Early Locomotor Training in Tetraplegic Post-Surgical Dogs with Cervical Intervertebral Disc Disease
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
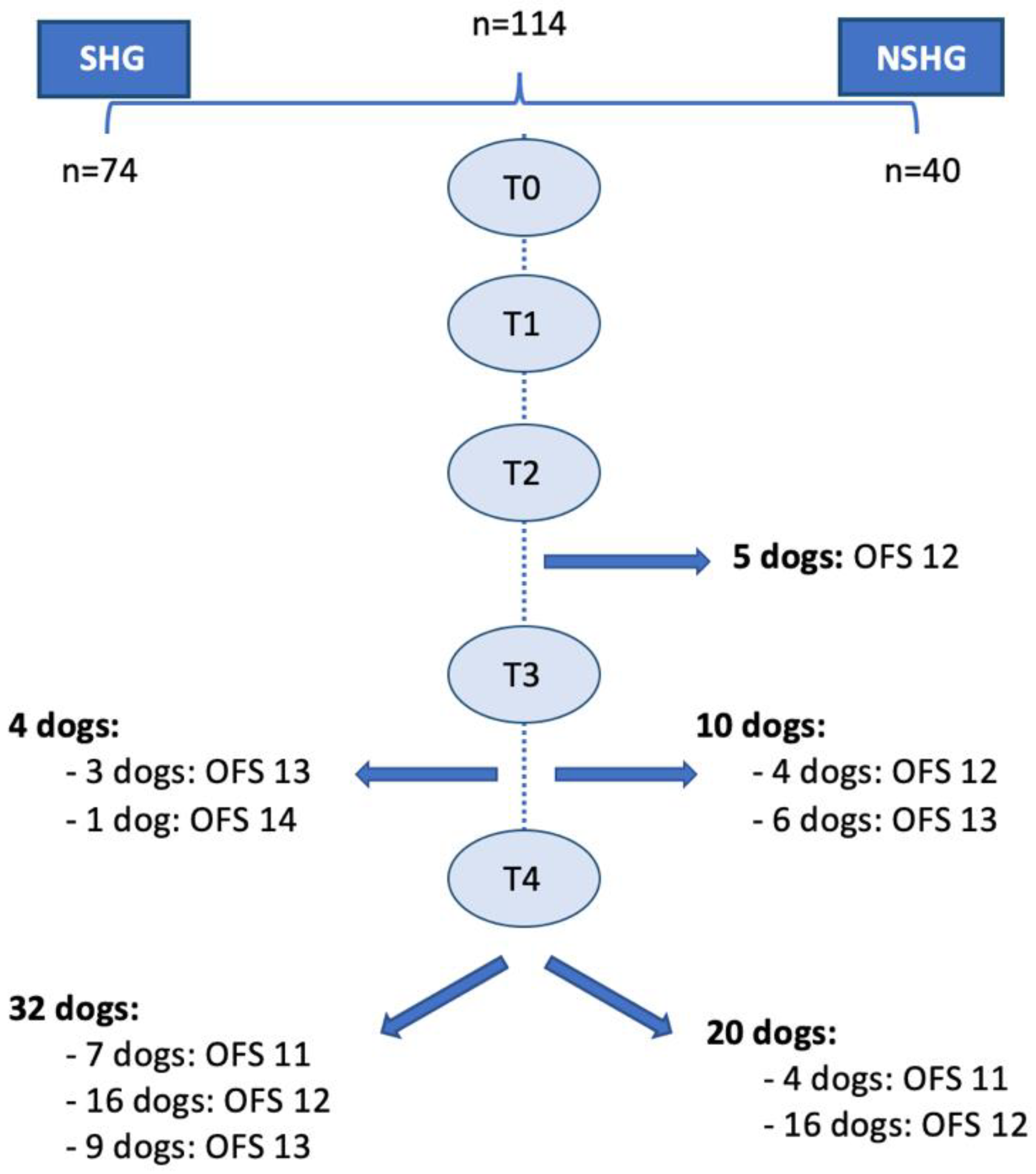
2.1. Participants
2.2. Study Design
2.3. Outcomes
2.4. Primary Supportive Care
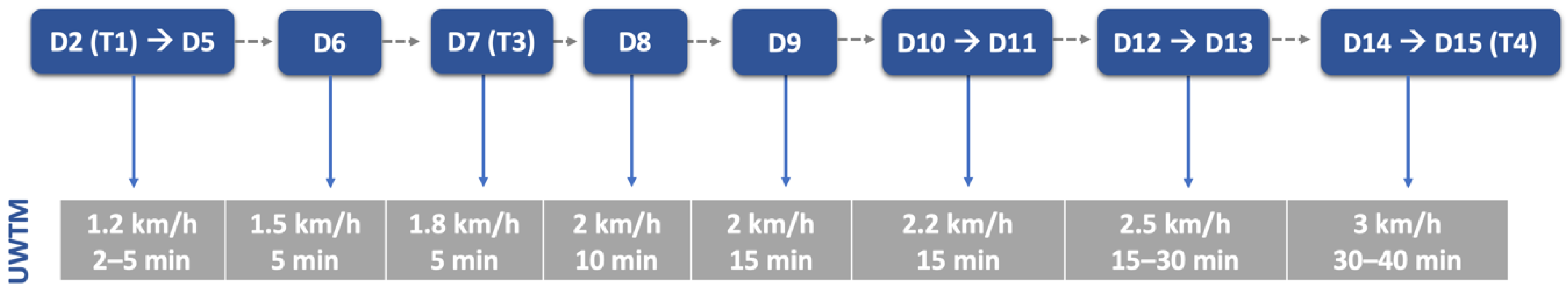
2.5. Locomotor Training Procedures
2.6. Data Collection
2.7. Statistical Analysis
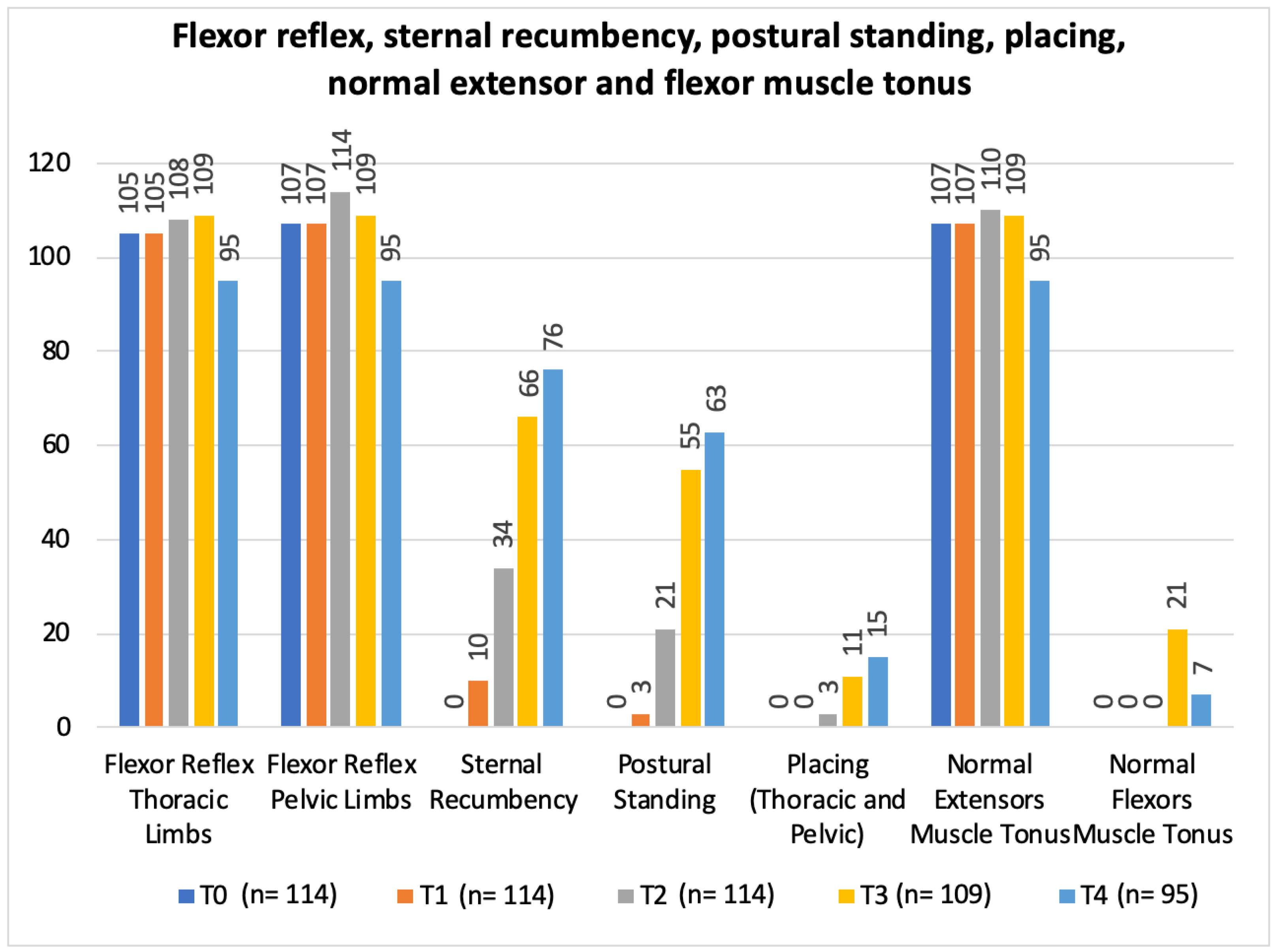
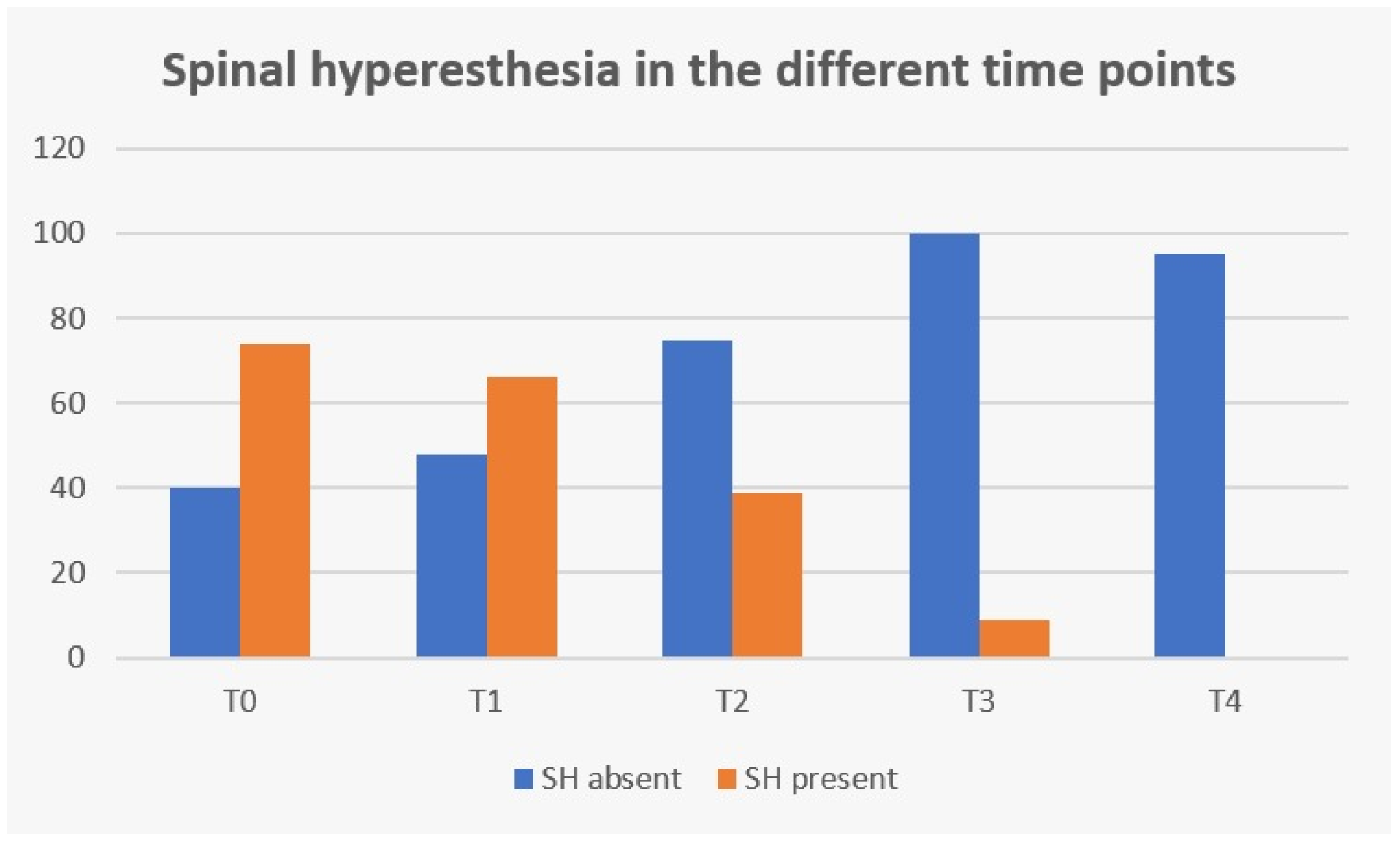
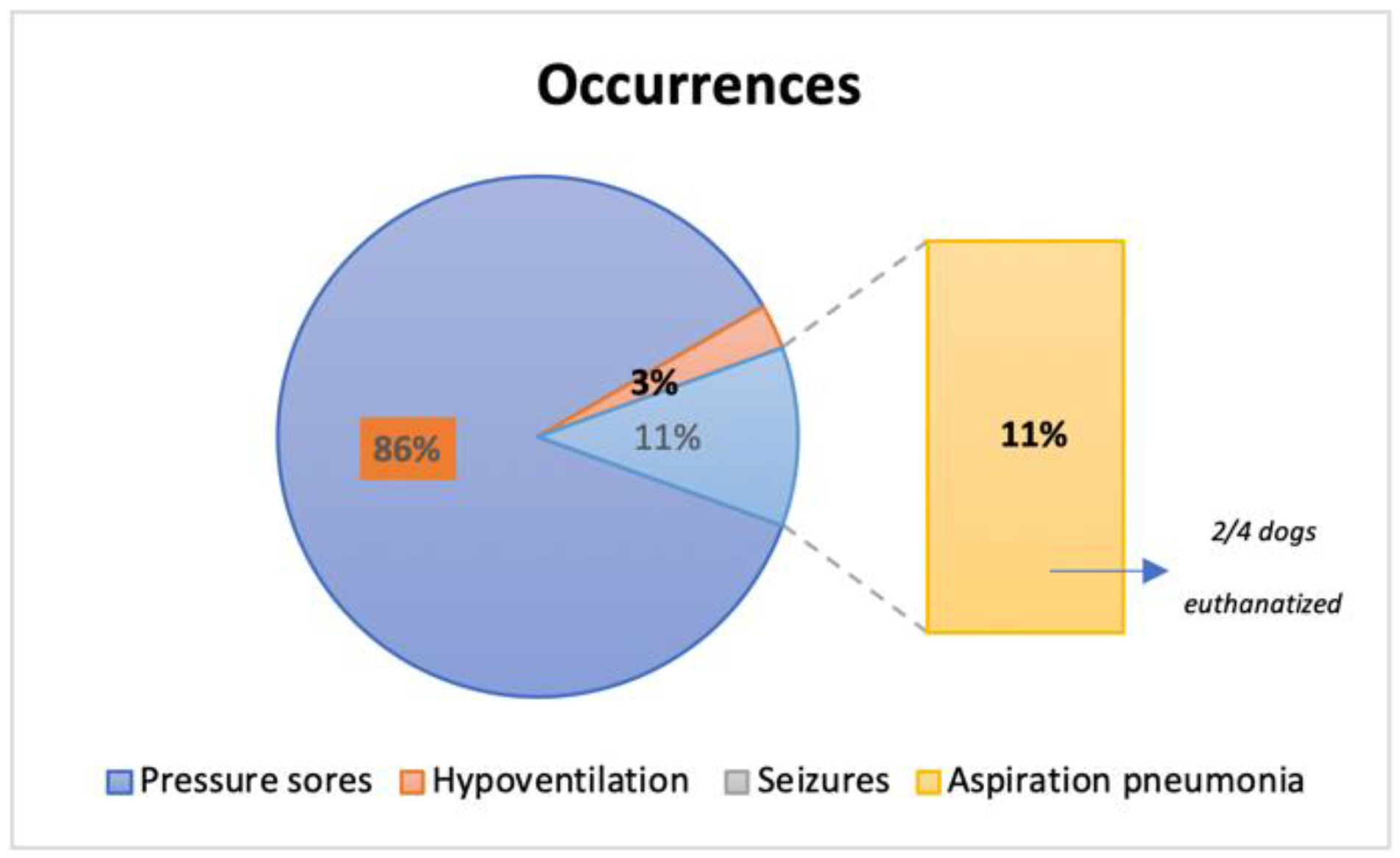
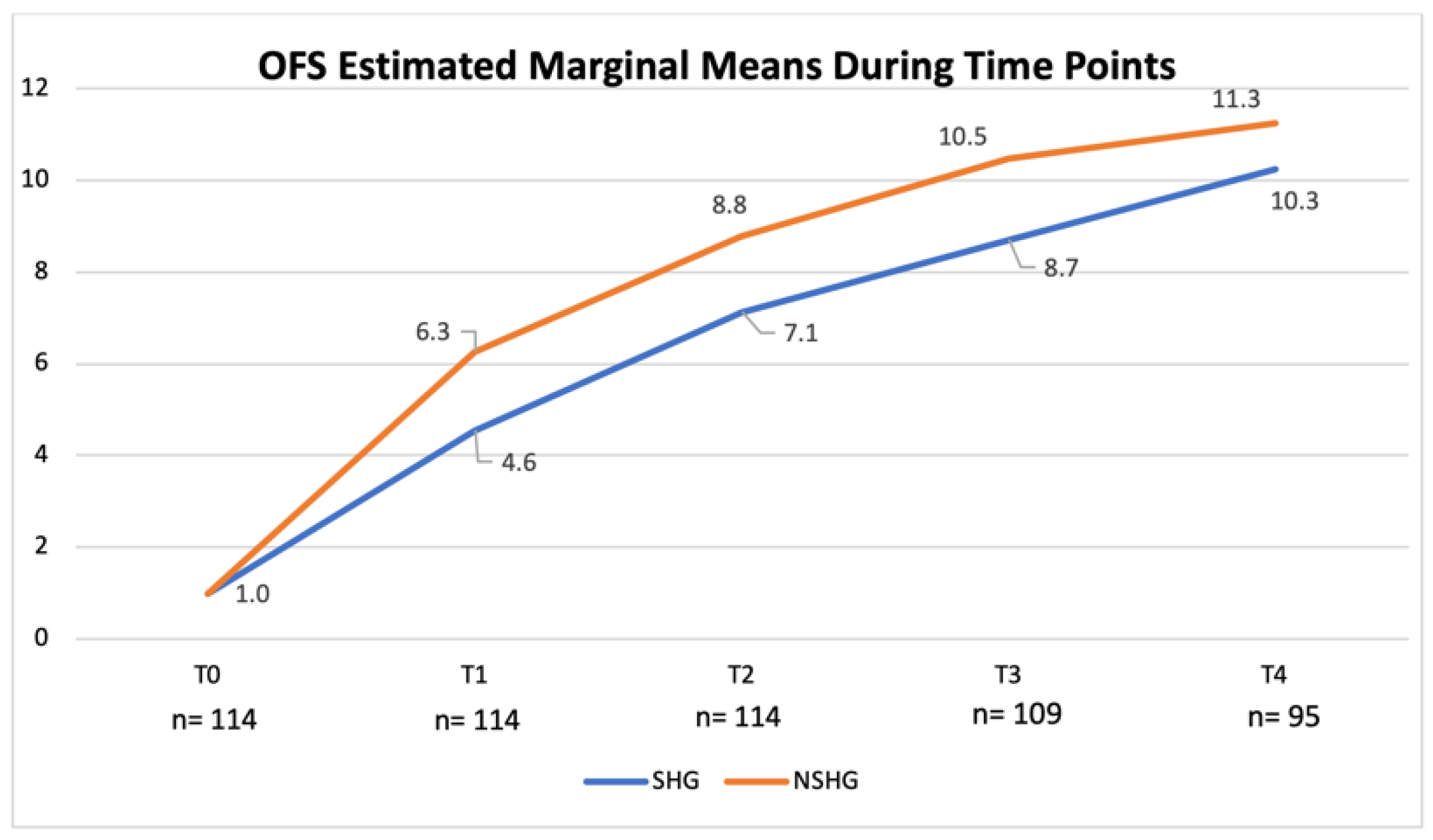
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Toombs, J.P. Cervical intervertebral disk disease in dogs. Compend. Contin. Educ. Pract. Vet. 1992, 14, 1477–1486. [Google Scholar]
- Seim, H.B. Surgery of the cervical spine. In Small Animal Surgery; Duncan, L.L., Ed.; Mosby Publishing Co.: St. Louis, MO, USA, 2002; pp. 1228–1237. [Google Scholar]
- Beal, M.W.; Paglia, D.T.; Griffin, G.M.; Hughes, D.; King, L.G. Ventilatory failure, ventilator management, and outcome in dogs with cervical spinal disorders: 14 cases (1991–1999). J. Am. Vet. Med. Assoc. 2001, 218, 1598–1602. [Google Scholar] [CrossRef] [PubMed]
- Java, M.A.; Drobatz, K.J.; Gilley, R.S.; Long, S.N.; Kushner, L.I.; King, L.G. Incidence of and risk factors for postoperative pneumonia in dogs anesthetized for diagnosis or treatment of intervertebral disk disease. J. Am. Vet. Med. Assoc. 2009, 235, 281–287. [Google Scholar] [CrossRef]
- Kube, S.; Owen, T.; Hanson, S. Severe respiratory compromise secondary to cervical disk herniation in two dogs. J. Am. Anim. Hosp. Assoc. 2003, 39, 513–517. [Google Scholar] [CrossRef]
- Posner, L.P.; Mariani, C.L.; Swanson, C.; Asakawa, M.; Campbell, N.; King, A.S. Perianesthetic morbidity and mortality in dogs undergoing cervical and thoracolumbar spinal surgery. Vet. Anaesth. Analg. 2014, 41, 137–144. [Google Scholar] [CrossRef]
- Rossmeisl, J.H.; White, C.; Pancotto, T.E.; Bays, A.; Henao-Guerrero, P.N. Acute adverse events associated with ventral slot decompression in 546 dogs with cervical intervertebral disc disease. Vet. Surg. 2013, 42, 795–806. [Google Scholar]
- Andruzzi, N.M.; Simon, B.T.; Boudreau, E. Subclinical hypoventilation in dogs undergoing ventral slot decompressive surgery for cervical myelopathy due to intervertebral disc herniation. Front. Vet. Sci. 2021, 8, 777052. [Google Scholar] [CrossRef]
- Hansen, H.J. A pathologic-anatomical study on disk degeneration in the dog. Acta Orthop. Scand. 1952, 11, 1–117. [Google Scholar]
- Jerram, R.M.; Dewey, C.W. Acute thoracolumbar disk extrusion in dogs—Part I. J. Am. Anim. Hosp. Assoc. 1999, 21, 922–930. [Google Scholar]
- Cheronne, K.L.; Dewey, C.W.; Coates, J.R.; Bergman, R.L. A retrospective comparison of cervical intervertebral disk disease in nonchondrodystrophic large dogs versus small dogs. J. Am. Anim. Hosp. Assoc. 2004, 40, 316–320. [Google Scholar] [CrossRef]
- Dewey, C.W.; Costa, R.C. Myelopathies: Disorders of the spinal cord. In Practical Guide to Canine and Feline Neurology, 3rd ed.; Dewey, C.W., da Costa, R.C., Eds.; Wiley Blackwell: New York, NY, USA, 2016; pp. 329–403. [Google Scholar]
