Inoculation with Mycorrhizal Fungi and Irrigation Management Shape the Bacterial and Fungal Communities and Networks in Vineyard Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description and Experimental Design
2.2. Soil Sampling, DNA Extraction, and Sequencing
2.3. Edaphic Factors of the Plots Subjected to Different Irrigation Amounts and AMF Inoculation
2.4. Statistical Analysis
3. Results
3.1. Richness and Diversity of Bacterial and Fungal Communities in Vineyard Plots before and after AMF Inoculation and Irrigation Systems Application
3.2. Bacterial and Fungal Taxa Distribution in the Merlot Vineyard Bulk Soil Is Significantly Affected by AMF Inoculation, Irrigation Treatments, and Sampling Time
3.3. Bacterial and Fungal Clade Proportions in the Merlot Vineyard after Different Treatments of AMF Inoculation and/or Irrigation
3.4. Edaphic Factors Barely Affected in Bacterial and Fungal Distribution
4. Discussion
4.1. Differential Responses of Bacterial and Fungi Alpha and Beta Diversities to AMF Inoculation and Irrigation Treatments
4.2. Bacterial and Fungal Composition after AMF Inoculation and Irrigation Treatment Application during the Season
4.3. Irrigation Treatments and AMF Inoculation Shifted Microbial Communities but Not through Changes in Soil Edaphic Factors
4.4. Time Course and Management Practices Affect Bacterial and Fungal Co-Occurrence Networks
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Compant, S.; Samad, A.; Faist, H.; Sessitsch, A. A review on the plant microbiome: Ecology, functions, and emerging trends in microbial application. J. Adv. Res. 2019, 19, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Karimi, B.; Cahurel, J.-Y.; Gontier, L.; Charlier, L.; Chovelon, M.; Mahé, H.; Ranjard, L. A meta-analysis of the ecotoxicological impact of viticultural practices on soil biodiversity. Environ. Chem. Lett. 2020, 18, 1947–1966. [Google Scholar] [CrossRef]
- Lazcano, C.; Decock, C.; Wilson, S.G. Defining and Managing for Healthy Vineyard Soils, Intersections with the Concept of Terroir. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Zarraonaindia, I.; Owens, S.M.; Weisenhorn, P.; West, K.; Hampton-Marcell, J.; Lax, S.; Bokulich, N.A.; Mills, D.A.; Martin, G.; Taghavi, S.; et al. The Soil Microbiome Influences Grapevine-Associated Microbiota. mBio 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Grangeteau, C.; Roullier-Gall, C.; Rousseaux, S.; Gougeon, R.D.; Schmitt-Kopplin, P.; Alexandre, H.; Guilloux-Benatier, M. Wine microbiology is driven by vineyard and winery anthropogenic factors. Microb. Biotechnol. 2017, 10, 354–370. [Google Scholar] [CrossRef] [PubMed]
- Knight, S.J.; Karon, O.; Goddard, M.R. Small scale fungal community differentiation in a vineyard system. Food Microbiol. 2020, 87, 103358. [Google Scholar] [CrossRef] [PubMed]
- Gopu, V.; Meena, C.K.; Shetty, P.H. Quercetin influences quorum sensing in food borne bacteria: In-vitro and in-silico evi-dence. PLoS ONE 2015, 10, e0134684. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, C.; Roby, J.-P.; De Rességuier, L. Soil-related terroir factors: A review. OENO One 2018, 52, 173–188. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Thorngate, J.H.; Richardson, P.M.; Mills, D.A. PNAS Plus: From the Cover: Microbial biogeography of wine grapes is conditioned by cultivar, vintage, and climate. Proc. Natl. Acad. Sci. USA 2014, 111, E139–E148. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Collins, T.S.; Masarweh, C.; Allen, G.; Heymann, H.; Ebeler, S.E.; Mills, D.A. Associations among Wine Grape Microbiome, Metabolome, and Fermentation Behavior Suggest Microbial Contribution to Regional Wine Characteristics. mBio 2016, 7. [Google Scholar] [CrossRef]
- Burns, K.N.; Kluepfel, D.A.; Strauss, S.L.; Bokulich, N.A.; Cantu, D.; Steenwerth, K.L. Vineyard soil bacterial diversity and composition revealed by 16S rRNA genes: Differentiation by geographic features. Soil Biol. Biochem. 2015, 91, 232–247. [Google Scholar] [CrossRef]
- Burns, K.N.; Bokulich, N.A.; Cantu, D.; Greenhut, R.F.; Kluepfel, D.A.; O’Geen, A.T.; Strauss, S.L.; Steenwerth, K.L. Vineyard soil bacterial diversity and composition revealed by 16S rRNA genes: Differentiation by vineyard management. Soil Biol. Biochem. 2016, 103, 337–348. [Google Scholar] [CrossRef]
- Likar, M.; Stres, B.; Rusjan, D.; Potisek, M.; Regvar, M. Ecological and conventional viticulture gives rise to distinct fungal and bacterial microbial communities in vineyard soils. Appl. Soil Ecol. 2017, 113, 86–95. [Google Scholar] [CrossRef]
- Coller, E.; Cestaro, A.; Zanzotti, R.; Bertoldi, D.; Pindo, M.; Larger, S.; Albanese, D.; Mescalchin, E.; Donati, C. Microbiome of vineyard soils is shaped by geography and management. Microbiome 2019, 7, 1–15. [Google Scholar] [CrossRef]
- Rienth, M.; Scholasch, T. State-of-the-art of tools and methods to assess vine water status. OENO One 2019, 53, 4. [Google Scholar] [CrossRef]
- Hartman, K.; Van Der Heijden, M.G.A.; Wittwer, R.A.; Banerjee, S.; Walser, J.-C.; Schlaeppi, K. Cropping practices manipulate abundance patterns of root and soil microbiome members paving the way to smart farming. Microbiome 2018, 6, 1–14. [Google Scholar] [CrossRef]
