Assessing the Genetic Background and Selection Signatures of Huaxi Cattle Using High-Density SNP Array
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Selection
2.2. Genotyping and Quality Control
2.3. Genetic Diversity, ROH Detection, and Linkage Disequilibrium Analysis
2.4. Population Structure and Phylogenetic Analysis
2.5. Identification of Selection Signatures
2.6. Gene Annotation and Enrichment Analysis
3. Results
3.1. Genetic Diversity
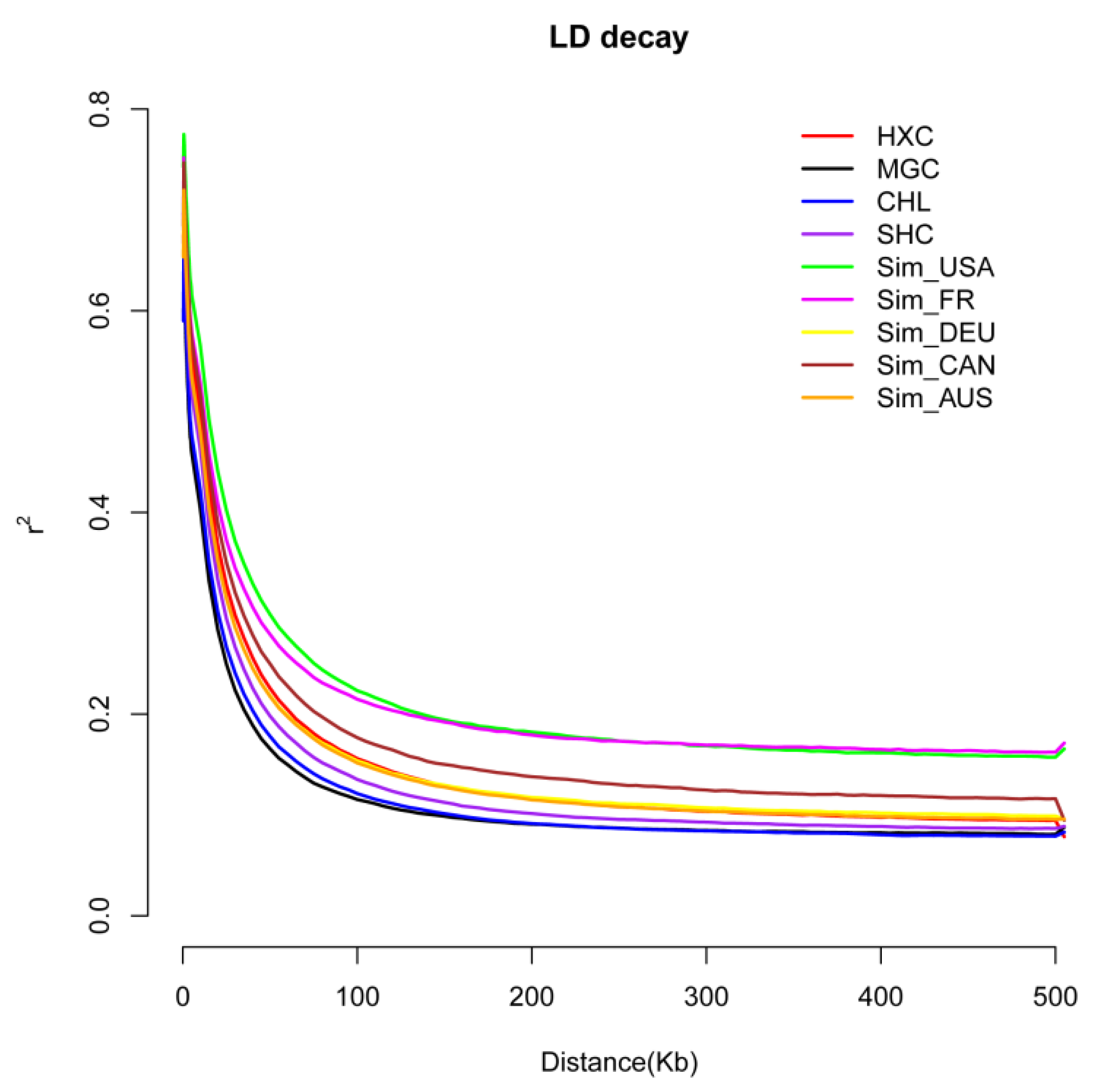
3.2. Linkage Disequilibrium
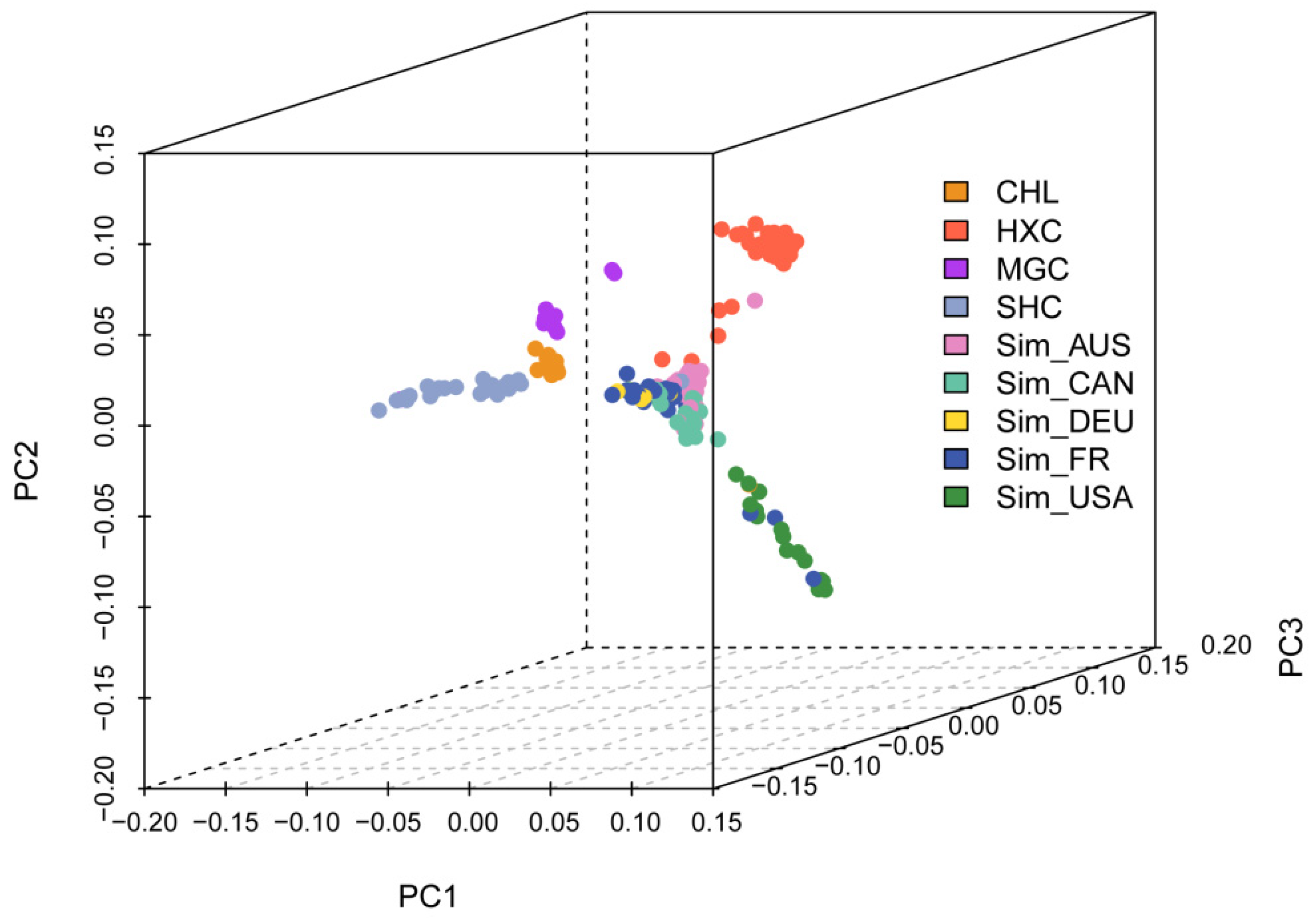
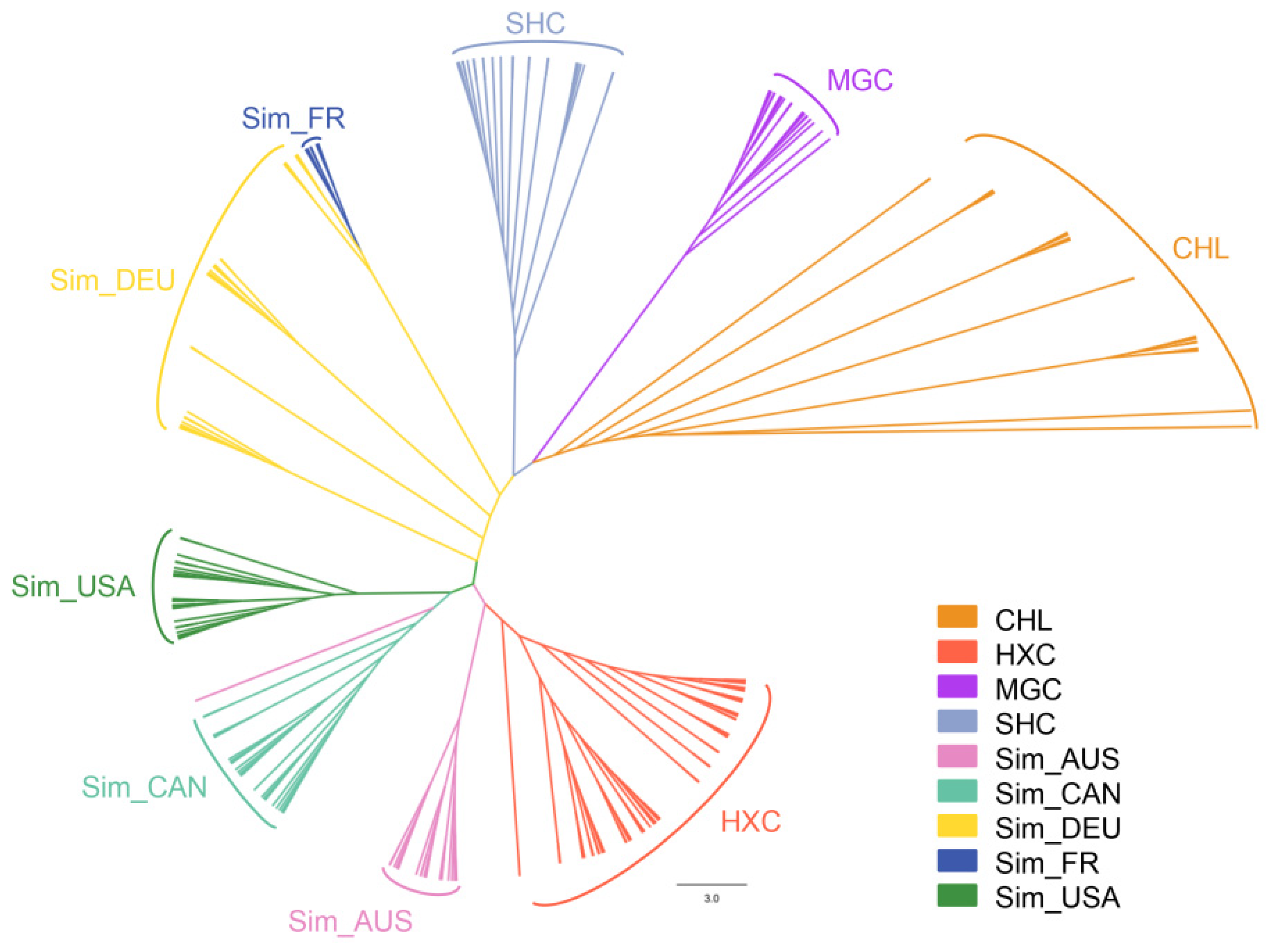
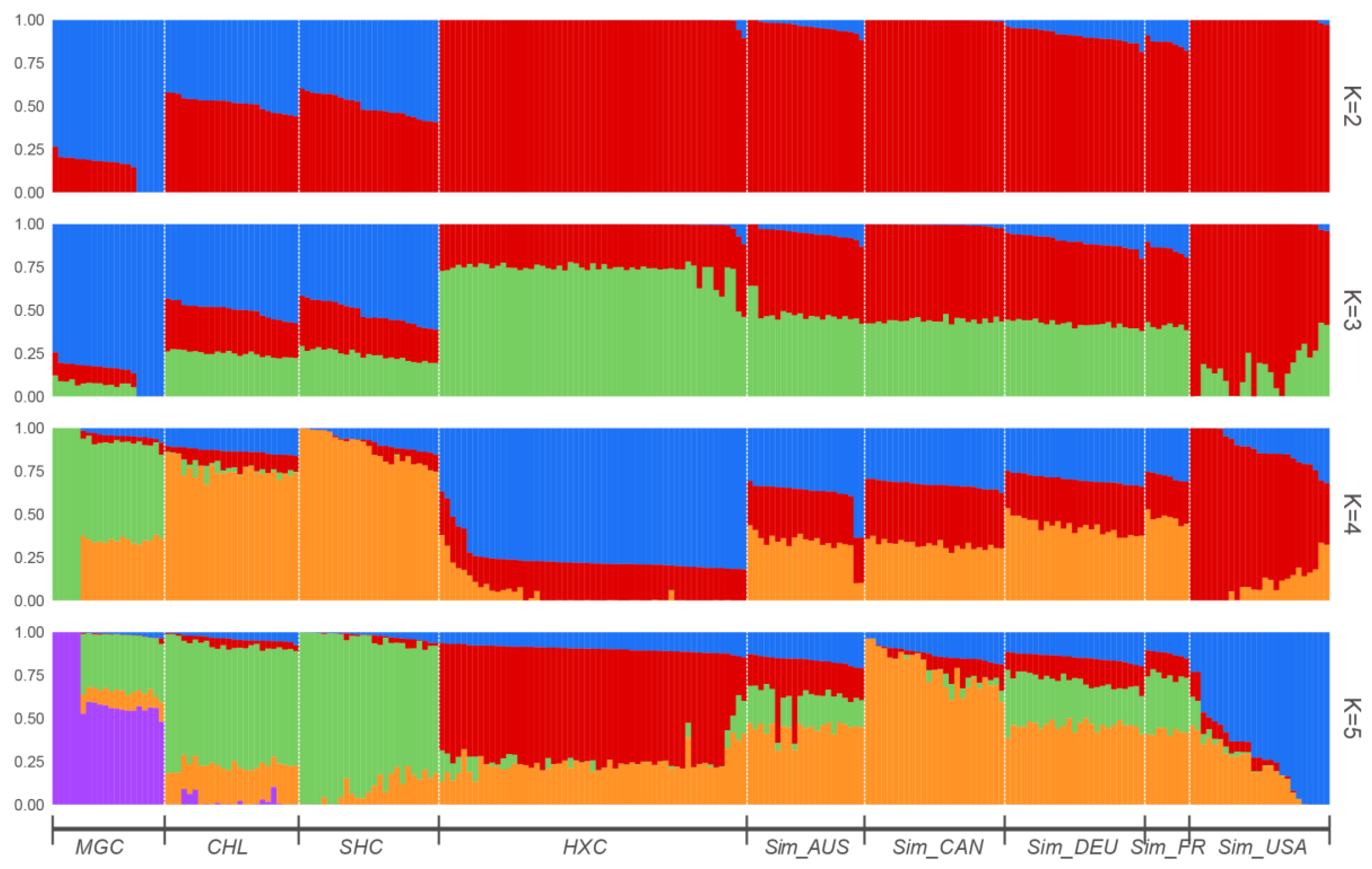
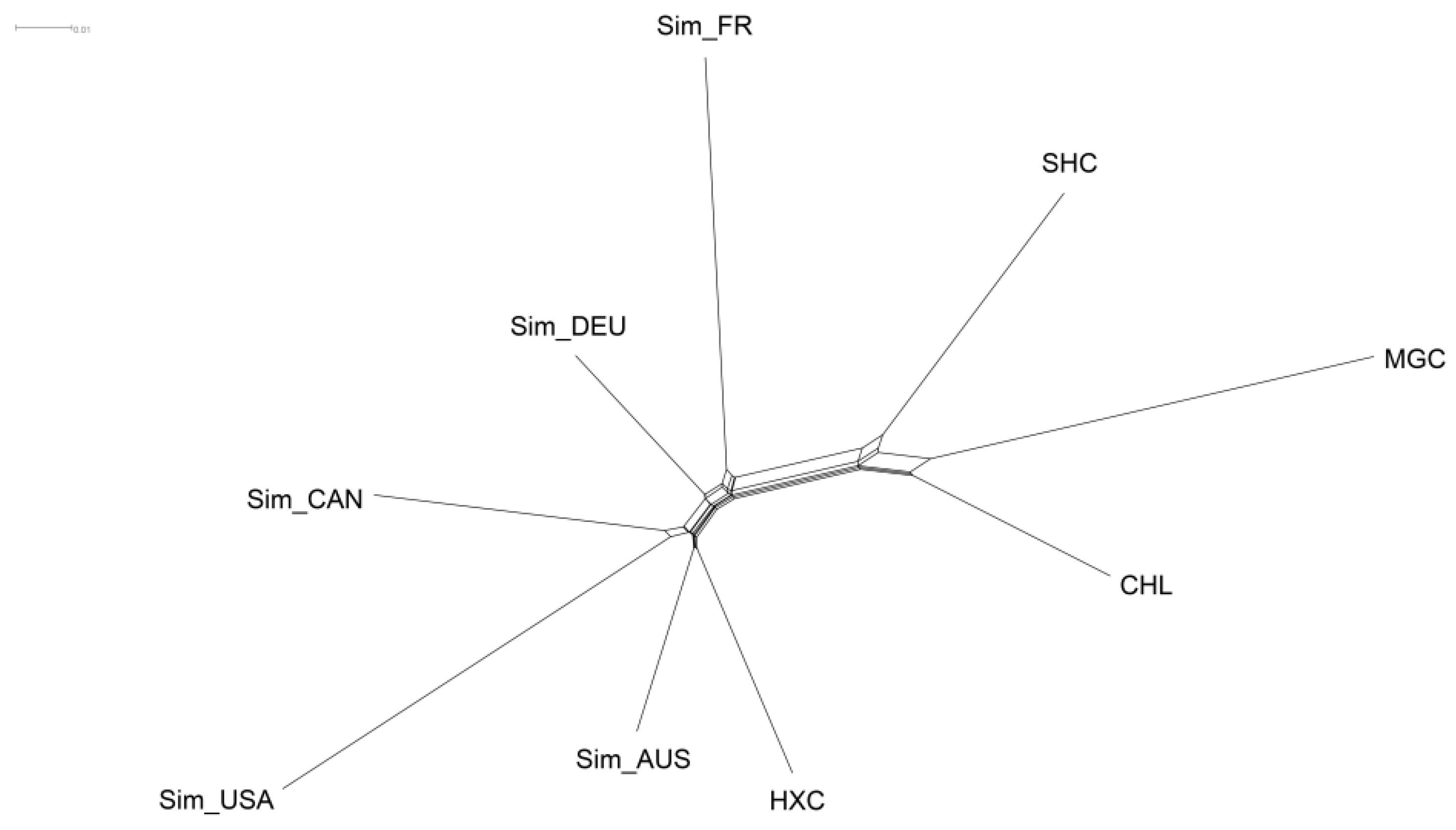
3.3. Population Structure, Admixture, and Phylogenetic Analysis
3.4. Population Genetic Distance Measure
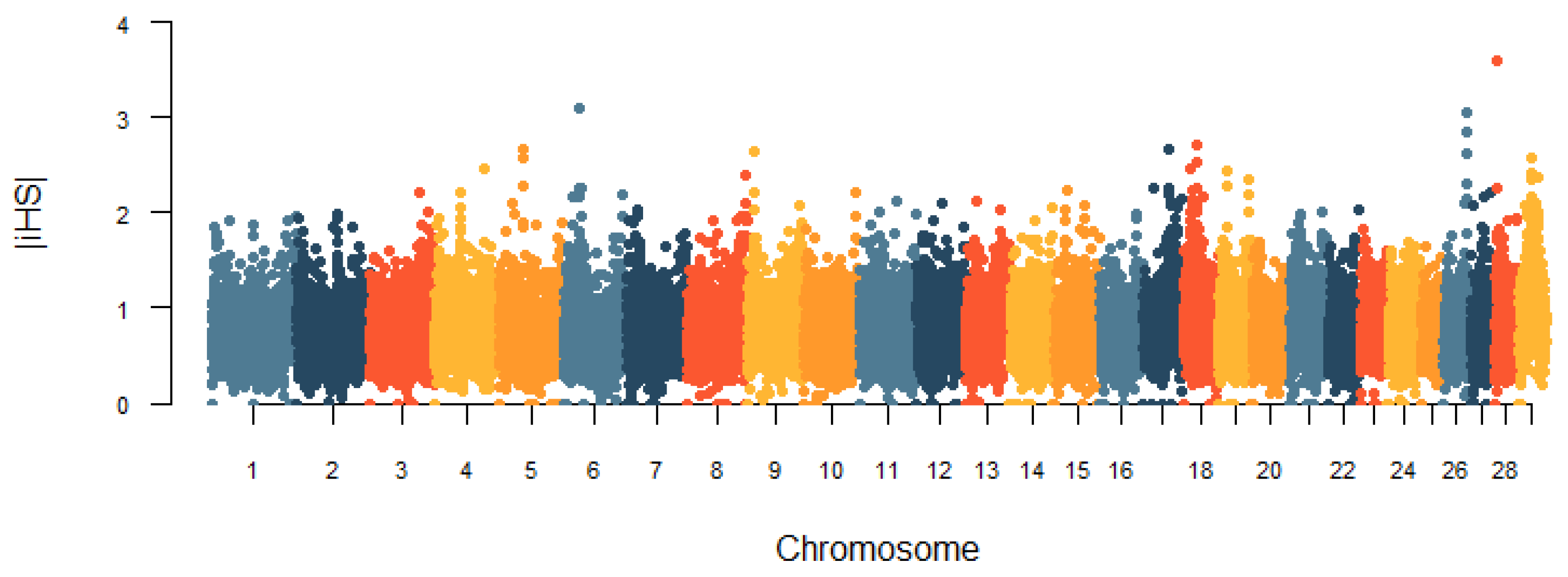
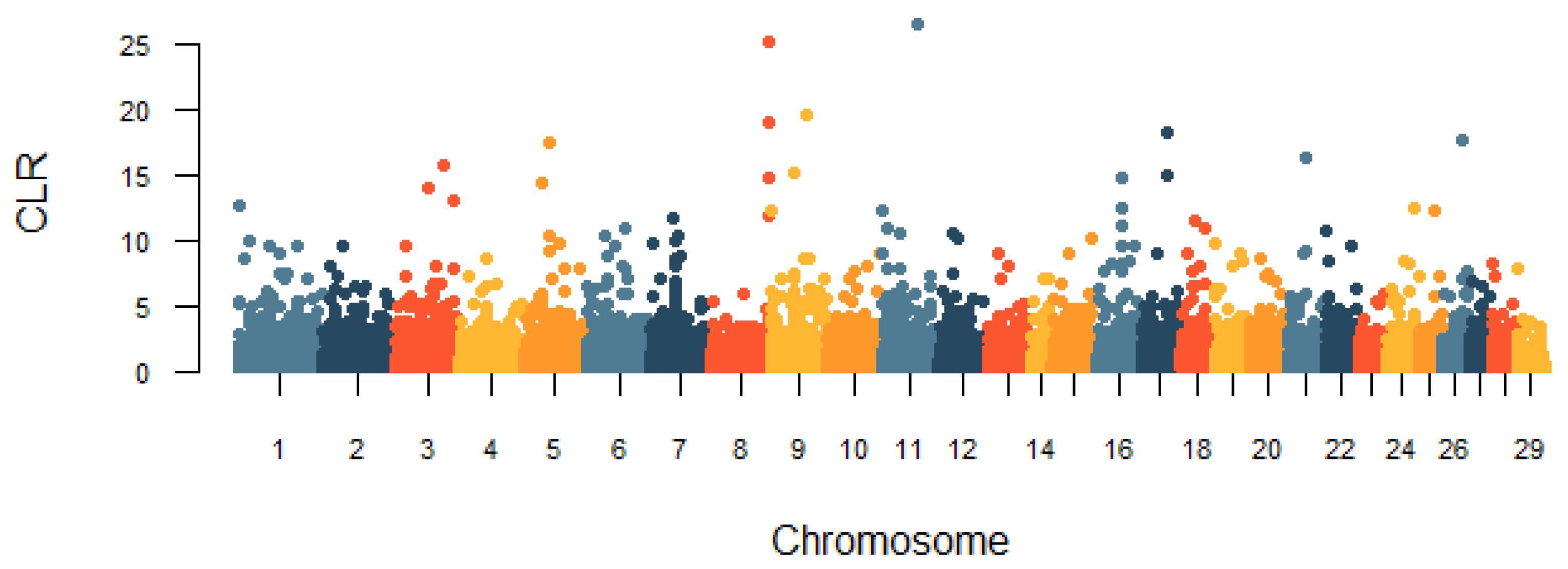
3.5. Identification of Selection Signatures
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Meng, X.; Gao, Z.; Liang, Y.; Zhang, C.; Chen, Z.; Mao, Y.; Huang, B.; Kui, K.; Yang, Z. Longissimus Dorsi Muscle Transcriptomic Analysis of Simmental and Chinese Native Cattle Differing in Meat Quality. Front. Vet. Sci. 2020, 7, 601064. [Google Scholar] [CrossRef]
- Paim, T.D.P.; Hay, E.H.A.; Wilson, C.; Thomas, M.G.; Kuehn, L.A.; Paiva, S.R.; McManus, C.; Blackburn, H.D. Dynamics of genomic architecture during composite breed development in cattle. Anim. Genet. 2020, 51, 224–234. [Google Scholar] [CrossRef] [Green Version]
- Frankham, R. Genetic rescue of small inbred populations: Meta-analysis reveals large and consistent benefits of gene flow. Mol. Ecol. 2015, 24, 2610–2618. [Google Scholar] [CrossRef]
- van der Nest, M.A.; Hlongwane, N.; Hadebe, K.; Chan, W.-Y.; van der Merwe, N.A.; De Vos, L.; Greyling, B.; Kooverjee, B.B.; Soma, P.; Dzomba, E.F.; et al. Breed Ancestry, Divergence, Admixture, and Selection Patterns of the Simbra Crossbreed. Front. Genet. 2021, 11, 608650. [Google Scholar] [CrossRef]
- Duan, X.; An, B.; Du, L.; Chang, T.; Liang, M.; Yang, B.G.; Xu, L.; Zhang, L.; Li, J.; Gao, H. Genome-Wide Association Analysis of Growth Curve Parameters in Chinese Simmental Beef Cattle. Animals 2021, 11, 192. [Google Scholar] [CrossRef]
- Zhuang, Z.; Xu, L.; Yang, J.; Gao, H.; Zhang, L.; Gao, X.; Li, J.; Zhu, B. Gao Weighted Single-Step Genome-Wide Association Study for Growth Traits in Chinese Simmental Beef Cattle. Genes 2020, 11, 189. [Google Scholar] [CrossRef] [Green Version]
- Song, Y.; Xu, L.; Chen, Y.; Zhang, L.; Gao, H.; Zhu, B.; Niu, H.; Zhang, W.; Xia, J.; Gao, X.; et al. Genome-Wide Association Study Reveals the PLAG1 Gene for Knuckle, Biceps and Shank Weight in Simmental Beef Cattle. PLoS ONE 2016, 11, e0168316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, T.; Xia, J.; Xu, L.; Wang, X.; Zhu, B.; Zhang, L.; Gao, X.; Chen, Y.; Li, J.; Gao, H. A genome-wide association study suggests several novel candidate genes for carcass traits in Chinese Simmental beef cattle. Anim. Genet. 2018, 49, 312–316. [Google Scholar] [CrossRef]
