Longitudinal Improvements in Zoo-Housed Elephant Welfare: A Case Study at ZSL Whipsnade Zoo
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
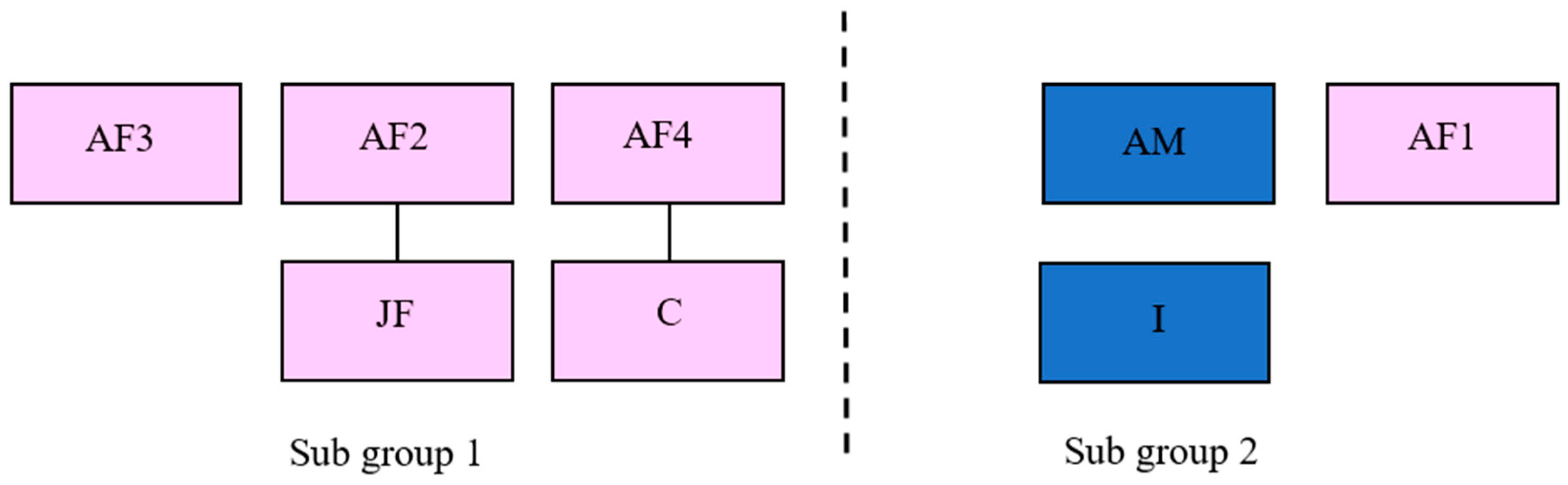
2.1. Study Subjects
2.2. Data Collection
2.2.1. Day Observation Data Collection
2.2.2. Night Observation Data Collection
2.2.3. Social Behaviour
2.3. Enclosure
2.3.1. Daytime Housing
Day Time Housing Changed Between Seasons
2.3.2. Nighttime Housing
2.4. Data Analysis
2.4.1. Activity Budgets
2.4.2. Data Analysis–Sociograms
3. Results
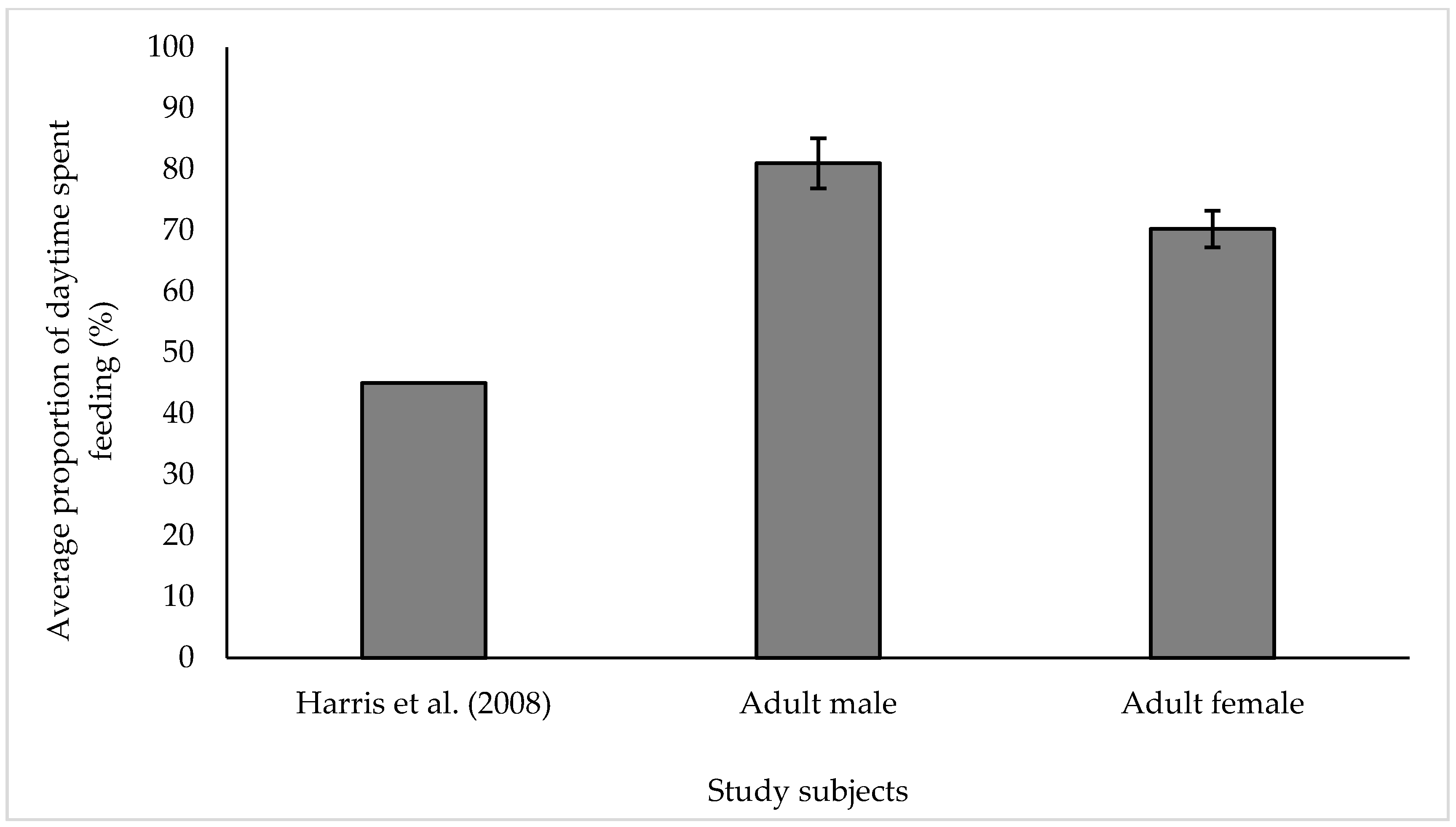
3.1. Feeding
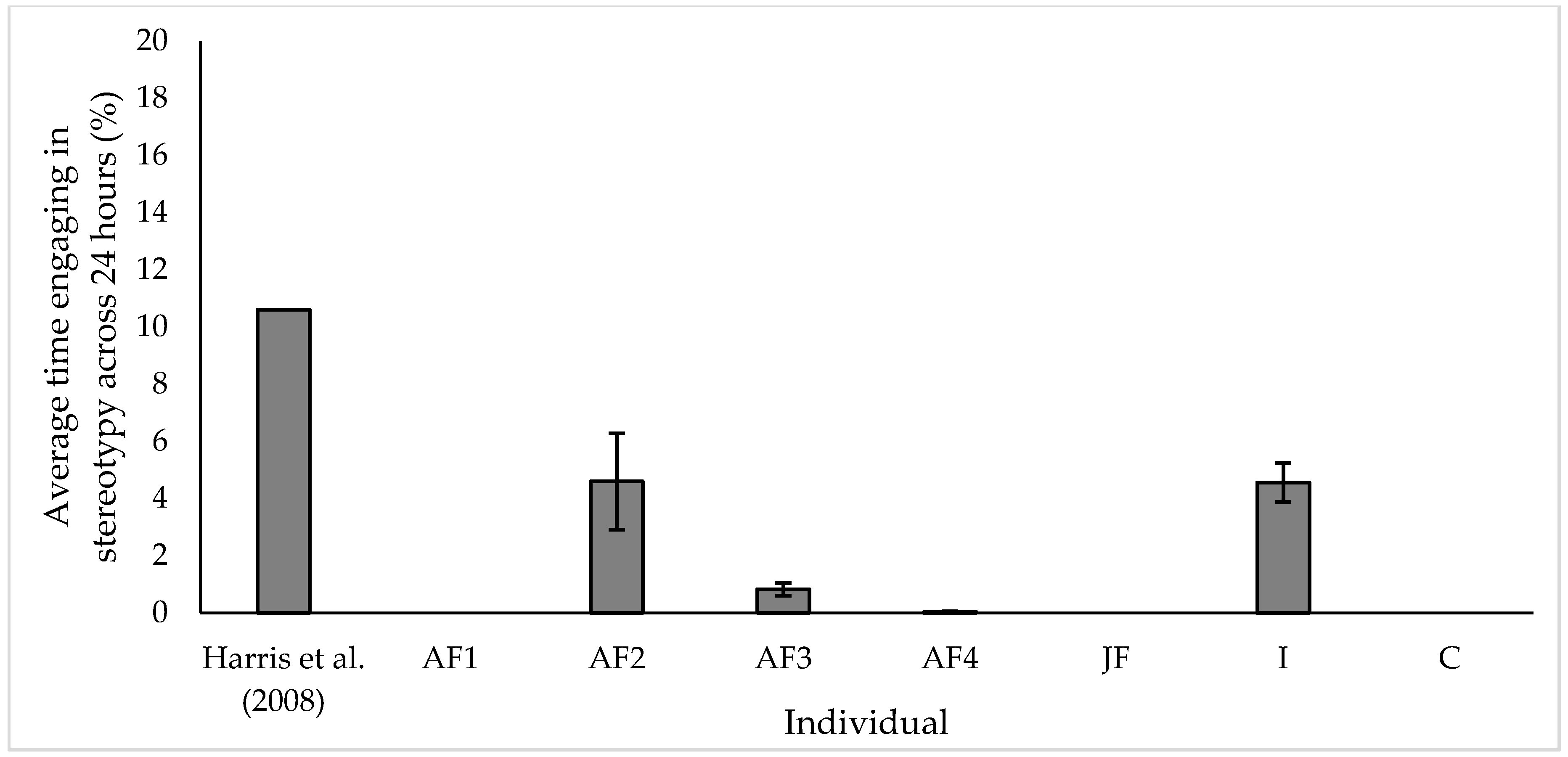
3.2. Stereotypic Behaviour
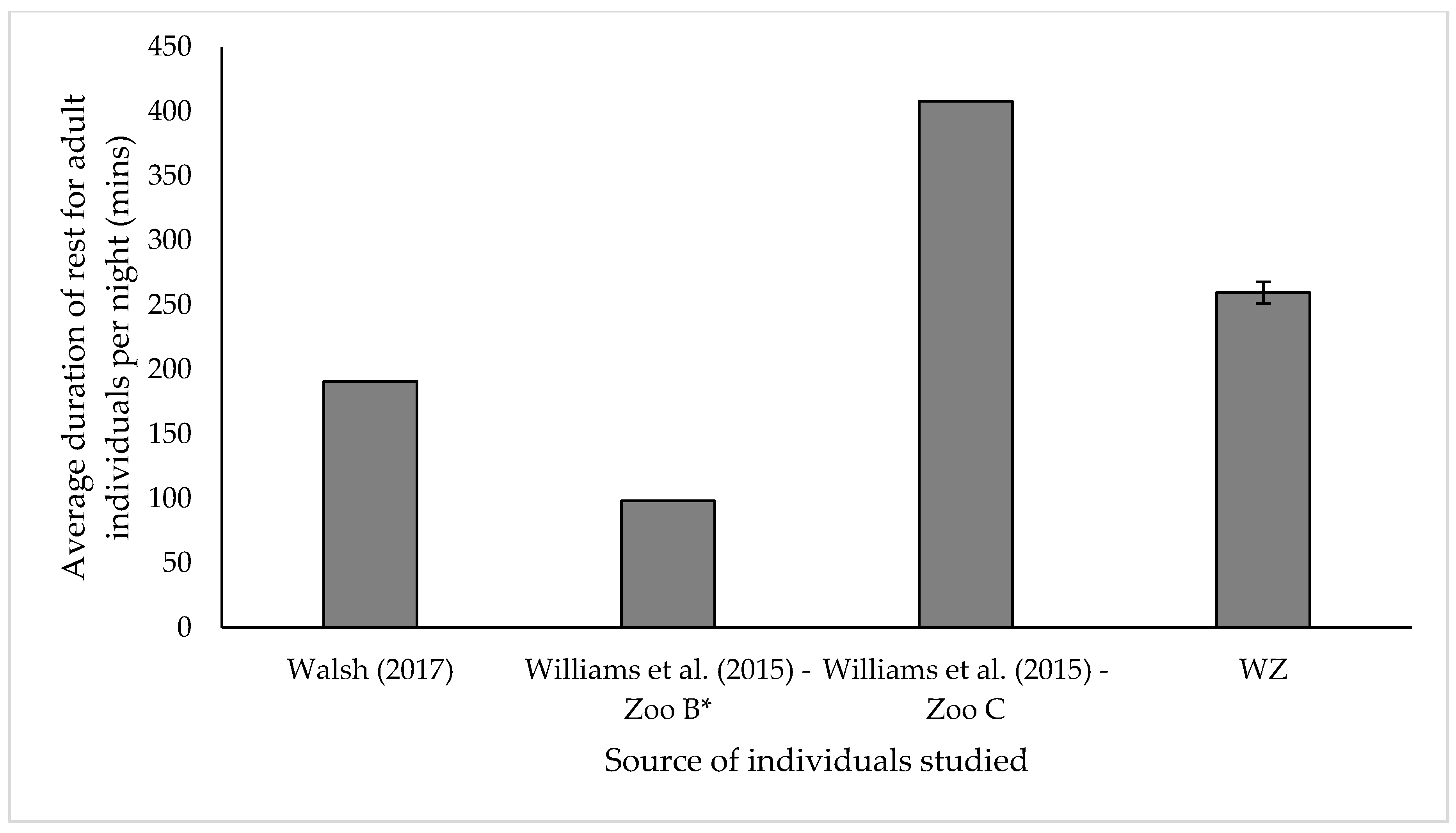
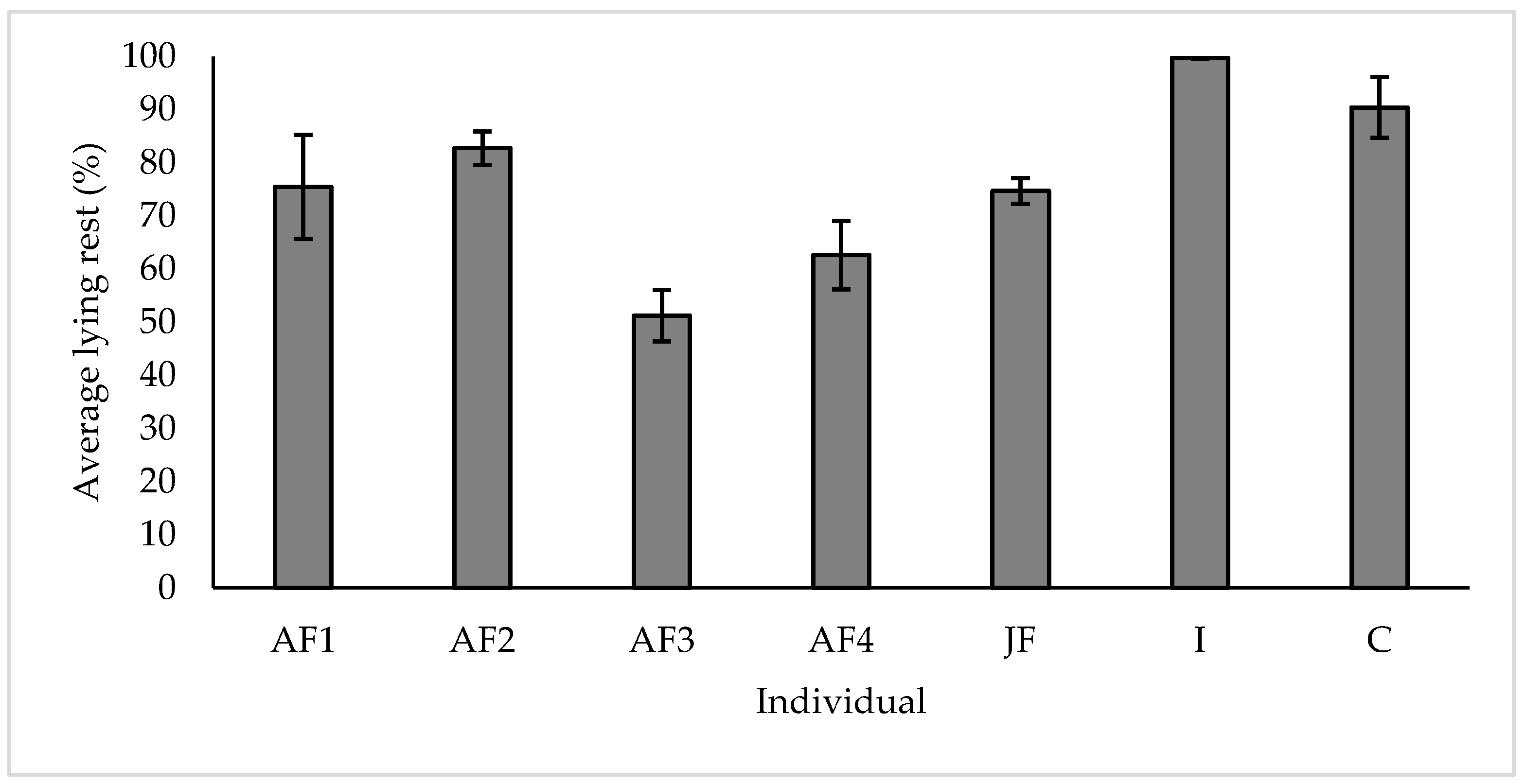
3.3. Resting
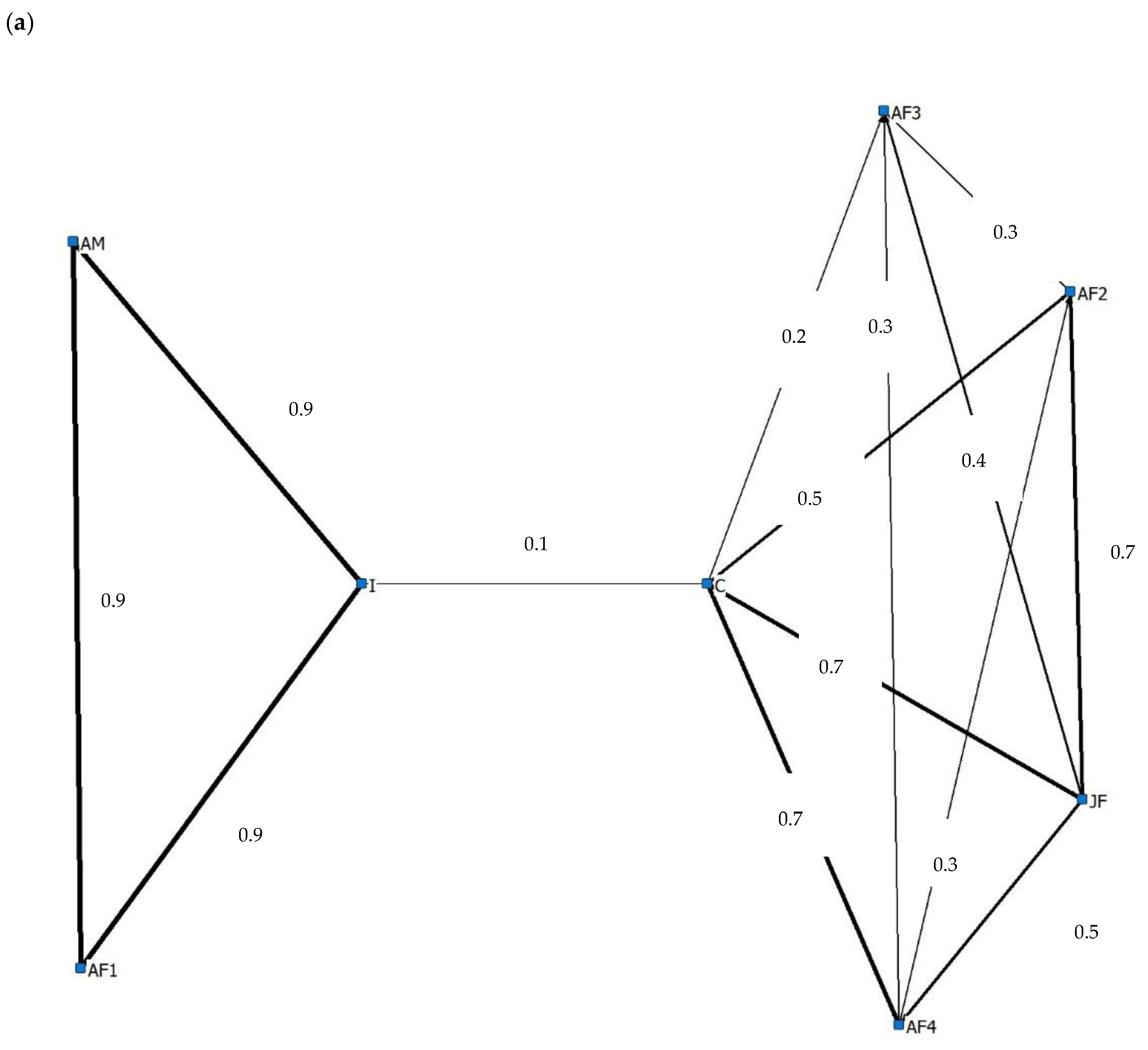
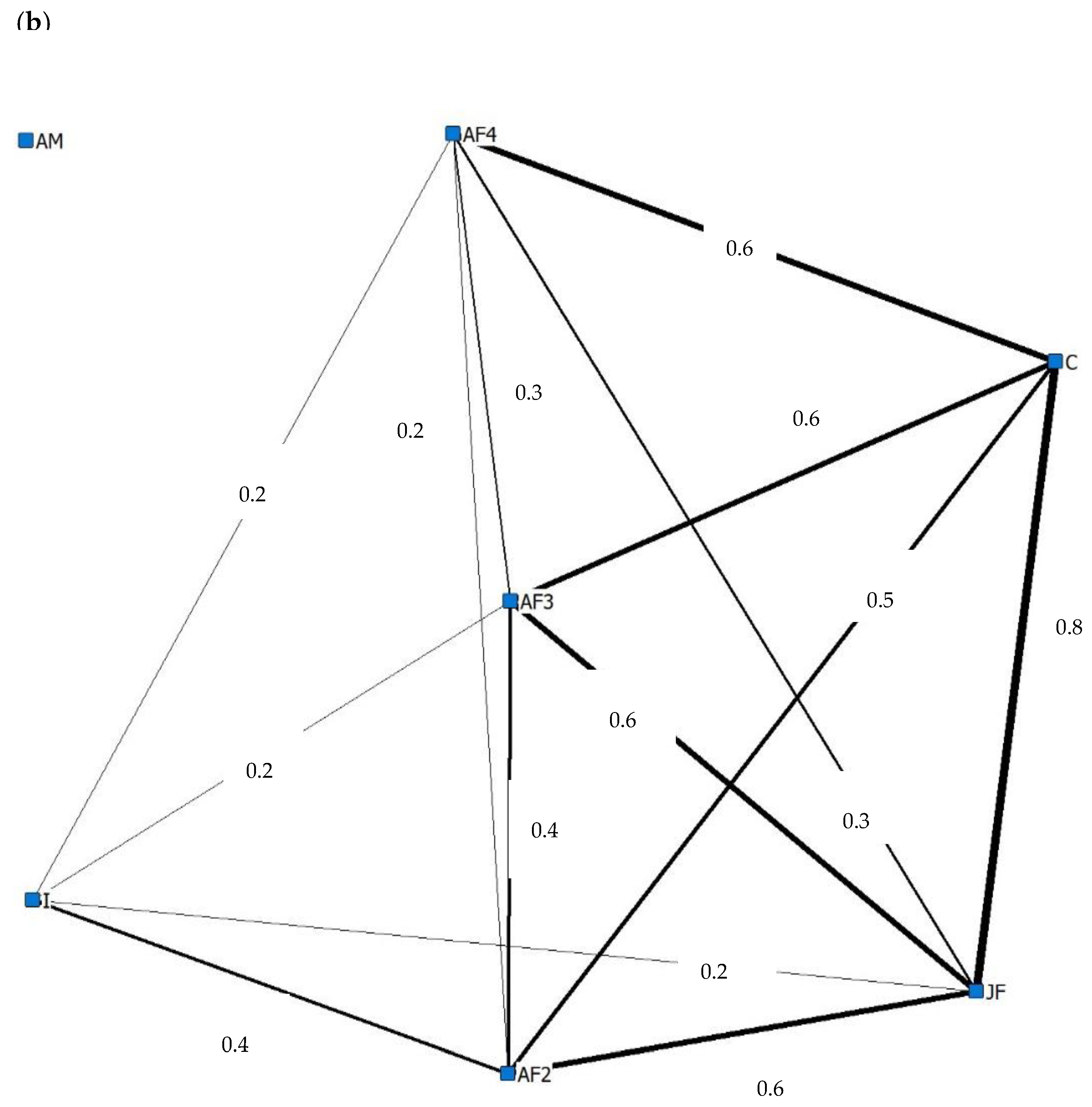
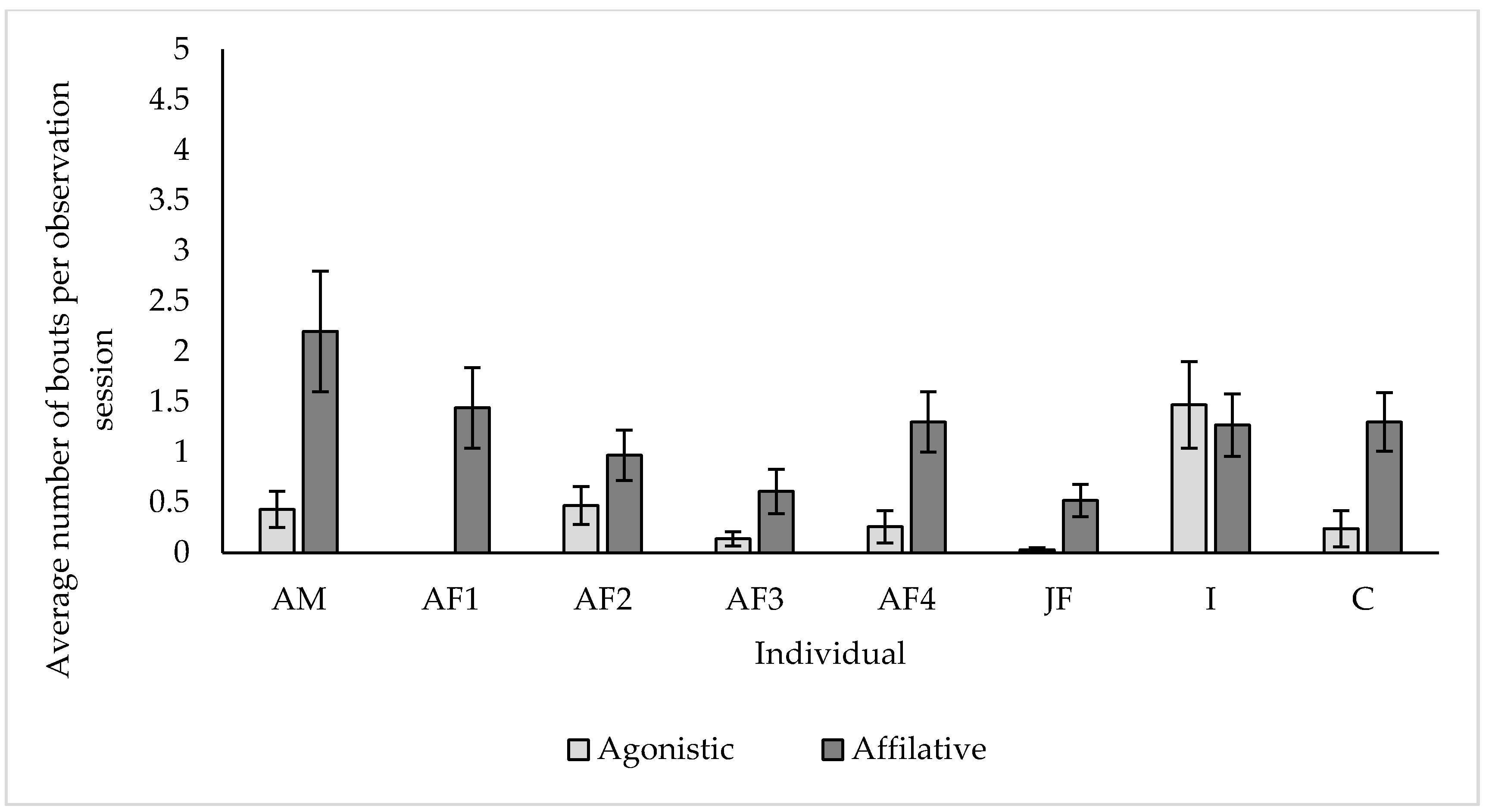
3.4. Social Behaviour
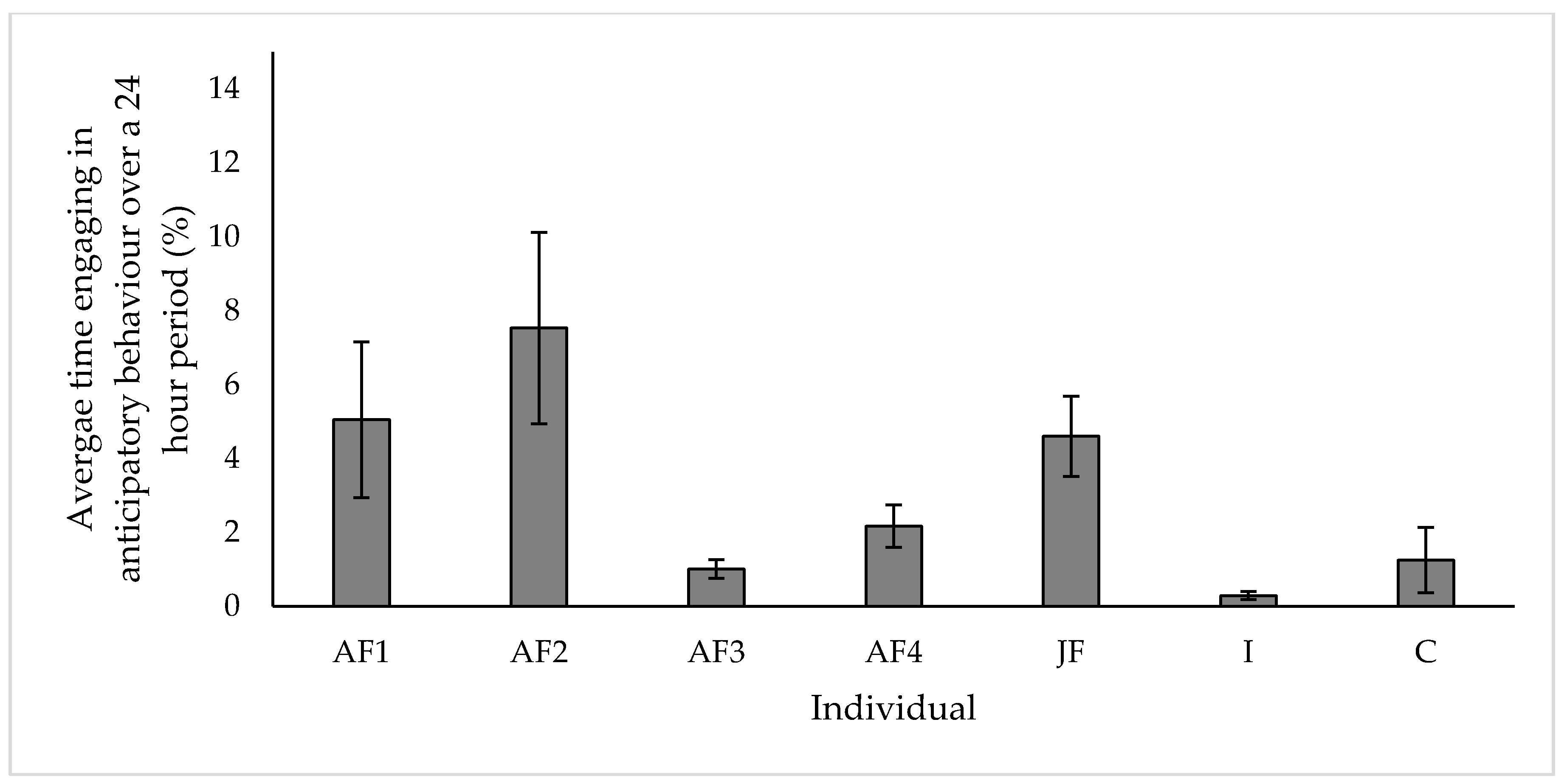
3.5. Anticipatory Behaviour
4. Discussion
4.1. Feeding
4.2. Stereotypy
4.3. Resting
4.4. Social Behaviour
4.5. Anticipatory Behaviour
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Behaviour Category | Behaviour | Description |
|---|---|---|
| Anticipatory | Anticipatory | Individual stands in a stationary, quadrupedal position at a fixed point within the exhibit. Attention is often fixed at this point, with head orientated towards it and no other discernible behaviours taking place. |
| Natural self-maintenance | Digging | Individual using foot to displace substrate. |
| Natural self-maintenance | Dust bathing | Individual uses trunk to pick up sand, dust or other substrate and throw it over the body. |
| Natural self-maintenance | Grooming self | Individual is scratching or rubbing itself using trunk or against an enclosure furnishing. |
| Natural self-maintenance | Wallowing | Individual is interacting with the mud wallows, rolling their body in the muddy water. |
