Administration of Dietary Microalgae Ameliorates Intestinal Parameters, Improves Body Weight, and Reduces Thawing Loss of Fillets in Broiler Chickens: A Pilot Study
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Microalgae Biomass
2.2. Experimental Setup
2.3. Body Weight
2.4. Morphometrical Analyses
2.5. Meat Quality Parameters
2.6. Statistical Analyses
3. Results
3.1. Effects of Diet on Body Weight
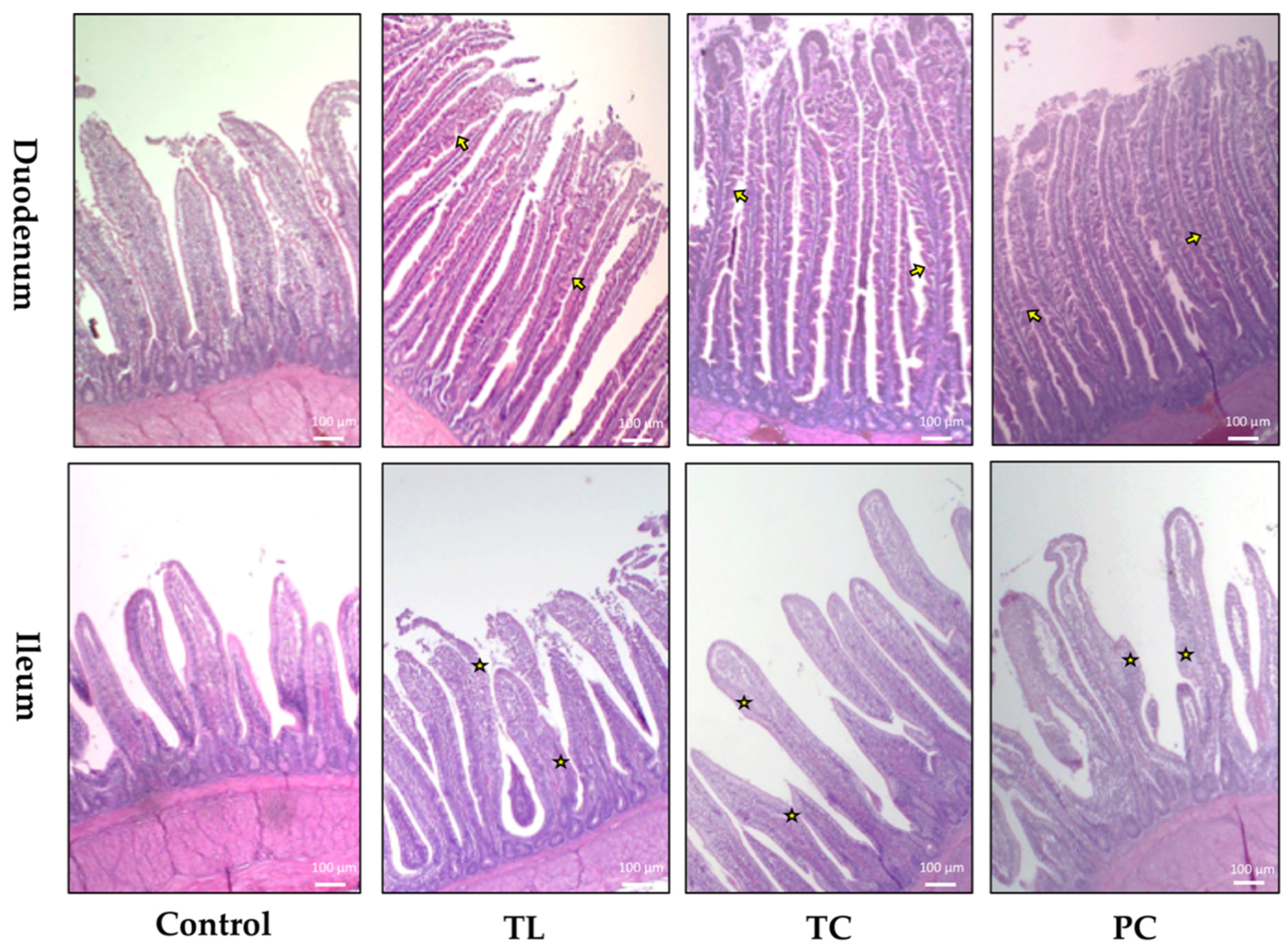
3.2. Histological Findings and Small Intestine Morphometric Measures
3.3. Meat Quality Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marshall, B.M.; Levy, S.B. Food animals and antimicrobials: Impacts on human health. Clin. Microbiol. Rev. 2011, 24, 718–733. [Google Scholar] [CrossRef] [Green Version]
- Lin, J. Antibiotic growth promoters enhance animal production by targeting intestinal bile salt hydrolase and its producers. Front. Microbiol. 2014, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Millet, S.; Maertens, L. The European ban on antibiotic growth promoters in animal feed: From challenges to opportunities. Vet. J. 2011, 187, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Hicks, M.H. Results of new FDA rules on antibiotic use in US food-producing animals. J. Public Health 2020, 42, E573–E574. [Google Scholar] [CrossRef]
- Humphreys, G.; Fleck, F. United Nations meeting on antimicrobial resistance. Bull. World Health 2016, 94, 638. [Google Scholar]
- Cox, J.A.; Vlieghe, E.; Mendelson, M.; Wertheim, H.; Ndegwa, L.; Villegas, M.V.; Gould, I.; Levy Hara, G. Antibiotic stewardship in low- and middle-income countries: The same but different? Clin. Microbiol. Infect. 2017, 23, 812–818. [Google Scholar] [CrossRef] [Green Version]
- Šefcová, M.; Larrea-Álvarez, M.; Larrea-Álvarez, C.; Revajová, V.; Karaffová, V.; Koščová, J.; Nemcová, R.; Ortega-Paredes, D.; Vinueza-Burgos, C.; Levkut, M.; et al. Effects of Lactobacillus fermentum supplementation on body weight and pro-inflammatory cytokine expression in Campylobacter jejuni-challenged chickens. Vet. Sci. 2020, 7, 121. [Google Scholar] [CrossRef]
- Šefcová, M.A.; Larrea-Álvarez, M.; Larrea-Álvarez, C.M.; Karaffová, V.; Ortega-Paredes, D.; Vinueza-Burgos, C.; Ševčíková, Z.; Levkut, M.; Herich, R.; Revajová, V. The probiotic Lactobacillus fermentum Biocenol CCM 7514 moderates Campylobacter jejuni-induced body weight impairment by improving gut morphometry and regulating cecal cytokine abundance in broiler chickens. Animals 2021, 11, 235. [Google Scholar] [CrossRef] [PubMed]
- Šefcová, M.; Larrea-Álvarez, M.; Larrea-Álvarez, C.; Karaffová, V.; Revajová, V.; Gancarčíková, S.; Ševčíková, Z.; Herich, R. Lactobacillus fermentum Administration Modulates Cytokine Expression and Lymphocyte Subpopulation Levels in Broiler Chickens Challenged with Campylobacter coli. Foodborne Pathog. Dis. 2020, 17, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Tian, Y.; Cao, Y.; Li, J.; Guo, H.; Su, Y.; Tian, Y.; Wang, C.; Wang, T.; Zhang, L. Probiotic properties of Lactobacillus paracasei subsp. Paracasei L1 and its growth performance-promotion in chicken by improving the intestinal microflora. Front. Physiol. 