Evaluation of Fermented Oat and Black Soldier Fly Larva as Food Ingredients in Senior Dog Diets
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. BSFL and FO Preparation
2.2. Animals, Designs, Diets, and Housing
2.3. Sampling and Analysis
2.4. Statistical Analysis
3. Results
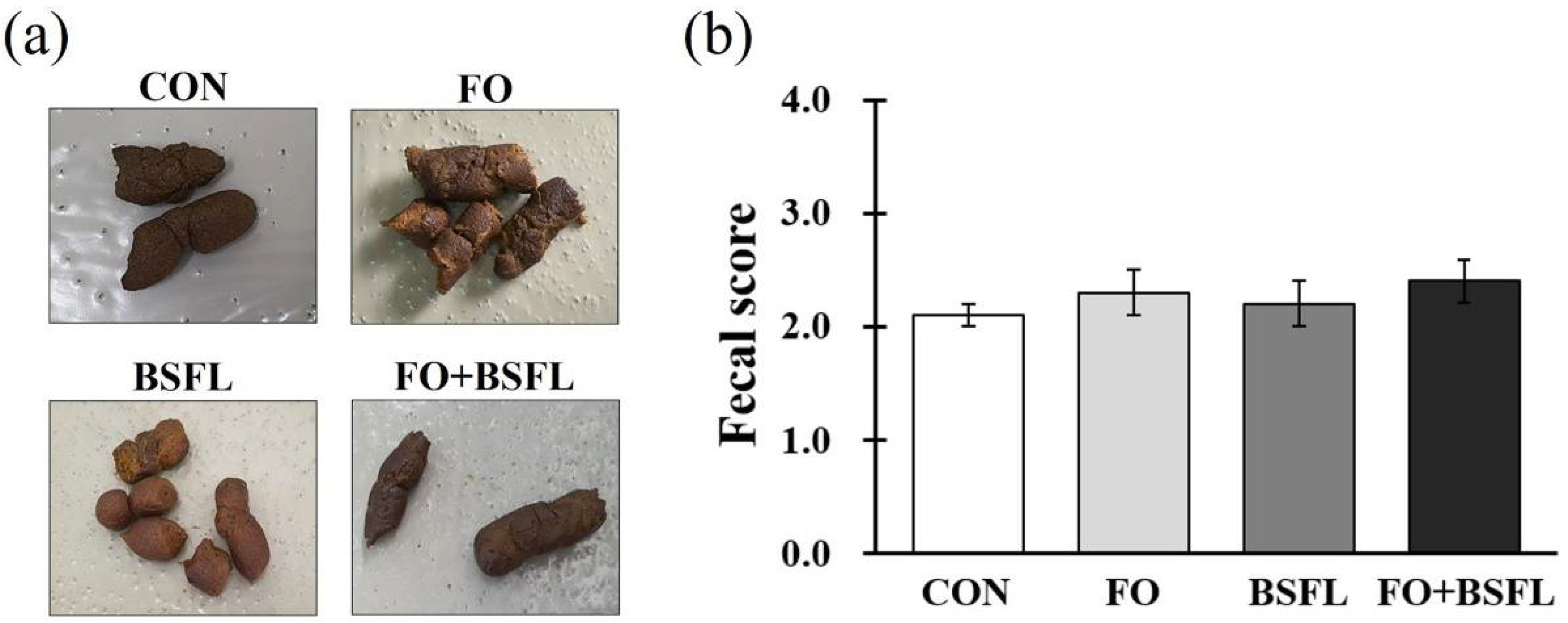
3.1. Food Intake, Body Parameters, and Fecal Score
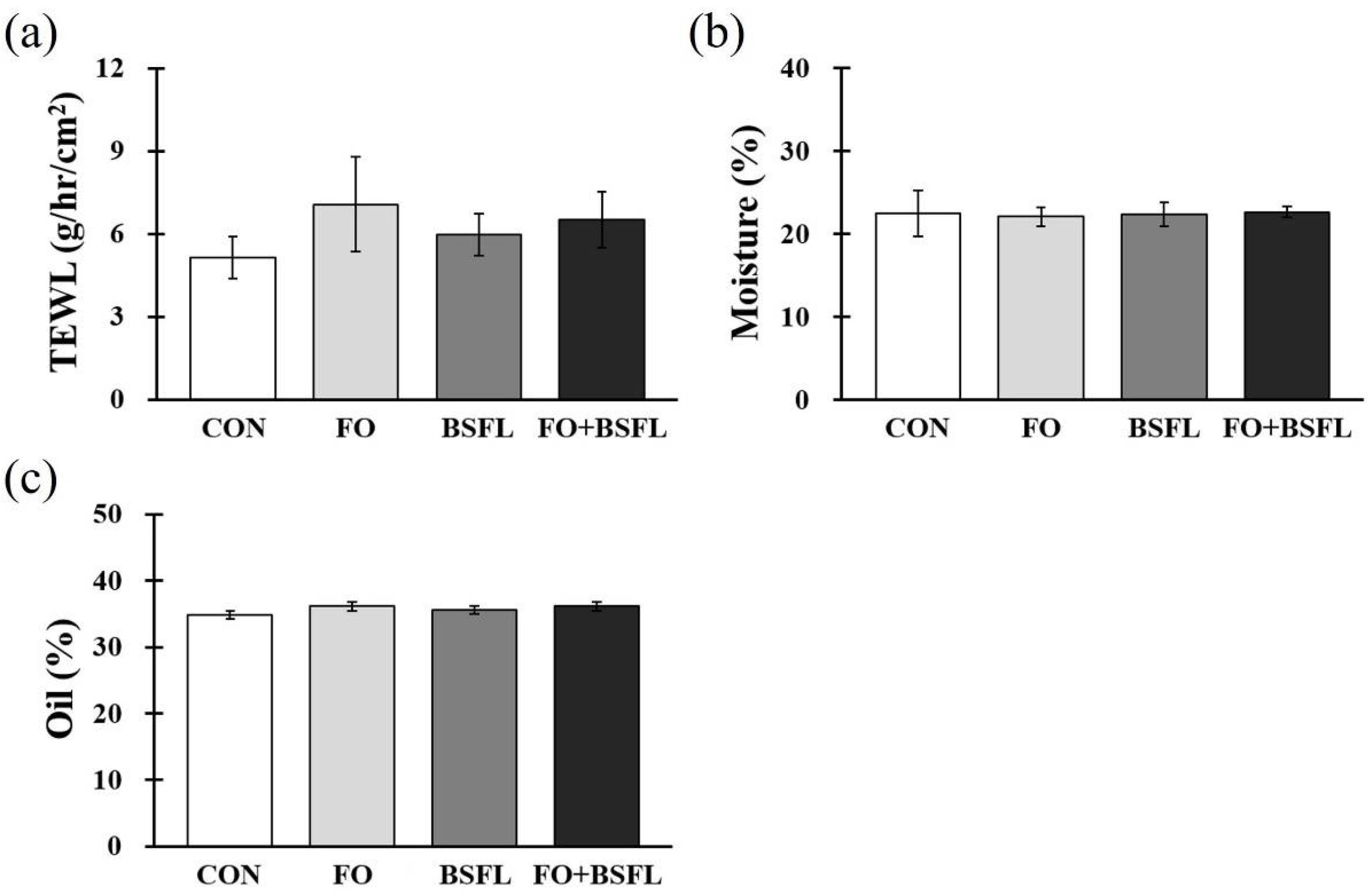
3.2. Skin Status
3.3. Hematological and Biochemical Parameters
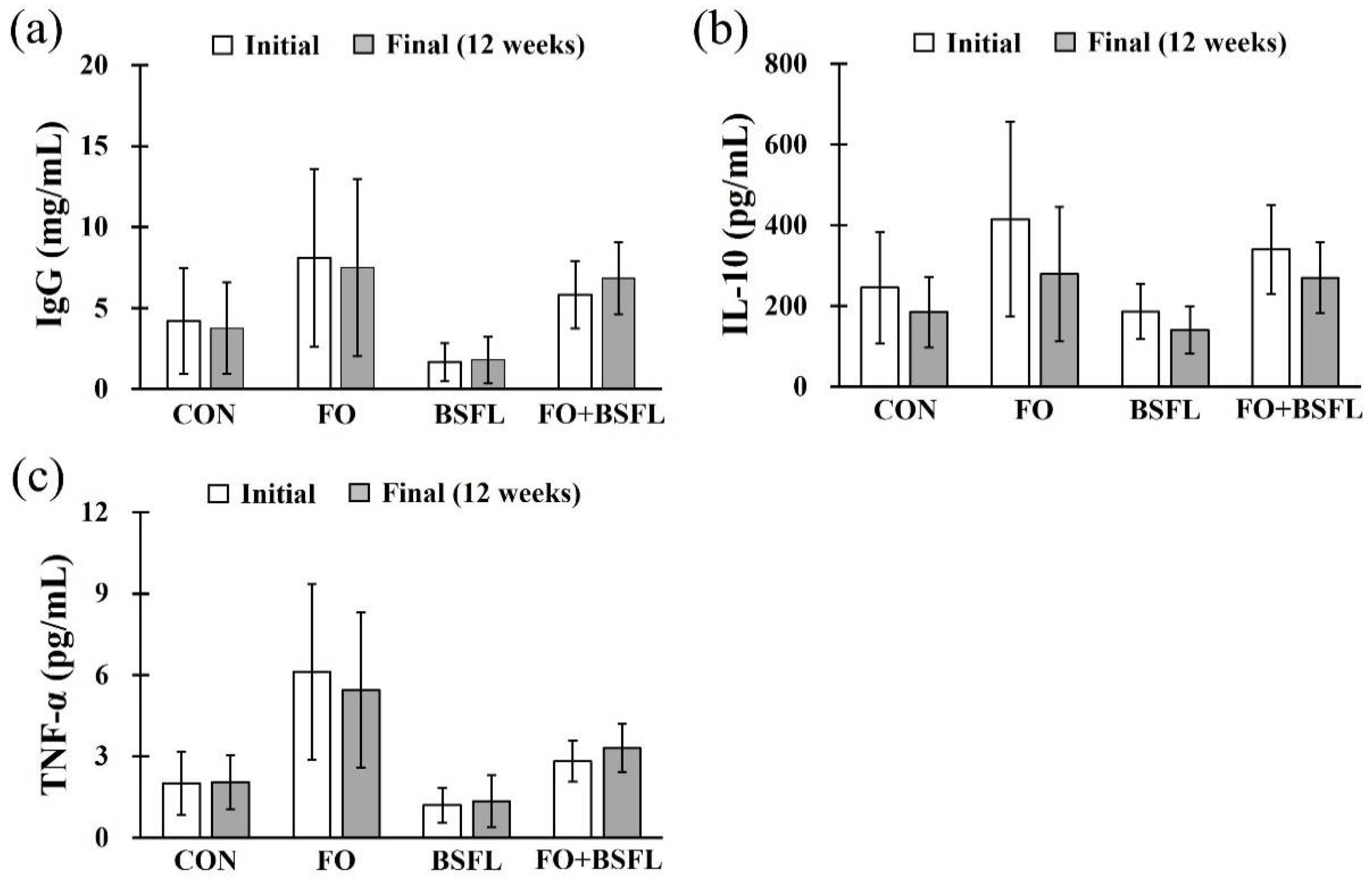
3.4. IgG and Cytokines
4. Discussion
4.1. Feeding and Body Parameters
4.2. Safety and Health Parameters
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations (FAO). High-Level Expert Forum—How to Feed the World in 2050; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2009. [Google Scholar]
- Boland, M.J.; Rae, A.N.; Vereijken, J.M.; Meuwissen, M.P.; Fischer, A.R.; van Boekel, M.A.; Rutherfurd, S.M.; Gruppen, H.; Moughan, P.J.; Hendriks, W.H. The future supply of animal-derived protein for human consumption. Trends Food Sci. Technol. 2013, 29, 62–73. [Google Scholar] [CrossRef]
- Alvarenga, C.I.; Aldrich, C.G. The effect of increasing levels of dehulled faba beans (Vicia faba L.) on extrusion and product parameters for dry expanded dog food. Foods 2019, 8, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCusker, S.; Buff, P.R.; Yu, Z.; Fascetti, A.J. Amino acid content of selected plant, algae and insect species: A search for alternative protein sources for use in pet foods. J. Nutr. Sci. 2014, 3, e39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verlinden, A.; Hesta, M.; Millet, S.; Janssens, G.P.J. Food allergy in dogs and cats: A review. Crit. Rev. Food Sci. Nutr. 2006, 46, 259–273. [Google Scholar] [CrossRef]
- Nadeau, L.; Nadeau, I.; Franklin, F.; Dunkel, F. The potential for entomophagy to address undernutrition. Ecol. Food Nutr. 2014, 54, 200–208. [Google Scholar] [CrossRef]
- Verneau, F.; Amato, M.; La Barbera, F. Edible insects and global food security. Insects 2021, 12, 472. [Google Scholar] [CrossRef]
- Areerat, S.; Chundang, P.; Lekcharoensuk, C.; Kovitvadhi, A. Possibility of using house cricket (Acheta domesticus) or mulberry silkworm (Bombyx mori) pupae meal to replace poultry meal in canine diets based on health and nutrient digestibility. Animals 2021, 11, 2680. [Google Scholar] [CrossRef]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2017, 97, 2594–2600. [Google Scholar] [CrossRef]
- Wang, Y.-S.; Shelomi, M. Review of black soldier fly (Hermetia illucens) as animal feed and human food. Foods 2017, 6, 91. [Google Scholar] [CrossRef] [Green Version]
