Courtship and Reproduction of the Whitetip Reef Shark Triaenodon obesus (Carcharhiniformes: Carcharhinidae) in an Ex Situ Environment, with a Description of the Late Embryonic Developmental Stage
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
Ultrasound Examination
3. Results
3.1. Courtship, Copulation, and Pregnancy
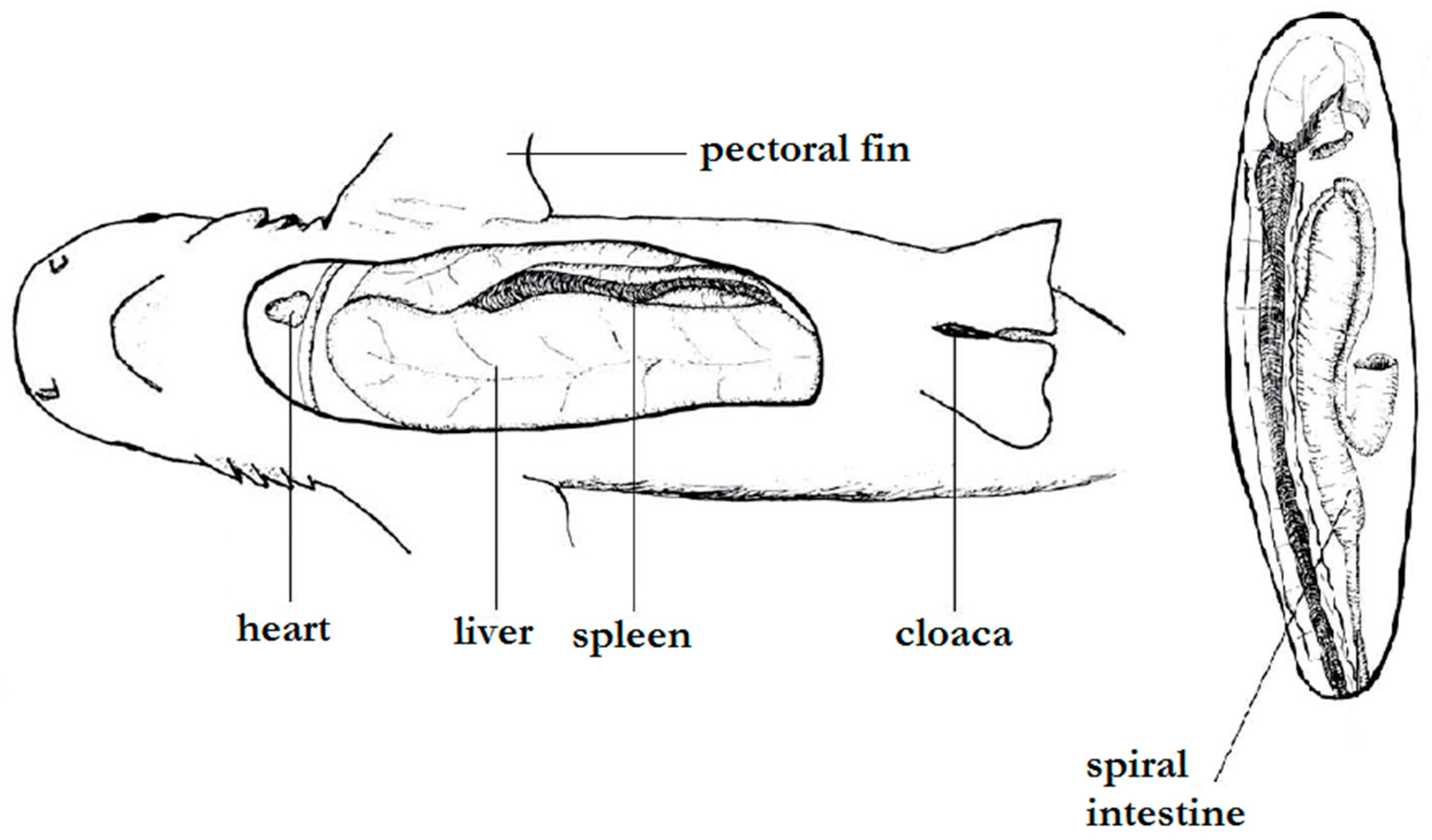
3.2. Internal Morphology
3.3. Birth and Feeding
4. Discussion
4.1. Courtship, Copulation, and Pregnancy
4.2. Internal Morphology
4.3. Birth and Feeding
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robbins, W.D. Abundance, Demography and Population Structure of the Grey Reef Shark (Carcharhinus amblyrhynchos) and the White Tip Reef Shark (Triaenodon obesus) (Fam. Carcharhinidae). Ph.D. Thesis, James Cook University, Douglas, Australia, 2006. [Google Scholar]
- Ferretti, F.; Worm, B.; Britten, G.L.; Heithaus, M.R.; Lotze, H.K. Patterns and ecosystem consequences of shark declines in the ocean. Ecol. Lett. 2010, 13, 1055–1071. [Google Scholar] [CrossRef] [PubMed]
- Pistevos, J.C.A.; Nagelkerken, I.; Rossi, T.; Olmos, M.; Connell, S.D. Ocean acidification and global warming impair shark hunting behavior and growth. Sci. Rep. 2015, 5, 16293. [Google Scholar] [CrossRef] [PubMed]
- Pacoureau, N.; Rigby, C.L.; Kyne, P.M.; Sherley, R.B.; Winker, H.; Carlson, J.K.; Fordham, S.V.; Barreto, R.; Fernando, D.; Francis, M.P.; et al. Half a century of global decline in oceanic sharks and rays. Nature 2021, 587, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Ruppert, J.L.W.; Travers, M.J.; Smith, L.L.; Fortin, M.-J.; Meekan, M.G. Caught in the Middle: Combined Impacts of Shark Removal and Coral Loss on the Fish Communities of Coral Reefs. PLoS ONE 2013, 8, e74648. [Google Scholar] [CrossRef] [PubMed]
- Heithaus, M.R.; Alcoverro, T.; Arthur, R.; Burkholder, D.A.; Coates, K.A.; Christianen, M.J.A.; Kelkar, N.; Manuel, S.A.; Wirsing, A.J. Seagrasses in the age of sea turtle conservation and shark overfishing. Front. Mar. Sci. 2014, 1, 28. [Google Scholar] [CrossRef]
- Roff, G.; Doropoulos, C.; Rogers, A.; Bozec, Y.-M.; Krueck, N.C.; Aurellado, E.; Priest, M.; Birrell, C.; Mumby, P.J. The ecological role of sharks on coral reefs. Trends Ecol. Evol. 2016, 31, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Ruppert, J.L.W.; Fortin, M.-J.; Meekan, M.G. The ecological role of sharks on coral reefs: Response to Roff et al. Trends Ecol. Evol. 2016, 31, 586–587. [Google Scholar] [CrossRef]
- Pauly, D.; Zeller, D. Accurate catches and the sustainability of coral reef fisheries. Curr. Opin. Environ. Sustain. 2014, 7, 44–51. [Google Scholar] [CrossRef]
- Barange, M. Ecosystem science and the sustainable management of marine resources: From Rio to Johannesburg. Front. Ecol. Environ. 2003, 1, 190–196. [Google Scholar] [CrossRef]
