Impact of Heat Stress on Poultry Health and Performances, and Potential Mitigation Strategies
Simple Summary
Abstract
1. Introduction
2. Biological Changes in Poultry Due to Heat Stress
2.1. Physiological Changes
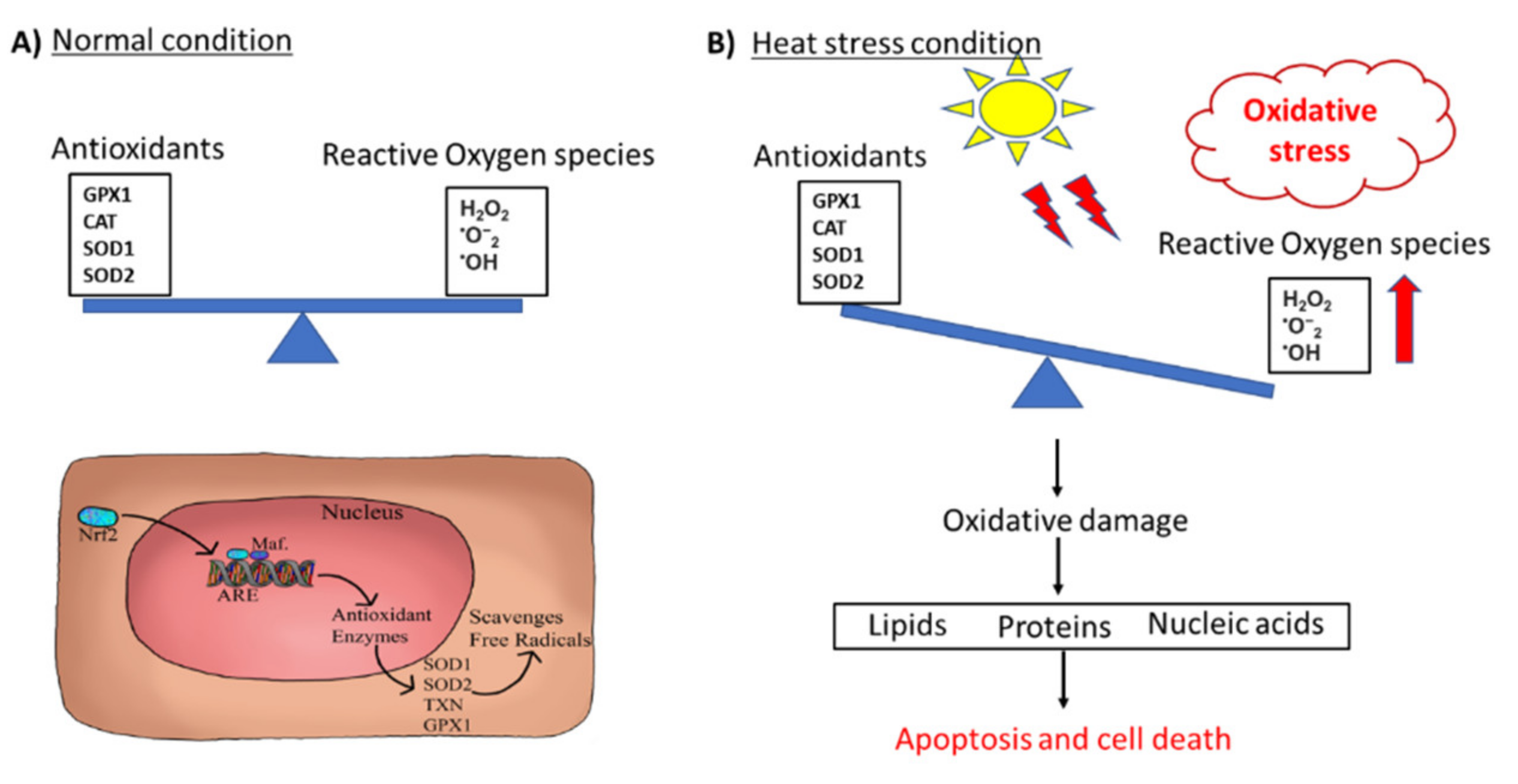
2.1.1. Oxidative Stress
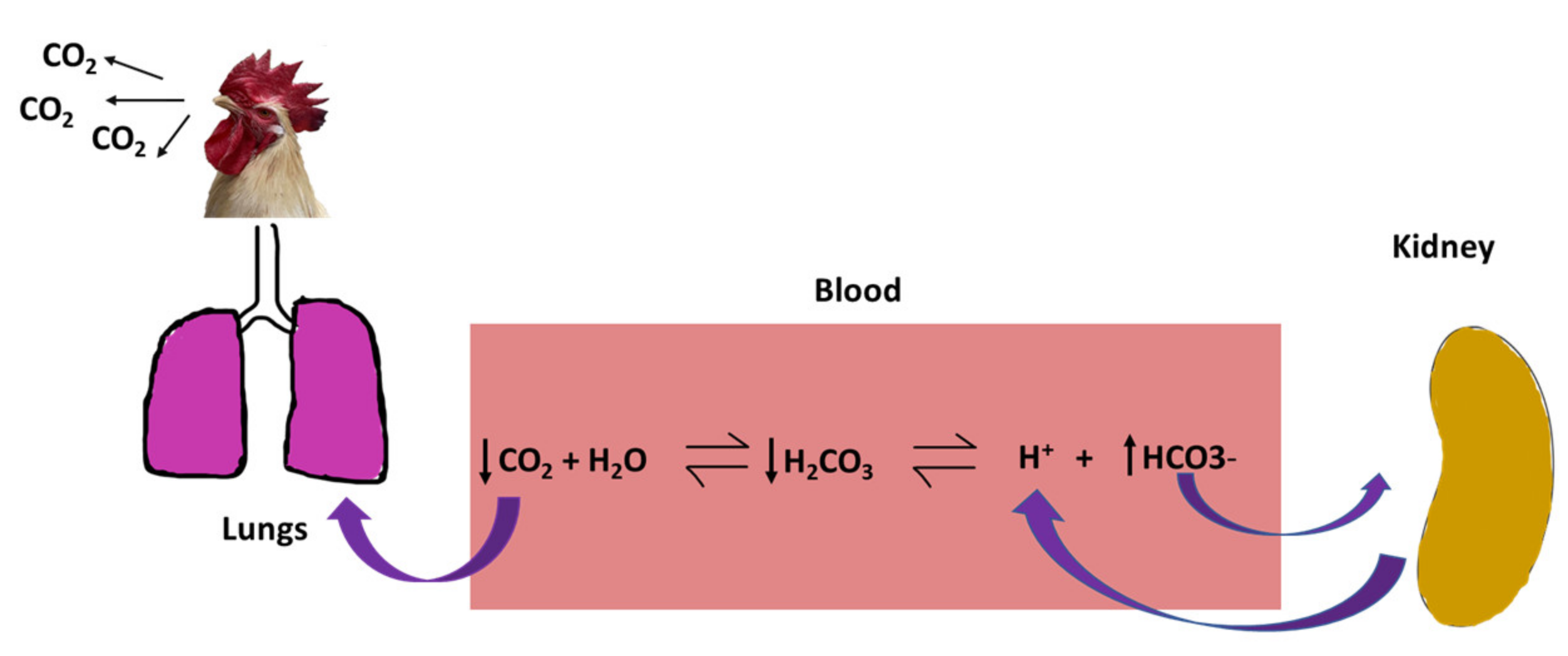
2.1.2. Acid-Base Imbalance
2.1.3. Suppressed Immunocompetence
2.2. Neuroendocrine Changes
2.3. Behavioral Changes
3. Potential Strategies to Mitigate Heat Stress in Poultry
3.1. Feeding Strategies
3.1.1. Feed Restriction
3.1.2. Dual Feeding Regime
3.1.3. Wet Feeding
3.1.4. Adding Fat in the Diet
3.1.5. Supplementation of Vitamins, Minerals, and Electrolytes
Vitamin E
Vitamin A
Vitamin C
Zinc
Chromium
Selenium
Electrolytes
3.1.6. Supplementation of Phytochemicals
Lycopene
Resveratrol
Epigallocatechin Gallate (EGCG)
Curcumin
3.1.7. Supplementation of Osmolytes
Betaine
Taurine
| Supplements | Beneficial Effects on Heat-Stressed Birds | References |
|---|---|---|
| Vitamin E |
| [62] |
| [64] | |
| Vitamin A |
| [68] |
| [69] | |
| Vitamin C |
| [72] |
| [73] | |
| [74] | |
| Zinc |
| [79] |
| [80] | |
| [81] | |
| [82,83] | |
| Chromium |
| [86] |
| [87] | |
| [88,89] | |
| [90] | |
| Selenium |
| [95] |
| [97] | |
| [98] | |
| Sodium Bicarbonate |
| [77] |
| KCL |
| [103] |
| Lycopene |
| [105] |
| [104] | |
| [104] | |
| Resveratrol |
| [107] |
| [108] | |
| [109] | |
| [110] | |
| Epigallocatechin gallate (EGCG) |
| [111] |
| [112] | |
