A Review of Non-Invasive Sampling in Wildlife Disease and Health Research: What’s New?
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
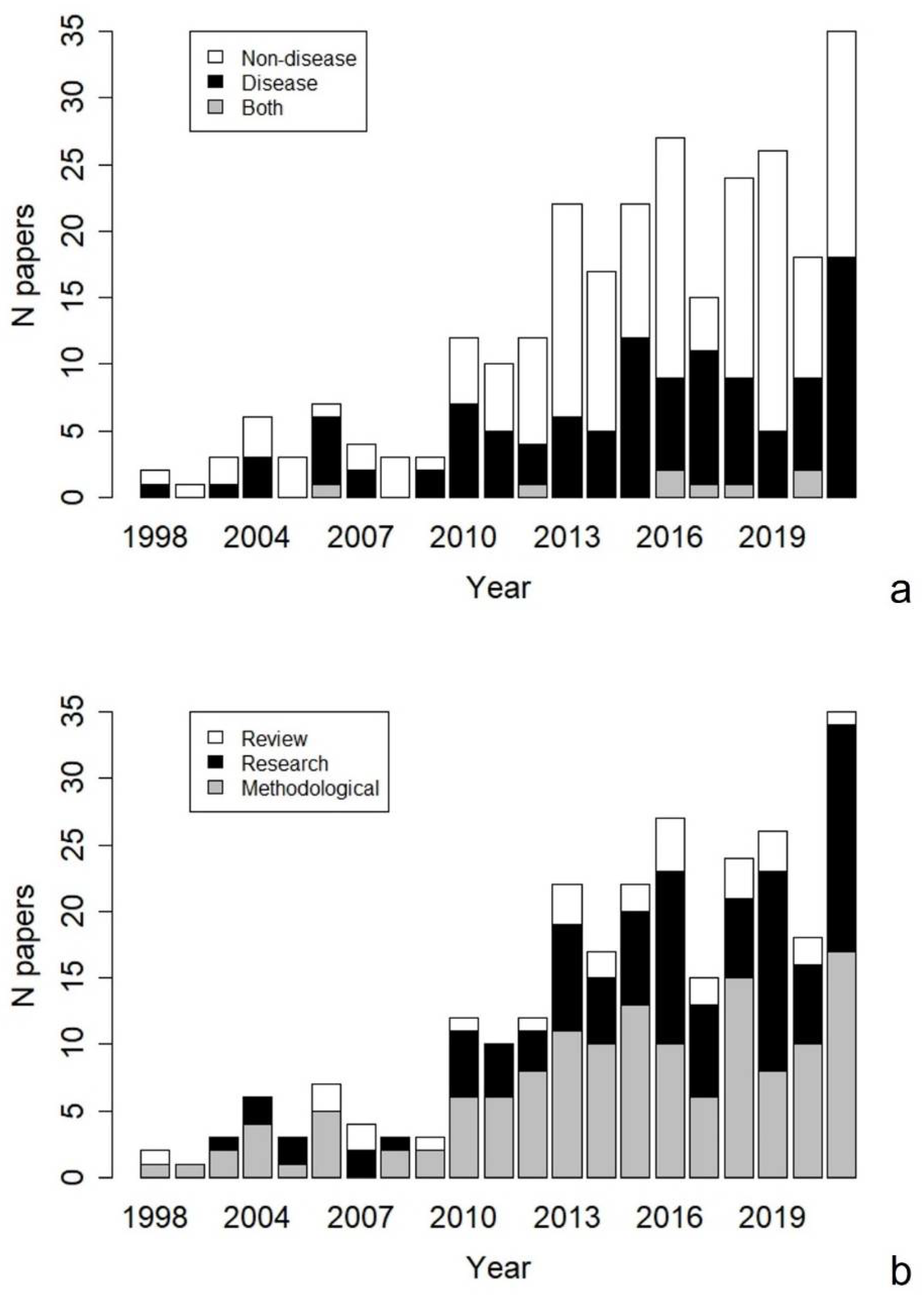
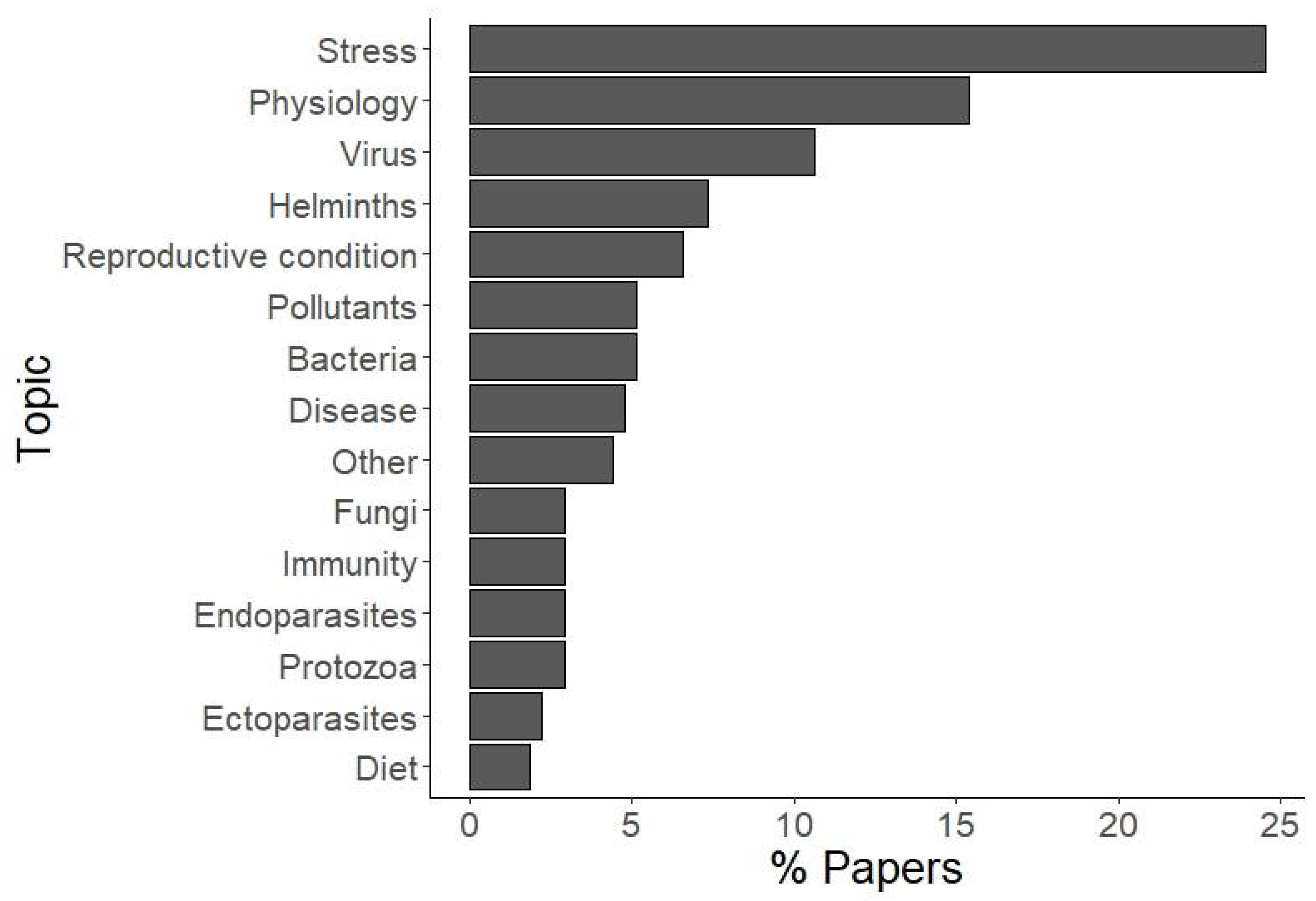
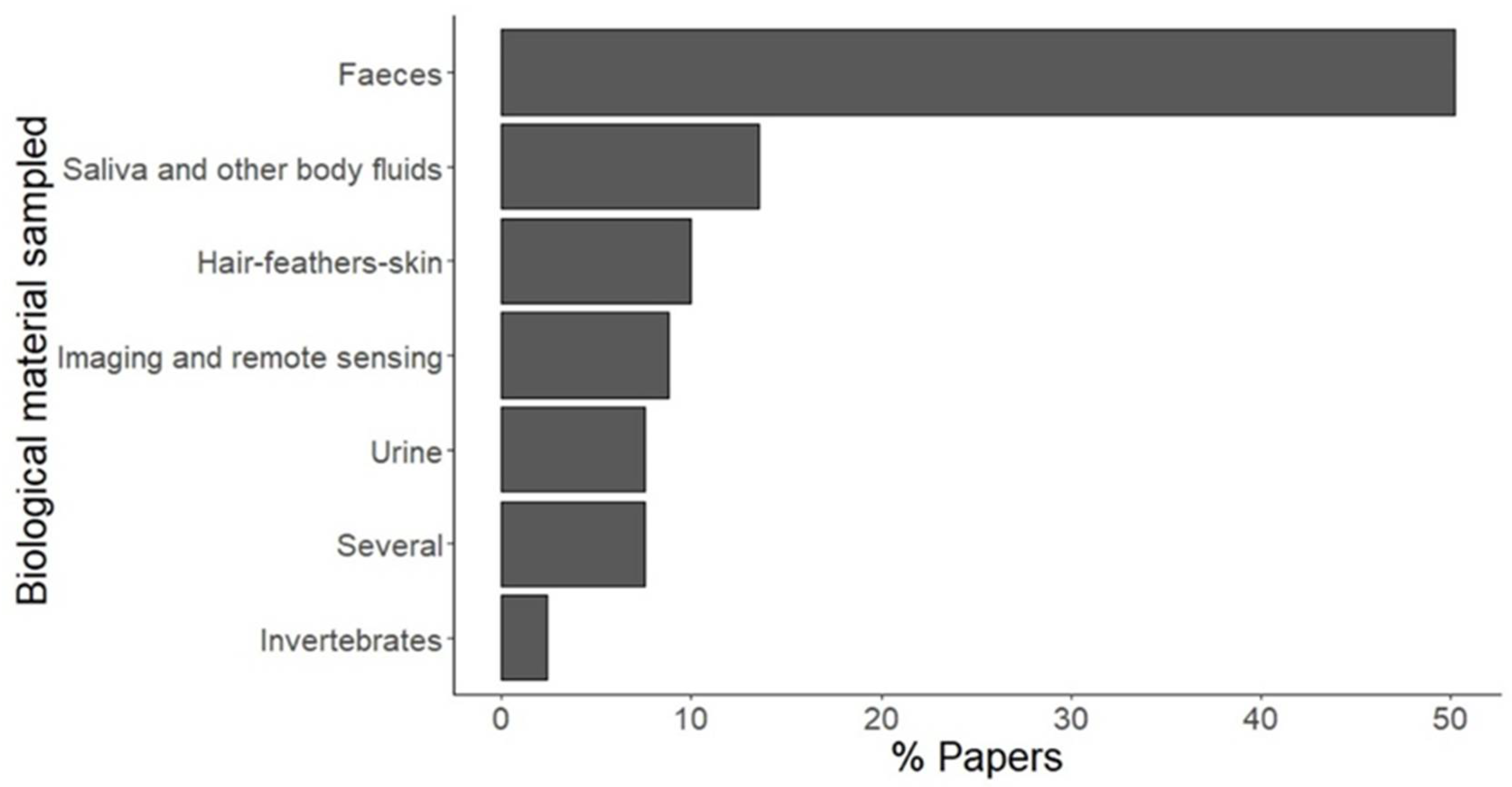
3. Results
4. Discussion
4.1. Faeces
4.2. Urine
4.3. Saliva and Other Body Fluids
4.4. Hair, Feathers, and Skin
4.5. Imaging and Remote Sensing
4.6. Other
4.7. Additional Considerations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Destoumieux-Garzón, D.; Mavingui, P.; Boetsch, G.; Boissier, J.; Darriet, F.; Duboz, P.; Fritsch, C.; Giraudoux, P.; Le Roux, F.; Morand, S.; et al. The One Health Concept: 10 Years Old and a Long Road Ahead. Front. Vet. Sci. 2018, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Watsa, M. Wildlife Disease Surveillance Focus Group Rigorous Wildlife Disease Surveillance. Science 2020, 369, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Blehert, D.S.; Hicks, A.C.; Behr, M.; Meteyer, C.U.; Berlowski-Zier, B.M.; Buckles, E.L.; Coleman, J.T.H.; Darling, S.R.; Gargas, A.; Niver, R.; et al. Bat White-Nose Syndrome: An Emerging Fungal Pathogen? Science 2009, 323, 227. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.C.; Garner, T.W.J.; Walker, S.F. Global Emergence of Batrachochytrium dendrobatidis and Amphibian chytridiomycosis in Space, Time, and Host. Annu. Rev. Microbiol. 2009, 63, 291–310. [Google Scholar] [CrossRef]
- Quigley, B.L.; Timms, P. Helping Koalas Battle Disease—Recent Advances in Chlamydia and Koala Retrovirus (KoRV) Disease Understanding and Treatment in Koalas. FEMS Microbiol. Rev. 2020, 44, 583–605. [Google Scholar] [CrossRef]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Emerging Infectious Diseases of Wildlife–Threats to Biodiversity and Human Health. Science 2000, 287, 443–449. [Google Scholar] [CrossRef]
- Thompson, R.C.A.; Lymbery, A.J.; Smith, A. Parasites, Emerging Disease and Wildlife Conservation. Int. J. Parasitol. 2010, 40, 1163–1170. [Google Scholar] [CrossRef]
- Beldomenico, P.M.; Begon, M. Disease Spread, Susceptibility and Infection Intensity: Vicious Circles? Trends Ecol. Evol. 2010, 25, 21–27. [Google Scholar] [CrossRef]
- Hawley, D.M.; Altizer, S.M. Disease Ecology Meets Ecological Immunology: Understanding the Links between Organismal Immunity and Infection Dynamics in Natural Populations. Funct. Ecol. 2011, 25, 48–60. [Google Scholar] [CrossRef]
- Pedersen, A.B.; Babayan, S.A. Wild Immunology. Mol. Ecol. 2011, 20, 872–880. [Google Scholar] [CrossRef]
- Hing, S.; Narayan, E.J.; Thompson, R.C.A.; Godfrey, S.S.; Hing, S.; Narayan, E.J.; Thompson, R.C.A.; Godfrey, S.S. The Relationship between Physiological Stress and Wildlife Disease: Consequences for Health and Conservation. Wildl. Res. 2016, 43, 51–60. [Google Scholar] [CrossRef]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Universities Federation for Animal Welfare: Wheathampstaed, UK, 1959. [Google Scholar]
- The European Parliament; The Council of The European Union. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the Protection of Animals Used for Scientific Purposes Text with EEA Relevance. Off. J. Eur. Union 2010, 276, 32010L0063. [Google Scholar]
- Lindsjö, J.; Fahlman, Å.; Törnqvist, E. Animal Welfare from Mouse to Moose—Implementing the Principles of the 3Rs in Wildlife Research. J. Wildl. Dis. 2016, 52, S65–S77. [Google Scholar] [CrossRef] [PubMed]
- Field, K.A.; Paquet, P.C.; Artelle, K.; Proulx, G.; Brook, R.K.; Darimont, C.T. Publication Reform to Safeguard Wildlife from Researcher Harm. PLoS Biol. 2019, 17, e3000193. [Google Scholar] [CrossRef] [PubMed]
- Zemanova, M.A. Towards More Compassionate Wildlife Research through the 3Rs Principles: Moving from Invasive to Non-Invasive Methods. Wildl. Biol. 2020, 2020, wlb.00607. [Google Scholar] [CrossRef]
- Fusillo, R.; Romanucci, M.; Marcelli, M.; Massimini, M.; Della Salda, L. Health and Mortality Monitoring in Threatened Mammals: A First Post Mortem Study of Otters (Lutra lutra L.) in Italy. Animals 2022, 12, 609. [Google Scholar] [CrossRef]
- Rohner, S.; Wohlsein, P.; Prenger-Berninghoff, E.; Ewers, C.; Waindok, P.; Strube, C.; Baechlein, C.; Becher, P.; Wilmes, D.; Rickerts, V.; et al. Pathological Findings in Eurasian Otters (Lutra lutra) Found Dead between 2015−2020 in Schleswig-Holstein, Germany. Animals 2022, 12, 59. [Google Scholar] [CrossRef]
- Shuttleworth, C.M.; Everest, D.; Holmes, P.; Bell, S.; Cripps, R. An Opportunistic Assessment of the Impact of Squirrelpox Disease Outbreaks upon a Red Squirrel Population Sympatric with Grey Squirrels in Wales. Animals 2022, 12, 99. [Google Scholar] [CrossRef]
- Vengušt, G.; Kuhar, U.; Jerina, K.; Švara, T.; Gombač, M.; Bandelj, P.; Vengušt, D.Ž. Passive Disease Surveillance of Alpine Chamois (Rupicapra r. rupicapra) in Slovenia between 2000 and 2020. Animals 2022, 12, 1119. [Google Scholar] [CrossRef]
- Nusser, S.M.; Clark, W.R.; Otis, D.L.; Huang, L. Sampling Considerations for Disease Surveillance in Wildlife Populations. J. Wildl. Manag. 2008, 72, 52–60. [Google Scholar] [CrossRef]
- Lachish, S.; Murray, K.A. The Certainty of Uncertainty: Potential Sources of Bias and Imprecision in Disease Ecology Studies. Front. Vet. Sci. 2018, 5, 90. [Google Scholar] [CrossRef] [PubMed]
- Ryser-Degiorgis, M.-P. Wildlife Health Investigations: Needs, Challenges and Recommendations. BMC Vet. Res. 2013, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.A.; May, R.M. Taxonomic Bias in Conservation Research. Science 2002, 297, 191–192. [Google Scholar] [CrossRef] [PubMed]
- Pyšek, P.; Richardson, D.M.; Pergl, J.; Jarošík, V.; Sixtová, Z.; Weber, E. Geographical and Taxonomic Biases in Invasion Ecology. Trends Ecol. Evol. 2008, 23, 237–244. [Google Scholar] [CrossRef]
- Meyer, C.; Kreft, H.; Guralnick, R.; Jetz, W. Global Priorities for an Effective Information Basis of Biodiversity Distributions. Nat. Commun. 2015, 6, 8221. [Google Scholar] [CrossRef]
- Rosenthal, M.F.; Gertler, M.; Hamilton, A.D.; Prasad, S.; Andrade, M.C.B. Taxonomic Bias in Animal Behaviour Publications. Anim. Behav. 2017, 127, 83–89. [Google Scholar] [CrossRef]
- Han, B.A.; Kramer, A.M.; Drake, J.M. Global Patterns of Zoonotic Disease in Mammals. Trends Parasitol. 2016, 32, 565–577. [Google Scholar] [CrossRef]
- Plourde, B.T.; Burgess, T.L.; Eskew, E.A.; Roth, T.M.; Stephenson, N.; Foley, J.E. Are Disease Reservoirs Special? Taxonomic and Life History Characteristics. PLoS ONE 2017, 12, e0180716. [Google Scholar] [CrossRef]
- Hoby, S.; Schwarzenberger, F.; Doherr, M.G.; Robert, N.; Walzer, C. Steroid Hormone Related Male Biased Parasitism in Chamois, Rupicapra Rupicapra Rupicapra. Vet. Parasitol. 2006, 138, 337–348. [Google Scholar] [CrossRef]
- Stringer, A.P.; Smith, D.; Kerley, G.I.H.; Linklater, W.L. Reducing Sampling Error in Faecal Egg Counts from Black Rhinoceros (Diceros bicornis). Int. J. Parasitol. Parasites Wildl. 2014, 3, 1–5. [Google Scholar] [CrossRef][Green Version]
- Catalano, S.; Symeou, A.; Marsh, K.J.; Borlase, A.; Léger, E.; Fall, C.B.; Sène, M.; Diouf, N.D.; Ianniello, D.; Cringoli, G.; et al. Mini-FLOTAC as an Alternative, Non-Invasive Diagnostic Tool for Schistosoma Mansoni and Other Trematode Infections in Wildlife Reservoirs. Parasit. Vectors 2019, 12, 439. [Google Scholar] [CrossRef] [PubMed]