| Natural self-maintenance | Water bathing | Individual using trunk to suck up water then spray and throw over the body. |
| Natural exploratory | Enrichment manipulation | Interaction with pieces of enrichment provided by the keepers as part of their enrichment plan. |
| Natural exploratory | Object manipulation | Interaction with other objects within the enclosure other than enrichment. |
| Feeding | Feeding | Active consumption or manipulation of feed items or ingestion of water. Includes foraging for grass, browse and hay. |
| Keeper interaction | Keeper interaction | Any interaction between the individual and members of the animal keeping team. |
| Stereotypy | Stereotypy | Individual is displaying an abnormal, repetitive behaviour which does not have a clear purpose or outcome. This can include: Linear pacing—Individual is walking forwards then backwards in a repetitive fashion. Pacing—Individual is locomoting in a repetitive fashion in a set pattern or route which serves no specific function. Swaying—Individual transfers weight onto alternate limbs in a repetitive pattern while in a fixed position, direction of travel can be from side to side or front to back. No head movement. Weaving—Individual transfers weight onto alternate limbs while simultaneously moving the head from side to side. |
| Locomotion | Walk | Individual moving across the enclosure to get to another specific location at a walking pace. Only one foot is removed from the ground at any one time. |
| Locomotion | Run | Individual moving across the enclosure to get to another specific location at a running pace, more than one foot in removed from the ground at any one time. |
| Resting | Lying rest | Individual is relaxed in lateral recumbence. |
| Resting | Standing rest | Individual is upright and stationary with 3 or 4 feet on the ground. Individual is relaxed with no other behaviour being displayed. |