- De Vries, F.T.; Griffiths, R.I.; Bailey, M.; Craig, H.; Girlanda, M.; Gweon, H.S.; Hallin, S.; Kaisermann, A.; Keith, A.M.; Kretzschmar, M.; et al. Soil bacterial networks are less stable under drought than fungal networks. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Uroz, S.; Courty, P.E.; Oger, P. Plant Symbionts Are Engineers of the Plant-Associated Microbiome. Trends Plant Sci. 2019, 24, 905–916. [Google Scholar] [CrossRef]
- Finkel, O.M.; Castrillo, G.; Paredes, S.H.; González, I.S.; Dangl, J.L. Understanding and exploiting plant beneficial microbes. Curr. Opin. Plant Biol. 2017, 38, 155–163. [Google Scholar] [CrossRef]
- Del Buono, D. Can biostimulants be used to mitigate the effect of anthropogenic climate change on agriculture? It is time to respond. Sci. Total Environ. 2021, 751, 141763. [Google Scholar] [CrossRef]
- Akyol, T.Y.; Niwa, R.; Hirakawa, H.; Maruyama, H.; Sato, T.; Suzuki, T.; Fukunaga, A.; Sato, T.; Yoshida, S.; Tawaraya, K.; et al. Impact of Introduction of Arbuscular Mycorrhizal Fungi on the Root Microbial Community in Agricultural Fields. Microbes Environ. 2019, 34, 23–32. [Google Scholar] [CrossRef]
- Shang, J.; Liu, B. Application of a microbial consortium improves the growth of Camellia sinensis and influences the indigenous rhizosphere bacterial communities. J. Appl. Microbiol. 2021, 130, 2029–2040. [Google Scholar] [CrossRef]
- Xu, H.; Yang, Y.; Tian, Y.; Xu, R.; Zhong, Y.; Liao, H. Rhizobium Inoculation Drives the Shifting of Rhizosphere Fungal Community in a Host Genotype Dependent Manner. Front. Microbiol. 2020, 10, 3135. [Google Scholar] [CrossRef]
- Belda, I.; Zarraonaindia, I.; Perisin, M.; Palacios, A.; Acedo, A. From Vineyard Soil to Wine Fermentation: Microbiome Approximations to Explain the “terroir” Concept. Front. Microbiol. 2017, 8, 821. [Google Scholar] [CrossRef]
- Belda, I.; Palacios, A.; Fresno, J.; Ortega, H.; Acedo, A. WineSeq®: A new tool for the study of the functional biodiversity of soils, and its use as a biomarker and guide for vitiviniculture practices. BIO Web Conf. 2017, 9, 1012. [Google Scholar] [CrossRef]
- Feld, L.; Nielsen, T.; Hansen, L.H.; Aamand, J.; Albers, C.N. Establishment of Bacterial Herbicide Degraders in a Rapid Sand Filter for Bioremediation of Phenoxypropionate-Polluted Groundwater. Appl. Environ. Microbiol. 2015, 82, 878–887. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Quince, C.; Mahé, F. VSEARCH: A versatile open source tool for metagenomics. PeerJ 2016, 4, e2584. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME Improves Sensitivity and Speed of Chimera Detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef]
- Edgar, R.C. SINTAX: A simple non-Bayesian taxonomy classifier for 16S and ITS sequences. BioRxiv 2016. [Google Scholar] [CrossRef]
- Glöckner, F.O.; Yilmaz, P.; Quast, C.; Gerken, J.; Beccati, A.; Ciuprina, A.; Bruns, G.; Yarza, P.; Peplies, J.; Westram, R.; et al. 25 years of serving the community with ribosomal RNA gene reference databases and tools. J. Biotechnol. 2017, 261, 169–176. [Google Scholar] [CrossRef]
- Kaplan, I.; Bokulich, N.A.; Caporaso, J.G.; Enders, L.S.; Ghanem, W.; Ingerslew, K.S. Phylogenetic farming: Can evolutionary history predict crop rotation via the soil microbiome? Evol. Appl. 2020, 13, 1984–1999. [Google Scholar] [CrossRef]
- Chen, H. Package ‘VennDiagram’. Generate High-Resolution Venn and Euler Plots, Version 1.6.20. 2018. Available online: https://cran.r-project.org/web/packages/VennDiagram/VennDiagram.pdf (accessed on 4 April 2020).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S.; Christensen, R.H.B.; Singmann, H. Package ‘lme4’. Linear Mixed-Effects Models Using ‘Eigen’ and S4. 2020. Available online: https://cran.r-project.org/web/packages/lme4/lme4.pdf (accessed on 8 September 2020).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Lenth, R. Package ‘lsmeans’. Least-Squares Means, Version 2.30-0. 2018. Available online: https://cran.r-project.org/web/packages/lsmeans/lsmeans.pdf (accessed on 9 July 2020).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D. Package ‘Vegan’. Community Ecology Package, Version 2.5-6; 2019. Available online: https://cran.r-project.org/web/packages/vegan/vegan.pdf (accessed on 10 July 2020).
- Foster, Z.; Grunwald, N.; Gilmore, R. Package ‘Metacoder’. Tools for Parsing; Manipulating; and Graphing Taxonomic Abundance Data, Version 0.3.4. 2020. Available online: https://cran.r-project.org/web/packages/metacoder/metacoder.pdf (accessed on 20 January 2021).
- Roberts, D.W. Package ‘labdsv’. Ordination and Multivariate Analysis for Ecology, Version 2.0-1. 2019. Available online: https://cran.r-project.org/web/packages/labdsv/labdsv.pdf (accessed on 8 September 2020).
- Griffith, D.M.; Veech, J.A.; Marsh, C. Cooccur: Probabilistic Species Co-Occurrence Analysis in R. J. Stat. Softw. 2016, 69, 1–17. [Google Scholar] [CrossRef]
- Veech, J.A. A probabilistic model for analysing species co-occurrence. Glob. Ecol. Biogeogr. 2012, 22, 252–260. [Google Scholar] [CrossRef]
- Almende, B.V.; Benoit, T.; Titouan, R. Package ‘visNetwork’. Network Visualization Using ‘vis.js’ Library, Version 2.0.9. 2019. Available online: https://cran.r-project.org/web/packages/visNetwork/visNetwork.pdf (accessed on 21 January 2021).