- Ronthal, M.A.; Pain, N. Bradley’s Neurology in Clinical Practice, 7th ed.; Daroff, R.B., Jankovic, J., Mazziotia, J.C., Pomeroy, S.L., Eds.; Elsevier: London, UK, 2016; pp. 324–331. [Google Scholar]
- Jeffery, N.D.; Mckee, W.M. Surgery for disc-associated wobbler syndrome in the dog—An examination of the controversy. J. Small Anim. Pract. 2001, 42, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Coates, J.R. Paraparesis. In BSAVA Manual of Canine and Feline Neurology, 3rd ed.; Platt, S.R., Olby, N.J., Eds.; BSAVA: Gloucester, UK, 2004; pp. 237–264. [Google Scholar]
- Bruecker, K.A.; Seim, H.B., III; Withrow, S.J. Clinical evaluation of three surgical methods for treatment of caudal cervical spondylomyelopathy of dogs. Vet. Surg. 1989, 18, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.R.; Aron, D.N.; Roberts, R.E. Observation of a secondary compressive lesion after treatment of caudal cervical spondylomyelopathy in a dog. J. Am. Vet. Med. Assoc. 1994, 205, 1297–1299. [Google Scholar] [PubMed]
- da Costa, R.C.; Parent, J.M. One-year clinical and magnetic resonance imaging follow-up of Doberman Pinschers with cervical spondylomyelopathy treated medically or surgically. J. Am. Vet. Med. Assoc. 2007, 231, 243–250. [Google Scholar] [CrossRef] [PubMed]
- McKee, W.M.; Sharp, N.J.H. Cervical spondylopathy. In Textbook of Small Animal Surgery, 3rd ed.; Slatter, D.H., Ed.; Saunders: Philadelphia, PA, USA, 2003; pp. 1180–1193. [Google Scholar]
- Rusbridge, C.; Wheeler, S.J.; Torrington, A.M.; Pead, M.J.; Carmichael, S. Comparison of two surgical techniques for the management of cervical spondylomyelopathy in Dobermans. J. Small. Anim. Pract. 1998, 39, 425–431. [Google Scholar] [CrossRef]
- Fauber, A.E. Cervical disc disease: Ventral slot versus hemilaminectomy versus dorsal laminectomy. In Advances in Intervertebral Disc Disease in Dogs and Cats; Fingeroth, J.M., Thomas, W.B., Eds.; Wiley Blackwell: Oxford, UK, 2015; pp. 226–231. [Google Scholar]
- Seim, H.B.; Withrow, S.J. Pathophysiology and diagnosis of caudal cervical spondylomyelopathy with emphasis on the dobermann pinscher. J. Am. Anim. Hosp. Assoc. 1982, 18, 241–251. [Google Scholar]
- Dewey, C.W.; da Costa, R.C.; Ducoté, J.M. Neurodiagnostics. In Practical Guide to Canine and Feline Neurology, 3rd ed.; Dewey, C.W., da Costa, R.C., Eds.; Wiley Blackwell: New York, NY, USA, 2016; pp. 61–86. [Google Scholar]
- Gavin, P.R.; Levine, J.M. Advances in Imaging for Intervertebral Disc Disease. In Advances in Intervertebral Disc Disease in Dogs and Cats; Fingeroth, J.M., Thomas, W.B., Eds.; Wiley Blackwell: Oxford, UK, 2015; pp. 135–146. [Google Scholar]
- Robertson, I.; Thrall, D.E. Imaging dogs with suspected disc herniation: Pros and cons of myelography, computed tomography, and magnetic resonance. Vet. Radiol. Ultrasound 2011, 52, 81–84. [Google Scholar] [CrossRef]
- Da Costa, R.C.; Parent, J.M.; Holmberg, D.L.; Sinclair, D.; Monteith, G. Outcome of medical and surgical treatment in dogs with cervical spondylomyelopathy: 104 cases (1988–2004). J. Am. Vet. Med. Assoc. 2008, 233, 1284–1290. [Google Scholar] [CrossRef]