| [113] | |
| Curcumin |
| [117] |
| [115,118] | |
| Betaine |
| [122] |
| [123] | |
| Taurine |
| [128] |
| [129] |
3.2. Genetic Approach
3.2.1. Naked Neck (Na) Gene
3.2.2. Frizzle Gene
3.2.3. Dwarf (dw) Gene
3.3. Housing
3.4. Others
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Marangoni, F.; Corsello, G.; Cricelli, C.; Ferrara, N.; Ghiselli, A.; Lucchin, L.; Poli, A. Role of poultry meat in a balanced diet aimed at maintaining health and wellbeing: An Italian consensus document. Food Nutr. Res. 2015, 59, 27606. [Google Scholar] [CrossRef] [PubMed]
- American Egg Board. Egg Protein Comparision and Costs. Available online: https://www.aeb.org/farmers-and-marketers/protein-comparison (accessed on 7 June 2020).
- Zaheer, K. An updated review on chicken eggs: Production, consumption, management aspects and nutritional benefits to human health. Food Nutr. Sci. 2015, 6, 1280. [Google Scholar] [CrossRef]
- Alexandratos, N.; Bruinsma, J. World Agriculture towards 2030/2050: The 2012 Revision; ESA Working Papar No. 12-03; FAO: Rome, Italy, 2012. [Google Scholar]
- Zuidhof, M.J.; Schneider, B.L.; Carney, V.L.; Korver, D.R.; Robinson, F.E. Growth, efficiency, and yield of commercial broilers from 1957, 1978, and 2005. Poult. Sci. 2014, 93, 2970–2982. [Google Scholar] [CrossRef] [PubMed]
- American Egg Board. History of Commercial Egg Production. Available online: https://www.aeb.org/farmers-and-marketers/history-of-egg-production (accessed on 7 June 2020).