- Berlanas, C.; Berbegal, M.; Elena, G.; Laidani, M.; Cibriain, J.F.; Sagües, A.; Gramaje, D. The Fungal and Bacterial Rhizosphere Microbiome Associated With Grapevine Rootstock Genotypes in Mature and Young Vineyards. Front. Microbiol. 2019, 10, 1142. [Google Scholar] [CrossRef]
- Aguilar, M.O.; Gobbi, A.; Browne, P.D.; Ellegaard-Jensen, L.; Hansen, L.H.; Semorile, L.; Pistorio, M. Influence of vintage, geographic location and cultivar on the structure of microbial communities associated with the grapevine rhizosphere in vineyards of San Juan Province, Argentina. PLoS ONE 2020, 15, e0243848. [Google Scholar] [CrossRef]
- Ortiz-Álvarez, R.; Ortega-Arranz, H.; Vicente, J. Ontiveros VJ; Ravarani C; Acedo A; Belda I. Emergent properties in microbiome networks reveal the anthropogenic disturbance of farming practices in vineyard soil fungal communities. BioRxiv 2020. [Google Scholar] [CrossRef]
- Novello, G.; Gamalero, E.; Bona, E.; Boatti, L.; Mignone, F.; Massa, N.; Cesaro, P.; Lingua, G.; Berta, G. The Rhizosphere Bacterial Microbiota of Vitis vinifera cv. Pinot Noir in an Integrated Pest Management Vineyard. Front. Microbiol. 2017, 8, 1528. [Google Scholar] [CrossRef]
- Marasco, R.; Rolli, E.; Fusi, M.; Michoud, G.; Daffonchio, D. Grapevine rootstocks shape underground bacterial microbiome and networking but not potential functionality. Microbiome 2018, 6, 1–17. [Google Scholar] [CrossRef]
- Canfora, L.; Vendramin, E.; Felici, B.; Tarricone, L.; Florio, A.; Benedetti, A. Vineyard microbiome variations during different fertilisation practices revealed by 16s rRNA gene sequencing. Appl. Soil Ecol. 2018, 125, 71–80. [Google Scholar] [CrossRef]
- Gupta, V.V.S.R.; Bramley, R.; Greenfield, P.; Yu, J.; Herderich, M.J. Vineyard Soil Microbiome Composition Related to Rotundone Concentration in Australian Cool Climate ‘Peppery’ Shiraz Grapes. Front. Microbiol. 2019, 10, 1607. [Google Scholar] [CrossRef]
- Willing, C.E.; Pierroz, G.; Coleman-Derr, D.; Dawson, T.E. The generalizability of water-deficit on bacterial community composition; Site-specific water-availability predicts the bacterial community associated with coast redwood roots. Mol. Ecol. 2020, 29. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Wang, X.; Yan, J.; Luo, L. Characterizing the Intra-Vineyard Variation of Soil Bacterial and Fungal Communities. Front. Microbiol. 2019, 10, 1239. [Google Scholar] [CrossRef]
