A Genome-Wide Association Study for Calving Interval in Holstein Dairy Cows Using Weighted Single-Step Genomic BLUP Approach
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Phenotypic and Genotypic Data
2.2. Variance Components Estimation
2.3. Weighted Single-Step Genome-Wide Association Study
2.4. Gene Prospection
3. Results and Discussion
Variance Components and Accuracy of Genomic Predictions
4. Conclusion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ahlman, T.; Berglund, B.; Rydhmer, L.; Strandberg, E. Culling reasons in organic and conventional dairy herds and genotype by environment interaction for longevity. J. Dairy Sci. 2011, 94, 1568–1575. [Google Scholar] [CrossRef] [PubMed]
- Monti, G.; Tenhagen, B.; Heuwieser, W. Culling Policies in Dairy Herds. A. Review. J. Vet. Med. Ser. A 1999, 46, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.; Williams, E.; Evans, A. A review of the causes of poor fertility in high milk producing dairy cows. Anim. Reprod. Sci. 2011, 123, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Wang, Y.; Sahana, G.; Zhang, Q.; Liu, L.; Lund, M.S.; Su, G. Genome-wide association studies for female fertility traits in Chinese and Nordic Holsteins. Sci. Rep. 2017, 7, 8487. [Google Scholar] [CrossRef]
- Lucy, M. Reproductive loss in high-producing dairy cattle: Where will it end? J. Dairy Sci. 2001, 84, 1277–1293. [Google Scholar] [CrossRef]
- Nebel, R.; Dransfield, M.; Jobst, S.; Bame, J. Automated electronic systems for the detection of oestrus and timing of AI in cattle. Anim. Reprod. Sci. 2000, 60, 713–723. [Google Scholar] [CrossRef]
- Atashi, H.; Zamiri, M.J.; Sayyadnejad, M.B.; Akhlaghi, A. Trends in the reproductive performance of Holstein dairy cows in Iran. Trop. Anim. Health Prod. 2012, 44, 2001–2006. [Google Scholar] [CrossRef]
- Norman, H.; Wright, J.; Hubbard, S.; Miller, R.; Hutchison, J. Reproductive status of Holstein and Jersey cows in the United States. J. Dairy Sci. 2009, 92, 3517–3528. [Google Scholar] [CrossRef]
- Veerkamp, R.; Beerda, B. Genetics and genomics to improve fertility in high producing dairy cows. Theriogenology 2007, 68, S266–S273. [Google Scholar] [CrossRef]
- Pryce, J.; Coffey, M.; Brotherstone, S.; Woolliams, J. Genetic relationships between calving interval and body condition score conditional on milk yield. J. Dairy Sci. 2002, 85, 1590–1595. [Google Scholar] [CrossRef]
