Search for Associations of FSHR, INHA, INHAB, PRL, TNP2 and SPEF2 Genes Polymorphisms with Semen Quality in Russian Holstein Bulls (Pilot Study)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Experimental Animals and Phenotypes
2.3. Isolation of Genomic DNA
2.4. PCR Conditions
2.5. Sequencing and RFLP Reactions
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Murphy, E.M.; Kelly, A.K.; O’Meara, C.; Eivers, B.; Lonergan, P.; Fair, S. Influence of bull age, ejaculate number, and season of collection on semen production and sperm motility parameters in holstein friesian bulls in a commercial artificial insemination centre. J. Anim. Sci. 2018, 96, 2408–2418. [Google Scholar] [CrossRef]
- Schenk, J.L. Review: Principles of maximizing bull semen production at genetic centers. Animal 2018, 12, s142–s147. [Google Scholar] [CrossRef] [PubMed]
- Olsen, H.B.; Heringstad, B.; Klemetsdal, G. Genetic analysis of semen characteristic traits in young Norwegian Red bulls. J. Dairy Sci. 2020, 103, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Saksa, E.I. Efficiency of use of bulls evaluated by various methods for improvement of highly productive herds. Dairy Beef Cattle Breed. 2018, 1, 5–8. [Google Scholar]
- Yin, H.; Fang, L.; Qin, C.; Zhang, S. Estimation of the genetic parameters for semen traits in Chinese Holstein bulls. BMC Genet. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Jing, J.; Shao, Y.; Zeng, R.; Wang, C.; Yao, B.; Hang, D. Circulating sex hormone levels in relation to male sperm quality. BMC Urol. 2020, 20. [Google Scholar] [CrossRef]
- Salas-Huetos, A.; Maghsoumi-Norouzabad, L.; James, E.R.; Carrell, D.T.; Aston, K.I.; Jenkins, T.G.; Becerra-Tomás, N.; Javid, A.Z.; Abed, R.; Torres, P.J.; et al. Male adiposity, sperm parameters and reproductive hormones: An updated systematic review and collaborative meta-analysis. Obes. Rev. 2021, 22. [Google Scholar] [CrossRef]
- Sun, L.P.; Du, Q.Z.; Song, Y.P.; Yu, J.N.; Wang, S.J.; Sang, L.; Song, L.W.; Yue, Y.M.; Lian, Y.Z.; Zhang, S.L.; et al. Polymorphisms in luteinizing hormone receptor and hypothalamic gonadotropin-releasing hormone genes and their effects on sperm quality traits in Chinese Holstein bulls. Mol. Biol. Rep. 2012, 39, 7117–7123. [Google Scholar] [CrossRef]
- Pengo, M.; Ferlin, A.; Arredi, B.; Ganz, F.; Selice, R.; Garolla, A.; Foresta, C. FSH receptor gene polymorphisms in fertile and infertile Italian men. Reprod. Biomed. Online 2006, 13, 795–800. [Google Scholar] [CrossRef]
- Huhtaniemi, I. Mutations along the pituitary-gonadal axis affecting sexual maturation: Novel information from transgenic and knockout mice. Mol. Cell. Endocrinol. 2006, 254–255, 84–90. [Google Scholar] [CrossRef]
- Tapanainen, J.S.; Aittomäki, K.; Min, J.; Vaskivuo, T.; Huhtaniemi, I.T. Men homozygous for an inactivating mutation of the follicle-stimulating hormone (FSH) receptor gene present variable suppression of spermatogenesis and fertility. Nat. Genet. 1997, 15, 205–206. [Google Scholar] [CrossRef]
- Krishnamurthy, H.; Danilovich, N.; Morales, C.R.; Sairam, M.R. Qualitative and quantitative decline in spermatogenesis of the follicle- stimulating hormone receptor knockout (FORKO) mouse. Biol. Reprod. 2000, 62, 1146–1159. [Google Scholar] [CrossRef]
- Von Eckardstein, S.; Simoni, M.; Bergmann, M.; Weinbauer, G.F.; Gassner, P.; Schepers, A.G.; Nieschlag, E. Serum inhibin B in combination with serum follicle-stimulating hormone (FSH) is a more sensitive marker than serum FSH alone for impaired spermatogenesis in men, but cannot predict the presence of sperm in testicular tissue samples. J. Clin. Endocrinol. Metab. 1999, 84, 2496–2501. [Google Scholar] [CrossRef] [PubMed]