- Reints Bok, T.E.; Willemsen, K.; van Rijen, M.H.; Grinwis, G.C.; Tryfonidou, M.A.; Meij, B.P. Instrumented cervical fusion in nine dogs with caudal cervical spondylomyelopathy. Vet. Surg. 2019, 48, 1287–1298. [Google Scholar] [CrossRef]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Viegas, I.; Gamboa, Ó.; Ferreira, A. A comparison between body weight-supported treadmill training and conventional over-ground training in dogs with incomplete spinal cord injury. Front. Vet. Sci. 2021, 8, 1–14. [Google Scholar] [CrossRef]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Carvalho, C.; Coelho, T.; Silva, C.; Viegas, I.; Gamboa, Ó.; Ferreira, A. Controlled clinical study of intensive neurorehabilitation in post-surgical dogs with severe acute intervertebral disc extrusion. Animals 2021, 11, 3034. [Google Scholar] [CrossRef]
- Dietz, V.; Wirz, M.; Curt, A.; Colombo, G. Locomotor pattern in paraplegic patients: Training effects and recovery of spinal cord function. Spinal Cord 1998, 36, 380–390. [Google Scholar] [CrossRef]
- Nam, K.Y.; Kim, H.J.; Kwon, B.S.; Park, J.-W.; Lee, H.J.; Yoo, A. Robot-assisted gait training (Lokomat) improves walking function and activity in people with spinal cord injury: A systematic review. J. Neuroeng. Rehabil. 2017, 14, 24. [Google Scholar] [CrossRef] [PubMed]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Carvalho, C.; Silva, C.; Coelho, T.; Gamboa, Ó.; Ferreira, A. Functional neurorehabilitation in dogs with an incomplete recovery 3 months following intervertebral disc surgery: A case series. Animals 2021, 11, 2442. [Google Scholar] [CrossRef]
- Côté, M.-P.; Murray, M.; Lemay, M.A. Rehabilitation Strategies after Spinal Cord Injury: Inquiry into the Mechanisms of Success and Failure. J. Neurotrauma 2017, 34, 1841–1857. [Google Scholar] [CrossRef]
- Huie, J.R.; Morioka, K.; Haefeli, J.; Ferguson, A.R. What is being trained? How divergent forms of plasticity compete to shape locomotor recovery after spinal cord injury. J. Neurotrama 2017, 34, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.T.; Huntingford, J.L. Nursing care and rehabilitation therapy for patients with neurologic disease. In Practical Guide to Canine and Feline Neurology; Dewey, C.W., da Costa, R.C., Eds.; Wiley Blackweel: Oxford, UK, 2016; pp. 559–583. [Google Scholar]
- Martins, Â.; Ferreira, A. Neuroreabilitação funcional em lesões medulares. In Fisiatria em Pequenos Animais; Lopes, R.S., Diniz, R., Eds.; Editora Inteligente: São Paulo, Brazil, 2018; pp. 287–298. [Google Scholar]
- De Decker, S.; Gielen, I.; Duchateau, L.; Volk, H.A.; Van Ham, L. Intervertebral disk width in dogs with and without clinical signs of disk associated spondylomyelopathy. BMC Vet. Res. 2012, 8, 126. [Google Scholar] [CrossRef]
- da Costa, R.C.; Parent, J.M.; Partlow, G.; Dobson, H.; Holmberg, D.L.; Lamarre, J. Morphologic and morphometric magnetic resonance imaging features of Dobermann Pinschers with and without clinical signs of cervical spondylomyelopathy. Am. J. Vet. Res. 2006, 67, 1601–1610. [Google Scholar] [CrossRef]
- Bonelli, M.A.; da Costa, R.C. Clinical and magnetic resonance imaging characterization of cervical spondylomyelopathy in juvenile dogs. J. Vet. Intern. Med. 2019, 33, 2160–2166. [Google Scholar] [CrossRef] [PubMed]