- Carrau, T.; Martínez-Carrasco, C.; Garijo, M.M.; Alonso, F.; de Ybáñez, R.R.; Tizzani, P. Evaluation of the Baermann–Wetzel Method for Detecting Lungworm Larvae in Wild Ruminants from Faecal Samples. J. Helminthol. 2021, 95. [Google Scholar] [CrossRef] [PubMed]
- Liccioli, S.; Rogers, S.; Greco, C.; Kutz, S.J.; Chan, F.; Ruckstuhl, K.E.; Massolo, A. Assessing Individual Patterns of Echinococcus Multilocularis Infection in Urban Coyotes: Non-Invasive Genetic Sampling as an Epidemiological Tool. J. Appl. Ecol. 2015, 52, 434–442. [Google Scholar] [CrossRef]
- Al-Sabi, M.N.S.; Deplazes, P.; Webster, P.; Willesen, J.L.; Davidson, R.K.; Kapel, C.M.O. PCR Detection of Angiostrongylus vasorum in Faecal Samples of Dogs and Foxes. Parasitol. Res. 2010, 107, 135–140. [Google Scholar] [CrossRef]
- Poglayen, G.; Gori, F.; Morandi, B.; Galuppi, R.; Fabbri, E.; Caniglia, R.; Milanesi, P.; Galaverni, M.; Randi, E.; Marchesi, B.; et al. Italian Wolves (Canis lupus italicus altobello, 1921) and Molecular Detection of Taeniids in the Foreste Casentinesi National Park, Northern Italian Apennines. Int. J. Parasitol. Parasites Wildl. 2017, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Solórzano-García, B.; Pérez-Ponce de León, G. Helminth Parasites of Howler and Spider Monkeys in Mexico: Insights into Molecular Diagnostic Methods and Their Importance for Zoonotic Diseases and Host Conservation. Int. J. Parasitol. Parasites Wildl. 2017, 6, 76–84. [Google Scholar] [CrossRef]
- Bertram, M.R.; Hamer, G.L.; Snowden, K.F.; Hartup, B.K.; Hamer, S.A. Coccidian Parasites and Conservation Implications for the Endangered Whooping Crane (Grus americana). PLoS ONE 2015, 10, e0127679. [Google Scholar] [CrossRef]
- Teixeira, W.F.P.; De Oliveira, M.L.; de Faria Peres, P.H.; Nagata, W.B.; Santana, B.N.; Oliveira, B.C.M.; Duarte, J.M.B.; Cardoso, T.C.; Lopes, W.D.Z.; Bresciani, K.D.S. Cryptosporidium Parvum in Brown Brocket (Mazama gouazoubira) from Brazil: First Report of the Subtype IIaA16G3R1 in Cervids. Parasitol. Int. 2021, 80, 102216. [Google Scholar] [CrossRef]
- Aivelo, T.; Medlar, A. Opportunities and Challenges in Metabarcoding Approaches for Helminth Community Identification in Wild Mammals. Parasitology 2018, 145, 608–621. [Google Scholar] [CrossRef]
- De Carvalho Ferreira, H.C.; Weesendorp, E.; Quak, S.; Stegeman, J.A.; Loeffen, W.L.A. Suitability of Faeces and Tissue Samples as a Basis for Non-Invasive Sampling for African Swine Fever in Wild Boar. Vet. Microbiol. 2014, 172, 449–454. [Google Scholar] [CrossRef]
- Bandoo, R.A.; Bautista, J.; Lund, M.; Newkirk, E.; Squires, J.; Varsani, A.; Kraberger, S. Identification of Novel Circovirus and Anelloviruses from Wolverines Using a Non-Invasive Faecal Sampling Approach. Infect. Genet. Evol. 2021, 93, 104914. [Google Scholar] [CrossRef] [PubMed]
- Hardmeier, I.; Aeberhard, N.; Qi, W.; Schoenbaechler, K.; Kraettli, H.; Hatt, J.-M.; Fraefel, C.; Kubacki, J. Metagenomic Analysis of Fecal and Tissue Samples from 18 Endemic Bat Species in Switzerland Revealed a Diverse Virus Composition Including Potentially Zoonotic Viruses. PLoS ONE 2021, 16, e0252534. [Google Scholar] [CrossRef] [PubMed]
- Wood, R.; Bangura, U.; Mariën, J.; Douno, M.; Fichet-Calvet, E. Detection of Lassa Virus in Wild Rodent Feces: Implications for Lassa Fever Burden within Households in the Endemic Region of Faranah, Guinea. One Health 2021, 13, 100317. [Google Scholar] [CrossRef]
- Bataille, A.; Kwiatek, O.; Belfkhi, S.; Mounier, L.; Parida, S.; Mahapatra, M.; Caron, A.; Chubwa, C.C.; Keyyu, J.; Kock, R.; et al. Optimization and Evaluation of a Non-Invasive Tool for Peste Des Petits Ruminants Surveillance and Control. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.C.; Hannaoui, S.; John, T.R.; Dudas, S.; Czub, S.; Gilch, S. Early and Non-Invasive Detection of Chronic Wasting Disease Prions in Elk Feces by Real-Time Quaking Induced Conversion. PLoS ONE 2016, 11, e0166187. [Google Scholar] [CrossRef] [PubMed]
- Fujita, S.; Kageyama, T. Polymerase Chain Reaction Detection of Clostridium perfringens in Feces from Captive and Wild Chimpanzees, Pan Troglodytes. J. Med. Primatol. 2007, 36, 25–32. [Google Scholar] [CrossRef]
- Keita, A.K.; Socolovschi, C.; Ahuka-Mundeke, S.; Ratmanov, P.; Butel, C.; Ayouba, A.; Inogwabini, B.-I.; Muyembe-Tamfum, J.-J.; Mpoudi-Ngole, E.; Delaporte, E.; et al. Molecular Evidence for the Presence of Rickettsia felis in the Feces of Wild-Living African Apes. PLoS ONE 2013, 8, e54679. [Google Scholar] [CrossRef]
- Smith, A.; Ismail, H.; Henton, M.; Keddy, K. GERMS-SA Surveillance Network Similarities between Salmonella enteritidis Isolated from Humans and Captive Wild Animals in South Africa. J. Infect. Dev. Ctries. 2014, 8, 1615–1619. [Google Scholar] [CrossRef]
- Grant, D.; Todd, P.A.; Pennycott, T. Monitoring Wild Greenfinch (Carduelis chloris) for Salmonella enterica typhimurium. Ecol. Res. 2007, 22, 571–574. [Google Scholar] [CrossRef]
- King, H.C.; Murphy, A.; James, P.; Travis, E.; Porter, D.; Sawyer, J.; Cork, J.; Delahay, R.J.; Gaze, W.; Courtenay, O.; et al. Performance of a Noninvasive Test for Detecting Mycobacterium bovis Shedding in European Badger (Meles meles) Populations. J. Clin. Microbiol. 2015, 53, 2316–2323. [Google Scholar] [CrossRef][Green Version]
- Knutie, S.A.; Gotanda, K.M. A Non-Invasive Method to Collect Fecal Samples from Wild Birds for Microbiome Studies. Microb. Ecol. 2018, 76, 851–855. [Google Scholar] [CrossRef] [PubMed]
- Wolf, J.F.; Kriss, K.D.; MacAulay, K.M.; Munro, K.; Patterson, B.R.; Shafer, A.B.A. Gut Microbiome Composition Predicts Summer Core Range Size in Two Divergent Ungulates. FEMS Microbiol. Ecol. 2021, 97, fiab048. [Google Scholar] [CrossRef] [PubMed]
- Schaumburg, F.; Mugisha, L.; Kappeller, P.; Fichtel, C.; Köck, R.; Köndgen, S.; Becker, K.; Boesch, C.; Peters, G.; Leendertz, F. Evaluation of Non-Invasive Biological Samples to Monitor Staphylococcus aureus Colonization in Great Apes and Lemurs. PLoS ONE 2013, 8, e78046. [Google Scholar] [CrossRef]
- Palme, R.; Touma, C.; Arias, N.; Dominchin, M.F.; Lepschy, M. Steroid Extraction: Get the Best out of Faecal Samples. Wien. Tierärztl. Monatsschrift 2013, 9, 238–246. [Google Scholar]
- Behringer, V.; Deimel, C.; Hohmann, G.; Negrey, J.; Schaebs, F.S.; Deschner, T. Applications for Non-Invasive Thyroid Hormone Measurements in Mammalian Ecology, Growth, and Maintenance. Horm. Behav. 2018, 105, 66–85. [Google Scholar] [CrossRef]
- Sheriff, M.J.; Dantzer, B.; Delehanty, B.; Palme, R.; Boonstra, R. Measuring Stress in Wildlife: Techniques for Quantifying Glucocorticoids. Oecologia 2011, 166, 869–887. [Google Scholar] [CrossRef]
- Gesquiere, L.R.; Ziegler, T.E.; Chen, P.A.; Epstein, K.A.; Alberts, S.C.; Altmann, J. Measuring Fecal Testosterone in Females and Fecal Estrogens in Males: Comparison of RIA and LC/MS/MS Methods for Wild Baboons (Papio cynocephalus). Gen. Comp. Endocrinol. 2014, 204, 141–149. [Google Scholar] [CrossRef]
- Touma, C.; Palme, R. Measuring Fecal Glucocorticoid Metabolites in Mammals and Birds: The Importance of Validation. Ann. N. Y. Acad. Sci. 2005, 1046, 54–74. [Google Scholar] [CrossRef]
- Laver, P.N.; Ganswindt, A.; Ganswindt, S.B.; Alexander, K.A. Non-Invasive Monitoring of Glucocorticoid Metabolites in Banded Mongooses (Mungos mungo) in Response to Physiological and Biological Challenges. Gen. Comp. Endocrinol. 2012, 179, 178–183. [Google Scholar] [CrossRef]