- Onsongo, V.O.; Osuga, I.M.; Gachuiri, C.K.; Wachira, A.M.; Miano, D.M.; Tanga, C.M.; Ekesi, S.; Nakimbugwe, D.; Fiaboe, K.K.M. Insects for income generation through animal feed: Effect of dietary replacement of soybean and fish meal with black soldier fly meal on broiler growth and economic performance. J. Econ. Èntomol. 2018, 111, 1966–1973. [Google Scholar] [CrossRef] [Green Version]
- Marono, S.; Loponte, R.; Lombardi, P.; Vassalotti, G.; Pero, M.E.; Russo, F.; Gasco, L.; Parisi, G.; Piccolo, G.; Nizza, S.; et al. Productive performance and blood profiles of laying hens fed Hermetia illucens larvae meal as total replacement of soybean meal from 24 to 45 weeks of age. Poult. Sci. 2017, 96, 1783–1790. [Google Scholar] [CrossRef]
- Renna, M.; Schiavone, A.; Gai, F.; Dabbou, S.; Lussiana, C.; Malfatto, V.; Prearo, M.; Capucchio, M.T.; Biasato, I.; Biasibetti, E.; et al. Evaluation of the suitability of a partially defatted black soldier fly (Hermetia illucens L.) larvae meal as ingredient for rainbow trout (Oncorhynchus mykiss Walbaum) diets. J. Anim. Sci. Biotechnol. 2017, 8, 57. [Google Scholar] [CrossRef]
- Belghit, I.; Liland, N.S.; Gjesdal, P.; Biancarosa, I.; Menchetti, E.; Li, Y.; Waagbo, R.; Krogdahl, Å.; Lock, E.-J. Black soldier fly larvae meal can replace fish meal in diets of sea-water phase Atlantic salmon (Salmo salar). Aquaculture 2019, 503, 609–619. [Google Scholar] [CrossRef]
- Chia, S.; Tanga, C.; Osuga, I.; Alaru, A.; Mwangi, D.; Githinji, M.; Dubois, T.; Ekesi, S.; van Loon, J.; Dicke, M. Black soldier fly larval meal in feed enhances growth performance, carcass yield and meat quality of finishing pigs. J. Insects Food Feed. 2021, 7, 433–447. [Google Scholar] [CrossRef]
- Lei, X.; Kim, T.; Park, J.; Kim, I. Evaluation of Supplementation of Defatted Black Soldier Fly (Hermetia illucens) Larvae Meal in Beagle Dogs. Ann. Anim. Sci. 2019, 19, 767–777. [Google Scholar] [CrossRef] [Green Version]
- Kröger, S.; Heide, C.; Zentek, J. Evaluation of an extruded diet for adult dogs containing larvae meal from the Black soldier fly (Hermetia illucens). Anim. Feed. Sci. Technol. 2020, 270, 114699. [Google Scholar] [CrossRef]
- Freel, T.A.; McComb, A.; Koutsos, E.A. Digestibility and safety of dry black soldier fly larvae meal and black soldier fly larvae oil in dogs. J. Anim. Sci. 2021, 99, 47. [Google Scholar] [CrossRef]
- Menon, R.; Gonzalez, T.; Ferruzzi, M.; Jackson, E.; Winderl, D.; Watson, J. Oats-from farm to fork. Adv. Food Nutr. Res. 2016, 77, 1–55. [Google Scholar] [CrossRef]
