Energy Requirements for Growth in the Norfolk Terrier
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Husbandry
2.2. Diet Management
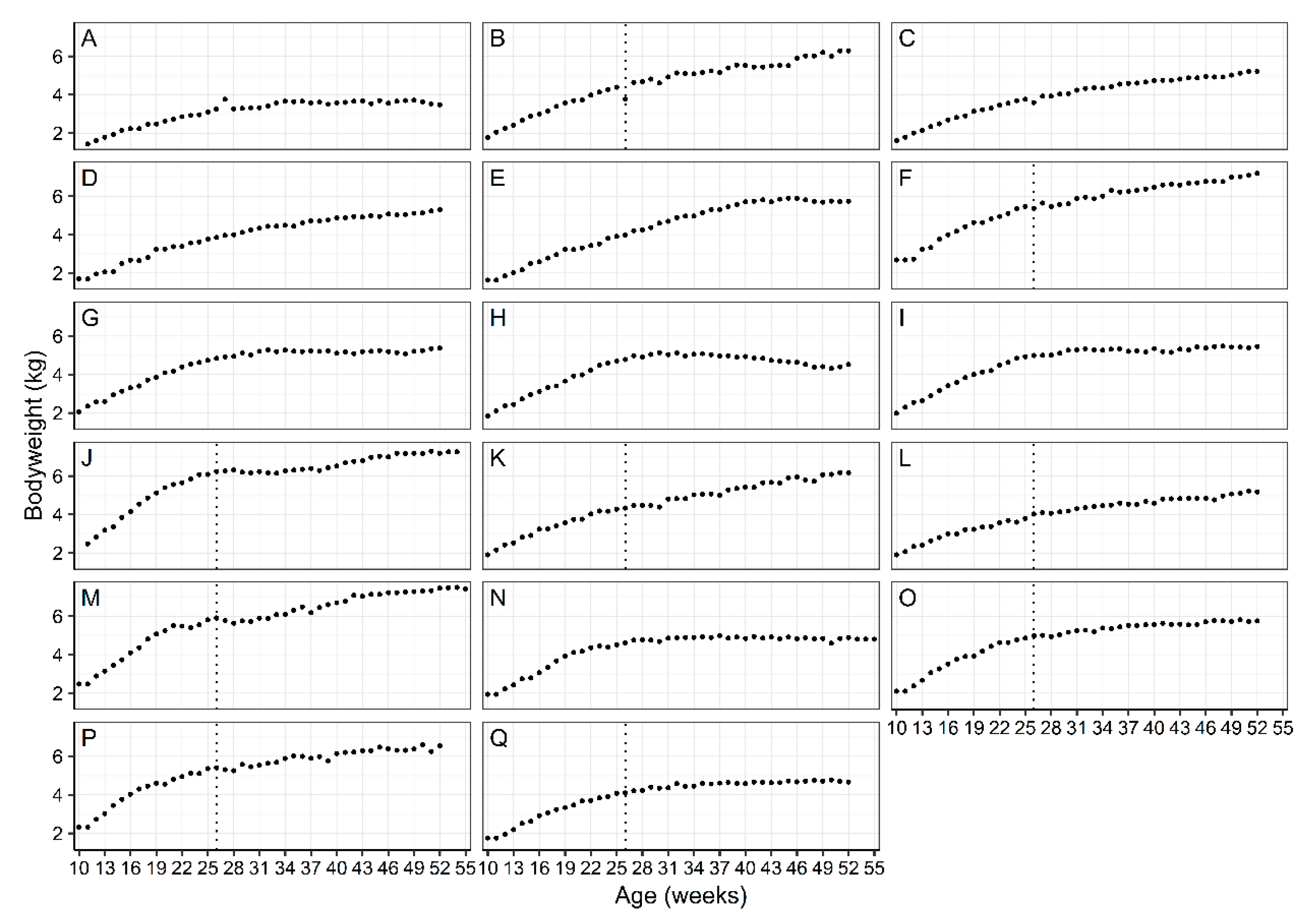
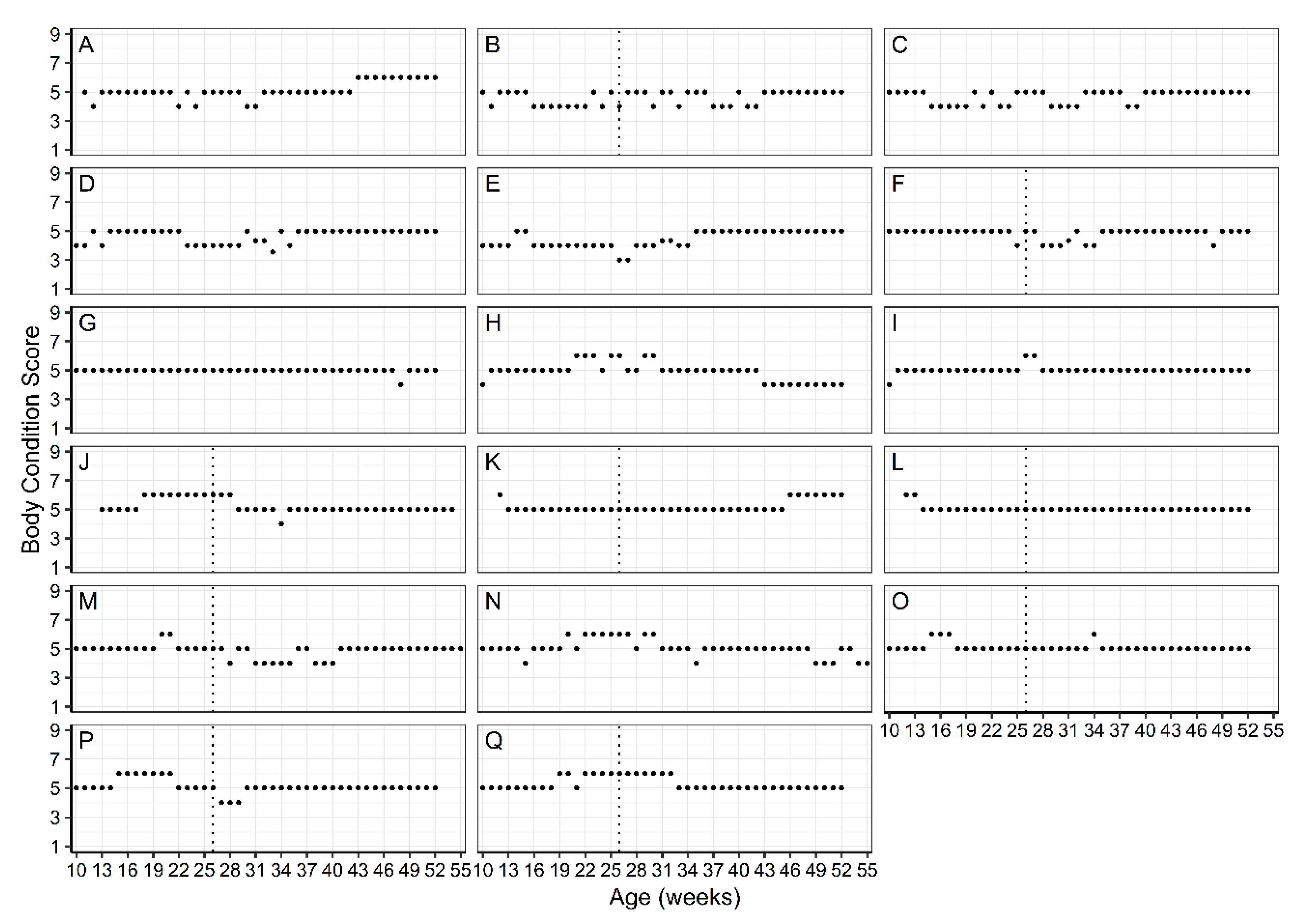
2.3. Bodyweight Management
2.4. Sample Collection and Analysis
2.5. Statistical Analysis
3. Results
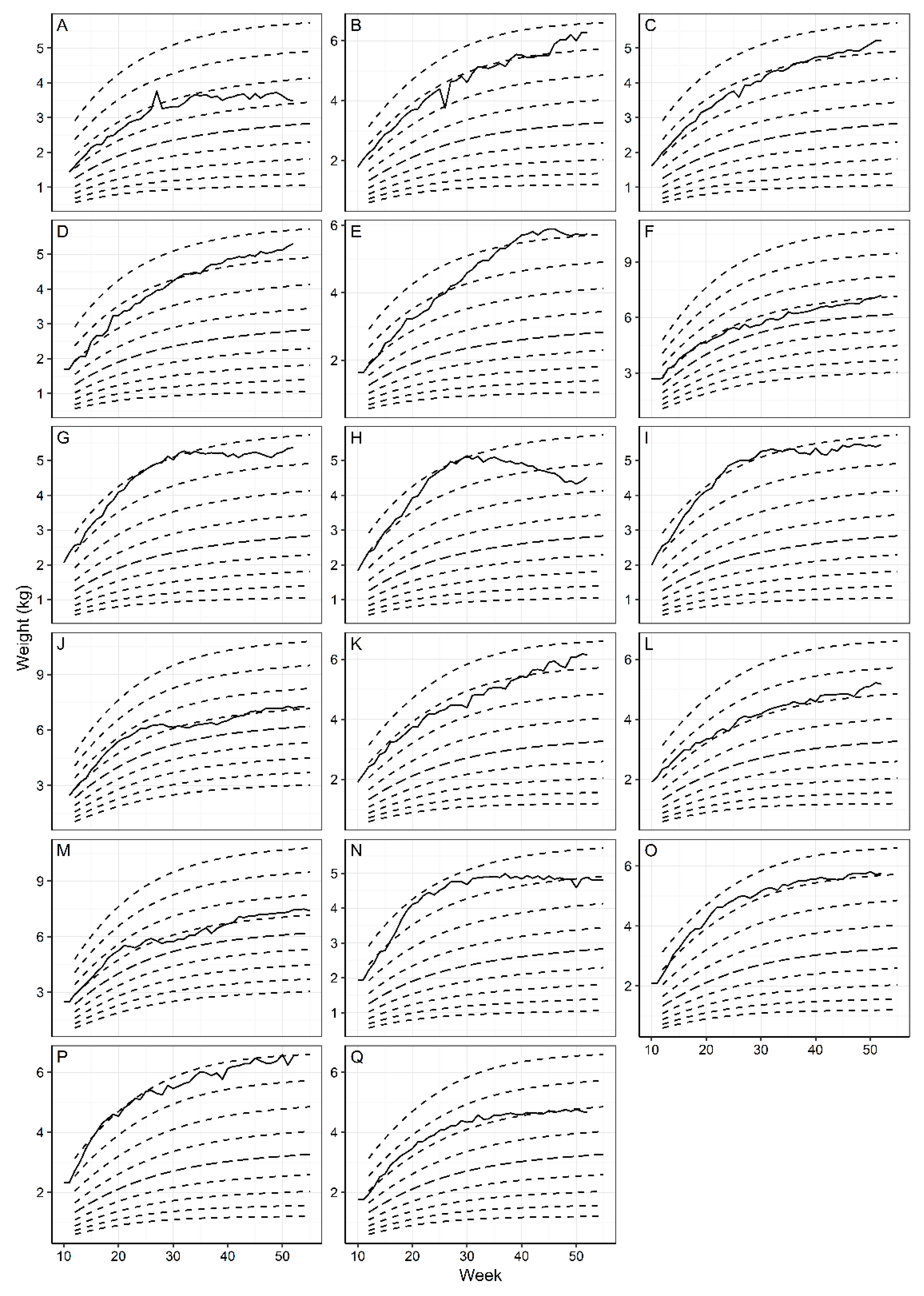
3.1. WALTHAM™ Puppy Growth Charts
3.2. Energy Intake
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lund, E.M.; Armstrong, P.J.; Kirk, C.A.; Klausner, J.S. Prevalence and risk factors for obesity in adult dogs from private US veterinary practices. Intern. J. Appl. Res. Vet. Med. 2006, 4, 177. [Google Scholar]
- Courcier, E.A.; Thomson, R.M.; Mellor, D.J.; Yam, P.S. An epidemiological study of environmental factors associated with canine obesity. J. Small Anim. Pract. 2010, 51, 362–367. [Google Scholar] [CrossRef]
- Mao, J.; Xia, Z.; Chen, J.; Yu, J. Prevalence and risk factors for canine obesity surveyed in veterinary practices in Beijing, China. Prev. Vet. Med. 2013, 112, 438–442. [Google Scholar] [CrossRef]
- Montoya-Alonso, J.A.; Bautista-Castaño, I.; Peña, C.; Suárez, L.; Juste, M.C.; Tvarijonaviciute, A. Prevalence of Canine Obesity, Obesity-Related Metabolic Dysfunction, and Relationship with Owner Obesity in an Obesogenic Region of Spain. Front. Vet. Sci. 2017, 4, 59. [Google Scholar] [CrossRef]
- Biagi, G.; Cipollini, I.; Grandi, M.; Sarti, D.; Zaghini, G. Preliminary Investigation of the Incidence of Obesity in a Canine Population in the USA. In Trends in Veterinary Sciences; Springer Science and Business Media LLC: New York, NY, USA, 2013; pp. 171–175. [Google Scholar]
- Payan-Carreira, R.; Sargo, T.; Nascimento, M.M. Canine obesity in Portugal: Perceptions on occurrence and treatment determinants. Acta Vet. Scand. 2015, 57, 1. [Google Scholar] [CrossRef]
