Spinal Locomotion in Cats Following Spinal Cord Injury: A Prospective Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Characterization of the Population
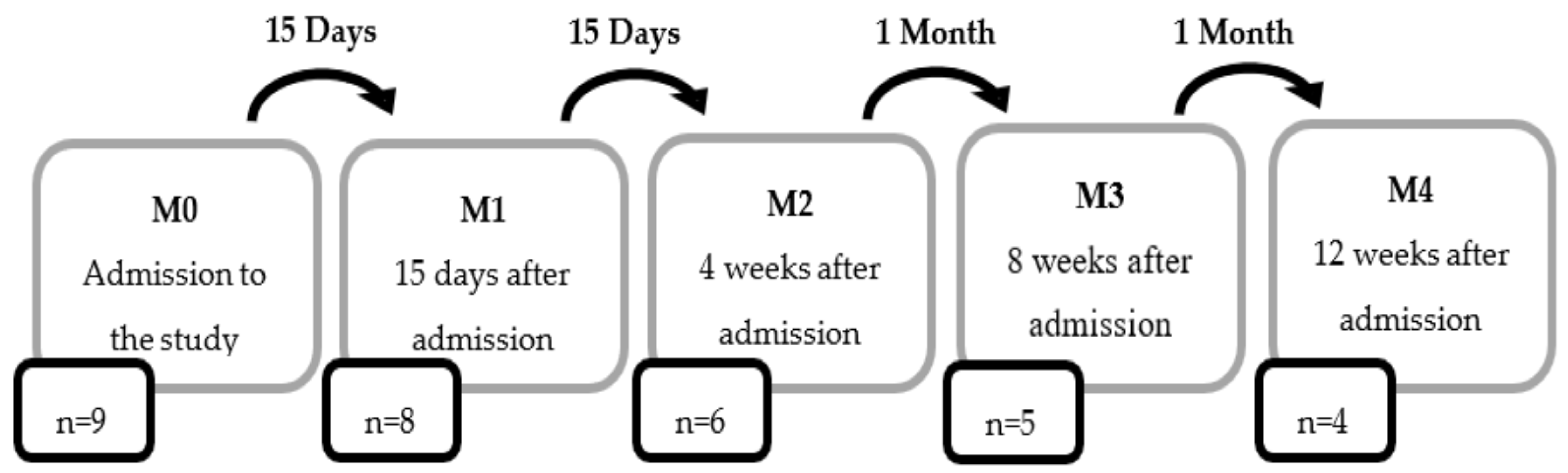
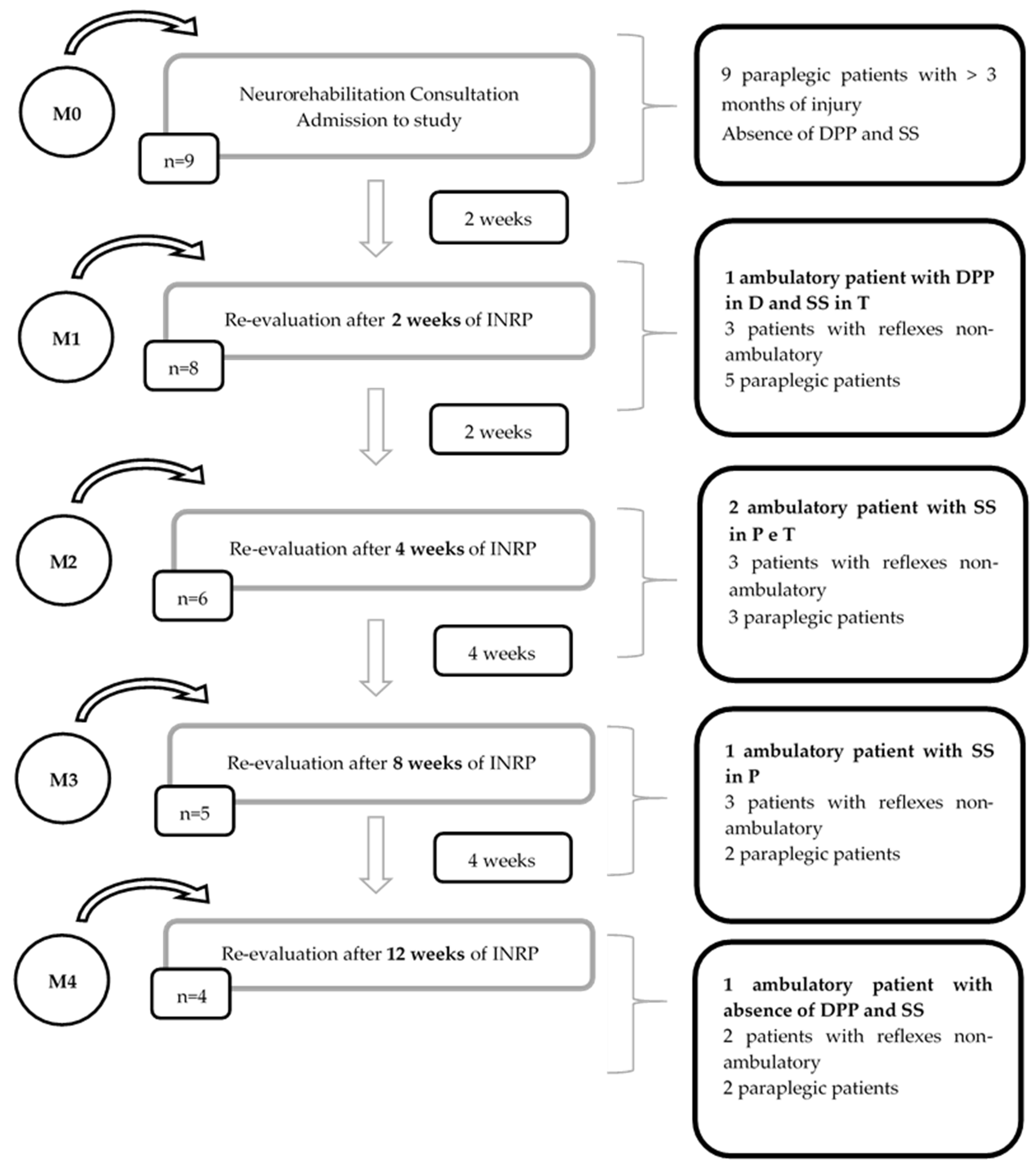
2.2. Plan of the Study
2.3. Neurorehabilitation Consultation at Study Admission
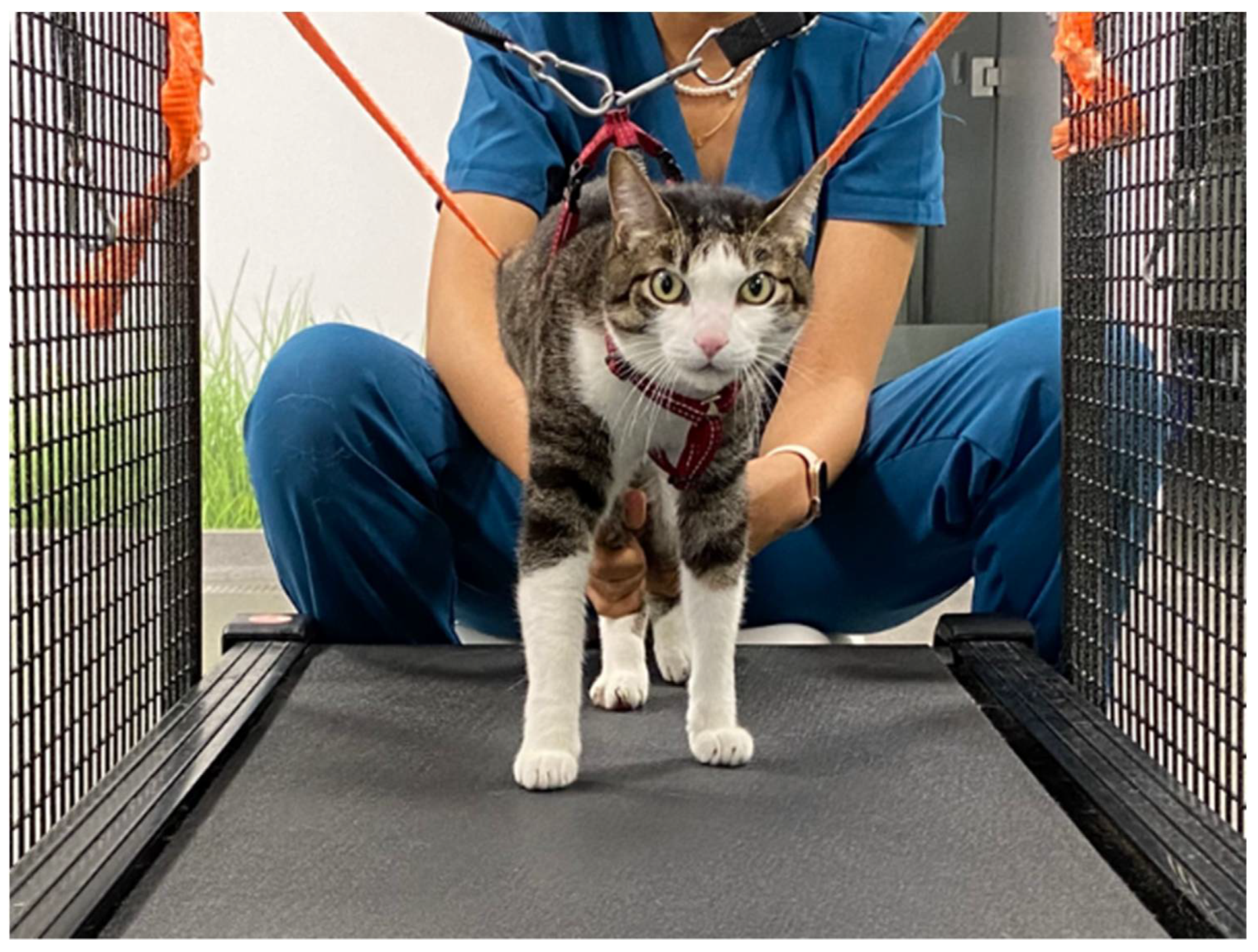
2.4. Intensive Neurorehabilitation Protocol
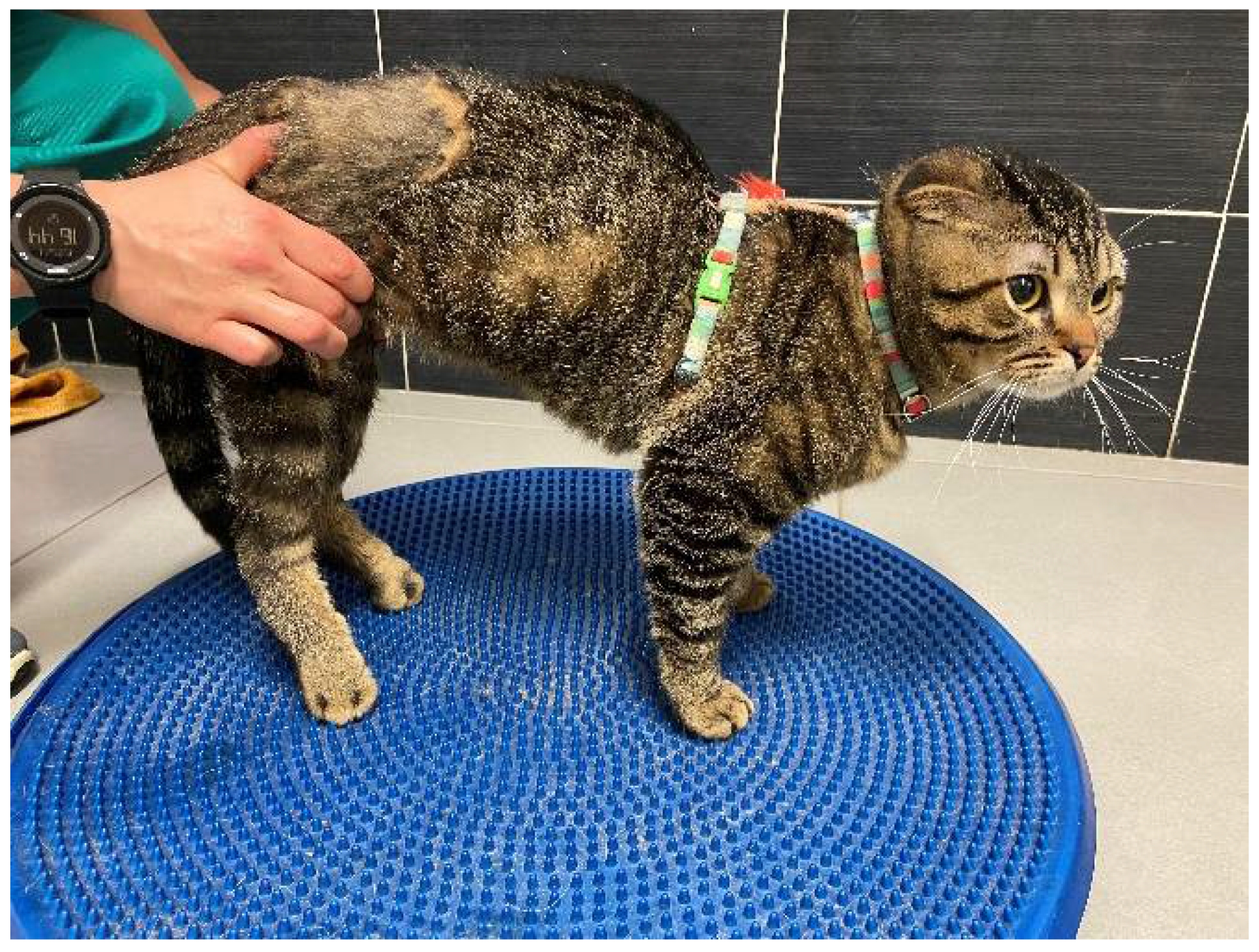
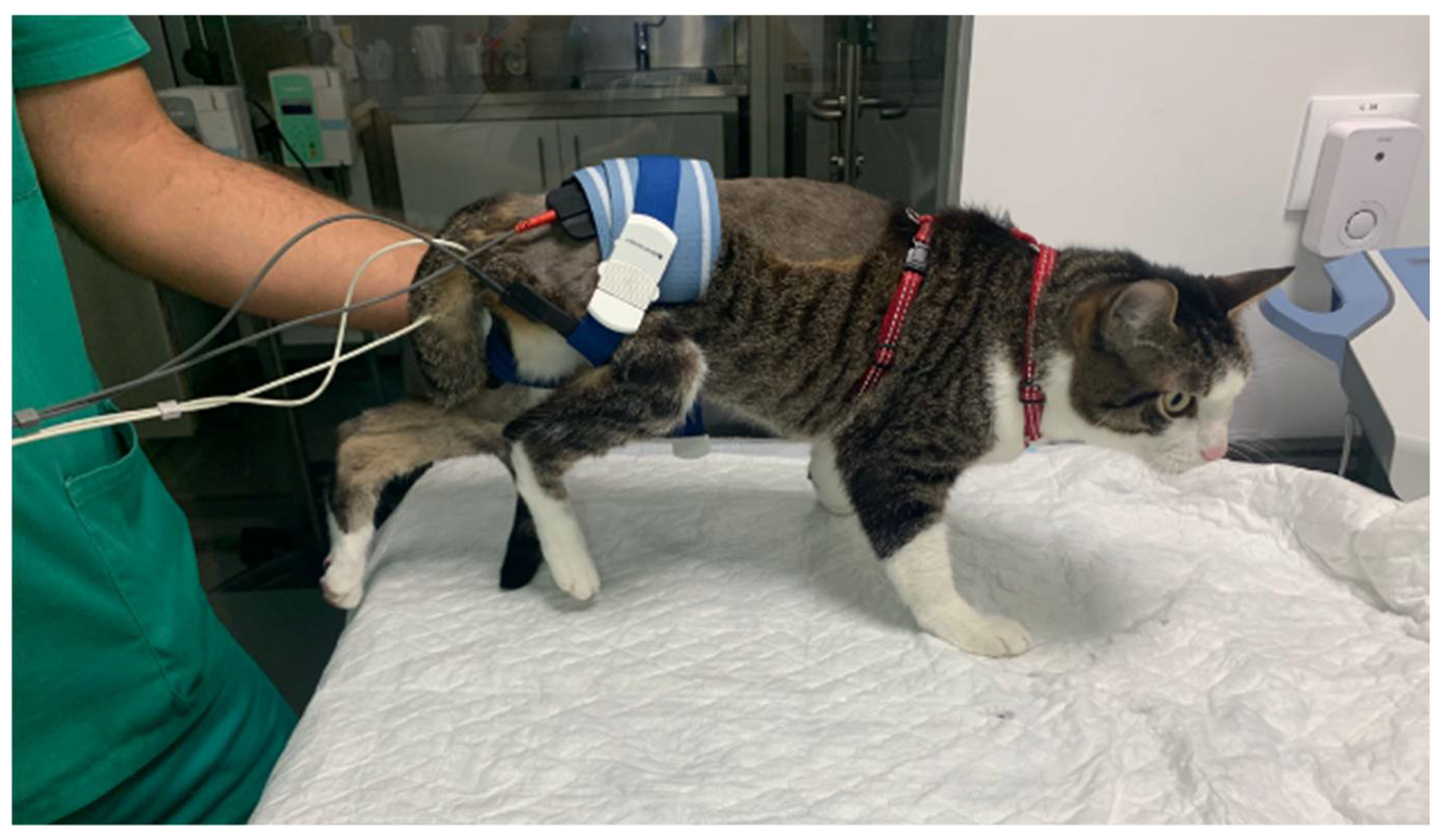
2.5. Monitoring of the Study Population
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thomson, C.; Hahn, C. Veterinary Neuroanatomy A Clinical Approach; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Uemura, E. Sistema Motor. In Dukes: Fisiologia Dos Animais Domésticos, 13th ed.; Dukes, H.H., Reece, W.O., Eds.; Guanabara Koogan: Rio De Janeiro, Brasil, 2017. [Google Scholar]
- Mackay-Lyons, M. Central Pattern Generation of Locomotion: A Review of the Evidence. Phys. Ther. 2002, 82, 69–83. Available online: https://academic.oup.com/ptj/article-abstract/82/1/69/2837028 (accessed on 10 June 2021). [CrossRef] [PubMed]
- Rossignol, S.; Frigon, A. Recovery of locomotion after spinal cord injury: Some facts and mechanisms. Annu. Rev. Neurosci. 2011, 34, 413–440. [Google Scholar] [CrossRef] [PubMed]
- Forssberg, H.; Grillner, S.; Halbertsma, J. The locomotion of the low spinal cat I. Coordination within a hindlimb. Acta Physiol. Scand. 1980, 108, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Forssberg, H.; Grillner, S.; Halbertsma, J.; Rossignol, S. The locomotion of the low spinal cat. II. Interlimb coordination. Acta Physiol. Scand. 1980, 108, 283–295. [Google Scholar] [CrossRef]
- Barbeau, H.; Rossignol, S. Recovery of locomotion after chronic spinalization in the adult cat. Brain Res. 1987, 412, 84–95. [Google Scholar] [CrossRef]
- Belanger, M.; Drew, T.; Provencher, J.; Rossignol, S. A Comparison of Treadmill Locomotion in Adult Cats Before and After Spinal Transection. J. Neurophysiol. 1996, 76, 471–491. Available online: www.physiology.org/journal/jn (accessed on 10 June 2021). [CrossRef] [PubMed]
- Barthélemy, D.; Leblond, H.; Provencher, J.; Rossignol, S. Nonlocomotor and locomotor hindlimb responses evoked by electrical microstimulation of the lumbar cord in spinalized cats. J. Neurophysiol. 2006, 96, 3273–3292. [Google Scholar] [CrossRef]
- Chau, C.; Barbeau, H.; Rossignol, S. Early Locomotor Training With Clonidine in Spinal Cats. J. Neurophysiol. 1998, 79, 392–409. [Google Scholar] [CrossRef]