- Soycan, G.; Schär, M.Y.; Kristek, A.; Boberska, J.; Alsharif, S.N.; Corona, G.; Shewry, P.R.; Spencer, J.P. Composition and content of phenolic acids and avenanthramides in commercial oat products: Are oats an important polyphenol source for consumers? Food Chem. X 2019, 3, 100047. [Google Scholar] [CrossRef]
- Ferreira, L.G.; Endrighi, M.; Lisenko, K.G.; Oliveira, M.R.D.; Damasceno, M.R.; Claudino, J.A.; Gutierres, P.G.; Peconick, A.P.; Saad, F.M.O.B.; Zangeronimo, M.G. Oat beta-glucan as a dietary supplement for dogs. PLoS ONE 2018, 13, e0201133. [Google Scholar] [CrossRef]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef]
- Whitehead, A.; Beck, E.J.; Tosh, S.; Wolever, T.M. Cholesterol-lowering effects of oat β-glucan: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 100, 1413–1421. [Google Scholar] [CrossRef] [Green Version]
- Bao, L.; Cai, X.; Xu, M.; Li, Y. Effect of oat intake on glycaemic control and insulin sensitivity: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2014, 112, 457–466. [Google Scholar] [CrossRef]
- Ho, H.V.T.; Sievenpiper, J.L.; Zurbau, A.; Mejia, S.B.; Jovanovski, E.; Au-Yeung, F.; Jenkins, A.L.; Vuksan, V. The effect of oatβ-glucan on LDL-cholesterol, non-HDL-cholesterol and apoB for CVD risk reduction: A systematic review and meta-analysis of randomised-controlled trials. Br. J. Nutr. 2016, 116, 1369–1382. [Google Scholar] [CrossRef] [Green Version]
- Chang, H.-C.; Huang, C.-N.; Yeh, D.-M.; Wang, S.-J.; Peng, C.-H.; Wang, C.-J. Oat prevents obesity and abdominal fat distribution, and improves liver function in humans. Plant Foods Hum. Nutr. 2013, 68, 18–23. [Google Scholar] [CrossRef]
- Angelov, A.; Gotcheva, V.; Kuncheva, R.; Hristozova, T. Development of a new oat-based probiotic drink. Int. J. Food Microbiol. 2006, 112, 75–80. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N. Probiotic fermentation of plant based products: Possibilities and opportunities. Crit. Rev. Food Sci. Nutr. 2012, 52, 183–199. [Google Scholar] [CrossRef]
- Asadzadeh, A.; Jalali, H.; Azizi, M.H.; Nafchi, A.M. Production of oat bran functional probiotic beverage using Bifidobacterium lactis. J. Food Meas. Charact. 2021, 15, 1301–1309. [Google Scholar] [CrossRef]
- Russo, P.; de Chiara, M.L.V.; Capozzi, V.; Arena, M.P.; Amodio, M.L.; Rascón, A.; Dueñas, M.T.; López, P.; Spano, G. Lactobacillus plantarum strains for multifunctional oat-based foods. LWT. 2016, 68, 288–294. [Google Scholar] [CrossRef] [Green Version]
- Bei, Q.; Wu, Z.; Chen, G. Dynamic changes in the phenolic composition and antioxidant activity of oats during simultaneous hydrolysis and fermentation. Food Chem. 2020, 305, 125269. [Google Scholar] [CrossRef]