- Shutt, K.; Setchell, J.M.; Heistermann, M. Non-Invasive Monitoring of Physiological Stress in the Western Lowland Gorilla (Gorilla gorilla gorilla): Validation of a Fecal Glucocorticoid Assay and Methods for Practical Application in the Field. Gen. Comp. Endocrinol. 2012, 179, 167–177. [Google Scholar] [CrossRef]
- Ludwig, C.; Wachter, B.; Silinski-Mehr, S.; Ganswindt, A.; Bertschinger, H.; Hofer, H.; Dehnhard, M. Characterisation and Validation of an Enzyme-Immunoassay for the Non-Invasive Assessment of Faecal Glucocorticoid Metabolites in Cheetahs (Acinonyx jubatus). Gen. Comp. Endocrinol. 2013, 180, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Negrin, A.; Coyohua-Fuentes, A.; Chavira, R.; Canales-Espinosa, D.; Dias, P. Primates Living Outside Protected Habitats Are More Stressed: The Case of Black Howler Monkeys in the Yucatan Peninsula. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Narayan, E. Physiological Stress Levels in Wild Koala Sub-Populations Facing Anthropogenic Induced Environmental Trauma and Disease. Sci. Rep. 2019, 9, 6031. [Google Scholar] [CrossRef]
- Ramahlo, M.; Chimimba, C.; Pirk, C.; Ganswindt, A. Non-Invasive Monitoring of Adrenocortical Activity in Free-Ranging Namaqua Rock Mice Micaelamys namaquensis from South Africa in Response to Anthropogenic Land Use and Season. Wildl. Biol. 2019, 2019, 1–6. [Google Scholar] [CrossRef]
- Navarro-Castilla, Á.; Garrido, M.; Hawlena, H.; Barja, I. Non-Invasive Monitoring of Adrenocortical Activity in Three Sympatric Desert Gerbil Species. Animals 2021, 11, 75. [Google Scholar] [CrossRef] [PubMed]
- Serres-Corral, P.; Fernandez-Bellon, H.; Padilla-Sole, P.; Carbajal, A.; Lopez-Bejar, M. Evaluation of Fecal Glucocorticoid Metabolite Levels in Response to a Change in Social and Handling Conditions in African Lions (Panthera leo bleyenberghi). Animals 2021, 11, 1877. [Google Scholar] [CrossRef] [PubMed]
- Baltic, M.; Jenni-Eiermann, S.; Arlettaz, R.; Palme, R. A Noninvasive Technique to Evaluate Human-Generated Stress in the Black Grouse. Ann. N. Y. Acad. Sci. 2005, 1046, 81–95. [Google Scholar] [CrossRef]
- Gladbach, A.; Gladbach, D.J.; Koch, M.; Kuchar, A.; Möstl, E.; Quillfeldt, P. Can Faecal Glucocorticoid Metabolites Be Used to Monitor Body Condition in Wild Upland Geese Chloephaga picta leucoptera? Behav. Ecol. Sociobiol. 2011, 65, 1491–1498. [Google Scholar] [CrossRef]
- Ozella, L.; Anfossi, L.; Di Nardo, F.; Pessani, D. Non-Invasive Monitoring of Adrenocortical Activity in Captive African Penguin (Spheniscus demersus) by Measuring Faecal Glucocorticoid Metabolites. Gen. Comp. Endocrinol. 2015, 224, 104–112. [Google Scholar] [CrossRef]
- Ganswindt, S.B.; Myburgh, J.G.; Cameron, E.Z.; Ganswindt, A. Non-Invasive Assessment of Adrenocortical Function in Captive Nile Crocodiles (Crocodylus niloticus). Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2014, 177, 11–17. [Google Scholar] [CrossRef]
- Bautista, L.M.; Silván, G.; Cáceres, S.; Martínez-Fernández, L.; Bravo, C.; Illera, J.C.; Alonso, J.C.; Blanco, G. Faecal Sexual Steroids in Sex Typing and Endocrine Status of Great Bustards. Eur. J. Wildl. Res. 2013, 59, 815–822. [Google Scholar] [CrossRef]
- Pribbenow, S.; Wachter, B.; Ludwig, C.; Weigold, A.; Dehnhard, M. Validation of an Enzyme-Immunoassay for the Non-Invasive Monitoring of Faecal Testosterone Metabolites in Male Cheetahs (Acinonyx jubatus). Gen. Comp. Endocrinol. 2016, 228, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Khonmee, J.; Rojanasthien, S.; Thitaram, C.; Sumretprasong, J.; Aunsusin, A.; Chaisongkram, C.; Songsasen, N. Non-Invasive Endocrine Monitoring Indicates Seasonal Variations in Gonadal Hormone Metabolites in Dholes (Cuon alpinus). Conserv. Physiol. 2017, 5, cox001. [Google Scholar] [CrossRef]
- Lemos, L.S.; Olsen, A.; Smith, A.; Chandler, T.E.; Larson, S.; Hunt, K.; Torres, L.G. Assessment of Fecal Steroid and Thyroid Hormone Metabolites in Eastern North Pacific Gray Whales. Conserv. Physiol. 2020, 8, coaa110. [Google Scholar] [CrossRef] [PubMed]
- Paris, M.C.J.; White, A.; Reiss, A.; West, M.; Schwarzenberger, F. Faecal Progesterone Metabolites and Behavioural Observations for the Non-Invasive Assessment of Oestrous Cycles in the Common Wombat (Vombatus ursinus) and the Southern Hairy-Nosed Wombat (Lasiorhinus latifrons). Anim. Reprod. Sci. 2002, 72, 245–257. [Google Scholar] [CrossRef]
- Van der Goot, A.; Dalerum, F.; Ganswindt, A.; Martin, G.; Millar, R.; Paris, M. Faecal Progestagen Profiles in Wild Southern White Rhinoceros (Ceratotherium simum simum). Afr. Zool. 2013, 48, 143–151. [Google Scholar] [CrossRef][Green Version]
- Mayor, P.; Guimaraes, D.A.; da Silva, J.; Jori, F.; Lopez-Bejar, M. Reproductive Monitoring of Collared Peccary Females (Pecari tajacu) by Analysis of Fecal Progesterone Metabolites. Theriogenology 2019, 134, 11–17. [Google Scholar] [CrossRef]
- Mondol, S.; Booth, R.K.; Wasser, S.K. Fecal Stress, Nutrition and Reproductive Hormones for Monitoring Environmental Impacts on Tigers (Panthera tigris). Conserv. Physiol. 2020, 8, coz091. [Google Scholar] [CrossRef]
- Seamster, V.; Waits, L.; Macko, S.; Shugart, H. Coyote (Canis latrans) Mammalian Prey Diet Shifts in Response to Seasonal Vegetation Change. Isotopes Environ. Health Stud. 2014, 50, 343–360. [Google Scholar] [CrossRef]
- Monterroso, P.; Godinho, R.; Oliveira, T.; Ferreras, P.; Kelly, M.J.; Morin, D.J.; Waits, L.P.; Alves, P.C.; Mills, L.S. Feeding Ecological Knowledge: The Underutilised Power of Faecal DNA Approaches for Carnivore Diet Analysis. Mammal Rev. 2019, 49, 97–112. [Google Scholar] [CrossRef]
- O’Rourke, D.R.; Mangan, M.T.; Mangan, K.E.; Bokulich, N.A.; MacManes, M.D.; Foster, J.T. Lord of the Diptera (and Moths and a Spider): Molecular Diet Analyses and Foraging Ecology of Indiana Bats in Illinois. Front. Ecol. Evol. 2021, 9, 12. [Google Scholar] [CrossRef]
- Ando, H.; Mukai, H.; Komura, T.; Dewi, T.; Ando, M.; Isagi, Y. Methodological Trends and Perspectives of Animal Dietary Studies by Noninvasive Fecal DNA Metabarcoding. Environ. DNA 2020, 2, 391–406. [Google Scholar] [CrossRef]
- Sage, M.; Fourel, I.; Cœurdassier, M.; Barrat, J.; Berny, P.; Giraudoux, P. Determination of Bromadiolone Residues in Fox Faeces by LC/ESI-MS in Relationship with Toxicological Data and Clinical Signs after Repeated Exposure. Environ. Res. 2010, 110, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Webster, A.B.; Callealta, F.J.; Ganswindt, A.; Bennett, N.C. A Non-Invasive Assessment of Essential Trace Element Utilization at Different Trophic Levels in African Wildlife. J. Environ. Manage. 2021, 293, 112820. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Anile, S.; Arrabito, C.; Scornavacca, D.; Mazzamuto, M.V.; Gaglio, G.; Otranto, D.; Giannetto, S.; Brianti, E. Survey on Parasitic Infections in Wildcat (Felis silvestris silvestris schreber, 1777) by Scat Collection. Parasitol. Res. 2016, 115, 255–261. [Google Scholar] [CrossRef]
- Stewart, L.; Tort, N.; Meakin, P.; Argudo, J.; Nzuma, R.; Reid, N.; Delahay, R.; Ashford, R.; Montgomery, W.; Grant, I. Development of a Novel Immunochromatographic Lateral Flow Assay Specific for Mycobacterium bovis Cells and Its Application in Combination with Immunomagnetic Separation to Test Badger Faeces. BMC Vet. Res. 2017, 13. [Google Scholar] [CrossRef]
- Lynch, M.A.; Youngflesh, C.; Agha, N.H.; Ottinger, M.A.; Lynch, H.J. Tourism and Stress Hormone Measures in Gentoo Penguins on the Antarctic Peninsula. Polar Biol. 2019, 42, 1299–1306. [Google Scholar] [CrossRef]