- Mee, J.F. Temporal trends in reproductive performance in Irish dairy herds and associated risk factors. Ir. Vet. J. 2004, 57, 158. [Google Scholar] [CrossRef] [PubMed]
- Hermans, K.; Opsomer, G.; Waegeman, W.; Moerman, S.; De Koster, J.; Van Eetvelde, M.; Van Ranst, B.; Hostens, M. Interpretation and visualisation of data from dairy herds. In Pract. 2018, 40, 195–203. [Google Scholar] [CrossRef]
- Lopez, B.I.; Son, J.-H.; Seo, K.; Lim, D. Estimation of Genetic Parameters for Reproductive Traits in Hanwoo (Korean Cattle). Animals 2019, 9, 715. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, M.; Buckley, F.; Evans, R.; Berry, D.P. Additive genetic, non-additive genetic and permanent environmental effects for female reproductive performance in seasonal calving dairy females. Ir. J. Agric. Food Res. 2016, 55, 10–23. [Google Scholar] [CrossRef][Green Version]
- Rydhmer, L.; Berglund, B. Selection for reproduction: Developments in several species. In Proceedings of the 8th World Congress on Genetics Applied to Livestock Production, Belo Horizonte, Minas Gerais, Brazil, 13–18 August 2006; p. 11-01. [Google Scholar]
- Berglund, B. Genetic improvement of dairy cow reproductive performance. Reprod. Domest. Anim. 2008, 43, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Minozzi, G.; Nicolazzi, E.L.; Stella, A.; Biffani, S.; Negrini, R.; Lazzari, B.; Ajmone-Marsan, P.; Williams, J.L. Genome wide analysis of fertility and production traits in Italian Holstein cattle. PLoS ONE 2013, 8, e80219. [Google Scholar] [CrossRef] [PubMed]
- Nayeri, S.; Sargolzaei, M.; Abo-Ismail, M.K.; May, N.; Miller, S.P.; Schenkel, F.; Moore, S.S.; Stothard, P. Genome-wide association for milk production and female fertility traits in Canadian dairy Holstein cattle. BMC Genet. 2016, 17, 75. [Google Scholar] [CrossRef]
- Frischknecht, M.; Bapst, B.; Seefried, F.R.; Signer-Hasler, H.; Garrick, D.; Stricker, C.; Fries, R.; Russ, I.; Sölkner, J.; Bieber, A. Genome-wide association studies of fertility and calving traits in Brown Swiss cattle using imputed whole-genome sequences. BMC Genom. 2017, 18, 910. [Google Scholar] [CrossRef]
- Aguilar, I.; Misztal, I.; Johnson, D.; Legarra, A.; Tsuruta, S.; Lawlor, T. Hot topic: A unified approach to utilize phenotypic, full pedigree, and genomic information for genetic evaluation of Holstein final score. J. Dairy Sci. 2010, 93, 743–752. [Google Scholar] [CrossRef]