- Alonso, A.; De Celis, M.; Ruiz, J.; Vicente, J.; Navascués, E.; Acedo, A.; Ortiz-Álvarez, R.; Belda, I.; Santos, A.; Gómez-Flechoso, M.Á.; et al. Looking at the Origin: Some Insights into the General and Fermentative Microbiota of Vineyard Soils. Fermentation 2019, 5, 78. [Google Scholar] [CrossRef]
- Zhang, R.; Gu, J.; Wang, X. Responses of soil bacteria and fungi after 36 years fertilizer, straw cover and irrigation management practices in northwest China. Soil Use Manag. 2020. [Google Scholar] [CrossRef]
- Garcia, J.A.L.; Barbas, C.; Probanza, A.; Barrientos, M.L.; Manero, F.J.G. Low molecular weight organic acids and fatty acids in root exudates of two Lupinus cultivars at flowering and fruiting stages. Phytochem. Anal. 2001, 12, 305–311. [Google Scholar] [CrossRef]
- Goss-Souza, D.; Mendes, L.W.; Borges, C.D.; Baretta, D.; Tsai, S.M.; Rodrigues, J.L.M. Soil microbial community dynamics and assembly under long-term land use change. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef]
- Holland, T.C.; Bowen, P.; Bogdanoff, C.; Hart, M.M. How distinct are arbuscular mycorrhizal fungal communities associating with grapevines? Biol. Fertil. Soils 2014, 50, 667–674. [Google Scholar] [CrossRef]
- Torres, N.; Yu, R.; Martínez-Lüscher, J.; Kostaki, E.; Kurtural, S.K. Application of Fractions of Crop Evapotranspiration Affects Carbon Partitioning of Grapevine Differentially in a Hot Climate. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Karlowsky, S.; Augusti, A.; Ingrisch, J.; Akanda, M.K.U.; Bahn, M.; Gleixner, G. Drought-Induced Accumulation of Root Exudates Supports Post-drought Recovery of Microbes in Mountain Grassland. Front. Plant Sci. 2018, 9, 1593. [Google Scholar] [CrossRef]
