Effects of Artificial Light at Night (ALAN) on European Hedgehog Activity at Supplementary Feeding Stations
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Survey
2.2. Statistical Analysis
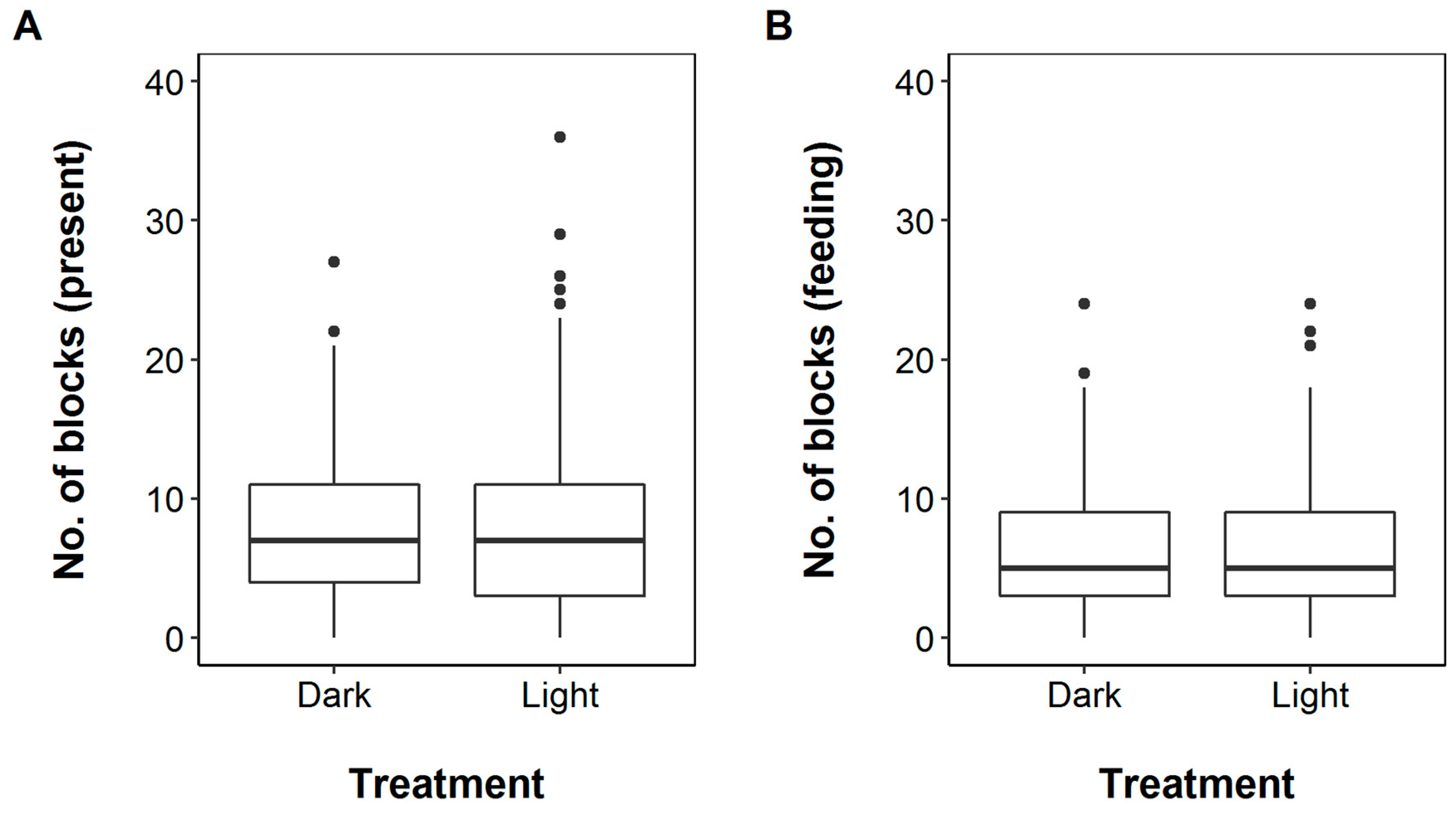
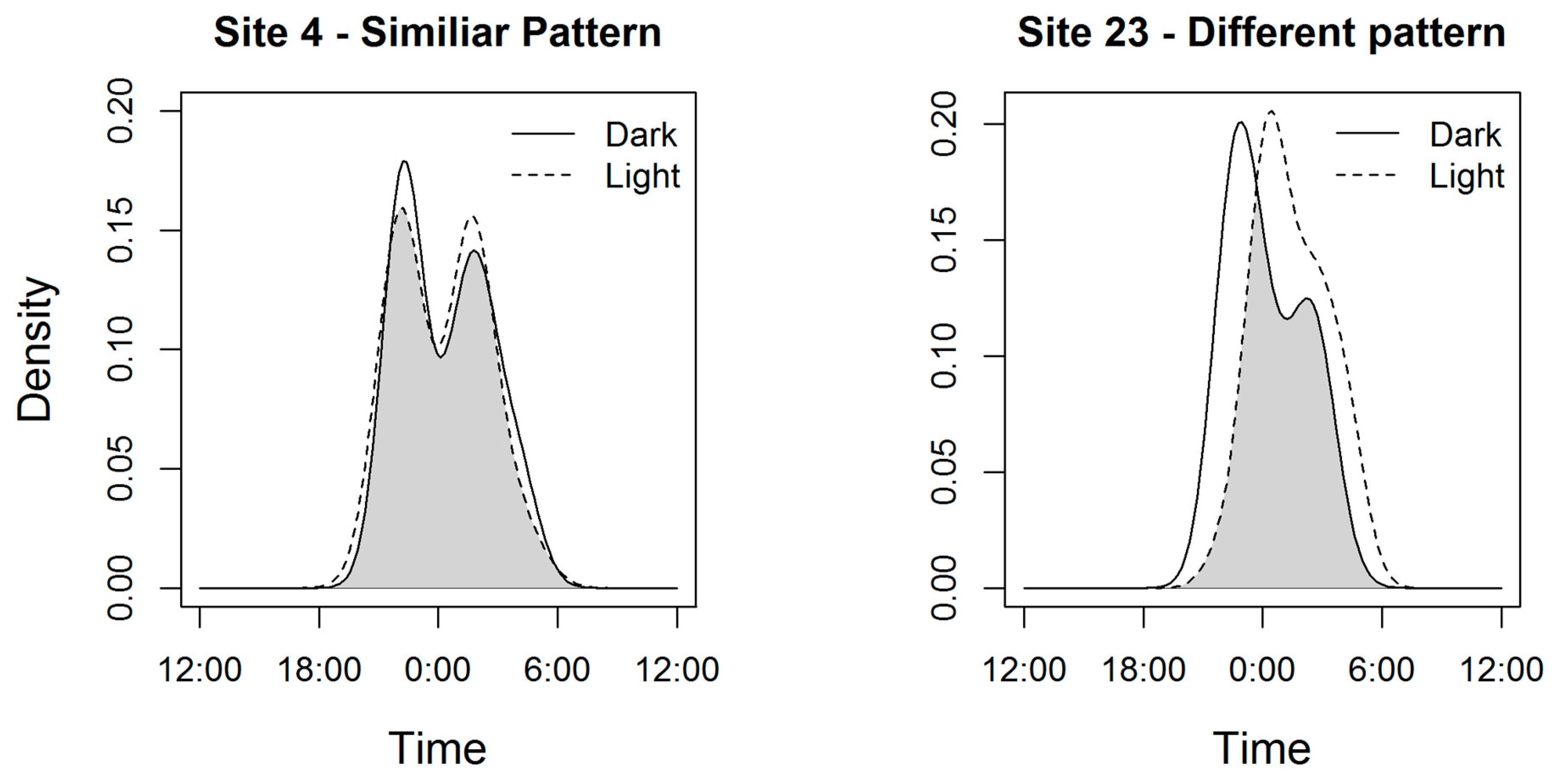
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- UN DESA. World Urbanization Prospects 2018: Highlights; UN DESA: New York, NY, USA, 2019. [Google Scholar] [CrossRef]
- Jung, K.; Kalko, E.K. Where forest meets urbanization: Foraging plasticity of aerial insectivorous bats in an anthropogenically altered environment. J. Mammal. 2010, 91, 144–153. [Google Scholar] [CrossRef]
- Penone, C.; Kerbiriou, C.; Julien, J.F.; Julliard, R.; Machon, N.; Le Viol, I. Urbanisation effect on O rthoptera: Which scale matters? Insect Conserv. Divers. 2013, 6, 319–327. [Google Scholar] [CrossRef]
- Gaston, K.J.; Duffy, J.P.; Gaston, S.; Bennie, J.; Davies, T.W. Human alteration of natural light cycles: Causes and ecological consequences. Oecologia 2014, 176, 917–931. [Google Scholar] [CrossRef] [PubMed]
- Bennie, J.; Davies, T.W.; Cruse, D.; Inger, R.; Gaston, K.J. Cascading effects of artificial light at night: Resource-mediated control of herbivores in a grassland ecosystem. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140131. [Google Scholar] [CrossRef]
- Lewanzik, D.; Voigt, C.C. Artificial light puts ecosystem services of frugivorous bats at risk. J. Appl. Ecol. 2014, 51, 388–394. [Google Scholar] [CrossRef]
- Becker, A.; Whitfield, A.K.; Cowley, P.D.; Järnegren, J.; Næsje, T.F. Potential effects of artificial light associated with anthropogenic infrastructure on the abundance and foraging behaviour of estuary-associated fishes. J. Appl. Ecol. 2013, 50, 43–50. [Google Scholar] [CrossRef]