- Christensen, O.F.; Lund, M.S. Genomic prediction when some animals are not genotyped. Genet. Sel. Evol. 2010, 42, 2. [Google Scholar] [CrossRef]
- Onogi, A.; Ogino, A.; Komatsu, T.; Shoji, N.; Shimizu, K.; Kurogi, K.; Yasumori, T.; Togashi, K.; Iwata, H. Whole-genome prediction of fatty acid composition in meat of Japanese Black cattle. Anim. Genet. 2015, 46, 557–559. [Google Scholar] [CrossRef] [PubMed]
- Matilainen, K.; Koivula, M.; Strandén, I.; Aamand, G.P.; Mäntysaari, E.A. Managing genetic groups in single-step genomic evaluations applied on female fertility traits in Nordic Red Dairy cattle. Interbull Bull. 2016, 50, 71–75. [Google Scholar]
- Wang, H.; Misztal, I.; Aguilar, I.; Legarra, A.; Muir, W. Genome-wide association mapping including phenotypes from relatives without genotypes. Genet. Res. 2012, 94, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Sargolzaei, M.; Chesnais, J.P.; Schenkel, F.S. A new approach for efficient genotype imputation using information from relatives. BMC Genom. 2014, 15, 478. [Google Scholar] [CrossRef]
- Misztal, I.; Tsuruta, S.; Strabel, T.; Auvray, B.; Druet, T.; Lee, D. BLUPF90 and related programs (BGF90). In Proceedings of the 7th world congress on genetics applied to livestock production, Montpellier, France, 19–23 August 2002; pp. 743–744. [Google Scholar]
- VanRaden, P.M. Efficient methods to compute genomic predictions. J. Dairy Sci. 2008, 91, 4414–4423. [Google Scholar] [CrossRef]
- Chen, E.Y.; Tan, C.M.; Kou, Y.; Duan, Q.; Wang, Z.; Meirelles, G.V.; Clark, N.R.; Ma’ayan, A. Enrichr: Interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 2013, 14, 128. [Google Scholar] [CrossRef]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S.L.; Jagodnik, K.M.; Lachmann, A. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef]
- Tenghe, A.; Bouwman, A.; Berglund, B.; Strandberg, E.; de Koning, D.; Veerkamp, R. Genome-wide association study for endocrine fertility traits using single nucleotide polymorphism arrays and sequence variants in dairy cattle. J. Dairy Sci. 2016, 99, 5470–5485. [Google Scholar] [CrossRef]
- Zaabza, H.B.; Gara, A.B.; Hammami, H.; Jemmali, B.; Ferchichi, M.A.; Rekik, B. Genetic parameters of reproductive traits in Tunisian Holsteins. Arch. Anim. Breed. (Arch. Tierz.) 2016, 59, 209–213. [Google Scholar] [CrossRef]
