Can Agro-Industrial By-Products Rich in Polyphenols be Advantageously Used in the Feeding and Nutrition of Dairy Small Ruminants?
Simple Summary
Abstract
1. Introduction
2. By-Products Naturally Rich in Polyphenols
2.1. Chemical Composition of Agro-Industrial By-Products Naturally Rich in Polyphenols
2.2. Phenolic Compounds of Some Agro-Industrial By-Products Naturally Rich in Polyphenols
3. By-Products Naturally Rich in Polyphenols in Small Ruminant Feeding and Nutrition
3.1. Effect on Voluntary Feed Intake
3.2. Effect on Digestibility
3.3. Effect on Blood Metabolites
3.4. Effect on Rumen Parameters
3.5. Effect on Milk Production and Composition
3.6. Effect on Mik and Cheese Fatty Acid Profile
4. Systemic Perspective of Using By-Products Rich in Polyphenols in Ruminant Nutrition
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Environment Action Programme—European Commission. Available online: https://ec.europa.eu/environment/action-programme/index.htm (accessed on 12 September 2019).
- Scotto, A.L. Impatto Ambientale dei Rifiuti e Degli Sprechi Agroalimentari in Europa e in Italia; University of Bologna: Bologna, Italy, 2012. [Google Scholar]
- EUROSTAT 2019 (Statistical Office of the European Union). Available online: https://ec.europa.eu/eurostat/data/database (accessed on 12 August 2019).
- FAOSTAT (Food and Agriculture Organization of the United Nations Statistics Division). Statistical Database of the Food and Agriculture Organization of the United Nations. 2019. Available online: http://www.fao.org/faostat/en/?#data/GE (accessed on 8 June 2019).
- Federici, F.; Fava, F.; Kalogerakis, N.; Mantzavinos, D. Valorisation of agro-industrial by-products, effluents and waste: concept, opportunities and the case of olive mill wastewaters. J. Chem. Technol. Biotechnol. 2009, 84, 895–900. [Google Scholar] [CrossRef]
- Mirzaei-Aghsaghali, A.; Maheri-Sis, N. Nutritive value of some agro-industrial by-products for ruminants—A review. World J. Zool. 2008, 3, 40–46. [Google Scholar]
- Schieber, A.; Stintzing, F.C.; Carle, R. By-products of plant food processing as a source of functional compounds—Recent developments. Trends Food Sci. Technol. 2001, 12, 401–413. [Google Scholar] [CrossRef]
- Vasta, V.; Luciano, G. The effects of dietary consumption of plants secondary compounds on small ruminants’ products quality. Small Rumin. Res. 2011, 101, 150–159. [Google Scholar] [CrossRef]
- Min, B.R.; Barry, T.N.; Attwood, G.T.; McNabb, W.C. The effect of condensed tannins on the nutrition and health of ruminants fed fresh temperate forages: A review. Anim. Feed Sci. Technol. 2003, 106, 3–19. [Google Scholar] [CrossRef]
- Vasta, V.; Daghio, M.; Cappucci, A.; Buccioni, A.; Serra, A.; Viti, C.; Mele, M. Invited review: Plant polyphenols and rumen microbiota responsible for fatty acid biohydrogenation, fiber digestion, and methane emission: Experimental evidence and methodological approaches. J. Dairy Sci. 2019, 102, 3781–3804. [Google Scholar] [CrossRef]
- Descalzo, A.M.; Sancho, A.M. A review of natural antioxidants and their effects on oxidative status, odor and quality of fresh beef produced in Argentina. Meat Sci. 2008, 79, 423–436. [Google Scholar] [CrossRef]
- Provenza, F.D.; Kronberg, S.L.; Gregorini, P. Is Grassfed Meat and Dairy Better for Human and Environmental Health? Front. Nutr. 2019, 6, 26. [Google Scholar] [CrossRef]
- Salami, S.A.; Luciano, G.; O’Grady, M.N.; Biondi, L.; Newbold, C.J.; Kerry, J.P.; Priolo, A. Sustainability of feeding plant by-products: A review of the implications for ruminant meat production. Anim. Feed Sci. Technol. 2019, 251, 37–55. [Google Scholar] [CrossRef]
- Halmemies-Beauchet-Filleau, A.; Rinne, M.; Lamminen, M.; Mapato, C.; Ampapon, T.; Wanapat, M.; Vanhatalo, A. Review: Alternative and novel feeds for ruminants: Nutritive value, product quality and environmental aspects. Animal 2018, 12, s295–s309. [Google Scholar] [CrossRef]
- Castrica, M.; Rebucci, R.; Giromini, C.; Tretola, M.; Cattaneo, D.; Baldi, A. Total phenolic content and antioxidant capacity of agri-food waste and by-products. Ital. J. Anim. Sci. 2019, 18, 336–341. [Google Scholar] [CrossRef]
- Nudda, A.; Correddu, F.; Atzori, A.S.; Marzano, A.; Battacone, G.; Nicolussi, P.; Bonelli, P.; Pulina, G. Whole exhausted berries of Myrtus communis L. supplied to dairy ewes: Effects on milk production traits and blood metabolites. Small Rumin. Res. 2017, 155, 33–38. [Google Scholar] [CrossRef]
- Nudda, A.; Buffa, G.; Atzori, A.S.; Cappai, M.G.; Caboni, P.; Fais, G.; Pulina, G. Small amounts of agro-industrial byproducts in dairy ewes diets affects milk production traits and hematological parameters. Anim. Feed Sci. Technol. 2019, 251, 76–85. [Google Scholar] [CrossRef]
- Rodrigues, M.A.M.; Guedes, C.M.; Rodrigues, A.; Cone, J.W.; van Gelder, A.H.; Ferreira, L.M.M. Evaluation of the nutritive value of apple pulp mixed with different amounts of wheat straw. Livest. Res. Rural Dev. 2008, 20, 376400. [Google Scholar]
- Fegeros, K.; Zervas, G.; Stamouli, S.; Apostolaki, E. Nutritive Value of Dried Citrus Pulp and Its Effect on Milk Yield and Milk Composition of Lactating Ewes. J. Dairy Sci. 1995, 78, 1116–1121. [Google Scholar] [CrossRef]
- Dey, A.; De, P.S. Influence of Condensed Tannins from Ficus bengalensis Leaves on Feed Utilization, Milk Production and Antioxidant Status of Crossbred Cows. Asian-Australas. J. Anim. Sci. 2014, 27, 342–348. [Google Scholar] [CrossRef]
- Tsiplakou, E.; Zervas, G. The effect of dietary inclusion of olive tree leaves and grape marc on the content of conjugated linoleic acid and vaccenic acid in the milk of dairy sheep and goats. J. Dairy Res. 2008, 75, 270–278. [Google Scholar] [CrossRef]
- Abarghuei, M.J.; Rouzbehan, Y.; Alipour, D. The influence of the grape pomace on the ruminal parameters of sheep. Livest. Sci. 2010, 132, 73–79. [Google Scholar] [CrossRef]
- Bahrami, Y.; Foroozandeh, A.D.; Zamani, F.; Modarresi, M.; Eghbal-Saeid, S.; Chekani-Azar, S. Effect of diet with varying levels of dried grape pomace on dry matter digestibility and growth performance of male lambs. J. Anim. Plant Sci. 2010, 6, 605–610. [Google Scholar]
- Ishida, K.; Kishi, Y.; Oishi, K.; Hirooka, H.; Kumagai, H. Effects of feeding polyphenol-rich winery wastes on digestibility, nitrogen utilization, ruminal fermentation, antioxidant status and oxidative stress in wethers. Anim. Sci. J. 2015, 86, 260–269. [Google Scholar] [CrossRef]
- Guerra-Rivas, C.; Vieira, C.; Rubio, B.; Martínez, B.; Gallardo, B.; Mantecón, A.R.; Lavín, P.; Manso, T. Effects of grape pomace in growing lamb diets compared with vitamin E and grape seed extract on meat shelf life. Meat Sci. 2016, 116, 221–229. [Google Scholar] [CrossRef]
- Manso, T.; Gallardo, B.; Salvá, A.; Guerra-Rivas, C.; Mantecón, A.R.; Lavín, P.; de la Fuente, M.A. Influence of dietary grape pomace combined with linseed oil on fatty acid profile and milk composition. J. Dairy Sci. 2016, 99, 1111–1120. [Google Scholar] [CrossRef]
- Guerra-Rivas, C.; Gallardo, B.; Mantecón, Á.R.; Álamo-Sanza, M.; del Manso, T. Evaluation of grape pomace from red wine by-product as feed for sheep. J. Sci. Food Agric. 2017, 97, 1885–1893. [Google Scholar] [CrossRef]