- Bockstahler, B.A.; Hulse, D.A.; Carmichael, S.; Marshall, W.G. A review of osteoarthritis and obesity: Current understanding of the relationship and benefit of obesity treatment and prevention in the dog. Vet. Comp. Orthop. Traumatol. 2009, 22, 339–345. [Google Scholar] [CrossRef]
- Marshall, W.G.; Hazewinkel, H.A.W.; Mullen, D.; De Meyer, G.; Baert, K.; Carmichael, S. The effect of weight loss on lameness in obese dogs with osteoarthritis. Veter- Res. Commun. 2010, 34, 241–253. [Google Scholar] [CrossRef]
- Pettitt, R.A.; German, A.J. Investigation and management of canine osteoarthritis. Practice. 2015, 37, 1–8. [Google Scholar] [CrossRef]
- Christmann, U.; Bečvářová, I.; Werre, S.; Meyer, H.P. Effectiveness of a New Dietetic Food to Achieve Weight Loss and to Improve Mobility in Client-Owned Obese Dogs with Osteoarthritis. Int. J. Appl. Res. Vet. Med. 2018, 16. [Google Scholar] [CrossRef]
- Johnson, L.R.; Pollard, R.E. Tracheal collapse and bronchomalacia in dogs: 58 cases (7/2001–1/2008). J. Vet. Intern. Med. 2010, 24, 298–305. [Google Scholar] [CrossRef]
- Tappin, S.W. Canine tracheal collapse. J. Small Anim. Pr. 2016, 57, 9–17. [Google Scholar] [CrossRef]
- Zatloukal, J.; Lorenzová, J.; Tichý, F.; Necas, A.; Kecova, H.; Kohout, P. Breed and Age as Risk Factors for Canine Mammary Tumours. Acta Vet. Brno 2005, 74, 103–109. [Google Scholar] [CrossRef]
- Lim, H.-Y.; Im, K.-S.; Kim, N.-H.; Kim, H.-W.; Shin, J.-I.; Yhee, J.-Y.; Sur, J.-H. Effects of Obesity and Obesity-Related Molecules on Canine Mammary Gland Tumors. Vet. Pathol. 2015, 52, 1045–1051. [Google Scholar] [CrossRef]
- German, A.J. The Growing Problem of Obesity in Dogs and Cats. J. Nutr. 2006, 136, 1940S–1946S. [Google Scholar] [CrossRef]
- Klinkenberg, H.; Sallander, M.H.; Hedhammar, A. Feeding, Exercise, and Weight Identified as Risk Factors in Canine Diabetes Mellitus. J. Nutr. 2006, 136, 1985S–1987S. [Google Scholar] [CrossRef]