- Graham Brown, T. On The Nature of The Fundamental Activity of The Nervous Centers; Together With an Analysis of The Conditioning of Rhythimic Activity in Progression, and a Theory of The Evolution of Functional in The Nervous System. J. Physiol. 1914, 48, 18–46. [Google Scholar] [CrossRef] [PubMed]
- Grillner, S.; Zangger, P. On the Central Generation of Locomotion in the Low Spinal Cat. Exp. Brain Res. 1979, 34, 241–261. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, E.; Jukes, M.G.M.; Lund, S.; Lundberg, A. The Effect of DOPA on the Spinal Cord 6. Half-centre organization of interneurones transmitting effects from the flexor reflex afferents. Acta Physiol. Scand. 1967, 70, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Harkema, S.; Edgerton, R. Basic concepts of activity-based interventions for improved recovery of motor function after spinal cord injury. Arch. Phys. Med. Rehabil. 2012, 93, 1487–1497. [Google Scholar] [CrossRef] [PubMed]
- Burns, A.S.; Marino, R.J.; Flanders, A.E.; Flett, H. Clinical diagnosis and prognosis following spinal cord injury. Handb. Clin. Neurol. 2012, 109, 47–62. [Google Scholar] [CrossRef]
- Burns, A.S.; Marino, R.J.; Kalsi-Ryan, S.; Middleton, J.W.; Tetreault, L.A.; Dettori, J.R.; Mihalovich, K.E.; Fehlings, M.G. Type and Timing of Rehabilitation Following Acute and Subacute Spinal Cord Injury: A Systematic Review. Glob. Spine J. 2017, 7, 175S–194S. [Google Scholar] [CrossRef]
- Edgerton, V.R.; de Leon, R.D.; Tillakaratne, N.; Recktenwald, M.R.; Hodgson, J.A.; Roy, R.R. Use-dependent plasticity in spinal stepping and standing. Adv. Neurol. 1997, 72, 233–247. [Google Scholar]
- Lovely, R.G.; Gregor, R.J.; Roy, R.R.; Edgerton, V.R. Effects of training on the recovery of full-weight-bearing stepping in the adult spinal cat. Exp. Neurol. 1986, 92, 421–435. [Google Scholar] [CrossRef]
- Rossignol, S.; Bouyer, L. Adaptive Mechanisms of Spinal Locomotion in Cats. Integr. Comp. Biol. 2004, 44, 71–79. Available online: https://academic.oup.com/icb/article/44/1/71/599761 (accessed on 10 June 2021). [CrossRef]
- Côté, M.P.; Ménard, A.; Gossard, J.P. Spinal cats on the treadmill: Changes in load pathways. J. Neurosci. Off. J. Soc. Neurosci. 2003, 23, 2789–2796. [Google Scholar] [CrossRef]
- De Leon, R.D.; Hodgson, J.A.; Roy, R.R.; Edgerton, V.R. Retention of hindlimb stepping ability in adult spinal cats after the cessation of step training. J. Neurophysiol. 1999, 81, 85–94. [Google Scholar] [CrossRef]