2019, 10, 937. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zeng, D.; Wang, H.; Qing, X.; Sun, N.; Xin, J.; Luo, M.; Khalique, A.; Pan, K.; Shu, G.; et al. Dietary Probiotic Bacillus licheniformis H2 Enhanced Growth Performance, Morphology of Small Intestine and Liver, and Antioxidant Capacity of Broiler Chickens Against Clostridium perfringens–Induced Subclinical Necrotic Enteritis. Probiotics Antimicrob. Proteins 2020, 12, 883–895. [Google Scholar] [CrossRef]
- Omar, A.E.; Al-Khalaifah, H.S.; Mohamed, W.A.M.; Gharib, H.S.A.; Osman, A.; Al-Gabri, N.A.; Amer, S.A. Effects of Phenolic-Rich Onion (Allium cepa L.) Extract on the Growth Performance, Behavior, Intestinal Histology, Amino Acid Digestibility, Antioxidant Activity, and the Immune Status of Broiler Chickens. Front. Vet. Sci. 2020, 7, 582612. [Google Scholar] [CrossRef]
- Amer, S.A.; Mohamed, W.A.M.; Gharib, H.S.A.; Al-Gabri, N.A.; Gouda, A.; Elabbasy, M.T.; Abd El-Rahman, G.I.; Omar, A.E. Changes in the growth, ileal digestibility, intestinal histology, behavior, fatty acid composition of the breast muscles, and blood biochemical parameters of broiler chickens by dietary inclusion of safflower oil and vitamin C. BMC Vet. Res. 2021, 17, 68. [Google Scholar] [CrossRef] [PubMed]
- Amad, A.A.; Männer, K.; Wendler, K.R.; Neumann, K.; Zentek, J. Effects of a phytogenic feed additive on growth performance and ileal nutrient digestibility in broiler chickens. Poult. Sci. 2011, 90, 2811–2816. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Zhu, Y.R.; Zhao, Z.H.; Jiang, P.; Yin, F.Q. Effects of dietary supplementation of algae-derived polysaccharides on morphology, tight junctions, antioxidant capacity and immune response of duodenum in broilers under heat stress. Animals 2021, 11, 2279. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.C.; Guo, Y.; Zhao, Z.H.; Jha, R.; Balasubramanian, B. Algae-Derived Polysaccharides Promote Growth Performance by Improving Antioxidant Capacity and Intestinal Barrier Function in Broiler Chickens. Front. Vet. Sci. 2020, 7, 601336. [Google Scholar] [CrossRef]
- Guiry, M.D. How many species of algae are there? J. Phycol. 2012, 48, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- Coudert, E.; Baéza, E.; Berri, C. Use of algae in poultry production: A review. Worlds Poult. Sci. J. 2020, 76, 767–786. [Google Scholar] [CrossRef]
- Abdelnour, S.A.; Abd El-Hack, M.E.; Arif, M.; Khafaga, A.F.; Taha, A.E. The application of the microalgae Chlorella spp. As a supplement in broiler feed. Worlds Poult. Sci. J. 2019, 75, 305–318. [Google Scholar] [CrossRef]
- Khan, I.A.; Parker, N.B.; Löhr, C.V.; Cherian, G. Docosahexaenoic acid (22:6 n-3)-rich microalgae along with methionine supplementation in broiler chickens: Effects on production performance, breast muscle quality attributes, lipid profile, and incidence of white striping and myopathy. Poult. Sci. 2021, 100, 865–874. [Google Scholar] [CrossRef]