| Affiliative social | Affiliative social behaviour * | Any social behaviour which maintains or strengthens positive social relationships within the group. |
| Agonistic social | Agonistic social behaviour * | Individuals interacting in a way which may disrupt or weaken social bonds within the group. |
Appendix B
| Behaviour Category | Behaviour | Description |
|---|---|---|
| Affiliative social | Trunk hold | Individual has its trunk intertwined with conspecific’s trunk in a non-aggressive manner. |
| Affiliative social | Trunk touch | Individual places trunk in a non-aggressive manner on the body, mouth or genital area of a conspecific. |
| Affiliative social | Body rub or nudge | Individuals have gentle physical contact with one another, head-head, head-body, body-body. |
| Affiliative social | Play | A positive interaction which could include sparring, wrestling, mounting, chasing, and rolling on another elephant in a playful manner. |
| Affiliative social | Leaning | Elephant gently leaning on a conspecific with their head, side or rump. |
| Affiliative social | Urine and faecal inspection | Individual inspects the urine or faeces of another elephant. |
| Affiliative social | Nursing | Mother stands for calf to suckle. |
| Agonistic social | Kick | Individual strikes out and hits another elephant with their foot in an aggressive manner. |
| Agonistic social | Trunk slap | Hitting a conspecific with trunk, head orientated towards another elephant violently swinging trunk in an aggressive display. |
| Agonistic social | Charge | Individual moving at a fast pace towards conspecific for more than three steps, head held high with some contact made. |
| Agonistic social | Mock charge | Individual moves at a fast pace towards conspecific for more than three steps, contact does not occur. |
| Agonistic social | Chase | Follows on from ‘charge’ behaviour leading to the pursuit of an individual. |
| Agonistic social | Push | Elephant forces or pushes body of another elephant, resulting in elephant that is pushed moving at least two steps. |