- Chou, M.-Y.; Heuvel, J.V.; Bell, T.; Panke-Buisse, K.; Kao-Kniffin, J. Vineyard under-vine floor management alters soil microbial composition, while the fruit microbiome shows no corresponding shifts. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Simoni, S.; Gagnarli, E.; Goggioli, D.; Guidi, S.; Tarchi, T.; D’Avino, L. Impact of Agricultural Management on Communities of Oribatida; Gamasina and Collembola in Italian and French Vineyards. EQA 2018, 31, 27–32. [Google Scholar] [CrossRef]
- Vink, S.N.; Chrysargyris, A.; Tzortzakis, N.; Salles, J.F. Bacterial community dynamics varies with soil management and irrigation practices in grapevines (Vitis vinifera L.). Appl. Soil Ecol. 2021, 158, 103807. [Google Scholar] [CrossRef]
- Redford, A.J.; Fierer, N. Bacterial Succession on the Leaf Surface: A Novel System for Studying Successional Dynamics. Microb. Ecol. 2009, 58, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Bona, E.; Massa, N.; Novello, G.; Boatti, L.; Cesaro, P.; Todeschini, V. Metaproteomic characterization of the Vitis vinifera rhizosphere. FEMS Microbiol. Ecol. 2019, 95. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, S.; Waite, I.S.; Blackburn, A.; Husband, R.; Rushton, S.P.; Manning, D.C.; O’Donnell, A.G. Actinobacterial community dynamics in long term managed grasslands. Antonie Leeuwenhoek 2009, 95, 319–334. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Espíndola, I.P.; Ferrara-Guerrero, M.J.; Luna-Guido, M.L.; Ramírez-Villanueva, D.A.; De León-Lorenzana, A.S.; Gómez-Acata, S.; González-Terreros, E.; Ramírez-Barajas, B.; Navarro-Noya, Y.E.; Sánchez-Rodríguez, L.M.; et al. The Bacterial Community Structure and Microbial Activity in a Traditional Organic Milpa Farming System Under Different Soil Moisture Conditions. Front. Microbiol. 2018, 9, 2737. [Google Scholar] [CrossRef]
- Santos-Medellín, C.; Edwards, J.; Liechty, Z.; Nguyen, B.; Sundaresan, V. Drought Stress Results in a Compartment-Specific Restructuring of the Rice Root-Associated Microbiomes. mBio 2017, 8. [Google Scholar] [CrossRef]
- De Vries, F.T.; Williams, A.; Stringer, F.; Willcocks, R.; McEwing, R.; Langridge, H.; Straathof, A.L. Changes in root-exudate-induced respiration reveal a novel mechanism through which drought affects ecosystem carbon cycling. New Phytol. 2019, 224, 132–145. [Google Scholar] [CrossRef]
