Leptin System in Obese Dog Skin: A Pilot Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Complete Blood Count (CBC) and Serum Biochemistry
2.2. Sample Collection
2.3. Immunohistochemistry
2.4. RNA Extraction and Real-Time PCR
2.5. Statistical Analysis
3. Results
3.1. Hemato-Chemical Parameters
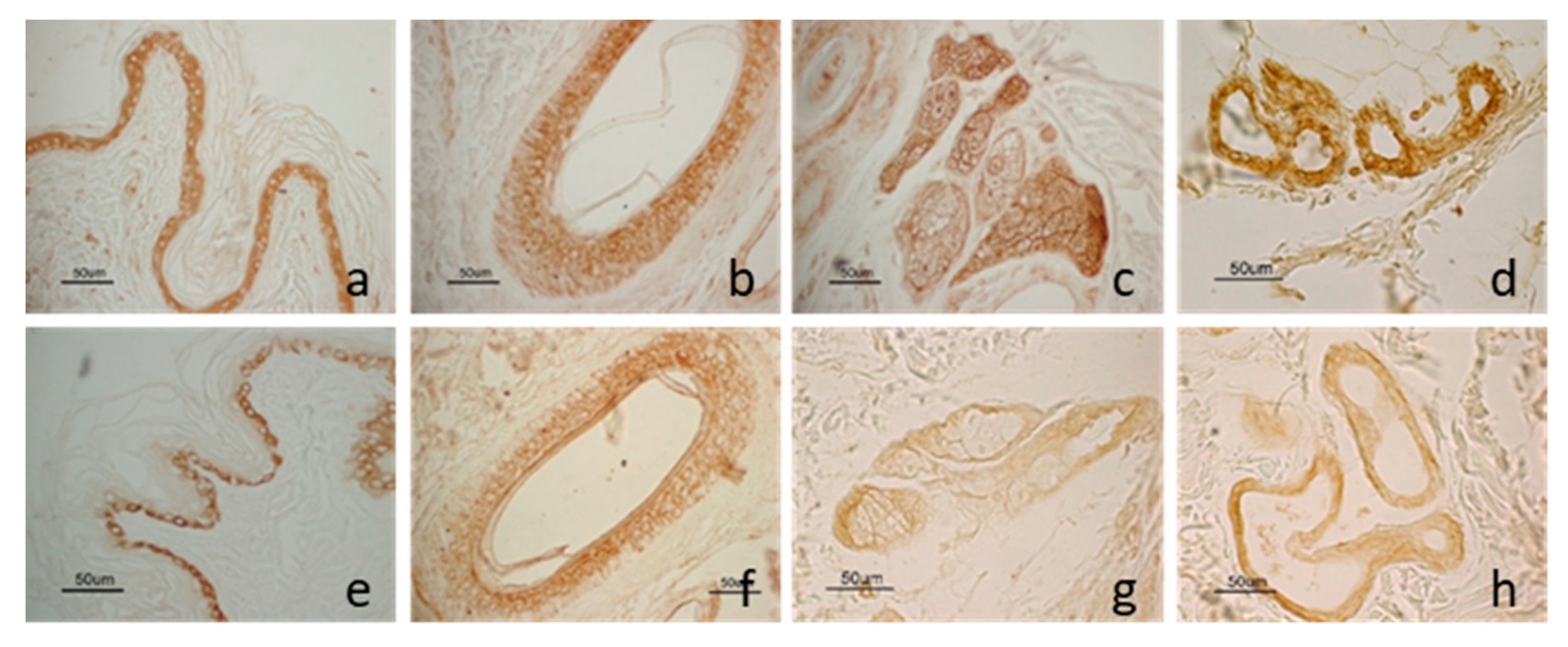
3.2. Immunohistochemistry
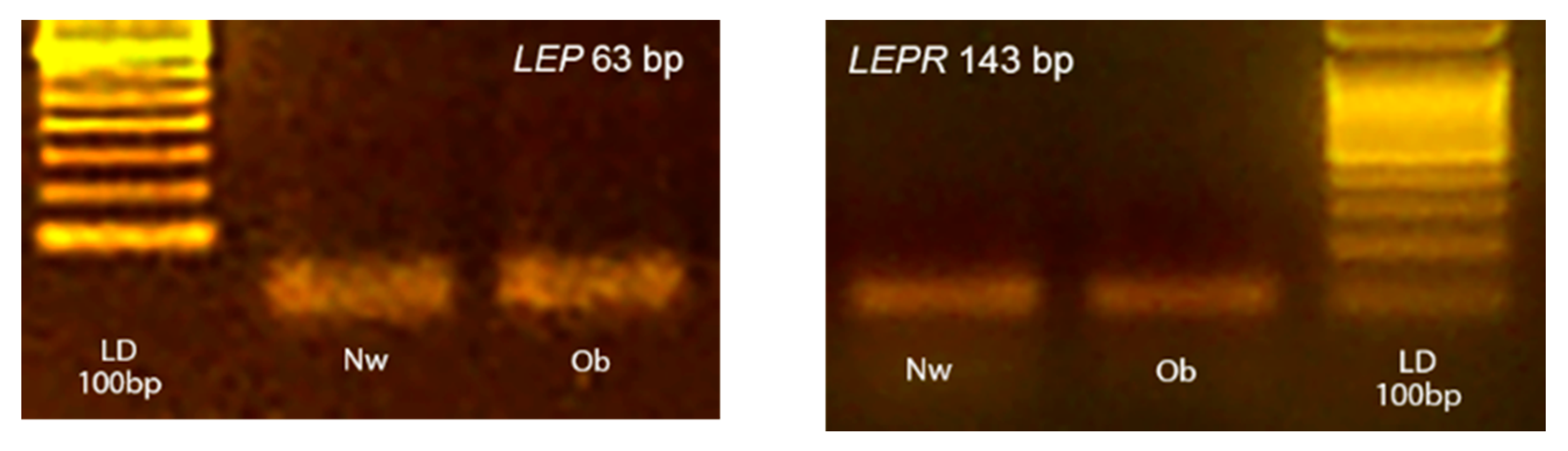
3.3. LEP and LEPR Gene Expression by Real-Time PCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chandler, M.; Cunningham, S.; Lund, E.; Khanna, C.; Naramore, R.; Patel, A.; Day, M.J. Obesity and Associated Comorbidities in People and Companion Animals: A One Health Perspective. J. Comp. Pathol. 2017, 156, 296–309. [Google Scholar] [CrossRef]
- Laflamme, D. Companion animals symposium: Obesity in dogs and cats: What is wrong with being fat? J. Anim. Sci. 2012, 90, 1653–1662. [Google Scholar] [CrossRef] [PubMed]
- German, A.J. The Growing Problem of Obesity in Dogs and Cats. J. Nutr. 2006, 136, 1940S–1946S. [Google Scholar] [CrossRef] [PubMed]
- German, A.J.; Ryan, V.H.; German, A.C.; Wood, I.S.; Trayhurn, P. Obesity, its associated disorders and the role of inflammatory adipokines in companion animals. Veter J. 2010, 185, 4–9. [Google Scholar] [CrossRef]
- Muñoz-Prieto, A.; Nielsen, L.R.; Dąbrowski, R.; Bjørnvad, C.R.; Söder, J.; Lamy, E.; Monkeviciene, I.; Ljubić, B.B.; Vasiu, I.; Savic, S.; et al. European dog owner perceptions of obesity and factors associated with human and canine obesity. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- McGreevy, P.D.; Thomson, P.C.; Pride, C.; Fawcett, A.; Grassi, T.; Jones, B. Prevalence of obesity in dogs examined by Australian veterinary practices and the risk factors involved. Veter Rec. 2005, 156, 695–702. [Google Scholar] [CrossRef]
- Tvarijonaviciute, A.; Cerón, J.J.; Holden, S.L.; Cuthbertson, D.J.; Biourge, V.C.; Morris, P.J.; German, A.J. Obesity-related metabolic dysfunction in dogs: A comparison with human metabolic syndrome. BMC Veter Res. 2012, 8, 147. [Google Scholar] [CrossRef]
- German, A.J.; Hervera, M.; Hunter, L.; Holden, S.L.; Morris, P.J.; Biourge, V.; Trayhurn, P. Improvement in insulin resistance and reduction in plasma inflammatory cytokines after weight loss in obese dogs. Domest. Anim. Endocrinol. 2009, 37, 214–226. [Google Scholar] [CrossRef]