- Albarrán-Portillo, B.; Pollott, G. The relationship between fertility and lactation characteristics in Holstein cows on United Kingdom commercial dairy farms. J. Dairy Sci. 2013, 96, 635–646. [Google Scholar] [CrossRef]
- Hossein-Zadeh, N.G. Genetic parameters and trends for calving interval in the first three lactations of Iranian Holsteins. Trop. Anim. Health Prod. 2011, 43, 1111–1115. [Google Scholar] [CrossRef] [PubMed]
- Miglior, F.; Fleming, A.; Malchiodi, F.; Brito, L.F.; Martin, P.; Baes, C.F. A 100-Year Review: Identification and genetic selection of economically important traits in dairy cattle. J. Dairy Sci. 2017, 100, 10251–10271. [Google Scholar] [CrossRef] [PubMed]
- Teissier, M.; Larroque, H.; Robert-Granié, C. Weighted single-step genomic BLUP improves accuracy of genomic breeding values for protein content in French dairy goats: A quantitative trait influenced by a major gene. Genet. Sel. Evol. 2018, 50, 31. [Google Scholar] [CrossRef] [PubMed]
- Teissier, M.; Larroque, H.; Robert-Granie, C. Accuracy of genomic evaluation with weighted single-step genomic best linear unbiased prediction for milk production traits, udder type traits, and somatic cell scores in French dairy goats. J. Dairy Sci. 2019, 102, 3142–3154. [Google Scholar] [CrossRef]
- Wang, H.; Misztal, I.; Aguilar, I.; Legarra, A.; Fernando, R.L.; Vitezica, Z.; Okimoto, R.; Wing, T.; Hawken, R.; Muir, W.M. Genome-wide association mapping including phenotypes from relatives without genotypes in a single-step (ssGWAS) for 6-week body weight in broiler chickens. Front. Genet. 2014, 5, 134. [Google Scholar] [CrossRef]
- Zhang, X.; Lourenco, D.; Aguilar, I.; Legarra, A.; Misztal, I. Weighting strategies for single-step genomic BLUP: An iterative approach for accurate calculation of GEBV and GWAS. Front. Genet. 2016, 7, 151. [Google Scholar] [CrossRef]
- Aguilar, I.; Legarra, A.; Cardoso, F.; Masuda, Y.; Lourenco, D.; Misztal, I. Frequentist p-values for large-scale-single step genome-wide association, with an application to birth weight in American Angus cattle. Genet. Sel. Evol. 2019, 51, 28. [Google Scholar] [CrossRef]
- Myles, S.; Tang, K.; Somel, M.; Green, R.E.; Kelso, J.; Stoneking, M. Identification and analysis of genomic regions with large between-population differentiation in humans. Ann. Hum. Genet. 2008, 72, 99–110. [Google Scholar] [CrossRef]