- Tallentire, C.W.; Leinonen, I.; Kyriazakis, I. Breeding for efficiency in the broiler chicken: A review. Agron. Sustain. Dev. 2016, 36, 66. [Google Scholar] [CrossRef]
- Goo, D.; Kim, J.H.; Park, G.H.; Delos Reyes, J.B.; Kil, D.Y. Effect of Heat Stress and Stocking Density on Growth Performance, Breast Meat Quality, and Intestinal Barrier Function in Broiler Chickens. Animals 2019, 9, 107. [Google Scholar] [CrossRef]
- St-Pierre, N.R.; Cobanov, B.; Schnitkey, G. Economic Losses from Heat Stress by US Livestock Industries. J. Dairy Sci. 2003, 86, E52–E77. [Google Scholar] [CrossRef]
- Lara, L.J.; Rostagno, M.H. Impact of heat stress on poultry production. Animals 2013, 3, 356–369. [Google Scholar] [CrossRef]
- Naga Raja Kumari, K.; Narendra Nath, D. Ameliorative measures to counter heat stress in poultry. World’s Poult. Sci. J. 2018, 74, 117–130. [Google Scholar] [CrossRef]
- Donkoh, A. Ambient temperature: A factor affecting performance and physiological response of broiler chickens. Int. J. Biometeorol. 1989, 33, 259–265. [Google Scholar] [CrossRef]
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Kidd, M.T. Antioxidant Defence Systems and Oxidative Stress in Poultry Biology: An Update. Antioxidants 2019, 8, 235. [Google Scholar] [CrossRef]
- Betteridge, D.J. What is oxidative stress? Metab. Exp. 2000, 49, 3–8. [Google Scholar] [CrossRef]
- Mishra, B.; Jha, R. Oxidative stress in the poultry gut: Potential challenges and interventions. Front. Vet. Sci. 2019, 6, 60. [Google Scholar] [CrossRef]
- Estévez, M. Oxidative damage to poultry: From farm to fork. Poult. Sci. 2015, 94, 1368–1378. [Google Scholar] [CrossRef] [PubMed]
- Lennon, S.V.; Martin, S.J.; Cotter, T.G. Dose-dependent induction of apoptosis in human tumour cell lines by widely diverging stimuli. Cell Prolif. 1991, 24, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.A. Physiology of thermal panting in birds. Ann. Biol. Anim. Biochim. Biophys. 1970, 10, 151–168. [Google Scholar] [CrossRef]
- Borges, S.A.; Da Silva, A.F.; Maiorka, A. Acid-base balance in broilers. World’s Poult. Sci. J. 2007, 63, 73–81. [Google Scholar] [CrossRef]
- Badruzzaman, A.T.M.; Noor, M.; Mamun, M.A.L.; Husna, A.; Islam, K.M.; Alam, K.J.; Rahman, M.M. Prevalence of diseases in commercial chickens at Sylhet Division of Bangladesh. Int. Clin. Pathol. J. 2015, 1, 00023. [Google Scholar]
- Ghazi, S.H.; Habibian, M.; Moeini, M.M.; Abdolmohammadi, A.R. Effects of different levels of organic and inorganic chromium on growth performance and immunocompetence of broilers under heat stress. Biol. Trace Elem. Res. 2012, 146, 309–317. [Google Scholar] [CrossRef]
- Quinteiro-Filho, W.M.; Ribeiro, A.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Sakai, M.; Sá, L.R.M.D.; Ferreira, A.J.P.; Palermo-Neto, J. Heat stress impairs performance parameters, induces intestinal injury, and decreases macrophage activity in broiler chickens. Poult. Sci. 2010, 89, 1905–1914. [Google Scholar] [CrossRef]
- Bartlett, J.R.; Smith, M.O. Effects of different levels of zinc on the performance and immunocompetence of broilers under heat stress. Poult. Sci. 2003, 82, 1580–1588. [Google Scholar] [CrossRef]
- Mashaly, M.M.; Hendricks, G.L.; Kalama, M.A.; Gehad, A.E.; Abbas, A.O.; Patterson, P.H. Effect of heat stress on production parameters and immune responses of commercial laying hens. Poult. Sci. 2004, 83, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Siegel, H.V.; Van Kampen, M. Energy relationships in growing chickens given daily injections of corticosterone. Br. Poult. Sci. 1984, 25, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Vale, W.W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 2006, 8, 383. [Google Scholar]
- Etches, R.J.; John, T.M.; Gibbins, A.V. Behavioural, physiological, neuroendocrine and molecular responses to heat stress. In Poultry Production in Hot Climates, 2nd ed.; CABI: Oxfordshire, UK, 2008; pp. 31–66. [Google Scholar]
- Quinteiro-Filho, W.M.; Gomes, A.V.S.; Pinheiro, M.L.; Ribeiro, A.; Ferraz-de-Paula, V.; Astolfi-Ferreira, C.S.; Ferreira, A.J.P.; Palermo-Neto, J. Heat stress impairs performance and induces intestinal inflammation in broiler chickens infected with Salmonella Enteritidis. Avian Pathol. 2012, 41, 421–4127. [Google Scholar] [CrossRef]
- Star, L.; Decuypere, E.; Parmentier, H.K.; Kemp, B. Effect of single or combined climatic and hygienic stress in four layer lines: 2. Endocrine and oxidative stress responses. Poult. Sci. 2008, 87, 1031–1038. [Google Scholar] [CrossRef]
- Decuypere, E.; Kuhn, E.R. Thyroid Hormone Physiology in Galliformes: Age and Strain Related Changes in Physiological Control 1. Am. Zool. 1988, 28, 401–415. [Google Scholar] [CrossRef]
- Hoshino, S.; Suzuki, M.; Kakegawa, T.; Wakita, M.; Kobayashi, Y. Thyroid hormone response to thyrotrophin releasing hormone (TRH) in the sex-linked dwarf chicken. Endocrinol. Jpn. 1986, 33, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Melesse, A.; Maak, S.; Von Lengerken, G. Effects of genetic group× ambient temperature interactions on performance and physiological responses of Naked-neck chickens and their F1 crosses with Lohmann White and New Hampshire laying hens. J. Anim. Feed Sci. 2011, 20, 599–612. [Google Scholar] [CrossRef]
- Nawab, A.; Ibtisham, F.; Li, G.; Kieser, B.; Wu, J.; Liu, W. Heat stress in poultry production: Mitigation strategies to overcome the future challenges facing the global poultry industry. J. Therm. Biol. 2018, 78, 131–139. [Google Scholar] [CrossRef]