- Long, S.F.; Kang, S.; Wang, Q.Q.; Xu, Y.T.; Pan, L.; Hu, J.X.; Li, M.; Piao, X.S. Dietary supplementation with DHA-rich microalgae improves performance, serum composition, carcass trait, antioxidant status, and fatty acid profile of broilers. Poult. Sci. 2018, 97, 1881–1890. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.K.; Salim, H.M.; Akter, N.; Kim, D.W.; Kim, J.H.; Bang, H.T.; Kim, M.J.; Na, J.C.; Hwangbo, J.; Choi, H.C.; et al. Effect of various forms of dietary Chlorella supplementation on growth performance, immune characteristics, and intestinal microflora population of broiler chickens. J. Appl. Poult. Res. 2013, 22, 100–108. [Google Scholar] [CrossRef]
- Madeira, M.S.; Cardoso, C.; Lopes, P.A.; Coelho, D.; Afonso, C.; Bandarra, N.M.; Prates, J.A.M. Microalgae as feed ingredients for livestock production and meat quality: A review. Livest. Sci. 2017, 205, 111–121. [Google Scholar] [CrossRef]
- El-Bahr, S.; Shousha, S.; Shehab, A.; Khattab, W.; Ahmed-Farid, O.; Sabike, I.; El-Garhy, O.; Albokhadaim, I.; Albosadah, K. Effect of dietary microalgae on growth performance, profiles of amino and fatty acids, antioxidant status, and meat quality of broiler chickens. Animals 2020, 10, 761. [Google Scholar] [CrossRef]
- Yan, L.; Kim, I.H. Effects of dietary ω -3 fatty acid-enriched microalgae supplementation on growth performance, blood profiles, meat quality, and fatty acid composition of meat in broilers. J. Appl. Anim. Res. 2013, 41, 392–397. [Google Scholar] [CrossRef] [Green Version]
- Alkhamis, Y.; Qin, J.G. Comparison of pigment and proximate compositions of Tisochrysis lutea in phototrophic and mixotrophic cultures. J. Appl. Phycol. 2016, 28, 35–42. [Google Scholar] [CrossRef]
- Lu, L.; Wang, J.; Yang, G.; Zhu, B.; Pan, K. Heterotrophic growth and nutrient productivities of Tetraselmis chuii using glucose as a carbon source under different C/N ratios. J. Appl. Phycol. 2017, 29, 15–21. [Google Scholar] [CrossRef]
- Erol, H.B.U.; Menegazzo, M.L.; Sandefur, H.; Gottberg, E.; Vaden, J.; Asgharpour, M.; Hestekin, C.N.; Hestekin, J.A. Porphyridium cruentum grown in ultra-filtered swine wastewater and its effects on microalgae growth productivity and fatty acid composition. Energies 2020, 13, 3194. [Google Scholar] [CrossRef]
- Casas-Arrojo, V.; Decara, J.; de los Ángeles Arrojo-Agudo, M.; Pérez-Manríquez, C.; Abdala-Díaz, R.T. Immunomodulatory, antioxidant activity and cytotoxic effect of sulfated polysaccharides from Porphyridium cruentum. (s.f. Gray) Nägeli. Biomolecules 2021, 11, 488. [Google Scholar] [CrossRef] [PubMed]
- Lazic, S.E.; Clarke-Williams, C.J.; Munafò, M.R. What exactly is ‘N’ in cell culture and animal experiments? PLoS Biol. 2018, 16, e2005282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aviagen Ross Broiler Guide. Available online: https://en.aviagen.com (accessed on 5 August 2021).