- Dib, L.V.; Palmer, J.P.S.; de Souza Carvalho Class, C.; Pinheiro, J.L.; Ramos, R.C.F.; dos Santos, C.R.; Fonseca, A.B.M.; Rodríguez-Castro, K.G.; Gonçalves, C.F.; Galetti, P.M.; et al. Non-Invasive Sampling in Itatiaia National Park, Brazil: Wild Mammal Parasite Detection. BMC Vet. Res. 2020, 16, 295. [Google Scholar] [CrossRef]
- Hou, Z.; Peng, Z.; Ning, Y.; Liu, D.; Chai, H.; Jiang, G. An Initial Coprological Survey of Parasitic Fauna in the Wild Amur Leopard (Panthera pardus orientalis). Integr. Zool. 2020, 15, 375–384. [Google Scholar] [CrossRef]
- Di Francesco, C.E.; Smoglica, C.; Paoletti, B.; Angelucci, S.; Innocenti, M.; Antonucci, A.; Di Domenico, G.; Marsilio, F. Detection of Selected Pathogens in Apennine Wolf (Canis lupus italicus) by a Non-Invasive GPS-Based Telemetry Sampling of Two Packs from Majella National Park, Italy. Eur. J. Wildl. Res. 2019, 65, 84. [Google Scholar] [CrossRef]
- Afonso, E.; Tournant, P.; Foltête, J.-C.; Giraudoux, P.; Baurand, P.-E.; Roué, S.; Canella, V.; Vey, D.; Scheifler, R. Is the Lesser Horseshoe Bat (Rhinolophus hipposideros) Exposed to Causes That May Have Contributed to Its Decline? A Non-Invasive Approach. Glob. Ecol. Conserv. 2016, 8, 123–137. [Google Scholar] [CrossRef]
- Maréchal, L.; Semple, S.; Majolo, B.; MacLarnon, A. Assessing the Effects of Tourist Provisioning on the Health of Wild Barbary Macaques in Morocco. PLoS ONE 2016, 11, e0155920. [Google Scholar] [CrossRef]
- Albery, G.F.; Kenyon, F.; Morris, A.; Morris, S.; Nussey, D.H.; Pemberton, J.M. Seasonality of Helminth Infection in Wild Red Deer Varies between Individuals and between Parasite Taxa. Parasitology 2018, 145, 1410–1420. [Google Scholar] [CrossRef]
- Jepsen, E.M.; Ganswindt, A.; Ngcamphalala, C.A.; Bourne, A.R.; Ridley, A.R.; McKechnie, A.E. Non-Invasive Monitoring of Physiological Stress in an Afrotropical Arid-Zone Passerine Bird, the Southern Pied Babbler. Gen. Comp. Endocrinol. 2019, 276, 60–68. [Google Scholar] [CrossRef]
- Wells, K.; Smales, L.R.; Kalko, E.K.V.; Pfeiffer, M. Impact of Rain-Forest Logging on Helminth Assemblages in Small Mammals (Muridae, Tupaiidae) from Borneo. J. Trop. Ecol. 2007, 23, 35–43. [Google Scholar] [CrossRef]
- Foreyt, W.J. Veterinary Parasitology Reference Manual; John Wiley & Sons: Hoboken, NJ, USA, 2013; ISBN 9781118682265. [Google Scholar]
- Biswas, S.; Bhatt, S.; Paul, S.; Modi, S.; Ghosh, T.; Habib, B.; Nigam, P.; Talukdar, G.; Pandav, B.; Mondol, S. A Practive Faeces Collection Protocol for Multidisciplinary Research in Wildlife Science. Curr. Sci. 2019, 116, 537803. [Google Scholar] [CrossRef]
- Marrero, P.; Fregel, R.; Cabrera, V.M.; Nogales, M. Extraction of High-Quality Host DNA from Feces and Regurgitated Seeds: A Useful Tool for Vertebrate Ecological Studies. Biol. Res. 2009, 42, 147–151. [Google Scholar] [CrossRef]
- Ramón-Laca, A.; Soriano, L.; Gleeson, D.; Godoy, J.A. A Simple and Effective Method for Obtaining Mammal DNA from Faeces. Wildl. Biol. 2015, 21, wlb.00855. [Google Scholar] [CrossRef]
- Santa, M.A.; Pastran, S.; Klein, C.; Ruckstuhl, K.; Massolo, A. Evaluation of an Automated Magnetic Bead-Based DNA Extraction and Real-Time PCR in Fecal Samples as a Pre-Screening Test for Detection of Echinococcus multilocularis and Echinococcus canadensis in Coyotes. Parasitol. Res. 2019, 118, 119–125. [Google Scholar] [CrossRef]
- Fernández-Vizcaíno, E.; Martínez-Carrasco, C.; Moratal, S.; Barroso, P.; Vicente, J. Detection of Stephanurus dentatus in Wild Boar Urine Using Different Parasitological Techniques. Int. J. Parasitol. Parasites Wildl. 2021, 15, 31–34. [Google Scholar] [CrossRef]
- Schneider-Crease, I.; Griffin, R.H.; Gomery, M.A.; Dorny, P.; Noh, J.C.; Handali, S.; Chastain, H.M.; Wilkins, P.P.; Nunn, C.L.; Snyder-Mackler, N.; et al. Identifying Wildlife Reservoirs of Neglected Taeniid Tapeworms: Non-Invasive Diagnosis of Endemic Taenia serialis Infection in a Wild Primate Population. PLoS Negl. Trop. Dis. 2017, 11, e0005709. [Google Scholar] [CrossRef] [PubMed]
- Seidlova, V.; Nemcova, M.; Pikula, J.; Bartonička, T.; Ghazaryan, A.; Heger, T.; Kokurewicz, T.; Orlov, O.L.; Patra, S.; Piacek, V.; et al. Urinary Shedding of Leptospires in Palearctic Bats. Transbound. Emerg. Dis. 2021, 68, 3089–3095. [Google Scholar] [CrossRef]
- Leendertz, F.H.; Boesch, C.; Ellerbrok, H.; Rietschel, W.; Couacy-Hymann, E.; Pauli, G. Non-Invasive Testing Reveals a High Prevalence of Simian T-Lymphotropic Virus Type 1 Antibodies in Wild Adult Chimpanzees of the Taï National Park, Côte d’Ivoire. J. Gen. Virol. 2004, 85, 3305–3312. [Google Scholar] [CrossRef] [PubMed]
- Baker, K.S.; Tachedjian, M.; Barr, J.; Marsh, G.A.; Todd, S.; Crameri, G.; Crameri, S.; Smith, I.; Holmes, C.E.G.; Suu-Ire, R.; et al. Achimota Pararubulavirus 3: A New Bat-Derived Paramyxovirus of the Genus Pararubulavirus. Viruses 2020, 12, 1236. [Google Scholar] [CrossRef] [PubMed]
- Baker, K.S.; Todd, S.; Marsh, G.; Fernandez-Loras, A.; Suu-Ire, R.; Wood, J.L.N.; Wang, L.F.; Murcia, P.R.; Cunningham, A.A.Y. 2012 Co-Circulation of Diverse Paramyxoviruses in an Urban African Fruit Bat Population. J. Gen. Virol. 2012, 93, 850–856. [Google Scholar] [CrossRef]
- Muller, M.N.; Wrangham, R.W. Dominance, Cortisol and Stress in Wild Chimpanzees (Pan troglodytes schweinfurthii). Behav. Ecol. Sociobiol. 2004, 55, 332–340. [Google Scholar] [CrossRef]
- Narayan, E.; Molinia, F.; Christi, K.; Morley, C.; Cockrem, J.; Narayan, E.; Molinia, F.; Christi, K.; Morley, C.; Cockrem, J. Urinary Corticosterone Metabolite Responses to Capture, and Annual Patterns of Urinary Corticosterone in Wild and Captive Endangered Fijian Ground Frogs (Platymantis vitiana). Aust. J. Zool. 2010, 58, 189–197. [Google Scholar] [CrossRef]
- Narayan, E.; Hero, J.-M. Urinary Corticosterone Responses and Haematological Stress Indicators in the Endangered Fijian Ground Frog (Platymantis vitiana) during Transportation and Captivity. Aust. J. Zool. 2011, 59, 79. [Google Scholar] [CrossRef]
- Kindermann, C.; Narayan, E.J.; Hero, J.-M. Urinary Corticosterone Metabolites and Chytridiomycosis Disease Prevalence in a Free-Living Population of Male Stony Creek Frogs (Litoria wilcoxii). Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 2012, 162, 171–176. [Google Scholar] [CrossRef]
- Narayan, E.J. Non-Invasive Reproductive and Stress Endocrinology in Amphibian Conservation Physiology. Conserv. Physiol. 2013, 1, cot011. [Google Scholar] [CrossRef]
- Narayan, E.J.; Gramapurohit, N.P. Sexual Dimorphism in Baseline Urinary Corticosterone Metabolites and Their Association with Body-Condition Indices in a Peri-Urban Population of the Common Asian Toad (Duttaphrynus melanostictus). Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 2016, 191, 174–179. [Google Scholar] [CrossRef]
- Sadoughi, B.; Girard-Buttoz, C.; Engelhardt, A.; Heistermann, M.; Ostner, J. Non-Invasive Assessment of Metabolic Responses to Food Restriction Using Urinary Triiodothyronine and Cortisol Measurement in Macaques. Gen. Comp. Endocrinol. 2021, 306. [Google Scholar] [CrossRef]
- Kinoshita, K.; Miyazaki, M.; Morita, H.; Vassileva, M.; Tang, C.; Li, D.; Ishikawa, O.; Kusunoki, H.; Tsenkova, R. Spectral Pattern of Urinary Water as a Biomarker of Estrus in the Giant Panda. Sci. Rep. 2012, 2, 856. [Google Scholar] [CrossRef]
- Mouri, K.; Shimizu, K. Stability of Chimpanzee (Pan troglodytes) Urinary Reproductive Hormones during Long-Term Preservation on Filter Paper. Primates 2021, 62, 289–296. [Google Scholar] [CrossRef]
