First Assessment of Micro-Litter Ingested by Dolphins, Sea Turtles and Monk Seals Found Stranded along the Coasts of Samos Island, Greece
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sample Collection
2.3. Sample Preparation
2.4. Filtration
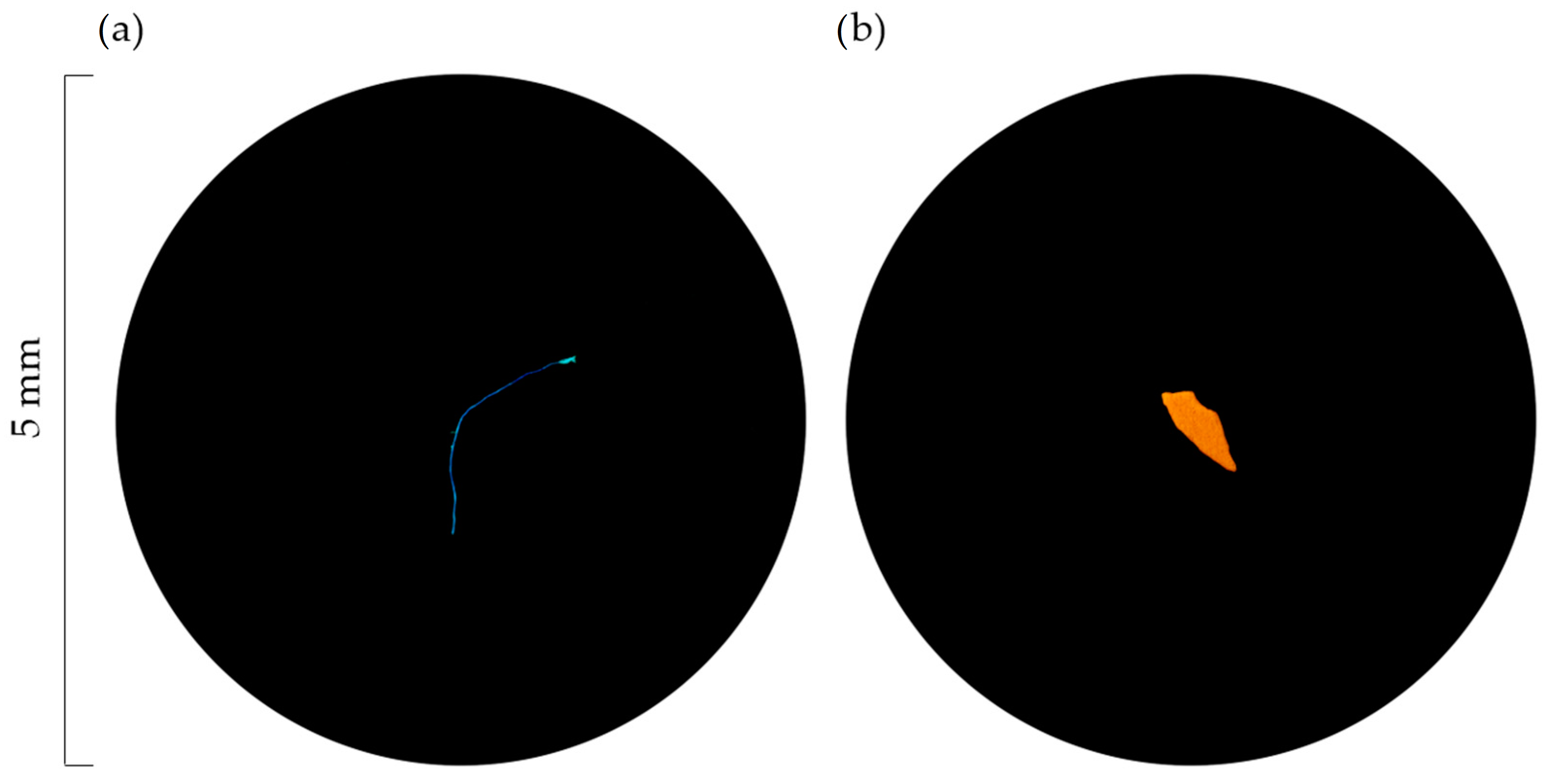
2.5. Particle Identification and Categorisation
2.6. Reagent and Laboratory Contamination
2.7. Data Analysis
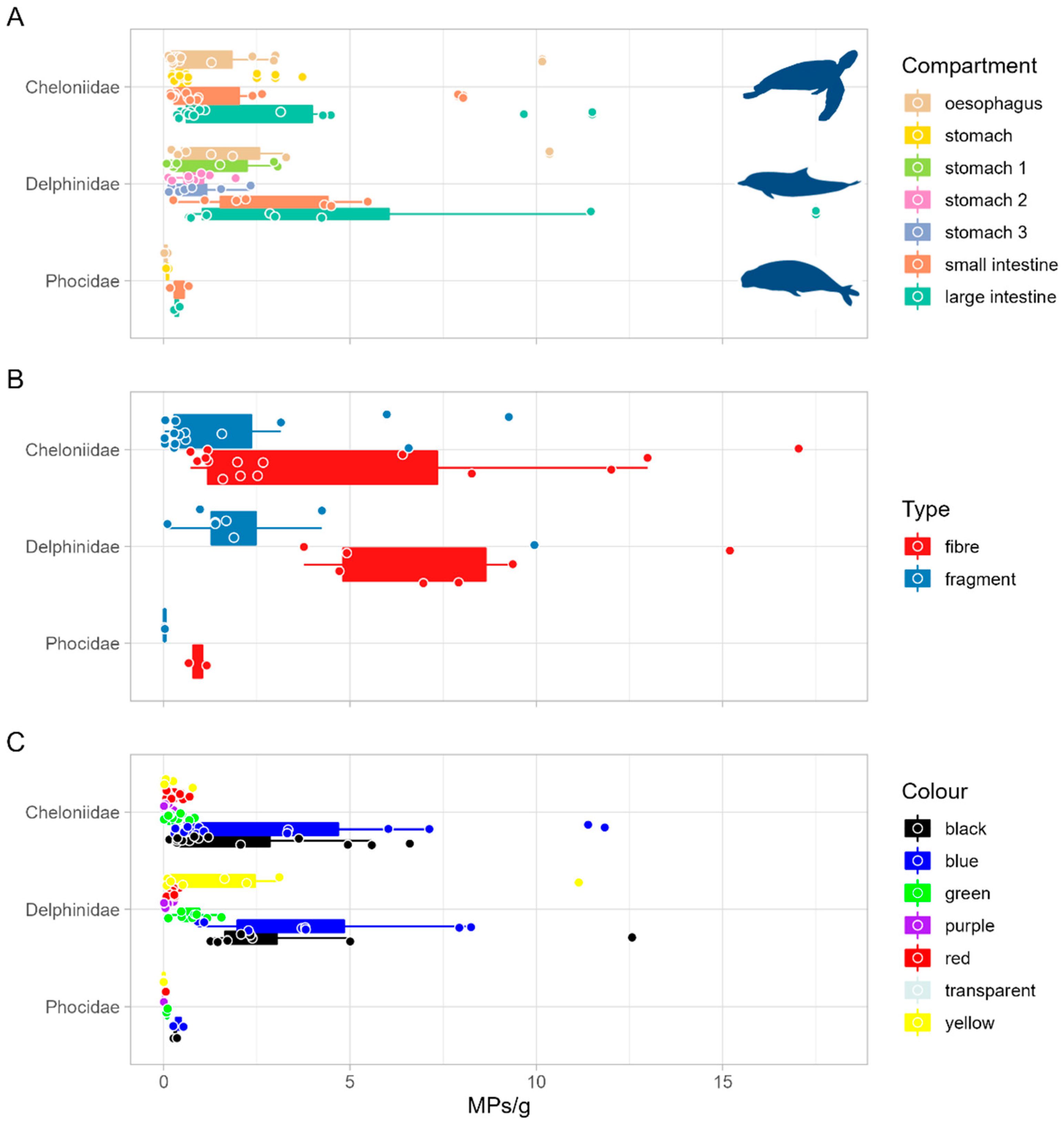
3. Results
Reagent and Laboratory Contamination
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moore, C.J. Synthetic Polymers in the Marine Environment: A Rapidly Increasing, Long-Term Threat. Environ. Res. 2008, 108, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.K. Remote Islands Reveal Rapid Rise of Southern Hemisphere Sea Debris. Sci. World J. 2005, 5, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and Fragmentation of Plastic Debris in Global Environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef]
- Pham, C.K.; Ramirez-Llodra, E.; Alt, C.H.S.; Amaro, T.; Bergmann, M.; Canals, M.; Company, J.B.; Davies, J.; Duineveld, G.; Galgani, F.; et al. Marine Litter Distribution and Density in European Seas, from the Shelves to Deep Basins. PLoS ONE 2014, 9, e95839. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.C.; Moore, C.J.; Saal, F.S.V.; Swan, S.H. Plastics, the Environment and Human Health: Current Consensus and Future Trends. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2153–2166. [Google Scholar] [CrossRef] [PubMed]
- Woodall, L.C.; Sanchez-Vidal, A.; Canals, M.; Paterson, G.L.J.; Coppock, R.; Sleight, V.; Calafat, A.; Rogers, A.D.; Narayanaswamy, B.E.; Thompson, R.C. The Deep Sea Is a Major Sink for Microplastic Debris. R. Soc. Open Sci. 2014, 1, 140317. [Google Scholar] [CrossRef] [PubMed]
- Galgani, F.; Hanke, G.; Maes, T. Global Distribution, Composition and Abundance of Marine Litter. In Marine Anthropogenic Litter; Springer: Cham, Switzerland, 2015; pp. 29–56. [Google Scholar]
- De Ruijter, V.N.; Milou, A.; Costa, V. Assessment of Microplastics Distribution and Stratification in the Shallow Marine Sediments of Samos Island, Eastern Mediterranean Sea, Greece. Mediterr. Mar. Sci. 2019, 20, 736–744. [Google Scholar] [CrossRef]
- Unger, B.; Rebolledo, E.L.B.; Deaville, R.; Gröne, A.; IJsseldijk, L.L.; Leopold, M.F.; Siebert, U.; Spitz, J.; Wohlsein, P.; Herr, H. Large Amounts of Marine Debris Found in Sperm Whales Stranded along the North Sea Coast in Early 2016. Mar. Pollut. Bull. 2016, 112, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Kraus, S.D. Entanglement of Whales in Fishing Gear. In Encyclopedia of Marine Mammals; Elsevier: Amsterdam, The Netherlands, 2018; p. 336. [Google Scholar]