- Piantedosi, D.; Di Loria, A.; Guccione, J.; De Rosa, A.; Fabbri, S.; Cortese, L.; Carta, S.; Ciaramella, P. Serum biochemistry profile, inflammatory cytokines, adipokines and cardiovascular findings in obese dogs. Veter J. 2016, 216, 72–78. [Google Scholar] [CrossRef]
- Trayhurn, P.; Bing, C.; Wood, I.S. Adipose Tissue and Adipokines—Energy Regulation from the Human Perspective. J. Nutr. 2006, 136, 1935S–1939S. [Google Scholar] [CrossRef]
- Wozniak, S.E.; Gee, L.L.; Wachtel, M.S.; Frezza, E.E. Adipose Tissue: The New Endocrine Organ? A Review Article. Dig. Dis. Sci. 2009, 54, 1847–1856. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Is obesity an inflammatory condition? Nutrition 2001, 17, 953–966. [Google Scholar] [CrossRef]
- Münzberg, H.; Morrison, C.D. Structure, production and signaling of leptin. Metabolism 2015, 64, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chua, S. Leptin Function and Regulation. Compr. Physiol. 2017, 8, 351–369. [Google Scholar] [CrossRef] [PubMed]
- Frühbeck, G. Intracellular signalling pathways activated by leptin. Biochem. J. 2006, 393, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Lago, F.; Diéguez, C.; Gómez-Reino, J.; Gualillo, O. The emerging role of adipokines as mediators of inflammation and immune responses. Cytokine Growth Factor Rev. 2007, 18, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Tadokoro, S.; Ide, S.; Tokuyama, R.; Umeki, H.; Tatehara, S.; Kataoka, S.; Satomura, K. Leptin Promotes Wound Healing in the Skin. PLoS ONE 2015, 10, e0121242. [Google Scholar] [CrossRef] [PubMed]
- Mercati, F.; Dall’Aglio, C.; Timperi, L.; Scocco, P.; De Felice, E.; Maranesi, M. Epithelial expression of the hormone leptin by bovine skin. Eur. J. Histochem. 2019, 63. [Google Scholar] [CrossRef]
- Mercati, F.; Maranesi, M.; Dall’Aglio, C.; Scocco, P.; Pascucci, L.; Boiti, C.; Ceccarelli, P. Leptin receptor is expressed by epidermis and skin appendages in dog. Acta Histochem. 2014, 116, 1270–1275. [Google Scholar] [CrossRef]
- Poeggeler, B.; Schulz, C.; Pappolla, M.A.; Bodã, E.; Tiede, S.; Lehnert, H.; Paus, R. Leptin and the skin: A new frontier. Exp. Dermatol. 2010, 19, 12–18. [Google Scholar] [CrossRef]