- Fouad, K.; Tetzlaff, W. Rehabilitative training and plasticity following spinal cord injury. Exp. Neurol. 2012, 235, 91–99. [Google Scholar] [CrossRef]
- Barbeau, I.; Chau, C.; Rossignol, S.; Noradrenergic, S. Noradrenergic Agonists and Locomotor Training Affect Locomotor Recovery After Cord Transection in Adult Cats. Brain Res. Bull. 1993, 30, 387–393. [Google Scholar] [CrossRef]
- Rossignol, S.; Barbeau, H.; Julien, C. Locomotion of the adult chronic spinal cat and its modification by monoaminergic agonists and antagonists. In Development and Plasticity of the Mammalian Spinal Cord; Goldberger, M., Gorio, A., Murray, M., Eds.; Liviana Press Padova: Padova, Italy, 1986; pp. 323–345. [Google Scholar]
- Frigon, A. Central pattern generators of the mammalian spinal cord. Neuroscientist 2012, 18, 56–69. [Google Scholar] [CrossRef]
- Lovely, R.G.; Gregor, R.J.; Roy, R.R.; Edgerton, V.R. Weight-bearing hindlimb stepping in treadmill-exercised adult spinal cats. Brain Res. 1990, 514, 206–218. [Google Scholar] [CrossRef]
- Barriére, G.; Leblond, H.; Provencher, J.; Rossignol, S. Prominent role of the spinal central pattern generator in the recovery of locomotion after partial spinal cord injuries. J. Neurosci. 2008, 28, 3976–3987. [Google Scholar] [CrossRef] [PubMed]
- Edgerton, V.R.; Roy, R.R.; Hodgson, J.A.; Prober, R.J.; de Guzman, C.P.; de Leon, R. A physiological basis for the development of rehabilitative strategies for spinally injured patients. J. Am. Paraplegia Soc. 1991, 14, 150–157. [Google Scholar] [CrossRef]
- Barbeau, H.; Rossignol, S. The effects of serotonergic drugs on the locomotor pattern and on cutaneous reflexes of the adult chronic spinal cat. Brain Res. 1991, 514, 55–67. [Google Scholar] [CrossRef]
- Barbeau, H.; Rossignol, S. Enhancement of locomotor recovery following spinal cord injury. Curr. Opin. Neurol. 1994, 7, 517–524. [Google Scholar] [CrossRef]
- Gerasimenko, Y.; Gad, P.; Sayenko, D.; Mckinney, Z.; Gorodnichev, R.; Puhov, A.; Moshonkina, T.; Savochin, A.; Selionov, V.; Shigueva, T.; et al. Rapid Report Integration of sensory, spinal, and volitional descending inputs in regulation of human locomotion. J. Neurophysiol. 2016, 116, 98–105. [Google Scholar] [CrossRef]
- Smith, J.L.; Smith, L.A.; Zernicke, R.F.; Hoy, M. Locomotion in Exercised and Nonexercised Cats Cordotomized at Two or Twelve Weeks of Age. Exp. Neurol. 1982, 76, 393–413. [Google Scholar] [CrossRef]