- Andreolli, M.; Lampis, S.; Zapparoli, G.; Angelini, E.; Vallini, G. Diversity of bacterial endophytes in 3 and 15 year-old grapevines of Vitis vinifera cv. Corvina and their potential for plant growth promotion and phytopathogen control. Microbiol. Res. 2016, 183, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Pérez, J.M.; González-García, S.; Cobos, R.; Olego, M.Á.; Ibañez, A.; Díez-Galán, A.; Garzón-Jimeno, E.; Coque, J.J.R. Use of Endophytic and Rhizosphere Actinobacteria from Grapevine Plants to Reduce Nursery Fungal Graft Infections That Lead to Young Grapevine Decline. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef] [PubMed]
- Gałązka, A.; Niedźwiecki, J.; Grządziel, J.; Gawryjołek, K. Evaluation of Changes in Glomalin-Related Soil Proteins (GRSP) Content, Microbial Diversity and Physical Properties Depending on the Type of Soil as the Important Biotic Determinants of Soil Quality. Agronomy 2020, 10, 1279. [Google Scholar] [CrossRef]
- Frąc, M.; Hannula, S.E.; Bełka, M.; Jędryczka, M. Fungal Biodiversity and Their Role in Soil Health. Front. Microbiol. 2018, 9, 707. [Google Scholar] [CrossRef]
- Green, J.L.; Holmes, A.J.; Westoby, M.; Oliver, I.; Briscoe, D.; Dangerfield, M.; Gillings, M.; Beattie, A.J. Spatial scaling of microbial eukaryote diversity. Nat. Cell Biol. 2004, 432, 747–750. [Google Scholar] [CrossRef]
- Xiong, W.; Li, R.; Ren, Y.; Liu, C.; Zhao, Q.; Wu, H.; Jousset, A.; Shen, Q. Distinct roles for soil fungal and bacterial communities associated with the suppression of vanilla Fusarium wilt disease. Soil Biol. Biochem. 2017, 107, 198–207. [Google Scholar] [CrossRef]
- Krishnan, R.; Lang, E.; Midha, S.; Patil, P.; Rameshkumar, N. Isolation and characterization of a novel 1-aminocyclopropane-1-carboxylate (ACC) deaminase producing plant growth promoting marine Gammaproteobacteria from crops grown in brackish environments. Proposal for Pokkaliibacter plantistimulans gen. nov., sp. nov., Balneatrichaceae fam. nov. in the order Oceanospirillales and an emended description of the genus Balneatrix. Syst. Appl. Microbiol. 2018, 41, 570–580. [Google Scholar] [CrossRef]