- Beissinger, T.M.; Rosa, G.J.; Kaeppler, S.M.; Gianola, D.; De Leon, N. Defining window-boundaries for genomic analyses using smoothing spline techniques. Genet. Sel. Evol. 2015, 47, 30. [Google Scholar] [CrossRef]
- Fragomeni, B.d.O.; Misztal, I.; Lourenco, D.L.; Aguilar, I.; Okimoto, R.; Muir, W.M. Changes in variance explained by top SNP windows over generations for three traits in broiler chicken. Front. Genet. 2014, 5, 332. [Google Scholar] [CrossRef]
- Braz, C.U.; Taylor, J.F.; Bresolin, T.; Espigolan, R.; Feitosa, F.L.; Carvalheiro, R.; Baldi, F.; Lucia, G.; de Oliveira, H.N. Sliding window haplotype approaches overcome single SNP analysis limitations in identifying genes for meat tenderness in Nelore cattle. BMC Genet. 2019, 20, 8. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Li, J.; Bonham, A.J.; Wang, Y.; Deng, H. Gains in power for exhaustive analyses of haplotypes using variable-sized sliding window strategy: A comparison of association-mapping strategies. Eur. J. Hum. Genet. 2009, 17, 785. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tang, R.; Feng, T.; Sha, Q.; Zhang, S. A variable-sized sliding-window approach for genetic association studies via principal component analysis. Ann. Hum. Genet. 2009, 73, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Peñagaricano, F. Unravelling the genomic architecture of bull fertility in Holstein cattle. BMC Genet. 2016, 17, 143. [Google Scholar] [CrossRef]
- Suwannasing, R.; Duangjinda, M.; Boonkum, W.; Taharnklaew, R.; Tuangsithtanon, K. The identification of novel regions for reproduction trait in Landrace and Large White pigs using a single step genome-wide association study. Asian Australas. J. Anim. Sci. 2018, 31, 1852. [Google Scholar] [CrossRef]
- de Oliveira Silva, R.M.; Stafuzza, N.B.; de Oliveira Fragomeni, B.; de Camargo, G.M.F.; Ceacero, T.M.; Cyrillo, J.N.d.S.G.; Baldi, F.; Boligon, A.A.; Mercadante, M.E.Z.; Lourenco, D.L. Genome-wide association study for carcass traits in an experimental Nelore cattle population. PLoS ONE 2017, 12, e0169860. [Google Scholar] [CrossRef]
- Zhou, C.; Li, C.; Cai, W.; Liu, S.; Yin, H.W.; Shi, S.; Zhang, Q.; Zhang, S. Genome-wide association study for milk protein composition traits in a Chinese Holstein population using a single-step approach. Front. Genet. 2019, 10, 72. [Google Scholar] [CrossRef]