- Liu, T.; Wang, C.; Wu, X.; Ren, M.; Hu, Q.; Jin, E.; Gu, Y. Effect of Boron on Microstructure, Immune Function, Expression of Tight Junction Protein, Cell Proliferation and Apoptosis of Duodenum in Rats. Biol. Trace Elem. Res. 2021, 199, 205–215. [Google Scholar] [CrossRef]
- Ginzberg, A.; Cohen, M.; Sod-Moriah, U.A.; Shany, S.; Rosenshtrauch, A.; Arad, S. Chickens fed with biomass of the red microalga Porphyridium sp. have reduced blood cholesterol level and modified fatty acid composition in egg yolk. J. Appl. Phycol. 2000, 12, 325–330. [Google Scholar] [CrossRef]
- Khatoon, H.; Penz, K.P.; Banerjee, S.; Mahmud, A.I.; Rahman, M.R.; Mian, S.; Minhaz, T.M.; Hossain, S. Improvement of water quality, survivality, growth performance, and proximate composition of Penaeus monodon postlarvae through immobilizing Tetraselmis chuii. Bioresour. Technol. Rep. 2021, 15, 100755. [Google Scholar] [CrossRef]
- Zhao, W.; Fang, H.H.; Liu, Z.Z.; Chen, J.M.; Zhang, C.W.; Gao, B.Y.; Niu, J. Responses in growth performance, enzymatic activity, immune function and liver health after dietary supplementation of Porphyridium sp. in juvenile golden pompano (Trachinotus ovatus). Aquac. Nutr. 2021, 27, 679–690. [Google Scholar] [CrossRef]
- Ribeiro, T.; Lordelo, M.M.; Alves, S.P.; Bessa, R.J.B.; Costa, P.; Lemos, J.P.C.; Ferreira, L.M.A.; Fontes, C.M.G.A.; Prates, J.A.M. Direct supplementation of diet is the most efficient way of enriching broiler meat with n-3 long-chain polyunsaturated fatty acids. Br. Poult. Sci. 2013, 54, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Pulido-Rodriguez, L.F.; Cardinaletti, G.; Secci, G.; Randazzo, B.; Bruni, L.; Cerri, R.; Olivotto, I.; Tibaldi, E.; Parisi, G. Appetite regulation, growth performances and fish quality are modulated by alternative dietary protein ingredients in gilthead sea bream (Sparus aurata) culture. Animals 2021, 11, 1919. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.K.; Park, S.B.; Kim, C.H. Effects of dietary supplementation with a Chlorella by-product on the growth performance, immune response, intestinal microflora and intestinal mucosal morphology in broiler chickens. J. Anim. Physiol. Anim. Nutr. 2017, 101, 208–214. [Google Scholar] [CrossRef]
- Mirzaie, S.; Sharifi, S.D.; Zirak-Khattab, F. The effect of a Chlorella by-product dietary supplement on immune response, antioxidant status, and intestinal mucosal morphology of broiler chickens. J. Appl. Phycol. 2020, 32, 1771–1777. [Google Scholar] [CrossRef]
- Chang, C.W.T.; Takemoto, J.Y.; Chang, P.E.; AlFindee, M.N.; Lin, Y.Y. Effects of Mesobiliverdin IXα-Enriched Microalgae Feed on Gut Health and Microbiota of Broilers. Front. Vet. Sci. 2021, 7, 586813. [Google Scholar] [CrossRef]
- Messina, M.; Bulfon, C.; Beraldo, P.; Tibaldi, E.; Cardinaletti, G. Intestinal morpho-physiology and innate immune status of European sea bass (Dicentrarchus labrax) in response to diets including a blend of two marine microalgae, Tisochrysis lutea and Tetraselmis suecica. Aquaculture 2019, 500, 660–669. [Google Scholar] [CrossRef]