- Evans, W.R.; Akashi, Y.; Altman, N.S.; Manville, A.M. Response of night-migrating songbirds in cloud to colored and flashing light. N. Am. Birds 2007, 60, 476–488. [Google Scholar]
- Cresswell, W.; Harris, S. The effects of weather conditions on the movements and activity of badgers (Meles meles) in a suburban environment. J. Zool. 1988, 216, 187–194. [Google Scholar] [CrossRef]
- Elangovan, V.; Marimuthu, G. Effect of moonlight on the foraging behaviour of a megachiropteran bat Cynopterus sphinx. J. Zool. 2001, 253, 347–350. [Google Scholar] [CrossRef]
- Preston, E.F.; Johnson, P.J.; Macdonald, D.W.; Loveridge, A.J. Hunting success of lions affected by the moon’s phase in a wooded habitat. Afr. J. Ecol. 2019, 57, 586–594. [Google Scholar] [CrossRef]
- Mathews, F.; Roche, N.; Aughney, T.; Jones, N.; Day, J.; Baker, J.; Langton, S. Barriers and benefits: Implications of artificial night-lighting for the distribution of common bats in Britain and Ireland. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140124. [Google Scholar] [CrossRef] [PubMed]
- Day, J.; Baker, J.; Schofield, H.; Mathews, F.; Gaston, K. Part-night lighting: Implications for bat conservation. Anim. Conserv. 2015, 18, 512–516. [Google Scholar] [CrossRef]
- Mathews, F.; Kubasiewicz, L.; Gurnell, J.; Harrower, C.; McDonald, R.; Shore, R. A Review of the Population and Conservation Status of British Mammals. A Report by the Mammal Society under Contract to Natural England, Natural Resources Wales and Scottish Natural Heritage; Natural England: Peterborough, UK, 2018. [Google Scholar]
- Huijser, M.P.; Bergers, P.J. The effect of roads and traffic on hedgehog (Erinaceus europaeus) populations. Biol. Conserv. 2000, 95, 111–116. [Google Scholar] [CrossRef]