- Kahane-Rapport, S.R.; Czapanskiy, M.F.; Fahlbusch, J.A.; Friedlaender, A.S.; Calambokidis, J.; Hazen, E.L.; Goldbogen, J.A.; Savoca, M.S. Field Measurements Reveal Exposure Risk to Microplastic Ingestion by Filter-Feeding Megafauna. Nat. Commun. 2022, 13, 6327. [Google Scholar] [CrossRef]
- Nelms, S.E.; Duncan, E.M.; Broderick, A.C.; Galloway, T.S.; Godfrey, M.H.; Hamann, M.; Lindeque, P.K.; Godley, B.J. Plastic and Marine Turtles: A Review and Call for Research. ICES J. Mar. Sci. 2016, 73, 165–181. [Google Scholar] [CrossRef]
- PlasticsEurope. Plastics—The Facts 2015. An Analysis of European Plastics Production, Demand and Waste Data; PlasticEurope: Brussels, Belgium, 2015. [Google Scholar]
- Tsangaris, C.; Panti, C.; Compa, M.; Pedà, C.; Digka, N.; Baini, M.; D’Alessandro, M.; Alomar, C.; Patsiou, D.; Giani, D.; et al. Interlaboratory Comparison of Microplastic Extraction Methods from Marine Biota Tissues: A Harmonization Exercise of the Plastic Busters MPAs Project. Mar. Pollut. Bull. 2021, 164, 111992. [Google Scholar] [CrossRef] [PubMed]
- Zantis, L.J.; Carroll, E.L.; Nelms, S.E.; Bosker, T. Marine Mammals and Microplastics: A Systematic Review and Call for Standardisation. Environ. Pollut. 2021, 269, 116142. [Google Scholar] [CrossRef]
- Barnett, S.; Evans, R.; Quintana Martín-Montalvo, B.; Miliou, A.; Pietroluongo, G. An Environmentally Friendly Method for the Identification of Microplastics Using Density Analysis. Environ. Toxicol. Chem. 2021, 40, 3299–3305. [Google Scholar] [CrossRef] [PubMed]
- Avio, C.G.; Gorbi, S.; Milan, M.; Benedetti, M.; Fattorini, D.; D’Errico, G.; Pauletto, M.; Bargelloni, L.; Regoli, F. Pollutants Bioavailability and Toxicological Risk from Microplastics to Marine Mussels. Environ. Pollut. 2015, 198, 211–222. [Google Scholar] [CrossRef]
- Vandermeersch, G.; Van Cauwenberghe, L.; Janssen, C.R.; Marques, A.; Granby, K.; Fait, G.; Kotterman, M.J.J.; Diogène, J.; Bekaert, K.; Robbens, J.; et al. A Critical View on Microplastic Quantification in Aquatic Organisms. Environ. Res. 2015, 143, 46–55. [Google Scholar] [CrossRef]
- Provencher, J.F.; Bond, A.L.; Avery-Gomm, S.; Borrelle, S.B.; Bravo Rebolledo, E.L.; Hammer, S.; Kühn, S.; Lavers, J.L.; Mallory, M.L.; Trevail, A.; et al. Quantifying Ingested Debris in Marine Megafauna: A Review and Recommendations for Standardization. Anal. Methods 2017, 9, 1454–1469. [Google Scholar] [CrossRef]
- Consoli, P.; Scotti, G.; Romeo, T.; Cristina, M.; Esposito, V.; Alessandro, M.D.; Battaglia, P.; Galgani, F.; Figurella, F.; Pragnell-raasch, H.; et al. Characterization of Seafloor Litter on Mediterranean Shallow Coastal Waters: Evidence from Dive Against Debris®, a Citizen Science Monitoring Approach. Mar. Pollut. Bull. 2020, 150, 110763. [Google Scholar] [CrossRef]