- Wang, M.; Chen, S.; Chen, L.; Wang, D. Responses of soil microbial communities and their network interactions to saline-alkaline stress in Cd-contaminated soils. Environ. Pollut. 2019, 252, 1609–1621. [Google Scholar] [CrossRef]
- Manici, L.M.; Saccà, M.; Caputo, F.; Zanzotto, A.; Gardiman, M.; Fila, G. Long- term grapevine cultivation and agro-environment affect rhizosphere microbiome rather than plant age. Appl. Soil Ecol. 2017, 119, 214–225. [Google Scholar] [CrossRef]
- Phạm, H.T.T.; Suwannapan, W.; Koomsiri, W.; Inahashi, Y.; Také, A.; Matsumoto, A.; Thamchaipenet, A. Fodinicola acaciae sp. nov., an Endophytic Actinomycete Isolated from the Roots of Acacia mangium Willd. and Its Genome Analysis. Microorganisms 2020, 8, 467. [Google Scholar] [CrossRef]
- Trujillo, M.E.; Riesco, R.; Benito, P.; Carro, L. Endophytic Actinobacteria and the Interaction of Micromonospora and Nitrogen Fixing Plants. Front. Microbiol. 2015, 6, 1341. [Google Scholar] [CrossRef]
- Poomthongdee, N.; Duangmal, K.; Pathom-Aree, W. Acidophilic actinomycetes from rhizosphere soil: Diversity and properties beneficial to plants. J. Antibiot. 2015, 68, 106–114. [Google Scholar] [CrossRef]
- Shen, G.; Zhang, S.; Liu, X.; Jiang, Q.; Ding, W. Soil acidification amendments change the rhizosphere bacterial community of tobacco in a bacterial wilt affected field. Appl. Microbiol. Biotechnol. 2018, 102, 9781–9791. [Google Scholar] [CrossRef]
- Fernández-González, A.J.; Cardoni, M.; Cabanás, C.G.-L.; Valverde-Corredor, A.; Villadas, P.J.; Fernández-López, M.; Mercado-Blanco, J. Linking belowground microbial network changes to different tolerance level towards Verticillium wilt of olive. Microbiome 2020, 8, 1–19. [Google Scholar] [CrossRef]
- Essakhi, S.; Mugnai, L.; Crous, P.; Groenewald, J.; Surico, G. Molecular and phenotypic characterisation of novel Phaeoacremonium species isolated from esca diseased grapevines. Persoonia 2008, 21, 119–134. [Google Scholar] [CrossRef]
- Viswanathan, R.; Selvakumar, R.; Manivannan, K.; Nithyanantham, R.; Kaverinathan, K. Behaviour of Soil Borne Inoculum of Colletotrichum falcatum in Causing Red Rot in Sugarcane Varieties with Varying Disease Resistance. Sugar Tech 2020, 22, 485–497. [Google Scholar] [CrossRef]