- Weerakoon, W.W.P.N.; Sakase, M.; Kawate, N.; Hannan, M.A.; Kohama, N.; Tamada, H. Plasma IGF-I, INSL3, testosterone, inhibin concentrations and scrotal circumferences surrounding puberty in Japanese Black beef bulls with normal and abnormal semen. Theriogenology 2018, 114, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Hiendleder, S.; Dodds, K.G.; Wassmuth, R. Linkage mapping of the ovine α-inhibin (INHA), β(A)-inhibin/activin (INHBA), and β(B)-inhibin/activin (INHBB) genes. J. Hered. 2000, 91, 343–345. [Google Scholar] [CrossRef][Green Version]
- Giesecke, K.; Hamann, H.; Sieme, H.; Distl, O. INHBA-Associated markers as candidates for stallion fertility. Reprod. Domest. Anim. 2010, 45, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Bartke, A. Prolactin in the male: 25 Years later. J. Androl. 2004, 25, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Aiman, J.; McAsey, M.; Harms, L. Serum and seminal plasma prolactin concentrations in men with normospermia, oligospermia, or azoospermia. Fertil. Steril. 1988, 49, 133–137. [Google Scholar] [CrossRef]
- Steger, R.W.; Chandrashekar, V.; Zhao, W.; Bartke, A.; Horseman, N.D. Neuroendocrine and reproductive functions in male mice with targeted disruption of the prolactin gene. Endocrinology 1998, 139, 3691–3695. [Google Scholar] [CrossRef]
- Giesecke, K.; Hamann, H.; Sieme, H.; Distl, O. Evaluation of prolactin receptor (prlr) as candidate gene for male fertility in hanoverian warmblood horses. Reprod. Domest. Anim. 2010, 45. [Google Scholar] [CrossRef]
- Meistrich, M.L. Nuclear Morphogenesis during Spermiogenesis. In Molecular Biology of the Male Reproductive System; Academic Press: Cambridge, MA, USA, 1993; pp. 67–97. [Google Scholar]
- Meistrich, M.L. Histone and basic nuclear protein transitions in mammalian spermatogenesis. In Histones and Other Basic Nuclear Proteins; CRC Press: Boca Raton, FL, USA, 1989; pp. 165–182. [Google Scholar]
- Baskaran, R.; Rao, M.R.S. Interaction of spermatid-specific protein TP2 with nucleic acids, in vitro. A comparative study with TP1. J. Biol. Chem. 1990, 265, 21039–21047. [Google Scholar] [CrossRef]
- Adham, I.M.; Nayernia, K.; Burkhardt-Göttges, E.; Topaloglu, Ö.; Dixkens, C.; Holstein, A.F.; Engel, W. Teratozoospermia in mice lacking the transition protein 2 (Tnp2). Mol. Hum. Reprod. 2001, 7, 513–520. [Google Scholar] [CrossRef]
- Heidari, M.M.; Danafar, A.; Moezzi, F.; Khatami, M.; Talebi, A.R. The association between TNP2 gene polymorphisms and iranian infertile men with varicocele: A case-control study. Int. J. Reprod. Biomed. 2019, 17, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Sironen, A.; Hansen, J.; Thomsen, B.; Andersson, M.; Vilkki, J.; Toppari, J.; Kotaja, N. Expression of SPEF2 during Mouse Spermatogenesis and Identification of IFT20 as an Interacting Protein. Biol. Reprod. 2010, 82, 580–590. [Google Scholar] [CrossRef]
- Sironen, A.; Uimari, P.; Iso-Touru, T.; Vilkki, J. L1 insertion within SPEF2 gene is associated with increased litter size in the Finnish Yorkshire population. J. Anim. Breed. Genet. 2012, 129, 92–97. [Google Scholar] [CrossRef]
- Chen, M.; Yan, H.; Wang, K.; Cui, Y.; Chen, R.; Liu, J.; Zhu, H.; Qu, L.; Pan, C. Goat SPEF2: Expression profile, indel variants identification and association analysis with litter size. Theriogenology 2019, 139, 147–155. [Google Scholar] [CrossRef]
- Sironen, A.; Kotaja, N.; Mulhern, H.; Wyatt, T.A.; Sisson, J.H.; Pavlik, J.A.; Miiluniemi, M.; Fleming, M.D.; Lee, L. Loss of SPEF2 function in mice results in spermatogenesis defects and primary ciliary dyskinesia. Biol. Reprod. 2011, 85, 690–701. [Google Scholar] [CrossRef]
- Sha, Y.; Liu, W.; Wei, X.; Zhu, X.; Luo, X.; Liang, L.; Guo, T. Biallelic mutations in Sperm flagellum 2 cause human multiple morphological abnormalities of the sperm flagella (MMAF) phenotype. Clin. Genet. 2019, 96, 385–393. [Google Scholar] [CrossRef]
- Ensembl. Available online: https://www.ensembl.org/Bos_taurus/Variation/Explore?db=core;r=4:79295681-79296681;v=rs43408735;vdb=variation;vf=9956999 (accessed on 25 January 2021).