- Hoenig, M. Comparative Aspects of Human, Canine, and Feline Obesity and Factors Predicting Progression to Diabetes. Vet. Sci. 2014, 1, 121–135. [Google Scholar] [CrossRef]
- Sandøe, P.; Palmer, C.; Corr, S.; Astrup, A.; Bjørnvad, C.R. Canine and feline obesity: A One Health perspective. Vet. Rec. 2014, 175, 610–616. [Google Scholar] [CrossRef]
- Salt, C.; Morris, P.J.; Wilson, D.; Lund, E.M.; German, A.J. Association between life span and body condition in neutered client-owned dogs. J. Vet. Intern. Med. 2018, 33, 89–99. [Google Scholar] [CrossRef]
- Reilly, J.J.; Armstrong, J.; Dorosty, A.R.; Emmett, P.M.; Ness, A.; Rogers, I.; Steer, C.; Sherriff, A. Early life risk factors for obesity in childhood: Cohort study. BMJ 2005, 330, 1357. [Google Scholar] [CrossRef]
- Dennison, B.A.; Edmunds, L.S.; Stratton, H.H.; Pruzek, R.M. Rapid Infant Weight Gain Predicts Childhood Overweight. Obesity 2006, 14, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Dubois, L.; Girard, M. Early determinants of overweight at 4.5 years in a population-based longitudinal study. Int. J. Obes. 2006, 30, 610–617. [Google Scholar] [CrossRef]
- Serisier, S.; Weber, M.; Feugier, A.; Fardet, M.-O.; Garnier, F.; Biourge, V.; German, A.J. Maintenance energy requirements in miniature colony dogs. J. Anim. Physiol. Anim. Nutr. 2013, 97, 60–67. [Google Scholar] [CrossRef]
- Salt, C.; Morris, P.J.; Butterwick, R.F.; Lund, E.M.; Cole, T.J.; German, A.J. Comparison of growth patterns in healthy dogs and dogs in abnormal body condition using growth standards. PLoS ONE 2020, 15, e0238521. [Google Scholar] [CrossRef] [PubMed]
- Kasström, H. Nutrition, weight gain and development of hip dysplasia. An experimental investigation in growing dogs with special reference to the effect of feeding intensity. Acta Radiol. 1975, 344, 135–179. [Google Scholar]