- Xia, J.; Qi, X.; Wu, Y.; Zhu, B.; Xu, L.; Zhang, L.; Gao, X.; Chen, Y.; Li, J.; Gao, H. Genome-wide association study identifies loci and candidate genes for meat quality traits in Simmental beef cattle. Mamm. Genome 2016, 27, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.; Niu, H.; Zhang, W.; Wang, Z.; Liang, Y.; Guan, L.; Guo, P.; Chen, Y.; Zhang, L.; Guo, Y.; et al. Genome wide association study and genomic prediction for fatty acid composition in Chinese Simmental beef cattle using high density SNP array. BMC Genom. 2017, 18, 464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Xu, L.; Wang, Z.; Xu, L.; Chen, Y.; Zhang, L.; Xu, L.; Gao, X.; Gao, H.; Zhu, B.; et al. Genomic Prediction and Association Analysis with Models Including Dominance Effects for Important Traits in Chinese Simmental Beef Cattle. Animals 2019, 9, 1055. [Google Scholar] [CrossRef] [Green Version]
- Guo, P.; Zhu, B.; Niu, H.; Wang, Z.; Liang, Y.; Chen, Y.; Zhang, L.; Ni, H.; Guo, Y.; Hay, E.H.A.; et al. Fast genomic prediction of breeding values using parallel Markov chain Monte Carlo with convergence diagnosis. BMC Bioinform. 2018, 19, 3. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Zhu, B.; Xu, L.; Wang, Z.; Xu, L.; Zhou, P.; Gao, H.; Guo, P.; Chen, Y.; Gao, X.; et al. Genomic Prediction Using LD-Based Haplotypes Inferred From High-Density Chip and Imputed Sequence Variants in Chinese Simmental Beef Cattle. Front. Genet. 2021, 12, 665382. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Miao, J.; Chang, T.; Xia, J.; An, B.; Li, Y.; Xu, L.; Zhang, L.; Gao, X.; Li, J.; et al. Evaluation of GBLUP, BayesB and elastic net for genomic prediction in Chinese Simmental beef cattle. PLoS ONE 2019, 14, e0210442. [Google Scholar] [CrossRef]
- An, B.; Liang, M.; Chang, T.; Duan, X.; Du, L.; Xu, L.; Zhang, L.; Gao, X.; Li, J.; Gao, H. KCRR: A nonlinear machine learning with a modified genomic similarity matrix improved the genomic prediction efficiency. Brief. Bioinform. 2021, 22, bbab132. [Google Scholar] [CrossRef]
- Liang, M.; Chang, T.; An, B.; Duan, X.; Du, L.; Wang, X.; Miao, J.; Xu, L.; Gao, X.; Zhang, L.; et al. A Stacking Ensemble Learning Framework for Genomic Prediction. Front. Genet. 2021, 12, 600040. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Sonstegard, T.S.; Cole, J.B.; Van Tassell, C.P.; Wiggans, G.R.; Crooker, B.A.; Tan, C.; Prakapenka, D.; Liu, G.E.; Da, Y. Genome changes due to artificial selection in U.S. Holstein cattle. BMC Genom. 2019, 20, 1–14. [Google Scholar] [CrossRef]
- Fan, H.; Wu, Y.; Qi, X.; Zhang, J.; Li, J.; Gao, X.; Zhang, L.; Li, J.; Gao, H. Genome-wide detection of selective signatures in Simmental cattle. J. Appl. Genet. 2014, 55, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, G.; Barria, A.; Correa, K.; Cáceres, G.; Jedlicki, A.; Cadiz, M.I.; Lhorente, J.P.; Yáñez, J.M. Genome-Wide Patterns of Population Structure and Linkage Disequilibrium in Farmed Nile Tilapia (Oreochromis niloticus). Front. Genet. 2019, 10, 745. [Google Scholar] [CrossRef] [Green Version]