- Alba, D.F.; Campigotto, G.; Cazarotto, C.J.; dos Santos, D.S.; Gebert, R.R.; Reis, J.H.; Souza, C.F.; Baldissera, M.D.; Gindri, A.L.; Kempka, A.P.; et al. Use of grape residue flour in lactating dairy sheep in heat stress: Effects on health, milk production and quality. J. Therm. Biol. 2019, 82, 197–205. [Google Scholar] [CrossRef]
- Correddu, F.; Nudda, A.; Battacone, G.; Boe, R.; Francesconi, A.H.D.; Pulina, G. Effects of grape seed supplementation, alone or associated with linseed, on ruminal metabolism in Sarda dairy sheep. Anim. Feed Sci. Technol. 2015, 199, 61–72. [Google Scholar] [CrossRef]
- Chiofalo, B.; Liotta, L.; Zumbo, A.; Chiofalo, V. Administration of olive cake for ewe feeding: Effect on milk yield and composition. Small Rumin. Res. 2004, 55, 169–176. [Google Scholar] [CrossRef]
- Molina-Alcaide, E.; Yáñez-Ruiz, D.R. Potential use of olive by-products in ruminant feeding: A review. Anim. Feed Sci. Technol. 2008, 147, 247–264. [Google Scholar] [CrossRef]
- Abbeddou, S.; Rischkowsky, B.; Richter, E.K.; Hess, H.D.; Kreuzer, M. Modification of milk fatty acid composition by feeding forages and agro-industrial byproducts from dry areas to Awassi sheep. J. Dairy Sci. 2011, 94, 4657–4668. [Google Scholar] [CrossRef]
- Tufarelli, V.; Introna, M.; Cazzato, E.; Mazzei, D.; Laudadio, V. Suitability of partly destoned exhausted olive cake as by-product feed ingredient for lamb production. J. Anim. Sci. 2013, 91, 872–877. [Google Scholar] [CrossRef]
- Villanueva, Z.; Ibarra, M.A.; Briones, F.; Escamilla, O.S. Productive performance of hair lambs fed fresh orange (Citrus sinensis) residues substituting sorghum (Sorghum vulgare) grains. Cuban J. Agric. Sci. 2013, 47, 27–31. [Google Scholar]
- Ghasemi, S.; Naserian, A.A.; Valizadeh, R.; Tahmasebi, A.M.; Vakili, A.R.; Behgar, M.; Ghovvati, S. Inclusion of pistachio hulls as a replacement for alfalfa hay in the diet of sheep causes a shift in the rumen cellulolytic bacterial population. Small Rumin. Res. 2012, 104, 94–98. [Google Scholar] [CrossRef]
- Mirzaei-Aghsaghali, A.; Maheri-Sis, N.; Mansouri, H.; Razeghi, M.E.; Mirza-Aghazadeh, A.; Cheraghi, H.; Aghajanzadeh-Golshani, A. Evaluating potential nutritive value of pomegranate processing by-products for ruminants using in vitro gas production technique. ARPN J. Agric. Biol. Sci. 2011, 6, 45–51. [Google Scholar]
- Valenti, B.; Luciano, G.; Morbidini, L.; Rossetti, U.; Codini, M.; Avondo, M.; Priolo, A.; Bella, M.; Natalello, A.; Pauselli, M. Dietary pomegranate pulp: Effect on ewe milk quality during late lactation. Animals 2019, 9, 283. [Google Scholar] [CrossRef]
- Ventura, M.R.; Pieltain, M.C.; Castanon, J.I.R. Evaluation of tomato crop by-products as feed for goats. Anim. Feed Sci. Technol. 2009, 154, 271–275. [Google Scholar] [CrossRef]
- Shdaifat, M.M.; Al-Barakah, F.S.; Kanan, A.Q.; Obeidat, B.S. The effect of feeding agricultural by-products on performance of lactating Awassi ewes. Small Rumin. Res. 2013, 113, 11–14. [Google Scholar] [CrossRef]
- Razzaghi, A.; Naserian, A.A.; Valizadeh, R.; Ebrahimi, S.H.; Khorrami, B.; Malekkhahi, M.; Khiaosa-ard, R. Pomegranate seed pulp, pistachio hulls, and tomato pomace as replacement of wheat bran increased milk conjugated linoleic acid concentrations without adverse effects on ruminal fermentation and performance of Saanen dairy goats. Anim. Feed Sci. Technol. 2015, 210, 46–55. [Google Scholar] [CrossRef]
- Denek, N.; Can, A. Feeding value of wet tomato pomace ensiled with wheat straw and wheat grain for Awassi sheep. Small Rumin. Res. 2006, 65, 260–265. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef]
- Frutos, P.; Hervás, G.; Giráldez, F.J.; Mantecón, A.R. Review. Tannins and ruminant nutrition. Span. J. Agric. Res. 2004, 2, 191–202. [Google Scholar] [CrossRef]
- Montoro, P.; Tuberoso, C.I.G.; Piacente, S.; Perrone, A.; De Feo, V.; Cabras, P.; Pizza, C. Stability and antioxidant activity of polyphenols in extracts of Myrtus communis L. berries used for the preparation of myrtle liqueur. J. Pharm. Biomed. Anal. 2006, 41, 1614–1619. [Google Scholar] [CrossRef]
- Maldini, M.; Chessa, M.; Petretto, G.L.; Montoro, P.; Rourke, J.P.; Foddai, M.; Nicoletti, M.; Pintore, G. Profiling and simultaneous quantitative determination of anthocyanins in wild Myrtus communis L. Berries from different geographical areas in Sardinia and their comparative evaluation. Phytochem. Anal. 2016, 27, 249–256. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Rosa, A.; Bifulco, E.; Melis, M.P.; Atzeri, A.; Pirisi, F.M.; Dessì, M.A. Chemical composition and antioxidant activities of Myrtus communis L. berries extracts. Food Chem. 2010, 123, 1242–1251. [Google Scholar] [CrossRef]
- Correddu, F.; Maldini, M.; Addis, R.; Petretto, G.L.; Palomba, M.; Battacone, G.; Pulina, G.; Nudda, A.; Pintore, G. Myrtus communis liquor byproduct as a source of bioactive compounds. Foods 2019, 8, 237. [Google Scholar] [CrossRef]
- Ozturk, B.; Parkinson, C.; Gonzalez-Miquel, M. Extraction of polyphenolic antioxidants from orange peel waste using deep eutectic solvents. Sep. Purif. Technol. 2018, 206, 1–13. [Google Scholar] [CrossRef]
- Besbes, S.; Blecker, C.; Deroanne, C.; Bahloul, N.; Lognay, G.; Drira, N.-E.; Attia, H. Date seed oil: Phenolic, tocopherol and sterol profiles. J. Food Lipids 2004, 11, 251–265. [Google Scholar] [CrossRef]
- Al-Farsi, M.A.; Lee, C.Y. Optimization of phenolics and dietary fibre extraction from date seeds. Food Chem. 2008, 108, 977–985. [Google Scholar] [CrossRef]
- Shams Ardekani, M.R.; Khanavi, M.; Hajimahmoodi, M.; Jahangiri, M.; Hadjiakhoondi, A. Comparison of Antioxidant Activity and Total Phenol Contents of some Date Seed Varieties from Iran. Iran. J. Pharm. Res. 2010, 9, 141–146. [Google Scholar]
- Platat, C.; M Habib, H.; AL Maqbali, F.D.; Jaber, N.N.; Ibrahim, W.H. Identification of date seeds varieties patterns to optimize nutritional benefits of date seeds. Nutr. Food Sci. 2014, 8, 2. [Google Scholar] [CrossRef]
- Bonilla, F.; Mayen, M.; Merida, J.; Medina, M. Extraction of phenolic compounds from red grape marc for use as food lipid antioxidants. Food Chem. 1999, 66, 209–215. [Google Scholar] [CrossRef]
- Alonso, Á.M.; Guillén, D.A.; Barroso, C.G.; Puertas, B.; García, A. Determination of Antioxidant Activity of Wine Byproducts and Its Correlation with Polyphenolic Content. J. Agric. Food Chem. 2002, 50, 5832–5836. [Google Scholar] [CrossRef]
- Spanghero, M.; Salem, A.Z.M.; Robinson, P.H. Chemical composition, including secondary metabolites, and rumen fermentability of seeds and pulp of Californian (USA) and Italian grape pomaces. Anim. Feed Sci. Technol. 2009, 152, 243–255. [Google Scholar] [CrossRef]
- Lutterodt, H.; Slavin, M.; Whent, M.; Turner, E.; Yu, L. Fatty acid composition, oxidative stability, antioxidant and antiproliferative properties of selected cold-pressed grape seed oils and flours. Food Chem. 2011, 128, 391–399. [Google Scholar] [CrossRef]
- Correddu, F.; Nudda, A.; Pulina, G. Effect of Myrtus communis liquor by-product on milk FA profile of Sarda dairy sheep. Unpublished.