- Delivet-Mongrain, H.; Dea, M.; Gossard, J.-P.; Rossignol, S. Recovery of locomotion in cats after severe contusion of the low thoracic spinal cord. J. Neurophysiol. 2020, 123, 1504–1525. [Google Scholar] [CrossRef]
- Lewis, M.J.; Jeffery, N.D.; Olby, N.J.; The Canine Spinal Cord Injury Consortium (CANSORT-SCI). Ambulation in Dogs With Absent Pain Perception After Acute Thoracolumbar Spinal Cord Injury. Front. Vet. Sci. 2020, 7, 560. [Google Scholar] [CrossRef]
- Dimitrijevic, M.R.; Dimitrijevic, M.M.; Faganel, J.; Sherwood, A.M. Suprasegmentally Induced Motor Unit Activity in Paralyzed Muscles of Patients with Established Spinal Cord Injury. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 1984, 16, 216–221. [Google Scholar] [CrossRef]
- Dimitrijevic, M.R.; Haller, J.A.; Sharkey, P.C.; Sherwood, A.M. Epidural Spinal Cord Stimulation and Carry-Over Effect in Chronic Spinal Cord Injury Patients. Appl. Neurophysiol. 1987, 50, 449–450. [Google Scholar] [CrossRef]
- Gerasimenko, Y.; Sayenko, D.; Gad, P.; Liu, C.T.; Tillakaratne, N.J.K.; Roy, R.R.; Kozlovskaya, I.; Edgerton, V.R. Feed-Forwardness of Spinal Networks in Posture and Locomotion. Neuroscientist 2017, 23, 441–453. [Google Scholar] [CrossRef]
- Militskova, A.; Mukhametova, E.; Fatykhova, E.; Sharifullin, S.; Cuellar, C.A.; Calvert, J.S.; Grahn, P.J.; Baltina, T.; Lavrov, I. Supraspinal and Afferent Signaling Facilitate Spinal Sensorimotor Network Excitability After Discomplete Spinal Cord Injury: A Case Report. Front. Neurosci. 2020, 14, 552. [Google Scholar] [CrossRef]
- Forssberg, H.; Grillner, S. The locomotion of the acute spinal cat injected with clonidine iv. Brain Res. 1973, 50, 184–186. [Google Scholar] [CrossRef]
- Giroux, N.; Chau, C.; Barbeau, H.; Reader, T.A.; Rossignol, S. Effects of intrathecal glutamatergic drugs on locomotion. II. NMDA and AP-5 in intact and late spinal cats. J. Neurophysiol. 2003, 90, 1027–1045. [Google Scholar] [CrossRef]
- Delcomyn, F. Neural basis of rhythmic behavior in animals. Science 1980, 210, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Pearson, K.G.; Rossignol, S. Fictive Motor Patterns in Chronic Spinal Cats. J. Neurophysiol. 1991, 66, 1874–1887. Available online: www.physiology.org/journal/jn (accessed on 10 June 2021). [CrossRef] [PubMed]
- Saltiel, P.; Rossignol, S. Critical points in the forelimb fictive locomotor cycle and motor coordination: Effects of phasic retractions and protractions of the shoulder in the cat. J. Neurophysiol. 2004, 92, 1342–1356. [Google Scholar] [CrossRef]