- Lu, D.; Sargolzaei, M.; Kelly, M.; Li, C.; Voort, G.V.; Wang, Z.; Plastow, G.; Moore, S.; Miller, S.P. Linkage disequilibrium in Angus, Charolais, and Crossbred beef cattle. Front. Genet. 2012, 3, 152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, W.; Dekkers, J.C.; Lee, W.R.; Abasht, B. Linkage disequilibrium in crossbred and pure line chickens. Genet. Sel. Evol. 2015, 47, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ai, H.; Huang, L.; Ren, J. Genetic Diversity, Linkage Disequilibrium and Selection Signatures in Chinese and Western Pigs Revealed by Genome-Wide SNP Markers. PLoS ONE 2013, 8, e56001. [Google Scholar] [CrossRef] [Green Version]
- Ma, H.; Wang, S.; Zeng, G.; Guo, J.; Guo, M.; Dong, X.; Hua, G.; Liu, Y.; Wang, M.; Ling, Y.; et al. The Origin of a Coastal Indigenous Horse Breed in China Revealed by Genome-Wide SNP Data. Genes 2019, 10, 241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, L.; Yang, L.; Zhu, B.; Zhang, W.; Wang, Z.; Chen, Y.; Zhang, L.; Gao, X.; Gao, H.; Liu, G.E.; et al. Genome-wide scan reveals genetic divergence and diverse adaptive selection in Chinese local cattle. BMC Genom. 2019, 20, 494. [Google Scholar] [CrossRef] [Green Version]
- Khayatzadeh, N.; Mészáros, G.; Utsunomiya, Y.T.; Garcia, J.F.; Schnyder, U.; Gredler, B.; Curik, I.; Sölkner, J. Locus-specific ancestry to detect recent response to selection in admixed Swiss Fleckvieh cattle. Anim. Genet. 2016, 47, 637–646. [Google Scholar] [CrossRef]
- Singh, A.; Mehrotra, A.; Gondro, C.; Romero, A.R.D.S.; Pandey, A.K.; Karthikeyan, A.; Bashir, A.; Mishra, B.P.; Dutt, T.; Kumar, A. Signatures of Selection in Composite Vrindavani Cattle of India. Front. Genet. 2020, 11, 589496. [Google Scholar] [CrossRef] [PubMed]
- Bahbahani, H.; Clifford, H.; Wragg, D.; Mbole-Kariuki, M.N.; Van Tassell, C.; Sonstegard, T.; Woolhouse, M.; Hanotte, O. Signatures of positive selection in East African Shorthorn Zebu: A genome-wide single nucleotide polymorphism analysis. Sci. Rep. 2015, 5, 11729. [Google Scholar] [CrossRef] [Green Version]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [Green Version]
- McQuillan, R.; Leutenegger, A.-L.; Abdel-Rahman, R.; Franklin, C.S.; Pericic, M.; Barac-Lauc, L.; Smolej-Narancic, N.; Janicijevic, B.; Polasek, O.; Tenesa, A.; et al. Runs of Homozygosity in European Populations. Am. J. Hum. Genet. 2008, 83, 359–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, L.; Zhao, G.; Yang, L.; Zhu, B.; Chen, Y.; Zhang, L.; Gao, X.; Gao, H.; Liu, G.E.; Li, J. Genomic Patterns of Homozygosity in Chinese Local Cattle. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Dong, S.-S.; Xu, J.-Y.; He, W.-M.; Yang, T.-L. PopLDdecay: A fast and effective tool for linkage disequilibrium decay analysis based on variant call format files. Bioinformatics 2019, 35, 1786–1788. [Google Scholar] [CrossRef]
- Baum, B.R. PHYLIP: Phylogeny Inference Package. Version 3.2. Joel Felsenstein. Q. Rev. Biol. 1989, 64, 539–541. [Google Scholar] [CrossRef]