- Fossi, M.C.; Coppola, D.; Baini, M.; Giannetti, M.; Guerranti, C.; Marsili, L.; Panti, C.; de Sabata, E.; Clò, S. Large Filter Feeding Marine Organisms as Indicators of Microplastic in the Pelagic Environment: The Case Studies of the Mediterranean Basking Shark (Cetorhinus maximus) and Fin Whale (Balaenoptera physalus). Mar. Environ. Res. 2014, 100, 17–24. [Google Scholar] [CrossRef]
- Lusher, A. Microplastics in the Marine Environment: Distribution, Interactions and Effects. In Marine Anthropogenic Litter; Springer International Publishing: Berlin/Heidelberg, Germany, 2015; pp. 245–307. ISBN 978-3-319-16510-3. [Google Scholar]
- GESAMP. Guidelines for the Monitoring and Assessment of Plastic Litter in the Ocean; United Nations Environment Programme (UNEP): Cambridge, UK, 2019; p. 138. [Google Scholar]
- Sun, X.; Li, Q.; Zhu, M.; Liang, J.; Zheng, S.; Zhao, Y. Ingestion of Microplastics by Natural Zooplankton Groups in the Northern South China Sea. Mar. Pollut. Bull. 2017, 115, 217–224. [Google Scholar] [CrossRef]
- Romeo, T.; Pietro, B.; Pedà, C.; Consoli, P.; Andaloro, F.; Fossi, M.C. First Evidence of Presence of Plastic Debris in Stomach of Large Pelagic Fish in the Mediterranean Sea. Mar. Pollut. Bull. 2015, 95, 358–361. [Google Scholar] [CrossRef]
- Thompson, R.C. Lost at Sea: Where Is All the Plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef] [PubMed]
- Caron, A.G.M.; Thomas, C.R.; Berry, K.L.E.; Motti, C.A.; Ariel, E.; Brodie, J.E. Ingestion of Microplastic Debris by Green Sea Turtles (Chelonia mydas) in the Great Barrier Reef: Validation of a Sequential Extraction Protocol. Mar. Pollut. Bull. 2018, 127, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Philipp, C.; Unger, B.; Fischer, E.K.; Schnitzler, J.G.; Siebert, U. Handle with Care—Microplastic Particles in Intestine Samples of Seals from German Waters. Sustainability 2020, 12, 10424. [Google Scholar] [CrossRef]
- Hernandez-Gonzalez, A.; Saavedra, C.; Gago, J.; Covelo, P.; Santos, M.B.; Pierce, G.J. Microplastics in the Stomach Contents of Common Dolphin (Delphinus Delphis) Stranded on the Galician Coasts (NW Spain, 2005–2010). Mar. Pollut. Bull. 2018, 137, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Lusher, A.L.; Hernandez-Milian, G.; Berrow, S.; Rogan, E.; O’Connor, I. Incidence of Marine Debris in Cetaceans Stranded and Bycaught in Ireland: Recent Findings and a Review of Historical Knowledge. Environ. Pollut. 2018, 232, 467–476. [Google Scholar] [CrossRef]
- Nelms, S.E.; Barnett, J.; Brownlow, A.; Davison, N.J.; Deaville, R.; Galloway, T.S.; Lindeque, P.K.; Santillo, D.; Godley, B.J. Microplastics in Marine Mammals Stranded around the British Coast: Ubiquitous but Transitory? Sci. Rep. 2019, 9, 1075. [Google Scholar] [CrossRef]
- Nelms, S.E.; Galloway, T.S.; Godley, B.J.; Jarvis, D.S.; Lindeque, P.K. Investigating Microplastic Trophic Transfer in Marine Top Predators. Environ. Pollut. 2018, 238, 999–1007. [Google Scholar] [CrossRef]