- Bocchi, S.; Rocchetti, G.; Elli, M.; Lucini, L.; Lim, C.-Y.; Morelli, L. The combined effect of fermentation of lactic acid bacteria and in vitro digestion on metabolomic and oligosaccharide profile of oat beverage. Food Res. Int. 2021, 142, 110216. [Google Scholar] [CrossRef] [PubMed]
- Association of American Feed Control Officials (AAFCO). Model Bill and Regulations; 2016 Official Publication; Association of American Feed Control Officials: Oxford, IN, USA, 2016; pp. 107–234. [Google Scholar]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2006. [Google Scholar]
- Laflamme, D.R.P.C. Development and validation of a body condition score system for dogs. Canine Pract. 1997, 22, 10–15. [Google Scholar]
- Moxham, G. Waltham feces scoring system-A tool for veterinarians and pet owners: How does your pet rate. Walth. Focus 2001, 11, 24–25. [Google Scholar]
- Traughber, Z.T.; He, F.; Hoke, J.M.; Davenport, G.M.; Rodriguez-Zas, S.L.; Southey, B.R.; de Godoy, M.R.C. Ancient grains as novel dietary carbohydrate sources in canine diets. J. Anim. Sci. 2021, 99, 080. [Google Scholar] [CrossRef]
- Inal, F.; Alatas, M.S.; Kahraman, O.; Inal, S.; Uludag, M.; Gurbuz, E.; Polat, E.S. Barley as an alternative to rice in dog food. Turk. J. Vet. Anim. Sci. 2017, 41, 770–774. [Google Scholar] [CrossRef]
- El-Wahab, A.A.; Wilke, V.; Grone, R.; Visscher, C. Nutrient digestibility of a vegetarian diet with or without the supplementation of feather meal and either corn meal, fermented rye or rye and its effect on fecal quality in dogs. Animals 2021, 11, 496. [Google Scholar] [CrossRef]
- Yang, X.; Chen, H.; Gao, H.; Li, Z. Bioconversion of corn straw by coupling ensiling and solid-state fermentation. Bioresour. Technol. 2001, 78, 277–280. [Google Scholar] [CrossRef]
- Lee, E.; Nam, K.-T.; Lee, K.-W.; Lee, S.-R. Pediococcus spp.-fermented chicken meat for dogs. J. Anim. Sci. Technol. 2020, 62, 84–93. [Google Scholar] [CrossRef] [Green Version]
- Gaschen, F.P.; Merchant, S.R. Adverse food reactions in dogs and cats. Veter-Clin. North Am. Small Anim. Pract. 2011, 41, 361–379. [Google Scholar] [CrossRef]
- Bashir, S.J.; Chew, A.-L.; Anigbogu, A.; Dreher, F.; Maibach, H.I. Physical and physiological effects of stratum corneum tape stripping. Ski. Res. Technol. 2001, 7, 40–48. [Google Scholar] [CrossRef]
- Shimada, K.; Yoshihara, T.; Yamamoto, M.; Konno, K.; Momoi, Y.; Nishifuji, K.; Iwasaki, T. Transepidermal water loss (TEWL) reflects skin barrier function of dog. J. Veter-Med Sci. 2008, 70, 841–843. [Google Scholar] [CrossRef] [Green Version]
- Bhagat, R.; Sheikh, A.A.; Wazir, V.S.; Mishra, A.; Maibam, U. Food allergy in canines: A review. J. Entomol. Zool. Stud. 2017, 5, 1522–1525. [Google Scholar]