- Obied, H.K.; Bedgood, D.R.; Prenzler, P.D.; Robards, K. Bioscreening of Australian olive mill waste extracts: Biophenol content, antioxidant, antimicrobial and molluscicidal activities. Food Chem. Toxicol. 2007, 45, 1238–1248. [Google Scholar] [CrossRef]
- Suárez, M.; Romero, M.-P.; Ramo, T.; Macià, A.; Motilva, M.-J. Methods for Preparing Phenolic Extracts from Olive Cake for Potential Application as Food Antioxidants. J. Agric. Food Chem. 2009, 57, 1463–1472. [Google Scholar] [CrossRef]
- Leouifoudi, I.; Harnafi, H.; Zyad, A. Olive mill waste extracts: Polyphenols content, antioxidant, and antimicrobial activities. Adv. Pharmacol. Sci. 2015, 2015, 714138. [Google Scholar] [CrossRef]
- Ryan, D.; Prenzler, P.D.; Lavee, S.; Antolovich, M.; Robards, K. Quantitative Changes in Phenolic Content during Physiological Development of the Olive (Olea europaea) Cultivar Hardy’s Mammoth. J. Agric. Food Chem. 2003, 51, 2532–2538. [Google Scholar] [CrossRef]
- Erşan, S.; Güçlü Üstündağ, Ö.; Carle, R.; Schweiggert, R.M. Identification of Phenolic Compounds in Red and Green Pistachio (Pistacia vera L.) Hulls (Exo- and Mesocarp) by HPLC-DAD-ESI-(HR)-MSn. J. Agric. Food Chem. 2016, 64, 5334–5344. [Google Scholar] [CrossRef]
- Grace, M.H.; Esposito, D.; Timmers, M.A.; Xiong, J.; Yousef, G.; Komarnytsky, S.; Lila, M.A. Chemical composition, antioxidant and anti-inflammatory properties of pistachio hull extracts. Food Chem. 2016, 210, 85–95. [Google Scholar] [CrossRef]
- Elfalleh, W. Total phenolic contents and antioxidant activities of pomegranate peel, seed, leaf and flower. J. Med. Plants Res. 2012, 6, 4724–4730. [Google Scholar] [CrossRef]
- Ambigaipalan, P.; de Camargo, A.C.; Shahidi, F. Phenolic Compounds of Pomegranate Byproducts (Outer Skin, Mesocarp, Divider Membrane) and Their Antioxidant Activities. J. Agric. Food Chem. 2016, 64, 6584–6604. [Google Scholar] [CrossRef]
- Gullon, B.; Pintado, M.E.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Assessment of polyphenolic profile and antibacterial activity of pomegranate peel (Punica granatum) flour obtained from co-product of juice extraction. Food Control 2016, 59, 94–98. [Google Scholar] [CrossRef]
- Abarghuei, M.J.; Rouzbehan, Y.; Salem, A.Z.M.; Zamiri, M.J. Nitrogen balance, blood metabolites and milk fatty acid composition of dairy cows fed pomegranate-peel extract. Livest. Sci. 2014, 164, 72–80. [Google Scholar] [CrossRef]
- Rice-evans, C.A.; Miller, N.J.; Bolwell, P.G.; Bramley, P.M.; Pridham, J.B. The relative antioxidant activities of plant-derived polyphenolic Flavonoids. Free Radic. Res. 1995, 22, 375–383. [Google Scholar] [CrossRef]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Duarte, J.; Jimenez, R.; Santos-Buelga, C.; Osuna, A. Antihypertensive effects of the flavonoid quercetin. Pharmacol. Rep. 2009, 61, 67–75. [Google Scholar] [CrossRef]
- Kubena, K.S.; McMurray, D.N. Nutrition and the Immune System: A Review of Nutrient–Nutrient Interactions. J. Am. Diet. Assoc. 1996, 96, 1156–1164. [Google Scholar] [CrossRef]
- Bahadoran, Z.; Mirmiran, P.; Azizi, F. Dietary polyphenols as potential nutraceuticals in management of diabetes: A review. J. Diabetes Metab. Disord. 2013, 12, 43. [Google Scholar] [CrossRef]
- Stoner, G.D.; Mukhtar, H. Polyphenols as cancer chemopreventive agents. J. Cell. Biochem. 1995, 59, 169–180. [Google Scholar] [CrossRef]
- Forester, S.C.; Choy, Y.Y.; Waterhouse, A.L.; Oteiza, P.I. The anthocyanin metabolites gallic acid, 3-O-methylgallic acid, and 2,4,6-trihydroxybenzaldehyde decrease human colon cancer cell viability by regulating pro-oncogenic signals. Mol. Carcinog. 2014, 53, 432–439. [Google Scholar] [CrossRef]
- Koeberle, A.; Werz, O. Multi-target approach for natural products in inflammation. Drug Discov. Today 2014, 19, 1871–1882. [Google Scholar] [CrossRef]
- Makkar, H.P.S. Effects and fate of tannins in ruminant animals, adaptation to tannins, and strategies to overcome detrimental effects of feeding tannin-rich feeds. Small Rumin. Res. 2003, 49, 241–256. [Google Scholar] [CrossRef]
- Wang, Y.; Waghorn, G.C.; McNabb, W.C.; Barry, T.N.; Hedley, M.J.; Shelton, I.D. Effect of condensed tannins in Lotus corniculatus upon the digestion of methionine and cysteine in the small intestine of sheep. J. Agric. Sci. 1996, 127, 413–421. [Google Scholar] [CrossRef]
- Toral, P.G.; Hervás, G.; Bichi, E.; Belenguer, Á.; Frutos, P. Tannins as feed additives to modulate ruminal biohydrogenation: Effects on animal performance, milk fatty acid composition and ruminal fermentation in dairy ewes fed a diet containing sunflower oil. Anim. Feed Sci. Technol. 2011, 164, 199–206. [Google Scholar] [CrossRef]
- Toral, P.G.; Monahan, F.J.; Hervás, G.; Frutos, P.; Moloney, A.P. Review: Modulating ruminal lipid metabolism to improve the fatty acid composition of meat and milk. Challenges and opportunities. Animal 2018, 12, s272–s281. [Google Scholar] [CrossRef]
- Athanasiadou, S.; Kyriazakis, I.; Jackson, F.; Coop, R.L. Direct anthelmintic effects of condensed tannins towards different gastrointestinal nematodes of sheep: In vitro and in vivo studies. Vet. Parasitol. 2001, 99, 205–219. [Google Scholar] [CrossRef]
- Athanasiadou, S.; Kyriazakis, I.; Jackson, F. Can plant secondary metabolites have a role in controlling gastrointestinal nematode parasitism in small ruminants? BSAP Occas. Publ. 2006, 34, 197–207. [Google Scholar] [CrossRef]
- Pathak, A.K. Potential of using condensed tannins to control gastrointestinal nematodes and improve small ruminant performance. Int. J. Mol. Vet. Res. 2013, 3. [Google Scholar] [CrossRef]
- Gessner, D.K.; Ringseis, R.; Eder, K. Potential of plant polyphenols to combat oxidative stress and inflammatory processes in farm animals. J. Anim. Physiol. Anim. Nutr. 2017, 101, 605–628. [Google Scholar] [CrossRef]
- Liu, H.W.; Zhou, D.W.; Li, K. Effects of chestnut tannins on performance and antioxidative status of transition dairy cows. J. Dairy Sci. 2013, 96, 5901–5907. [Google Scholar] [CrossRef]
- López-Andrés, P.; Luciano, G.; Vasta, V.; Gibson, T.M.; Biondi, L.; Priolo, A.; Mueller-Harvey, I. Dietary quebracho tannins are not absorbed, but increase the antioxidant capacity of liver and plasma in sheep. Br. J. Nutr. 2013, 110, 632–639. [Google Scholar] [CrossRef]
- Alcaide, E.M.; Ruiz, D.Y.; Moumen, A.; Garcıa, A.M. Ruminal degradability and in vitro intestinal digestibility of sunflower meal and in vitro digestibility of olive by-products supplemented with urea or sunflower meal: comparison between goats and sheep. Anim. Feed Sci. Technol. 2003, 110, 3–15. [Google Scholar] [CrossRef]
- Yáñez Ruiz, D.R.; Moumen, A.; Martin Garcia, A.I.; Molina Alcaide, E. Ruminal fermentation and degradation patterns, protozoa population, and urinary purine derivatives excretion in goats and wethers fed diets based on two-stage olive cake: Effect of PEG supply. J. Anim. Sci. 2004, 82, 2023–2032. [Google Scholar] [CrossRef]
- Ghasemi, S.; Naserian, A.A.; Valizadeh, R.; Tahmasebi, A.M.; Vakili, A.R.; Behgar, M. Effects of pistachio by-product in replacement of lucerne hay on microbial protein synthesis and fermentative parameters in the rumen of sheep. Anim. Prod. Sci. 2012, 52, 1052–1057. [Google Scholar] [CrossRef]
- Nudda, A.; Correddu, F.; Marzano, A.; Battacone, G.; Nicolussi, P.; Bonelli, P.; Pulina, G. Effects of diets containing grape seed, linseed, or both on milk production traits, liver and kidney activities, and immunity of lactating dairy ewes. J. Dairy Sci. 2015, 98, 1157–1166. [Google Scholar] [CrossRef]