- Côté, M.P.; Azzam, G.A.; Lemay, M.A.; Zhukareva, V.; Houlé, J.D. Activity-dependent increase in neurotrophic factors is associated with an enhanced modulation of spinal reflexes after spinal cord injury. J. Neurotrauma 2011, 28, 299–309. [Google Scholar] [CrossRef]
- Rossignol, S.; Barrière, G.; Frigon, A.; Barthélemy, D.; Bouyer, L.; Provencher, J.; Leblond, H.; Bernard, G. Plasticity of locomotor sensorimotor interactions after peripheral and/or spinal lesions. Brain Res. Rev. 2008, 57, 228–240. [Google Scholar] [CrossRef]
- Viala, D.; Vidal, C. Evidence for distinct spinal locomotion generators supplying respectively fore-and hindlimbs in the rabbit. Brain Res. 1978, 155, 182–186. [Google Scholar] [CrossRef]
- Juvin, L.; Simmers, J.; Morin, D. Propriospinal circuitry underlying interlimb coordination in mammalian quadrupedal locomotion. J. Neurosci. 2005, 25, 6025–6035. [Google Scholar] [CrossRef]
- Gerasimenko, Y.; Musienko, P.; Bogacheva, I.; Moshonkina, T.; Savochin, A.; Lavrov, I.; Roy, R.R.; Edgerton, V.R. Propriospinal bypass of the serotonergic system that can facilitate stepping. J. Neurosci. 2009, 29, 5681–5689. [Google Scholar] [CrossRef]
- Gosgnach, S. The role of genetically-defined interneurons in generating the mammalian locomotor rhythm. Integr. Comp. Biol. 2011, 51, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Rybak, I.A.; Dougherty, K.J.; Shevtsova, N.A. Organization of the mammalian locomotor CPG: Review of computational model and circuit architectures based on genetically identified spinal interneurons. eNeuro 2015, 2. [Google Scholar] [CrossRef]
- Fawcett, J.W.; Curt, A.; Steeves, J.D.; Coleman, W.P.; Tuszynski, M.H.; Lammertse, D.; Bartlett, P.F.; Blight, A.R.; Dietz, V.; Ditunno, J.; et al. Guidelines for the conduct of clinical trials for spinal cord injury as developed by the ICCP panel: Spontaneous recovery after spinal cord injury and statistical power needed for therapeutic clinical trials. Spinal Cord 2007, 45, 190–205. [Google Scholar] [CrossRef] [PubMed]
- Angeli, C.A.; Edgerton, V.R.; Gerasimenko, Y.P.; Harkema, S.J. Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 2014, 137, 1394–1409. [Google Scholar] [CrossRef] [PubMed]
- Escalona, M.; Delivet-Mongrain, H.; Kundu, A.; Gossard, J.P.; Rossignol, S. Ladder treadmill: A method to assess locomotion in cats with an intact or lesioned spinal cord. J. Neurosci. 2017, 37, 5429–5446. [Google Scholar] [CrossRef] [PubMed]