- Moscovice, L.R.; Ziegler, T.E. Peripheral Oxytocin in Female Baboons Relates to Estrous State and Maintenance of Sexual Consortships. Horm. Behav. 2012, 62, 592–597. [Google Scholar] [CrossRef]
- Heistermann, M.; Higham, J.P. Urinary Neopterin, a Non-Invasive Marker of Mammalian Cellular Immune Activation, Is Highly Stable under Field Conditions. Sci. Rep. 2015, 5, 16308. [Google Scholar] [CrossRef]
- Behringer, V.; Stevens, J.; Leendertz, F.; Hohmann, G.; Deschner, T. Validation of a Method for the Assessment of Urinary Neopterin Levels to Monitor Health Status in Non-Human-Primate Species. Front. Physiol. 2017, 8, 51. [Google Scholar] [CrossRef]
- Löhrich, T.; Behringer, V.; Wittig, R.M.; Deschner, T.; Leendertz, F.H. The Use of Neopterin as a Noninvasive Marker in Monitoring Diseases in Wild Chimpanzees. EcoHealth 2018, 15, 792–803. [Google Scholar] [CrossRef]
- Wu, D.F.; Behringer, V.; Wittig, R.M.; Leendertz, F.H.; Deschner, T. Urinary Neopterin Levels Increase and Predict Survival during a Respiratory Outbreak in Wild Chimpanzees (Taï National Park, Côte d’Ivoire). Sci. Rep. 2018, 8, 13346. [Google Scholar] [CrossRef]
- Löhrich, T. Health Monitoring in Great Apes: The Use of Neopterin as a Non-Invasive Marker in Monitoring Diseases in Wild Chimpanzees (Pan troglodytes verus). Ph.D. Thesis, Freien Universitat Berlin, Berlin, Germany, 2020. [Google Scholar] [CrossRef]
- Sacco, A.J.; Mayhew, J.A.; Watsa, M.; Erkenswick, G.; Binder, A.K. Detection of Neopterin in the Urine of Captive and Wild Platyrrhines. BMC Zool. 2020, 5, 2. [Google Scholar] [CrossRef]
- Emery Thompson, M.; Knott, C.D. Urinary C-Peptide of Insulin as a Non-Invasive Marker of Energy Balance in Wild Orangutans. Horm. Behav. 2008, 53, 526–535. [Google Scholar] [CrossRef]
- Peterson, R.O.; Hoy, S.R.; Vucetich, L.M.; Vucetich, J.A.; Kraus, V.B.; Huebner, J.L. Urinary Biomarkers to Non-Invasively Evaluate Health in Wild Moose (Alces alces). Osteoarthr. Cartil. 2018, 26, S196–S197. [Google Scholar] [CrossRef]
- Rosenblatt, E.; DeBow, J.; Blouin, J.; Donovan, T.; Murdoch, J.; Creel, S.; Rogers, W.; Gieder, K.; Fortin, N.; Alexander, C. Juvenile Moose Stress and Nutrition Dynamics Related to Winter Ticks, Landscape Characteristics, Climate-Mediated Factors and Survival. Conserv. Physiol. 2021, 9. [Google Scholar] [CrossRef]
- Millspaugh, J.J.; Washburn, B.E.; Milanick, M.A.; Beringer, J.; Hansen, L.P.; Meyer, T.M. Non-Invasive Techniques for Stress Assessment in White-Tailed Deer. Wildl. Soc. Bull. 1973–2006 2002, 30, 899–907. [Google Scholar]
- Menargues, A.; Urios, V.; Mauri, M. Welfare Assessment of Captive Asian Elephants (Elephas maximus) and Indian Rhinoceros (Rhinoceros unicornis) Using Salivary Cortisol Measurement. Anim. Welf. 2008, 17, 305–312. [Google Scholar]
- Staley, M.; Conners, M.G.; Hall, K.; Miller, L.J. Linking Stress and Immunity: Immunoglobulin A as a Non-Invasive Physiological Biomarker in Animal Welfare Studies. Horm. Behav. 2018, 102, 55–68. [Google Scholar] [CrossRef]
- Emami-Khoyi, A.; Agnew, T.W.; Adair, M.G.; Murphy, E.C.; Benmazouz, I.; Monsanto, D.M.; Parbhu, S.P.; Main, D.C.; Le Roux, R.; Golla, T.R.; et al. A New Non-Invasive Method for Collecting DNA From Small Mammals in the Field, and Its Application in Simultaneous Vector and Disease Monitoring in Brushtail Possums. Front. Environ. Sci. 2021, 9, 318. [Google Scholar] [CrossRef]
- Filek, K.; Trotta, A.; Gračan, R.; Di Bello, A.; Corrente, M.; Bosak, S. Characterization of Oral and Cloacal Microbial Communities of Wild and Rehabilitated Loggerhead Sea Turtles (Caretta caretta). Anim. Microbiome 2021, 3, 59. [Google Scholar] [CrossRef]
- Menegotto, A. Potential Role of Telemetry and Non-Invasive Sampling in Wild Boar in the Surveillance and the Control of Foot-and-Mouth Disease in Bulgaria; Università di Padova: Padua, Italy, 2013. [Google Scholar]
- Mouchantat, S.; Haas, B.; Böhle, W.; Globig, A.; Lange, E.; Mettenleiter, T.C.; Depner, K. Proof of Principle: Non-Invasive Sampling for Early Detection of Foot-and-Mouth Disease Virus Infection in Wild Boar using a Rope-in-a-Bait Sampling Technique. Vet. Microbiol. 2014, 172, 329–333. [Google Scholar] [CrossRef]
- Mouchantat, S.; Globig, A.; Böhle, W.; Petrov, A.; Strebelow, H.-G.; Mettenleiter, T.C.; Depner, K. Novel Rope-Based Sampling of Classical Swine Fever Shedding in a Group of Wild Boar Showing Low Contagiosity upon Experimental Infection with a Classical Swine Fever Field Strain of Genotype 2.3. Vet. Microbiol. 2014, 170, 425–429. [Google Scholar] [CrossRef]
- Männistö, H.E. Collection of Oral Fluid Samples from Wild Boar in the Field Conditions to Detect African Swine Fever Virus (ASFV). Ph.D. Thesis, Estonian University of Life Sciences, Tartu, Estonia, 2018. [Google Scholar]
- Common, S.M.; Yun, Y.; Silva-Fletcher, A.; Thitaram, C.; Janyamethakul, T.; Khammesri, S.; Molenaar, F.M. Developing a Non-Invasive Method of Detecting Elephant Endotheliotropic Herpesvirus Infections Using Faecal Samples. Vet. Rec. 2021, 190, e833. [Google Scholar] [CrossRef]
- Evans, T.S.; Barry, P.A.; Gilardi, K.V.; Goldstein, T.; Deere, J.D.; Fike, J.; Yee, J.; Ssebide, B.J.; Karmacharya, D.; Cranfield, M.R.; et al. Optimization of a Novel Non-Invasive Oral Sampling Technique for Zoonotic Pathogen Surveillance in Nonhuman Primates. PLoS Negl. Trop. Dis. 2015, 9, e0003813. [Google Scholar] [CrossRef]
- Smiley Evans, T.; Gilardi, K.V.K.; Barry, P.A.; Ssebide, B.J.; Kinani, J.F.; Nizeyimana, F.; Noheri, J.B.; Byarugaba, D.K.; Mudakikwa, A.; Cranfield, M.R.; et al. Detection of Viruses Using Discarded Plants from Wild Mountain Gorillas and Golden Monkeys. Am. J. Primatol. 2016, 78, 1222–1234. [Google Scholar] [CrossRef]
- Seeber, P.A.; Soilemetzidou, S.E.; East, M.L.; Walzer, C.; Greenwood, A.D. Equine Behavioral Enrichment Toys as Tools for Non-Invasive Recovery of Viral and Host DNA. Zoo Biol. 2017, 36, 341–344. [Google Scholar] [CrossRef]
- Barasona, J.A.; Barroso-Arévalo, S.; Rivera, B.; Gortázar, C.; Sánchez-Vizcaíno, J.M. Detection of Antibodies against Mycobacterium bovis in Oral Fluid from Eurasian Wild Boar. Pathogens 2020, 9, 242. [Google Scholar] [CrossRef]
- Thompson, L.A.; Spoon, T.R.; Goertz, C.E.C.; Hobbs, R.C.; Romano, T.A. Blow Collection as a Non-Invasive Method for Measuring Cortisol in the Beluga (Delphinapterus leucas). PLoS ONE 2014, 9, e114062. [Google Scholar] [CrossRef]
- Groch, K.R.; Blazquez, D.N.H.; Marcondes, M.C.C.; Santos, J.; Colosio, A.; Díaz Delgado, J.; Catão-Dias, J.L. Cetacean Morbillivirus in Humpback Whales’ Exhaled Breath. Transbound. Emerg. Dis. 2021, 68, 1736–1743. [Google Scholar] [CrossRef]
- Reckendorf, A.; Schmicke, M.; Bunskoek, P.; Hansen, K.; Thybo, M.; Strube, C.; Siebert, U. Is Harbor Porpoise (Phocoena phocoena) Exhaled Breath Sampling Suitable for Hormonal Assessments? Animals 2021, 11, 907. [Google Scholar] [CrossRef]
- Acevedo-Whitehouse, K.; Rocha-Gosselin, A.; Gendron, D. A Novel Non-Invasive Tool for Disease Surveillance of Free-Ranging Whales and Its Relevance to Conservation Programs. Anim. Conserv. 2010, 13, 217–225. [Google Scholar] [CrossRef]
- Lima, N.; Rogers, T.; Acevedo-Whitehouse, K.; Brown, M.V. Temporal Stability and Species Specificity in Bacteria Associated with the Bottlenose Dolphins Respiratory System. Environ. Microbiol. Rep. 2012, 4, 89–96. [Google Scholar] [CrossRef]