- Dobenecker, B.; Kienzle, E.; Köstlin, R.; Matis, U. Mal- and overnutrition in puppies with or without clinical disorders of skeletal development. J. Anim. Physiol. Anim. Nutr. 1998, 80, 76–81. [Google Scholar] [CrossRef]
- Dobenecker, B.; Endres, V.; Kienzle, E. Energy requirements of puppies of two different breeds for ideal growth from weaning to 28 weeks of age. J. Anim. Physiol. Anim. Nutr. 2011, 97, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J. Feeding large-breed puppies. Compend. Cont. Educ. Vet. 2010, 32, E1–E4. [Google Scholar]
- Bjørnvad, C.; Gloor, S.; Johansen, S.; Sandøe, P.; Lund, T. Neutering increases the risk of obesity in male dogs but not in bitches —A cross-sectional study of dog- and owner-related risk factors for obesity in Danish companion dogs. Prev. Vet. Med. 2019, 170, 104730. [Google Scholar] [CrossRef]
- Edney, A.T.; Smith, P.M. Study of obesity in dogs visiting veterinary practices in the United Kingdom. Vet. Rec. 1986, 118, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Robertson, I.D. The association of exercise, diet and other factors with owner-perceived obesity in privately owned dogs from metropolitan Perth, WA. Prev. Vet. Med. 2003, 58, 75–83. [Google Scholar] [CrossRef]
- McGreevy, P.D.; Thomson, P.C.; Pride, C.; Fawcett, A.; Grassi, T.; Jones, B. Prevalence of obesity in dogs examined by Australian veterinary practices and the risk factors involved. Veter Rec. 2005, 156, 695–702. [Google Scholar] [CrossRef]
- German, A.J.; Blackwell, E.; Evans, M.; Westgarth, C. Overweight dogs exercise less frequently and for shorter periods: Results of a large online survey of dog owners from the UK. J. Nutr. Sci. 2017, 6, e11. [Google Scholar] [CrossRef] [PubMed]