- Sang, L.; Du, Q.-Z.; Yang, W.-C.; Tang, K.-Q.; Yu, J.-N.; Hua, G.-h.; Zhang, X.-X.; Yang, L.-G. Polymorphisms in follicle stimulation hormone receptor, inhibin alpha, inhibin bata A, and prolactin genes, and their association with sperm quality in Chinese Holstein bulls. Anim. Reprod. Sci. 2011, 126, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Kuroi, N.; Sano, M.; Tabara, Y.; Katsuya, T.; Ogihara, T.; Makita, Y.; Hata, A.; Yamada, M.; Takahashi, N.; et al. Mutation of the follicle-stimulating hormone receptor gene 5′-untranslated region associated with female hypertension. Hypertension 2006, 48, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Seok, O.S.; Ahn, J.M.; Mayo, K.E.; Cho, B.N. Developmental changes in inhibin-α gene expression in the mouse testis. Mol. Cells 2004, 17, 67–72. [Google Scholar]
- Tang, K.Q.; Li, S.J.; Yang, W.C.; Yu, J.N.; Han, L.; Li, X.; Yang, L.G. An MspI polymorphism in the inhibin alpha gene and its associations with superovulation traits in Chinese Holstein cows. Mol. Biol. Rep. 2011, 38, 17–21. [Google Scholar] [CrossRef]
- Wang, J.; Li, Z.J.; Lan, X.Y.; Hua, L.S.; Huai, Y.T.; Huang, Y.Z.; Ma, L.; Zhao, M.; Jing, Y.J.; Chen, H.; et al. Two novel SNPs in the coding region of the bovine PRDM16 gene and its associations with growth traits. Mol. Biol. Rep. 2010, 37, 571–577. [Google Scholar] [CrossRef]
- Krause, W.; Bohring, C. Inhibin B as a marker of spermatogenesis. A new dimension in andrology. Hautarzt 2002, 53, 5–10. [Google Scholar] [CrossRef]
- Meehan, T.; Schlatt, S.; O’Bryan, M.K.; De Kretser, D.M.; Loveland, K.L. Regulation of germ cell and sertoli cell development by activin, follistatin, and FSH. Dev. Biol. 2000, 220, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Boitani, C.; Stefanini, M.; Fragale, A.; Morena, A.R. Activin stimulates sertoli cell proliferation in a defined period of rat testis development. Endocrinology 1995, 136, 5438–5444. [Google Scholar] [CrossRef]
