Poult Enteritis and Mortality Syndrome in Turkey Poults: Causes, Diagnosis and Preventive Measures
Simple Summary
Abstract
1. Introduction
2. Causes of PEMS
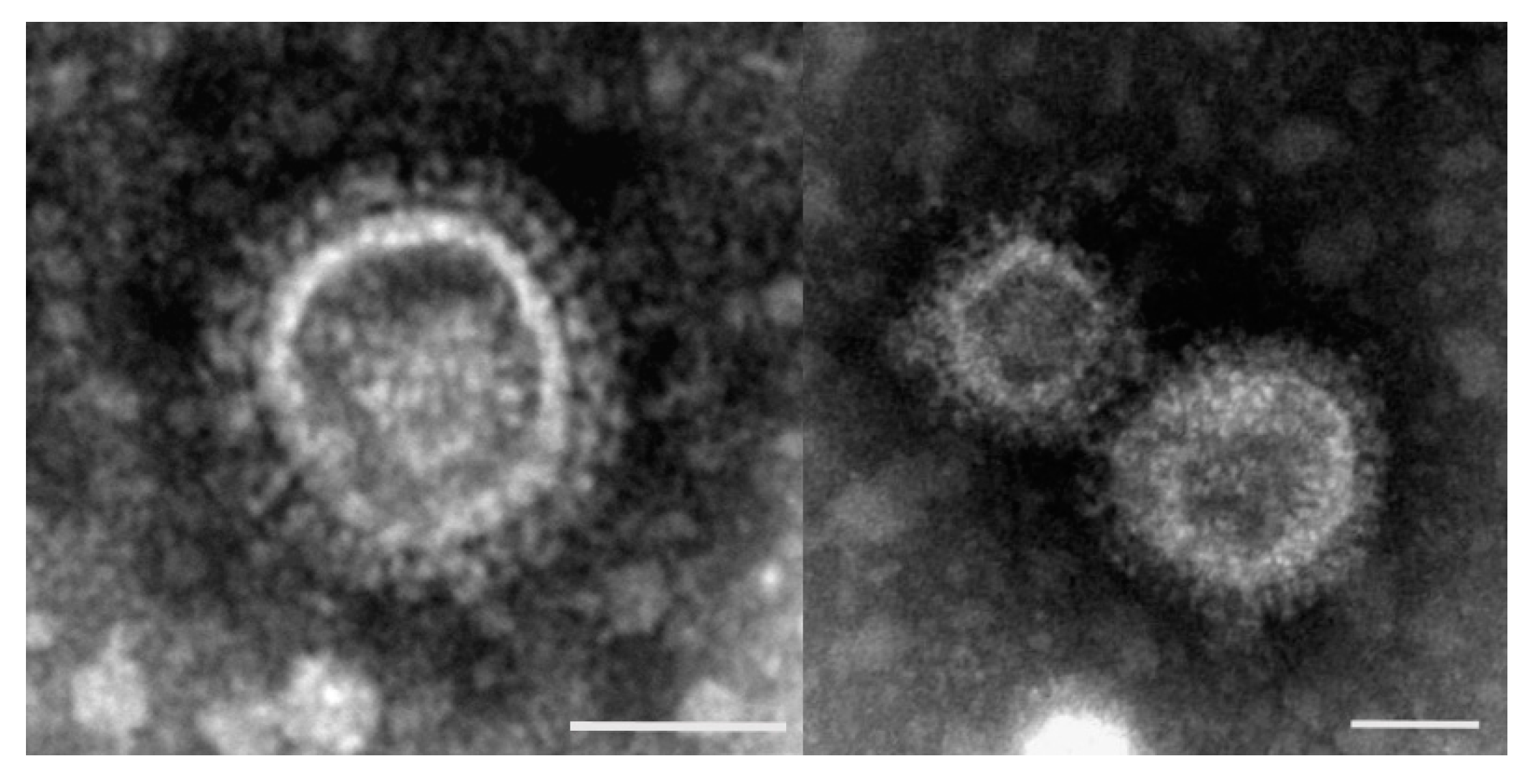
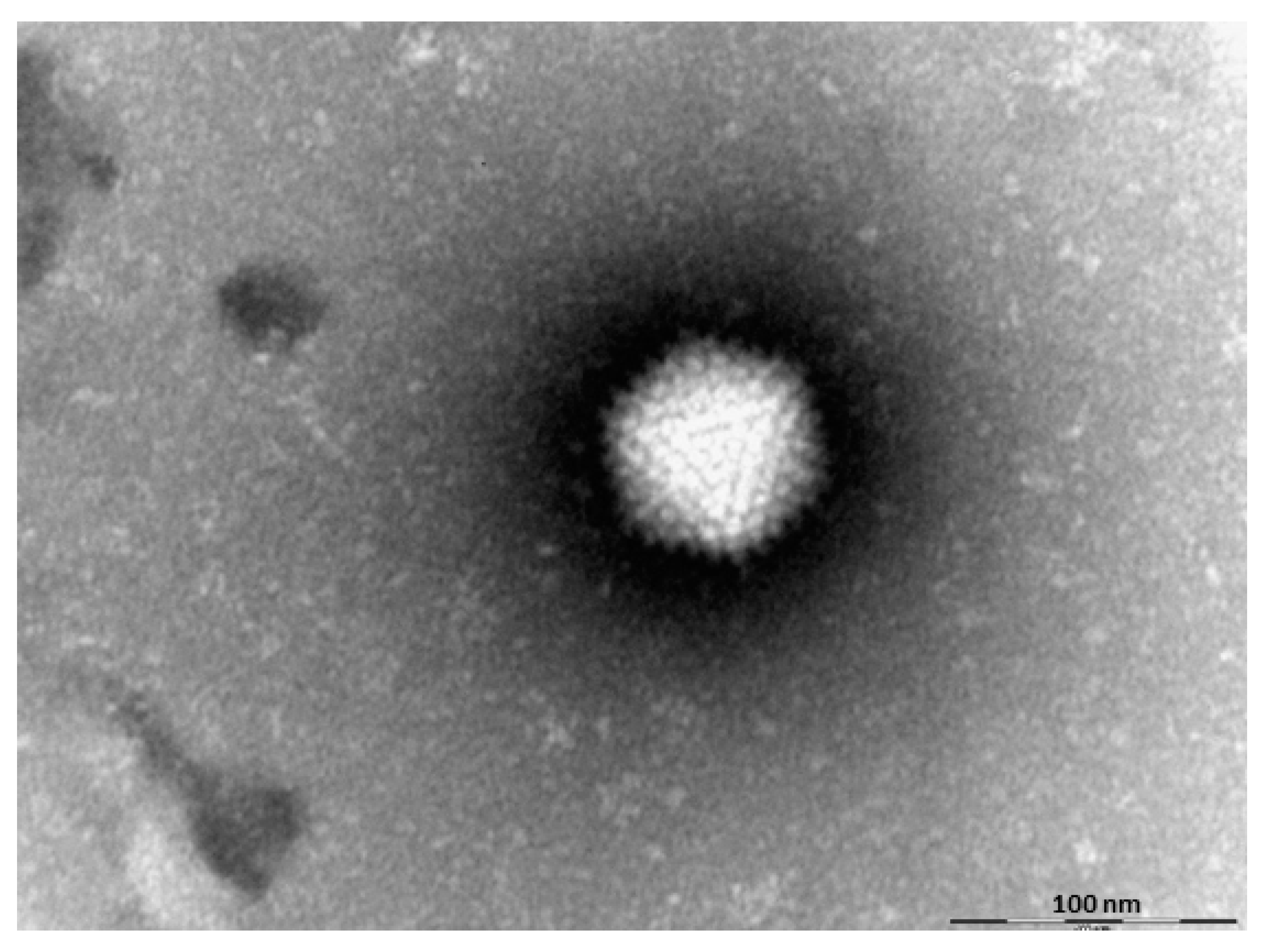
2.1. Turkey Coronavirus
2.2. Astrovirus
2.3. Adenovirus
2.4. Rotaviruses
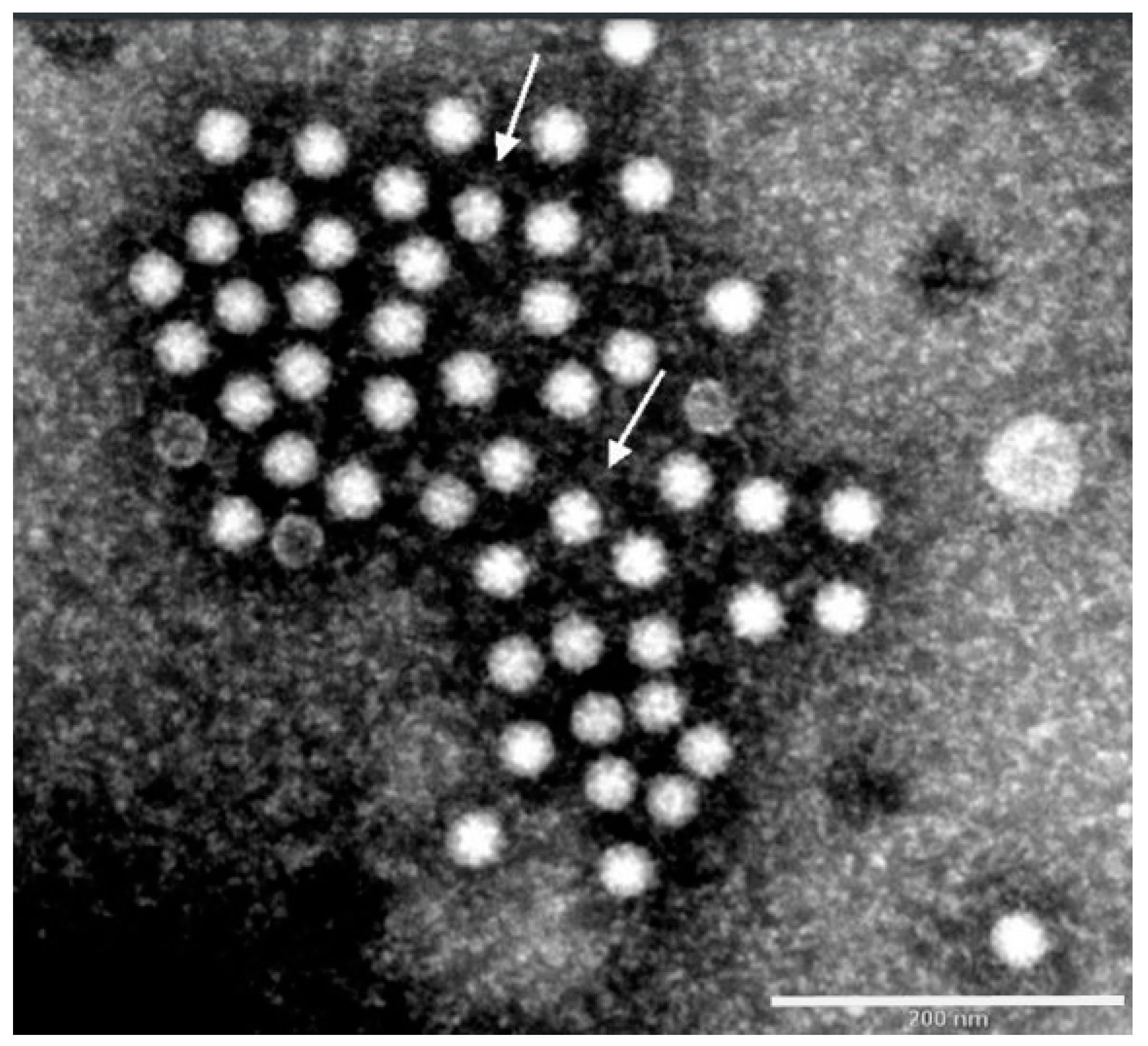
2.5. Reovirus
3. Diagnosis of Viruses Implicated in Turkey Poult Enteritis
4. Prevention and Control of Enteritis in Turkeys
5. Conclusions and Recommendations
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Hafez, H.M.; Shehata, A.A. Turkey production and health: Current challenges. Ger. J. Vet. Res. 2021, 1, 3–14. [Google Scholar] [CrossRef]
- Abd El-Wahab, A.; Visscher, C.; Haider, W.; Dimitri, R. A case study of histomoniasis in fattening turkeys identified in histopathological investigations. Ger. J. Vet. Res. 2021, 3, 13–18. [Google Scholar] [CrossRef]
- Hafez, H.M. Overview about currently ongoing viral diseases of turkeys. In Proceedings of the 7th International Symposium on Turkey Diseases, Berlin, Germany, 19–21 June 2008; Verlag der DVG Service GmbH: Gießen, Germany, 2008. ISBN 978-3-939902-96-6. [Google Scholar]
- Barnes, H.J.; Guy, J.S.; Vaillancourt, J.P. Poult enteritis complex. Rev. Sci. Tech. 2000, 19, 565–588. [Google Scholar] [CrossRef]
- Spackman, E.; Kapczynski, D.; Sellers, H. Multiplex real-time reverse transcription–polymerase chain reaction for the detection of three viruses associated with poult enteritis complex: Turkey astrovirus, turkey coronavirus, and turkey reovirus. Avian Dis. 2005, 49, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Jindal, N.; Patnayak, D.P.; Ziegler, A.F.; Lago, A.; Goyal, S.M. A retrospective study on poult enteritis syndrome in minnesota. Avian Dis. 2009, 53, 268–275. [Google Scholar] [CrossRef]
- Mor, S.K.; Sharafeldin, T.A.; Abin, M.; Kromm, M.; Porter, R.E.; Goyal, S.M.; Patnayak, D.P. The occurrence of enteric viruses in light turkey syndrome. Avian Pathol. 2013, 42, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Carver, D.K.; Vaillancourt, J.P.; Stringham, M.; Guy, J.S.; Barnes, H.J. Mortality patterns associated with poult enteritis mortality syndrome (PEMS) and coronaviral enteritis in turkey flocks raised in pems-affected regions. Avian Dis. 2001, 45, 985–991. [Google Scholar] [CrossRef]