- Cumeras, R.; Cheung, W.H.K.; Gulland, F.; Goley, D.; Davis, C.E. Chemical Analysis of Whale Breath Volatiles: A Case Study for Non-Invasive Field Health Diagnostics of Marine Mammals. Metabolites 2014, 4, 790–806. [Google Scholar] [CrossRef] [PubMed]
- Peled, N.; Ionescu, R.; Nol, P.; Barash, O.; McCollum, M.; VerCauteren, K.; Koslow, M.; Stahl, R.; Rhyan, J.; Haick, H. Detection of Volatile Organic Compounds in Cattle Naturally Infected with Mycobacterium bovis. Sens. Actuators B Chem. 2012, 171–172, 588–594. [Google Scholar] [CrossRef]
- Gundlach, N.H.; Piechotta, M.; Siebert, U. Is Lachrymal Fluid a Potential Method for Cortisol Measurement in Wild Harbor Seals? A Pilot Study. Int. J. Vet. Med. Res. Rep. 2016, 1–10. [Google Scholar] [CrossRef]
- Woodhams, D.; Brandt, H.; Baumgartner, S.; Kielgast, J.; Kupfer, E.; Tobler, U.; Davis, L.; Schmidt, B.; Bel, C.; Hodel, S.; et al. Interacting Symbionts and Immunity in the Amphibian Skin Mucosome Predict Disease Risk and Probiotic Effectiveness. PLoS ONE 2014, 9, e96375. [Google Scholar] [CrossRef]
- Zhu, W.; Fan, L.; Soto-Azat, C.; Yan, S.; Gao, X.; Liu, X.; Wang, S.; Liu, C.; Yang, X.; Li, Y. Filling a Gap in the Distribution of Batrachochytrium dendrobatidis: Evidence in Amphibians from Northern China. Dis. Aquat. Organ. 2016, 118, 259–265. [Google Scholar] [CrossRef]
- DiRenzo, G.V.; Campbell Grant, E.H.; Longo, A.V.; Che-Castaldo, C.; Zamudio, K.R.; Lips, K.R. Imperfect Pathogen Detection from Non-Invasive Skin Swabs Biases Disease Inference. Methods Ecol. Evol. 2018, 9, 380–389. [Google Scholar] [CrossRef]
- Santymire, R.M.; Manjerovic, M.B.; Sacerdote-Velat, A. A Novel Method for the Measurement of Glucocorticoids in Dermal Secretions of Amphibians. Conserv. Physiol. 2018, 6, coy008. [Google Scholar] [CrossRef]
- Smith, H.; Pasmans, F.; Dhaenens, M.; Deforce, D.; Bonte, D.; Verheyen, K.; Lens, L.; Martel, A. Skin Mucosome Activity as an Indicator of Batrachochytrium salamandrivorans Susceptibility in Salamanders. PLoS ONE 2018, 13. [Google Scholar] [CrossRef]
- Ghosh, P.N.; Verster, R.; Sewell, T.R.; O’Hanlon, S.J.; Brookes, L.M.; Rieux, A.; Garner, T.W.J.; Weldon, C.; Fisher, M.C. Discriminating Lineages of Batrachochytrium Dendrobatidis Using Quantitative PCR. Mol. Ecol. Resour. 2021, 21, 1452–1459. [Google Scholar] [CrossRef]
- Rothstein, A.; Byrne, A.; Knapp, R.; Briggs, C.; Voyles, J.; Richards-Zawacki, C.; Rosenblum, E. Divergent Regional Evolutionary Histories of a Devastating Global Amphibian Pathogen. Proc. R. Soc. B-Biol. Sci. 2021, 288, 20210782. [Google Scholar] [CrossRef]
- Dzul-Caamal, R.; Olivares-Rubio, H.F.; Salazar-Coria, L.; Rocha-Gómez, M.A.; Vega-López, A. Multivariate Analysis of Biochemical Responses using Non-Invasive Methods to Evaluate the Health Status of the Endangered Blackfin Goodeid (Girardinichthys viviparus). Ecol. Indic. 2016, 60, 1118–1129. [Google Scholar] [CrossRef]
- Fernández-Alacid, L.; Sanahuja, I.; Ordóñez-Grande, B.; Sánchez-Nuño, S.; Viscor, G.; Gisbert, E.; Herrera, M.; Ibarz, A. Skin Mucus Metabolites in Response to Physiological Challenges: A Valuable Non-Invasive Method to Study Teleost Marine Species. Sci. Total Environ. 2018, 644, 1323–1335. [Google Scholar] [CrossRef] [PubMed]
- Bulloch, P.; Schur, S.; Muthumuni, D.; Xia, Z.; Johnson, W.; Chu, M.; Palace, V.; Su, G.; Letcher, R.; Tomy, G.T. F2-Isoprostanes in Fish Mucus: A New, Non-Invasive Method for Analyzing a Biomarker of Oxidative Stress. Chemosphere 2020, 239, 124797. [Google Scholar] [CrossRef] [PubMed]
- Zuberi, A.; Ali, S.; Brown, C. A Non-Invasive Assay for Monitoring Stress Responses: A Comparison between Wild and Captive-Reared Rainbowfish (Melanoteania duboulayi). Aquaculture 2011, 321, 267–272. [Google Scholar] [CrossRef]
- Gabor, C.R.; Fisher, M.C.; Bosch, J. A Non-Invasive Stress Assay Shows That Tadpole Populations Infected with Batrachochytrium dendrobatidis Have Elevated Corticosterone Levels. PLoS ONE 2013, 8, e56054. [Google Scholar] [CrossRef] [PubMed]
- Baugh, A.T.; Bastien, B.; Still, M.B.; Stowell, N. Validation of Water-Borne Steroid Hormones in a Tropical Frog (Physalaemus pustulosus). Gen. Comp. Endocrinol. 2018, 261, 67–80. [Google Scholar] [CrossRef]
- Tornabene, B.; Hossack, B.; Crespi, E.; Breuner, C. Evaluating Corticosterone as a Biomarker for Amphibians Exposed to Increased Salinity and Ambient Corticosterone. Conserv. Physiol. 2021, 9, 1–15. [Google Scholar] [CrossRef]
- Sobańska, M.A. Wild Boar Hair (Sus scrofa) as a Non-Invasive Indicator of Mercury Pollution. Sci. Total Environ. 2005, 339, 81–88. [Google Scholar] [CrossRef]
- Bechshoft, T.; Derocher, A.E.; Richardson, E.; Mislan, P.; Lunn, N.J.; Sonne, C.; Dietz, R.; Janz, D.M.; St. Louis, V.L. Mercury and Cortisol in Western Hudson Bay Polar Bear Hair. Ecotoxicology 2015, 24, 1315–1321. [Google Scholar] [CrossRef]
- Hernout, B.V.; McClean, C.J.; Arnold, K.E.; Walls, M.; Baxter, M.; Boxall, A.B.A. Fur: A Non-Invasive Approach to Monitor Metal Exposure in Bats. Chemosphere 2016, 147, 376–381. [Google Scholar] [CrossRef]
- Treu, G.; Krone, O.; Unnsteinsdóttir, E.R.; Greenwood, A.D.; Czirják, G.Á. Correlations between Hair and Tissue Mercury Concentrations in Icelandic Arctic Foxes (Vulpes lagopus). Sci. Total Environ. 2018, 619–620, 1589–1598. [Google Scholar] [CrossRef] [PubMed]
- Montillo, M.; Caslini, C.; Peric, T.; Prandi, A.; Netto, P.; Tubaro, F.; Pedrotti, L.; Bianchi, A.; Mattiello, S. Analysis of 19 Minerals and Cortisol in Red Deer Hair in Two Different Areas of the Stelvio National Park: A Preliminary Study. Animals 2019, 9, 492. [Google Scholar] [CrossRef] [PubMed]
- Stoklasová, L.; Váhala, J.; Hejcmanová, P. Minerals in the Blood, Hair, and Faeces of the Critically Endangered Western Derby Eland under Human Care in Two Wildlife Reserves in Senegal. Biol. Trace Elem. Res. 2020, 195, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Richards, N.; Cook, G.; Simpson, V.; Hall, S.; Harrison, N.; Scott, K. Qualitative Detection of the NSAIDs Diclofenac and Ibuprofen in the Hair of Eurasian Otters (Lutra lutra) Occupying UK Waterways with GC-MS. Eur. J. Wildl. Res. 2011, 57, 1107–1114. [Google Scholar] [CrossRef]
- Keckeis, K.; Lepschy, M.; Schöpper, H.; Moser, L.; Troxler, J.; Palme, R. Hair Cortisol: A Parameter of Chronic Stress? Insights from a Radiometabolism Study in Guinea Pigs. J. Comp. Physiol. B 2012, 182, 985–996. [Google Scholar] [CrossRef]
- Terwissen, C.V.; Mastromonaco, G.F.; Murray, D.L. Influence of Adrenocorticotrophin Hormone Challenge and External Factors (Age, Sex, and Body Region) on Hair Cortisol Concentration in Canada Lynx (Lynx canadensis). Gen. Comp. Endocrinol. 2013, 194, 162–167. [Google Scholar] [CrossRef]
- Caslini, C.; Comin, A.; Peric, T.; Prandi, A.; Pedrotti, L.; Mattiello, S. Use of Hair Cortisol Analysis for Comparing Population Status in Wild Red Deer (Cervus elaphus) Living in Areas with Different Characteristics. Eur. J. Wildl. Res. 2016, 62, 713–723. [Google Scholar] [CrossRef]
- Charalambous, R.; Narayan, E. Cortisol Measurement in Koala (Phascolarctos cinereus) Fur. JOVE-J. Vis. Exp. 2019. [Google Scholar] [CrossRef]
- Crill, C.; Janz, D.M.; Kusch, J.M.; Santymire, R.M.; Heyer, G.P.; Shury, T.K.; Lane, J.E. Investigation of the Utility of Feces and Hair as Non-Invasive Measures of Glucocorticoids in Wild Black-Tailed Prairie Dogs (Cynomys ludovicianus). Gen. Comp. Endocrinol. 2019, 275, 15–24. [Google Scholar] [CrossRef]