- Humphreys, J.; Clark, R.W.E. A critical history of marine protected areas. In Marine Protected Areas: Science, Policy and Management, 1st ed.; Humphreys, J., Clark, R.W.E., Eds.; Elsevier Ltd.: Amsterdam, The Netherlands, 2020; pp. 1–12. [Google Scholar]
- Whitney, N.M.; Robbins, W.D.; Schultz, J.K.; Bowen, B.W.; Holland, K.N. Oceanic dispersal in a sedentary reef shark (Triaenodon obesus): Genetic evidence for extensive connectivity without a pelagic larval stage. J. Biogeogr. 2012, 39, 1144–1156. [Google Scholar] [CrossRef]
- Bornatowski, H.; Loose, R.; Sampaio, C.L.S.; Gadig, O.B.F.; Carvalho-Filho, A.; Domingues, R.R. Human introduction or natural dispersion? Atlantic Ocean occurrence of the Indo-Pacific whitetip reef shark Triaenodon obesus. J. Fish Biol. 2018, 92, 537–542. [Google Scholar] [CrossRef] [PubMed]
- López-Garro, A.; Zanella, I.; Golfín-Duarte, G.; Pérez-Montero, M. Residencia del tiburón punta blanca de arrecife, Triaenodon obesus, (Carcharhiniformes: Carcharhinidae) en las bahías Chatam y Wafer del Parque Nacional Isla del Coco, Costa Rica. Rev. Biol. Trop. 2020, 68, S330–S339. [Google Scholar] [CrossRef]
- Dillon, E.M.; McCauley, D.J.; Morales-Saldaña, J.M.; Leonard, N.D.; Zhao, J. Fossil dermal denticles reveal the preexploitation baseline of a Caribbean coral reef shark community. Proc. Natl. Acad. Sci. USA 2021, 118, e2017735118. [Google Scholar] [CrossRef]
- Graham, N.A.J.; Wilson, S.K.; Jennings, S.; Polunin, N.V.C.; Robinson, J.; Bijoux, J.P.; Daw, T.M. Lag effects in the impacts of mass coral bleaching on coral reef fish, fisheries, and ecosystems. Conserv. Biol. 2007, 21, 1291–1300. [Google Scholar] [CrossRef]
- Heron, S.F.; Maynard, J.A.; van Hooidonk, R.; Eakin, C.M. Warming trends and bleaching stress of the world’s coral reefs 1985–2012. Sci. Rep. 2016, 6, 38402. [Google Scholar] [CrossRef] [PubMed]
- Cornwall, C.E.; Comeau, S.; Kornder, N.A.; Perry, C.T.; van Hooidonk, R.; DeCarlo, T.M.; Pratchett, M.S.; Anderson, K.D.; Browne, N.; Carpenter, R.; et al. Global declines in coral reef calcium carbonate production under ocean acidification and warming. Proc. Natl. Acad. Sci. USA 2021, 118, e2015265118. [Google Scholar] [CrossRef]
- Simpfendorfer, C.; Yuneni, R.R.; Tanay, D.; Seyha, L.; Haque, A.B.; Bineesh, K.K.; Bin Ali, A.; Gautama, D.A.; Maung, A.; Sianipar, A.; et al. Triaenodon obesus. IUCN Red List. Threat. Species 2020, 2020, e.T39384A173436715. [Google Scholar] [CrossRef]
- Whitney, N.M.; Pyle, R.L.; Holland, K.N.; Barez, J.T. Movements, reproductive seasonality, and fisheries interactions in the whitetip reef shark (Triaenodon obesus) from community-contributed photographs. Environ. Biol. Fishes 2012, 93, 121–136. [Google Scholar] [CrossRef]
- Ebert, D.A.; Fowler, S.; Compagno, L. Sharks of the World: A Fully Illustrated Guide, 1st ed.; Wild Nature Press: Plymouth, UK, 2013. [Google Scholar]
- Buddle, A.L.; Van Dyke, J.U.; Thompson, M.B.; Simpfendorfer, C.A.; Whittington, C.M. Evolution of placentotrophy: Using viviparous sharks as a model to understand vertebrate placental evolution. Mar. Freshw. Res. 2018, 70, 908–924. [Google Scholar] [CrossRef]