- Day, J.M.; Ballard, L.L.; Duke, M.V.; Scheffler, B.E.; Zsak, L. Metagenomic analysis of the turkey gut RNA virus community. Virol. J. 2010, 7, 313. [Google Scholar] [CrossRef]
- Guy, J.S.; Smith, L.G.; Breslin, J.J.; Vaillancourt, J.P.; Barnes, H.J. High mortality and growth depression experimentally produced in young turkeys by dual infection with enteropathogenic Escherichia coli and turkey coronavirus. Avian Dis. 2000, 44, 105–113. [Google Scholar] [CrossRef]
- Guy, J.S.; Barnes, H.J. Partial characterization of a turkey enterovirus-like virus. Avian Dis. 1991, 35, 197–200. [Google Scholar] [CrossRef]
- Pakpinyo, S.; Ley, D.H.; Barnes, H.J.; Vaillancourt, J.P.; Guy, J.S. Prevalence of enteropathogenic Escherichia coli in naturally occurring cases of poult enteritis-mortality syndrome. Avian Dis. 2002, 46, 360–369. [Google Scholar] [CrossRef]
- Woolcock, P.; Shivaprasad, H. Electron microscopic identification of viruses associated with poult enteritis in turkeys grown in california 1993–2003. Avian Dis. 2008, 52, 209–213. [Google Scholar] [CrossRef]
- Yu, M.; Ismail, M.M.; Qureshi, M.A.; Dearth, R.N.; Barnes, H.J.; Saif, Y.M. Viral agents associated with poult enteritis and mortality syndrome: The role of a small round virus and a turkey coronavirus. Avian Dis. 2000, 44, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Spackman, E.; Day, J.M.; Pantin-Jackwood, M.J. Astrovirus, reovirus, and rotavirus concomitant infection causes decreased weight gain in broad-breasted white poults. Avian Dis. 2010, 54, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Zsak, L.; Strother, K.O.; Kisary, J. Partial genome sequence analysis of parvoviruses associated with enteric disease in poultry. Avian Pathol. 2008, 37, 435–441. [Google Scholar] [CrossRef]