- Rozenboim, I.; Tako, E.; Gal-Garber, O.; Proudman, J.A.; Uni, Z. The effect of heat stress on ovarian function of laying hens. Poult. Sci. 2007, 86, 1760–1765. [Google Scholar] [CrossRef]
- Yoshida, N.; Fujita, M.; Nakahara, M.; Kuwahara, T.; Kawakami, S.I.; Bungo, T. Effect of high environmental temperature on egg production, serum lipoproteins and follicle steroid hormones in laying hens. J. Poult. Sci. 2011, 48, 207–211. [Google Scholar] [CrossRef]
- Lin, H.; Jiao, H.C.; Buyse, J.; Decuypere, E. Strategies for preventing heat stress in poultry. World’s Poult. Sci. J. 2006, 62, 71–86. [Google Scholar] [CrossRef]
- Abhu-Dieyeh, Z.H. Effect of Chronic Heat Stress and Long-Term Feed Restriction on Broiler Performance. Int. J. Poult. Sci. 2006, 5, 185–190. [Google Scholar]
- Uzum, M.H.; Toplu, H.D.O. Effects of stocking density and feed restriction on performance, carcass, meat quality characteristics and some stress parameters in broilers under heat stress. Rev. Med. Vet. (Toulouse) 2013, 164, 546–554. [Google Scholar]
- Mohamed, A.S.A.; Lozovskiy, A.R.; Ali, A.M.A. Strategies to combat the deleterious impacts of heat stress through feed restrictions and dietary supplementation (vitamins, minerals) in broilers. J. Indones. Trop. Anim. Agric. 2019, 44, 155–166. [Google Scholar] [CrossRef]
- MacLeod, M.G.; Hocking, P.M. Thermoregulation at high ambient temperature in genetically fat and lean broiler hens fed ad libitum or on a controlled-feeding regime. Br. Poult. Sci. 1993, 34, 589–596. [Google Scholar] [CrossRef]
- Francis, C.A.; Macleod, M.G.; Anderson, J.E. Alleviation of acute heat stress by food withdrawal or darkness. Br. Poult. Sci. 1991, 32, 219–225. [Google Scholar] [CrossRef]
- Wiernusz, C.J.; Teeter, R.G. Acclimation effects on fed and fasted broiler thermobalance during thermoneutral and high ambient temperature exposure. Br. Poult. Sci. 1996, 4, 677–687. [Google Scholar] [CrossRef]
- Westerterp, K.R. Diet induced thermogenesis. Nutr. Metab. (Lond.) 2004, 1, 1–5. [Google Scholar] [CrossRef]
- Basilio, V.; De Vilarin, M.; Yahav, S.; Picard, M. Early Age Thermal Conditioning and a Dual Feeding Program for Male Broilers Challenged by Heat Stress. Poult. Sci. 2001, 80, 29–36. [Google Scholar] [CrossRef]
- Lozano, C.; De Basilio, V.; Oliveros, I.; Alvarez, R.; Colina, I.; Bastianelli, D.; Yahav, S.; Picard, M. Is sequential feeding a suitable technique to compensate for the negative effects of a tropical climate in finishing broilers? Anim. Res. 2006, 55, 71–76. [Google Scholar] [CrossRef]
- Syafwan, S.; Kwakkel, R.P.; Group, A.N. Heat stress and feeding strategies in meat- type chickens. World’s Poult. Sci. Assoc. 2011, 67, 653–674. [Google Scholar] [CrossRef]
- Khoa, M.A. Wet and Coarse Diets in Broiler Nutrition: Development of the GI Tract and Performance. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2007. [Google Scholar]
- Moritz, J.S.; Beyer, R.S.; Wilson, K.J.; Cramer, K.R. Effect of moisture addition at the mixer to a corn-soybean-based diet on broiler performance. J. Appl. Poult. Res. 2001, 10, 347–353. [Google Scholar] [CrossRef]
- Shariatmadari, F.; Forbes, J.M. Performance of broiler chickens given whey in the food and/or drinking water. Br. Poult. Sci. 2005, 46, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Musharaf, N.A.; Latshaw, J.D. Heat increment as affected by protein and amino acid nutrition. World’s Poult. Sci. J. 1999, 55, 233–240. [Google Scholar] [CrossRef]
- Mateos, G.G.; Sell, J.L.; Eastwood, J.A. Rate of food passage (transit time) as influenced by level of supplemental fat. Poult. Sci. 1982, 61, 94–100. [Google Scholar] [CrossRef]
- Mateos, G.G.; Sell, J.L. Influence of fat and carbohydrate source on rate of food passage of semi-purified diets for laying hens. Poult. Sci. 1981, 60, 2114–2119. [Google Scholar] [CrossRef]
- Attia, Y.A.; Al-Harthi, M.A.; Sh Elnaggar, A. Productive, physiological and immunological responses of two broiler strains fed different dietary regimens and exposed to heat stress. Ital. J. Anim. Sci. 2018, 17, 686–697. [Google Scholar] [CrossRef]
- Daghir, N.J. (Ed.) Poultry Production in Hot Climates, 2nd ed.; CABI: Oxfordshire, UK, 2008. [Google Scholar]
- Ghazalah, A.A.; Abd-Elsamee, M.O.; Ali, A.M. Influence of dietary energy and poultry fat on the response of broiler chicks to heat therm. Int. J. Poult. Sci. 2008, 7, 355–359. [Google Scholar]
- Attia, Y.A.; Hassan, S.S. Broiler tolerance to heat stress at various dietary protein/energy levels. Eur. Poult. Sci. 2017, 81. [Google Scholar] [CrossRef]
- Dalólio, F.S.; Albino, L.F.T.; Lima, H.J.; Silva, J.N.D.; Moreira, J. Heat stress and vitamin E in diets for broilers as a mitigating measure. Acta Sci. Anim. Sci. 2015, 37, 419–427. [Google Scholar] [CrossRef]
- Meydani, S.N.; Blumberg, J.B. Vitamin E and the immune response. Nutr. Modul. Immune Response 1992, 16, 223–228. [Google Scholar]
- Puthpongsiriporn, U.; Scheideler, S.E.; Sell, J.L.; Beck, M.M. Effects of vitamin E and C supplementation on performance, in vitro lymphocyte proliferation, and antioxidant status of laying hens during heat stress. Poult. Sci. 2001, 80, 1190–1200. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.U.; Naz, S.; Nikousefat, Z.; Tufarelli, V.; Javdani, M.; Rana, N.; Laudadio, V. Effect of vitamin E in heat-stressed poultry. World’s Poult. Sci. J. 2011, 67, 469–478. [Google Scholar] [CrossRef]
- Bollengier-Lee, S. Optimal dietary concentration of vitamin E for alleviating the effect of heat stress on egg production in laying hens. Br. Poult. Sci. 1999, 40, 102–107. [Google Scholar] [CrossRef]