- Xu, L.; Bickhart, D.M.; Cole, J.B.; Schroeder, S.G.; Song, J.; Van Tassell, C.P.; Sonstegard, T.S.; Liu, G.E. Genomic Signatures Reveal New Evidences for Selection of Important Traits in Domestic Cattle. Mol. Biol. Evol. 2014, 32, 711–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jombart, T.; Ahmed, I. adegenet 1.3-1: New tools for the analysis of genome-wide SNP data. Bioinformatics 2011, 27, 3070–3071. [Google Scholar] [CrossRef] [Green Version]
- Huson, D.H.; Bryant, D. Application of Phylogenetic Networks in Evolutionary Studies. Mol. Biol. Evol. 2005, 23, 254–267. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar] [CrossRef]
- Msalya, G.; Kim, E.-S.; Laisser, E.; Kipanyula, M.J.; Karimuribo, E.D.; Kusiluka, L.J.M.; Chenyambuga, S.W.; Rothschild, M.F. Determination of Genetic Structure and Signatures of Selection in Three Strains of Tanzania Shorthorn Zebu, Boran and Friesian Cattle by Genome-Wide SNP Analyses. PLoS ONE 2017, 12, e0171088. [Google Scholar] [CrossRef] [Green Version]
- Voight, B.F.; Kudaravalli, S.; Wen, X.; Pritchard, J.K. A Map of Recent Positive Selection in the Human Genome. PLoS Biol. 2006, 4, e72. [Google Scholar] [CrossRef] [Green Version]
- Williamson, S.H.; Hubisz, M.J.; Clark, A.G.; Payseur, B.A.; Bustamante, C.D.; Nielsen, R. Localizing Recent Adaptive Evolution in the Human Genome. PLoS Genet. 2007, 3, e90. [Google Scholar] [CrossRef]
- Browning, S.; Browning, B. Rapid and Accurate Haplotype Phasing and Missing-Data Inference for Whole-Genome Association Studies By Use of Localized Haplotype Clustering. Am. J. Hum. Genet. 2007, 81, 1084–1097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szpiech, Z.A.; Hernandez, R.D. selscan: An Efficient Multithreaded Program to Perform EHH-Based Scans for Positive Selection. Mol. Biol. Evol. 2014, 31, 2824–2827. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Xu, L.; Yang, L.; Zhao, G.; Li, J.; Liu, D.; Li, Y. Discovery of Genomic Characteristics and Selection Signatures in Southern Chinese Local Cattle. Front. Genet. 2020, 11, 533052. [Google Scholar] [CrossRef]
- Pavlidis, P.; Živković, D.; Stamatakis, A.; Alachiotis, N. SweeD: Likelihood-Based Detection of Selective Sweeps in Thousands of Genomes. Mol. Biol. Evol. 2013, 30, 2224–2234. [Google Scholar] [CrossRef] [Green Version]
- Pickrell, J.K.; Coop, G.; Novembre, J.; Kudaravalli, S.; Li, J.Z.; Absher, D.; Srinivasan, B.S.; Barsh, G.S.; Myers, R.M.; Feldman, M.W.; et al. Signals of recent positive selection in a worldwide sample of human populations. Genome Res. 2009, 19, 826–837. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; Wang, J.; Wang, Y.; Wu, Y.; Fu, J.; Liu, J.-F. Genome-wide detection of selection signatures in Chinese indigenous Laiwu pigs revealed candidate genes regulating fat deposition in muscle. BMC Genet. 2018, 19, 31. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Hao, P.; Yu, Y.; Feng, W.; Du, H.; Yu, J.; Kang, H.; Zheng, X.; Wang, Z.; Liu, G.E.; Ernst, C.W.; et al. Evidence of evolutionary history and selective sweeps in the genome of Meishan pig reveals its genetic and phenotypic characterization. GigaScience 2018, 7, giy058. [Google Scholar] [CrossRef]