- Harkema, S.; Gerasimenko, Y.; Hodes, J.; Burdick, J.; Angeli, C.; Chen, Y.; Ferreira, C.; Willhite, A.; Rejc, E.; Grossman, R.G.; et al. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: A case study. Lancet 2011, 377, 1938–1947. [Google Scholar] [CrossRef]
- Cramer, S.C.; Sur, M.; Dobkin, B.H.; O’Brien, C.; Sanger, T.D.; Trojanowski, J.Q.; Rumsey, J.M.; Hicks, R.; Cameron, J.; Chen, D.; et al. Harnessing neuroplasticity for clinical applications. Brain 2011, 134, 1591–1609. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Amatya, B.; Galea, M.P.; Gonzenbach, R.; Kesselring, J. Neurorehabilitation: Applied neuroplasticity. J. Neurol. 2017, 264, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, S.; Dubuc, R.; Gossard, J.P. Dynamic sensorimotor interactions in locomotion. Physiol. Rev. 2006, 86, 89–154. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, S.; Chau, C.; Giroux, N.; Brustein, E.; Bouyer, L.; Marcoux, J.; Langlet, C.; Barthel6my, D.; Provencher, J.; Leblond, H.; et al. The cat model of spinal injury. Prog. Brain Res. 2002, 137, 151–168. [Google Scholar] [PubMed]
- Jaggy, A.; Platt, S. Small Animal Neurology An Illustrated Text; Schlütersche Verlagsgesellschaft mbH & Co. KG: Hannover, Germany, 2010. [Google Scholar]
- Maier, I.C.; Ichiyama, R.M.; Courtine, G.; Schnell, L.; Lavrov, I.; Edgerton, V.R.; Schwab, M.E. Differential effects of anti-Nogo-A antibody treatment and treadmill training in rats with incomplete spinal cord injury. Brain 2009, 132, 1426–1440. [Google Scholar] [CrossRef]
- Tillakaratne, N.J.K.; Duru, P.; Fujino, H.; Zhong, H.; Xiao, M.S.; Edgerton, V.R.; Roy, R.R. Identification of interneurons activated at different inclines during treadmill locomotion in adult rats. J. Neurosci. Res. 2014, 92, 1714–1722. [Google Scholar] [CrossRef]
- Edgerton, R.; de Leon, R.; Harkema, S.; Hodgson, J.; London, N.; Reinkensmeyer, D.; Roy, R.; Talmadge, R.; Tillakaratne, N.; Timoszyk, W.; et al. Retraining the injured spinal cord. J. Physiol. 2001, 533, 15–22. [Google Scholar] [CrossRef]
- Martinez, M.; Delivet-Mongrain, H.; Leblond, H.; Rossignol, S. Effect of locomotor training in completely spinalized cats previously submitted to a spinal hemisection. J. Neurosci. 2012, 32, 10961–10970. [Google Scholar] [CrossRef]
- Forssberg, H. Stumbling corrective reaction: A phase-dependent compensatory reaction during locomotion. J. Neurophysiol. 1979, 42, 936–953. [Google Scholar] [CrossRef] [PubMed]
- Mccrea, D.A. Topical Review Spinal circuitry of sensorimotor control of locomotion. J. Physiol. Symp. Spinal Cord Funct. Rehabil. 2001, 533, 41–50. [Google Scholar] [CrossRef]