- Domańska-Blicharz, K.; Bocian, Ł.; Lisowska, A.; Jacukowicz, A.; Pikuła, A.; Minta, Z. Cross-sectional survey of selected enteric viruses in polish turkey flocks between 2008 and 2011. BMC Vet. Res. 2017, 13, 108. [Google Scholar] [CrossRef]
- Peterson, E.H.; Hymas, T.A. Antibiotics in the treatment of an unfamiliar turkey disease. Poult. Sci. 1951, 30, 466–468. [Google Scholar] [CrossRef]
- Circella, E.; Camarda, A.; Martella, V.; Bruni, G.; Lavazza, A.; Buonavoglia, C. Coronavirus associated with an enteric syndrome on a quail farm. Avian Pathol. 2007, 36, 251–258. [Google Scholar] [CrossRef]
- Culver, F.; Dziva, F.; Cavanagh, D.; Stevens, M.P. Poult enteritis and mortality syndrome in turkeys in great britain. Vet. Rec. 2006, 159, 209–210. [Google Scholar] [CrossRef]
- Maurel, S.; Toquin, D.; Briand, F.X.; Quéguiner, M.; Allée, C.; Bertin, J.; Ravillion, L.; Retaux, C.; Turblin, V.; Morvan, H.; et al. First full-length sequences of the s gene of european isolates reveal further diversity among turkey coronaviruses. Avian Pathol. 2011, 40, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.C.B.; Luvizotto, M.C.R.; Ferrari, H.F.; Mendes, A.R.; da Silva, S.E.L.; Cardoso, T.C. Detection of turkey coronavirus in commercial turkey poults in brazil. Avian Pathol. 2007, 36, 29–33. [Google Scholar] [CrossRef][Green Version]
- Villarreal, L.Y.B.; Assayag, M.S.; Brandão, P.E.; Chacón, J.L.V.; Bunger, A.N.D.; Astolfi-Ferreira, C.S.; Gomes, C.R.; Jones, R.C.; Ferreira, A.J.P. Identification of turkey astrovirus and turkey coronavirus in an outbreak of poult enteritis and mortality syndrome. Braz. J. Poult. Sci. 2006, 8, 131–135. [Google Scholar] [CrossRef]
- Wooming, B. A coronavirus outbreak in northwest arkansas. In Proceedings of the Annual meeting of American Association of Avian Pathologist, Chicago, IL, USA, 20–23 July 2013. [Google Scholar]
- Houta, M.H.; Awe, O.O.; Ali, A. Infection with the turkey coronavirus: A recurring problem in turkeys. Ger. J. Vet. Res. 2021, 3, 19–27. [Google Scholar] [CrossRef]
- International Committee on Taxonomy of Viruses (ICTV) Master Species List 2019 V1. Available online: https://talk.ictvonline.org/taxonomy (accessed on 15 June 2021).