- Watts, A.J.R.; Urbina, M.A.; Corr, S.; Lewis, C.; Galloway, T.S. Ingestion of Plastic Microfibers by the Crab Carcinus Maenas and Its Effect on Food Consumption and Energy Balance. Environ. Sci. Technol. 2015, 49, 14597–14604. [Google Scholar] [CrossRef]
- Galloway, T.S.; Cole, M.; Lewis, C. Interactions of Microplastic Debris throughout the Marine Ecosystem. Nat. Ecol. Evol. 2017, 1, 1–8. [Google Scholar] [CrossRef]
- Besseling, E.; Wegner, A.; Foekema, E.M.; Van Den Heuvel-Greve, M.J.; Koelmans, A.A. Effects of Microplastic on Fitness and PCB Bioaccumulation by the Lugworm Arenicola marina (L.). Environ. Sci. Technol. 2013, 47, 593–600. [Google Scholar] [CrossRef]
- Rochman, C.M.; Brookson, C.; Bikker, J.; Djuric, N.; Earn, A.; Bucci, K.; Athey, S.; Huntington, A.; McIlwraith, H.; Munno, K.; et al. Rethinking Microplastics as a Diverse Contaminant Suite. Environ. Toxicol. Chem. 2019, 38, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Fossi, M.C.; Panti, C. Sentinel Species of Marine Ecosystems. In Oxford Research Encyclopedia of Environmental Science; Oxford University Press: Oxford, UK, 2017. [Google Scholar]
- Fossi, M.C.; Panti, C.; Baini, M.; Lavers, J.L. A Review of Plastic-Associated Pressures: Cetaceans of the Mediterranean Sea and Eastern Australian Shearwaters as Case Studies. Front. Mar. Sci. 2018, 5, 173. [Google Scholar] [CrossRef]
- Perez-Venegas, D.J.; Toro-Valdivieso, C.; Ayala, F.; Brito, B.; Iturra, L.; Arriagada, M.; Seguel, M.; Barrios, C.; Sepúlveda, M.; Oliva, D.; et al. Monitoring the Occurrence of Microplastic Ingestion in Otariids along the Peruvian and Chilean Coasts. Mar. Pollut. Bull. 2020, 153, 110966. [Google Scholar] [CrossRef] [PubMed]
- Miliou, A.; Höfer, S.; Maridakis, C.; Almeida, M.; Cox, R.; Interior, B.; Edward, P. Assessment of Microplastic Fibre Contamination in the Eastern Aegean Sea, with the Use of Holothurians as Indicator Species. Rapp. Comm. Int. Mer. Medit. 2016, 41, 232. [Google Scholar] [CrossRef]
- Miliou, A.; Mentzel, S.; Almeida, M.; Maridakis, C.; Cox, R. Microplastic Fibre Presence in the Food Chain of Sphyraena Viridensis in the Eastern Aegean Sea, Greece. Rapp. Comm. Int. Mer. Medit. 2016, 41, 222. [Google Scholar]
- Digka, N.; Bray, L.; Tsangaris, C.; Andreanidou, K.; Kasimati, E.; Kofidou, E.; Komnenou, A.; Kaberi, H. Evidence of Ingested Plastics in Stranded Loggerhead Sea Turtles along the Greek Coastline, East Mediterranean Sea. Environ. Pollut. 2020, 263, 114596. [Google Scholar] [CrossRef]
- IUCN-MMPATF. Hellenic Trench IMMA. Full Accounts of Mediterranean IMMA Factsheet; IUCN Joint SSC/WCPA Marine Mammal Protected Areas Task Force: Italy, 2017; Available online: https://www.marinemammalhabitat.org/wp-content/uploads/imma-factsheets/Mediterranean/Hellenic-Trench-Mediterranean.pdf (accessed on 17 November 2022).