- Ghaffari, M.H.; Tahmasbi, A.M.; Khorvash, M.; Naserian, A.A.; Ghaffari, A.H.; Valizadeh, H. Effects of pistachio by-products in replacement of alfalfa hay on populations of rumen bacteria involved in biohydrogenation and fermentative parameters in the rumen of sheep. J. Anim. Physiol. Anim. Nutr. 2014, 98, 578–586. [Google Scholar] [CrossRef]
- Sedighi-Vesagh, R.; Naserian, A.A.; Ghaffari, M.H.; Petit, H.V. Effects of pistachio by-products on digestibility, milk production, milk fatty acid profile and blood metabolites in Saanen dairy goats. J. Anim. Physiol. Anim. Nutr. 2015, 99, 777–787. [Google Scholar] [CrossRef]
- Sharifi, M.; Bashtani, M.; Naserian, A.A.; Farhangfar, H. The effect of feeding low quality date palm (Phoenix dactylifera L.) on the performance, antioxidant status and ruminal fermentation of mid-lactating Saanen dairy goats. Small Rumin. Res. 2015, 130, 95–100. [Google Scholar] [CrossRef]
- Hofmann, R.R. Evolutionary steps of ecophysiological adaptation and diversification of ruminants: A comparative view of their digestive system. Oecologia 1989, 78, 443–457. [Google Scholar] [CrossRef]
- Alonso-Díaz, M.A.; Torres-Acosta, J.F.J.; Sandoval-Castro, C.A.; Capetillo-Leal, C.M. Amino acid profile of the protein from whole saliva of goats and sheep and its interaction with tannic acid and tannins extracted from the fodder of tropical plants. Small Rumin. Res. 2012, 103, 69–74. [Google Scholar] [CrossRef]
- Domingue, B.M.F.; Dellow, D.W.; Barry, T.N. The efficiency of chewing during eating and ruminating in goats and sheep. Br. J. Nutr. 1991, 65, 355–363. [Google Scholar] [CrossRef]
- Lamy, E.; da Costa, G.; e Silva, F.C.; Potes, J.; Coelho, A.V.; Baptista, E.S. Comparison of Electrophoretic Protein Profiles from Sheep and Goat Parotid Saliva. J. Chem. Ecol. 2008, 34, 388–397. [Google Scholar] [CrossRef]
- Lamy, E.; Rawel, H.; Schweigert, F.J.; Capela e Silva, F.; Ferreira, A.; Costa, A.R.; Antunes, C.; Almeida, A.M.; Coelho, A.V.; Sales-Baptista, E. The Effect of Tannins on Mediterranean Ruminant Ingestive Behavior: The Role of the Oral Cavity. Molecules 2011, 16, 2766–2784. [Google Scholar] [CrossRef]
- Leparmarai, P.T.; Sinz, S.; Kunz, C.; Liesegang, A.; Ortmann, S.; Kreuzer, M.; Marquardt, S. Transfer of total phenols from a grapeseed-supplemented diet to dairy sheep and goat milk, and effects on performance and milk quality. J. Anim. Sci. 2019, 97, 1840–1851. [Google Scholar] [CrossRef]
- Austin, P.J.; Suchar, L.A.; Robbins, C.T.; Hagerman, A.E. Tannin-binding proteins in saliva of deer and their absence in saliva of sheep and cattle. J. Chem. Ecol. 1989, 15, 1335–1347. [Google Scholar] [CrossRef]
- Mancilla-Leytón, J.M.; Vicente, A.M.; Delgado-Pertíñez, M. Summer diet selection of dairy goats grazing in a Mediterranean shrubland and the quality of secreted fat. Small Rumin. Res. 2013, 113, 437–445. [Google Scholar] [CrossRef]
- Silanikove, N. Why goats raised on harsh environment perform better than other domesticated animals. Options Mediterr. 1997, 34, 185–194. [Google Scholar]
- McMahon, L.R.; McAllister, T.A.; Berg, B.P.; Majak, W.; Acharya, S.N.; Popp, J.D.; Coulman, B.E.; Wang, Y.; Cheng, K.J. A review of the effects of forage condensed tannins on ruminal fermentation and bloat in grazing cattle. Can. J. Plant Sci. 2000, 80, 469–485. [Google Scholar] [CrossRef]
- Schreurs, N.M.; Tavendale, M.H.; Lane, G.A.; Barry, T.N.; Lopez-Villalobos, N.; McNabb, W.C. Effect of different condensed tannin-containing forages, forage maturity and nitrogen fertiliser application on the formation of indole and skatole in in vitro rumen fermentations. J. Sci. Food Agric. 2007, 87, 1076–1087. [Google Scholar] [CrossRef]
- Cabiddu, A.; Salis, L.; Tweed, J.K.; Molle, G.; Decandia, M.; Lee, M.R. The influence of plant polyphenols on lipolysis and biohydrogenation in dried forages at different phenological stages: in vitro study. J. Sci. Food Agric. 2010, 90, 829–835. [Google Scholar]
- Ghaffari, M.H.; Tahmasbi, A.M.; Khorvash, M.; Naserian, A.A.; Vakili, A.R. Effects of pistachio by-products in replacement of alfalfa hay on ruminal fermentation, blood metabolites, and milk fatty acid composition in Saanen dairy goats fed a diet containing fish oil. J. Appl. Anim. Res. 2014, 42, 186–193. [Google Scholar] [CrossRef]
- Romero, M.J.; Madrid, J.; Hernández, F.; Cerón, J.J. Digestibility and voluntary intake of vine leaves (Vitis vinifera L.) by sheep. Small Rumin. Res. 2000, 38, 191–195. [Google Scholar] [CrossRef]
- García, A.M.; Moumen, A.; Ruiz, D.Y.; Alcaide, E.M. Chemical composition and nutrients availability for goats and sheep of two-stage olive cake and olive leaves. Anim. Feed Sci. Technol. 2003, 107, 61–74. [Google Scholar] [CrossRef]
- Awawdeh, M.S. Alternative feedstuffs and their effects on performance of Awassi sheep: A review. Trop. Anim. Health Prod. 2011, 43, 1297–1309. [Google Scholar] [CrossRef]
- Molina Alcaide, E.; Yáñez Ruiz, D.; Moumen, A.; Martín García, I. Chemical composition and nitrogen availability for goats and sheep of some olive by-products. Small Rumin. Res. 2003, 49, 329–336. [Google Scholar] [CrossRef]
- Yáñez-Ruiz, D.R.; Molina-Alcaide, E. A comparative study of the effect of two-stage olive cake added to alfalfa on digestion and nitrogen losses in sheep and goats. Animal 2007, 1, 227–232. [Google Scholar] [CrossRef]
- Perez-Maldonado, R.A.; Norton, B.W. Digestion of 14 C-labelled condensed tannins from Desmodium intortum in sheep and goats. Br. J. Nutr. 1996, 76, 501–513. [Google Scholar] [CrossRef]
- Brooker, J.D.; O’donovan, L.A.; Skene, I.; Clarke, K.; Blackall, L.; Muslera, P. Streptococcus caprinus sp. nov., a tannin-resistant ruminal bacterium from feral goats. Lett. Appl. Microbiol. 1994, 18, 313–318. [Google Scholar] [CrossRef]
- Paraskevakis, N. Effects of dietary dried Greek Oregano (Origanum vulgare ssp. hirtum) supplementation on blood and milk enzymatic antioxidant indices, on milk total antioxidant capacity and on productivity in goats. Anim. Feed Sci. Technol. 2015, 209, 90–97. [Google Scholar] [CrossRef]
- Moñino, I.; Martínez, C.; Sotomayor, J.A.; Lafuente, A.; Jordán, M.J. Polyphenolic Transmission to Segureño Lamb Meat from Ewes’ Diet Supplemented with the Distillate from Rosemary (Rosmarinus officinalis) Leaves. J. Agric. Food Chem. 2008, 56, 3363–3367. [Google Scholar] [CrossRef]
- Jordán, M.J.; Moñino, M.I.; Martínez, C.; Lafuente, A.; Sotomayor, J.A. Introduction of Distillate Rosemary Leaves into the Diet of the Murciano-Granadina Goat: Transfer of Polyphenolic Compounds to Goats’ Milk and the Plasma of Suckling Goat Kids. J. Agric. Food Chem. 2010, 58, 8265–8270. [Google Scholar] [CrossRef]
- Halliwell, B.; Rafter, J.; Jenner, A. Health promotion by flavonoids, tocopherols, tocotrienols, and other phenols: Direct or indirect effects? Antioxidant or not? Am. J. Clin. Nutr. 2005, 81, 268S–276S. [Google Scholar] [CrossRef]
- Kerem, Z.; Chetrit, D.; Shoseyov, O.; Regev-Shoshani, G. Protection of Lipids from Oxidation by Epicatechin, trans-Resveratrol, and Gallic and Caffeic Acids in Intestinal Model Systems. J. Agric. Food Chem. 2006, 54, 10288–10293. [Google Scholar] [CrossRef]
- Torres-Acosta, J.F.J.; Hoste, H. Alternative or improved methods to limit gastro-intestinal parasitism in grazing sheep and goats. Small Rumin. Res. 2008, 77, 159–173. [Google Scholar] [CrossRef]