- Sallander, M.; Hagberg, M.; Hedhammar, Å.; Rundgren, M.; Lindberg, J.E. Energy-intake and activity risk factors for owner-perceived obesity in a defined population of Swedish dogs. Prev. Vet. Med. 2010, 96, 132–141. [Google Scholar] [CrossRef]
- Kienzle, E.; Bergler, R.; Mandernach, A. A Comparison of the Feeding Behavior and the Human–Animal Relationship in Owners of Normal and Obese Dogs. J. Nutr. 1998, 128, 2779S–2782S. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Nutrient Requirements of Dogs and Cats; National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Blanchard, G.; Grandjean, D.; Paragon, B.-M. Calculation of a dietary plan for puppies. J. Anim. Physiol. Anim. Nutr. 1998, 80, 54–59. [Google Scholar] [CrossRef]
- Meyer, H.; Zentek, J. Ernährung des Hundes [Nutrition of the Dog], 3rd ed.; Eugen Ulmer: Stuttgart, Germany, 1998. (in German) [Google Scholar]
- Klein, C.; Thes, M.; Böswald, L.F.; Kienzle, E. Metabolisable energy intake and growth of privately owned growing dogs in comparison with official recommendations on the growth curve and energy supply. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1952–1958. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.E.; Colyer, A.; Morris, P.J. Energy requirements for growth in the Yorkshire terrier. J. Nutr. Sci. 2017, 6, 6. [Google Scholar] [CrossRef]
- Laflamme, D. Development and validation of a body condition score system for dogs. Canine Pract. 1997, 22, 10–15. [Google Scholar]
- Salt, C.; Morris, P.J.; German, A.J.; Wilson, D.; Lund, E.M.; Cole, T.J.; Butterwick, R.F. Growth standard charts for monitoring bodyweight in dogs of different sizes. PLoS ONE 2017, 12, e0182064. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. Available online: https://www.r-project.org (accessed on 10 November 2020).
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. arXiv. 2014. arXiv preprint. Available online: https://arxiv.org/abs/1406.5823 (accessed on 10 November 2020).