- Kara, M.; Beser, O.; Konukoglu, D.; Cokugras, H.C.; Erkan, T.; Kutlu, T.; Cokugras, F. The utility of TNF-α, IL-6 and IL-10 in the diagnosis and/or follow-up food allergy. Allergol. Immunopathol. 2020, 48, 48–55. [Google Scholar] [CrossRef]
- Stanton, C.; Ross, R.P.; Fitzgerald, G.F.; Van Sinderen, D. Fermented functional foods based on probiotics and their biogenic metabolites. Curr. Opin. Biotechnol. 2005, 16, 198–203. [Google Scholar] [CrossRef]
- Park, D.H.; Kothari, D.; Niu, K.M.; Han, S.G.; Yoon, J.E.; Lee, H.G.; Kim, S.K. Effect of fermented medicinal plants as dietary additives on food preference and fecal microbial quality in dogs. Animals 2019, 9, 690. [Google Scholar] [CrossRef] [Green Version]
- Mikawa, S.; Matsuda, A.; Kamemori, Y.; Asanuma, S.; Kitagawa, H. Enhancement of natural killer cell activity by oral administration of a fermented soybean product in dogs. Open. Vet. J. 2021, 11, 394–400. [Google Scholar] [CrossRef]
- He, W.; Gao, Y.; Guo, Z.; Yang, Z.; Wang, X.; Liu, H.; Sun, H.; Shi, B. Effects of fermented wheat bran and yeast culture on growth performance, immunity and intestinal microflora in growing-finishing pigs. J. Anim. Sci. 2021, 11, skab308. [Google Scholar] [CrossRef]
- Whalan, J.E. A toxicologist’s Guide to Clinical Pathology in Animals: Hematology, Clinical Chemistry, Urinalysis; Springer: Cham, Switzerland, 2015; pp. 1–94. [Google Scholar]
- Gizzarelli, M.; Calabrò, S.; Vastolo, A.; Molinaro, G.; Balestrino, I.; Cutrignelli, M.I. Clinical findings in healthy dogs fed with diets characterized by different carbohydrates sources. Front. Veter.-Sci. 2021, 8, 382. [Google Scholar] [CrossRef]
- Fielder, S.E. Serum Biochemical Reference Ranges: MSD Manual Veterinary Manual, DVM, MS, DACVP (Clinical Pathology); Merck & Co., Inc., Oklahoma State University: Kenilworth, NJ, USA, 2015. [Google Scholar]
- Dall’Aglio, C.; Maranesi, M.; Di Loria, A.; Piantedosi, D.; Ciaramella, P.; Alterisio, M.C.; Lepri, E.; Mercati, F. Effects of Obesity on Adiponectin System Skin Expression in Dogs: A Comparative Study. Animals 2021, 11, 2308. [Google Scholar] [CrossRef]
- Chang, Y.M.; Hadox, E.; Szladovits, B.; Garden, O.A. Serum biochemical phenotypes in the domestic dog. PLoS ONE 2016, 11, e0149650. [Google Scholar] [CrossRef]
- Wood, J.D.; Migicovsky, B.B. The effect of dietary oils and fatty acids on cholesterol metabolism in the rat. Can. J. Biochem. Physiol. 1958, 36, 433–438. [Google Scholar] [CrossRef]
- Hashim, S.A.; Arteaga, A.; Van, I.T.B.; Cozanitis, D.A. Effect of a saturated medium-chain triglyceride on serum lipids in man. Lancet 1960, 1, 1105–1108. [Google Scholar] [CrossRef]
- Denke, M.A. Dietary fats, fatty acids, and their effects on lipoproteins. Curr. Atheroscler. Rep. 2006, 8, 466–471. [Google Scholar] [CrossRef]