- Borges, D.G.L.; Borges, F.D.A. Plants and their medicinal potential for controlling gastrointestinal nematodes in ruminants. Nematoda 2016, 3, 1–7. [Google Scholar] [CrossRef]
- Windisch, W.; Schedle, K.; Plitzner, C.; Kroismayr, A. Use of phytogenic products as feed additives for swine and poultry. J. Anim. Sci. 2008, 86, E140–E148. [Google Scholar] [CrossRef]
- Correddu, F.; Fancello, F.; Chessa, L.; Atzori, A.S.; Pulina, G.; Nudda, A. Effects of supplementation with exhausted myrtle berries on rumen function of dairy sheep. Small Rumin. Res. 2019, 170, 51–61. [Google Scholar] [CrossRef]
- Tavendale, M.H.; Meagher, L.P.; Pacheco, D.; Walker, N.; Attwood, G.T.; Sivakumaran, S. Methane production from in vitro rumen incubations with Lotus pedunculatus and Medicago sativa, and effects of extractable condensed tannin fractions on methanogenesis. Anim. Feed Sci. Technol. 2005, 123–124, 403–419. [Google Scholar] [CrossRef]
- Goel, G.; Makkar, H.P.S. Methane mitigation from ruminants using tannins and saponins. Trop. Anim. Health Prod. 2012, 44, 729–739. [Google Scholar] [CrossRef]
- Bhatta, R.; Uyeno, Y.; Tajima, K.; Takenaka, A.; Yabumoto, Y.; Nonaka, I.; Enishi, O.; Kurihara, M. Difference in the nature of tannins on in vitro ruminal methane and volatile fatty acid production and on methanogenic archaea and protozoal populations. J. Dairy Sci. 2009, 92, 5512–5522. [Google Scholar] [CrossRef]
- Arco-Pérez, A.; Ramos-Morales, E.; Yáñez-Ruiz, D.R.; Abecia, L.; Martín-García, A.I. Nutritive evaluation and milk quality of including of tomato or olive by-products silages with sunflower oil in the diet of dairy goats. Anim. Feed Sci. Technol. 2017, 232, 57–70. [Google Scholar] [CrossRef]
- Mannelli, F.; Cappucci, A.; Pini, F.; Pastorelli, R.; Decorosi, F.; Giovannetti, L.; Mele, M.; Minieri, S.; Conte, G.; Pauselli, M.; et al. Effect of different types of olive oil pomace dietary supplementation on the rumen microbial community profile in Comisana ewes. Sci. Rep. 2018, 8, 8455. [Google Scholar] [CrossRef]
- Marcos, C.N.; de Evan, T.; Molina-Alcaide, E.; Carro, M.D. Nutritive Value of Tomato Pomace for Ruminants and Its Influence on In Vitro Methane Production. Animals 2019, 9, 343. [Google Scholar] [CrossRef]
- Modaresi, J.; Fathi Nasri, M.H.; Rashidi, L.; Dayani, O.; Kebreab, E. Short communication: Effects of supplementation with pomegranate seed pulp on concentrations of conjugated linoleic acid and punicic acid in goat milk. J. Dairy Sci. 2011, 94, 4075–4080. [Google Scholar] [CrossRef]
- Boutoial, K.; Ferrandini, E.; Rovira, S.; García, V.; López, M.B. Effect of feeding goats with rosemary (Rosmarinus officinalis spp.) by-product on milk and cheese properties. Small Rumin. Res. 2013, 112, 147–153. [Google Scholar] [CrossRef]
- Abbeddou, S.; Rischkowsky, B.; Hilali, M.E.-D.; Haylani, M.; Hess, H.D.; Kreuzer, M. Supplementing diets of Awassi ewes with olive cake and tomato pomace: on-farm recovery of effects on yield, composition and fatty acid profile of the milk. Trop. Anim. Health Prod. 2015, 47, 145–152. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): A prospective cohort study. Lancet 2017, 390, 2050–2062. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Banu, J.; Rahman, M.; Causey, J.; Fernandes, G. Biological effects of conjugated linoleic acids in health and disease. J. Nutr. Biochem. 2006, 17, 789–810. [Google Scholar] [CrossRef]
- Sofi, F.; Buccioni, A.; Cesari, F.; Gori, A.M.; Minieri, S.; Mannini, L.; Casini, A.; Gensini, G.F.; Abbate, R.; Antongiovanni, M. Effects of a dairy product (pecorino cheese) naturally rich in cis-9, trans-11 conjugated linoleic acid on lipid, inflammatory and haemorheological variables: A dietary intervention study. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 117–124. [Google Scholar] [CrossRef]
- Pintus, S.; Murru, E.; Carta, G.; Cordeddu, L.; Batetta, B.; Accossu, S.; Pistis, D.; Uda, S.; Ghiani, M.E.; Mele, M.; et al. Sheep cheese naturally enriched in α-linolenic, conjugated linoleic and vaccenic acids improves the lipid profile and reduces anandamide in the plasma of hypercholesterolaemic subjects. Br. J. Nutr. 2013, 109, 1453–1462. [Google Scholar] [CrossRef]
- Jenkins, T.C.; Wallace, R.J.; Moate, P.J.; Mosley, E.E. Board-invited review: Recent advances in biohydrogenation of unsaturated fatty acids within the rumen microbial ecosystem. J. Anim. Sci. 2008, 86, 397–412. [Google Scholar] [CrossRef]
- Vasta, V.; Makkar, H.P.S.; Mele, M.; Priolo, A. Ruminal biohydrogenation as affected by tannins in vitro. Br. J. Nutr. 2008, 102, 82–92. [Google Scholar] [CrossRef]
- Khiaosa-Ard, R.; Bryner, S.F.; Scheeder, M.R.L.; Wettstein, H.-R.; Leiber, F.; Kreuzer, M.; Soliva, C.R. Evidence for the inhibition of the terminal step of ruminal α-linolenic acid biohydrogenation by condensed tannins. J. Dairy Sci. 2009, 92, 177–188. [Google Scholar] [CrossRef]
- Correddu, F.; Gaspa, G.; Pulina, G.; Nudda, A. Grape seed and linseed, alone and in combination, enhance unsaturated fatty acids in the milk of Sarda dairy sheep. J. Dairy Sci. 2016, 99, 1725–1735. [Google Scholar] [CrossRef]
- Moate, P.J.; Williams, S.R.O.; Torok, V.A.; Hannah, M.C.; Ribaux, B.E.; Tavendale, M.H.; Eckard, R.J.; Jacobs, J.L.; Auldist, M.J.; Wales, W.J. Grape marc reduces methane emissions when fed to dairy cows. J. Dairy Sci. 2014, 97, 5073–5087. [Google Scholar] [CrossRef]
- Bodas, R.; Manso, T.; Mantecón, Á.R.; Juárez, M.; De la Fuente, M.Á.; Gómez-Cortés, P. Comparison of the Fatty Acid Profiles in Cheeses from Ewes Fed Diets Supplemented with Different Plant Oils. J. Agric. Food Chem. 2010, 58, 10493–10502. [Google Scholar] [CrossRef]
- Kliem, K.E.; Shingfield, K.J. Manipulation of milk fatty acid composition in lactating cows: Opportunities and challenges. Eur. J. Lipid Sci. Technol. 2016, 118, 1661–1683. [Google Scholar] [CrossRef]
- Nudda, A.; Battacone, G.; Boaventura Neto, O.; Cannas, A.; Francesconi, A.H.D.; Atzori, A.S.; Pulina, G. Feeding strategies to design the fatty acid profile of sheep milk and cheese. Revista Brasileira de Zootecnia 2014, 43, 445–456. [Google Scholar] [CrossRef]
- Correddu, F.; Nudda, A.; Manca, M.G.; Pulina, G.; Dalsgaard, T.K. Light-Induced Lipid Oxidation in Sheep Milk: Effects of Dietary Grape Seed and Linseed, Alone or in Combination, on Milk Oxidative Stability. J. Agric. Food Chem. 2015, 63, 3980–3986. [Google Scholar] [CrossRef]
- Armendáriz, V.; Armenia, S.; Atzori, A.S. Systemic Analysis of Food Supply and Distribution Systems in City-Region Systems—An Examination of FAO’s Policy Guidelines towards Sustainable Agri-Food Systems. Agriculture 2016, 6, 65. [Google Scholar] [CrossRef]
- Turner, B.L.; Menendez, H.M.; Gates, R.; Tedeschi, L.O.; Atzori, A.S. System Dynamics Modeling for Agricultural and Natural Resource Management Issues: Review of Some Past Cases and Forecasting Future Roles. Resources 2016, 5, 40. [Google Scholar] [CrossRef]
- Molina Benavides, R.A.; Sánchez Guerrero, H.; Campos Gaona, R.; Atzori, A.S.; Morales, J.D.; Molina Benavides, R.A.; Sánchez Guerrero, H.; Campos Gaona, R.; Atzori, A.S.; Morales, J.D. Dynamic estimation of greenhouse gas emissions from bovine livestock of Valle del Cauca, Colombia. Acta Agronómica 2017, 66, 422–429. [Google Scholar] [CrossRef]
- Sterman, J. Business Dynamics: Systems Thinking and Modeling for a Complex World 2000. Available online: https://dk.um.si/IzpisGradiva.php?id=28601 (accessed on 12 September 2019).