- Seidenspinner, T.; Bennewitz, J.; Reinhardt, F.; Thaller, G. Need for sharp phenotypes in QTL detection for calving traits in dairy cattle. J. Anim. Breed. Genet. 2009, 126, 455–462. [Google Scholar] [CrossRef]
- McClure, M.; Morsci, N.; Schnabel, R.; Kim, J.; Yao, P.; Rolf, M.; McKay, S.; Gregg, S.; Chapple, R.; Northcutt, S. A genome scan for quantitative trait loci influencing carcass, post-natal growth and reproductive traits in commercial Angus cattle. Anim. Genet. 2010, 41, 597–607. [Google Scholar] [CrossRef]
- Rodriguez-Zas, S.L.; Southey, B.; Heyen, D.; Lewin, H. Interval and composite interval mapping of somatic cell score, yield, and components of milk in dairy cattle. J. Dairy Sci. 2002, 85, 3081–3091. [Google Scholar] [CrossRef]
- Sherman, E.; Nkrumah, J.; Li, C.; Bartusiak, R.; Murdoch, B.; Moore, S. Fine mapping quantitative trait loci for feed intake and feed efficiency in beef cattle. J. Anim. Sci. 2009, 87, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Van Winkle, L.J. Amino acid transport regulation and early embryo development. Biol. Reprod. 2001, 64, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Van Winkle, L.J.; Campione, A.L. Amino acid transport regulation in preimplantation mouse embryos: Effects on amino acid content and pre-and peri-implantation development. Theriogenology 1996, 45, 69–80. [Google Scholar] [CrossRef]
- Van Winkle, L.J.; Dickinson, H.R. Differences in amino acid content of preimplantation mouse embryos that develop In Vitro versus In Vivo: In Vitro effects of five amino acids that are abundant in oviductal secretions. Biol. Reprod. 1995, 52, 96–104. [Google Scholar] [CrossRef]
- Van Winkle, L.J.; Christensen, H.; Campione, A. Na+-dependent transport of basic, zwitterionic, and bicyclic amino acids by a broad-scope system in mouse blastocysts. J. Biol. Chem. 1985, 260, 12118–12123. [Google Scholar]
- Lane, M.; Gardner, D. Differential regulation of mouse embryo development and viability by amino acids. Reproduction 1997, 109, 153–164. [Google Scholar] [CrossRef]
- Moore, K.; Bondioli, K.R. Glycine and alanine supplementation of culture medium enhances development of in vitro matured and fertilized cattle embryos. Biol. Reprod. 1993, 48, 833–840. [Google Scholar] [CrossRef]