- Pietroluongo, G.; Cipriano, G.; Ashok, K.; Antichi, S.; Carlier, H.; Miliou, A.; Maglietta, R.; Fanizza, C.; Carlucci, R. Density and Abundance of Delphinus Delphis in Waters South of Samos Island, Greece (Eastern Mediterranean Sea). J. Mar. Sci. Eng. 2020, 8, 218. [Google Scholar] [CrossRef]
- Pietroluongo, G.; Quintana Martín-Montalvo, B.Q.; Ashok, K.; Miliou, A.; Fosberry, J.; Antichi, S.; Moscatelli, S.; Tsimpidis, T.; Carlucci, R.; Azzolin, M. Combining Monitoring Approaches as a Tool to Assess the Occurrence of the Mediterranean Monk Seal in Samos Island, Greece. Hydrobiology 2022, 1, 440–450. [Google Scholar] [CrossRef]
- Almpanidou, V.; Tsapalou, V.; Chatzimentor, A.; Cardona, L.; Claro, F.; Hostetter, P.; Kaska, Y.; Liu, W.; Mansui, J.; Miliou, A.; et al. Foraging Grounds of Adult Loggerhead Sea Turtles across the Mediterranean Sea: Key Sites and Hotspots of Risk. Biodivers. Conserv. 2022, 31, 143–160. [Google Scholar] [CrossRef]
- Wolke, R.E.; George, A. Sea Turtle Necropsy Manual; NOAA Technical Memorandum NMFS-SEFC-24: La Jolla, CA, USA, 1981.
- Geraci, J.; Lounsbury, V. Marine Mammals Ashore: A Field Guide for Strandings; National Aquarium in Baltimore: Baltimore, MD, USA, 2005. [Google Scholar]
- INDICIT. Monitoring Marine Litter Impacts on Sea Turtles. Protocol for the Collection of Data on Ingestion and Entanglement in the Loggerhead Turtle (Caretta Caretta Linnaeus, 1758). Deliverable D2.6 of the European Project Implementation of the Indicator of Mari; Grant Agreement 11.0661/2016/748064/SUB/ENV.C2; European Commission: Bruxelles, Belgium, 2018; p. 22. [Google Scholar]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the Marine Environment: A Review of the Methods Used for Identification and Quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef]
- De Witte, B.; Devriese, L.; Bekaert, K.; Hoffman, S.; Vandermeersch, G.; Cooreman, K.; Robbens, J. Quality Assessment of the Blue Mussel (Mytilus Edulis): Comparison between Commercial and Wild Types. Mar. Pollut. Bull. 2014, 85, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; PRIMER-E Ltd.: Devon, UK, 2008. [Google Scholar]
- Clarke, K.R. Non-Parametric Multivariate Analyses of Changes in Community Structure. Austral Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Wickham, H.K.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.D.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 43, 1686. [Google Scholar] [CrossRef]
- Dinno, A. Dunn. Test: Dunn’s Test of Multiple Comparisons Using Rank Sums 2017. R Package Version 1.3.5. Available online: https://cran.r-project.org/web/packages/dunn.test (accessed on 17 November 2022).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; Mcglinn, D.; Minchin, P.R.; Hara, R.B.O.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package 2018. R Package Version 2.6-4. Available online: https://cran.r-project.org/web/packages/vegan (accessed on 17 November 2022).