- Aikawa, T.; Shibata, M.; Asano, M.; Hara, Y.; Tagawa, M.; Orima, H. A comparison of thoracolumbar intervertebral disc extrusion in French Bulldogs and Dachshunds and association with congenital vertebral anomalies. Vet. Surg. VS 2014, 43, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Olby, N.; Harris, T.; Burr, J.; Muñana, K.; Sharp, N.; Keene, B. Recovery of pelvic limb function in dogs following acute intervertebral disc herniations. J. Neurotrauma 2004, 21, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Olby, N.J.; da Costa, R.C.; Levine, J.M.; Stein, V.M.; The Canine Spinal Cord Injury Consortium (CANSORT SCI). Prognostic Factors in Canine Acute Intervertebral Disc Disease. Front. Vet. Sci. 2020, 7, 596059. [Google Scholar] [CrossRef]
| Locomotor Training | ||
|---|---|---|
| Animal | Treadmill | Underwater Treadmill |
| I | Bipedal first 15 days and then quadrupedal | Bipedal first 15 days and then quadrupedal |
| II | Bipedal | Not tolerated |
| III | Bipedal first 15 days and then quadrupedal | Bipedal first 15 days and then quadrupedal |
| IV | Bipedal first 15 days and then quadrupedal | Bipedal first 7 days and then quadrupedal |
| V | Quadrupedal | Not tolerated |
| VI | Quadrupedal | Quadrupedal |
| VII | Bipedal first 15 days and then quadrupedal | Not tolerated |
| VIII | Bipedal first 15 days and then quadrupedal | Not tolerated |
| IX | Bipedal | Bipedal |
| Animal | FR | CTR | CER | RA | ||||
|---|---|---|---|---|---|---|---|---|
| Before INRP | After INRP | Before INRP | After INRP | Before INRP | After INRP | Before INRP | After INRP | |
| I | Present bilateral | Normal | Present at the site of lesion * | Recovery (since the lumbosacral region) | Present bilateral | Normal/Modeled with flexor reflex | Normal | Normal |
| II | Present bilateral exaggerated | Clonic | Present at the site of lesion * | Present at the site of lesion * | Present bilateral | Normal/Modeled with flexor reflex | Decreased | Normal |
| III | Present bilateral | Normal | Present at the site of lesion * | Recovery (since the lumbosacral region) | Present bilateral | Normal/Modeled with flexor reflex | Decreased | Normal |
| IV | Present bilateral decreased | Normal | Present since the lumbosacral region | Recovery (since the lumbosacral region) | Present bilateral decreased | Normal/Modeled with flexor reflex | Decreased | Normal |