- O’brien, A.M.P.; Utsunomiya, Y.T.; Mészáros, G.; Bickhart, D.M.; Liu, G.E.; Van Tassell, C.P.; Sonstegard, T.S.; Da Silva, M.V.; Garcia, J.F.; Sölkner, J. Assessing signatures of selection through variation in linkage disequilibrium between taurine and indicine cattle. Genet. Sel. Evol. 2014, 46, 19. [Google Scholar] [CrossRef] [Green Version]
- Jia, X.; Ding, P.; Chen, S.; Zhao, S.; Wang, J.; Lai, S. Analysis of MC1R, MITF, TYR, TYRP1, and MLPH Genes Polymorphism in Four Rabbit Breeds with Different Coat Colors. Animals 2021, 11, 81. [Google Scholar] [CrossRef]
- Zhang, Q.; Calus, M.P.L.; Guldbrandtsen, B.; Lund, M.S.; Sahana, G. Estimation of inbreeding using pedigree, 50k SNP chip genotypes and full sequence data in three cattle breeds. BMC Genet. 2015, 16, 88. [Google Scholar] [CrossRef] [Green Version]
- Maiorano, A.M.; Lourenco, D.L.; Tsuruta, S.; Ospina, A.M.; Stafuzza, N.B.; Masuda, Y.; Filho, A.E.; Cyrillo, J.N.; Curi, R.A.; Silva, J.A. Assessing genetic architecture and signatures of selection of dual purpose Gir cattle populations using genomic information. PLoS ONE 2018, 13, e0200694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakoev, S.; Kolosov, A.; Bakoev, F.; Kostyunina, O.; Bakoev, N.; Romanets, T.; Koshkina, O.; Getmantseva, L. Analysis of Homozygous-by-Descent (HBD) Segments for Purebred and Crossbred Pigs in Russia. Life 2021, 11, 861. [Google Scholar] [CrossRef] [PubMed]
- Lan, D.; Xiong, X.; Mipam, T.-D.; Fu, C.; Li, Q.; Ai, Y.; Hou, D.; Chai, Z.; Zhong, J.; Li, J. Genetic Diversity, Molecular Phylogeny, and Selection Evidence of Jinchuan Yak Revealed by Whole-Genome Resequencing. G3 Genes Genomes Genet. 2018, 8, 945–952. [Google Scholar] [CrossRef] [Green Version]
- Biegelmeyer, P.; Gulias-Gomes, C.C.; Caetano, A.R.; Steibel, J.P.; Cardoso, F.F. Linkage disequilibrium, persistence of phase and effective population size estimates in Hereford and Braford cattle. BMC Genet. 2016, 17, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boschiero, C.; Moreira, G.C.M.; Gheyas, A.A.; Godoy, T.F.; Gasparin, G.; Mariani, P.D.S.C.; Paduan, M.; Cesar, A.S.M.; Ledur, M.C.; Coutinho, L. Genome-wide characterization of genetic variants and putative regions under selection in meat and egg-type chicken lines. BMC Genom. 2018, 19, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, Z.-H.; Pausch, H. Multi-trait meta-analyses reveal 25 quantitative trait loci for economically important traits in Brown Swiss cattle. BMC Genom. 2019, 20, 695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guan, D.; Luo, N.; Tan, X.; Zhao, Z.; Huang, Y.; Na, R.; Zhang, J.; Zhao, Y. Scanning of selection signature provides a glimpse into important economic traits in goats (Capra hircus). Sci. Rep. 2016, 6, 36372. [Google Scholar] [CrossRef] [Green Version]
- . López, M.-E.; Cádiz, M.I.; Rondeau, E.B.; Koop, B.F.; Yáñez, J.M. Detection of selection signatures in farmed coho salmon (Oncorhynchus kisutch) using dense genome-wide information. Sci. Rep. 2021, 11, 1–13. [Google Scholar] [CrossRef]