- Mylniczenko, N.D. A1: Anatomy and taxonomy. In Clinical Guide to Fish Medicine, 1st ed.; Hadfield, C.A., Clayton, L.A., Eds.; John Willey & Sons, Inc.: Hoboken, NJ, USA, 2021; pp. 3–34. [Google Scholar]
- Schaller, P. Husbandry and reproduction of whitetip reef sharks Triaenodon obesus at Steinhart Aquarium, San Francisco. Int. Zoo Yearb. 2006, 40, 232–240. [Google Scholar] [CrossRef]
- Anderson, B.; Belcher, C.; Slack, J.A.; Gelsleichter, J. Evaluation of the use of portable ultrasonography to determine pregnancy status and fecundity in bonnethead shark Sphyrna tiburo. J. Fish Biol. 2018, 93, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Carrier, J.C.; Murru, F.L.; Walsh, M.T.; Pratt, H.L., Jr. Assessing reproductive potential and gestation in nurse sharks (Ginglymostoma cirratum) using ultrasonography and endoscopy: An example of bridging the gap between field research and captive studies. Zoo Biol. 2003, 22, 179–187. [Google Scholar] [CrossRef]
- Grant, K.R.; Campbell, T.W.; Silver, T.I.; Olea-Popelka, F.J. Validation of an ultrasound-guided technique to establish a liver-to-coelom ratio and a comparative analysis of the ratios among acclimated and recently wild-caught southern stingrays, Dasyatis americana. Zoo Biol. 2012, 32, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Janse, M.; Zimmerman, B.; Geerlings, L.; Brown, C.; Nagelkerke, L.A.J. Sustainable species management of the elasmobranch populations within European aquariums: A conservation challenge. J. Zoo Aquar. Res. 2017, 5, 172–181. [Google Scholar] [CrossRef]
- Compagno, L.J.V. FAO Species Catalogue Vol. 4. Sharks of the World. An annotated and illustrated catalogue of shark species known to date. Part 2. Carcharhiniformes. FAO Fish. Synop. 1984, 125, 251–655. [Google Scholar]
- Henningsen, A.D. Tonic immobility in 12 elasmobranchs: Use as an aid in captive husbandry. Zoo Biol. 1994, 13, 325–332. [Google Scholar] [CrossRef]
- Kessel, S.T.; Hussey, N.E. Tonic immobility as an anaesthetic for elasmobranchs during surgical implantation procedures. Can. J. Fish. Aquat. Sci. 2015, 72, 1287–1291. [Google Scholar] [CrossRef]
- Crow, G.L.; Brock, J.A. Chapter 30: Necropsy methods and procedures for elasmobranchs. In The Elasmobranch Husbandry Manual: Captive Care of Sharks, Rays and Their Relatives, 1st ed.; Smith, M., Warmolts, D., Thoney, D., Hueter, R., Eds.; Ohio Biological Survey, Inc.: Columbus, OH, USA, 2004; pp. 467–472. [Google Scholar]
- White, W.T. Catch composition and reproductive biology of whaler sharks (Carcharhiniformes: Carcharhinidae) caught by fisheries in Indonesia. J. Fish Biol. 2007, 71, 1512–1540. [Google Scholar] [CrossRef]
- Whitney, N.M.; Pratt, H.L., Jr.; Carrier, J.C. Group courtship, mating behaviour and siphon sac function in the whitetip reef shark, Triaenodon obesus. Anim. Behav. 2004, 68, 1435–1442. [Google Scholar] [CrossRef]