- Bogucka, J.; Ribeiro, D.M.; Bogusławska-Tryk, M.; Dankowiakowska, A.; da Costa, R.P.R.; Bednarczyk, M. Microstructure of the small intestine in broiler chickens fed a diet with probiotic or synbiotic supplementation. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1785–1791. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Jia, M.; Wong, E.A. Delayed access to feed affects broiler small intestinal morphology and goblet cell ontogeny. Poult. Sci. 2020, 99, 5275–5285. [Google Scholar] [CrossRef] [PubMed]
- Poloni, V.; Magnoli, A.; Fochesato, A.; Cristofolini, A.; Caverzan, M.; Merkis, C.; Montenegro, M.; Cavaglieri, L. A Saccharomyces cerevisiae RC016-based feed additive reduces liver toxicity, residual aflatoxin B1 levels and positively influences intestinal morphology in broiler chickens fed chronic aflatoxin B1-contaminated diets. Anim. Nutr. 2020, 6, 31–38. [Google Scholar] [CrossRef]
- Park, I.; Lee, Y.; Goo, D.; Zimmerman, N.P.; Smith, A.H.; Rehberger, T.; Lillehoj, H.S. The effects of dietary Bacillus subtilis supplementation, as an alternative to antibiotics, on growth performance, intestinal immunity, and epithelial barrier integrity in broiler chickens infected with Eimeria maxima. Poult. Sci. 2020, 99, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Clark, D.L.; Jacobi, S.K.; Velleman, S.G. Supplementation of vitamin E and omega-3 fatty acids during the early posthatch period on intestinal morphology and gene expression differentiation in broilers. Poult. Sci. 2021, 100, 100954. [Google Scholar] [CrossRef]
- Alagawany, M.; Elnesr, S.S.; Farag, M.R.; Abd El-Hack, M.E.; Khafaga, A.F.; Taha, A.E.; Tiwari, R.; Iqbal Yatoo, M.; Bhatt, P.; Khurana, S.K.; et al. Omega-3 and omega-6 fatty acids in poultry nutrition: Effect on production performance and health. Animals 2019, 9, 573. [Google Scholar] [CrossRef] [Green Version]
- Asgharpour, M.; Rodgers, B.; Hestekin, J.A. Eicosapentaenoic acid from Porphyridium cruentum: Increasing growth and productivity of microalgae for pharmaceutical products. Energies 2015, 8, 10487–10503. [Google Scholar] [CrossRef] [Green Version]
- Csernus, B.; Biro, S.; Babinszky, L. Effect of Carotenoids, Oligosaccharides and Anthocyanins on Growth Performance, Immunological Parameters and Intestinal Morphology in Broiler Chickens Challenged with Escherichia coli Lipopolysaccharide. Animals 2020, 10, 347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lokaewmanee, K.; Yamauchi, K.; Okuda, N. Effects of dietary red pepper on egg yolk colour and histological intestinal morphology in laying hens. J. Anim. Physiol. Anim. Nutr. 2013, 97, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.S.; Vantharam Venkata, H.G.R.; Raju, M.V.L.N.; Rama Rao, S.V.; Nori, S.S.; Suryanarayan, S.; Kumar, V.; Perveen, Z.; Prasad, C.S. Dietary supplementation of extracts of red sea weed (Kappaphycus alvarezii) improves growth, intestinal morphology, expression of intestinal genes and immune responses in broiler chickens. J. Sci. Food Agric. 2021, 101, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.F.; Chen, Y.P.; Chen, R.; Su, Y.; Zhang, R.Q.; He, Q.F.; Wang, K.; Wen, C.; Zhou, Y.M. Dietary mannan oligosaccharide ameliorates cyclic heat stress-induced damages on intestinal oxidative status and barrier integrity of broilers. Poult. Sci. 2019, 98, 4767–4776. [Google Scholar] [CrossRef]