- German, J.B.; Dillard, C.J. Saturated fats: What dietary intake? Am. J. Clin. Nutr. 2004, 80, 550–559. [Google Scholar] [CrossRef]
- Dayrit, F.M. The properties of lauric acid and their significance in coconut oil. J. Am. Oil. Chem. Soc. 2015, 92, 1–15. [Google Scholar] [CrossRef]
| Items | Ingredients | Groups 1 | ||||
|---|---|---|---|---|---|---|
| FO | BSFL | CON | FO | BSFL | FO + BSFL | |
| Ingredients (%) | ||||||
| Rice powder | 58.03 | 48.82 | 56.07 | 47.23 | ||
| Poultry meal | 22.5 | 22.0 | 20.0 | 19.22 | ||
| BSFL | - | - | 5 | 5 | ||
| FO | - | 10 | - | 10 | ||
| Egg yolk powder | 13.12 | 13.08 | 13.13 | 13.25 | ||
| Calcium carbonate | 1.35 | 1.3 | 1.0 | 0.9 | ||
| Lard | 1.0 | 1.0 | 0.9 | 0.6 | ||
| Cabbage powder | 1.0 | 1.0 | 1.0 | 1.0 | ||
| Seaweed (Enteromorpha) powder | 1.0 | 1.0 | 1.0 | 1.0 | ||
| Potassium citrate | 0.6 | 0.6 | 0.6 | 0.6 | ||
| Vit. and Min. premix 2 | 0.5 | 0.5 | 0.5 | 0.5 | ||
| Salt | 0.4 | 0.4 | 0.4 | 0.4 | ||
| Calcium phosphate | 0.3 | 0.3 | 0.4 | 0.3 | ||
| Analyzed composition (%DM) | ||||||
| Moisture | 4.36 | 3.62 | 10.9 | 10.24 | 10.18 | 12.21 |
| Crude protein (CP) | 18.95 | 46.83 | 31.32 | 32.11 | 31.17 | 31.79 |
| Ether extract (EE) | 8.82 | 26.87 | 11.65 | 12.19 | 12.51 | 13.01 |
| Crude fiber (CF) | 3.69 | 12.71 | 0.19 | 0.78 | 0.86 | 1.44 |
| Crude ash (CA) | 1.62 | 10.68 | 2.27 | 2.36 | 2.73 | 2.83 |
| Nitrogen-free extract (NFE) | 66.92 | 2.91 | 54.58 | 52.55 | 52.72 | 50.93 |
| Calcium (Ca) | 0.08 | 3.6 | 0.68 | 0.66 | 0.75 | 0.69 |
| Phosphorus (P) | 0.38 | 0.67 | 0.46 | 0.44 | 0.50 | 0.46 |
| Ca/P ratio | 0.21 | 5.37 | 1.47 | 1.47 | 1.50 | 1.49 |
| Calculated ME, kcal/kg 3 | 3755 | 4025 | 3997 | 3999 | 4000 | 4001 |
| Items | CON 1 | FO | BSFL | FO + BSFL | p-Value |
|---|---|---|---|---|---|
| ADFI 2, g/d | 98.0 ± 9.2 | 100.4 ± 12.0 | 102.3 ± 10.2 | 100.2 ± 12.0 | 0.735 |
| Body weight, kg | |||||
| Initial | 4.13 ± 0.75 | 4.22 ± 0.67 | 4.20 ± 0.67 | 4.17 ± 0.67 | 0.999 |
| Final | 4.49 ± 0.83 | 4.52 ± 0.76 | 4.62 ± 0.76 | 4.51 ± 0.73 | 0.999 |
| Rate of BWG 3, % | 108.7 ± 1.2 | 106.5 ± 1.2 | 109.8 ± 2.1 | 108.0 ± 1.0 | 0.437 |
| BCS 4 | |||||
| Initial | 4.20 ± 0.73 | 4.20 ± 0.58 | 3.60 ± 0.93 | 3.40 ± 0.87 | 0.850 |
| Final | 4.60 ± 0.68 | 4.20 ± 0.49 | 3.60 ± 0.81 | 3.40 ± 0.68 | 0.593 |
| Items | CON | FO | BSFL | FO + BSFL | p-Value |
|---|---|---|---|---|---|
| WBC, ×106/mL (Ref. range: 5.05–16.76) | |||||
| Initial | 8.08 ± 0.80 | 8.10 ± 1.89 | 10.26 ± 2.15 | 6.28 ± 1.00 | 0.385 |
| Final | 7.48 ± 0.81 | 9.48 ± 2.88 | 12.24 ± 1.10 * | 7.65 ± 1.08 | 0.194 |
| NEU, ×106/mL (Ref. range: 2.95–11.64) | |||||