- van de Poel, J.L.; Dekker, J.; van Langevelde, F. Dutch hedgehogs Erinaceus europaeus are nowadays mainly found in urban areas, possibly due to the negative effects of badgers Meles meles. Wildl. Biol. 2015, 21, 51–55. [Google Scholar] [CrossRef]
- Holsbeek, L.; Rodts, J.; Muyldermans, S. Hedgehog and other animal traffic victims in Belgium: Results of a countrywide survey. Lutra 1999, 42, 111–119. [Google Scholar]
- Krange, M. Change in the Occurrence of the West European Hedgehog (Erinaceus europaeus) in Western Sweden during 1950–2010; Karlstad University: Karlstad, Sweden, 2015. [Google Scholar]
- Pettett, C.E.; Johnson, P.J.; Moorhouse, T.P.; Macdonald, D.W. National predictors of hedgehog Erinaceus europaeus distribution and decline in Britain. Mammal Rev. 2018, 48, 1–6. [Google Scholar] [CrossRef]
- Williams, B.M.; Baker, P.J.; Thomas, E.; Wilson, G.; Judge, J.; Yarnell, R.W. Reduced occupancy of hedgehogs (Erinaceus europaeus) in rural England and Wales: The influence of habitat and an asymmetric intra-guild predator. Sci. Rep. 2018, 8, 12156. [Google Scholar] [CrossRef]
- Hof, A.R.; Allen, A.M.; Bright, P.W. Investigating the role of the eurasian badger (Meles meles) in the nationwide distribution of the western european hedgehog (Erinaceus europaeus) in England. Animals 2019, 9, 759. [Google Scholar] [CrossRef]
- Wembridge, D.E.; Newman, M.R.; Bright, P.W.; Morris, P.A. An estimate of the annual number of hedgehog (Erinaceus europaeus) road casualties in Great Britain. Mammal Commun. 2016, 2, 8–14. [Google Scholar]
- Wright, P.G.; Coomber, F.G.; Bellamy, C.C.; Perkins, S.E.; Mathews, F. Predicting hedgehog mortality risks on British roads using habitat suitability modelling. PeerJ 2020, 7, e8154. [Google Scholar] [CrossRef]