- Bonelli, M.A.; da Costa, R.C. Comparison of the percentage of the C3-C7 vertebral canal occupied by the spinal cord in small breed dogs with that in Doberman Pinschers and Great Danes with and without cervical spondylomyelopathy. Am. J. Vet. Res. 2018, 79, 83–89. [Google Scholar] [CrossRef]
- da Costa, R.C. Cervical spondylomyelopathy (wobbler syndrome) in dogs. Vet. Clin. N. Am. Small Anim. Pract. 2010, 40, 881–913. [Google Scholar] [CrossRef] [PubMed]
- Chambers, J.N.; Oliver, J.E.; Bjorling, D.E. Update on ventral decompression for caudal cervical disk herniation in Doberman Pinschers. J. Am. Anim. Hosp. Assoc. 1986, 22, 775–778. [Google Scholar]
- Bonelli, M.A.; da Costa, L.B.; da Costa, R.C. Magnetic resonance imaging and neurological findings in dogs with disc-associated cervical spondylomyelopathy: A case series. BMC Vet. Res. 2021, 17, 145. [Google Scholar] [CrossRef] [PubMed]
- De Decker, S.; Bhatti, S.F.M.; Duchateau, L.; Martle, V.A.; Van Soens, I.; Van Meervenne, S.A.E.; Saunders, J.H.; Van Ham, L.M.L. Clinical evaluation of 51 dogs treated conservatively for disc-associated wobbler syndrome. J. Small Anim. Pract. 2009, 50, 136–142. [Google Scholar] [CrossRef]
- Stabile, F.; Bernardini, M.; Bevilacqua, G.; Ekiri, A.B.; de Stefani, A.; De Risio, L. Neurological signs and pre- and post-traction low-field MRI findings in Dobermanns with disc-associated cervical spondylomyelopathy. J. Small Anim. Pract. 2015, 56, 331–338. [Google Scholar] [CrossRef]
- Solano, M.A.; Fitzpatrick, N.; Bertran, J. Cervical distraction-stabilization using an intervertebral spacer screw and string-of pearl (SOPTM) plates in 16 dogs with disc-associated wobbler syndrome. Vet. Surg. 2015, 44, 627–641. [Google Scholar] [CrossRef]
- Coates, J.R. Intervertebral disk disease. Vet. Clin. N. Am. Small Anim. Pract. 2000, 30, 77–93. [Google Scholar] [CrossRef]
- Fitch, R.B.; Kerwin, S.C.; Hosgood, G. Caudal cervical intervertebral disk disease in the small dog: Role of distraction and stabilization in ventral slot decompression. J. Am. Anim. Hosp. Assoc. 2000, 36, 68–74. [Google Scholar] [CrossRef]
- Bonelli, M.A.; da Costa, L.B.; da Costa, R.C. Association of neurologic signs with high-field MRI findings in 100 dogs with osseous-associated cervical spondylomyelopathy. Vet. Radiol. Ultrasound 2021, 62, 678–686. [Google Scholar] [CrossRef]
- Frank, L.R.; Roynard, P.F.P. Veterinary neurologic rehabilitation: The rationale for a comprehensive approach. Top. Companion Anim. Med. 2018, 33, 49–57. [Google Scholar] [CrossRef]
- Solopova, I.A.; Selionov, V.A.; Sylos-Labini, F.; Gurfinkel, V.S.; Lacquaniti, F.; Ivanenko, Y.P. Tapping into rhythm generation circuitry in humans during simulated weightlessness conditions. Front. Syst. Neurosci. 2015, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Pang, M.Y.; Yang, J.F. The initiation of the swing phase in human infant stepping: Importance of hip position and leg loading. J. Physiol. 2000, 528, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Martins, Â.; Gouveia, D.; Cardoso, A.; Gamboa, Ó.; Millis, D.; Ferreira, A. Nervous system modulation through electrical stimulation in companion animals. Acta Veter. Scand. 2021, 63, 1–12. [Google Scholar]