- Dvir, I.; Chayoth, R.; Sod-Moriah, U.; Shany, S.; Nyska, A.; Stark, A.H.; Madar, Z.; Arad, S.M. Soluble polysaccharide and biomass of red microalga Porphyridium sp. alter intestinal morphology and reduce serum cholesterol in rats. Br. J. Nutr. 2000, 84, 469–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomes, A.M.P.; Malcata, F.X. Bifidobacterium spp. and Lactobacillus acidophilus: Biological, biochemical, technological and therapeutical properties relevant for use as probiotics. Trends Food Sci. Technol. 1999, 10, 139–157. [Google Scholar] [CrossRef]
- Koh, A.; De Vadder, F.; Kovatcheva-Datchary, P.; Bäckhed, F. From dietary fiber to host physiology: Short-chain fatty acids as key bacterial metabolites. Cell 2016, 165, 1332–1345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cornick, S.; Tawiah, A.; Chadee, K. Roles and regulation of the mucus barrier in the gut. Tissue Barriers 2015, 3, e982426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansson, M.E.V.; Hansson, G.C. Immunological aspects of intestinal mucus and mucins. Nat. Rev. Immunol. 2016, 16, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Baurhoo, B.; Phillip, L.; Ruiz-Feria, C.A. Effects of purified lignin and mannan oligosaccharides on intestinal integrity and microbial populations in the ceca and litter of broiler chickens. Poult. Sci. 2007, 86, 1070–1078. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Zhao, S.; Jiang, L.; Lu, L.; Yang, Q.; Yu, Q. Lactobacillus reuteri Stimulates Intestinal Epithelial Proliferation and Induces Differentiation into Goblet Cells in Young Chickens. J. Agric. Food Chem. 2019, 67, 13758–13766. [Google Scholar] [CrossRef] [PubMed]
- Mullenix, G.J.; Maynard, C.J.; Owens, C.M.; Rochell, S.J.; Bottje, W.G.; Brister, R.D.; Kidd, M.T. Spirulina platensis meal inclusion effects on broilers fed a reduced protein diet. J. Appl. Poult. Res. 2021, 8, 100199. [Google Scholar] [CrossRef]
- Yan, G.L.; Guo, Y.M.; Yuan, J.M.; Liu, D.; Zhang, B.K. Sodium alginate oligosaccharides from brown algae inhibit Salmonella Enteritidis colonization in broiler chickens. Poult. Sci. 2011, 90, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Altmann, B.A.; Wigger, R.; Ciulu, M.; Mörlein, D. The effect of insect or microalga alternative protein feeds on broiler meat quality. J. Sci. Food Agric. 2020, 100, 4292–4302. [Google Scholar] [CrossRef] [PubMed]
- Larrea-Alvarez, M.; Purton, S. Multigenic engineering of the chloroplast genome in the green alga Chlamydomonas reinhardtii. Microbiology 2020, 166, 510–515. [Google Scholar] [CrossRef]