- Dede, A.; Salman, A.; Tonay, A.M. Stomach Contents of By-Caught Striped Dolphins (Stenella coeruleoalba) in the Eastern Mediterranean Sea. J. Mar. Biol. Assoc. UK 2016, 96, 869–875. [Google Scholar] [CrossRef]
- Pierce, G.J.; Hernandez-Milian, G.; Santos, M.B.; Dendrinos, P.; Psaradellis, M.; Tounta, E.; Androukaki, E.; Edridge, A. Diet of the Monk Seal (Monachus monachus) in Greek Waters. Aquat. Mamm. 2011, 37, 284–297. [Google Scholar] [CrossRef]
- Tomas, J.; Aznar, F.J.; Raga, J.A. Feeding Ecology of the Loggerhead Turtle Caretta caretta in the Western Mediterranean. J. Zool. 2001, 255, 525–532. [Google Scholar] [CrossRef]
- Di Beneditto, A.P.M.; Awabdi, D.R. How Marine Debris Ingestion Differs among Megafauna Species in a Tropical Coastal Area. Mar. Pollut. Bull. 2014, 88, 86–90. [Google Scholar] [CrossRef]
- Sanchez-Vidal, A.; Canals, M.; de Haan, W.P.; Romero, J.; Veny, M. Seagrasses Provide a Novel Ecosystem Service by Trapping Marine Plastics. Sci. Rep. 2021, 11, 254. [Google Scholar] [CrossRef] [PubMed]
- Teuten, E.L.; Saquing, J.M.; Knappe, D.R.U.; Barlaz, M.A.; Jonsson, S.; Björn, A.; Rowland, S.J.; Thompson, R.C.; Galloway, T.S.; Yamashita, R.; et al. Transport and Release of Chemicals from Plastics to the Environment and to Wildlife. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2027–2045. [Google Scholar] [CrossRef] [PubMed]
- Egbeocha, C.; Malek, S.; Emenike, C.; Milow, P. Feasting on Microplastics: Ingestion by and Effects on Marine Organisms. Aquat. Biol. 2018, 27, 93–106. [Google Scholar] [CrossRef]
- Battaglia, F.M.; Beckingham, B.A.; McFee, W.E. First Report from North America of Microplastics in the Gastrointestinal Tract of Stranded Bottlenose Dolphins (Tursiops truncatus). Mar. Pollut. Bull. 2020, 160, 111677. [Google Scholar] [CrossRef] [PubMed]
- Duncan, E.M.; Broderick, A.C.; Fuller, W.J.; Galloway, T.S.; Godfrey, M.H.; Hamann, M.; Limpus, C.J.; Lindeque, P.K.; Mayes, A.G.; Omeyer, L.C.M.; et al. Microplastic Ingestion Ubiquitous in Marine Turtles. Glob. Change Biol. 2019, 25, 744–752. [Google Scholar] [CrossRef] [PubMed]
- Salvador Cesa, F.; Turra, A.; Baruque-Ramos, J. Synthetic Fibers as Microplastics in the Marine Environment: A Review from Textile Perspective with a Focus on Domestic Washings. Sci. Total Environ. 2017, 598, 1116–1129. [Google Scholar] [CrossRef]
- Bravo Rebolledo, E.L.; Van Franeker, J.A.; Jansen, O.E.; Brasseur, S.M. Plastic Ingestion by Harbour Seals (Phoca vitulina) in The Netherlands. Mar. Pollut. Bull. 2013, 67, 200–202. [Google Scholar] [CrossRef] [PubMed]