- Sabeti, P.C.; He, Y.; Varilly, P.; Fry, B.; Lohmueller, J.; Hostetter, E.; Cotsapas, C.; Xie, X.; Byrne, E.H.; McCarroll, S.A.; et al. Genome-wide detection and characterization of positive selection in human populations. Nature 2007, 449, 913–918. [Google Scholar] [CrossRef]
- Distefano, M.B.; Haugen, L.H.; Wang, Y.; Perdreau-Dahl, H.; Kjos, I.; Jia, D.; Morth, J.P.; Neefjes, J.; Bakke, O.; Progida, C. TBC1D5 controls the GTPase cycle of Rab7b. J. Cell Sci. 2018, 131, jcs.216630. [Google Scholar] [CrossRef] [Green Version]
- . Liu, Y.; Zan, L.; Cui, W.; Xin, Y.; Jiao, Y.; Li, K. Molecular cloning, characterization and association analysis of the promoter region of the bovine CDK6 gene. Genet. Mol. Res. 2011, 10, 1777–1786. [Google Scholar] [CrossRef]
- Paredes-Sánchez, F.A.; Sifuentes-Rincón, A.M.; Cabrera, A.S.; Pérez, C.A.G.; Bracamonte, G.M.P.; Morales, P.A. Associations of SNPs located at candidate genes to bovine growth traits, prioritized with an interaction networks construction approach. BMC Genet. 2015, 16, 91. [Google Scholar] [CrossRef] [Green Version]
- Goszczynski, D.E.; Mazzucco, J.P.; Ripoli, M.V.; Villarreal, E.L.; Rogberg-Muñoz, A.; Mezzadra, C.A.; Melucci, L.M.; Giovambattista, G. Genetic characterisation of PPARG, CEBPA and RXRA, and their influence on meat quality traits in cattle. J. Anim. Sci. Technol. 2016, 58, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, R.; Li, X. Association between IGF-IR, m-calpain and UCP-3 gene polymorphisms and growth traits in Nanyang cattle. Mol. Biol. Rep. 2010, 38, 2179–2184. [Google Scholar] [CrossRef]
- Pryce, J.E.; Hayes, B.; Bolormaa, S.; Goddard, M. Polymorphic Regions Affecting Human Height Also Control Stature in Cattle. Genetics 2011, 187, 981–984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindholm-Perry, A.K.; Sexten, A.K.; Kuehn, L.A.; Smith, T.P.; King, D.A.; Shackelford, S.D.; Wheeler, T.L.; Ferrell, C.L.; Jenkins, T.G.; Snelling, W.M.; et al. Association, effects and validation of polymorphisms within the NCAPG-LCORL locus located on BTA6 with feed intake, gain, meat and carcass traits in beef cattle. BMC Genet. 2011, 12, 103. [Google Scholar] [CrossRef] [Green Version]
- Xia, J.; Fan, H.; Chang, T.; Xu, L.; Zhang, W.; Song, Y.; Zhu, B.; Zhang, L.; Gao, X.; Chen, Y.; et al. Searching for new loci and candidate genes for economically important traits through gene-based association analysis of Simmental cattle. Sci. Rep. 2017, 7, 42048. [Google Scholar] [CrossRef] [Green Version]
- Buitenhuis, A.; Lund, M.; Thomasen, J.; Thomsen, B.; Nielsen, V.H.; Bendixen, C.; Guldbrandtsen, B. Detection of Quantitative Trait Loci Affecting Lameness and Leg Conformation Traits in Danish Holstein Cattle. J. Dairy Sci. 2007, 90, 472–481. [Google Scholar] [CrossRef] [Green Version]
- Bedhane, M.; Van Der Werf, J.; Gondro, C.; Duijvesteijn, N.; Lim, D.; Park, B.; Na Park, M.; Hee, R.S.; Clark, S. Genome-Wide Association Study of Meat Quality Traits in Hanwoo Beef Cattle Using Imputed Whole-Genome Sequence Data. Front. Genet. 2019, 10, 1235. [Google Scholar] [CrossRef]