- Lewis, M.J.; Jeffery, N.D.; Olby, N.J. The Canine Spinal Cord Injury Consortium (CANSORT-SCI). Ambulation in Dogs with Absent Pain Perception after Acute Thoracolumbar Spinal Cord Injury. Front. Veter. Sci. 2020, 7, 560. [Google Scholar] [CrossRef] [PubMed]
- Dolbow, D.R.; Gorgey, A.S.; Sutor, T.W.; Bochkezanian, V.; Musselman, K. Invasive and non-invasive approaches of electrical stimulation to improve physical functioning after spinal cord injury. J. Clin. Med. 2021, 10, 5356. [Google Scholar] [CrossRef] [PubMed]
- DiMarco, A.F.; Kowalski, K.E. Intercostal muscle pacing with high frequency spinal cord stimulation in dogs. Respir. Physiol. Neurobiol. 2010, 171, 218–224. [Google Scholar] [CrossRef][Green Version]
- DiMarco, A.F. Restoration of respiratory muscle function following spinal cord injury: Review of electrical and magnetic stimulation techniques. Respir. Physiol. Neurobiol. 2005, 147, 273–287. [Google Scholar] [CrossRef]
- Zidan, N.; Sims, C.; Fenn, J.; Williams, K.; Griffith, E.; Early, P.J.; Mariani, C.; Munana, K.R.; Guevar, J.; Olby, N.J. A randomized, blinded, prospective clinical trial of postoperative rehabilitation in dogs after surgical decompression of acute thoracolumbar intervertebral disc herniation. J. Veter. Intern. Med. 2018, 32, 1133–1144. [Google Scholar] [CrossRef]
- McLaughlin, R.M. Kinetic and kinematic gait analysis in dogs. Vet. Clin. N. Am. Small Anim. Pract. 2001, 31, 193–201. [Google Scholar] [CrossRef]
- Foss, K.D.; Smith, R.L.; da Costa, R.C. Kinetic and kinematic follow-up gate analysis in Doberman Pinschers with cervical spondylomyelopathy treated medically and surgically. J. Vet. Intern. Med. 2018, 32, 1126–1132. [Google Scholar] [CrossRef]
- Mann, F.; Wagner-Mann, C.; Dunphy, E.; Ruben, D.; Rochat, M.; Bartels, K. Recurrence rate of presumed thoracolumbar intervertebral disc disease in ambulatory dogs with spinal hyperpathia treated with anti-inflammatory drugs: 78 cases (1997–2000). J. Vet. Emerg. Crit. Care. 2007, 17, 53–60. [Google Scholar] [CrossRef]
- Van Den Brand, R.; Heutschi, J.; Barraud, Q.; DiGiovanna, J.; Bartholdi, K.; Huerlimann, M.; Friedliisabel, L.; Vollenweider, I.; Martin, E.M.; Duis, M.; et al. Restoring voluntary control of locomotion after paralyzing spinal cord injury. Science 2012, 336, 1182–1185. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Cohen, E.B.; Olby, N.J. Magnetic resonance imaging features of dogs with incomplete recovery after acute, severe spinal cord injury. Spinal Cord 2018, 56, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Villalobos, A.E. Assessment and treatment of nonpain conditions in life-limiting disease. Vet. Clin. Small Anim. 2011, 41, 551–563. [Google Scholar] [CrossRef]
- Siddiqui, A.M.; Khazaei, M.; Fehlings, M.G. Translating mechanisms of neuroprotection, regeneration, and repair to treatment of spinal cord injury. Prog. Brain Res. 2015, 218, 15–54. [Google Scholar]
- Branco, É.; Alves, J.G.R.; Pinheiro, L.L.; Coutinho, L.N.; Gomes, C.R.M.; Galvão, G.R.; Santos, G.R.D.O.D.; Moreira, L.F.; David, M.B.; Martins, D.M.; et al. Can Paraplegia by Disruption of the Spinal Cord Tissue Be Reversed? The Signs of a New Perspective. Anat. Rec. 2019, 303, 1812–1820. [Google Scholar] [CrossRef]