- Yardibi, H.; HOŞTÜRK, G.T. The effects of vitamin E on the antioxidant system, egg production, and egg quality in heat stressed laying hens. Turk. J. Vet. Anim. Sci. 2008, 32, 319–325. [Google Scholar]
- Mishra, B.; Sah, N.; Wasti, S. Genetic and Hormonal Regulation of Egg Formation in the Oviduct of Laying Hens. In Poultry; IntechOpen: London, UK, 2019. [Google Scholar]
- Sahin, K.; Kucuk, O. Effects of vitamin E and selenium on performance, digestibility of nutrients, and carcass characteristics of Japanese quails reared under heat stress (34 C). J. Anim. Physiol. Anim. Nutr. (Berl.) 2001, 85, 342–348. [Google Scholar] [CrossRef]
- Attia, Y.A.; Al-Harthi, M.A.; El-Shafey, A.S.; Rehab, Y.A.; Kim, W.K. Enhancing tolerance of broiler chickens to heat stress by supplementation with Vitamin E, Vitamin C and/or probiotics. Ann. Anim. Sci. 2017, 17, 1155–1169. [Google Scholar] [CrossRef]
- Sklan, D.; Melamed, D.; Friedman, A. The effect of varying levels of dietary vitamin A on immune response in the chick. Poult. Sci. 1994, 73, 843–847. [Google Scholar] [CrossRef]
- Palace, V.P.; Khaper, N.; Qin, Q.; Singal, P.K. Antioxidant potentials of vitamin A and carotenoids and their relevance to heart disease. Free Radic. Biol. Med. 1999, 26, 746–761. [Google Scholar] [CrossRef]
- Lin, H.; Wang, L.F.; Song, J.L.; Xie, Y.M.; Yang, Q.M. Effect of dietary supplemental levels of vitamin A on the egg production and immune responses of heat-stressed laying hens. Poult. Sci. 2002, 81, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Kucuk, O.; Sahin, N.; Sahin, K. Supplemental zinc and vitamin A can alleviate negative effects of heat stress in broiler chickens. Biol. Trace Elem. Res. 2003, 94, 225–235. [Google Scholar] [CrossRef]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.U.; Naz, S.; Nikousefat, Z.; Selvaggi, M.; Laudadio, V.; Tufarelli, V. Effect of ascorbic acid in heat-stressed poultry. World’s Poult. Sci. J. 2012, 68, 477–490. [Google Scholar] [CrossRef]
- Sahin, K.; Onderci, M.; Sahin, N.; Gursu, M.F.; Kucuk, O. Dietary vitamin C and folic acid supplementation ameliorates the detrimental effects of heat stress in Japanese quail. J. Nutr. 2003, 133, 1882–1886. [Google Scholar] [CrossRef]
- Njoku, P.C. Effect of dietary ascorbic acid (vitamin C) supplementation on the performance of broiler chickens in a tropical environment. Anim. Feed Sci. Technol. 1986, 16, 17–24. [Google Scholar] [CrossRef]
- Prasad, A.S.; Kucuk, O. Zinc in cancer prevention. Cancer Metastasis Rev. 2002, 21, 291–295. [Google Scholar] [CrossRef]
- Oteiza, P.L.; Olin, K.L.; Fraga, C.G.; Keen, C.L. Oxidant defense systems in testes from Zn deficient rats. Proc. Soc. Exp. Biol. Med. 1996, 213, 85–91. [Google Scholar] [CrossRef]
- Balnave, D.; Muheereza, S.K. Improving eggshell quality at high temperatures with dietary sodium bicarbonate. Poult. Sci. 1997, 76, 588–593. [Google Scholar] [CrossRef]
- Lee, S.R. Critical role of zinc as either an antioxidant or a prooxidant in cellular systems. Oxid. Med. Cell. Longev. 2018, 2018, 9156285. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.V.R.; Prakash, B.; Raju, M.V.L.N.; Panda, A.K.; Kumari, R.K.; Reddy, E.P.K. Effect of Supplementing Organic Forms of Zinc, Selenium and Chromium on Performance, Anti-Oxidant and Immune Responses in Broiler Chicken Reared in Tropical Summer. Biol. Trace Elem. Res. 2016, 172, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Kucuk, O. Zinc in a combination with magnesium helps reducing negative effects of heat stress in quails. Biol. Trace Elem. Res. 2008, 123, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Kucuk, O. Zinc Supplementation Alleviates Heat Stress in Laying Japanese. J. Nutr. 2003, 133, 2808–2811. [Google Scholar] [CrossRef]
- Moreng, R.E.; Balnave, D.; Zhang, D. Dietary zinc methionine effect on eggshell quality of hens drinking saline water. Poult. Sci. 1992, 71, 1163–1167. [Google Scholar] [CrossRef] [PubMed]
- Balnave, D.; Zhang, D. Research note: Responses of laying hens on saline drinking water to dietary supplementation with various zinc compounds. Poult. Sci. 1993, 72, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.B. The biochemistry of chromium. J. Nutr. 2000, 130, 715–718. [Google Scholar] [CrossRef]
- Hayirli, A. Chromium nutrition of livestock species. Nutr. Abstr. Rev. Ser. B Livest. Feed. Feed. 2005, 75, 1N–14N. [Google Scholar]
- Sahin, K.; Sahin, N.; Onderci, M.; Gursu, F.; Cikim, G. Optimal dietary concentration of chromium for alleviating the effect of heat stress on growth, carcass qualities, and some serum metabolites of broiler chickens. Biol. Trace Elem. Res. 2002, 89, 53–64. [Google Scholar] [CrossRef]
- Jahanian, R.; Rasouli, E. Dietary chromium methionine supplementation could alleviate immunosuppressive effects of heat stress in broiler chicks. J. Anim. Sci. 2015, 93, 3355–3363. [Google Scholar] [CrossRef]
- Li, S.; Luo, X.; Liu, B.; Shao, G.; Guo, X.; Yu, S. Effect of organic chromium on egg production and immune responses in heat stressed layers. (Ying Yang Xue Bao) Acta Nutrimenta Sinica 2001, 23, 117–121. [Google Scholar]
- Sahin, K.; Ozbey, O.; Onderci, M.; Cikim, G.; Aysondu, M.H. Chromium supplementation can alleviate negative effects of heat stress on egg production, egg quality and some serum metabolites of laying Japanese quail. J. Nutr. 2002, 132, 1265–1268. [Google Scholar] [CrossRef] [PubMed]
- Torki, M.; Zangeneh, S.; Habibian, M. Performance, egg quality traits, and serum metabolite concentrations of laying hens affected by dietary supplemental chromium picolinate and vitamin C under a heat-stress condition. Biol. Trace Elem. Res. 2014, 157, 120–129. [Google Scholar] [CrossRef]