- Kiser, J.N.; Lawrence, T.E.; Neupane, M.; Seabury, C.M.; Taylor, J.F.; Womack, J.E.; Neibergs, H.L. Rapid Communication: Subclinical bovine respiratory disease-loci and pathogens associated with lung lesions in feedlot cattle. J. Anim. Sci. 2017, 95, 2726–2731. [Google Scholar] [CrossRef]
- Dear, T.; Boehm, T. Identification and characterization of two novel calpain large subunit genes. Gene 2001, 274, 245–252. [Google Scholar] [CrossRef]
- Curran, J.; Makara, M.A.; Little, S.C.; Musa, H.; Liu, B.; Wu, X.; Polina, I.; Alecusan, J.S.; Wright, P.; Li, J.; et al. EHD3-Dependent Endosome Pathway Regulates Cardiac Membrane Excitability and Physiology. Circ. Res. 2014, 115, 68–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Çetinkaya, A.; Taşkıran, E.; Soyer, T.; Şimşek-Kiper, P.; Özlem; Utine, G.E.; Tunçbilek, G.; Boduroğlu, K.; Alikaşifoğlu, M. Dermal fibroblast transcriptome indicates contribution of wnt signaling pathways in the pathogenesis of apert syndrome. Turk. J. Pediatr. 2017, 59, 619. [Google Scholar] [CrossRef] [PubMed]
- Sabri, A.; Lai, D.; D’Silva, A.; Seeho, S.; Kaur, J.; Ng, C.; Hyett, J. Differential placental gene expression in term pregnancies affected by fetal growth restriction and macrosomia. Fetal Diagn. Ther. 2014, 36, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Kang, H.; Liu, H.; Wang, J.; Guo, Q.; Song, C.; Sun, Y.; Zhang, Y.; Zhang, H.; Zhang, Z.; et al. Myoferlin, a Membrane Protein with Emerging Oncogenic Roles. BioMed Res. Int. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Yasumizu, Y.; Sakaue, S.; Konuma, T.; Suzuki, K.; Matsuda, K.; Murakami, Y.; Kubo, M.; Palamara, P.F.; Kamatani, Y.; Okada, Y. Genome-Wide Natural Selection Signatures Are Linked to Genetic Risk of Modern Phenotypes in the Japanese Population. Mol. Biol. Evol. 2020, 37, 1306–1316. [Google Scholar] [CrossRef]
- Huang, X.; Qu, R.; Ouyang, J.; Zhong, S.; Dai, J. An Overview of the Cytoskeleton-Associated Role of PDLIM5. Front. Physiol. 2020, 11, 975. [Google Scholar] [CrossRef]
- Sajan, S.A.; Ganesh, J.; Shinde, D.N.; Powis, Z.; Scarano, M.I.; Stone, J.; Winter, S.; Tang, S. Biallelic disruption of PKDCC is associated with a skeletal disorder characterised by rhizomelic shortening of extremities and dysmorphic features. J. Med Genet. 2019, 56, 850–854. [Google Scholar] [CrossRef]
- Kinoshita, M.; Era, T.; Jakt, L.M.; Nishikawa, S.-I. The novel protein kinase Vlk is essential for stromal function of mesenchymal cells. Development 2009, 136, 2069–2079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chatelain, F.C.; Bichet, D.; Douguet, D.; Feliciangeli, S.; Bendahhou, S.; Reichold, M.; Warth, R.; Barhanin, J.; Lesage, F. TWIK1, a unique background channel with variable ion selectivity. Proc. Natl. Acad. Sci. USA 2012, 109, 5499–5504. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Li, H.; Huo, Q.; Cui, M.; Ge, C.; Zhao, F.; Tian, H.; Chen, T.; Yao, M.; Li, J. The transcription factor FOXN3 inhibits cell proliferation by downregulating E2F5 expression in hepatocellular carcinoma cells. Oncotarget 2016, 7, 43534–43545. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Population | Abbreviation | Number | MAF 1 | Ho 2 | He 3 | FROH 4 |