- Frank, S.; Stallmeyer, B.; Kämpfer, H.; Kolb, N.; Pfeilschifter, J. Leptin enhances wound re-epithelialization and constitutes a direct function of leptin in skin repair. J. Clin. Investig. 2000, 106, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Murad, A.; Nath, A.K.; Cha, S.-T.; Demir, E.; Floras-Riveras, J.; Sierra-Honigmann, M.R. Leptin is an autocrine/paracrine regulator of wound healing. FASEB J. 2003, 17, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Çerman, A.; Bozkurt, S.; Sav, A.; Tulunay, A.; Elbaşı, M.; Ergun, T. Serum leptin levels, skin leptin and leptin receptor expression in psoriasis. Br. J. Dermatol. 2008, 159, 820–826. [Google Scholar] [CrossRef] [PubMed]
- Guida, B.; Nino, M.; Perrino, N.R.; Laccetti, R.; Trio, R.; Labella, S.; Balato, N. The impact of obesity on skin disease and epidermal permeability barrier status. J. Eur. Acad. Dermatol. Venereol. 2010, 24, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Shipman, A.; Millington, G. Obesity and the skin. Br. J. Dermatol. 2011, 165, 743–750. [Google Scholar] [CrossRef]
- Raffan, E. The big problem: Battling companion animal obesity. Veter Rec. 2013, 173, 287–291. [Google Scholar] [CrossRef]
- Lund, E.M.; Armstrong, P.J.; Kirk, C.A.; Klausner, J.S. Prevalence and Risk Factors for Obesity in Adult Dogs from Private US Veterinary Practices. Intern. J. Appl Res. Vet. Med. 2006, 4, 177–186. [Google Scholar]
- Brément, T.; Cossec, C.; Roux, C.; Knol, A.; Dréno, B.; Khammari, A.; Bourdeau, P.; Bruet, V. Expression of Three Adipokines (Adiponectin, Leptin and Resistin) in Normal Canine Skin: A Pilot Study. J. Comp. Pathol. 2019, 167, 82–90. [Google Scholar] [CrossRef]
- Laflamme, D.P. Development and validation of a body condition score system for dogs. Canine Pract. 1997, 22, 10–15. [Google Scholar]
- Mercati, F.; Scocco, P.; Maranesi, M.; Acuti, G.; Petrucci, L.; Cocci, P.; Renzi, A.; De Felice, E.; Dall’Aglio, C. Apelin system detection in the reproductive apparatus of ewes grazing on semi-natural pasture. Theriogenology 2019, 139, 156–166. [Google Scholar] [CrossRef]
- Dall’Aglio, C.; Scocco, P.; Maranesi, M.; Petrucci, L.; Acuti, G.; De Felice, E.; Mercati, F. Immunohistochemical identification of resistin in the uterus of ewes subjected to different diets: Preliminary results. Eur. J. Histochem. 2019, 63, 3020. [Google Scholar] [CrossRef] [PubMed]
- Porcellato, I.; Silvestri, S.; Menchetti, L.; Dvm, F.R.; Mechelli, L.; Sforna, M.; Iussich, S.; Bongiovanni, L.; Lepri, E.; Brachelente, C. Tumour-infiltrating lymphocytes in canine melanocytic tumours: An investigation on the prognostic role of CD3 + and CD20 + lymphocytic populations. Veter Comp. Oncol. 2019, 18, 370–380. [Google Scholar] [CrossRef]