- Heimbürge, S.; Kanitz, E.; Otten, W. The Use of Hair Cortisol for the Assessment of Stress in Animals. Gen. Comp. Endocrinol. 2019, 270, 10–17. [Google Scholar] [CrossRef]
- Potratz, E.J.; Brown, J.S.; Gallo, T.; Anchor, C.; Santymire, R.M. Effects of Demography and Urbanization on Stress and Body Condition in Urban White-Tailed Deer. Urban Ecosyst. 2019, 22, 807–816. [Google Scholar] [CrossRef]
- Cattet, M.; Stenhouse, G.B.; Boulanger, J.; Janz, D.M.; Kapronczai, L.; Swenson, J.E.; Zedrosser, A. Can Concentrations of Steroid Hormones in Brown Bear Hair Reveal Age Class? Conserv. Physiol. 2018, 6, coy001. [Google Scholar] [CrossRef] [PubMed]
- Everest, D.; Tolhurst-Cherriman, D.; Davies, H.; Dastjerdi, A.; Ashton, A.; Blackett, T.; Meredith, A.; Milne, E.; Mill, A.; Shuttleworth, C. Assessing a Potential Non-Invasive Method for Viral Diagnostic Purposes in European Squirrels. HYSTRIX—Ital. J. Mammal. 2019, 30, 44–50. [Google Scholar] [CrossRef]
- Muñoz-Madrid, R.; Belinchón-Lorenzo, S.; Iniesta, V.; Fernández-Cotrina, J.; Parejo, J.C.; Serrano, F.J.; Monroy, I.; Baz, V.; Gómez-Luque, A.; Gómez-Nieto, L.C. First Detection of Leishmania infantum Kinetoplast DNA in Hair of Wild Mammals: Application of QPCR Method to Determine Potential Parasite Reservoirs. Acta Trop. 2013, 128, 706–709. [Google Scholar] [CrossRef]
- Ortega, M.V.; Moreno, I.; Domínguez, M.; de la Cruz, M.L.; Martín, A.B.; Rodríguez-Bertos, A.; López, R.; Navarro, A.; González, S.; Mazariegos, M.; et al. Application of a Specific Quantitative Real-Time PCR (QPCR) to Identify Leishmania infantum DNA in Spleen, Skin and Hair Samples of Wild Leporidae. Vet. Parasitol. 2017, 243, 92–99. [Google Scholar] [CrossRef]
- Hopkins, J.B.; Whittington, J.; Clevenger, A.P.; Sawaya, M.A.; St. Clair, C.C. Stable Isotopes Reveal Rail-Associated Behavior in a Threatened Carnivore. Isotopes Environ. Health Stud. 2014, 50, 322–331. [Google Scholar] [CrossRef]
- Rysava, K.; McGill, R.; Matthiopoulos, J.; Hopcraft, J. Re-Constructing Nutritional History of Serengeti Wildebeest from Stable Isotopes in Tail Hair: Seasonal Starvation Patterns in an Obligate Grazer. Rapid Commun. Mass Spectrom. 2016, 30, 1461–1468. [Google Scholar] [CrossRef]
- Espín, S.; Martínez-López, E.; Gómez-Ramírez, P.; María-Mojica, P.; García-Fernández, A.J. Razorbills (Alca torda) as Bioindicators of Mercury Pollution in the Southwestern Mediterranean. Mar. Pollut. Bull. 2012, 64, 2461–2470. [Google Scholar] [CrossRef]
- Costa, R.A.; Eeva, T.; Eira, C.; Vaqueiro, J.; Vingada, J.V. Assessing Heavy Metal Pollution Using Great Tits (Parus major): Feathers and Excrements from Nestlings and Adults. Environ. Monit. Assess. 2013, 185, 5339–5344. [Google Scholar] [CrossRef]
- Di Marzio, A.; Gómez-Ramírez, P.; Barbar, F.; Lambertucci, S.A.; García-Fernández, A.J.; Martínez-López, E. Mercury in the Feathers of Bird Scavengers from Two Areas of Patagonia (Argentina) under the Influence of Different Anthropogenic Activities: A Preliminary Study. Environ. Sci. Pollut. Res. 2018, 25, 13906–13915. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, Y.; Maltby, L.; Zheng, Y.; Ma, Q. Trace Elements Exposure of Endangered Crested Ibis (Nipponia nippon) under in Situ and Ex Situ Conservations. Environ. Pollut. 2019, 253, 800–810. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.; Flavier, M.; Paller, V.; Rebancos, C.; Alcantara, A.; Sanchez, R.; Pelegrina, D. Presence of Heavy Metals in Feathers of Avian Species and in Soils of Barangay Ipil-Calancan Bay, Sta. Cruz, Marinduque Island, Philippines. PERTANIKA J. Trop. Agric. Sci. 2019, 42, 1009–1021. [Google Scholar]
- Bryan, C.E.; Christopher, S.J.; Balmer, B.C.; Wells, R.S. Establishing Baseline Levels of Trace Elements in Blood and Skin of Bottlenose Dolphins in Sarasota Bay, Florida: Implications for Non-Invasive Monitoring. Sci. Total Environ. 2007, 388, 325–342. [Google Scholar] [CrossRef] [PubMed]
- Ebmer, D.; Navarrete, M.J.; Muñoz, P.; Flores, L.M.; Gärtner, U.; Taubert, A.; Hermosilla, C. Antarctophthirus Microchir Infestation in Synanthropic South American Sea Lion (Otaria flavescens) Males Diagnosed by a Novel Non-Invasive Method. Parasitol. Res. 2019, 118, 1353–1361. [Google Scholar] [CrossRef]
- Fraser, T.A.; Martin, A.; Polkinghorne, A.; Carver, S. Comparative Diagnostics Reveals PCR Assays on Skin Scrapings Is the Most Reliable Method to Detect Sarcoptes Scabiei Infestations. Vet. Parasitol. 2018, 251, 119–124. [Google Scholar] [CrossRef]
- Schneider, L.; Eggins, S.; Maher, W.; Vogt, R.C.; Krikowa, F.; Kinsley, L.; Eggins, S.M.; Da Silveira, R. An Evaluation of the Use of Reptile Dermal Scutes as a Non-Invasive Method to Monitor Mercury Concentrations in the Environment. Chemosphere 2015, 119, 163–170. [Google Scholar] [CrossRef]
- Burns, T.J.; McCafferty, D.J.; Kennedy, M.W. Core and Body Surface Temperatures of Nesting Leatherback Turtles (Dermochelys coriacea). J. Therm. Biol. 2015, 51, 15–22. [Google Scholar] [CrossRef]
- Horton, T.W.; Hauser, N.; Cassel, S.; Klaus, K.F.; Fettermann, T.; Key, N. Doctor Drone: Non-Invasive Measurement of Humpback Whale Vital Signs Using Unoccupied Aerial System Infrared Thermography. Front. Mar. Sci. 2019, 6, 466. [Google Scholar] [CrossRef]
- Valle, C.; Grijalva, C.; Calle, P.; Munoz-PereZ, J.; Quezada, G.; Vera, C.; Lewbart, G. Methods of Body Temperature Assessment in Conolophus subcristatus, Conolophus pallidus (Galapagos Land Iguanas), and Amblyrhynchus Cristatus X C. Subcristatus Hybrid. PEERJ 2019, 7. [Google Scholar] [CrossRef]
- Jerem, P.; Jenni-Eiermann, S.; Herborn, K.; McKeegan, D.; McCafferty, D.J.; Nager, R.G. Eye Region Surface Temperature Reflects Both Energy Reserves and Circulating Glucocorticoids in a Wild Bird. Sci. Rep. 2018, 8, 1907. [Google Scholar] [CrossRef]
- Mellish, J.; Nienaber, J.; Polasek, L.; Horning, M. Beneath the Surface: Profiling Blubber Depth in Pinnipeds with Infrared Imaging. J. Therm. Biol. 2013, 38, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Narayan, E.; Perakis, A.; Meikle, W. Using Thermal Imaging to Monitor Body Temperature of Koalas (Phascolarctos cinereus) in A Zoo Setting. Animals 2019, 9, 1094. [Google Scholar] [CrossRef] [PubMed]
- Mccafferty, D.J. The Value of Infrared Thermography for Research on Mammals: Previous Applications and Future Directions. Mammal Rev. 2007, 37, 207–223. [Google Scholar] [CrossRef]
- Arenas, A.J.; Gómez, F.; Salas, R.; Carrasco, P.; Borge, C.; Maldonado, A.; O’Brien, D.J.; Martínez-Moreno, F.J. An Evaluation of the Application of Infrared Thermal Imaging to the Tele-Diagnosis of Sarcoptic Mange in the Spanish Ibex (Capra pyrenaica). Vet. Parasitol. 2002, 109, 111–117. [Google Scholar] [CrossRef]
- Dunbar, M.R.; MacCarthy, K.A. Use of Infrared Thermography to Detect Signs of Rabies Infection in Raccoons (Procyon lotor). J. Zoo Wildl. Med. 2006, 37, 518–523. [Google Scholar] [CrossRef]
- Dunbar, M.R.; Johnson, S.R.; Rhyan, J.C.; McCollum, M. Use of Infrared Thermography to Detect Thermographic Changes in Mule Deer (Odocoileus hemionus) Experimentally Infected with Foot-and-Mouth Disease. J. Zoo Wildl. Med. 2009, 40, 296–301. [Google Scholar] [CrossRef]
- Galaverni, M.; Palumbo, D.; Fabbri, E.; Caniglia, R.; Greco, C.; Randi, E. Monitoring Wolves (Canis lupus) by Non-Invasive Genetics and Camera Trapping: A Small-Scale Pilot Study. Eur. J. Wildl. Res. 2012, 58, 47–58. [Google Scholar] [CrossRef]