- Mojarradi, A.; De Decker, S.; Backstrom, C.; Bergknut, N. Safety of early postoperative hydrotherapy in dogs undergoing thoracolumbar hemilaminectomy. J. Small Anim. Pract. 2021, 62, 1062–1069. [Google Scholar] [CrossRef]
- Gandini, G.; Cizinauskas, S.; Lang, J.; Fatzer, R.; Jaggy, A. Fibrocartilaginous embolism in 75 dogs: Clinical findings and factors influencing recovery rate. J. Sm. Anim. Pract. 2003, 44, 76–80. [Google Scholar] [CrossRef]
- Jeong, I.S.; Piao, Z.; Rahman, M.; Kim, S.; Kim, N.S. Canine thoracolumbar intervertebral disk herniation and rehabilitation therapy after surgical decompression: A retrospective study. J. Adv. Vet. Anim. Res. 2019, 6, 394–402. [Google Scholar] [CrossRef]
- Gordon-Evans, W.J.; Johnson, A.L.; Knap, K.E.; Griffon, D.J. The effect of body condition on postoperative recovery of dachshunds with intervertebral disc-disease treated with postoperative physical rehabilitation. Vet. Surg. 2019, 48, 159–163. [Google Scholar] [CrossRef]
- Gallucci, A.; Dragone, L.; Menchetti, M.; Gagliardo, T.; Pietra, M.; Cardinali, M.; Gandini, G. Acquisition of involuntary spinal locomotion (Spinal Walking) in dogs with irreversible thoracolumbar spinal cord lesion: 81 dogs. J. Vet. Intern. Med. 2017, 222, 762–766. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Bowditch, J.; Laflen, B.; Perry, N.; Yoquelet, R.; Thomovsky, S.A. Pilot Study on Feasibility of Sensory-Enhanced Rehabilitation in Canine Spinal Cord Injury. Front. Vet. Sci. 2022, 9, 921471. [Google Scholar] [CrossRef] [PubMed]
- Bennaim, M.; Porato, M.; Jarleton, A.; Hamon, M.; Carroll, J.D.; Gommeren, K.; Balligand, M. Preliminary evaluation of the effects of photobiomodulation therapy and physical rehabilitation on early postoperative recovery of dogs undergoing hemilaminectomy for treatment of thoracolumbar intervertebral disk disease. Am. J. Veter. Res. 2017, 78, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Hady, L.L.; Schwarz, P.D. Recovery times for dogs undergoing thoracolumbar hemilaminectomy with fenestration and physical rehabilitation: A review of 113 cases. JVMAH 2015, 7, 278–289. [Google Scholar]
- Drum, M.G. Physical rehabilitation of the canine neurologic patient. Vet. Clin. Small Anim. 2010, 40, 181–193. [Google Scholar] [CrossRef]
- Draper, W.E.; Schubert, T.A.; Clemmons, R.M.; Miles, S.A. Low-level laser therapy reduces time to ambulation in dogs after hemilaminectomy: A preliminary study. J. Small Anim. Pract. 2012, 53, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.A.; Tipold, A.; Olby, N.J.; Stein, V.; Granger, N. Cansort-Canine Spinal Cord Injury Consortium (CANSORTSCI) Current Approaches to the Management of Acute Thoracolumbar Disc Extrusion in Dogs. Front. Veter. Sci. 2020, 7, 610. [Google Scholar] [CrossRef]
- Bagley, R. Basics of treatment of important spinal cord diseases of dogs and cats. Fund. Vet. Clin. Neurol. 2005, 14, 323–349. [Google Scholar]
- Hall, J.E. Ch 54 Motor functions of the spinal cord reflexes. In Guyton and Hall Textbook of Medical Physiology, 12th ed.; Hall, J.E., Ed.; Saunders Elsevier: Philadephia, PA, USA, 2011; pp. 655–666. [Google Scholar]
- Uemura, E.E. Ch 16 Motor System. In Fundamentals of Canine Neuroanatomy and Neurophysiology; Uemura, E., Ed.; Wiley Blackwell: New York, NY, USA, 2015; pp. 257–287. [Google Scholar]