- Zhou, J.; Huang, K.; Lei, X.G. Selenium and diabetes—Evidence from animal studies. Free Radic. Biol. Med. 2013, 65, 1548–1556. [Google Scholar] [CrossRef]
- Nazıroğlu, M.; Yıldız, K.; Tamtürk, B.; Erturan, İ.; Flores-Arce, M. Selenium and psoriasis. Biol. Trace Elem. Res. 2012, 150, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.R.; Nicol, F.; Beckett, G.J. The role of selenium in thyroid hormone metabolism and effects of selenium deficiency on thyroid hormone and iodine metabolism. Biol. Trace Elem. Res. 1992, 33, 37–42. [Google Scholar] [CrossRef]
- Yang, Y.R.; Meng, F.C.; Wang, P.; Jiang, Y.B.; Yin, Q.Q.; Chang, J.; Zuo, R.Y.; Zheng, Q.H.; Liu, J.X. Effect of organic and inorganic selenium supplementation on growth performance, meat quality and antioxidant property of broilers. Afr. J. Biotechnol. 2012, 11, 3031–3036. [Google Scholar]
- Rahimi, S.; Farhadi, D.; Valipouri, A.R. Effect of organic and inorganic selenium sources and vitamin E on broiler performance and carcass characteristics in heat stress condition. Vet. J. 2011, 91, 25–35. [Google Scholar]
- Attia, Y.A.; Abdalah, A.A.; Zeweil, H.S.; Bovera, F.; Tag El-Din, A.A.; Araft, M.A. Effect of inorganic or organic selenium supplementation on productive performance, egg quality and some physiological traits of dual-purpose breeding hens. Czech J. Anim. Sci. 2010, 55, 505–519. [Google Scholar] [CrossRef]
- Siske, V.; Zeman, L.; Klecker, D. The egg shell: A case study in improving quality by altering mineral metabolism–naturally. In Biotechnology in the Feed Industry, Proceedings of the Alltech’s 16th Annual Symposium; Nottingham University Press: Nottingham, UK, 2000; pp. 327–346. [Google Scholar]
- Sahin, N.; Onderci, M.; Sahin, K.; Kucuk, O. Supplementation with organic or inorganic selenium in heat-distressed quail. Biol. Trace Elem. Res. 2008, 122, 229–237. [Google Scholar] [CrossRef]
- Ahmad, T.; Khalid, T.; Mushtaq, T.; Mirza, M.A.; Nadeem, A.; Babar, M.E.; Ahmad, G. Effect of potassium chloride supplementation in drinking water on broiler performance under heat stress conditions. Poult. Sci. 2008, 87, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, S.; Cohen, I.; Bar, A.; Bornstein, S. Sodium and Chloride Requirements of the Chick: Relationship to Acid-Base Balance. Poult. Sci. 1973, 52, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, M.M.H.; Pasha, T.N.; Mushtaq, T.; Parvin, R. Electrolytes, dietary electrolyte balance and salts in broilers: An updated review on growth performance, water intake and litter quality. World’s Poult. Sci. J. 2013, 69, 789–802. [Google Scholar] [CrossRef]
- Benton, C.E.; Balnave, D.; Pas, J.B.; Boyd, L.J.; Brown, M.A. The use of dietary minerals during heat stress in broilers. Prof. Anim. Sci. 1998, 14, 193–196. [Google Scholar] [CrossRef]
- Smith, M.O.; Teeter, R.G. Potassium balance of the 5 to 8-week-old broiler exposed to constant heat or cycling high temperature stress and the effects of supplemental potassium chloride on body weight gain and feed efficiency. Poult. Sci. 1987, 66, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Arain, M.A.; Mei, Z.; Hassan, F.U.; Saeed, M.; Alagawany, M.; Shar, A.H.; Rajput, I.R. Lycopene: A natural antioxidant for prevention of heat-induced oxidative stress in poultry. World’s Poult. Sci. J. 2018, 74, 89–100. [Google Scholar] [CrossRef]
- Sahin, K.; Orhan, C.; Tuzcu, M.; Sahin, N.; Hayirli, A.; Bilgili, S.; Kucuk, O. Lycopene activates antioxidant enzymes and nuclear transcription factor systems in heat-stressed broilers. Poult. Sci. 2016, 95, 1088–1095. [Google Scholar] [CrossRef]
- Hu, R.; He, Y.; Arowolo, M.A.; Wu, S.; He, J. Polyphenols as Potential Attenuators of Heat Stress in Poultry Production. Antioxidants 2019, 8, 67. [Google Scholar] [CrossRef]
- He, S.; Li, S.; Arowolo, M.A.; Yu, Q.; Chen, F.; Hu, R.; He, J. Effect of resveratrol on growth performance, rectal temperature and serum parameters of yellow-feather broilers under heat stress. Anim. Sci. J. 2019, 90, 401–411. [Google Scholar] [CrossRef]
- Zhang, C.; Zhao, X.H.; Yang, L.; Chen, X.Y.; Jiang, R.S.; Jin, S.H.; Geng, Z.Y. Resveratrol alleviates heat stress-induced impairment of intestinal morphology, microflora, and barrier integrity in broilers. Poult. Sci. 2017, 96, 4325–4332. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, L.; Zhao, X.H.; Chen, X.Y.; Yang, L.; Geng, Z.Y. Dietary resveratrol supplementation prevents transport-stress-impaired meat quality of broilers through maintaining muscle energy metabolism and antioxidant status. Poult. Sci. 2017, 96, 2219–2225. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Kang, X.; Zhang, T.; Huang, J. Positive Effects of Resveratrol on Egg-Laying Ability, Egg Quality, and Antioxidant Activity in Hens. J. Appl. Poult. Res. 2019, 28, 1099–1105. [Google Scholar] [CrossRef]
- Luo, J.; Song, J.; Liu, L.; Xue, B.; Tian, G.; Yang, Y. Effect of epigallocatechin gallate on growth performance and serum biochemical metabolites in heat-stressed broilers. Poult. Sci. 2017, 97, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Xue, B.; Song, J.; Liu, L.; Luo, J.; Tian, G.; Yang, Y. Effect of epigallocatechin gallate on growth performance and antioxidant capacity in heat-stressed broilers. Arch. Anim. Nutr. 2017, 71, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Orhan, C.; Tuzcu, M.; Ali, S.; Sahin, N.; Hayirli, A. Epigallocatechin-3-gallate prevents lipid peroxidation and enhances antioxidant defense system via modulating hepatic nuclear transcription factors in heat-stressed quails. Poult. Sci. 2010, 89, 2251–2258. [Google Scholar] [CrossRef]