- Elhassan, Y.; Wu, G.; Leanez, A.; Tasca, R.; Watson, A.; Westhusin, M. Amino acid concentrations in fluids from the bovine oviduct and uterus and in KSOM-based culture media. Theriogenology 2001, 55, 1907–1918. [Google Scholar] [CrossRef]
- Morris, D.G.; Grealy, M.; Leese, H.; Diskin, M.G.; Sreenan, J. Cattle Embryo Growth Development and Viability; Teagasc: Dublin, Ireland, 2001. [Google Scholar]
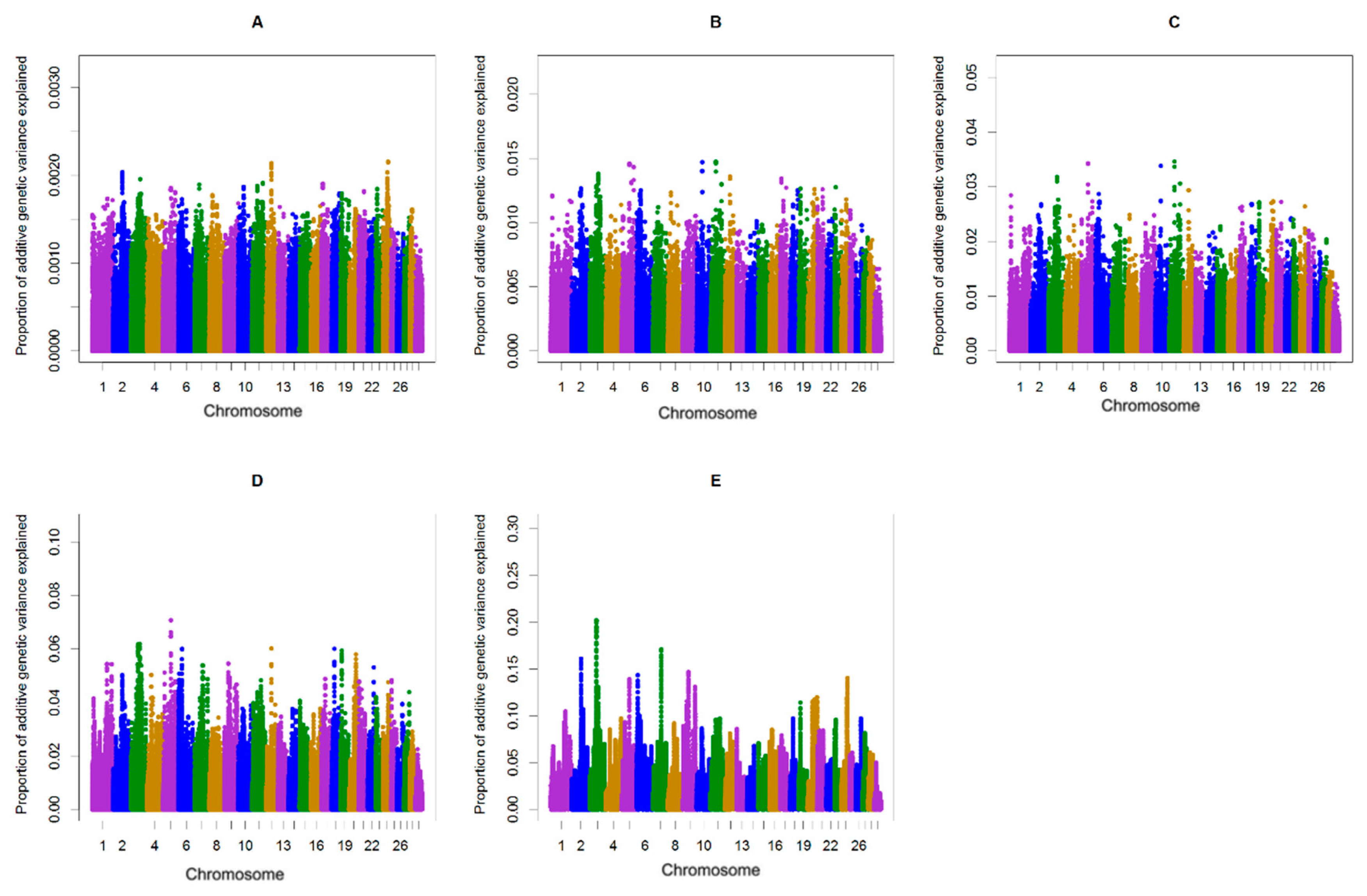
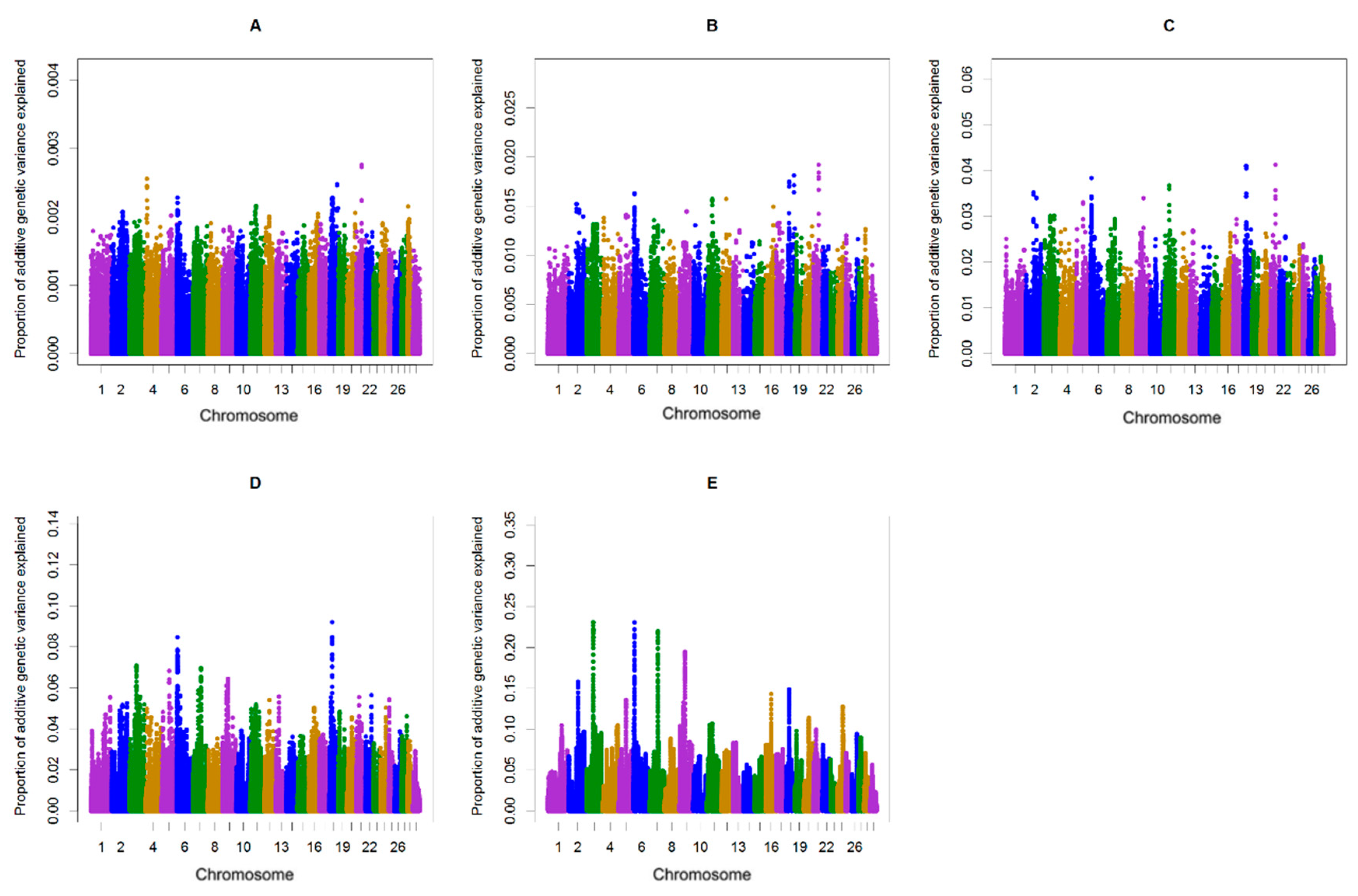
| Chromosome | Position | Genes 1 | The Proportion of Genetic Variance Explained in Primi- and Multiparous Cows |
|---|---|---|---|
| BTA3 | 49426411–49528260 | ARHGAP29 | 0.20 and 0.23 |
| BTA6 | 7734092–7861603 | SEC24D, METTL14 | 0.14 and 0.23 |
| BTA7 | 64522263–64602968 | SLC36A2, SLC36A3 | 0.17 and 0.22 |
| GO Term Description | Genes |
|---|---|
| L-alanine transport (GO:0015808) | SLC36A2, SLC36A3 |
| alanine transport (GO:0032328) | SLC36A2, SLC36A3 |
| proline transport (GO:0015824) | SLC36A2, SLC36A3 |
| glycine transport (GO:0015816) | SLC36A2, SLC36A3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atashi, H.; Salavati, M.; De Koster, J.; Crowe, M.A.; Opsomer, G.; Hostens, M.; The GplusE Consortium. A Genome-Wide Association Study for Calving Interval in Holstein Dairy Cows Using Weighted Single-Step Genomic BLUP Approach. Animals 2020, 10, 500. https://doi.org/10.3390/ani10030500
Atashi H, Salavati M, De Koster J, Crowe MA, Opsomer G, Hostens M, The GplusE Consortium. A Genome-Wide Association Study for Calving Interval in Holstein Dairy Cows Using Weighted Single-Step Genomic BLUP Approach. Animals. 2020; 10(3):500. https://doi.org/10.3390/ani10030500
Chicago/Turabian StyleAtashi, Hadi, Mazdak Salavati, Jenne De Koster, Mark A. Crowe, Geert Opsomer, Miel Hostens, and The GplusE Consortium. 2020. "A Genome-Wide Association Study for Calving Interval in Holstein Dairy Cows Using Weighted Single-Step Genomic BLUP Approach" Animals 10, no. 3: 500. https://doi.org/10.3390/ani10030500
APA StyleAtashi, H., Salavati, M., De Koster, J., Crowe, M. A., Opsomer, G., Hostens, M., & The GplusE Consortium. (2020). A Genome-Wide Association Study for Calving Interval in Holstein Dairy Cows Using Weighted Single-Step Genomic BLUP Approach. Animals, 10(3), 500. https://doi.org/10.3390/ani10030500