- Besseling, E.; Foekema, E.M.; Franeker, J.A.V.; Leopold, M.F.; Kühn, S.; Rebolledo, E.L.B.; Heße, E.; Mielke, L.; Ijzer, J.; Kamminga, P.; et al. Microplastic in a Macro Filter Feeder: Humpback Whale Megaptera Novaeangliae. Mar. Pollut. Bull. 2015, 95, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.G.; Cole, G.; Spiby, K.; Nel, R.; Osborne, A.; Perold, V. Impacts of Plastic Ingestion on Post-Hatchling Loggerhead Turtles off South Africa. Mar. Pollut. Bull. 2016, 107, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Pham, C.K.; Rodríguez, Y.; Dauphin, A.; Carriço, R.; Frias, J.P.G.L.; Vandeperre, F.; Otero, V.; Santos, M.R.; Martins, H.R.; Bolten, A.B.; et al. Plastic Ingestion in Oceanic-Stage Loggerhead Sea Turtles (Caretta caretta) off the North Atlantic Subtropical Gyre. Mar. Pollut. Bull. 2017, 121, 222–229. [Google Scholar] [CrossRef]
- van Franeker, J.A.; Bravo Rebolledo, E.L.; Hesse, E.; IJsseldijk, L.L.; Kühn, S.; Leopold, M.; Mielke, L. Plastic Ingestion by Harbour Porpoises Phocoena phocoena in the Netherlands: Establishing a Standardised Method. Ambio 2018, 47, 387–397. [Google Scholar] [CrossRef]
- Zhu, J.; Yu, X.; Zhang, Q.; Li, Y.; Tan, S.; Li, D.; Yang, Z.; Wang, J. Cetaceans and Microplastics: First Report of Microplastic Ingestion by a Coastal Delphinid, Sousa chinensis. Sci. Total Environ. 2019, 659, 649–654. [Google Scholar] [CrossRef]
- Novillo, O.; Raga, J.A.; Tomás, J. Evaluating the Presence of Microplastics in Striped Dolphins (Stenella coeruleoalba) Stranded in the Western Mediterranean Sea. Mar. Pollut. Bull. 2020, 160, 111557. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Luo, D.; Yu, R.Q.; Xie, Z.; He, L.; Wu, Y. Microplastics in the Endangered Indo-Pacific Humpback Dolphins (Sousa chinensis) from the Pearl River Estuary, China. Environ. Pollut. 2021, 270, 116057. [Google Scholar] [CrossRef] [PubMed]
- Philipp, C.; Unger, B.; Ehlers, S.M.; Koop, J.H.E.; Siebert, U. First Evidence of Retrospective Findings of Microplastics in Harbour Porpoises (Phocoena phocoena) From German Waters. Front. Mar. Sci. 2021, 8, 508. [Google Scholar] [CrossRef]
- Lusher, A.L.; Hernandez-Milian, G.; O’Brien, J.; Berrow, S.; O’Connor, I.; Officer, R. Microplastic and macroplastic ingestion by a deep diving, oceanic cetacean: The True’s beaked whale Mesoplodon mirus. Environ. Pollut. 2015, 199, 185–191. [Google Scholar]
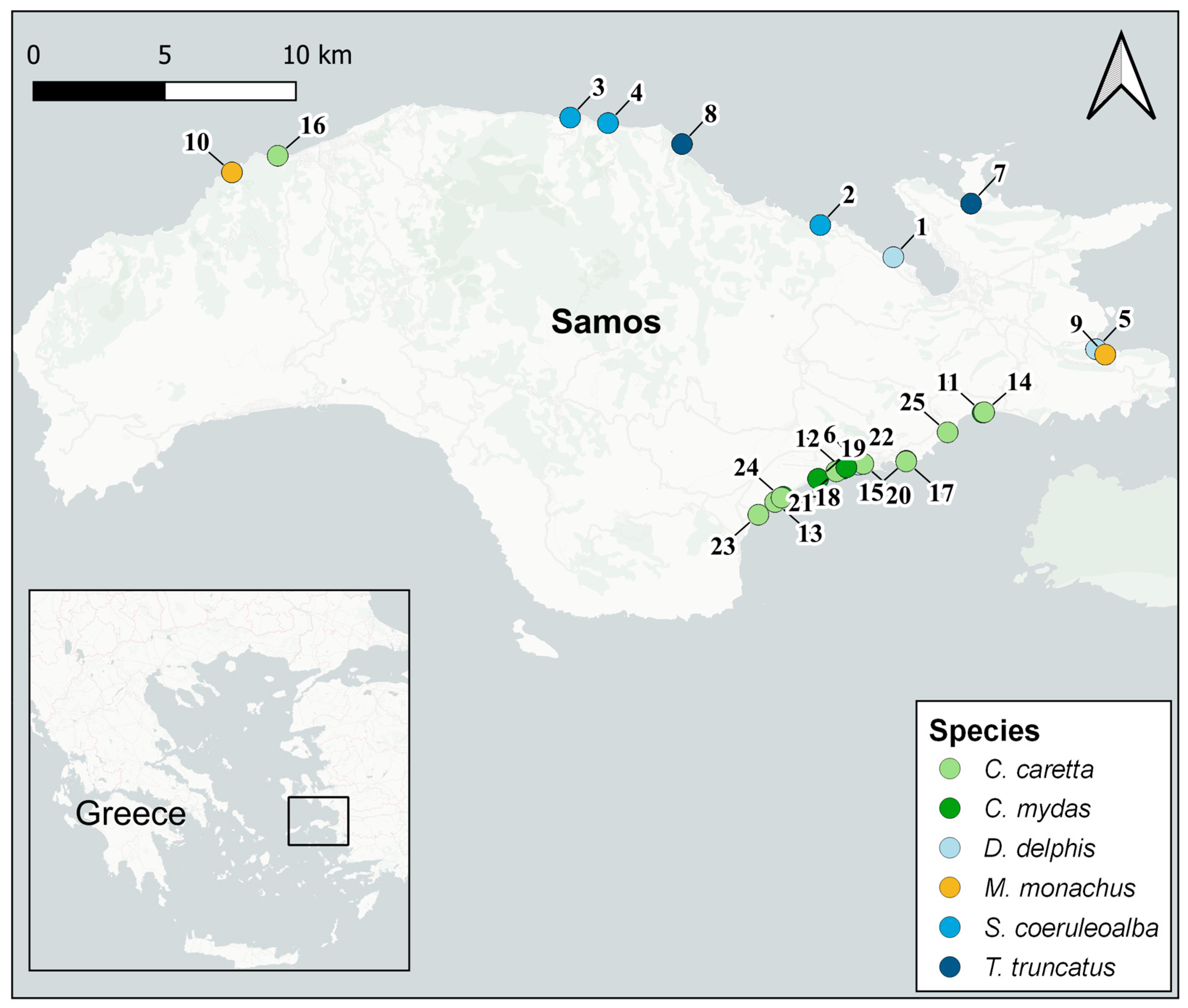

| ID | Stranding Date | Lat. (N) | Lon. (E) | Family | Species | Sex | Age Class | Total Length (cm) | MPs |
|---|---|---|---|---|---|---|---|---|---|
| 11 | 12 February2018 | 37.70749 | 26.98678 | Cheloniidae | Chelonia mydas | F | Adult | - | 133 |
| 12 | 23 February 2018 | 37.68833 | 26.92639 | Cheloniidae | Chelonia mydas | ND | Subadult | 54 | 214 |
| 13 | 08 March 2018 | 37.67701 | 26.89728 | Cheloniidae | Caretta caretta | F | Adult | 110 | 259 |
| 14 | 10 April 2018 | 37.70763 | 26.98737 | Cheloniidae | Caretta caretta | M | Adult | 108 | 237 |
| 15 | 17 April 2018 | 37.69104 | 26.95381 | Cheloniidae | Chelonia mydas | F | Adult | 85 | 292 |
| 16 | 22 April 2018 | 37.79510 | 26.68289 | Cheloniidae | Caretta caretta | F | Adult | 98 | 296 |
| 17 | 07 November 2018 | 37.69085 | 26.95379 | Cheloniidae | Caretta caretta | ND | Juvenile | 39 | 245 |
| 18 | 19 November 2018 | 37.67877 | 26.90080 | Cheloniidae | Chelonia mydas | F | Adult | 97 | 503 |