|---|---|---|---|---|---|---|
| Mongolian cattle | MGC | 20 | 0.289 | 0.366 | 0.377 | 0.049 |
| Sanhe cattle | SHC | 25 | 0.280 | 0.378 | 0.366 | 0.052 |
| Charolais | CHL | 24 | 0.290 | 0.388 | 0.377 | 0.034 |
| Australian Simmental cattle | Sim_AUS | 21 | 0.267 | 0.365 | 0.352 | 0.071 |
| Canadian Simmental cattle | Sim_CAN | 25 | 0.252 | 0.357 | 0.334 | 0.094 |
| American Simmental cattle | Sim_USA | 21 | 0.235 | 0.334 | 0.313 | 0.150 |
| Fleevicht cattle | Sim_DEU | 25 | 0.273 | 0.384 | 0.358 | 0.053 |
| Montbeliard cattle | Sim_FR | 8 | 0.261 | 0.387 | 0.342 | 0.046 |
| Huaxi cattle | HXC | 55 | 0.256 | 0.375 | 0.340 | 0.053 |
| Trait Class | Trait | Gene Detected by iHS | Gene Detected by CLR |
|---|---|---|---|
| Growth and development | Average daily feed intake | LCORL | |
| Body length | CDK6 | CAPN2 | |
| Body weight | RXRA, TBC1D5 | CAPN2 | |
| Carcass and meat quality | Carcass weight | ZNF280B | CA10, LCORL |
| Bone weight | LAP3, LCORL | ||
| Bone quality | POLB | ||
| Marbling score | HELB, IRAK3 | ||
| Longissimus muscle area | LCORL | ||
| Fat thickness at the 12th rib | RXRA | LCORL | |
| Monounsaturated fatty acid content | RXRA | ||
| Meat texture | ANO5 | ||
| Reproduction | Conception rate | DZIP3 | |
| Daughter pregnancy rate | AMN1, COQ9, KCNMB2, CACNA2D3 | ||
| Early embryonic survival | SLC18A2 | ||
| Milk | Milk yield | NCKAP1L | ABCA7, DNAJC21, IL20RA |
| Milk fat yield | TBX5, CNOT1, NDRG4, NCKAP1L, CNOT1 | AMN1, NELL2, PCED1B | |
| Milk protein yield | GRIN3A, NDRG4, VPS35 | DZIP3, IL12RB2, LAP3, MED28, SPSB1, CACNA2D3 | |
| Milking speed | SLC18A2 | ||
| Milk C14 index | ANO5 | ||
| Milk-conjugated linoleic acid content | ASIC2 | ||
| Milk cholesterol content | RBM19 | ||
| Health | Bovine respiratory disease susceptibility | KDR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, J.; Gao, X.; Li, J.; Gao, H.; Wang, Z.; Zhang, L.; Xu, L.; Gao, H.; Li, H.; Wang, Y.; et al. Assessing the Genetic Background and Selection Signatures of Huaxi Cattle Using High-Density SNP Array. Animals 2021, 11, 3469. https://doi.org/10.3390/ani11123469
Ma J, Gao X, Li J, Gao H, Wang Z, Zhang L, Xu L, Gao H, Li H, Wang Y, et al. Assessing the Genetic Background and Selection Signatures of Huaxi Cattle Using High-Density SNP Array. Animals. 2021; 11(12):3469. https://doi.org/10.3390/ani11123469
Chicago/Turabian StyleMa, Jun, Xue Gao, Junya Li, Huijiang Gao, Zezhao Wang, Lupei Zhang, Lingyang Xu, Han Gao, Hongwei Li, Yahui Wang, and et al. 2021. "Assessing the Genetic Background and Selection Signatures of Huaxi Cattle Using High-Density SNP Array" Animals 11, no. 12: 3469. https://doi.org/10.3390/ani11123469
APA StyleMa, J., Gao, X., Li, J., Gao, H., Wang, Z., Zhang, L., Xu, L., Gao, H., Li, H., Wang, Y., Zhu, B., Cai, W., Wang, C., & Chen, Y. (2021). Assessing the Genetic Background and Selection Signatures of Huaxi Cattle Using High-Density SNP Array. Animals, 11(12), 3469. https://doi.org/10.3390/ani11123469