| Agonistic social | Stand off | Two elephants standing facing in opposite directions, with foreheads pushing against each other. |
| Agonistic social | Lunge | Individual thrusts their body towards a conspecific in an aggressive manner taking less than three steps. If more steps taken then reclassify as ‘Charge/Mock charge’. |
| Agonistic social | Tusking | Individual pokes or jabs a conspecific with their tusk. |
| Agonistic social | Tail pulling | Individual pulls a conspecifics tail with trunk |
| Agonistic social | Contact displacement | Movement of an individual resulting in conspecific leaving its location (within 10s) caused by physical contact between individuals such as push or nudge. |
| Agonistic social | Non-contact displacement | Movement of one elephant towards the other, resulting in conspecific leaving its location (within 10s) no physical contact occurs between elephants. |
| Agonistic social | Aggressive display | Facing a conspecific in an aggressive posture, head bobbing up and down or side to side, ears wide and flapping. |
| Agonistic social | Biting | Individual bites a conspecific’s body, trunk or tail. Physical contact occurs between the mouth of the initiator and the body part of the recipient. |
| Agonistic social | Trunk dominance | Focal individual places their trunk over the top of conspecific, mouths usually close together. Individual actively tries to place trunk in a higher position than conspecific to assert dominance. |
| Agonistic social | Food stealing | Individual takes food away from a conspecific using its trunk or another part of the body. |
| Excretion | Urination | Elimination of urine from the body. |
| Excretion | Defecation | Elimination of faeces from the body. |
| Vocalisation | Trumpet call | Individual emits a loud alarm call. |
| Vocalisation | Rumble | Individual emits a low frequency rumble call. |