| 19 | 19 November 2018 | 37.68491 | 26.91581 | Cheloniidae | Chelonia mydas | F | Adult | 84 | 442 |
| 20 | 22 November 2018 | 37.69000 | 26.93533 | Cheloniidae | Caretta caretta | F | Adult | 93 | 165 |
| 21 | 27 November 2018 | 37.68743 | 26.92365 | Cheloniidae | Caretta caretta | M | Adult | - | 140 |
| 22 | 25 January 2019 | 37.68881 | 26.92802 | Cheloniidae | Chelonia mydas | ND | Subadult | 53 | 360 |
| 23 | 30 January 2019 | 37.67265 | 26.89008 | Cheloniidae | Caretta caretta | M | Adult | 96 | 287 |
| 24 | 30 January 2019 | 37.67848 | 26.90008 | Cheloniidae | Caretta caretta | F | Adult | - | 87 |
| 25 | 04 February 2019 | 37.70077 | 26.97165 | Cheloniidae | Caretta caretta | F | Adult | 93 | 253 |
| 1 | 09 February 2019 | 37.76050 | 26.94826 | Delphinidae | Delphinus delphis | F | Subadult | 152 | 592 |
| 2 | 25 February 2019 | 37.77147 | 26.91677 | Delphinidae | Stenella coeruleoalba | F | Juvenile | 163 | 287 |
| 3 | 01 March 2019 | 37.80810 | 26.80897 | Delphinidae | Stenella coeruleoalba | F | Subadult | 196 | 773 |
| 9 | 14 March 2019 | 37.72729 | 27.03958 | Phocidae | Monachus monachus | F | Adult | 205 | 277 |
| 4 | 16 March 2019 | 37.80622 | 26.82526 | Delphinidae | Stenella coeruleoalba | M | Subadult | 180 | 2056 |
| 5 | 24 March 2019 | 37.72913 | 27.03561 | Delphinidae | Delphinus delphis | F | Subadult | - | 448 |
| 6 | 12 April 2019 | 37.68975 | 26.93322 | Delphinidae | Delphinus delphis | M | Subadult | - | 402 |
| 10 | 17 April 2019 | 37.78942 | 26.66318 | Phocidae | Monachus monachus | M | Adult | 260 | 261 |
| 7 | 22 April 2019 | 37.77878 | 26.98176 | Delphinidae | Tursiops truncatus | F | Adult | 280 | 1056 |
| 8 | 04 May 2019 | 37.79908 | 26.85716 | Delphinidae | Tursiops truncatus | M | Juvenile | 180 | 574 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietroluongo, G.; Quintana Martín-Montalvo, B.; Antichi, S.; Miliou, A.; Costa, V. First Assessment of Micro-Litter Ingested by Dolphins, Sea Turtles and Monk Seals Found Stranded along the Coasts of Samos Island, Greece. Animals 2022, 12, 3499. https://doi.org/10.3390/ani12243499
Pietroluongo G, Quintana Martín-Montalvo B, Antichi S, Miliou A, Costa V. First Assessment of Micro-Litter Ingested by Dolphins, Sea Turtles and Monk Seals Found Stranded along the Coasts of Samos Island, Greece. Animals. 2022; 12(24):3499. https://doi.org/10.3390/ani12243499
Chicago/Turabian StylePietroluongo, Guido, Belén Quintana Martín-Montalvo, Simone Antichi, Anastasia Miliou, and Valentina Costa. 2022. "First Assessment of Micro-Litter Ingested by Dolphins, Sea Turtles and Monk Seals Found Stranded along the Coasts of Samos Island, Greece" Animals 12, no. 24: 3499. https://doi.org/10.3390/ani12243499
APA StylePietroluongo, G., Quintana Martín-Montalvo, B., Antichi, S., Miliou, A., & Costa, V. (2022). First Assessment of Micro-Litter Ingested by Dolphins, Sea Turtles and Monk Seals Found Stranded along the Coasts of Samos Island, Greece. Animals, 12(24), 3499. https://doi.org/10.3390/ani12243499