- Bradbury, E.J.; McMahon, S.B. Spinal cord repair strategies: Why do they work? Nat. Rev. Neurosci. 2006, 7, 644–653. [Google Scholar] [CrossRef]
- Bareyre, F.M.; Kerschensteiner, M.; Raineteau, O.; Mettenleiter, T.C.; Weinmann, O.; Schwab, M.E. The injured spinal cord spontaneously forms a new intraspinal circuit in adult rats. Nat. Neurosci. 2004, 7, 269–277. [Google Scholar] [CrossRef]
- Flynn, J.R.; Graham, B.A.; Galea, M.P.; Callister, R.J. The role of propriospinal interneurons in recovery from spinal cord injury. Neuropharmacology 2011, 60, 809–822. [Google Scholar] [CrossRef] [PubMed]
- Juvin, L.; Simmers, J.; Morin, D. Propriospinal circuitry underlying interlimb coordination in mammalian quadrupedal locomotion. J. Neurosci. 2005, 25, 6025–6035. [Google Scholar] [CrossRef] [PubMed]


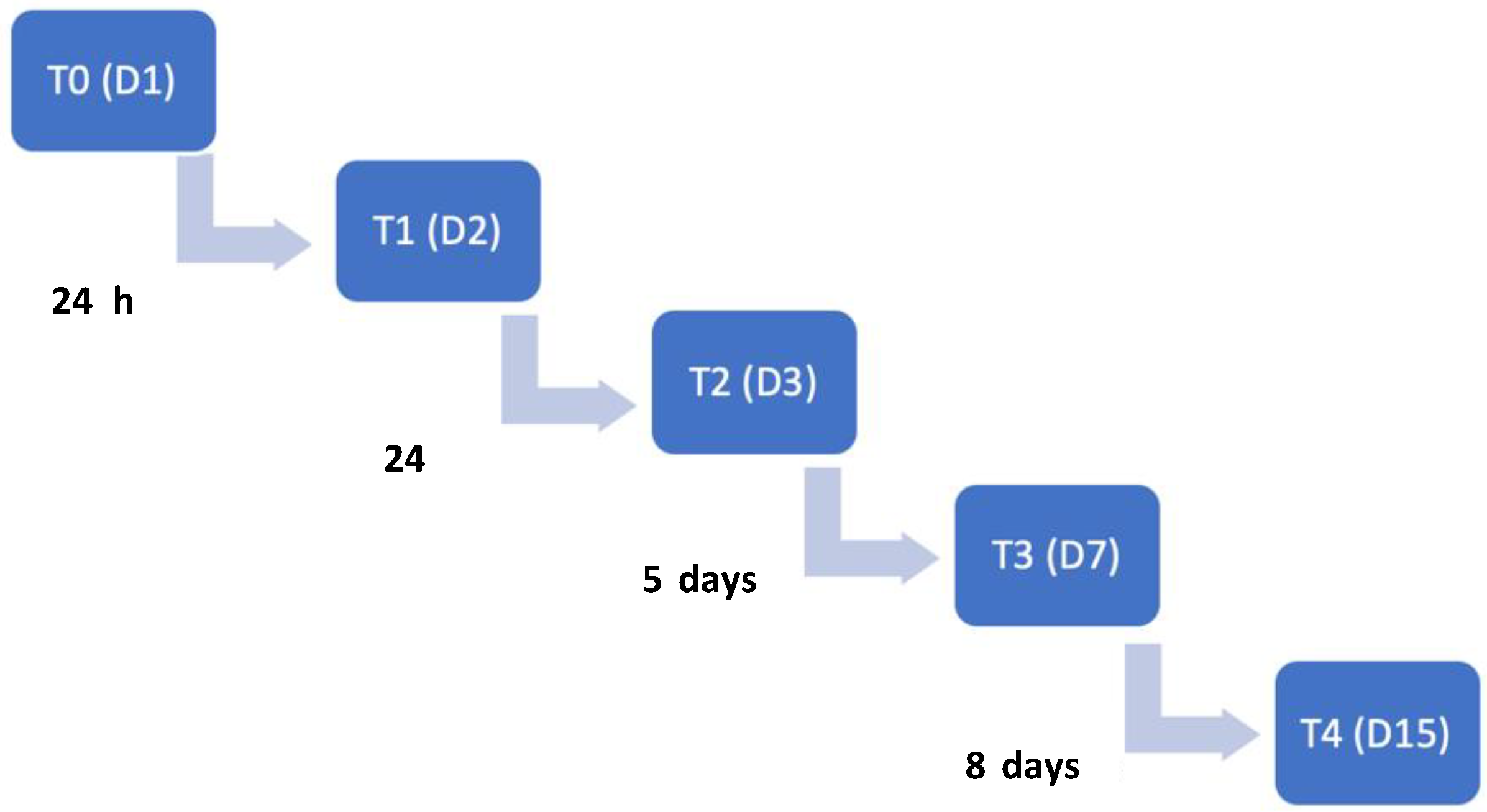

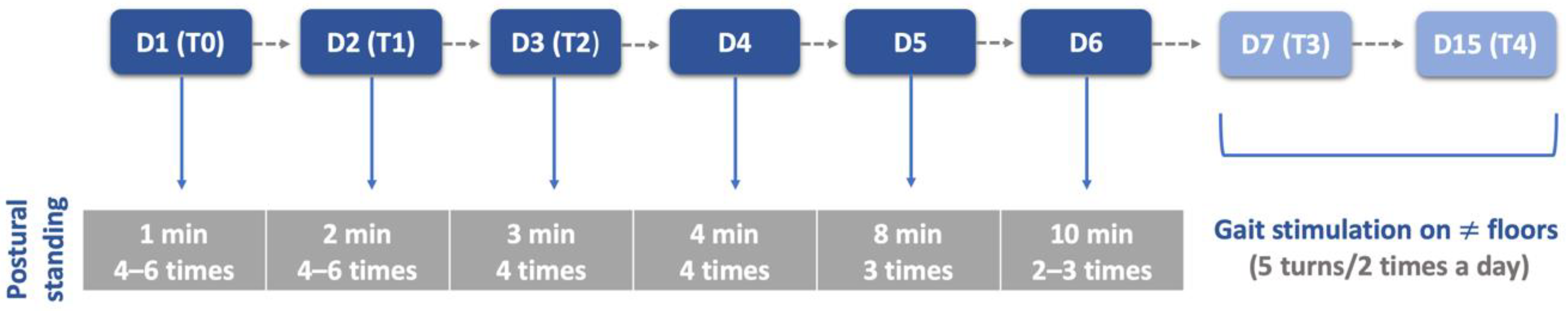
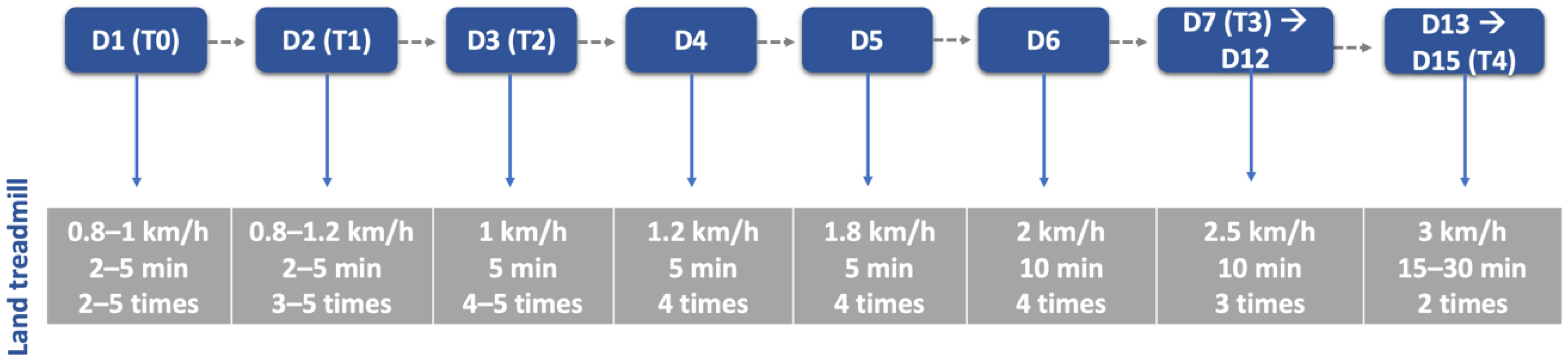
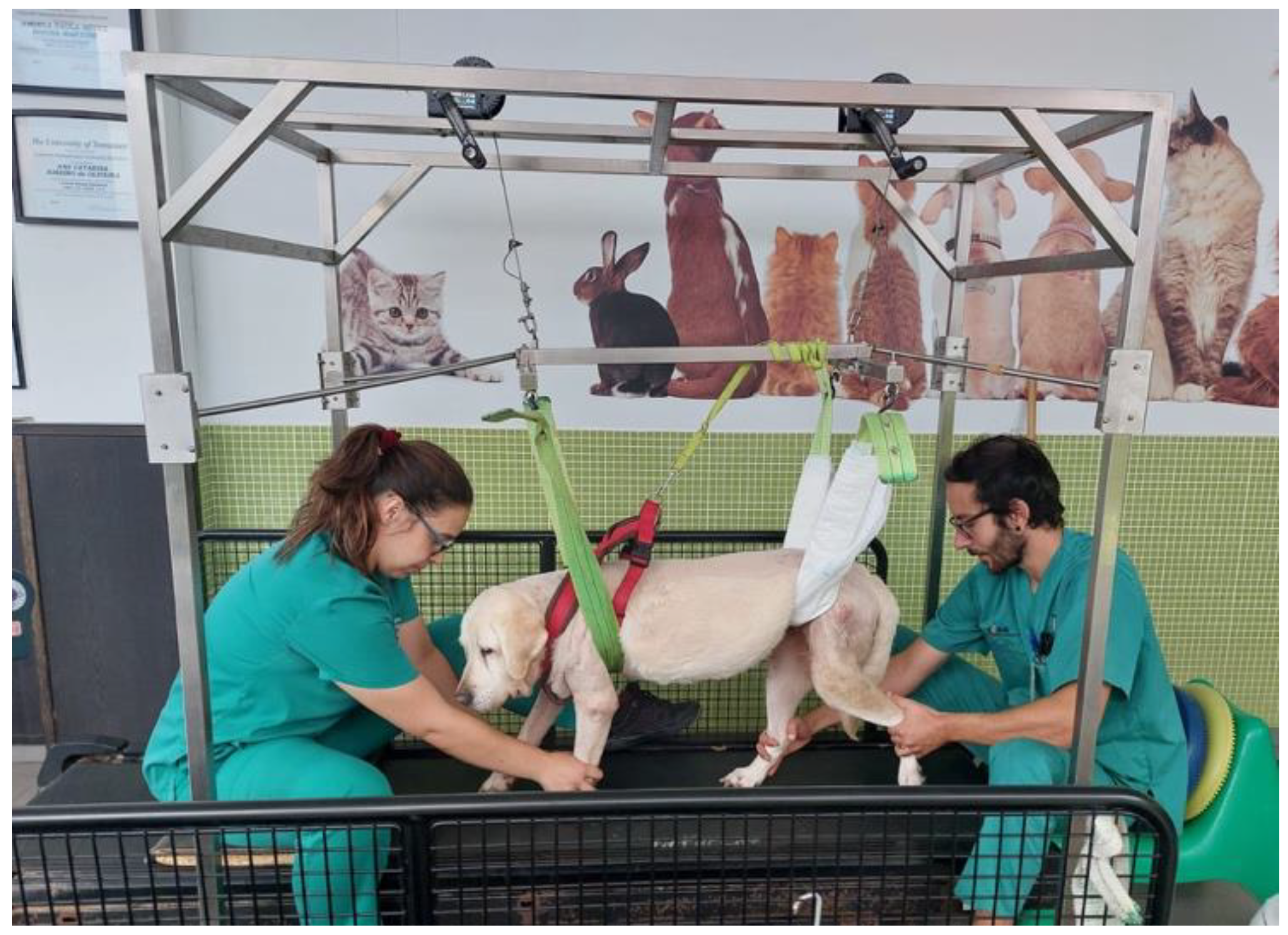


| Characterization Parameters | SHG (n = 74) | NSHG (n = 40) | Total (n = 114) |
|---|---|---|---|
| Breed | Breed: 63/74 (85.1%) Mixed breed: 11/74 (14.9%) | Breed: 23/40 (57.5%) Mixed breed: 17/40 (42.5%) | Breed: 86/114 (75.4%) Mixed breed: 28/114 (24.6%) |
| Chondrodystrophy | Present: 29/74 (39.2%) Absent: 45/74 (60.8%) | Present: 6/40 (15%) Absent: 34/40 (85%) | Present: 35/114 (30.7%) Absent: 79/114 (69.3%) |
| Sex | Male: 52/74 (70.3%) Female: 22/74 (29.7%) | Male: 17/40 (42.5%) Female: 23/40 (57.5%) | Male: 69/114 (60.5%) Female: 45/114 (39.5%) |
| Age | <7 years: 24/74 (32.4%) ≥7 years: 50/74 (67.6%) Mean: 7.78 years | <7 years: 17/40 (42.5%) ≥7 years: 23/40 (57.5%) Mean: 8.07 years | <7 years: 41/114 (36%) ≥7 years: 73/114 (64%) Mean: 7.89 years |
| Weight | ≤10 kg: 24/74 (32.4%) >10 kg: 50/74 (67.6%) Mean: 22.07 kg | ≤10 kg: 7/40 (17.5%) >10 kg: 33/40 (82.5%) Mean: 18.23 kg | ≤10 kg: 31/114 (27.2%) >10 kg: 83/114 (72.8%) Mean: 20.72 kg |
| Neuro-location | C1–C2: 8/74 (10.8%) C2–C3: 9/74 (12.2%) C3–C4: 15/74 (20.3%) C4–C5: 42/74 (56.8%) | C2–C3: 12/40 (30%) C3–C4: 16/40 (40%) C4–C5: 12/40 (30%) | C1–C2: 8/114 (7%) C2–C3: 21/114 (18.4%) C3–C4: 31/114 (27.2%) C4–C5: 54/114 (47.4%) |
| Spinal hyperesthesia | Present: 74/74 (100%) | Absent: 40/40 (100%) | Absent: 40/114 (35.1%) Present: 74/114 (64.9%) |
| Sternal recumbency | Absent: 74/74 (100%) | Absent: 40/40 (100%) | Absent: 114/114 (100%) |
| Postural standing | |||
| Neurogenic bladder | Absent: 17/74 (23%) Present: 57/74 (77%) | Absent: 6/40 (15%) Present: 34/40 (85%) | Absent: 23/114 (20.2%) Present: 91/114 (79.8%) |
| Neck movement | Absent: 26/74 (35.1%) Present: 48/74 (64.9%) | Present: 40/40 (100%) | Absent: 26/114 (22.8%) Present: 88/114 (77.2%) |
| Flexor reflex (forelimbs) | Present: 68/74 (91.9%) Absent: 6/74 (8.1%) | Present: 37/40 (92.5%) Absent: 3/40 (7.5%) | Present: 105/114 (92.1%) Absent: 9/114 (7.9%) |
| Flexor reflex (hindlimbs) | Present: 68/74 (91.9%) Absent: 6/74 (8.1%) | Present: 39/40 (97.5%) Absent: 1/40 (2.5%) | Present: 107/114 (93.9%) Absent: 7/114 (6.1%) |
| Ambulation | Absent: 74/74 (100%) | Absent: 40/40 (100%) | Absent: 114/114 (100%) |
| Placing (fore and hindlimbs) | |||