Proximity Categories
- 0—
- Individuals are touching but not engaging in a social interaction
- 1—
- Individual is one elephant body length away from the focal individual
- 2—
- Individual is two elephant body lengths away from the focal individual
- 3—
- Individual is three or more elephant body lengths away from the focal individual.
References
- Lee, H.S. Measurement of visitors’ satisfaction with public zoos in Korea using importance-performance analysis. Tour. Manag. 2015, 47, 251–260. [Google Scholar] [CrossRef]
- Moss, A.; Esson, M. Visitor interest in zoo animals and the implications for collection planning and zoo education programmes. Zoo Biol. 2010, 29, 715–731. [Google Scholar] [CrossRef] [PubMed]
- Hvilsom, C.; Welden, H.L.A.; Stelvig, M.; Nielsen, C.K.; Purcell, C.; Eckley, L.; Bertelsen, M.F. The contributions of EAZA zoos and aquariums to peer reviewed scientific research. JZAR 2020, 8, 133–138. [Google Scholar]
- Wolfensohn, S.; Shotton, J.; Bowley, H.; Davies, S.; Thompson, S.; Justice, W.S.M. Assessment of welfare in zoo animals: Towards optimum quality of life. Animals 2018, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Watters, J.V.; Margulis, S.W.; Atsalis, S. Behavioural monitoring in zoos and aquariums: A tool for guiding husbandry and directing research. Zoo Biol. 2008, 28, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Veasey, J. In pursuit of peak animal welfare; the need to prioritize the meaningful over the measurable. Zoo Biol. 2017, 36, 413–425. [Google Scholar] [CrossRef]
- Brando, S.; Buchanan-Smith, H.M. The 24/7 approach to promoting optimal welfare for captive wild animals. Behav. Proc. 2018, 156, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Broom, D.M. Animal welfare: Concepts and measurement. J. Anim. Sci. 1991, 69, 4167–4175. [Google Scholar] [CrossRef] [PubMed]