- Buzzard, J.J.; Farnworth, P.G.; De Kretser, D.M.; O’Connor, A.E.; Wreford, N.G.; Morrison, J.R. Proliferative phase Sertoli cells display a developmentally regulated response to activin in vitro. Endocrinology 2003, 144, 474–483. [Google Scholar] [CrossRef]
- Barakat, B.; O’Connor, A.E.; Gold, E.; de Kretser, D.M.; Loveland, K.L. Inhibin, activin, follistatin and FSH serum levels and testicular production are highly modulated during the first spermatogenic wave in mice. Reproduction 2008, 136, 345–359. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Ju, Z.; Zhang, Y.; Huang, J.; Zhang, X.; Qi, C.; Li, J.; Zhong, J.; Li, G.; Wang, C. Association of TNP2 gene polymorphisms of the bta-miR-154 target site with the semen quality traits of Chinese Holstein bulls. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Sironen, A.; Thomsen, B.; Andersson, M.; Ahola, V.; Vilkki, J. An intronic insertion in KPL2 results in aberrant splicing and causes the immotile short-tail sperm defect in the pig. Proc. Natl. Acad. Sci. USA 2006, 103, 5006–5011. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Yang, B.; Ju, Z.H.; Wang, X.G.; Qi, C.; Zhang, Y.; Wang, C.F.; Liu, H.D.; Feng, M.Y.; Chen, Y.; et al. Alternative splicing, promoter methylation, and functional SNPs of sperm flagella 2 gene in testis and mature spermatozoa of Holstein bulls. Reproduction 2014, 147, 241–252. [Google Scholar] [CrossRef] [PubMed]
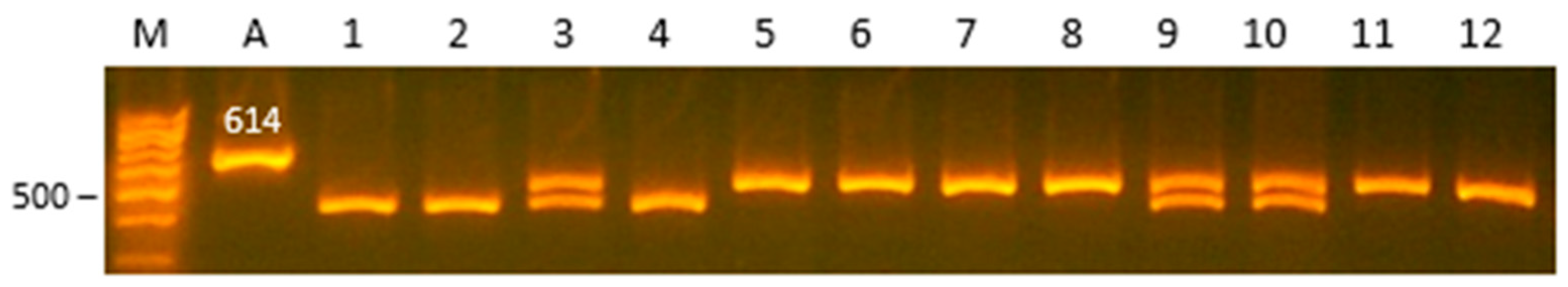
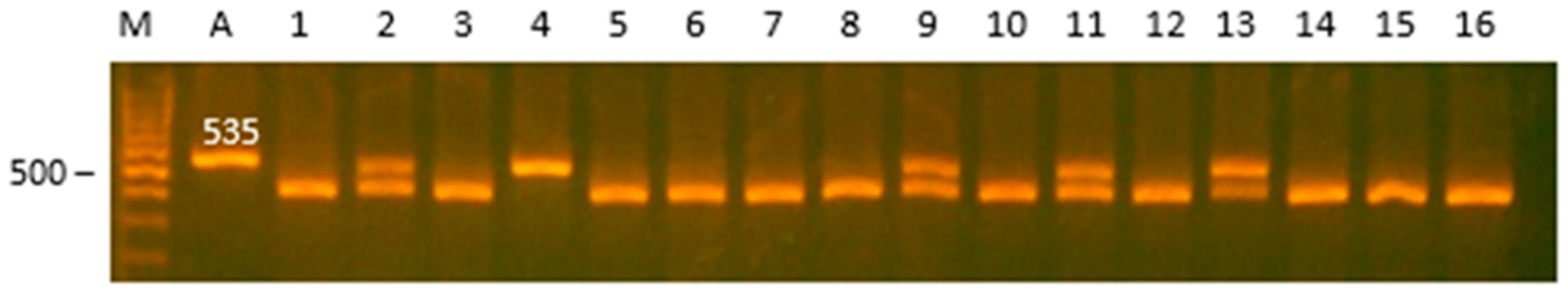
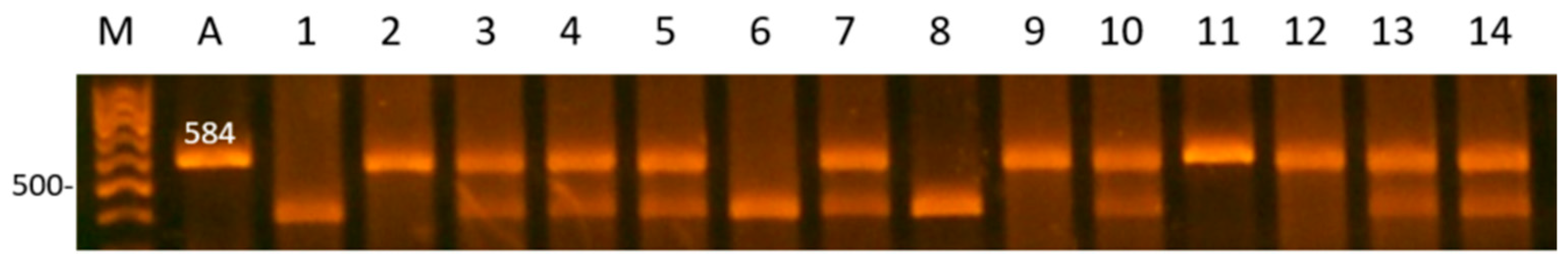
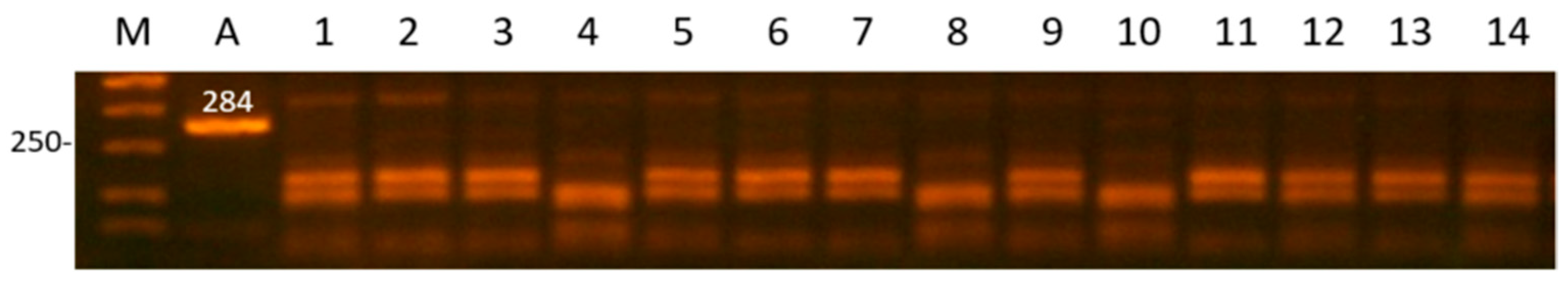
| Gene | Locus | Primer Sequence (5′ > 3′) | AT (C0) | RE | RES, bp/Genotype |
|---|---|---|---|---|---|
| FSHR | 5′-upstream region | F:TGTTCCCACTGACTCAGATACTT R:TCCCTGCCCTTCAGTGACGAA | 61 | TaqI | AA: 446, 97, 71 AT: 517, 446, 97, 71 TT: 517, 97 |
| INHA | Part of exon 1 | F:ATTCAACCCAACCTGCCTA R:GCCCTGTTTCTGGATGCC | 61 | MspI | AA: 123, 95, 31 AG: 123, 95, 79, 44, 31 GG: 95, 79, 44, 31 |