- Pratt, H.L., Jr.; Carrier, J.C. A review of elasmobranch reproductive behavior with a case study on the nurse shark, Ginglymostoma cirratum. Environ. Biol. Fishes 2001, 60, 157–188. [Google Scholar] [CrossRef]
- Tricas, T.C.; Le Feuvre, E.M. Mating in the reef white-tip shark Triaenodon obesus. Mar. Biol. 1985, 84, 233–237. [Google Scholar] [CrossRef]
- Ebert, D.A.; Ebert, T.B. Reproduction, diet and habitat use of leopard sharks Triakis semifasciata (Girard), in Humbolt Bay, California, USA. Mar. Freshw. Res. 2005, 56, 1089–1098. [Google Scholar] [CrossRef]
- Rangel, B.S.; Hammerschlag, N.; Sulikowski, J.A.; Moreira, R.G. Dietary and reproductive biomarkers in a generalist apex predator reveal differences in nutritional ecology across life stages. Mar. Ecol. Prog. Ser. 2021, 664, 149–163. [Google Scholar] [CrossRef]
- Whitney, N.M.; Papastamatiou, Y.P.; Holland, K.N.; Lowe, C.G. Use of an acceleration data logger to measure diel activity patterns in captive whitetip reef sharks, Triaenodon obesus. Aquat. Living Resour. 2007, 20, 299–305. [Google Scholar] [CrossRef]
- Mejía-Falla, P.A.; Navia, A.F.; Lozano, R.; Tóbon-López, A.; Narváez, K.; Muñoz-Osorio, L.A.; Mejía-Ladino, L.M.; López-García, J. Uso de hábitat de Triaenodon obesus (Carcharhiniformes: Carcharhinidae), Rhincodon typus (Orectolobiformes: Rhincodontidae) y Manta birostris (Myliobatiformes: Myliobatidae) en el Parque Nacional Gorgona, Pacífico Colombiano. Rev. Biol. Trop. 2014, 62, 329–342. [Google Scholar] [CrossRef]
- Robbins, W.D.; Hisano, M.; Connolly, S.R.; Choat, J.H. Ongoing collapse of coral-reef shark populations. Curr. Biol. 2006, 16, 2314–2319. [Google Scholar] [CrossRef] [PubMed]
- Onimaru, K.; Motone, F.; Kiyatake, I.; Nishida, K.; Kuraku, S. A staging table for the embryonic development of the brownbanded bamboo shark (Chiloscyllium punctatum). Dev. Dyn. 2018, 247, 712–723. [Google Scholar] [CrossRef]
- Guallart, J.; Vicent, J.J. Changes in composition during embryo development of the gulper shark, Centrophorus granulosus (Elasmobranchii, Centrophoridae): An assessment of maternal-embryonic nutritional relationship. Environ. Biol. Fishes 2001, 61, 135–150. [Google Scholar] [CrossRef]
- Sato, K.; Nakamura, M.; Tomita, T.; Toda, M.; Miyamoto, K.; Nozu, R. How great white sharks nourish their embryos to a large size: Evidence of lipid histotrophy in lamnoid shark reproduction. Biol. Open 2016, 5, 1211–1215. [Google Scholar] [CrossRef]
- Castro, J.I. Biology of the blacktip shark, Carcharhinus limbatus, off the southeastern United States. Bull. Mar. Sci. 1996, 59, 508–522. [Google Scholar]
- Macías-Cuyare, M.; Tavares, R.; Oddone, M.C. Reproductive biology and placentotrophic embryonic development of the smalleye smooth-hound shark, Mustelus higmani, from the south-eastern Caribbean. J. Mar. Biol. Assoc. U. K. 2020, 100, 1337–1347. [Google Scholar] [CrossRef]