- Botreau, R.; Veissier, I.; Butterworth, A.; Bracke, M.B.M.; Keeling, L.J. Definition of criteria for overall assessment of animal welfare. Anim. Welf. 2007, 16, 225–228. [Google Scholar]
- Whitham, J.C.; Wielebnowski, N. New directions for zoo animal welfare science. Appl. Anim. Behav. Sci. 2013, 147, 247–260. [Google Scholar] [CrossRef]
- Williams, E.; Carter, A.; Hall, C.; Bremner-Harrison, S. Social interactions in zoo housed elephants: Factors affecting social relationships. Animals 2019, 9, 747. [Google Scholar] [CrossRef] [PubMed]
- Yon, L.; Williams, E.; Harvey, N.; Asher, L. Development of a behavioural welfare assessment tool for routine use with captive elephants. PLoS ONE 2019, 14, e0210783. [Google Scholar] [CrossRef] [PubMed]
- Meehan, C.; Greco, B.; Lynn, B.; Morfeld, K.; Vicino, G.; Orban, D.; Gorsuch, C.; Quick, M.; Ripple, L.; Fournier, K.; et al. The Elephant Welfare Initiative: A model for advancing evidence-based zoo animal welfare monitoring, assessment and enhancement. Int. Zoo Yearb. 2019, 53, 45–61. [Google Scholar] [CrossRef]
- Swaisgood, R.R. The conservation-welfare nexus in reintroduction programmes: A role for sensory ecology. Anim. Welf. 2010, 19, 125–137. [Google Scholar]
- Mellor, D.J. Animal emotions, behaviour and the promotion of positive welfare states. NZ Vet. J. 2012, 60, 1–8. [Google Scholar] [CrossRef]
- Clayton, S.; Fraser, J.; Saunders, C.D. Zoo experiences: Conversations, connections and concern for animals. Zoo Biol. 2009, 28, 377–397. [Google Scholar] [CrossRef] [PubMed]
- Cooper, M.E. Zoo legislation. Int. Zoo Yearb. 2003, 38, 81–93. [Google Scholar] [CrossRef]
- Zoo Licencing Act 1981. Available online: http://www.legislation.gov.uk/ukpga/1981/37 (accessed on 6 July 2020).
- Secretary of State’s Standards of Modern Zoo Practice Appenidx 8—Specialist Exhibits, Elephants 2017. DEFRA: London, UK. Available online: https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/69596/standards-of-zoo-practice.pdf (accessed on 28 June 2020).