- Cui, J.; Li, F.; Shi, Z.-L. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019, 17, 181–192. [Google Scholar] [CrossRef] [PubMed]
- de Wit, J.J.; Cook, J.K.A. Spotlight on avian coronaviruses. Avian Pathol. 2020, 49, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, M.H.; Barta, J.R.; Ojkic, D.; Yoo, D. Complete genomic sequence of turkey coronavirus. Virus Res. 2008, 135, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Jackwood, M.W.; Boynton, T.O.; Hilt, D.A.; McKinley, E.T.; Kissinger, J.C.; Paterson, A.H.; Robertson, J.; Lemke, C.; McCall, A.W.; Williams, S.M.; et al. Emergence of a group 3 coronavirus through recombination. Virology 2010, 398, 98–108. [Google Scholar] [CrossRef]
- Brown, P.A.; Touzain, F.; Briand, F.X.; Gouilh, A.M.; Courtillon, C.; Allée, C.; Lemaitre, E.; De Boisséson, C.; Blanchard, Y.; Eterradossi, N. First complete genome sequence of european turkey coronavirus suggests complex recombination history related with us turkey and guinea fowl coronaviruses. J. Gen. Virol. 2016, 97, 110–120. [Google Scholar] [CrossRef]
- Domanska-Blicharz, K.; Sajewicz-Krukowska, J. Recombinant turkey coronavirus: Are some s gene structures of gammacoronaviruses especially prone to exchange? Poult. Sci. 2021, 100, 101018. [Google Scholar] [CrossRef]
- Ducatez, M.F.; Liais, E.; Croville, G.; Guérin, J.-L. Full genome sequence of guinea fowl coronavirus associated with fulminating disease. Virus Genes 2015, 50, 514–517. [Google Scholar] [CrossRef]
- Miłek, J.; Blicharz-Domańska, K. Coronaviruses in avian species—review with focus on epidemiology and diagnosis in wild birds. J. Vet. Res. 2018, 62, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Jackwood, M.W. Review of infectious bronchitis virus around the world. Avian Dis. 2012, 56, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-N.; Loa, C.C.; Ababneh, M.M.-K.; Wu, C.C.; Lin, T.L. Genotyping of turkey coronavirus field isolates from various geographic locations in the unites states based on the spike gene. Arch. Virol. 2015, 160, 2719–2726. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, X.; Chen, X.; Yang, S.; Ling, Y.; Song, Q.; Zhu, S.; Sun, L.; Li, C.; Li, Y.; et al. A recombinant infectious bronchitis virus from a chicken with a spike gene closely related to that of a turkey coronavirus. Arch. Virol. 2020, 165, 703–707. [Google Scholar] [CrossRef]
- Guy, J.S. Turkey coronavirus. In Diseases of Poultry; Saif, Y.M., Barnes, H.J., Glisson, J.R., Fadly, A.M., McDougald, L.R., Swayne, D.E., Eds.; Iowa State University Press: Ames, IA, USA, 2003; pp. 300–307. [Google Scholar]
- Guy, J.S.; Smith, L.G.; Breslin, J.J.; Pakpinyo, S. Development of a competitive enzyme-linked immunosorbent assay for detection of turkey coronavirus antibodies. Avian Dis. 2002, 46, 334–341. [Google Scholar] [CrossRef]
- Brown, P.A.; Courtillon, C.; Weerts, E.A.W.S.; Andraud, M.; Allée, C.; Vendembeuche, A.; Amelot, M.; Rose, N.; Verheije, M.H.; Eterradossi, N. Transmission kinetics and histopathology induced by European turkey coronavirus during experimental infection of specific pathogen free turkeys. Transbound. Emerg. Dis. 2019, 66, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Guy, J. Turkey Coronavirus Enteritis, 4th ed.; Swayne, D.E., Boulianne, M., McDougald, L.R., Nair, V., Suarez, D.L., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2020; pp. 402–408. [Google Scholar]
- Gomaa, M.H.; Yoo, D.; Ojkic, D.; Barta, J.R. Use of recombinant s1 spike polypeptide to develop a tcov-specific antibody ELISA. Vet. Microbiol. 2009, 138, 281–288. [Google Scholar] [CrossRef]
- Breslin, J.J.; Smith, L.G.; Fuller, F.J.; Guy, J.S. Sequence analysis of the turkey coronavirus nucleocapsid protein gene and 3′ untranslated region identifies the virus as a close relative of infectious bronchitis virus. Virus Res. 1999, 65, 187–193. [Google Scholar] [CrossRef]
- Larsen, C. Turkey Coronavirus (TCV): Cleanup and prevention of TCV enteritis. In Proceedings and Technical Supplement of Roche Animal Nutrition and Health-Turkey Coronavirus Workshop; Clark, S.R., Ed.; Roche: Raleigh, NC, USA, 1998; pp. 181–183. [Google Scholar]
- McNulty, M.S.; Curran, W.L.; McFerran, J.B. Detection of astroviruses in turkey faeces by direct electron microscopy. Vet. Rec. 1980, 106, 561. [Google Scholar] [CrossRef]
- Sellers, H.S.; Koci, M.D.; Linnemann, E.; Kelley, L.A.; Schultz-Cherry, S. Development of a multiplex reverse transcription–polymerase chain reaction diagnostic test specific for turkey astrovirus and coronavirus. Avian Dis. 2004, 48, 531–538. [Google Scholar] [CrossRef]
- Adebiyi, A.I.; Mcilwaine, K.; Oluwayelu, D.O.; Smyth, V.J. Detection and characterization of chicken astrovirus associated with hatchery disease in commercial day-old turkeys in Southwestern Nigeria. Arch. Virol. 2021, 166, 1607–1614. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, D.L.; Saif, Y.M. Astrovirus: A cause of an enteric disease in turkey poults. Avian Dis. 1986, 30, 728–735. [Google Scholar] [CrossRef]
- Koci, M.D.; Seal, B.S.; Schultz-Cherry, S. Development of an RT-PCR diagnostic test for an avian astrovirus. J. Virol. Methods 2000, 90, 79–83. [Google Scholar] [CrossRef]
- Jindal, N.; Patnayak, D.P.; Chander, Y.; Ziegler, A.F.; Goyal, S.M. Detection and molecular characterization of enteric viruses from poult enteritis syndrome in turkeys. Poult. Sci. 2010, 89, 217–226. [Google Scholar] [CrossRef]
- Pantin-Jackwood, M.J.; Spackman, E.; Day, J.M.; Rives, D. Periodic monitoring of commercial turkeys for enteric viruses indicates continuous presence of astrovirus and rotavirus on the farms. Avian Dis. 2007, 51, 674–680. [Google Scholar] [CrossRef]
- Lagan Tregaskis, P.; Devaney, R.; Smyth, V.J. The first whole genome sequence and characterisation of avian nephritis virus genotype 3. Viruses 2021, 13, 235. [Google Scholar] [CrossRef]