| V | Present bilateral decreased | Normal | Present at the site of lesion * | Recovery (since the lumbosacral region) | Present bilateral | Normal/Modeled with flexor reflex | Decreased | Normal |
| VI | Present bilateral | Normal | Present at the site of lesion * | Recovery (since the lumbosacral region) | Present bilateral | Normal/Modeled with flexor reflex | Normal | Normal |
| VII | Present bilateral | Normal | Absent at the site of lesion | Present at the site of lesion * | Present bilateral decreased | Decreased | Decreased | Normal |
| VIII | Present bilateral decreased | Normal | Absent at the site of lesion | Present at the site of lesion * | Absent | Decreased | Decreased | Decreased |
| IX | Present bilateral decreased | Normal | Absent at the site of lesion | Absent at the site of lesion | Present bilateral decreased | Decreased | Decreased | Decreased |
| Neurological Grade before INRP Neurological Grade after INRP | ||
|---|---|---|
| I | Paraplegic | Functional spinal locomotion by reflexes |
| II | Paraplegic | Non-functional spinal locomotion by reflexes |
| III | Paraplegic | Functional spinal locomotion by reflexes |
| IV | Paraplegic | Paraparesis ambulatory with proprioceptive ataxia |
| V | Paraplegic | Functional spinal locomotion by reflexes |
| VI | Paraplegic | Functional spinal locomotion by reflexes |
| VII | Paraplegic | Non-functional spinal locomotion by reflexes |
| VIII | Paraplegic | Paraplegic |
| IX | Paraplegic | Paraplegic |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, Â.; Silva, C.M.; Gouveia, D.; Cardoso, A.; Coelho, T.; Gamboa, Ó.; Marcelino, E.; Ferreira, A. Spinal Locomotion in Cats Following Spinal Cord Injury: A Prospective Study. Animals 2021, 11, 1994. https://doi.org/10.3390/ani11071994
Martins Â, Silva CM, Gouveia D, Cardoso A, Coelho T, Gamboa Ó, Marcelino E, Ferreira A. Spinal Locomotion in Cats Following Spinal Cord Injury: A Prospective Study. Animals. 2021; 11(7):1994. https://doi.org/10.3390/ani11071994
Chicago/Turabian StyleMartins, Ângela, Cátia Marina Silva, Débora Gouveia, Ana Cardoso, Tiago Coelho, Óscar Gamboa, Eduardo Marcelino, and António Ferreira. 2021. "Spinal Locomotion in Cats Following Spinal Cord Injury: A Prospective Study" Animals 11, no. 7: 1994. https://doi.org/10.3390/ani11071994
APA StyleMartins, Â., Silva, C. M., Gouveia, D., Cardoso, A., Coelho, T., Gamboa, Ó., Marcelino, E., & Ferreira, A. (2021). Spinal Locomotion in Cats Following Spinal Cord Injury: A Prospective Study. Animals, 11(7), 1994. https://doi.org/10.3390/ani11071994