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Flynn, M.F.; Hardie, E.M.; Armstrong, P.J. Effect of ovariohysterectomy on maintenance energy requirement in cats. J. Am. Vet. Med. Assoc. 1996, 209, 1572–1581. [Google Scholar]
- Harper, E.J.; Stack, D.M.; Watson, T.D.G.; Moxham, G. Effects of feeding regimens on bodyweight, composition and condition score in cats following ovariohysterectomy. J. Small Anim. Pr. 2001, 42, 433–438. [Google Scholar] [CrossRef]
- Kanchuk, M.L.; Backus, R.C.; Calvert, C.C.; Morris, J.G.; Rogers, Q.R. Weight Gain in Gonadectomized Normal and Lipoprotein Lipase–Deficient Male Domestic Cats Results from Increased Food Intake and Not Decreased Energy Expenditure. J. Nutr. 2003, 133, 1866–1874. [Google Scholar] [CrossRef]
- Wei, A.; Fascetti, A.J.; Kim, K.; Ramsey, J.J. Post-castration variations in weight gain in a cohort of young adult male cats. J. Nutr. Sci. 2014, 3, 37. [Google Scholar] [CrossRef]
- Belsito, K.R.; Vester, B.M.; Keel, T.; Graves, T.K.; Swanson, K.S. Impact of ovariohysterectomy and food intake on body composition, physical activity, and adipose gene expression in cats1. J. Anim. Sci. 2009, 87, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Alexander, L.G.; Salt, C.; Thomas, G.; Butterwick, R. Effects of neutering on food intake, body weight and body composition in growing female kittens. Br. J. Nutr. 2011, 106, S19–S23. [Google Scholar] [CrossRef]
- Allaway, D.; Gilham, M.; Colyer, A.; Morris, P.J. The impact of time of neutering on weight gain and energy intake in female kittens. J. Nutr. Sci. 2017, 6, 2031. [Google Scholar] [CrossRef]
- A Houpt, K.; Coren, B.; Hintz, H.F.; E Hilderbrant, J. Effect of sex and reproductive status on sucrose preference, food intake, and body weight of dogs. J. Am. Vet. Med. Assoc. 1979, 174, 1083–1085. [Google Scholar] [PubMed]
- Simpson, M.; Albright, S.; Wolfe, B.; Searfoss, E.; Street, K.; Diehl, K.; Page, R. Age at gonadectomy and risk of overweight/obesity and orthopedic injury in a cohort of Golden Retrievers. PLoS ONE 2019, 14, e0209131. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, S.L.; Yang, M.; Wang, M.; Elliott, D.A.; Buff, P.R.; Lund, E.M. Effect of age at gonadectomy on the probability of dogs becoming overweight. J. Am. Vet. Med. Assoc. 2013, 243, 236–243. [Google Scholar] [CrossRef]
- Phungviwatnikul, T.; Valentine, H.; de Godoy, M.R.C.; Swanson, K.S. Effects of diet on body weight, body composition, metabolic status, and physical activity levels of adult female dogs after spay surgery. J. Anim. Sci. 2020, 98. [Google Scholar] [CrossRef] [PubMed]
- Salmeri, K.R.; Bloomberg, M.S.; Scruggs, S.L.; Shille, V. Gonadectomy in immature dogs: Effects on skeletal, physical, and behavioral development. J. Am. Vet. Med. Assoc. 1991, 198, 1193–1203. [Google Scholar]
- Spain, C.V.; Scarlett, J.M.; Houpt, K.A. Long-term risks and benefits of early-age gonadectomy in dogs. J. Am. Vet. Med. Assoc. 2004, 224, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Mystkowski, P.; Schwartz, M.W. Gonadal steroids and energy homeostasis in the leptin era. Nutrrition 2000, 16, 937–946. [Google Scholar] [CrossRef]
- Colliard, L.; Paragon, B.-M.; Lemuet, B.; Bénet, J.-J.; Blanchard, G. Prevalence and risk factors of obesity in an urban population of healthy cats. J. Feline Med. Surg. 2009, 11, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Prieto, A.; Nielsen, L.R.; Dąbrowski, R.; Bjørnvad, C.R.; Söder, J.; Lamy, E.; Monkeviciene, I.; Ljubić, B.B.; Vasiu, I.; Savic, S.; et al. European dog owner perceptions of obesity and factors associated with human and canine obesity. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Courcier, E.; Mellor, D.; Thomson, R.; Yam, P. A cross sectional study of the prevalence and risk factors for owner misperception of canine body shape in first opinion practice in Glasgow. Prev. Vet. Med. 2011, 102, 66–74. [Google Scholar] [CrossRef]
- Heuberger, R.; Wakshlag, J. The relationship of feeding patterns and obesity in dogs. J. Anim. Physiol. Anim. Nutr. 2011, 95, 98–105. [Google Scholar] [CrossRef]
- Bermingham, E.N.; Thomas, D.G.; Cave, N.J.; Morris, P.J.; Butterwick, R.F.; German, A.J. Energy Requirements of Adult Dogs: A Meta-Analysis. PLoS ONE 2014, 9, e109681. [Google Scholar] [CrossRef]
- Hawthorne, A.J.; Booles, D.; Nugent, P.A.; Gettinby, G.; Wilkinson, J. Body-Weight Changes during Growth in Puppies of Different Breeds. J. Nutr. 2004, 134, 2027S–2030S. [Google Scholar] [CrossRef] [PubMed]
- Kienzle, E.; Rainbird, A. Maintenance Energy Requirement of Dogs: What is the Correct Value for the Calculation of Metabolic Body Weight in Dogs? J. Nutr. 1991, 121, S39–S40. [Google Scholar] [CrossRef]
- World Health Organisation: Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 14 January 2021).