- Castellanos-Huerta, I.; Bañuelos-Hernández, B.; Téllez, G.; Rosales-Mendoza, S.; Brieba, L.G.; Esquivel-Ramos, E.; Beltrán-López, J.I.; Velazquez, G.; Fernandez-Siurob, I. Recombinant Hemagglutinin of Avian Influenza Virus H5 Expressed in the Chloroplast of Chlamydomonas reinhardtii and Evaluation of Its Immunogenicity in Chickens. Avian Dis. 2016, 60, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Dyo, Y.M.; Purton, S. The algal chloroplast as a synthetic biology platform for production of therapeutic proteins. Microbiology 2018, 164, 113–121. [Google Scholar] [CrossRef]
| Ingredients (%) | Control | Experimental Groups | ||
|---|---|---|---|---|
| Starter | Finisher | Starter | Finisher | |
| Ground corn | 52.99 | 57.23 | 52.88 | 57.12 |
| Soybean meal (CP, 48.0%) | 36.3 | 32.07 | 36.23 | 32.01 |
| Calcium carbonate | 1.52 | 1.49 | 1.52 | 1.49 |
| Monocalcium phosphate | 1.07 | 0.82 | 1.07 | 0.82 |
| Sodium chloride | 0.31 | 0.26 | 0.31 | 0.26 |
| Crude (vegetal) fat | 7.4 | 7.71 | 7.39 | 7.69 |
| Antimycotic | 0.10 | 0.08 | 0.10 | 0.08 |
| Mycotoxin sequestrant | 0.05 | 0.05 | 0.05 | 0.05 |
| Antioxidant | 0.02 | 0.02 | 0.02 | 0.02 |
| Phytase | 0.01 | 0.01 | 0.01 | 0.01 |
| 1 Vitamin and mineral premix | 0.23 | 0.26 | 0.23 | 0.26 |
| Microalgae-derived protein | - | - | 0.08 | 0.08 |
| Microalgae-derived fat | - | - | 0.01 | 0.01 |
| Microalgae-derived crude ash | - | - | 0.05 | 0.05 |
| Microalgae-derived carbohydrate, fiber, rest of biomass | - | - | 0.06 | 0.06 |
| Nutrient specifications | ||||
| 2 ME Kcal/kg diet | 3000 | 3190 | 3000 | 3190 |
| Lysine (%) | 1.48 | 1.25 | 1.48 | 1.25 |
| Methionine + cysteine (%) | 1.07 | 0.99 | 1.07 | 0.99 |
| Methionine (%) | 0.59 | 0.50 | 0.59 | 0.50 |
| Threonine (%) | 0.97 | 0.85 | 0.97 | 0.85 |
| Valine (%) | 1.06 | 0.97 | 1.06 | 0.97 |
| Isoleucine (%) | 0.95 | 0.86 | 0.95 | 0.86 |
| Arginine (%) | 1.51 | 1.38 | 1.51 | 1.38 |
| Tryptophan (%) | 0.24 | 0.22 | 0.24 | 0.22 |
| Leucine (%) | 1.58 | 1.45 | 1.58 | 1.45 |
| Crude protein (%) | 22.50 | 21.00 | 22.50 | 21.00 |
| Ca (%) | 1.00 | 0.85 | 1.00 | 0.85 |
| Available P (%) | 0.50 | 0.40 | 0.50 | 0.40 |
| Mg (%) | 0.40 | 0.40 | 0.40 | 0.40 |
| Na (%) | 0.23 | 0.16 | 0.23 | 0.16 |
| Cl (%) | 0.22 | 0.19 | 0.22 | 0.19 |
| K (%) | 0.96 | 0.72 | 0.96 | 0.72 |
| Choline (mg/kg) | 1700 | 1600 | 1700 | 1600 |
| Linoleic acid (%) | 1.25 | 1.15 | 1.25 | 1.15 |
| Experimental Groups | ||||
|---|---|---|---|---|
| Control | TL | TC | PC | |
| Body weight (g) | ||||
| Day 5 | 108.67 ± 10.5 | 111.44 ± 13.3 | 116.78 ± 16.9 | 110.44 ± 15.7 |
| Day 7 | 154.78 ± 16.4 | 155 ± 18.4 | 172.11 ± 21 | 157.67 ± 18.7 |
| Day 14 | 391 ± 34.2 | 373.78 ± 38.7 | 416.67 ± 39.3 | 395.67 ± 42.3 |
| Day 21 | 800.67 ± 51.5 | 760 ± 61.3 | 891.11 ± 72.9 ■ | 867.33 ± 94.9 ■ |
| Day 28 | 1336 ± 56.7 | 1316.8 ± 95.8 | 1530.2 ± 85.3 *■ | 1433.9 ± 111.1 ■ |
| Day 29 | 1398.8 ± 64.8 | 1399 ± 105.8 | 1611.4 ±100.3 *■ | 1540.2 ± 125.3 *■ |