- Behling-Kelly, E.; Schultz-Cherry, S.; Koci, M.; Kelley, L.; Larsen, D.; Brown, C. Localization of astrovirus in experimentally infected turkeys as determined by in situ hybridization. Vet. Pathol. 2002, 39, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Murphy, F.A.; Gibbs, E.P.J.; Horzinek, M.C.; Studdert, M.J. Astroviridae. In Veterinary Virology, 3rd ed.; Murphy, F.A., Gibbs, E.P.J., Horzinek, M.C., Und Studdert, M.J., Eds.; Academic Press: San Diego, CA, USA, 1999. [Google Scholar]
- Ismail, M.M.; Tang, A.Y.; Saif, Y.M. Pathogenicity of turkey coronavirus in turkeys and chickens. Avian Dis. 2003, 47, 515–522. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thouvenelle, M.L.; Haynes, J.S.; Sell, J.L.; Reynolds, D.L. Astrovirus infection in hatchling turkeys: Alterations in intestinal maltase activity. Avian Dis. 1995, 39, 343–348. [Google Scholar] [CrossRef]
- Pantin-Jackwood, M.J.; Day, J.M.; Jackwood, M.W.; Spackman, E. Enteric viruses detected by molecular methods in commercial chicken and turkey flocks in the united states between 2005 and 2006. Avian Dis. 2008, 52, 235–244. [Google Scholar] [CrossRef]
- Koci, M.D.; Moser, L.A.; Kelley, L.A.; Larsen, D.; Brown, C.C.; Schultz-Cherry, S. Astrovirus induces diarrhea in the absence of inflammation and cell death. J. Virol. 2003, 77, 11798–11808. [Google Scholar] [CrossRef] [PubMed]
- Schultz-Cherry, S.; Kapczynski, D.R.; Simmons, V.M.; Koci, M.D.; Brown, C.; Barnes, H.J. Identifying agent(s) associated with poult enteritis mortality syndrome: Importance of the thymus. Avian Dis. 2000, 44, 256–265. [Google Scholar] [CrossRef]
- Doszpoly, A.; Harrach, B.; LaPatra, S.; Benkő, M. Unconventional gene arrangement and content revealed by full genome analysis of the white sturgeon adenovirus, the single member of the genus Ichtadenovirus. Infect. Genet. Evol. 2019, 75, 103976. [Google Scholar] [CrossRef] [PubMed]
- Jejesky de Oliveira, A.P.; Valdetaro Rangel, M.C.; Z. Vidovszky, M.; Rossi, J.L., Jr.; Vicentini, F.; Harrach, B.; L. Kaján, G. Identification of two novel adenoviruses in smooth-billed ani and tropical screech Owl. PLoS ONE 2020, 15, e0229415. [Google Scholar] [CrossRef]
- Wellehan, J.F.X.; Johnson, A.J.; Harrach, B.; Benkö, M.; Pessier, A.P.; Johnson, C.M.; Garner, M.M.; Childress, A.; Jacobson, E.R. Detection and analysis of six lizard adenoviruses by consensus primer PCR provides further evidence of a reptilian origin for the atadenoviruses. J. Virol. 2004, 78, 13366–13369. [Google Scholar] [CrossRef]
- Harrach, B.; Benkő, M.; Both, G.W.; Brown, M.; Davison, A.J.; Echavarrı’a, M. Family Adenoviridae. In King AMQ, Lefkowitz E; Adams, M.J., Carstens, E.B., Eds.; Virus Taxonomy. IXth Report of the International Committee on Taxonomy of Viruses; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Kaján, G.L.; Stefancsik, R.; Ursu, K.; Palya, V.; Benko, M. The first complete genome sequence of a non-chicken aviadenovirus, proposed to be turkey adenovirus 1. Virus Res. 2010, 153, 226–233. [Google Scholar] [CrossRef]
- Kleine, A.; Hafez, H.M.; Lüschow, D. Investigations on aviadenoviruses isolated from turkey flocks in Germany. Avian Pathol. 2017, 46, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Marek, A.; Ballmann, M.Z.; Kosiol, C.; Harrach, B.; Schlötterer, C.; Hess, M. Whole-genome sequences of two turkey adenovirus types reveal the existence of two unknown lineages that merit the establishment of novel species within the genus Aviadenovirus. J. Gen. Virol. 2014, 95, 156–170. [Google Scholar] [CrossRef]
- Guy, J.S.; Schaeffer, J.L.; Barnes, H.J. Inclusion-body hepatitis in day-old turkeys. Avian Dis. 1988, 32, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Shivaprasad, H.L.; Woolcock, P.R.; McFarland, M.D. Group I avian adenovirus and avian adeno-Associated virus in turkey poults with inclusion body hepatitis. Avian Pathol. 2001, 30, 661–666. [Google Scholar] [CrossRef]
- Guy, J.S. Virus infections of the gastrointestinal tract of poultry. Poult. Sci. 1998, 77, 1166–1175. [Google Scholar] [CrossRef]
- Tykałowski, B.; Śmiałek, M.; Koncicki, A.; Ognik, K.; Zduńczyk, Z.; Jankowski, J. The immune response of young turkeys to haemorrhagic enteritis virus infection at different levels and sources of methionine in the diet. BMC Vet. Res. 2019, 15, 387. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, S.D.; Piersonn, F.W. Hemorrhagic enteritis and related infections. In Disease of Poultry, 12th ed.; Fadly, A.M., Glisson, J.R., Mcdougald, L.R., Nolan, L.K., Swayne, D.E., Saif, Y.M., Eds.; Iowa State University Press: Ames, IA, USA, 2013. [Google Scholar]
- Estes, M.; Greenberg, H. Rotaviruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Martella, V.; Bányai, K.; Matthijnssens, J.; Buonavoglia, C.; Ciarlet, M. Zoonotic aspects of rotaviruses. Vet. Microbiol. 2010, 140, 246–255. [Google Scholar] [CrossRef]
- Matthijnssens, J.; Van Ranst, M. Genotype constellation and evolution of group a rotaviruses infecting humans. Curr. Opin. Virol. 2012, 2, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Bányai, K.; Kemenesi, G.; Budinski, I.; Földes, F.; Zana, B.; Marton, S.; Varga-Kugler, R.; Oldal, M.; Kurucz, K.; Jakab, F. Candidate new rotavirus species in schreiber’s bats, Serbia. Infect. Genet. Evol. 2017, 48, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Theil, K.W.; Reynolds, D.L.; Saif, Y.M. Comparison of immune electron microscopy and genome electropherotyping techniques for detection of turkey rotaviruses and rotaviruslike viruses in intestinal contents. J. Clin. Microbiol. 1986, 23, 695–699. [Google Scholar] [CrossRef]