| Normal flexor muscle group tonus | Abnormal: 74/74 (100%) | Abnormal: 40/40 (100%) | Abnormal: 114/114 (100%) |
| Normal extensor muscle group tonus | Normal: 68/74 (91.9%) Abnormal: 6/74 (8.1%) | Normal: 39/40 (97.5%) Abnormal: 1/40 (2.5%) | Normal: 107/114 (93.9%) Abnormal: 7/114 (6.1%) |
| Pressure sores | Absent: 74/74 (100%) | Absent: 40/40 (100%) | Absent: 114/114 (100%) |
| Material | Land treadmill; Underwater treadmill; Passive standing device; Harness; Four support straps; Cavaletti rail; Trampoline; Ramps; Different floor surfaces. |
| Rehabilitation Team | Dogs ≤ 10 kg: One technician (Two in the first week); Dogs > 10 kg: Two technicians (Four in the first week). |
| Time Points | T0 | T1 | T2 | T3 | T4 |
|---|---|---|---|---|---|
| Postural standing (30 s) | |||||
| Absence of spinal hyperesthesia (palpation of the vertebral column from C1–T2) | |||||
| Normal neck movement | |||||
| Normal flexor reflex (forelimbs and hindlimbs) | |||||
| Normal placing test (forelimbs and hindlimbs) | |||||
| Ability to perform until 10 steps without falling (OFS ≥ 11) | |||||
| Ability to perform ≥ 10 steps in different floors (OFS 13/14) | |||||
| Normal muscle tone (palpation of the extensor muscles) | |||||
| Normal muscle tone (palpation of the flexor muscles) | |||||
| Sternal recumbency | |||||
| OFS evaluation | |||||
| Pressure sores | |||||
| Neurogenic bladder |
| SHG (n = 74) | NSHG (n = 40) | Total (n = 114) | ||
|---|---|---|---|---|
| Age (years) | Mean | 7.78 | 8.07 | 7.89 |
| Median | 8 | 8 | 8 | |
| Variance | 11.514 | 14.533 | 12.474 | |
| SD | 3.393 | 3.812 | 3.532 | |
| Minimum | 1 | 1 | 1 | |
| Maximum | 14 | 16 | 16 | |
| SEM | 0.394 | 0.603 | 0.331 | |
| Normality Test | 0.009 (Kolmogorov–Smirnov) | 0.587 (Shapiro–Wilk) | 0.015 (Kolmogorov–Smirnov) | |
| Weight (kg) | Mean | 22.07 | 18.23 | 20.72 |
| Median | 21.5 | 19 | 20.5 | |
| Variance | 183.571 | 139.82 | 170.239 | |
| SD | 13.549 | 11.825 | 13.048 | |
| Minimum | 3 | 3 | 3 | |
| Maximum | 62 | 45 | 62 | |
| SEM | 1.575 | 1.87 | 1.222 | |
| Normality Test | 0.066 (Kolmogorov–Smirnov) | 0.019 (Shapiro–Wilk) | 0.033 (Kolmogorov–Smirnov) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gouveia, D.; Carvalho, C.; Cardoso, A.; Gamboa, Ó.; Almeida, A.; Ferreira, A.; Martins, Â. Early Locomotor Training in Tetraplegic Post-Surgical Dogs with Cervical Intervertebral Disc Disease. Animals 2022, 12, 2369. https://doi.org/10.3390/ani12182369
Gouveia D, Carvalho C, Cardoso A, Gamboa Ó, Almeida A, Ferreira A, Martins Â. Early Locomotor Training in Tetraplegic Post-Surgical Dogs with Cervical Intervertebral Disc Disease. Animals. 2022; 12(18):2369. https://doi.org/10.3390/ani12182369
Chicago/Turabian StyleGouveia, Débora, Carla Carvalho, Ana Cardoso, Óscar Gamboa, António Almeida, António Ferreira, and Ângela Martins. 2022. "Early Locomotor Training in Tetraplegic Post-Surgical Dogs with Cervical Intervertebral Disc Disease" Animals 12, no. 18: 2369. https://doi.org/10.3390/ani12182369
APA StyleGouveia, D., Carvalho, C., Cardoso, A., Gamboa, Ó., Almeida, A., Ferreira, A., & Martins, Â. (2022). Early Locomotor Training in Tetraplegic Post-Surgical Dogs with Cervical Intervertebral Disc Disease. Animals, 12(18), 2369. https://doi.org/10.3390/ani12182369