- Maranesi, M.; Petrucci, L.; Leonardi, L.; Piro, F.; Rebollar, P.G.; Millán, P.; Cocci, P.; Vullo, C.; Parillo, F.; Moura, A.; et al. New insights on aNGF-mediated pathway to induce ovulation in rabbits (Oryctolagus cuniculus). Biol. Reprod. 2018, 98, 634–643. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Mercati, F.; Maranesi, M.; Dall’Aglio, C.; Petrucci, L.; Pasquariello, R.; Tardella, F.M.; De Felice, E.; Scocco, P. Apelin System in Mammary Gland of Sheep Reared in Semi-Natural Pastures of the Central Apennines. Animals 2018, 8, 223. [Google Scholar] [CrossRef]
- Guelfi, G.; Zerani, M.; Brecchia, G.; Parillo, F.; Dall’Aglio, C.; Maranesi, M.; Boiti, C. Direct actions of ACTH on ovarian function of pseudopregnant rabbits. Mol. Cell. Endocrinol. 2011, 339, 63–71. [Google Scholar] [CrossRef][Green Version]
- Rizzi, T.E.; Meinkoth, J.H.; Clinkenbeard, K.D. Shalms Veterinary Hematology, 6th ed.; Weiss, D.J., Wardrop, K.J., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2010; p. 802. [Google Scholar]
- Kaneko, J.J.; Harvey, J.W.; Bruss, M.L. Textbook of Clinical Biochemistry of Domestic Animals, 6th ed.; Elsevier: St. Louis, MO, USA, 2008; pp. 889–895. [Google Scholar]
- Cortese, L.; Terrazzano, G.; Pelagalli, A. Leptin and Immunological Profile in Obesity and Its Associated Diseases in Dogs. Int. J. Mol. Sci. 2019, 20, 2392. [Google Scholar] [CrossRef]
- Di Loria, A.; Squillacioti, C.; De Luca, A.; Veneziano, V.; Mirabella, N.; Guccione, J.; Santoro, D. Increased leptin mRNA expression in the blood of dogs naturally infected by Leishmania infantum. Veter J. 2014, 202, 634–636. [Google Scholar] [CrossRef]
- Ishioka, K.; Hosoya, K.; Kitagawa, H.; Shibata, H.; Honjoh, T.; Kimura, K.; Saito, M. Plasma leptin concentration in dogs: Effects of body condition score, age, gender and breeds. Res. Veter Sci. 2007, 82, 11–15. [Google Scholar] [CrossRef]
- Jeusette, I.J.; Detilleux, J.D.; Shibata, H.; Saito, M.; Honjoh, T.; Delobel, A.; Istasse, L.; Diez, M. Effects of chronic obesity and weight loss on plasma gherlin and leptin concentrations in dogs. Res. Vet. Sci. 2005, 79, 169–175. [Google Scholar] [CrossRef]
- Maury, E.; Brichard, S.M. Adipokine dysregulation, adipose tissue inflammation and metabolic syndrome. Mol. Cell. Endocrinol. 2010, 314, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Tvarijonaviciute, A.; Tecles, F.; Martínez-Subiela, S.; Cerón, J.J. Effect of weight loss on inflammatory biomarkers in obese dogs. Veter J. 2012, 193, 570–572. [Google Scholar] [CrossRef] [PubMed]