- Holmes, K.L.; Morris, P.J.; Abdulla, Z.; Hackett, R.; Rawlings, J.M. Risk factors associated with excess body weight in dogs in the UK. J. Anim. Physiol. Anim. Nutr. 2007, 91, 166–167. [Google Scholar] [CrossRef]
- Nijland, M.L.; Stam, F.; Seidell, J.C. Overweight in dogs, but not in cats, is related to overweight in their owners. Public Health Nutr. 2009, 13, 102–106. [Google Scholar] [CrossRef]
- Richardson, D.C. The Role of Nutrition in Canine Hip Dysplasia. Vet. Clin. North Am. Small Anim. Pr. 1992, 22, 529–540. [Google Scholar] [CrossRef]
- Nap, R.; Hazewinkel, H. Growth and skeletal development in the dog in relation to nutrition; a review. Vet. Q. 1994, 16, 50–59. [Google Scholar] [CrossRef]
- Fries, C.L.; Remedios, A.M. The pathogenesis and diagnosis of canine hip dysplasia: A review. Can. Vet. J. 1995, 36, 494–502. [Google Scholar] [PubMed]
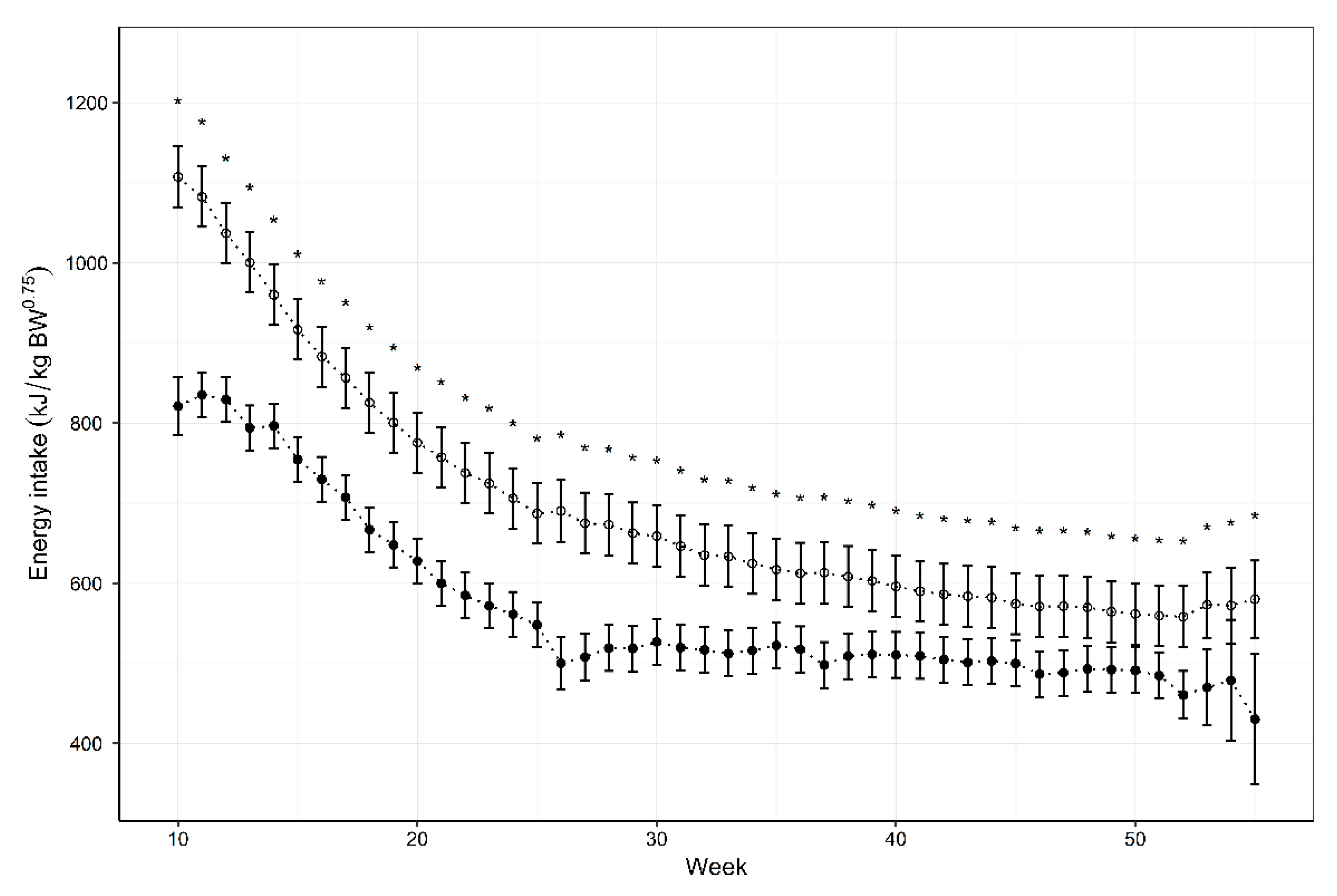
| Age (Weeks) | Mean Energy Intake (kJ/kg0.75) | NRC Estimate (kJ/kg0.75) | Energy Difference (kJ/kg0.75) | p-Value |
|---|---|---|---|---|
| 10 | 821.2 (784.7, 857.8) | 1107.4 (1068.6, 1146.2) | −286.0 (−334.4, −237.6) | <0.001 |
| 14 | 796.7 (768.7, 824.7) | 960.1 (922.5, 997.8) | −163.3 (−204.9, −121.6) | <0.001 |
| 18 | 667.0 (639.0, 695.1) | 825.7 (788.1, 863.4) | −158.6 (−200.2, −116.9) | <0.001 |
| 22 | 585.1 (557.1, 613.1) | 737.8 (700.1, 775.4) | −152.5 (−194.2, −110.9) | <0.001 |
| 26 | 500.3 (467.3, 533.4) | 690.4 (651.7, 729.0) | −190.0 (−235.9, −144.2) | <0.001 |