- McNulty, M.S.; Curran, W.L.; Todd, D.; McFerran, J.B. Detection of viruses in avian faeces by direct electron microscopy. Avian Pathol. 1979, 8, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Barrera, M.; Kumar, P.; Porter, R.E.; Goyal, S.M.; Mor, S.K. Retrospective analysis of turkey arthritis reovirus diagnostic submissions in minnesota. Avian Dis. 2019, 63, 681. [Google Scholar] [CrossRef]
- Day, J.M.; Spackman, E.; Pantin-Jackwood, M. A multiplex RT-PCR test for the differential identification of turkey astrovirus type 1, turkey astrovirus type 2, chicken astrovirus, avian nephritis virus, and avian rotavirus. Avian Dis. 2007, 51, 681–684. [Google Scholar] [CrossRef]
- Jones, R.C.; Islam, M.R.; Kelly, D.F. Early pathogenesis of experimental reovirus infection in chickens. Avian Pathol. 1989, 18, 239–253. [Google Scholar] [CrossRef]
- van der Heide, L. The history of avian reovirus. Avian Dis. 2000, 44, 638–641. [Google Scholar] [CrossRef]
- Mor, S.K.; Kumar, R.; Sobhy, N.M.; Singh, A.; Kakrudi, N.; Marusak, R.A.; Goyal, S.M.; Porter, R.E. Enteric viruses associated with mid-growth turkey enteritis. Avian Dis. 2020, 64, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Wooley, R.E.; Dees, T.A.; Cromack, A.S.; Gratzek, J.B. Infectious enteritis of turkeys: Characterization of two reoviruses isolated by sucrose density gradient centrifugation from turkeys with infectious enteritis. Am. J. Vet. Res. 1972, 33, 157–164. [Google Scholar]
- Sharafeldin, T.A.; Mor, S.K.; Bekele, A.Z.; Verma, H.; Noll, S.L.; Goyal, S.M.; Porter, R.E. Experimentally induced lameness in turkeys inoculated with a newly emergent turkey reovirus. Vet. Res. 2015, 46, 11. [Google Scholar] [CrossRef] [PubMed]
- Sharafeldin, T.A.; Mor, S.K.; Sobhy, N.M.; Xing, Z.; Reed, K.M.; Goyal, S.M.; Porter, R.E. A newly emergent turkey arthritis reovirus shows dominant enteric tropism and induces significantly elevated innate antiviral and t helper-1 cytokine responses. PLoS ONE 2015, 10, e0144085. [Google Scholar] [CrossRef] [PubMed]
- Palmer, E.L.; Martin, M.L. Electron Microscopy in Viral Diagnosis; CRC Press Inc.: Boca Raton, FL, USA, 1988. [Google Scholar]
- Doane, F.W.; Anderson, N. Electron microscopy in diagnostic virology; A practical guide and atlas; Cambridge University Press: New York, NY, USA, 1987. [Google Scholar]
- Madeley, C.R.; Field, A.M. Virus morphology, 2rd ed.; Churchill Livingstone: New York, NY, USA, 1988. [Google Scholar]
- Laue, M.; Bannert, N. Detection limit of negative staining electron microscopy for the diagnosis of bioterrorism-related micro-organisms. J. Appl. Microbiol. 2010, 109, 1159–1168. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Ismail, M.M.; Saif, Y.M. Development of antigen-capture enzyme-linked immunosorbent assay and RT-PCR for detection of turkey astroviruses. Avian Dis. 2005, 49, 182–188. [Google Scholar] [CrossRef]
- Gast, R.K.; Porter, R.E. Salmonella infections. In Diseases of Poultry; Swayne, D.E., Boulianne, M., Logue, C.M., McDougald, L.R., Nair, V., Suarez, D.L., Wit, S., Grimes, T., Johnson, D., Kromm, M., et al., Eds.; Wiley: Hoboken, NJ, USA, 2020; pp. 717–753. ISBN 978-1-119-37116-8. [Google Scholar]
- Bozzola, J.J.; Russell, L.D. Specimen staining and contrast methods for transmission electron microscopy. In Electron Microscopy. Principles and Techniques for Biologists, 2nd ed.; Bozzola, J.J., Und Russell, L.D., Eds.; Jones & Bartlett Publishers: Sudbery, MA, USA, 1998. [Google Scholar]
- Moura-Alvarez, J.; Chacon, J.V.; Scanavini, L.S.; Nuñez, L.F.N.; Astolfi-Ferreira, C.S.; Jones, R.C.; Piantino Ferreira, A.J. Enteric viruses in Brazilian turkey flocks: Single and multiple virus infection frequency according to age and clinical signs of intestinal disease. Poult. Sci. 2013, 92, 945–955. [Google Scholar] [CrossRef] [PubMed]
- Hess, M.; Raue, R.; Hafez, H.M. PCR for specific detection of haemorrhagic enteritis virus of turkeys, an avian adenovirus. J. Virol. Methods 1999, 81, 199–203. [Google Scholar] [CrossRef]
- Liu, H.J.; Giambrone, J.J.; Nielsen, B.L. Molecular characterization of avian reoviruses using nested PCR and nucleotide sequence analysis. J. Virol. Methods 1997, 65, 159–167. [Google Scholar] [CrossRef]
- Xie, Z.; Fadl, A.A.; Girshick, T.; Khan, M.I. Detection of avian adenovirus by polymerase chain reaction. Avian Dis. 1999, 43, 98. [Google Scholar] [CrossRef]
- Bunger, A.N.D.; Chacón, J.L.; Jones, R.C.; Ferreira, A.J.P. Detection and molecular characterization of gene 3 and 5 of turkey coronavirus from turkeys with severe enteritis in Brazil. Avian Dis. 2009, 53, 356–362. [Google Scholar] [CrossRef]
- Nuñez, L.F.N.; Ferreira, A.J.P. Viral agents related to enteric disease in commercial chicken flocks, with special reference to latin america. World’s Poult. Sci. J. 2013, 69, 853–864. [Google Scholar] [CrossRef]
- El-Adawy, H.; Hotzel, H.; Neubauer, H.; Hafez, H.M. Detection of some turkey enteric pathogens by single multiplex polymerase chain reaction. In Proceedings of the 9th “Hafez” International Symposium on Turkey Production, Berlin, Germany, 18–20 May 2017. [Google Scholar]
- Stephensen, C.B.; Casebolt, D.B.; Gangopadhyay, N.N. Phylogenetic analysis of a highly conserved region of the polymerase gene from 11 coronaviruses and development of a consensus polymerase chain reaction assay. Virus Res. 1999, 60, 181–189. [Google Scholar] [CrossRef]
- Cavanagh, D. Coronaviruses in poultry and other birds. Avian Pathol. 2005, 34, 439–448. [Google Scholar] [CrossRef]
- Jonassen, C.M.; Kofstad, T.; Larsen, I.-L.; Løvland, A.; Handeland, K.; Follestad, A.; Lillehaug, A. Molecular identification and characterization of novel coronaviruses infecting graylag geese (Anser anser), feral pigeons (Columbia livia) and mallards (Anas platyrhynchos). J. Gen. Virol. 2005, 86, 1597–1607. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, D.; Mawditt, K.; Sharma, M.; Drury, S.E.; Ainsworth, H.L.; Britton, P.; Gough, R.E. Detection of a coronavirus from turkey poults in europe genetically related to infectious bronchitis virus of chickens. Avian Pathol. 2001, 30, 355–368. [Google Scholar] [CrossRef]