- Hof, A.R.; Bright, P.W. The value of green-spaces in built-up areas for western hedgehogs. Lutra 2009, 52, 69–82. [Google Scholar]
- Doncaster, C.P. Factors regulating local variations in abundance: Field tests on hedgehogs, Erinaceus europaeus. Oikos 1994, 182–192. [Google Scholar] [CrossRef]
- Rondinini, C.; Doncaster, C. Roads as barriers to movement for hedgehogs. Funct. Ecol. 2002, 16, 504–509. [Google Scholar] [CrossRef]
- Orlowski, G.; Nowak, L. Road mortality of hedgehogs Erinaceus spp. in farmland in Lower Silesia (south-western Poland). Pol. J. Ecol. 2004, 52, 377–382. [Google Scholar]
- Bennie, J.; Davies, T.W.; Duffy, J.P.; Inger, R.; Gaston, K.J. Contrasting trends in light pollution across Europe based on satellite observed night time lights. Sci. Rep. 2014, 4, 3789. [Google Scholar] [CrossRef]
- Kyba, C.C.; Tong, K.P.; Bennie, J.; Birriel, I.; Birriel, J.J.; Cool, A.; Danielsen, A.; Davies, T.W.; Peter, N.; Edwards, W. Worldwide variations in artificial skyglow. Sci. Rep. 2015, 5, 8409. [Google Scholar] [CrossRef]
- Morton, D.; Rowland, C.; Wood, C.; Meek, L.; Marston, C.; Smith, G.; Wadsworth, R.; Simpson, I. Final Report for LCM2007-the New UK Land Cover Map; Countryside Survey Technical Report No 11/07; NERC/Centre for Ecology & Hydrology: Bailrigg, UK, 2011. [Google Scholar]
- ESRI. ArcGIS Release 10.5; Environmental Systems Research Institute: Redlands, CA, USA, 2016. [Google Scholar]
- Pettett, C.E.; Moorhouse, T.P.; Johnson, P.J.; Macdonald, D.W. Factors affecting hedgehog (Erinaceus europaeus) attraction to rural villages in arable landscapes. Eur. J. Wildl. Res. 2017, 63, 54. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: https://www.R-project.org/ (accessed on 6 December 2018).