- Murray, M.H.; Fidino, M.; Lehrer, E.W.; Simonis, J.L.; Magle, S.B. A Multi-State Occupancy Model to Non-Invasively Monitor Visible Signs of Wildlife Health with Camera Traps That Accounts for Image Quality. J. Anim. Ecol. 2021, 90, 1973–1984. [Google Scholar] [CrossRef]
- Lacroux, C.; Guma, N.; Krief, S. Facial Dysplasia in Wild Forest Olive Baboons (Papio anubis) in Sebitoli, Kibale National Park, Uganda: Use of Camera Traps to Detect Health Defects. J. Med. Primatol. 2019, 48, 143–153. [Google Scholar] [CrossRef]
- Hicks, L.L.; Schwab, N.A.; Homyack, J.A.; Jones, J.E.; Maxell, B.A.; Burkholder, B.O. A Statistical Approach to White-Nose Syndrome Surveillance Monitoring Using Acoustic Data. PLoS ONE 2020, 15, e0241052. [Google Scholar] [CrossRef]
- Kvapil, P.; Kastelic, M.; Jež, N.; Sedlák, K.; Kašpárková, N.; Jelovšek, M.; Avšič-Županc, T.; Bártová, E.; Račnik, J. Detection of Antibodies against Tick-Borne Encephalitis Virus in Zoo Animals Using Non-Invasive Blood Sampling with Medicinal Leeches (Hirudo medicinalis). Pathogens 2021, 10, 952. [Google Scholar] [CrossRef] [PubMed]
- Alfano, N.; Dayaram, A.; Axtner, J.; Tsangaras, K.; Kampmann, M.-L.; Mohamed, A.; Wong, S.T.; Gilbert, M.T.P.; Wilting, A.; Greenwood, A.D. Non-Invasive Surveys of Mammalian Viruses Using Environmental DNA. Methods Ecol. Evol. 2021, 12, 1941–1952. [Google Scholar] [CrossRef]
- Thomsen, R.; Voigt, C.C. Non-Invasive Blood Sampling from Primates using Laboratory-Bred Blood-Sucking Bugs (Dipetalogaster maximus; Reduviidae, Heteroptera). Primates 2006, 47, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Voigt, C.C.; Peschel, U.; Wibbelt, G.; Frölich, K. An Alternative, Less Invasive Blood Sample Collection Technique for Serologic Studies Utilizing Triatomine Bugs (Heteroptera; Insecta). J. Wildl. Dis. 2006, 42, 466–469. [Google Scholar] [CrossRef]
- Hoffmann, C.; Stockhausen, M.; Merkel, K.; Calvignac-Spencer, S.; Leendertz, F.H. Assessing the Feasibility of Fly Based Surveillance of Wildlife Infectious Diseases. Sci. Rep. 2016, 6, 37952. [Google Scholar] [CrossRef]
- Zechmeisterová, K.; de Bellocq, J.G.; Široký, P. Diversity of Karyolysus and Schellackia from the Iberian Lizard Lacerta schreiberi with Sequence Data from Engorged Ticks. Parasitology 2019, 146, 1690–1698. [Google Scholar] [CrossRef]
- Martens, J.M.; Stokes, H.S.; Berg, M.L.; Walder, K.; Raidal, S.R.; Magrath, M.J.L.; Bennett, A.T.D. A Non-Invasive Method to Assess Environmental Contamination with Avian Pathogens: Beak and Feather Disease Virus (BFDV) Detection in Nest Boxes. PeerJ 2020, 8, e9211. [Google Scholar] [CrossRef]
- De Oliveira, M.L.; Norris, D.; Ramírez, J.F.M.; de Peres, P.H.F.; Galetti, M.; Duarte, J.M.B. Dogs Can Detect Scat Samples More Efficiently than Humans: An Experiment in a Continuous Atlantic Forest Remnant. Zool. Curitiba 2012, 29, 183–186. [Google Scholar] [CrossRef]
- Curry, E.; Skogen, M.; Roth, T. Evaluation of an Odour Detection Dog for Non-Invasive Pregnancy Diagnosis in Polar Bears (Ursus maritimus): Considerations for Training Sniffer Dogs for Biomedical Investigations in Wildlife Species. J. Zoo Aquar. Res. 2021, 9, 1–7. [Google Scholar] [CrossRef]
- Lurz, P.W.W.; Krauze-Gryz, D.; Gryz, J.; Meredith, A.; Schilling, A.-K.; Thain, C.; Heller, E. Invisible Threats to Native Mammals—Mercury Levels in Three Eurasian Red Squirrel Populations. Hystrix Ital. J. Mammal. 2017, 28, 280–283. [Google Scholar] [CrossRef]
- Romeo, C.; Piscitelli, A.P.; Santicchia, F.; Martinoli, A.; Ferrari, N.; Wauters, L.A. Invading Parasites: Spillover of an Alien Nematode Reduces Survival in a Native Species. Biol. Invasions 2021, 23, 3847–3857. [Google Scholar] [CrossRef]
- Beard, D.; Stannard, H.J.; Old, J.M. Morphological Identification of Ticks and Molecular Detection of Tick-Borne Pathogens from Bare-Nosed Wombats (Vombatus ursinus). Parasit. Vectors 2021, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Segura-Göthlin, S.; Fernández, A.; Arbelo, M.; Felipe-Jiménez, I.; Colom-Rivero, A.; Almunia, J.; Sierra, E. The Validation of a Non-Invasive Skin Sampling Device for Detecting Cetacean Poxvirus. Animals 2021, 11, 2814. [Google Scholar] [CrossRef]
- Apprill, A.; Miller, C.A.; Moore, M.J.; Durban, J.W.; Fearnbach, H.; Barrett-Lennard, L.G. Extensive Core Microbiome in Drone-Captured Whale Blow Supports a Framework for Health Monitoring. mSystems 2017, 2, e00119-17. [Google Scholar] [CrossRef] [PubMed]
- Lebarbenchon, C.; Poulson, R.; Shannon, K.; Slagter, J.; Slusher, M.J.; Wilcox, B.R.; Berdeen, J.; Knutsen, G.A.; Cardona, C.J.; Stallknecht, D.E. Isolation of Influenza A Viruses from Wild Ducks and Feathers in Minnesota (2010–2011). Avian Dis. 2013, 57, 677–680. [Google Scholar] [CrossRef] [PubMed]
- Frongia, G.N.; Peric, T.; Leoni, G.; Satta, V.; Berlinguer, F.; Muzzeddu, M.; Prandi, A.; Naitana, S.; Comin, A. Assessment of Cortisol and DHEA Concentrations in Griffon Vulture (Gyps fulvus) Feathers to Evaluate its Allostatic Load. Ann. Anim. Sci. 2020, 20, 85–96. [Google Scholar] [CrossRef]
- Dipineto, L.; Bossa, L.M.D.L.; Pace, A.; Russo, T.P.; Gargiulo, A.; Ciccarelli, F.; Raia, P.; Caputo, V.; Fioretti, A. Microbiological Survey of Birds of Prey Pellets. Comp. Immunol. Microbiol. Infect. Dis. 2015, 41, 49–53. [Google Scholar] [CrossRef]
- Nguyen, Q. The Isolation of Presumptive Pathogens from the Pellets of Seven North American Owl Species. Merge 2019, 3, 1–21. [Google Scholar]
| Topic | Type of Article | Host’s Taxon | Material Collected | Collection Method 1 | Detection Method 2 | |
|---|---|---|---|---|---|---|
| Disease | Research 5 | Amphibia | Faeces | Collection from habitat | Analytical chemistry | |
| Bacteria | Method 6 | Aves | Hair-feathers-skin | Hair trapping | Coprological method | |
| Disease | Review | Fish | Imaging and remote sensing | New device | Coprological method combined 11 | |
| Ectoparasites | Mammalia | Invertebrates 8 | Other animals 9 | Imaging | ||
| Endoparasites | Marine mammal | Saliva and other body fluids | Post-mortem 10 | Immunoassay | ||
| Fungi | Reptilia | Urine | Trapping and handling | Molecular method | ||
| Helminths | Several 7 | Several 7 | Visual | Molecular method combined 12 | ||
| Protozoa | Several 7 | Other 3 | ||||
| Virus | Several 7 | |||||
| Other 3 | ||||||
| Non-disease | ||||||
| Diet | ||||||
| Immunity | ||||||
| Physiology 4 | ||||||
| Pollutants | ||||||
| Reproductive condition | ||||||
| Stress | ||||||
| Other 3 | ||||||
| Both | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schilling, A.-K.; Mazzamuto, M.V.; Romeo, C. A Review of Non-Invasive Sampling in Wildlife Disease and Health Research: What’s New? Animals 2022, 12, 1719. https://doi.org/10.3390/ani12131719
Schilling A-K, Mazzamuto MV, Romeo C. A Review of Non-Invasive Sampling in Wildlife Disease and Health Research: What’s New? Animals. 2022; 12(13):1719. https://doi.org/10.3390/ani12131719
Chicago/Turabian StyleSchilling, Anna-Katarina, Maria Vittoria Mazzamuto, and Claudia Romeo. 2022. "A Review of Non-Invasive Sampling in Wildlife Disease and Health Research: What’s New?" Animals 12, no. 13: 1719. https://doi.org/10.3390/ani12131719
APA StyleSchilling, A.-K., Mazzamuto, M. V., & Romeo, C. (2022). A Review of Non-Invasive Sampling in Wildlife Disease and Health Research: What’s New? Animals, 12(13), 1719. https://doi.org/10.3390/ani12131719