| INHAB | Intron 1 | F: CAGGGTTTCAGAAGTTGG R:GGTGGTTGTTACTGTTTATC | 61 | Seq 1 | |
| PRL-1 | Complete of exon 4 | F:GAGATTGTTTCTTGTGGTTGTTCAG R:TTCAAAACTCATTCCTCATTACAGA | 62 | RsaI | 284 n/p 2 |
| PRL-2 | Part of exon 5 | F:GATAAATAGAAAGAACAAAGATGAG R:AACCCATTAGAGCCAAGC | 60 | AluI | 372 n/p 2 |
| TNP2 | 3′-upstream region | F:ACTGGACCAATGAACGAA R:CTCCCTACCCAACCTCTT | 61 | HindIII | TT: 432, 103 TC: 535, 432, 103 CC: 535 |
| SPEF2 | 5′-upstream region | F: TAATCCTCCACCAGAATCTG R:GCCTTATGATGTGGGTATGA | 61 | TaqI | TT: 584 TG: 584, 460, 124 GG: 460, 124 |
| Gene | Genotype | Frequency of Genotypes | Alleles |
|---|---|---|---|
| FSHR (rs43676359) | AA (69) | 0.365 | A-0.651 |
| AT (108) | 0.571 | T-0.349 | |
| TT (12) | 0.063 | ||
| INHA (rs41257116) | AA (0) | 0.000 | A-0.413 |
| AG (156) | 0.825 | G-0.587 | |
| GG (33) | 0.175 | ||
| INHAB (rs43408735) | AA (69) | 0.365 | A-0.569 |
| AG (77) | 0.407 | G-0.431 | |
| GG (43) | 0.227 | ||
| TNP2 (g.1536) | CC (21) | 0.111 | C-0.196 |
| CT (32) | 0.169 | T-0.804 | |
| TT (136) | 0.720 | ||
| SPEF2 (c.2851G>T) | GG (65) | 0.344 | G-0.603 |
| GT (98) | 0.518 | T-0.397 | |
| TT (26) | 0.138 |
| Gene | DVOL (mL) | SCON (×108/mL) | TNS (×108) | PMOT (%) |
|---|---|---|---|---|
| Number of samples | 189 | 189 | 189 | 189 |
| means | 16.3 | 1.45 | 24.16 | 85.9 |
| standard deviation | 5.0 | 0.22 | 13.5 | 4.9 |
| minimum | 3 | 0.70 | 2.7 | 80.0 |
| maximum | 27 | 2.00 | 46.0 | 90.0 |
| Gene | Genotype | DVOL (mL) | SCON (×108/mL) | TNS (×108) | PMOT (%) |
|---|---|---|---|---|---|
| FSHR | AA (69) | 17.2 ± 5.43 a | 1.41 ± 0.21 a | 25.8 ± 10.55 a | 86.3 ± 5.21 |
| AT (108) | 16.8 ± 7.23 | 1.45 ± 0.27 | 25.2 ± 14.83 | 84.3 ± 4.27 | |
| TT (12) | 11.965 ± 7.58 b | 1.29 ± 0.32 b | 15.6 ± 12.16 b | 82.9 ± 5.13 | |
| INHA | AG (156) | 16.56 ± 5.67 | 1.39 ± 0.26 a | 23.55 ± 12.54 | 84.24 ± 4.67 |