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. arXiv 2014, arXiv:1406.5823. [Google Scholar]
- Ridout, M.S.; Linkie, M. Estimating overlap of daily activity patterns from camera trap data. J. Agric. Biol. Environ. Stat. 2009, 14, 322–337. [Google Scholar] [CrossRef]
- Rowcliffe, J.M.; Kays, R.; Kranstauber, B.; Carbone, C.; Jansen, P.A. Quantifying levels of animal activity using camera trap data. Methods Ecol. Evol. 2014, 5, 1170–1179. [Google Scholar] [CrossRef]
- Rowcliffe, M. Activity: Animal Activity Statistics, R package version 1.1; 2016; Available online: https://rdrr.io/cran/activity/ (accessed on 11 March 2019).
- Lashley, M.A.; Cove, M.V.; Chitwood, M.C.; Penido, G.; Gardner, B.; DePerno, C.S.; Moorman, C.E. Estimating wildlife activity curves: Comparison of methods and sample size. Sci. Rep. 2018, 8, 4173. [Google Scholar] [CrossRef]
- Gaston, K.J.; Bennie, J. Demographic effects of artificial nighttime lighting on animal populations. Environ. Rev. 2014, 22, 323–330. [Google Scholar] [CrossRef]
- Bennie, J.; Davies, T.W.; Cruse, D.; Bell, F.; Gaston, K.J. Artificial light at night alters grassland vegetation species composition and phenology. J. Appl. Ecol. 2018, 55, 442–450. [Google Scholar] [CrossRef]
- Bennie, J.; Davies, T.W.; Cruse, D.; Inger, R.; Gaston, K.J. Artificial light at night causes top-down and bottom-up trophic effects on invertebrate populations. J. Appl. Ecol. 2018, 55, 2698–2706. [Google Scholar] [CrossRef]
- Hoffmann, J.; Palme, R.; Eccard, J.A. Long-term dim light during nighttime changes activity patterns and space use in experimental small mammal populations. Environ. Pollut. 2018, 238, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Yurk, H.; Trites, A. Experimental attempts to reduce predation by harbor seals on out-migrating juvenile salmonids. Trans. Am. Fish. Soc. 2000, 129, 1360–1366. [Google Scholar] [CrossRef]
- De Molenaar, J.C.; Henkens, R.H.C.; ter Braak, C.; van Duyne, C.; Hoefsloot, C.; Jonkers, D.A. Road Illumination and Nature IV; Ministry of Transport Public Works and Water Management of The Netherlands: The Hague, The Netherlands, 2013; p. 72. [Google Scholar]
- Reeve, N. Hedgehogs; T & AD Poyser: Calton, UK, 1994. [Google Scholar]
- Wroot, A.J. Feeding Ecology of the European Hedgehog Erinaceus europaeus; Royal Holloway, University of London: London, UK, 1984. [Google Scholar]
- Schoeman, M. Light pollution at stadiums favors urban exploiter bats. Anim. Conserv. 2016, 19, 120–130. [Google Scholar] [CrossRef]
- Rydell, J. Seasonal use of illuminated areas by foraging northern bats Eptesicus nilssoni. Ecography 1991, 14, 203–207. [Google Scholar] [CrossRef]
- Rydell, J. Exploitation of insects around streetlamps by bats in Sweden. Funct. Ecol. 1992, 744–750. [Google Scholar] [CrossRef]
- Azam, C.; Le Viol, I.; Bas, Y.; Zissis, G.; Vernet, A.; Julien, J.-F.; Kerbiriou, C. Evidence for distance and illuminance thresholds in the effects of artificial lighting on bat activity. Landsc. Urban Plan. 2018, 175, 123–135. [Google Scholar] [CrossRef]
- Straka, T.M.; Wolf, M.; Gras, P.; Buchholz, S.; Voigt, C.C. Tree Cover Mediates the Effect of Artificial Light on Urban Bats. Front. Ecol. Evol. 2019, 7. [Google Scholar] [CrossRef]
