19):3105. https://doi.org/10.3390/ani13193105
Chicago/Turabian StylePutyora, Endre, Sarah Brocklehurst, and Victoria Sandilands. 2023. "The Effects of Commercially-Relevant Disturbances on Sleep Behaviour in Laying Hens" Animals 13, no. 19: 3105. https://doi.org/10.3390/ani13193105
APA StylePutyora, E., Brocklehurst, S., & Sandilands, V. (2023). The Effects of Commercially-Relevant Disturbances on Sleep Behaviour in Laying Hens. Animals, 13(19), 3105. https://doi.org/10.3390/ani13193105