- Loa, C.C.; Lin, T.L.; Wu, C.C.; Bryan, T.A.; Thacker, H.L.; Hooper, T.; Schrader, D. Detection of antibody to turkey coronavirus by antibody-capture enzyme-linked immunosorbent assay utilizing infectious bronchitis virus antigen. Avian Dis. 2000, 44, 498–506. [Google Scholar] [CrossRef]
- Abdelwahab, M.; Loa, C.C.; Wu, C.C.; Lin, T.L. Recombinant nucleocapsid protein-based enzyme-linked immunosorbent assay for detection of antibody to turkey coronavirus. J. Virol. Methods 2015, 217, 36–41. [Google Scholar] [CrossRef]
- Reynolds, D.; Schulz-Cherry, S. Astrovirus infections. In Diseases of Poultry, 12th ed.; Saif, Y.M., Fadly, A.M., Glisson, J.R., McDouglad, L.R., Nolan, L.K., und Swayne, D.E., Eds.; Iowa State University Press: Ames, IA, USA, 2008; pp. 351–356. [Google Scholar]
- Madeley, C.R.; Cosgrove, B.P. Letter: 28 nm particles in faeces in infantile gastroenteritis. Lancet 1975, 2, 451–452. [Google Scholar] [CrossRef]
- Iturriza-Gómara, M.; Green, J.; Brown, D.W.; Desselberger, U.; Gray, J.J. Diversity within the VP4 gene of rotavirus p[8] strains: Implications for reverse transcription-pcr genotyping. J. Clin. Microbiol. 2000, 38, 898–901. [Google Scholar] [CrossRef] [PubMed]
- Meulemans, G.; Boschmans, M.; Berg, T.P.; Decaesstecker, M. Polymerase chain reaction combined with restriction enzyme analysis for detection and differentiation of fowl adenoviruses. Avian Pathol. 2001, 30, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Coulouris, G.; Zaretskaya, I.; Cutcutache, I.; Rozen, S.; Madden, T.L. Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinform. 2012, 13, 134. [Google Scholar] [CrossRef]
- Kirkwood, C. Rotavirus. In PCR for Clinical Microbiology: An Australian and International Perspective; Schuller, M., Sloots, T.P., James, G.S., Halliday, C.L., Und, C.I.W.J., Eds.; Springer: Dordrecht, The Netherlands, 2010. [Google Scholar]
- Gouvea, V.; Glass, R.I.; Woods, P.; Taniguchi, K.; Clark, H.F.; Forrester, B.; Fang, Z.Y. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. J. Clin. Microbiol. 1990, 28, 276–282. [Google Scholar] [CrossRef]
- Rodríguez-Díaz, J.; Rubilar-Abreu, E.; Spitzner, M.; Hedlund, K.-O.; Liprandi, F.; Svensson, L. Design of a multiplex nested PCR for genotyping of the nsp4 from group a rotavirus. J. Virol. Methods 2008, 149, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Gernat, A.A.; Santos, F.B.O.; Grimes, J.L. Alternative antimicrobials in the turkey industry—Challenges and perspectives. Ger. J. Vet. Res. 2021, 3. in press. [Google Scholar]
- Batkowska, J.; Brodacki, A.; Tomczyk, G. The influence of probiotic bacteria (Bacillus Toyoi) on livability and performance of young meat-type turkeys. Rev. Bras. Cienc. Avic. 2015, 17, 433–438. [Google Scholar] [CrossRef]
- Grimes, J.L.; Rahimi, S.; Oviedo, E.; Sheldon, B.W.; Santos, F.B.O. Effects of a direct-fed microbial (Primalac) on turkey poult performance and susceptibility to oral Salmonella challenge. Poult. Sci. 2008, 87, 1464–1470. [Google Scholar] [CrossRef] [PubMed]
- Mohammadigheisar, M.; Shirley, R.B.; Barton, J.; Welsher, A.; Thiery, P.; Kiarie, E. Growth performance and gastrointestinal responses in heavy tom turkeys fed antibiotic free corn−soybean meal diets supplemented with multiple doses of a single strain Bacillus subtilis probiotic (DSM29784). Poult. Sci. 2019, 98, 5541–5550. [Google Scholar] [CrossRef]
- Russell, S.M.; Grimes, J.L. The Effect of a direct-fed microbial (primalac) on turkey live performance. J. Appl. Poult. Res. 2009, 18, 185–192. [Google Scholar] [CrossRef]
- Torres-Rodriguez, A.; Donoghue, A.M.; Donoghue, D.J.; Barton, J.T.; Tellez, G.; Hargis, B.M. Performance and condemnation rate analysis of commercial turkey flocks treated with a Lactobacillus spp.-based probiotic. Poult. Sci. 2007, 86, 444–446. [Google Scholar] [CrossRef] [PubMed]
- Tellez-Isaias, V.; Christine, N.V.; Brittany, D.G.; Callie, M.S.; Lucas, E.G.; Roberto, S.; Thaina, L.B.; Lesleigh, C.B.; Makenly, E.C.; Aaron, J.F.; et al. Developing probiotics, prebiotics, and organic acids to control Salmonella spp. in commercial turkeys at the University of Arkansas USA. Ger. J. Vet. Res. 2021, 3, 7–12. [Google Scholar] [CrossRef]
- Yang, Y.; Latorre, J.D.; Khatri, B.; Kwon, Y.M.; Kong, B.W.; Teague, K.D.; Graham, L.E.; Wolfenden, A.D.; Mahaffey, B.D.; Baxter, M.; et al. Characterization and evaluation of lactic acid bacteria candidates for intestinal epithelial permeability and Salmonella Typhimurium colonization in neonatal turkey poults. Poult. Sci. 2018, 97, 515–521. [Google Scholar] [CrossRef]
- Arreguin-Nava, M.A.; Hernández-Patlán, D.; Solis-Cruz, B.; Latorre, J.D.; Hernandez-Velasco, X.; Tellez, G.; El-Ashram, S.; Hargis, B.M.; Tellez-Isaias, G. Isolation and identification of lactic acid bacteria probiotic culture candidates for the treatment of Salmonella enterica serovar Enteritidis in neonatal turkey poults. Animals 2019, 9, 696. [Google Scholar] [CrossRef]
- Higgins, S.E.; Torres-Rodriguez, A.; Vicente, J.L.; Sartor, C.D.; Pixley, C.M.; Nava, G.M.; Tellez, G.; Barton, J.T.; Hargis, B.M. Evaluation of intervention strategies for idiopathic diarrhea in commercial turkey brooding houses. J. Appl. Poult. Res. 2005, 14, 345–348. [Google Scholar] [CrossRef]
- Leyva-Diaz, A.A.; Hernandez-Patlan, D.; Solis-Cruz, B.; Adhikari, B.; Kwon, Y.M.; Latorre, J.D.; Hernandez-Velasco, X.; Fuente-Martinez, B.; Hargis, B.M.; Lopez-Arellano, R.; et al. Evaluation of curcumin and copper acetate against Salmonella Typhimurium infection, intestinal permeability, and cecal microbiota composition in broiler chickens. J. Anim. Sci. Biotechnol. 2021, 12, 23. [Google Scholar] [CrossRef] [PubMed]