| Day 30 | 1425 ± 72.7 | 1494.6 ± 117 | 1658.6 ± 103.7 *■ | 1613 ± 146.1 * |
| Small Intestine Sections | Experimental Groups | |||
|---|---|---|---|---|
| Control | TL | TC | PC | |
| Duodenum | ||||
| Villus height (μm) | 2659.06 ± 324.9 | 3233.92 ± 210.1 * | 3454.45 ± 353.9 * | 3842.46 ± 243.1 *■ |
| Crypt depth (μm) 1 | 357.27(69.5) | 404.79(38.6) * | 395.32(52.8) | 484.90(48.7) *■▲ |
| Villus-height-to-crypt-depth ratio | 7.48 ± 1.0 | 8.15 ± 0.9 | 8.89 ± 1.0 * | 8.19 ± 0.9 |
| Goblet cell count | 20.82 ± 5.2 | 31.94 ± 4.9 * | 35.65 ± 7.2 * | 36.35 ± 4.0 * |
| Ileum | ||||
| Villus height (μm) | 1270.70 ± 143.7 | 1659.30 ± 228.6 * | 1981.50 ± 117.4 *■ | 2303.90 ± 180.7 *■▲ |
| Crypt depth (μm) | 323.92 ± 29.4 | 362.40 ± 45.4 | 374.63 ± 54.06 | 406.94 ± 57.9 * |
| Villus-height-to-crypt-depth ratio | 3.81 ± 0.6 | 4.17 ± 0.4 * | 5.51 ± 0.8 * | 5.8 ± 0.8 *■ |
| Goblet cell count | 41.37 ± 4.5 | 46.62 ± 9.6 | 45.55 ± 3.5 | 52.96 ± 5.6 * |
| Parameters | Experimental Groups | |||
|---|---|---|---|---|
| Control | TL | TC | PC | |
| Cooking loss (g) | 1.28 ± 0.1 | 1.30 ± 0.2 | 1.41 ± 0.2 | 1.42 ± 0.3 |
| Thawing loss (g) | 0.83 ± 0.1 | 0.71 ± 0.2 | 0.49 ± 0.1 *■ | 0.89 ± 0.3 ▲ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šefcová, M.A.; Santacruz, F.; Larrea-Álvarez, C.M.; Vinueza-Burgos, C.; Ortega-Paredes, D.; Molina-Cuasapaz, G.; Rodríguez, J.; Calero-Cáceres, W.; Revajová, V.; Fernández-Moreira, E.; et al. Administration of Dietary Microalgae Ameliorates Intestinal Parameters, Improves Body Weight, and Reduces Thawing Loss of Fillets in Broiler Chickens: A Pilot Study. Animals 2021, 11, 3601. https://doi.org/10.3390/ani11123601
Šefcová MA, Santacruz F, Larrea-Álvarez CM, Vinueza-Burgos C, Ortega-Paredes D, Molina-Cuasapaz G, Rodríguez J, Calero-Cáceres W, Revajová V, Fernández-Moreira E, et al. Administration of Dietary Microalgae Ameliorates Intestinal Parameters, Improves Body Weight, and Reduces Thawing Loss of Fillets in Broiler Chickens: A Pilot Study. Animals. 2021; 11(12):3601. https://doi.org/10.3390/ani11123601
Chicago/Turabian StyleŠefcová, Miroslava Anna, Francisco Santacruz, César Marcelo Larrea-Álvarez, Christian Vinueza-Burgos, David Ortega-Paredes, Gabriel Molina-Cuasapaz, Jessica Rodríguez, William Calero-Cáceres, Viera Revajová, Esteban Fernández-Moreira, and et al. 2021. "Administration of Dietary Microalgae Ameliorates Intestinal Parameters, Improves Body Weight, and Reduces Thawing Loss of Fillets in Broiler Chickens: A Pilot Study" Animals 11, no. 12: 3601. https://doi.org/10.3390/ani11123601
APA StyleŠefcová, M. A., Santacruz, F., Larrea-Álvarez, C. M., Vinueza-Burgos, C., Ortega-Paredes, D., Molina-Cuasapaz, G., Rodríguez, J., Calero-Cáceres, W., Revajová, V., Fernández-Moreira, E., & Larrea-Álvarez, M. (2021). Administration of Dietary Microalgae Ameliorates Intestinal Parameters, Improves Body Weight, and Reduces Thawing Loss of Fillets in Broiler Chickens: A Pilot Study. Animals, 11(12), 3601. https://doi.org/10.3390/ani11123601