- López, J.A.; Ryburn, J.A.; Fedrigo, O.; Naylor, G.J.P. Phylogeny of sharks of the family Triakidae (Carcharhiniformes) and its implications for the evolution of carcharhiniform placental viviparity. Mol. Phylogenet. Evol. 2006, 40, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, R.G.; Dodrill, J.W.; Linley, P.A. Reproduction and embryonic development of the sand tiger shark, Odontaspis taurus (Rafinesque). Fish. Bull. 1983, 81, 201–225. [Google Scholar]
- Joung, S.-J.; Hsu, H.-H. Reproduction and Embryonic Development of the Shortfin Mako, Isurus oxyrinchus Rafinesque, 1810, in the Northwestern Pacific. Zool. Stud. 2005, 44, 487–496. [Google Scholar]
- Buckley, K.A.; Crook, D.A.; Pillans, R.D.; Smith, L.; Kyne, P.M. Sustainability of threatened species displayed in public aquaria, with a case study of Australian sharks and rays. Rev. Fish Biol. Fish. 2018, 28, 137–151. [Google Scholar] [CrossRef]
- Portnoy, D.S.; Hollenbeck, C.M.; Johnston, J.S.; Casman, H.M.; Gold, J.R. Parthenogenesis in a whitetip reef shark Triaenodon obesus involves a reduction in ploidy. J. Fish Biol. 2014, 85, 502–508. [Google Scholar] [CrossRef]
- Frisch, A.J.; Ireland, M.; Rizzari, J.R.; Lönnstedt, O.M.; Magnenat, K.A.; Mirbach, C.E.; Hobbs, J.-P.A. Reassessing the trophic role of reef sharks as apex predators on coral reefs. Coral Reefs 2016, 35, 459–472. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos, S.R.; Takatsuka, V.; Bonatelli, S.P.; Amaral, N.L.L.; Goés, M.F.; Valle, R.F. Courtship and Reproduction of the Whitetip Reef Shark Triaenodon obesus (Carcharhiniformes: Carcharhinidae) in an Ex Situ Environment, with a Description of the Late Embryonic Developmental Stage. Animals 2022, 12, 3291. https://doi.org/10.3390/ani12233291
Santos SR, Takatsuka V, Bonatelli SP, Amaral NLL, Goés MF, Valle RF. Courtship and Reproduction of the Whitetip Reef Shark Triaenodon obesus (Carcharhiniformes: Carcharhinidae) in an Ex Situ Environment, with a Description of the Late Embryonic Developmental Stage. Animals. 2022; 12(23):3291. https://doi.org/10.3390/ani12233291
Chicago/Turabian StyleSantos, Sérgio Ricardo, Veronica Takatsuka, Shayra P. Bonatelli, Nicole L. L. Amaral, Matheus F. Goés, and Rafael F. Valle. 2022. "Courtship and Reproduction of the Whitetip Reef Shark Triaenodon obesus (Carcharhiniformes: Carcharhinidae) in an Ex Situ Environment, with a Description of the Late Embryonic Developmental Stage" Animals 12, no. 23: 3291. https://doi.org/10.3390/ani12233291
APA StyleSantos, S. R., Takatsuka, V., Bonatelli, S. P., Amaral, N. L. L., Goés, M. F., & Valle, R. F. (2022). Courtship and Reproduction of the Whitetip Reef Shark Triaenodon obesus (Carcharhiniformes: Carcharhinidae) in an Ex Situ Environment, with a Description of the Late Embryonic Developmental Stage. Animals, 12(23), 3291. https://doi.org/10.3390/ani12233291