| 30 | 527.1 (498.4, 555.7) | 658.9 (620.8, 697.0) | −131.9 (−174.4, −89.5) | <0.001 |
| 34 | 516.2 (487.6, 544.8) | 624.8 (586.7, 662.9) | −108.7 (−151.1, −66.3) | <0.001 |
| 38 | 509.1 (480.5, 537.7) | 608.5 (570.3, 646.6) | −99.6 (−142.0, −57.2) | <0.001 |
| 42 | 504.8 (476.2, 533.4) | 586.2 (548.1, 624.3) | −81.7 (−124.1, −39.2) | <0.001 |
| 46 | 486.7 (458.1, 515.4) | 571.1 (533.0, 609.3) | −84.7 (−127.1, −42.2) | <0.001 |
| 50 | 491.4 (462.8, 520.1) | 561.8 (523.7, 599.9) | −70.5 (−113, −28.1) | <0.001 |
| 52 | 460.6 (430.8, 490.5) | 558.3 (520.0, 596.5) | −97.8 (−141.2, −54.5) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bradley, S.; Alexander, J.; Haydock, R.; Bakke, A.M.; Watson, P. Energy Requirements for Growth in the Norfolk Terrier. Animals 2021, 11, 1380. https://doi.org/10.3390/ani11051380
Bradley S, Alexander J, Haydock R, Bakke AM, Watson P. Energy Requirements for Growth in the Norfolk Terrier. Animals. 2021; 11(5):1380. https://doi.org/10.3390/ani11051380
Chicago/Turabian StyleBradley, Sophie, Janet Alexander, Richard Haydock, Anne Marie Bakke, and Phillip Watson. 2021. "Energy Requirements for Growth in the Norfolk Terrier" Animals 11, no. 5: 1380. https://doi.org/10.3390/ani11051380
APA StyleBradley, S., Alexander, J., Haydock, R., Bakke, A. M., & Watson, P. (2021). Energy Requirements for Growth in the Norfolk Terrier. Animals, 11(5), 1380. https://doi.org/10.3390/ani11051380