- Pulina, G.; Milán, M.J.; Lavín, M.P.; Theodoridis, A.; Morin, E.; Capote, J.; Thomas, D.L.; Francesconi, A.H.D.; Caja, G. Invited review: Current production trends, farm structures, and economics of the dairy sheep and goat sectors. J. Dairy Sci. 2018, 101, 6715–6729. [Google Scholar] [CrossRef]
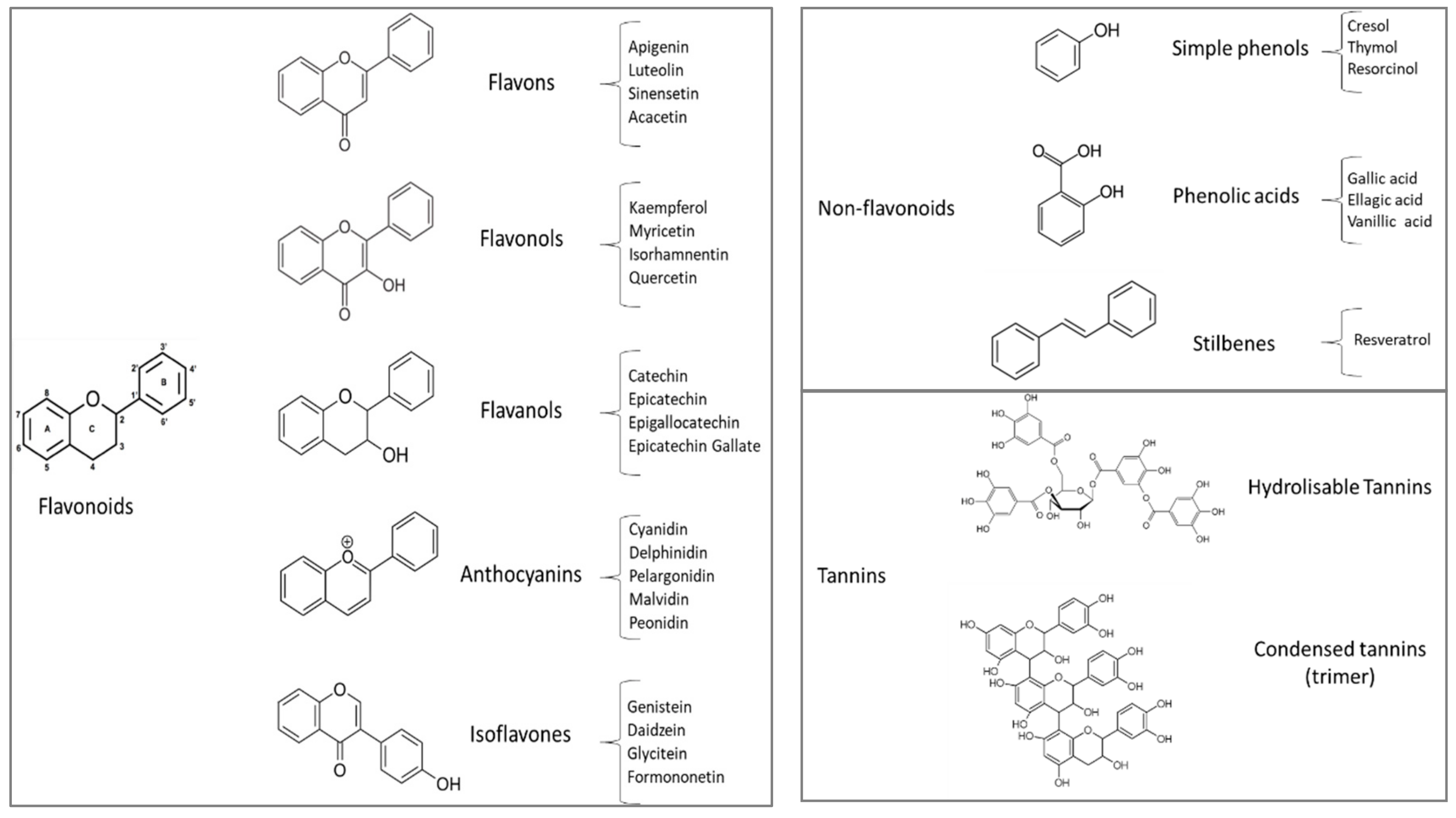
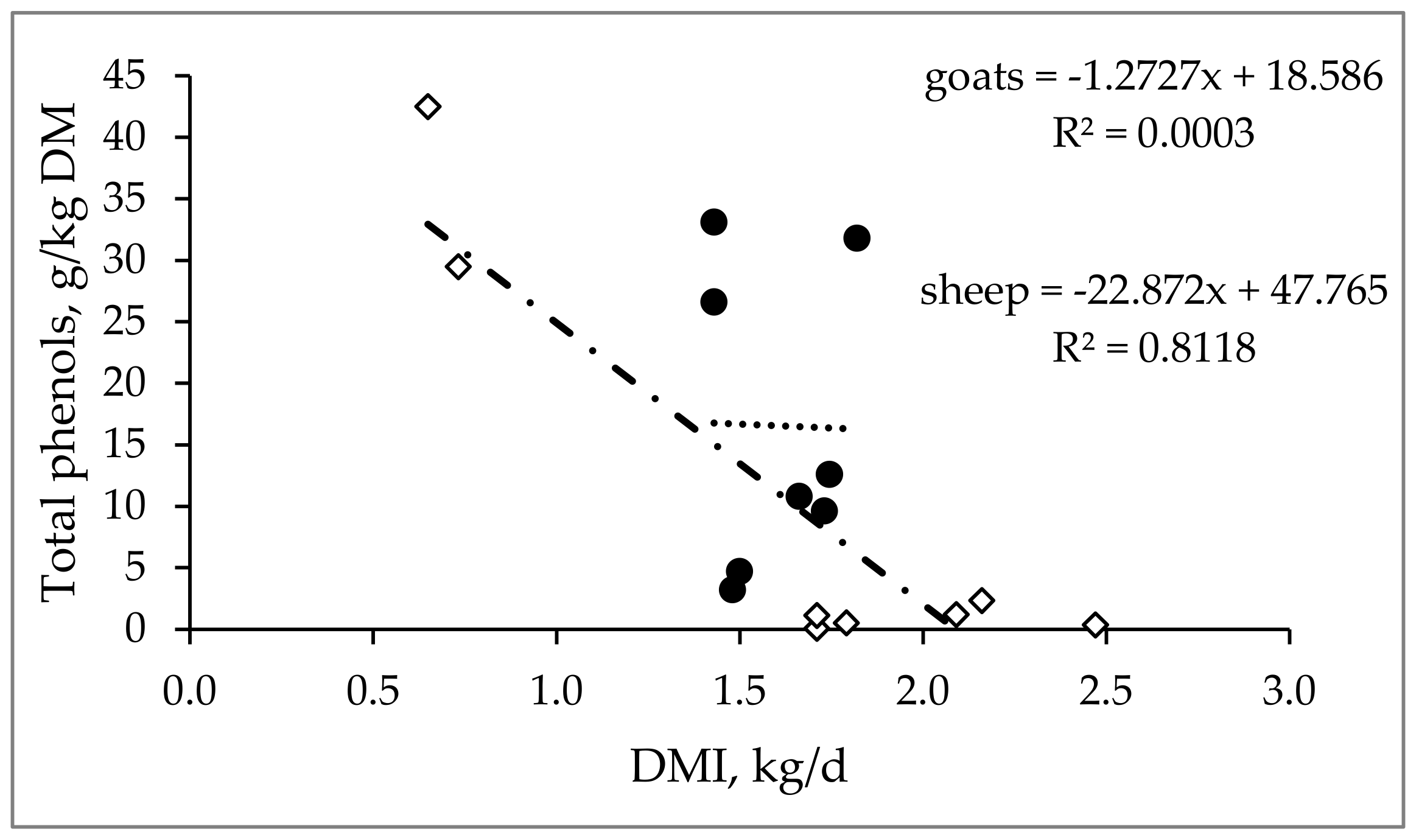
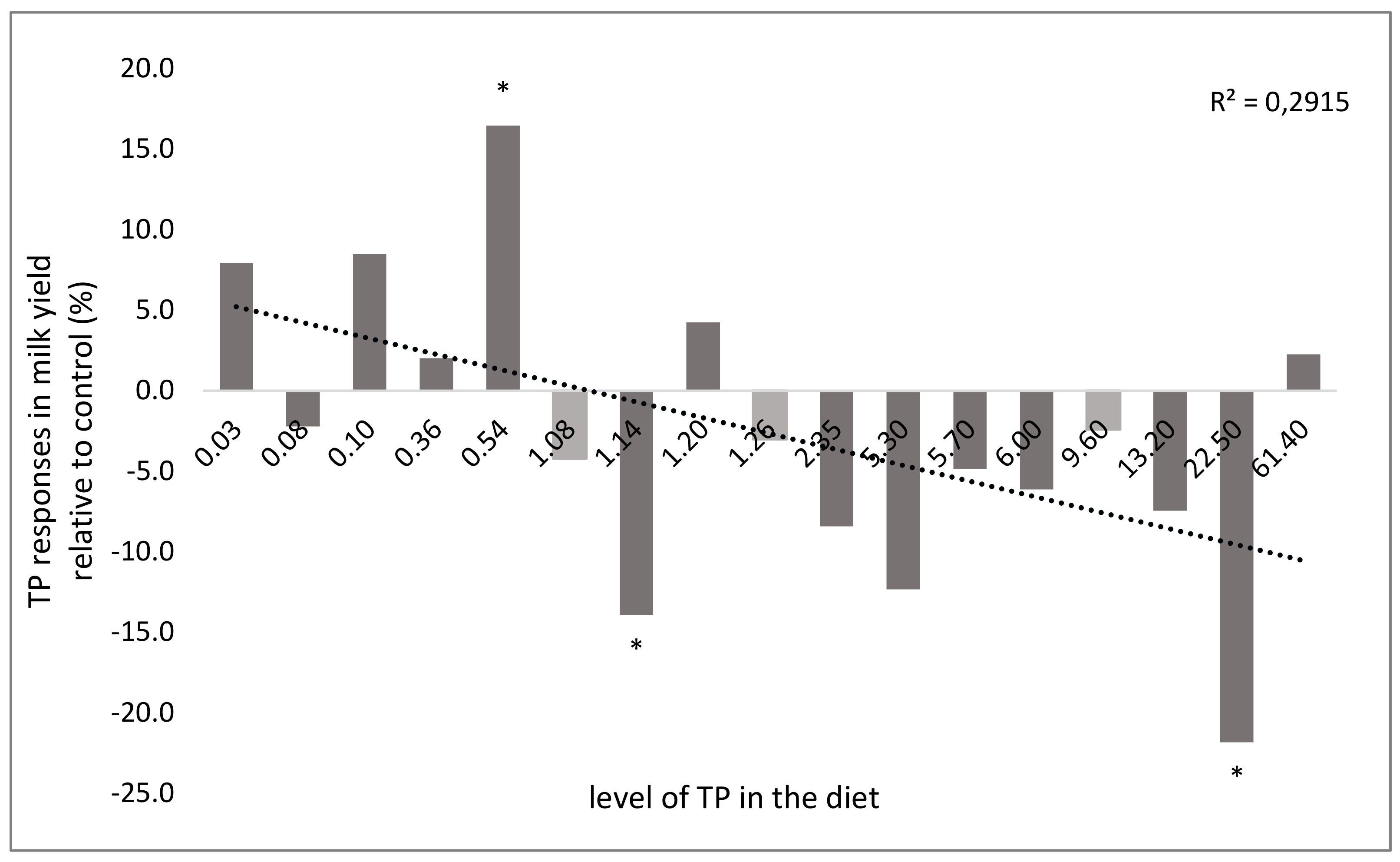
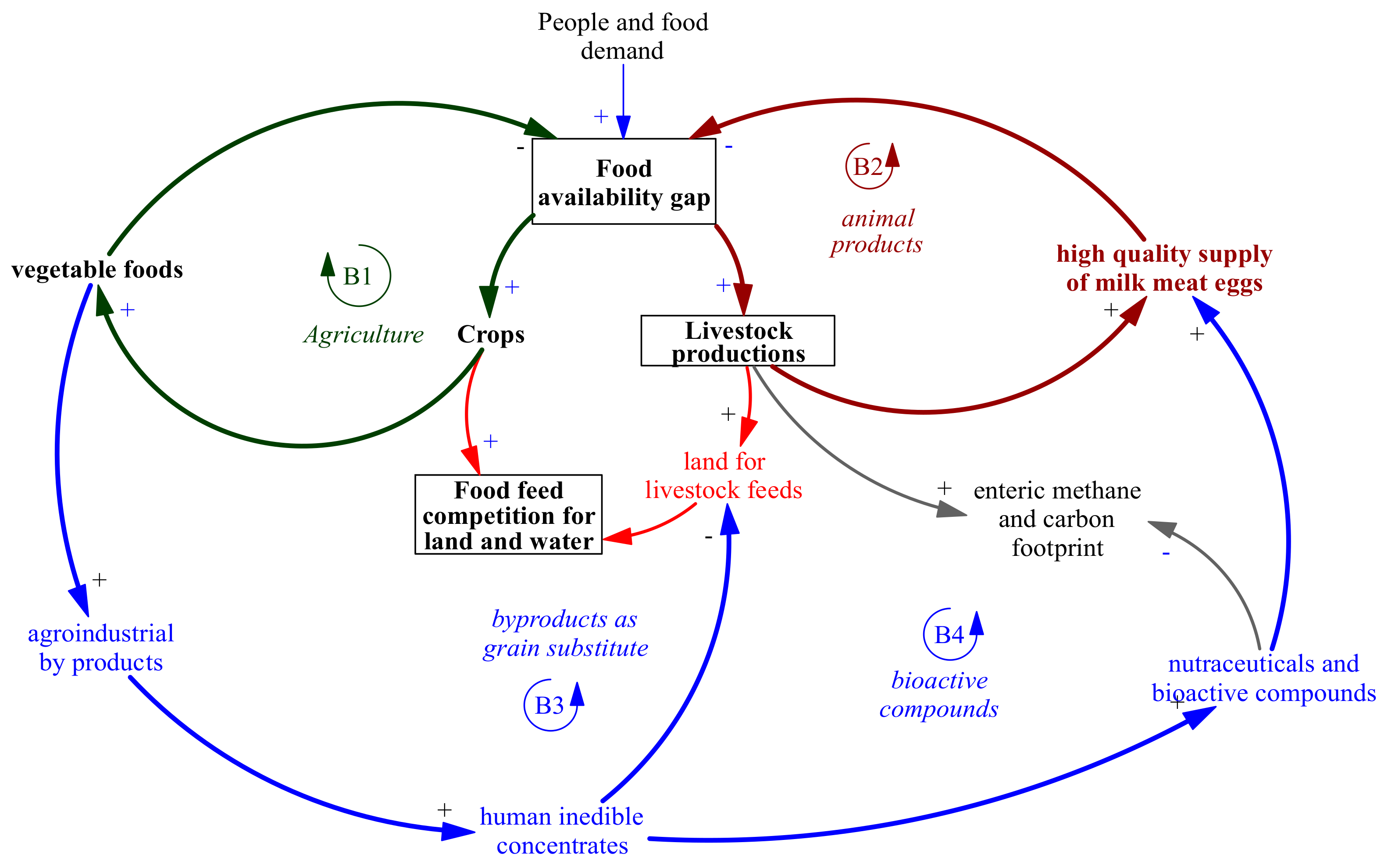
| Chemical Composition 1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| By-Products | DM | OM | NDF | ADF | NFC | CP | Lignin | EE | Ash | Reference |
| Apple | 179 | - | 107 | 80 | 842 | 4 | 24 | - | - | [18] |
| Citrus pulp | 904 | 831 | 194 | 128 | 510 * | 77 | - | 49 | 168 | [19] |
| Citrus pulp | 937 | - | 230 | 162 | 604 * | 50 | 38 | 26 | 90 | [15] |
| Exhausted myrtle berries | 970 | - | 670 | 533 | 292 | 80 | 348 | 110 | 28 | [16] |
| Exhausted myrtle berries | 943 | - | 648 | 517 | 183 | 78 | 308 | 54 | 37 | [17] |
| Ficus bengalensis | - | 852 | 425 | 369 | 279 * | 109 | - | 39 | 148 | [20] |
| Grape marc | 910 | 938 | 558 | 465 | - | 113 | - | 89 | - | [21] |
| Grape marc | 934 | - | 527 | 389 | 206 | 111 | 260 | 69 | 87 | [17] |
| Grape pomace | 525 | 940 | 568 | 476 | - | 94 | 200 | 52 | - | [22] |
| Grape pomace | 890 | 471 | 312 | - | 128 | - | - | - | [23] | |
| Grape pomace | 439 | 918 | 474 | 440 | 263 | 95 | - | 85 | 82 | [24] |
| Grape pomace | 950 | - | 376 | 317 | - | 119 | - | 73 | 89 | [25] |
| Grape pomace | - | 866 | 376 | 317 | - | 122 | 207 | 64 | - | [26] |
| Grape pulp | - | 811 | 243 | 193 | - | 138 | 75 | 32 | - | [27] |
| Grape residual flour | 934 | - | 333 | 217 | - | 103 | - | 50 | 124 | [28] |
| Grape seed | 974 | 539 | - | 231 | 93 | 411 | 109 | 27 | [29] | |
| Grape seed | - | 927 | 523 | 454 | - | 104 | 353 | 99 | - | [27] |
| Olive cake | 908 | - | 665 | 497 | - | 32.8 | 308 | 221 | 19 | [30] |
| Olive cake | 805 | 901 | 676 | 544 | - | 73 | 289 | 54 | - | [31] |
| Olive cake | 947 | 864 | 584 | 459 | 109 | 79 | 237 | 92 | 136 | [32] |
| Olive cake (exhausted) | 950 | - | 683 | 531 | 317 | 102 | 367 | 12 | 97 | [33] |
| Orange residue (fresh) | 219 | - | 227 | 171 | 657 * | 60 | 17 | 24 | 32 | [34] |
| Orange peel | 266 | - | 100 | 76 | 35 | 18 | 17 | 38 | [15] | |
| Pistachio | 900 | 755 | 259 | - | - | 153 | - | 58 | - | [35] |
| Pomegranate (peel) | 961 | - | 208 | 151 | 696 | 36 | - | 6 | 54 | [36] |
| Pomegranate pulp | 912 | - | 314 | 228 | - | 69 | 69 | 26 | 36 | [37] |
| Pomegranate (seeds) | 951 | - | 680 | 490 | 135 | 154 | - | 6 | 24 | [36] |
| Tomato fruit | 69 | - | 260 | 217 | 465 * | 170 | 195 | 28 | 77 | [38] |
| Tomato pomace | 952 | 952 | 552 | 462 | 109 | 191 | 259 | 100 | 48 | [32] |
| Tomato pomace | 85.1 | 966 | 500 | 340 | - | 194 | - | - | - | [39] |
| Tomato pomace | 94.1 | 955 | 554 | 422 | - | 217 | - | 93 | - | [40] |
| Tomato pomace | 926 | - | 616 | 507 | 121 | 157 | 313 | 62 | 44 | [17] |
| Tomato whole plant | 177 | - | 457 | 356 | 276 * | 74 | 128 | 12 | 181 | [38] |
| Wet tomato pomace | 142 | 962 | 636 | 435 | - | 195 | - | - | - | [41] |
| Winery sediment | 312 | 786 | 64 | 43 | 496 | 28 | - | 280 | 214 | [24] |
| Processed Food | By-Product | TP 1 | TT 2 | CT 3 | HT 4 | Polyphenols | References |
|---|---|---|---|---|---|---|---|
| Citrus fruit | Orange peel | 104–223 | - | - | - | Gallic acid, ferulic acid, p-coumaric, catechins, epicatechins, hesperidin, quercetin, kaempferol | [50] |