- Sach, F.; Fitzpatrick, M.; Masters, N.; Field, D. Financial planning required to keep elephants in zoos in the United Kingdom in accordance with the Secretary of State’s Standards of Modern Zoo Practice for the next 30 years. Int. Zoo Yearb. 2019, 53, 78–88. [Google Scholar] [CrossRef]
- Draper, C.; Harris, S. The assessment of animal welfare in British zoos by government-appointed inspectors. Animals 2012, 2, 507–5283. [Google Scholar] [CrossRef] [PubMed]
- Clubb, R.; Mason, G. A Review of the Welfare of Zoo Elephants in Europe. A Report Commissioned by The RSPCA; University of Oxford RSPCA Horsham: West Sussex, UK, 2002. [Google Scholar]
- Harris, M.; Sherwin, C.; Harris, S. The welfare, Housing and Husbandry of Elephants in UK Zoos; DEFRA Science and Research Project WC05007; Department of Food, The Environment and Rural Affairs: London, UK, 2008.
- Clubb, R.; Mason, G. Natural behavioural biology as a risk factor in carnivore welfare: How analysing species differences could help zoos improve enclosures. Appl. Anim. Behav. Sci. 2007, 102, 303–328. [Google Scholar] [CrossRef]
- Chadwick, C.L.; Williams, E.; Asher, L.; Yon, L. Incorporating stakeholder perspectives into the assessment and provision of captive elephant welfare. Anim. Welf. 2017, 26, 461–472. [Google Scholar] [CrossRef]
- Fischer, M.; Sach, F. Editorial: Conservation of elephants. Int. Zoo Yearb. 2019, 53, 9–16. [Google Scholar] [CrossRef]
- Schmidt, H.; Kappelhof, J. Review of the management of the Asian elephant Elephas maximus EEP: Current challenges and future solutions. Int. Zoo Yearb. 2019, 53, 31–44. [Google Scholar] [CrossRef]
- Martin, P.R.; Bateson, P.P.G. Measuring Behaviour: An Introductory Guide, 3rd ed.; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Walsh, B. Asian elephant (Elephas maximus) sleep study—Long-term quantitative research at Dublin Zoo. JZAR 2017, 5, 82–85. [Google Scholar]
- Williams, E.; Bremner-Harrison, S.; Harvey, N.; Evison, E.; Yon, L. An investigation into resting behaviour in Asian elephants in UK zoos. Zoo Biol. 2015, 34, 406–417. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2013. Available online: http://www.R-project.org (accessed on 6 July 2020).
- Borgatti, S.P.; Everett, M.G.; Freeman, L.C. Ucinet 6 for Windows: Software for Social Network Analysis. Harv. MA Anal. Technol. 2002, 6, 12–15. [Google Scholar]
- Sukumar, R. Ecology of the Asian elephant in southern India. J. Trop. Ecol. 1990, 6, 33–53. [Google Scholar] [CrossRef]
- Ullrey, D.E.; Crissey, S.D.; Hintz, H.F. Elephants: Nutrition and Dietary Husbandry; Nutrition Advisory Group Fact Sheet 004: East Lansing, MI, USA, 1997; pp. 1–20. [Google Scholar]
- Clauss, M.; Loehlein, W.; Kienzle, E.; Wiesner, H. Studies on feed digestibilities in captive Asian elephants (Elephas maximus). J. Anim. Physiol. Anim. Nutr. 2003, 87, 160–173. [Google Scholar] [CrossRef]
- Hatt, J.M.; Clauss, M. Feeding Asian and African elephants Elephas maximus and Loxodonta africana in captivity. Int. Zoo Yearb. 2006, 40, 88–95. [Google Scholar] [CrossRef]
- Sach, F.; Dierenfeld, E.S.; Langley-Evans, S.C.; Watts, M.J.; Yon, L. African savannah elephants (Loxodonta africana) as an example of a herbivore making movement choices based on nutritional needs. PeerJ 2019, 7, e6260. [Google Scholar] [CrossRef]
- Sach, F.; Tatchley, C.; Needham, N.; Pullen, K. Guidelines for the Management of Elephants within BIAZA Zoos: Incorporating BIAZA’s Policy on the Management of Elephants; BIAZA: London, UK, 2019. [Google Scholar]
- French, F.; Mancini, C.; Sharp, H. High tech cognitive and acoustic enrichment for captive elephants. J. Neurosci. Methods 2018, 300, 173–183. [Google Scholar] [CrossRef]
- Veasey, J. Assessing the psychological priorities for optimising captive Asian elephant (Elephas maximus) welfare. Animals 2020, 10, 39. [Google Scholar] [CrossRef]
- Mason, G.J. Species differences in responses to captivity: Stress, welfare and the comparative method. Trends Ecol. Evol. 2010, 25, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Haspeslagh, M.; Stevens, J.M.G.; De Groot, E.; Dewulf, J.; Kalmar, I.D.; Moons, C.P.H. A survey of foot problems, stereotypic behaviour and floor type in Asian elephants (Elephas maximus) in European zoos. Anim. Welf. 2013, 22, 437–443. [Google Scholar] [CrossRef]
- Greco, B.J.; Meehan, C.L.; Heinsius, J.L.; Mench, J.A. Why pace? The influence of social, housing, management, life history, and demographic characteristics on locomotor stereotypy in zoo elephants. Appl. Anim. Behav. Sci. 2017, 194, 104–111. [Google Scholar] [CrossRef]
- Marriner, L.M.; Drickamer, L.C. Factors influencing stereotyped behaviour of primates in a zoo. Zoo Biol. 1994, 13, 267–275. [Google Scholar] [CrossRef]
- Quirke, T.; O’Riordan, R.M.; Zuur, A. Factors influencing the prevalence of stereotypical behaviour in captive cheetahs (Acinonyx jubatus). Appl. Anim. Behav. Sci 2012, 142, 189–197. [Google Scholar] [CrossRef]
- Cooper, J.J.; Nicol, C.J. Neighbour effects on the development of locomotor stereotypies in bank voles (Clethrionomys glareolus). Anim. Behav. 1994, 41, 971–977. [Google Scholar] [CrossRef]
- Mason, G.; Latham, N.R. Can’t stop, won’t stop: Is stereotypy a reliable animal welfare indicator? Anim. Welf. 2004, 13, 57–69. [Google Scholar]
- Greco, B.J.; Meehan, C.L.; Hogan, L.N.; Leighty, K.A.; Mellen, J.; Mason, G.J.; Mench, J.A. The days and nights of zoo elephants: Using epidemiology to better understand stereotypic behavior of African elephants (Loxodonta africana) and Asian elephants (Elephas maximus) in North American zoos. PLoS ONE 2016, 11, 1–29. [Google Scholar] [CrossRef]
- Coleman, K.; Maier, A. The use of positive reinforcement training to reduce stereotypic behaviour in rhesus macaques. Appl. Anim. Behav. Sci. 2010, 124, 142–148. [Google Scholar] [CrossRef]
- Korte, S.M.; Olivier, B.; Koolhaas, J.M. A new animal welfare concept based on allostasis. Physiol. Behav. 2007, 92, 422–428. [Google Scholar] [CrossRef]
- Asher, L.; Williams, E.; Yon, L. Developing Behavioural Indicators as Part of a Wider Set of Indicators, to Assess the Welfare of Elephants in UK Zoos—DEFRA Project WC 1081 2015. Available online: http://sciencesearch.defra.gov.uk/Document.aspx?Document=12816_ElephantwelfarereportWC1081.pdf (accessed on 6 July 2020).