- Yamka, R.M.; Friesen, K.G.; Frantz, N.Z. Identification of canine markers related to obesity and the effects of weight loss on the markers of interest. Int. J. Appl. Res. Vet. 2006, 4, 282–292. [Google Scholar]
- Tribuddharatana, T.; Kongpiromchean, Y.; Sribhen, K.; Sribhen, C. Biochemical alterations and their relationships with the metabolic syndrome components in canine obesity. Nat. Sci. 2011, 45, 622–628. [Google Scholar]
- Center, S.A. Interpretation of Liver Enzymes. Veter Clin. N. Am. Small Anim. Pr. 2007, 37, 297–333. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Keogh, J. Metabolic effects of high-protein diets. Curr. Atheroscler. Rep. 2007, 9, 472–478. [Google Scholar] [CrossRef]
- Blüher, M. Metabolically Healthy Obesity. Endocr. Rev. 2020, 41, 405–420. [Google Scholar] [CrossRef]
- Carmina, E.; Chu, M.C.; Moran, C.; Tortoriello, D.; Vardhana, P.; Tena, G.; Preciado, R.; Lobo, R. Subcutaneous and omental fat expression of adiponectin and leptin in women with polycystic ovary syndrome. Fertil. Steril. 2008, 89, 642–648. [Google Scholar] [CrossRef]
- Collares, R.V.A.; Junior, W.S.; Tirapelli, D.P.D.C.; Dos Santos, J.S. The Expression of LEP, LEPR, IGF1 and IL10 in Obesity and the Relationship with microRNAs. PLoS ONE 2014, 9, e93512. [Google Scholar] [CrossRef]
- Sabol, F.; Bobrov, N.; Novotný, M.; Vasilenko, T.; Mozeš, Š.; Šefčíková, Z.; Molčányiová, A.; Tkacova, R.; Tomori, Z.; Gál, P. Skin wound healing in obese and lean male adolescent rats submitted to pre-weaning litter size manipulation. Folia Biol. 2014, 60, 21–27. [Google Scholar]
- Otero, M.; Lago, R.; Gomez, R.; Dieguez, C.; Lago, F.; Gomez-Reino, J.; Gualillo, O. Towards a pro-inflammatory and immunomodulatory emerging role of leptin. Rheumatology 2006, 45, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Bremmer, S.; Van Voorhees, A.S.; Hsu, S.; Korman, N.J.; Lebwohl, M.G.; Young, M.; Bebo, B.F.; Blauvelt, A. Obesity and psoriasis: From the Medical Board of the National Psoriasis Foundation. J. Am. Acad. Dermatol. 2010, 63, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Pierezan, F.; Mansell, J.; Ambrus, A.; Hoffmann, A.R. Immunohistochemical Expression of Ionized Calcium Binding Adapter Molecule 1 in Cutaneous Histiocytic Proliferative, Neoplastic and Inflammatory Disorders of Dogs and Cats. J. Comp. Pathol. 