- Nair, D.V.T.; Johnson, T.J.; Noll, S.L.; Kollanoor Johny, A. Effect of supplementation of a dairy-originated probiotic bacterium, Propionibacterium freudenreichii subsp. freudenreichii, on the cecal microbiome of turkeys challenged with multidrug-resistant Salmonella Heidelberg. Poult. Sci. 2021, 100, 283–295. [Google Scholar] [CrossRef]
- do Diniz Nascimento, L.; de Moraes, A.A.B.; da Costa, K.S.; Pereira Galúcio, J.M.; Taube, P.S.; Costa, C.M.L.; Neves Cruz, J.; de Aguiar Andrade, E.H.; de Faria, L.J.G. Bioactive natural compounds and antioxidant activity of essential oils from spice plants: New findings and potential applications. Biomolecules 2020, 10, 988. [Google Scholar] [CrossRef]
- Nieto, G. A review on applications and uses of thymus in the food industry. Plants 2020, 9, 961. [Google Scholar] [CrossRef]
- Bampidis, V.A.; Christodoulou, V.; Florou-Paneri, P.; Christaki, E.; Chatzopoulou, P.S.; Tsiligianni, T.; Spais, A.B. Effect of dietary dried oregano leaves on growth performance, carcase characteristics and serum cholesterol of female early maturing turkeys. Br. Poult. Sci. 2005, 46, 595–601. [Google Scholar] [CrossRef]
- Hafez, H.M.; Hauck, R. Efficacy of a herbal product against Histomonas Meleagridis after experimental infection of turkey poults. Arch. Anim. Nutr. 2006, 60, 436–442. [Google Scholar] [CrossRef]
- Zhang, S.; Shen, Y.R.; Wu, S.; Xiao, Y.Q.; He, Q.; Shi, S.R. The dietary combination of essential oils and organic acids reduces Salmonella Enteritidis in challenged chicks. Poult. Sci. 2019, 98, 6349–6355. [Google Scholar] [CrossRef] [PubMed]
- Giannenas, I.; Papaneophytou, C.P.; Tsalie, E.; Pappas, I.; Triantafillou, E.; Tontis, D.; Kontopidis, G.A. Dietary supplementation of benzoic acid and essential oil compounds affects buffering capacit y of the feeds, performance of turkey poults and their antioxidant status, ph in the digestive tract, intestinal microbiota and morphology. Asian Australas. J. Anim. Sci. 2014, 27, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Cetin, E.; Yibar, A.; Yesilbag, D.; Cetin, I.; Cengiz, S.S. The effect of volatile oil mixtures on the performance and ilio-caecal microflora of broiler chickens. Br. Poult. Sci. 2016, 57, 780–787. [Google Scholar] [CrossRef]
- Shehata, A.A.; Lüschow, D.; Hafez, H.M. History and current status of Marek’s disease in turkeys. Ger. J. Vet. Res. 2021, 3, 1–6. [Google Scholar] [CrossRef]
- Reynolds, J.A.; Kastello, M.D.; Harrington, D.G.; Crabbs, C.L.; Peters, C.J.; Jemski, J.V.; Scott, G.H.; Di Luzio, N.R. Glucan-induced enhancement of host resistance to selected infectious diseases. Infect. Immun. 1980, 30, 51–57. [Google Scholar] [CrossRef]
- Cleary, J.A.; Kelly, G.E.; Husband, A.J. The effect of molecular weight and β-1,6-linkages on priming of macrophage function in mice by (1,3)-β-d-Glucan. Immunol. Cell. Biol. 1999, 77, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Tzianabos, A.O. Polysaccharide immunomodulators as therapeutic agents: Structural aspects and biologic function. Clin. Microbiol. Rev. 2000, 13, 523–533. [Google Scholar] [CrossRef]
- Lowry, V.K.; Farnell, M.B.; Ferro, P.J.; Swaggerty, C.L.; Bahl, A.; Kogut, M.H. Purified β-Glucan as an abiotic feed additive up-regulates the innate immune response in immature chickens against Salmonella enterica serovar Enteritidis. Int. J. Food Microbiol. 2005, 98, 309–318. [Google Scholar] [CrossRef]
- Cetin, N.; Güçlü, B.K.; Cetin, E. The effects of probiotic and mannanoligosaccharide on some haematological and immunological parameters in turkeys. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2005, 52, 263–267. [Google Scholar] [CrossRef]
- Grimes, J.L.; Sharara, M.; Kolar, P. Considerations in selecting turkey bedding materials. Ger. J. Vet. Res. 2021, 3, 28–39. [Google Scholar] [CrossRef]

| Infectious | Viral | Newcastle disease virus (Paramyxoviridae) Avian influenza A (Orthomyxoviridae) Infectious bursal disease virus (Birnaviridae) Hemorrhagic enteritis virus (Adenoviridae) Coronavirus enteritis (Coronaviridae) Rotavirus (Reoviridae) Reovirus (Orthoreoviridae) Astrovirus (Astroviridiae) Enterovirus (Picornaviridae) Parvovirus (Parvoviridae) |
| Bacterial | Salmonella spp. E. coli Clostridia spp. Chlamydia spp. | |
| Mycotic | Candida | |
| Parasitic | Ascaridia Coccidia | |
| Non-infectious | Nutritional | Feed structure, palatability, energy content and pellet quality |
| Management | Temperature, stocking density, available feed space, available water space, distribution of feeders and air quality |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shehata, A.A.; Basiouni, S.; Sting, R.; Akimkin, V.; Hoferer, M.; Hafez, H.M. Poult Enteritis and Mortality Syndrome in Turkey Poults: Causes, Diagnosis and Preventive Measures. Animals 2021, 11, 2063. https://doi.org/10.3390/ani11072063
Shehata AA, Basiouni S, Sting R, Akimkin V, Hoferer M, Hafez HM. Poult Enteritis and Mortality Syndrome in Turkey Poults: Causes, Diagnosis and Preventive Measures. Animals. 2021; 11(7):2063. https://doi.org/10.3390/ani11072063
Chicago/Turabian StyleShehata, Awad A., Shereen Basiouni, Reinhard Sting, Valerij Akimkin, Marc Hoferer, and Hafez M. Hafez. 2021. "Poult Enteritis and Mortality Syndrome in Turkey Poults: Causes, Diagnosis and Preventive Measures" Animals 11, no. 7: 2063. https://doi.org/10.3390/ani11072063
APA StyleShehata, A. A., Basiouni, S., Sting, R., Akimkin, V., Hoferer, M., & Hafez, H. M. (2021). Poult Enteritis and Mortality Syndrome in Turkey Poults: Causes, Diagnosis and Preventive Measures. Animals, 11(7), 2063. https://doi.org/10.3390/ani11072063