| GG (33) | 17.20 ± 6.83 | 1.71 ± 0.29 b | 28.43 ± 13.76 | 86.76 ± 5.59 | |
| INHAB | AA (69) | 19.24 ± 6.35 a | 1.45 ± 0.22 | 32.54 ± 8.59 a | 87.13 ± 4.43 |
| AG (77) | 16.45 ± 8.67 b | 1.46 ± 0.33 | 23.35 ± 4.32 b | 83.21 ± 5.24 | |
| GG (43) | 11.32 ± 7.57 b | 1.30 ± 0.31 | 14.85 ± 1.98 b | 83.56 ± 5.37 | |
| TNP2 | CC (21) | 14.70 ± 8.65 | 1.35 ± 0.36 | 19.64 ± 16.93 | 83.48 ± 5.56 |
| CT (32) | 14.50 ± 9.72 | 1.26 ± 0.36 | 19.29 ± 16.06 | 86.23 ± 5.49 | |
| TT (136) | 16.21 ± 7.74 | 1.43 ± 0.24 | 25.37 ± 12.23 | 85.57 ± 5.34 | |
| SPEF2 | GG (65) | 16.34 ± 7.87 | 1.41 ± 0.23 | 23.33 ± 12.74 | 84.83 ± 5.23 |
| GT (98) | 17.00 ± 7.89 | 1.43 ± 0.23 | 25.84 ± 12.20 | 85.30 ± 4.89 | |
| TT (26) | 15.46 ± 9.64 | 1.38 ± 0.42 | 23.38 ± 17.99 | 85.12 ± 5.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikitkina, E.; Krutikova, A.; Musidray, A.; Plemyashov, K. Search for Associations of FSHR, INHA, INHAB, PRL, TNP2 and SPEF2 Genes Polymorphisms with Semen Quality in Russian Holstein Bulls (Pilot Study). Animals 2021, 11, 2882. https://doi.org/10.3390/ani11102882
Nikitkina E, Krutikova A, Musidray A, Plemyashov K. Search for Associations of FSHR, INHA, INHAB, PRL, TNP2 and SPEF2 Genes Polymorphisms with Semen Quality in Russian Holstein Bulls (Pilot Study). Animals. 2021; 11(10):2882. https://doi.org/10.3390/ani11102882
Chicago/Turabian StyleNikitkina, Elena, Anna Krutikova, Artem Musidray, and Kirill Plemyashov. 2021. "Search for Associations of FSHR, INHA, INHAB, PRL, TNP2 and SPEF2 Genes Polymorphisms with Semen Quality in Russian Holstein Bulls (Pilot Study)" Animals 11, no. 10: 2882. https://doi.org/10.3390/ani11102882
APA StyleNikitkina, E., Krutikova, A., Musidray, A., & Plemyashov, K. (2021). Search for Associations of FSHR, INHA, INHAB, PRL, TNP2 and SPEF2 Genes Polymorphisms with Semen Quality in Russian Holstein Bulls (Pilot Study). Animals, 11(10), 2882. https://doi.org/10.3390/ani11102882