2014, 151, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Palatucci, A.T.; Piantedosi, D.; Rubino, V.; Giovazzino, A.; Guccione, J.; Pernice, V.; Ruggiero, G.; Cortese, L.; Terrazzano, G. Circulating regulatory T cells (Treg), leptin and induction of proinflammatory activity in obese Labrador Retriever dogs. Veter Immunol. Immunopathol. 2018, 202, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.C.; Smith, S.J.; Tow, B.; Elias, P.M.; Farese, R.V., Jr. Leptin modulates the effects of acyl CoA: Diacylglycerol acyltransferase deficiency on murine fur and sebaceous glands. J. Clin. Investig. 2002, 109, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Löffler, J.A.H.; Aramaki, J.U.N.; Effendy, I. The influence of body mass index on skin susceptibility to sodium lauryl sulphate. Ski. Res. Technol. 2002, 8, 19–22. [Google Scholar] [CrossRef]
- Campbell, K.L.; Lichtensteiger, C.A. Structure and Function of the Skin in Small Animal Dermatology Secrets, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2004; pp. 1–9. [Google Scholar]
- Frank, L.A. Comparative dermatology—Canine endocrine dermatoses. Clin. Dermatol. 2006, 24, 317–325. [Google Scholar] [CrossRef]
- Dopytalska, K.; Baranowska-Bik, A.; Roszkiewicz, M.; Bik, W.; Walecka, I. The role of leptin in selected skin diseases. Lipids Health Dis. 2020, 19, 1–10. [Google Scholar] [CrossRef]
- Arnold, T.; Brandlhofer, S.; Vrtikapa, K.; Stangl, H.; Hermann, M.; Zwiauer, K.; Mangge, H.; Karwautz, A.; Huemer, J.; Koller, D.; et al. Effect of Obesity on Plasma Clusterin: A Proposed Modulator of Leptin Action. Pediatr. Res. 2011, 69, 237–242. [Google Scholar] [CrossRef]
- Liu, Z.-J.; Bian, J.; Liu, J.; Endoh, A. Obesity Reduced the Gene Expressions of Leptin Receptors in Hypothalamus and Liver. Horm. Metab. Res. 2007, 39, 489–494. [Google Scholar] [CrossRef]
- Haltiner, A.L.; Mitchell, T.D.; Harris, R.B.S. Leptin action is modified by an interaction between dietary fat content and ambient temperature. Am. J. Physiol. Integr. Comp. Physiol. 2004, 287, R1250–R1255. [Google Scholar] [CrossRef] [PubMed]
- Münzberg, H.; Flier, J.S.; Bjørbaek, C. Region-specific leptin resistance within the hypothalamus of diet-induced obese mice. Endocrinology 2004, 145, 4880–4889. [Google Scholar] [CrossRef] [PubMed]