- Attia, Y.A.; Al-Harthi, M.A.; Hassan, S.S. Turmeric (Curcuma longa Linn.) as a phytogenic growth promoter alternative for antibiotic and comparable to mannan oligosaccharides for broiler chicks. Rev. Mex. Cienc. Pecu. 2017, 8, 11–21. [Google Scholar] [CrossRef]
- Zhang, J.; Bai, K.W.; He, J.; Niu, Y.; Lu, Y.; Zhang, L.; Wang, T. Curcumin attenuates hepatic mitochondrial dysfunction through the maintenance of thiol pool, inhibition of mtDNA damage, and stimulation of the mitochondrial thioredoxin system in heat-stressed broilers. J. Anim. Sci. 2018, 96, 867–879. [Google Scholar] [CrossRef]
- Zhang, J.F.; Bai, K.W.; Su, W.P.; Wang, A.A.; Zhang, L.L.; Huang, K.H.; Wang, T. Curcumin attenuates heat-stress-induced oxidant damage by simultaneous activation of GSH-related antioxidant enzymes and Nrf2-mediated phase II detoxifying enzyme systems in broiler chickens. Poult. Sci. 2018, 97, 1209–1219. [Google Scholar] [CrossRef]
- Zhang, J.F.; Hu, Z.P.; Lu, C.H.; Yang, M.X.; Zhang, L.L.; Wang, T. Dietary curcumin supplementation protects against heat-stress-impaired growth performance of broilers possibly through a mitochondrial pathway. J. Anim. Sci. 2015, 93, 1656–1665. [Google Scholar] [CrossRef]
- Liu, M.; Lu, Y.; Gao, P.; Xie, X.; Li, D.; Yu, D.; Yu, M. Effect of curcumin on laying performance, egg quality, endocrine hormones, and immune activity in heat-stressed hens. Poult. Sci. 2020, 99, 2196–2202. [Google Scholar] [CrossRef]
- Craig, S.A. Betaine in human nutrition. Am. J. Clin. Nutr. 2004, 80, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Ratriyanto, A.; Mosenthin, R. Osmoregulatory function of betaine in alleviating heat stress in poultry. J. Anim. Physiol. Anim. Nutr. (Berl.) 2018, 102, 1634–1650. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; He, F.; Wu, C.; Li, P.; Li, N.; Deng, J.; Zhu, G.; Ren, W.; Peng, Y. Betaine in inflammation: Mechanistic aspects and applications. Front. Immunol. 2018, 9, 1070. [Google Scholar] [CrossRef] [PubMed]
- Chand, N.; Naz, S.; Maris, H.; Khan, R.U.; Khan, S.; Qureshi, M.S. Effect of betaine supplementation on the performance and immune response of heat stressed broilers. Pak. J. Zool. 2017, 49, 1857–1862. [Google Scholar] [CrossRef]
- Liu, W.; Yuan, Y.; Sun, C.; Balasubramanian, B.; Zhao, Z.; An, L. Effects of dietary betaine on growth performance, digestive function, carcass traits, and meat quality in indigenous yellow-feathered broilers under long-term heat stress. Animals 2019, 9, 506. [Google Scholar] [CrossRef] [PubMed]
- Attia, Y.A.; Hassan, R.A.; Qota, E.M.A. Recovery from adverse effects of heat stress on slow-growing chicks in the tropics 1: Effect of ascorbic acid and different levels of betaine. Trop. Anim. Health Prod. 2009, 41, 807–818. [Google Scholar] [CrossRef]
- Attia, Y.A.; Abd El-Hamid, A.E.H.E.; Abedalla, A.A.; Berika, M.A.; Al-Harthi, M.A.; Kucuk, O.; Sahin, K.; Abou-Shehema, B.M. Laying performance, digestibility and plasma hormones in laying hens exposed to chronic heat stress as affected by betaine, vitamin C, and/or vitamin E supplementation. Springerplus 2016, 5, 1–12. [Google Scholar] [CrossRef]
- Attia, Y.A.; El-Naggar, A.S.; Abou-Shehema, B.M.; Abdella, A.A. Effect of supplementation with trimethylglycine (Betaine) and/or vitamins on semen quality, fertility, antioxidant status, dna repair and welfare of roosters exposed to chronic heat stress. Animals 2019, 9, 547. [Google Scholar] [CrossRef]
- Ripps, H.; Shen, W. Review: Taurine: A “very essential” amino acid. Mol. Vis. 2012, 18, 2673–2686. [Google Scholar]
- Belal, S.A.; Kang, D.R.; Cho, E.S.R.; Park, G.H.; Shim, K.S. Taurine reduces heat stress by regulating the expression of heat shock proteins in broilers exposed to chronic heat. Rev. Bras. Cienc. Avic. 2018, 20, 479–486. [Google Scholar] [CrossRef]
- He, X.; Lu, Z.; Ma, B.; Zhang, L.; Li, J.; Jiang, Y.; Zhou, G.; Gao, F. Effects of dietary taurine supplementation on growth performance, jejunal morphology, appetite-related hormones, and genes expression in broilers subjected to chronic heat stress. Poult. Sci. 2019, 98, 2719–2728. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; He, X.; Ma, B.; Zhang, L.; Li, J.; Jiang, Y.; Zhou, G.; Gao, F. Dietary taurine supplementation decreases fat synthesis by suppressing the liver X receptor α pathway and alleviates lipid accumulation in the liver of chronic heat-stressed broilers. J. Sci. Food Agric. 2019, 99, 5631–5637. [Google Scholar] [CrossRef] [PubMed]
- Dai, B.; Zhang, Y.-S.; Ma, Z.-L.; Zheng, L.-H.; Li, S.-J.; Dou, X.-H.; Gong, J.-S.; Miao, J.-F. Influence of dietary taurine and housing density on oviduct function in laying hens. J. Zhejiang Univ. Sci. B 2015, 16, 456–464. [Google Scholar] [CrossRef]
- Merat, P. Potential usefulness of the Na (naked neck) gene in poultry production. World’s Poult. Sci. J. 1986, 42, 124–142. [Google Scholar] [CrossRef]
- Azoulay, Y.; Druyan, S.; Yadgary, L.; Hadad, Y.; Cahaner, A. The viability and performance under hot conditions of featherless broilers versus fully feathered broilers. Poult. Sci. 2011, 90, 19–29. [Google Scholar] [CrossRef]
- Cahaner, A.; Deeb, N.; Gutman, M. Effects of the plumage-reducing naked neck (Na) gene on the performance of fast-growing broilers at normal and high ambient temperatures. Poult. Sci. 1993, 72, 767–775. [Google Scholar] [CrossRef]