| Initial | 5.42 ± 0.53 | 4.97 ± 1.23 | 7.23 ± 1.71 | 4.11 ± 0.86 | 0.321 |
| Final | 5.58 ± 0.54 | 6.56 ± 2.21 | 8.38 ± 1.16 | 5.57 ± 0.64 | 0.415 |
| LYM, ×106/mL (Ref. range: 1.05–5.10) | |||||
| Initial | 1.69 ± 0.28 | 2.15 ± 0.38 | 1.82 ± 0.29 | 1.40 ± 0.16 | 0.362 |
| Final | 1.48 ± 0.26 | 2.06 ± 0.39 | 2.74 ± 0.79 | 1.34 ± 0.30 | 0.200 |
| MONO, ×106/mL (Ref. range: 0.16–1.12) | |||||
| Initial | 0.55 ± 0.24 | 0.65 ± 0.25 | 0.65 ± 0.25 | 0.46 ± 0.13 | 0.919 |
| Final | 0.19 ± 0.12 | 0.55 ± 0.29 | 0.74 ± 0.20 | 0.53 ± 0.18 | 0.324 |
| EOS, ×106/mL (Ref. range: 0.06–1.23) | |||||
| Initial | 0.43 ± 0.07 | 0.33 ± 0.10 | 0.56 ± 0.22 | 0.31 ± 0.08 | 0.542 |
| Final | 0.06 ± 0.01 | 0.16 ± 0.05 | 0.27 ± 0.15 | 0.14 ± 0.06 | 0.406 |
| BASO, ×106/mL (Ref. range: 0–0.1) | |||||
| Initial | 0.00 ± 0.00 | 0.002 ± 0.00 | 0.004 ± 0.00 | 0.00 ± 0.00 | 0.547 |
| Final | 0.05 ± 0.02 | 0.11 ± 0.05 | 0.10 ± 0.03 # | 0.07 ± 0.03 | 0.516 |
| RBC, ×109/mL (Ref. range: 5.65–8.87) | |||||
| Initial | 5.85 ± 0.26 | 5.84 ± 0.17 | 6.11 ± 0.09 | 5.88 ± 0.27 | 0.783 |
| Final | 6.27 ± 0.31 | 6.01 ± 0.11 | 6.33 ± 0.18 | 6.02 ± 0.31 | 0.702 |
| HGB, g/dL (Ref. range: 13.1–20.5) | |||||
| Initial | 14.26 ± 0.61 | 13.94 ± 0.47 | 14.32 ± 0.44 | 14.16 ± 0.64 | 0.963 |
| Final | 14.40 ± 0.69 | 13.82 ± 0.54 | 14.14 ± 0.41 | 14.18 ± 0.99 | 0.947 |
| HCT, % (Ref. range: 37.3–61.7) | |||||
| Initial | 41.28 ± 1.65 | 40.36 ± 1.03 | 42.24 ± 1.06 | 40.52 ± 1.76 | 0.777 |
| Final | 44.51 ± 1.98 | 42.27 ± 0.88 | 43.89 ± 0.93 | 43.16 ± 2.25 | 0.790 |
| Items | CON | FO | BSFL | FO + BSFL | p-Value |
|---|---|---|---|---|---|
| GLU, mg/dL (Ref. range: 70–138) | |||||
| Initial | 97.2 ± 8 | 98.6 ± 4.34 | 103.6 ± 6.45 | 95.8 ± 6.07 | 0.835 |
| Final | 94.6 ± 4.58 | 95.6 ± 4.34 | 91.8 ± 3.12 | 97 ± 3.51 | 0.816 |
| CREA, mg/dL (Ref. range: 0.5–1.6) | |||||
| Initial | 0.64 ± 0.09 | 0.64 ± 0.07 | 0.68 ± 0.05 | 0.6 ± 0.05 | 0.883 |
| Final | 0.72 ± 0.12 | 0.72 ± 0.09 | 0.72 ± 0.07 | 0.68 ± 0.07 | 0.984 |
| BUN, mg/dL (Ref. range: 6.0–31) | |||||
| Initial | 13.6 ± 0.93 | 14.4 ± 1.63 | 16.4 ± 1.47 | 15 ± 1.38 | 0.545 |
| Final | 14.6 ± 1.25 | 15 ± 1.22 | 15.8 ± 1.2 | 14 ± 1.41 | 0.789 |
| PHOS, mg/dL (Ref. range: 2.5–6.0) | |||||
| Initial | 4.74 ± 0.35 | 4.62 ± 0.37 | 4.22 ± 0.29 | 4.32 ± 0.32 | 0.655 |
| Final | 4.54 ± 0.26 | 4.54 ± 0.47 | 3.52 ± 0.31 * | 3.72 ± 0.25 | 0.049 |
| CA, mg/dL (Ref. range: 8.9–11.4) | |||||
| Initial | 9.18 ± 0.41 | 9.34 ± 0.29 | 9.64 ± 0.48 | 9.64 ± 0.21 | 0.295 |
| Final | 9.08 ± 0.36 | 9.38 ± 0.16 | 8.96 ± 0.32 | 8.9 ± 0.23 | 0.628 |
| T-Pro, g/dL (Ref. range: 5.0–7.4) | |||||
| Initial | 6.98 ± 0.37 | 6.76 ± 0.27 | 7.24 ± 0.17 | 6.24 ± 0.2 | 0.089 |