- Enriori, P.J.; Evans, A.E.; Sinnayah, P.; Jobst, E.E.; Tonelli-Lemos, L.; Billes, S.K.; Glavas, M.M.; Grayson, B.E.; Perello, M.; Nillni, E.A.; et al. Diet-Induced Obesity Causes Severe but Reversible Leptin Resistance in Arcuate Melanocortin Neurons. Cell Metab. 2007, 5, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Priego, T.; Sánchez, J.; Palou, A.; Picó, C. Effect of high-fat diet feeding on leptin receptor expression in white adipose tissue in rats: Depot- and sex-related differential response. Genes Nutr. 2009, 4, 151–156. [Google Scholar] [CrossRef]
- Considine, R.V.; Considine, E.L.; Williams, C.J.; Hyde, T.M.; Caro, J.F. The hypothalamic leptin receptor in humans: Identification of incidental sequence polymorphisms and absence of the db/db mouse and fa/fa rat mutations. Diabetes 1996, 45, 992–994. [Google Scholar] [CrossRef]
| Antisera | Working Dilutions | Sources |
|---|---|---|
| normal goat serum | 1:10 | S-1000, Vector Laboratories, Burlingame, CA, USA |
| normal horse serum | 1:10 | S-2000, Vector Laboratories, Burlingame, CA, USA |
| polyclonal rabbit anti-LEP | 1:100 | abx177326, Abbexa Ltd., Cambridge, UK |
| polyclonal goat anti LEPR | 1:200 | ab 50424, Abcam, Cambridge, UK |
| polyclonal rabbit anti-CD20 | 1:200 | RB9013-P; ThermoScientific, Waltham, MA, USA |
| polyclonal rabbit Anti-CD3 | 1:200 | A0452; DakoCytomation, Carpentera, CA, USA |
| monoclonal mouse anti-Iba-1 | 1:50 | MABN92; Merck Millipore, Damstadt, Germany |
| horse anti rabbit IgG biotin conjugated | 1:200 | BA-1100; Vector Laboratories, Burlingame, CA, USA |
| horse anti-goat IgG biotin conjugated | 1:200 | BA-9500; Vector Laboratories, Burlingame, CA, USA |
| goat anti-mouse IgG biotin conjugated | 1:200 | Ab6788 Abcam, Cambridge, UK |
| Gene | NCBI seq, ref. | Primers | |
|---|---|---|---|
| LEP | NM_001003070.1 | F | ACCGTATGGGTGTCCTTTATCCT |
| R | AGAGTGGCTCTGTGGTGTGAGA | ||
| LEPR [19] | NM_001024634.1 | F | CTTTTGCCTGCTGGAATCTC |
| R | TTGCTCCAAAAGCAACAGTG | ||
| BACT | NM_001195845.2 | F | CTTCCAGCCTTCCTTCCTGG |
| R | CCAGGGTACATGGTGGTTCC |
| Parameter | Unit | Reference Ranges | Ob Group | Nw Group | p |
|---|---|---|---|---|---|
| RBCs | (×106/µL) | 5.5–8.5 | 7.7 ± 0.57 | 7.6 ± 0.36 | 0.255 |
| HGB | g/dL | 12–18 | 16.8 ± 0.63 | 17.2 ± 0.49 | 0.061 |
| HCT | % | 37–55 | 52.1 ± 1.94 | 52.0 ± 2.08 | 0.456 |
| MCV | fL | 60–77 | 67.9 ± 6.92 | 68.8 ± 4.74 | 0.370 |
| MCH | pg | 20.5–24.2 | 21.8 ± 2.30 | 22.7 ± 1.36 | 0.162 |
| MCHC | % | 32–36 | 32.1 ± 1.25 | 33.0 ± 1.85 | 0.113 |
| WBCs | (×103/µL) | 6–17 | 10.7 ± 2.63 | 11.8 ± 1.70 | 0.151 |
| PLTs | (×103/µL) | 200–500 | 354.3 ± 78.96 | 311.8 ± 70.60 | 0.110 |