- Rajkumar, U.; Reddy, B.L.N.; Rajaravindra, K.S.; Niranjan, M.; Bhattacharya, T.K.; Chatterjee, R.N.; Panda, A.K.; Reddy, M.R.; Sharma, R.P. Effect of naked neck gene on immune competence, serum biochemical and carcass traits in chickens under a tropical climate. Asian-Australas. J. Anim. Sci. 2010, 23, 867–872. [Google Scholar] [CrossRef]
- Yalcin, S.; Testik, A.; Ozkan, S.; Settar, P.; Celen, F.; Cahaner, A. Performance of naked neck and normal broilers in hot, warm, and temperate climates. Poult. Sci. 1997, 76, 930–937. [Google Scholar] [CrossRef]
- Rajkumar, U.; Reddy, M.R.; Rao, S.V.; Radhika, K.; Shanmugam, M. Evaluation of growth, carcass, immune response and stress parameters in naked neck chicken and their normal siblings under tropical winter and summer temperatures. Asian-Australas. J. Anim. Sci. 2011, 24, 509–516. [Google Scholar] [CrossRef]
- Fathi, M.M.; Galal, A.; El-Safty, S.; Mahrous, M. Naked neck and frizzle genes for improving chickens raised under high ambient temperature: I. Growth performance and egg production. World’s Poult. Sci. J. 2013, 69, 813–832. [Google Scholar] [CrossRef]
- Zerjal, T.; Gourichon, D.; Rivet, B.; Bordas, A. Performance comparison of laying hens segregating for the frizzle gene under thermoneutral and high ambient temperatures. Poult. Sci. 2013, 92, 1474–1485. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, A.R.; Horst, P.; Simianer, H. The effect of frizzle gene and dwarf gene on reproductive performance of broiler breeder dams under high and normal ambient temperatures. Poult. Sci. 2010, 89, 2356–2369. [Google Scholar] [CrossRef] [PubMed]
- Yunis, R.; Cahaner, A. The effects of the naked neck (Na) and frizzle (F) genes on growth and meat yield of broilers and their interactions with ambient temperatures and potential growth rate. Poult. Sci. 1999, 78, 1347–1352. [Google Scholar] [CrossRef] [PubMed]
- Decuypere, E.; Huybrechts, L.M.; Kuhn, E.R.; Tixier-Boichard, M.; Merat, P. Physiological alterations associated with the chicken sex-linked dwarfing gene. Crit. Rev. Poult. Biol. (USA) 1991, 3, 191–221. [Google Scholar]
- Deeb, N.; Cahaner, A. Genotype-by-environment interaction with broiler genotypes differing in growth rate: 2. The effects of high ambient temperature on dwarf versus normal broilers. Poult. Sci. 2001, 80, 541–548. [Google Scholar] [CrossRef]
- Oloyo, A.; Ojerinde, A. Poultry Housing and Management. In Poultry; IntechOpen: London, UK, 2019. [Google Scholar]
- NaRanong, V. Structural Changes in Thailand’s Poultry Sector and Its Social Implications; Thailand Development Research Institute: Bangkok, Thailand, 2007. [Google Scholar]
- Glatz, P.; Pym, R. Poultry housing and management in developing countries. In Poultry Development Review; FAO: Rome, Italy, 2013; pp. 24–28. [Google Scholar]
- Park, B.S.; Um, K.H.; Park, S.O.; Zammit, V.A. Effect of stocking density on behavioral traits, blood biochemical parameters and immune responses in meat ducks exposed to heat stress. Arch. Anim. Breed. 2018, 61, 425–432. [Google Scholar] [CrossRef]
- Taouis, M.; De Basilio, V.; Mignon-Grasteau, S.; Crochet, S.; Bouchot, C.; Bigot, K.; Collin, A.; Picard, M. Early-age thermal conditioning reduces uncoupling protein messenger RNA expression in pectoral muscle of broiler chicks at seven days of age. Poult. Sci. 2002, 81, 1640–1643. [Google Scholar] [CrossRef]
- Liew, P.K.; Zulkifli, I.; Hair-Bejo, M.; Omar, A.R.; Israf, D.A. Effects of early age feed restriction and heat conditioning on heat shock protein 70 expression, resistance to infectious bursal disease, and growth in male broiler chickens subjected to heat stress. Poult. Sci. 2003, 82, 1879–1885. [Google Scholar] [CrossRef]
- Brosnan, J.T.; Brosnan, M.E. The sulfur-containing amino acids: An overview. J. Nutr. 2006, 136, 1636S–1640S. [Google Scholar] [CrossRef]
- Elnesr, S.S.; Elwan, H.A.M.; Xu, Q.Q.; Xie, C.; Dong, X.Y.; Zou, X.T. Effects of in ovo injection of sulfur-containing amino acids on heat shock protein 70, corticosterone hormone, antioxidant indices, and lipid profile of newly hatched broiler chicks exposed to heat stress during incubation. Poult. Sci. 2019, 98, 2290–2298. [Google Scholar] [CrossRef]
- Yi, D.; Hou, Y.; Tan, L.; Liao, M.; Xie, J.; Wang, L.; Ding, B.; Yang, Y.; Gong, J. N-acetylcysteine improves the growth performance and intestinal function in the heat-stressed broilers. Anim. Feed Sci. Technol. 2016, 220, 83–92. [Google Scholar] [CrossRef]
- Omid, K.; Amirali, S.; Ahmad, K. N-Acetyl cysteine improves performance, reproduction, antioxidant status, immunity and maternal antibody transmission in breeder Japanese quail under heat stress condition. Livest. Sci. 2018, 217, 55–64. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wasti, S.; Sah, N.; Mishra, B. Impact of Heat Stress on Poultry Health and Performances, and Potential Mitigation Strategies. Animals 2020, 10, 1266. https://doi.org/10.3390/ani10081266
Wasti S, Sah N, Mishra B. Impact of Heat Stress on Poultry Health and Performances, and Potential Mitigation Strategies. Animals. 2020; 10(8):1266. https://doi.org/10.3390/ani10081266
Chicago/Turabian StyleWasti, Sanjeev, Nirvay Sah, and Birendra Mishra. 2020. "Impact of Heat Stress on Poultry Health and Performances, and Potential Mitigation Strategies" Animals 10, no. 8: 1266. https://doi.org/10.3390/ani10081266
APA StyleWasti, S., Sah, N., & Mishra, B. (2020). Impact of Heat Stress on Poultry Health and Performances, and Potential Mitigation Strategies. Animals, 10(8), 1266. https://doi.org/10.3390/ani10081266