| Final | 7.34 ± 0.26 | 6.66 ± 0.25 | 6.62 ± 0.19 # | 6.56 ± 0.22 | 0.100 |
| ALB, g/dL (Ref. range: 2.7–4.4) | |||||
| Initial | 3.16 ± 0.12 | 3.22 ± 0.08 | 3.44 ± 0.11 | 2.94 ± 0.14 | 0.087 |
| Final | 3.22 ± 0.09 | 3.2 ± 0.09 | 3.14 ± 0.12 | 3.14 ± 0.13 | 0.935 |
| GLOB, g/dL (Ref. range: 1.6–3.6) | |||||
| Initial | 3.82 ± 0.28 | 3.54 ± 0.22 | 3.8 ± 0.17 | 3.3 ± 0.13 | 0.271 |
| Final | 4.12 ± 0.23 | 3.46 ± 0.29 | 3.48 ± 0.19 | 3.42 ± 0.12 * | 0.048 |
| A/G ratio (Ref. range: 0.8–2.0) | |||||
| Initial | 0.84 ± 0.05 | 0.92 ± 0.05 | 0.92 ± 0.07 | 0.88 ± 0.06 | 0.715 |
| Final | 0.78 ± 0.06 | 0.98 ± 0.09 | 0.92 ± 0.07 | 0.9 ± 0.03 | 0.221 |
| ALT, U/L (Ref. range: 12–118) | |||||
| Initial | 111.4 ± 48.17 | 58.8 ± 13.75 | 135.5 ± 43.56 | 41.2 ± 6.18 | 0.164 |
| Final | 87 ± 31.58 | 38.4 ± 9.9 | 97.5 ± 35.26 | 35.4 ± 8.07 | 0.246 |
| ALKP, U/L (Ref. range: 5.0–131) | |||||
| Initial | 60.6 ± 19.94 | 24.6 ± 3.61 | 62 ± 17.52 | 20.6 ± 4.55 | 0.079 |
| Final | 58.4 ± 7.93 | 36.4 ± 4.17 * | 70 ± 24.03 | 29 ± 5.39 * | 0.043 |
| GGT, U/L (Ref. range: 0–12) | |||||
| Initial | 0 ± 0 | 0 ± 0 | 3 ± 3 | 0 ± 0 | 0.418 |
| Final | 0 ± 0 | 0 ± 0 | 2.8 ± 2.8 | 0 ± 0 | 0.418 |
| T-BIL, mg/dL (Ref. range: 0.1–0.3) | |||||
| Initial | 0.28 ± 0.04 | 0.22 ± 0.04 | 0.4 ± 0.03 * | 0.3 ± 0.03 | 0.004 |
| Final | 0.28 ± 0.04 | 0.2 ± 0.04 | 0.3 ± 0.05 | 0.2 ± 0.03 | 0.254 |
| CHOL, mg/dL (Ref. range: 29–291) | |||||
| Initial | 131.4 ± 15.16 | 136.4 ± 8.89 | 177.6 ± 8.95 * | 132.8 ± 6.58 | 0.017 |
| Final | 143.2 ± 18.99 | 141.8 ± 10.94 | 159.4 ± 9.89 | 153 ± 10.29 | 0.749 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seo, K.; Cho, H.-W.; Chun, J.; Jeon, J.; Kim, C.; Kim, M.; Park, K.; Kim, K. Evaluation of Fermented Oat and Black Soldier Fly Larva as Food Ingredients in Senior Dog Diets. Animals 2021, 11, 3509. https://doi.org/10.3390/ani11123509
Seo K, Cho H-W, Chun J, Jeon J, Kim C, Kim M, Park K, Kim K. Evaluation of Fermented Oat and Black Soldier Fly Larva as Food Ingredients in Senior Dog Diets. Animals. 2021; 11(12):3509. https://doi.org/10.3390/ani11123509
Chicago/Turabian StyleSeo, Kangmin, Hyun-Woo Cho, Julan Chun, Junghwan Jeon, Chanho Kim, Minji Kim, Kwanho Park, and Kihyun Kim. 2021. "Evaluation of Fermented Oat and Black Soldier Fly Larva as Food Ingredients in Senior Dog Diets" Animals 11, no. 12: 3509. https://doi.org/10.3390/ani11123509
APA StyleSeo, K., Cho, H.-W., Chun, J., Jeon, J., Kim, C., Kim, M., Park, K., & Kim, K. (2021). Evaluation of Fermented Oat and Black Soldier Fly Larva as Food Ingredients in Senior Dog Diets. Animals, 11(12), 3509. https://doi.org/10.3390/ani11123509