| Date palm | Date seeds (pits) | 12.7–47.7 | Hydroxytyrosol, tyrosol oleuropein, gallic acid, ferulic acid, coumaric acids, p-hydroxybenzoic acid, flavonoids | [51,52,53,54] | |||
| Grape | Grape pomace | 14.8–70.5 | 39.1–105 | Anthocyanins, condensed tannins, catechin, epicatechin, gallic acids | [17,55] | ||
| Grape stalk | Flavanols, condensed tannins, flavonols and hydroxycinnamates | [56] | |||||
| Grape seeds | 3–90 | Condensed tannins, catechin, gallic, caffeic, and ferulic acids | [29,57,58] | ||||
| Myrtus communis | Whole exhausted Mirtle berries | 47 | nd | 0.0004 | 5.7 | Hydrolysable tannins, phenolic acids, flavanols, flavonols | [16,17,49] |
| Mirtle seeds | 39.3 | nd | 0 | phenolic acids, flavanols, flavonols | [49,59] | ||
| Mirtle pericarp | 13.7 | nd | 0.0004 | nd | phenolic acids, flavanols, flavonols | [49,59] | |
| Olive | Olive cake | 4.1–19.4 | 1.7 | Tyrosol, hydroxitirosol, oleuropein, verbacoside, rutin, luteolin, apigenin, quercetin | [32,60,61,62] | ||
| Olive waste water | 5.17–8.90 5 | Hydroxitirosol, oleuropein, tyrosol, syringing acid, caffeic acid, vanillic acid, verbacoside, catechol, rutin | [60,61,62] | ||||
| Olive stones and seeds | Tyrosol, hydroxitirosol, oleuropein, verbacoside (in seeds), nüzhenide in (seeds) | [63] | |||||
| Pistachio | Pistachio hulls | 78.5–103 | 31.6–63.9 | 8.5–12.0 | Gallotannins, flavonoids, anacardic acids. | [40,64,65] | |
| Pomegranate | Pomegranate seeds | 27.2 | 16.9 | 0.8 | - | Flavonoids, anthocyanins, hydrolizable tannins | [66] |
| Pomegranate peel | 48.3 | - | - | - | Gallic acid, flavonoids, hydrolizable tannins, condensed tannins, punicalagin | [67,68] | |
| Pomegranate pulp | 95.3 | 93.4 | - | - | Tannins | [69] | |
| Tomato | Tomato pomace | 6.1–6.4 | 4.0 | 0 | Naringenin, rutin, quercetin, kaempferol | [17,32,40] |
| BPRP | TP 1 in BPRP | TP 1 in Diet | Main Effect | Species | Reference |
|---|---|---|---|---|---|
| Date palm | - | 10.1, 12.6 g/kg DM | Increase pH, propionate and valerate; reduced acetate | goats | [94] |
| Olive by-product | - | - | Increase nutrient apparent digestibility and metabolizable energy | goat | [127] |
| Pistachio | - | 33.1 g/kg DM | Reduction of ammonia and acetic acid | goat | [107] |
| Pistachio hull | 103 g/kg DM | 26.6 g/kg DM | Reduction of ammonia and VFA | goat | [40] |
| Tomato silage | - | - | Increase nutrient apparent digestibility and metabolizable energy Reduce acetate concentration and (numerically) methane production | goat | [127] |
| Grape pomace | 70.5 g/kg DM | 40.7 g/kg DM | Reduction of ammonia, pH, CP digestibility | sheep | [22] |
| Grape seed | 3.0 g/kg DM | 0.4 g/kg DM | Increase rumen ammonia, rumenic acid, reduced linoleic and α-linolenic acids | sheep | [29] |
| Exhausted myrtle berries | 50 g/kg DM | 2.27 g/kg DM | Reduction of ammonia, VFA, Butyrivibrio group | sheep | [123] |
| Olive oil pomace | - | 4.9, 2.7 g/kg DM | Increase α-linolenic and rumenic acids | sheep | [128] |
| Pistachio by-product | 78.5 g/kg DM | 42.50 g/kg DM | Decrease total VFA, acetic acid | sheep | [90] |
| Pistachio hull | 78.5 g/kg DM | 42.50 g/kg DM | Increase pH, decrease ammonia, total VFA, acetate | sheep | [35] |
| Pistachio | 99.5 g/kg DM | 26.4, 35.2g/kg DM | Reduction of ammonia, VFA and acetate | sheep | [92] |
| Vine leaves | 50 g/kg DM | - | Reduce nutrient digestibility | sheep | [108] |
| By-Products | TP 1 in by-Products | By-Product in the Diet 2 | TP 1 in Diet g/kg DM | Milk | Fat | Protein | Lactose | Urea | Species | References |