- Voigt, C.; Rehnig, K.; Lindecke, O.; Pētersons, G. Migratory bats are attracted by red light but not by warm-white light: Implications for the protection of nocturnal migrants. Ecol. Evol. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Bird, B.L.; Branch, L.; Miller, D.L. Effects of Coastal Lighting on Foraging Behavior of Beach Mice. Conserv. Biol. 2004, 18, 1435–1439. [Google Scholar] [CrossRef]
- Yalden, D. The food of the hedgehog in England. Acta Theriol. 1976, 21, 401–424. [Google Scholar] [CrossRef]
- Dickman, C. Age-related dietary change in the European hedgehog, Erinaceus europaeus. J. Zool. 1988, 215, 1–14. [Google Scholar] [CrossRef]
- Davies, T.W.; Bennie, J.; Gaston, K.J. Street lighting changes the composition of invertebrate communities. Biol. Lett. 2012, 8, 764–767. [Google Scholar] [CrossRef]
- Doncaster, C.P. Testing the role of intraguild predation in regulating hedgehog populations. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1992, 249, 113–117. [Google Scholar] [CrossRef]
- Young, R.P.; Davison, J.; Trewby, I.D.; Wilson, G.J.; Delahay, R.J.; Doncaster, C.P. Abundance of hedgehogs (Erinaceus europaeus) in relation to the density and distribution of badgers (Meles meles). J. Zool. 2006, 269, 349–356. [Google Scholar] [CrossRef]
- Dowding, C.V.; Harris, S.; Poulton, S.; Baker, P.J. Nocturnal ranging behaviour of urban hedgehogs, Erinaceus europaeus, in relation to risk and reward. Anim. Behav. 2010, 80, 13–21. [Google Scholar] [CrossRef]
- Rautio, A.; Valtonen, A.; Kunnasranta, M. The Effects of Sex and Season on Home Range in European Hedgehogs at the Northern Edge of the Species Range; Finnish Zoological and Botanical Publishing Board: Helsink, Finland, 2013; pp. 107–123. [Google Scholar]
- Raap, T.; Pinxten, R.; Eens, M. Artificial light at night disrupts sleep in female great tits (Parus major) during the nestling period, and is followed by a sleep rebound. Environ. Pollut. 2016, 215, 125–134. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finch, D.; Smith, B.R.; Marshall, C.; Coomber, F.G.; Kubasiewicz, L.M.; Anderson, M.; Wright, P.G.R.; Mathews, F. Effects of Artificial Light at Night (ALAN) on European Hedgehog Activity at Supplementary Feeding Stations. Animals 2020, 10, 768. https://doi.org/10.3390/ani10050768
Finch D, Smith BR, Marshall C, Coomber FG, Kubasiewicz LM, Anderson M, Wright PGR, Mathews F. Effects of Artificial Light at Night (ALAN) on European Hedgehog Activity at Supplementary Feeding Stations. Animals. 2020; 10(5):768. https://doi.org/10.3390/ani10050768
Chicago/Turabian StyleFinch, Domhnall, Bethany R. Smith, Charlotte Marshall, Frazer G. Coomber, Laura M. Kubasiewicz, Max Anderson, Patrick G. R. Wright, and Fiona Mathews. 2020. "Effects of Artificial Light at Night (ALAN) on European Hedgehog Activity at Supplementary Feeding Stations" Animals 10, no. 5: 768. https://doi.org/10.3390/ani10050768
APA StyleFinch, D., Smith, B. R., Marshall, C., Coomber, F. G., Kubasiewicz, L. M., Anderson, M., Wright, P. G. R., & Mathews, F. (2020). Effects of Artificial Light at Night (ALAN) on European Hedgehog Activity at Supplementary Feeding Stations. Animals, 10(5), 768. https://doi.org/10.3390/ani10050768