| Parameter | Unit | Reference Ranges | Ob Group | Nw Group | p |
|---|---|---|---|---|---|
| Glucose | mg/dL | 65–118 | 82.6 ± 8.41 | 77.3 ± 10.14 | 0.110 |
| Urea | mg/dL | 21–59 | 41.5 ± 11.15 | 37.7 ± 7.56 | 0.192 |
| Creatinine | mg/dL | 0.5–1.5 | 1.4 ± 0.2 | 1.3 ± 0.14 | 0.067 |
| T-Chol | mg/dL | 135–270 | 151.2 ± 34.84 | 154.5 ± 30 | 0.411 |
| TG | mg/dL | 20–112 | 48.4 ± 15.99 | 38.9 ± 4.79 | 0.052 |
| ALT | U/IL | 21–102 | 39.3 ± 24.34 | 37.2 ± 10.72 | 0.403 |
| GGT | UI/L | 1.2–6.4 | 5.1 ± 2.85 | 3 ± 1.05 | 0.025 * |
| ALP | UI/L | 20–156 | 70.6 ± 49.15 | 66.7 ± 35.1 | 0.420 |
| T-Bil | mg/dL | 0.1–0.5 | 0.3 ± 0.19 | 0.1 ± 0.02 | 0.016 * |
| TP | g/dL | 5.4–7.1 | 7.1 ± 0.62 | 6.8 ± 0.42 | 0.144 |
| Alb | g/dL | 2.6–3.3 | 3.5 ± 0.27 | 3.4 ± 0.40 | 0.163 |
| α1-glob | g/dL | 0.2–0.5 | 0.2 ± 0.05 | 0.2 ± 0.02 | 0.004 ** |
| α2-glob | g/dL | 0.3–1.1 | 1.0 ± 0.13 | 0.9 ± 0.15 | 0.087 |
| β1-glob | g/dL | 0.7–1.3 | 0.8 ± 0.23 | 0.9 ± 0.35 | 0.135 |
| β2-glob | g/dL | 0.6–1.4 | 0.8 ± 0.15 | 0.8 ± 0.23 | 0.323 |
| γ-glob | g/dL | 0.5–1.3 | 0.7 ± 0.25 | 0.6 ± 0.14 | 0.054 |
| LEP | ng/mL | - | 14.6 ± 5.88 | 6.3 ± 4.07 | 0.003 ** |
| LEP | LEPR | |||
|---|---|---|---|---|
| Groups | Mean | SD | Mean | SD |
| Ob | 1.91 | 0.76 | 1.82 | 0.66 |
| Nw | 1.72 | 1.04 | 0.66 | 0.42 |
| p * Ob vs. Nw | 0.67 | 0.0016 | ||
| LEP | LEPR | |||
|---|---|---|---|---|
| Groups | Mean | SD | Mean | SD |
| Ob | 4.70 | 5.10 | 5.15 | 0.95 |
| Nw | 9.06 | 7.01 | 0.47 | 0.32 |
| p * Ob vs. Nw | 0.37 | 6.18 × 10−6 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maranesi, M.; Di Loria, A.; Dall’Aglio, C.; Piantedosi, D.; Lepri, E.; Ciaramella, P.; Mercati, F. Leptin System in Obese Dog Skin: A Pilot Study. Animals 2020, 10, 2338. https://doi.org/10.3390/ani10122338
Maranesi M, Di Loria A, Dall’Aglio C, Piantedosi D, Lepri E, Ciaramella P, Mercati F. Leptin System in Obese Dog Skin: A Pilot Study. Animals. 2020; 10(12):2338. https://doi.org/10.3390/ani10122338
Chicago/Turabian StyleMaranesi, Margherita, Antonio Di Loria, Cecilia Dall’Aglio, Diego Piantedosi, Elvio Lepri, Paolo Ciaramella, and Francesca Mercati. 2020. "Leptin System in Obese Dog Skin: A Pilot Study" Animals 10, no. 12: 2338. https://doi.org/10.3390/ani10122338
APA StyleMaranesi, M., Di Loria, A., Dall’Aglio, C., Piantedosi, D., Lepri, E., Ciaramella, P., & Mercati, F. (2020). Leptin System in Obese Dog Skin: A Pilot Study. Animals, 10(12), 2338. https://doi.org/10.3390/ani10122338