- Wuestenhagen, A.; Weisz, A.; Schwammer, H. Sleeping behaviour of six African elephants (Loxodonta africana) in the Schonbrunn zoological garden, Vienna, Austria. Zoologische Garten 2000, 70, 253–261. [Google Scholar]
- Schiffman, C.; Ho, S.; Wenker, C.; Hard, T.; Scholz, R.; Clauss, M.; Hatt, J.M. When elephants fall asleep: A literature review on elephant rest with case studies on elephant falling bouts, and practical solutions for zoo elephants. Zoo Biol. 2018, 37, 133–145. [Google Scholar] [CrossRef]
- Schiffman, C.; Knibbs, K.; Clauss, M.; Merrington, J.; Beasley, D. Unexpected resting behaviour in a geriatric zoo elephant. Gajah 2018, 48, 30–33. [Google Scholar]
- Miller, M.A.; Hogan, J.N.; Meehan, C.L. Housing and demographic risk factors impacting foot and musculoskeletal health in African elephants (Loxodonta africana) and Asian elephants (Elephas maximus) in North American Zoos. PLoS ONE 2016, 11, e0155223. [Google Scholar] [CrossRef]
- Williams, E.; Chadwick, C.L.; Yon, L.; Asher, L. A review of the current indicators of welfare in captive elephants (Loxodonta africana and Elephas maximus). Anim. Welf. J. 2018, 27, 235–249. [Google Scholar] [CrossRef]
- Archie, E.A.; Chiyo, P.I. Elephant behaviour and conservation: Social relationships, the effects of poaching, and genetic tools for management. Mol. Ecol. 2012, 21, 765–778. [Google Scholar] [CrossRef]
- Liu, S.; Yin, Y.; Li, J.; Cheng, F.; Dong, S.; Zhang, Y. Using cross-scale landscape connectivity indices to identify key habitat resource patches for Asian elephants in Xishuangbanna, China. Landsc. Urban Plan 2018, 171, 80–87. [Google Scholar] [CrossRef]
- Srinivasaiah, N.; Kumar, V.; Vaidyanathan, S.; Sukumar, R.; Sinha, A. All-Male groups in Asian elephants: A novel, adaptive social strategy in increasingly anthropogenic landscapes of southern India. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Meehan, C.L.; Mench, J.A.; Carlstead, K.; Hogan, J.N. Determining connections between the daily lives of zoo elephant and their welfare: An epidemiological approach. PLoS ONE 2016, 11, e0158124. [Google Scholar] [CrossRef]
- Harvey, N.D.; Daly, C.; Clark, N.; Ransford, E.; Wallace, S.; Yon, L. Social interactions in two groups of zoo-housed adult female Asian elephants (Elephas maximus) that differ in relatedness. Animals 2018, 8, 132. [Google Scholar] [CrossRef] [PubMed]
- Archie, E.A.; Moss, C.J.; Alberts, S.C. The ties that bind: Genetic relatedness predicts the fission and fusion of social groups in wild African elephants. Proc. R. Soc. B 2006, 273, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Veasey, J. Concepts in the care and welfare of captive elephants. Int. Zoo Yearb. 2006, 40, 63–79. [Google Scholar] [CrossRef]
- Slotow, R.; van Dyk, G.; Poole, J.; Page, B.; Klocke, A. Older bull elephants control young males. Nature 2000, 408, 425–426. [Google Scholar] [CrossRef]
- Evans, K.; Harris, S. Adolescence in male African elephants, Loxodonta africana, and the importance of sociality. Anim. Behav. 2000, 76, 779–787. [Google Scholar] [CrossRef]
- Bradshaw, G.A.; Schore, A.N. How elephants are opening doors: Developmental neuroethology, attachment and social context. Ethology 2007, 113, 426–436. [Google Scholar] [CrossRef]
- Hartley, M.; Wood, A.; Yon, L. Facilitating the social behaviour of bull elephants in zoos. Int. Zoo Yearb. 2019, 53, 62–77. [Google Scholar] [CrossRef]
- Clegg, I.L.K.; Rödel, H.G.; Boivin, X.; Delfour, F. Looking forward to interacting with their caretakers: Dolphins’ anticipatory behaviour indicates motivation to participate in specific events. Appl. Anim. Behav. Sci. 2018, 202, 85–93. [Google Scholar] [CrossRef]
- Ward, S.J.; Sherwen, S.; Clark, F.E. Advances in applied zoo animal welfare science. J. Appl. Anim. Welf. Sci. 2018, 21, 23–33. [Google Scholar] [CrossRef]
- Krebs, B.L.; Torres, E.; Chesney, C.; Moon, V.K.; Watters, J.V. Applying behavioural conditioning to identify anticipatory behaviours. J. Appl. Anim. Welf. Sci. 2017, 20, 155–175. [Google Scholar] [CrossRef]
- Watters, J.V. Searching for behavioural indicators of welfare in zoos: Uncovering anticipatory behaviour. Zoo Biol. 2014, 33, 251–256. [Google Scholar] [CrossRef]
- Spruijt, B.M.; van den Bos, R.; Pijlman, F.T.A. A concept of welfare based on reward evaluating mechanisms in the brain: Anticipatory behaviour as an indicator for the state of reward systems. Appl. Anim. Behav. Sci. 2001, 72, 145–171. [Google Scholar] [CrossRef]
| Individual | Average Proportion of Time Spent Resting per Night (% ± s.e) | Average Duration Spent Resting per Night (Minutes ± s.e) |
|---|---|---|
| AF1 | 24.9 (±2.6) | 243.8 (±12.6) |
| AF2 | 26 (±1.5) | 296.7 (±18.3) |
| AF3 | 24.2 (±2.8) | 246.1 (±15.8) |
| AF4 | 21 (±1.2) | 246.8 (±13.5) |
| JF | 28.6 (±0.9) | 343.9 (±10.5) |
| I | 31.5 (±0.8) | 338.3 (±10.5) |
| C | 29.8 (±2) | 343.2 (±16.7) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finch, K.; Sach, F.; Fitzpatrick, M.; Masters, N.; Rowden, L.J. Longitudinal Improvements in Zoo-Housed Elephant Welfare: A Case Study at ZSL Whipsnade Zoo. Animals 2020, 10, 2029. https://doi.org/10.3390/ani10112029
Finch K, Sach F, Fitzpatrick M, Masters N, Rowden LJ. Longitudinal Improvements in Zoo-Housed Elephant Welfare: A Case Study at ZSL Whipsnade Zoo. Animals. 2020; 10(11):2029. https://doi.org/10.3390/ani10112029
Chicago/Turabian StyleFinch, Katherine, Fiona Sach, Malcolm Fitzpatrick, Nic Masters, and Lewis J. Rowden. 2020. "Longitudinal Improvements in Zoo-Housed Elephant Welfare: A Case Study at ZSL Whipsnade Zoo" Animals 10, no. 11: 2029. https://doi.org/10.3390/ani10112029
APA StyleFinch, K., Sach, F., Fitzpatrick, M., Masters, N., & Rowden, L. J. (2020). Longitudinal Improvements in Zoo-Housed Elephant Welfare: A Case Study at ZSL Whipsnade Zoo. Animals, 10(11), 2029. https://doi.org/10.3390/ani10112029