|---|---|---|---|---|---|---|---|---|---|---|
| lentil straw | 2.8 TAE% on DM | 300.0 | 13.20 | ↓ ns | ↓ | ↑ ns | ↑ ns | - | sheep | [32] |
| atriplex leaves | 0.63 TAE% on DM | 300.0 | 5.70 | ↓ ns | ↓ ns | ↑ ns | ↑ | - | sheep | [32] |
| date palm | - | 60.0 | 9.60 | ns | ns | ns | ns | - | goats | [94] |
| date palm | - | 120.0 | 1.08 | ns | ns | ns | ns | - | goats | [94] |
| date palm | - | 180.0 | 1.26 | ns | ns | ns | ns | - | goats | [94] |
| exhausted myrtle berries | 5.30 g GAE/100gDM | 22.6 | 1.20 | ns | ns | ns | ns | ↓ ns | sheep | [16] |
| exhausted myrtle berries | 5.30 g GAE/100 g DM | 44.3 | 2.35 | ns | ns | ns | ns | ↓ | sheep | [16] |
| exhausted myrtle berries | 40.9 g/kg DM | 28.0 | 1.14 | ↓ | ns | ns | ↓ | ↑ ns | sheep | [17] |
| grape pomace | 14.8 g/kg DM | 36.5 | 0.54 | ↑ | ↓ | ↓ | ns | ↓ ns | sheep | [17] |
| grape pomace | 42.8 g/kg DM | 51.7 | 2.21 | ns | ns | ns | ↓ | - | sheep | [26] |
| grape pomace | 42.8 g/kg DM | 103.2 | 4.42 | ns | ns | ns | ↓ | - | sheep | [26] |
| grape residue flour | 87.4 mg GAE/g DM | 3.4 | 0.03 | ns | ↑ ns | ns | ns | - | sheep | [28] |
| grape residue flour | 87.4 mg GAE/g DM | 6.7 | 0.10 | ns | ↑ | ns | ns | - | sheep | [28] |
| grape seed | 0.3g/100 g DM | 121.5 | 0.36 | ns | ns | ns | ns | - | sheep | [91] |
| olive leaves | 6.35 TAE% on DM | 300.0 | 22.50 | ↓ | ↓ ns | ↑ ns | ns | - | sheep | [32] |
| olive cake | 0.41 TAE% on DM | 300.0 | 5.30 | ↓ ns | ns | ns | ↑ | - | sheep | [32] |
| olive silage | - | 202.0 | - | - | ↑ | ns | ns | - | goats | [127] |
| pomegranate seed | - | 60.0 | - | ns | ↑ | ns | ↑ ns | - | goats | [130] |
| pomegranate seed | - | 120.0 | - | ns | ↑ | ns | ↑ | - | goats | [130] |
| pomegranate pulp | 95.3 g/kg DM | 648.4 | 61.40 | ns | ns | ns | ns | ns | sheep | [37] |
| RO 3 by-product | - | 50.0 | - | - | ns | ns | ns | - | goats | [131] |
| RO 3 by-product | - | 100.0 | - | - | ns | ns | ↓ | - | goats | [131] |
| tomato pomace | 0.64 TAE% on DM | 300.0 | 6.00 | ↓ ns | ↓ ns | ↓ ns | ↑ | - | sheep | [32] |
| tomato | 2.3 g/kg DM | 36.2 | 0.08 | ns | ↓ | ↓ | ns | ↓ ns | sheep | [17] |
| tomato silage | - | 202.0 | - | - | ↑ | ns | ns | - | goats | [127] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Correddu, F.; Lunesu, M.F.; Buffa, G.; Atzori, A.S.; Nudda, A.; Battacone, G.; Pulina, G. Can Agro-Industrial By-Products Rich in Polyphenols be Advantageously Used in the Feeding and Nutrition of Dairy Small Ruminants? Animals 2020, 10, 131. https://doi.org/10.3390/ani10010131
Correddu F, Lunesu MF, Buffa G, Atzori AS, Nudda A, Battacone G, Pulina G. Can Agro-Industrial By-Products Rich in Polyphenols be Advantageously Used in the Feeding and Nutrition of Dairy Small Ruminants? Animals. 2020; 10(1):131. https://doi.org/10.3390/ani10010131
Chicago/Turabian StyleCorreddu, Fabio, Mondina Francesca Lunesu, Giovanna Buffa, Alberto Stanislao Atzori, Anna Nudda, Gianni Battacone, and Giuseppe Pulina. 2020. "Can Agro-Industrial By-Products Rich in Polyphenols be Advantageously Used in the Feeding and Nutrition of Dairy Small Ruminants?" Animals 10, no. 1: 131. https://doi.org/10.3390/ani10010131
APA StyleCorreddu, F., Lunesu, M. F., Buffa, G., Atzori, A. S., Nudda, A., Battacone, G., & Pulina, G. (2020). Can Agro-Industrial By-Products Rich in Polyphenols be Advantageously Used in the Feeding and Nutrition of Dairy Small Ruminants? Animals, 10(1), 131. https://doi.org/10.3390/ani10010131