- Brum, M.; Araújo, W.L.; Maki, C.; Azevedo, J. Endophytic fungi from Vitis labrusca L. ‘Niagara Rosada’ and its potential for the biological control of Fusarium oxysporum. Genet. Mol. Res. 2012, 11, 4187–4197. [Google Scholar] [CrossRef]
- Rabeendran, N.; Jones, E.; Moot, D.; Stewart, A. Biocontrol of Sclerotinia lettuce drop by Coniothyrium minitans and Trichoderma hamatum. Biol. Control 2006, 39, 352–362. [Google Scholar] [CrossRef]
- Martínez-Diz, M.D.P.; Andrés-Sodupe, M.; Bujanda, R.; Díaz-Losada, E.; Eichmeier, A.; Gramaje, D. Soil-plant compartments affect fungal microbiome diversity and composition in grapevine. Fungal Ecol. 2019, 41, 234–244. [Google Scholar] [CrossRef]
- Banerjee, S.; Walder, F.; Büchi, L.; Meyer, M.; Held, A.Y.; Gattinger, A.; Keller, T.; Charles, R.; Van Der Heijden, M.G.A. Agricultural intensification reduces microbial network complexity and the abundance of keystone taxa in roots. ISME J. 2019, 13, 1722–1736. [Google Scholar] [CrossRef]
- Dong, L.; Xu, J.; Feng, G.; Li, X.; Chen, S. Soil bacterial and fungal community dynamics in relation to Panax notoginseng death rate in a continuous cropping system. Sci. Rep. 2016, 6, 31802. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Deng, Y.; Luo, F.; He, Z.; Tu, Q.; Zhi, X. Functional Molecular Ecological Networks. mBio 2010, 1. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Meng, D.; Qin, C.; Liu, X.; Liang, Y.; Xiao, Y.; Liu, Z.; Gu, Y.; Li, J.; Yin, H. Integrated network analysis reveals the importance of microbial interactions for maize growth. Appl. Microbiol. Biotechnol. 2018, 102, 3805–3818. [Google Scholar] [CrossRef] [PubMed]
- Layeghifard, M.; Hwang, D.M.; Guttman, D.S. Disentangling Interactions in the Microbiome: A Network Perspective. Trends Microbiol. 2017, 25, 217–228. [Google Scholar] [CrossRef]
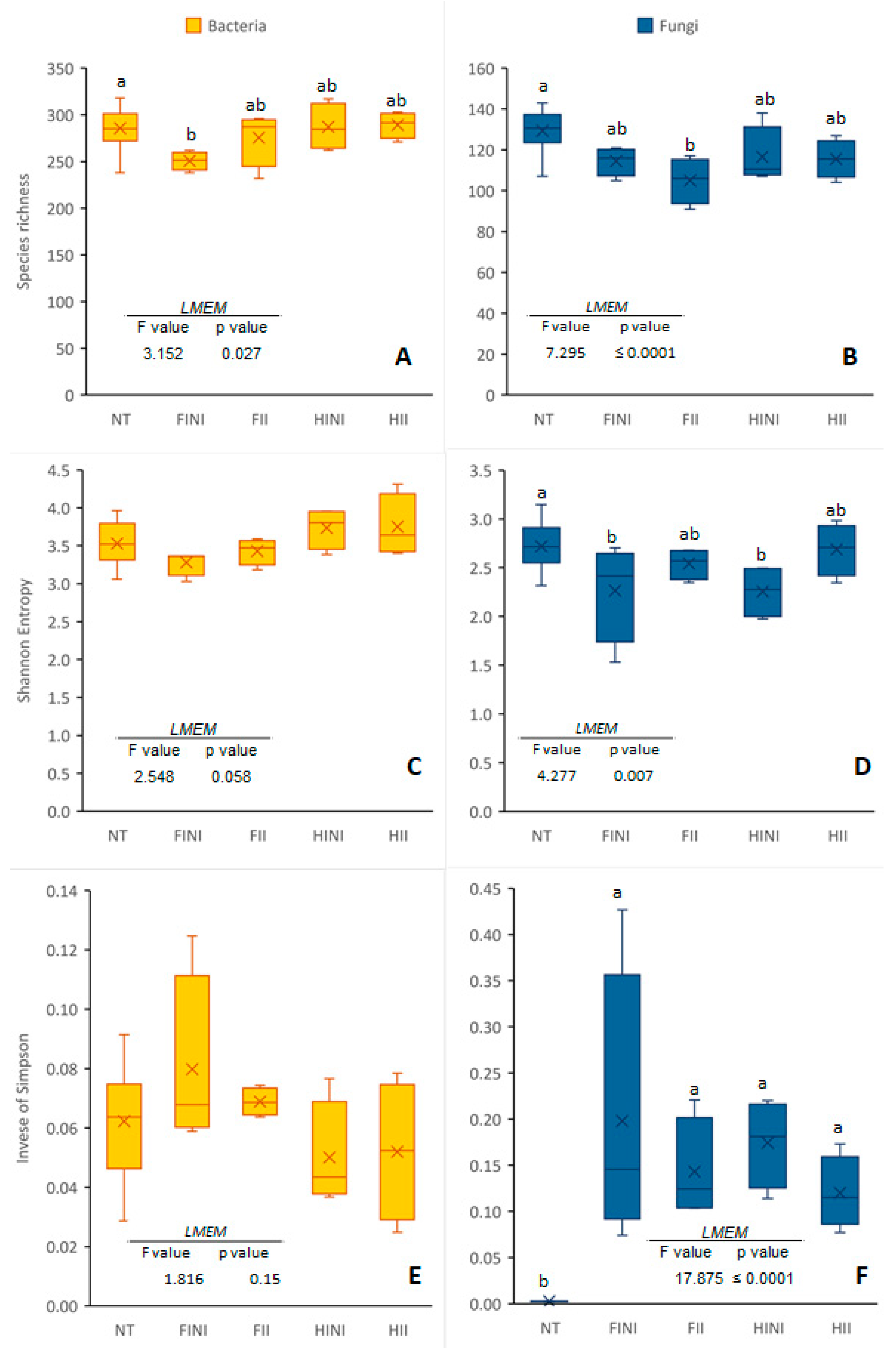
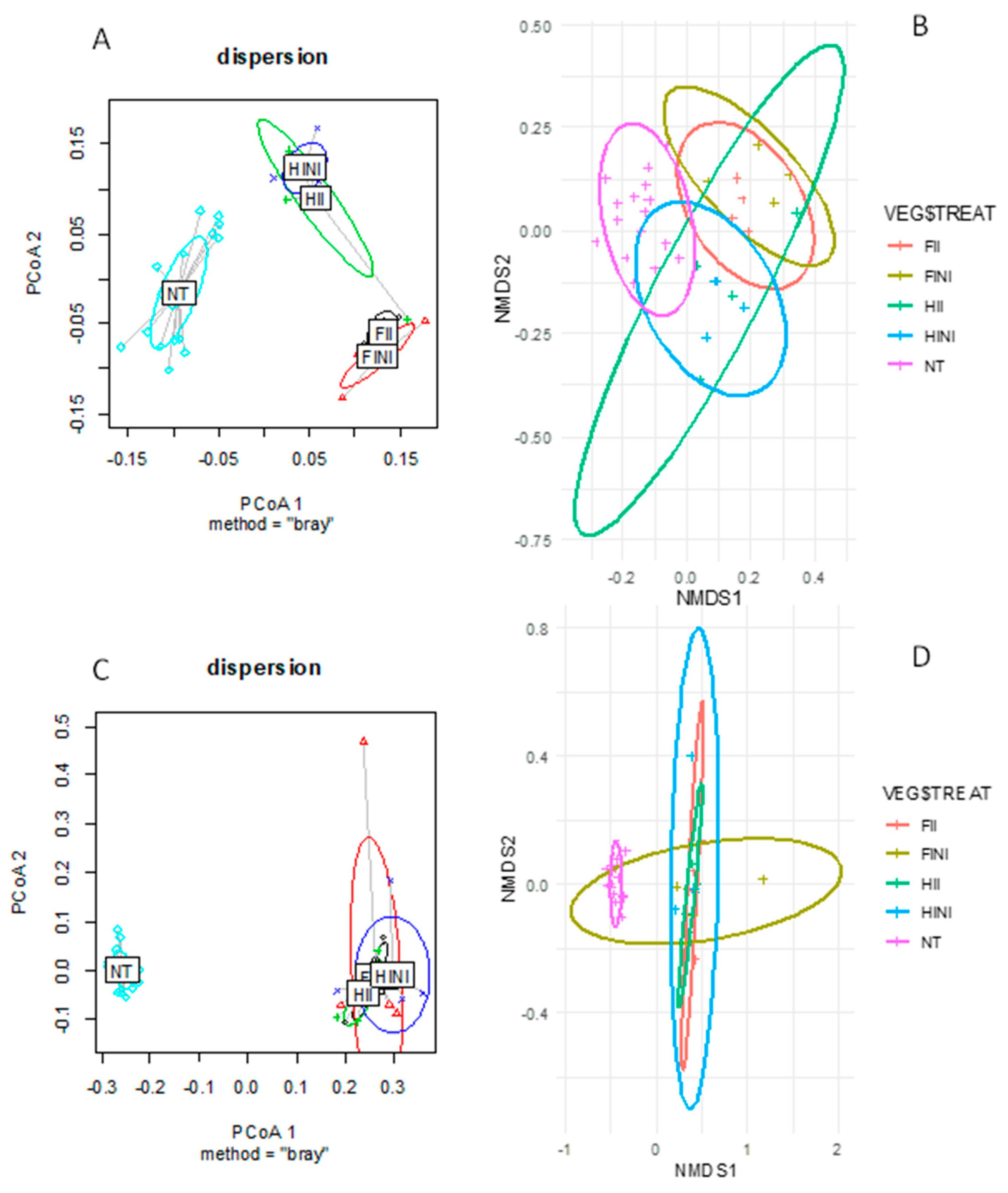
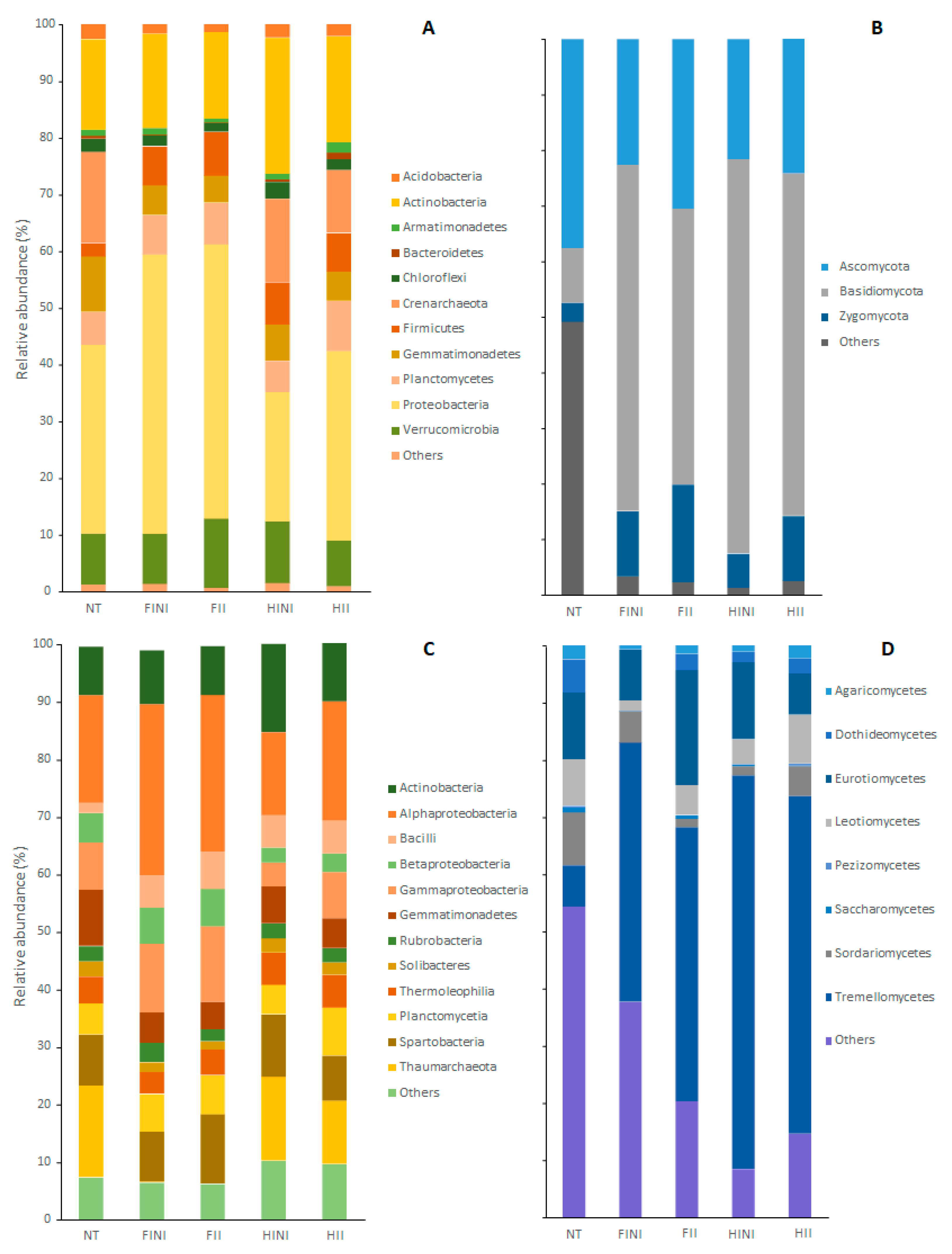
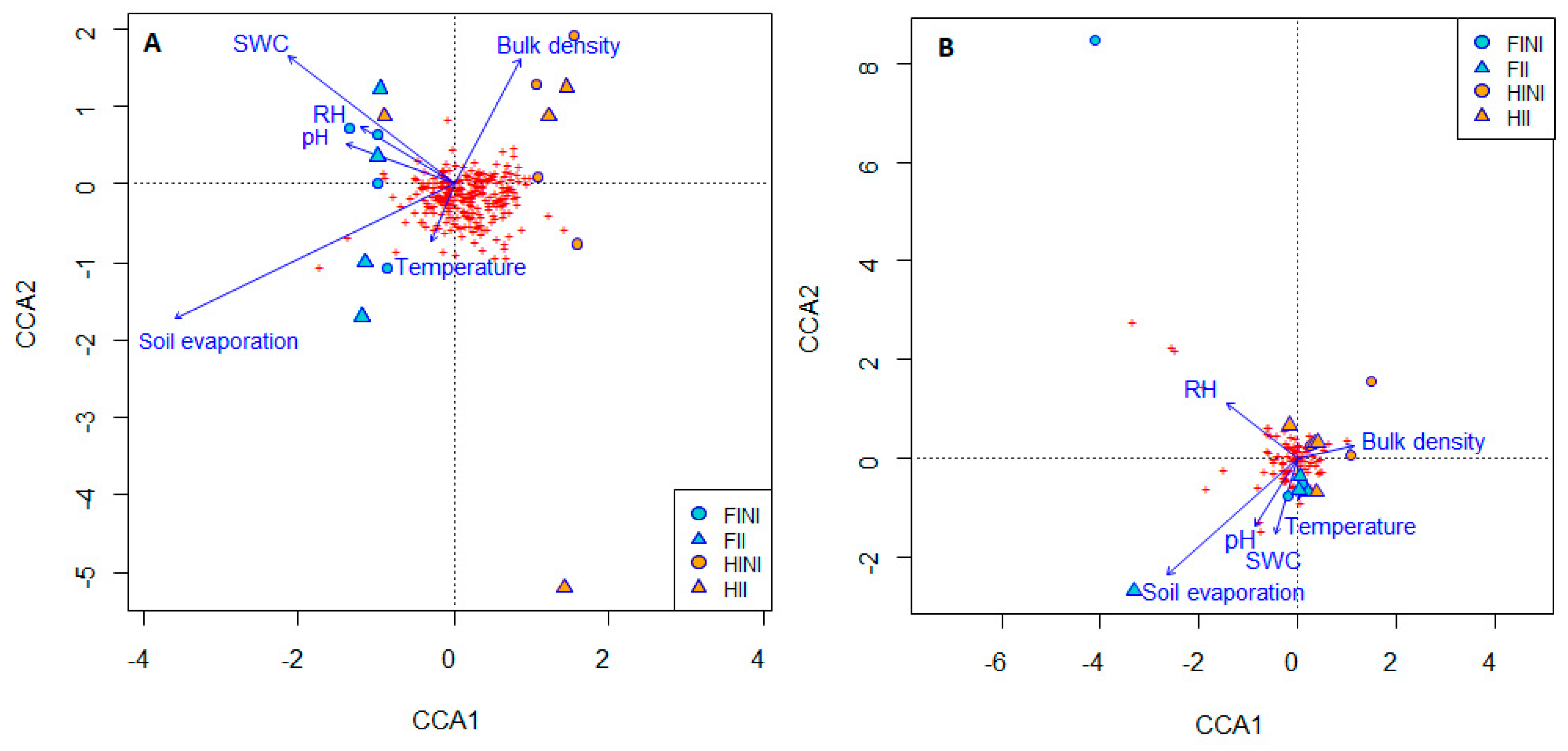
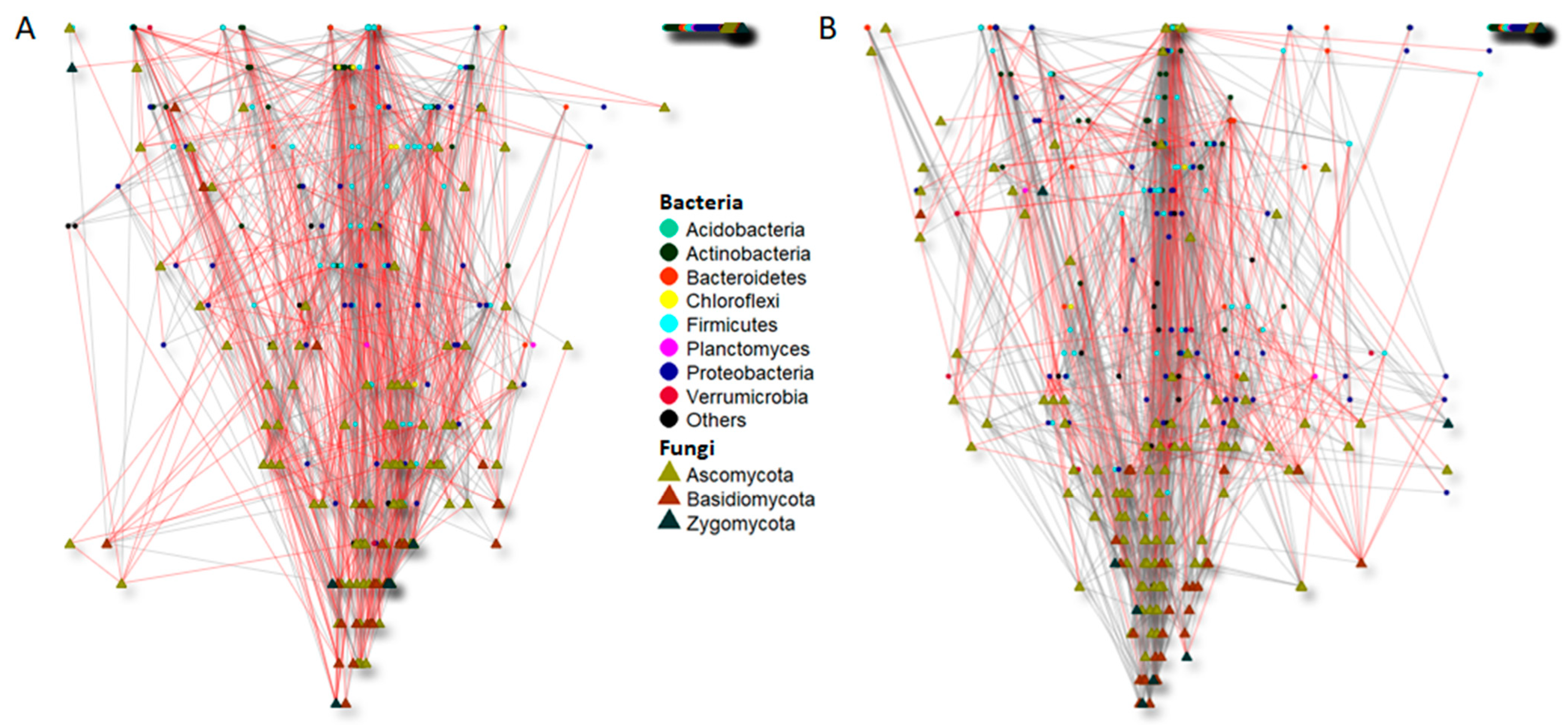
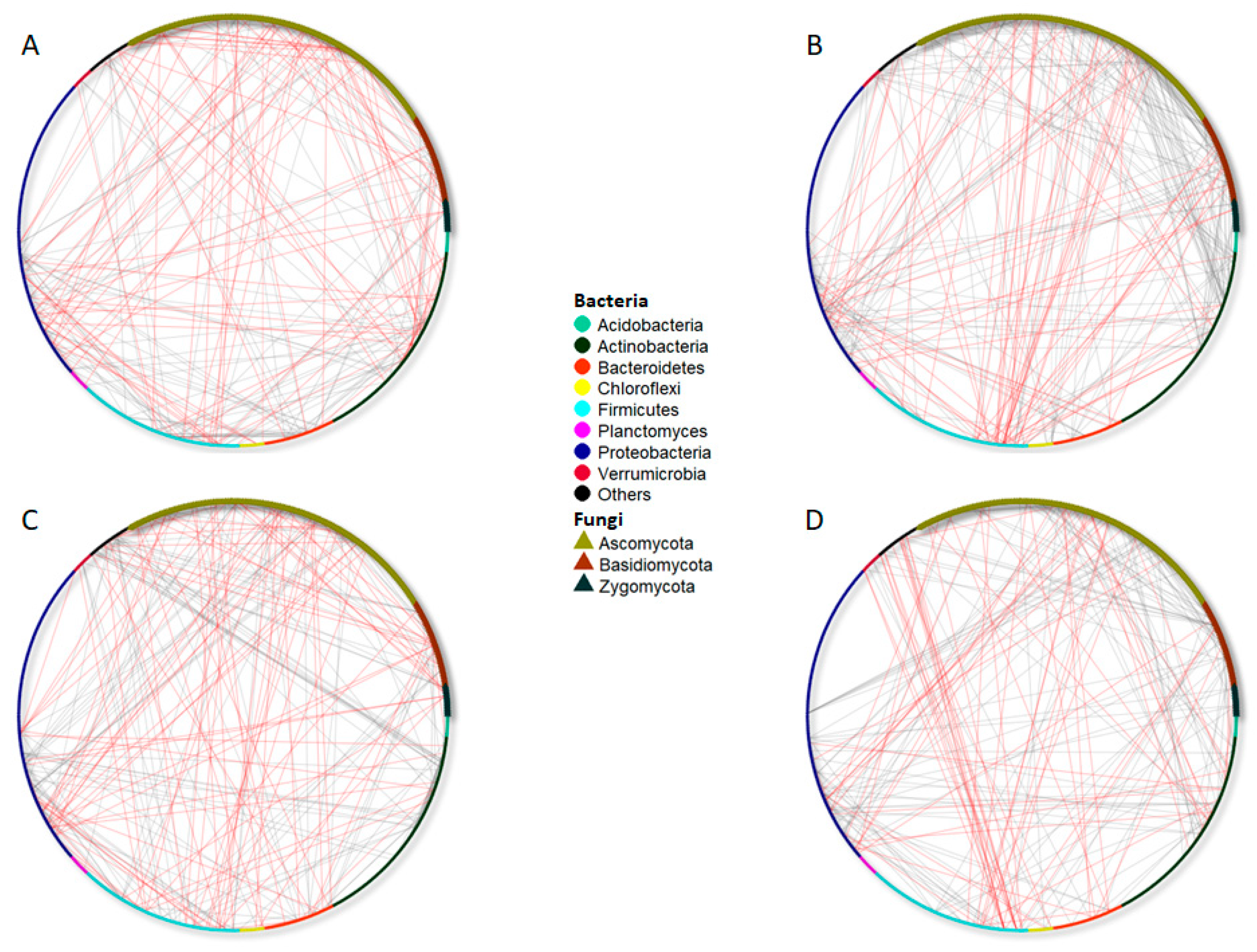
| Bray–Curtis | |||||
|---|---|---|---|---|---|
| Bacteria | Fungi | ||||
| Treatment | Comparison | F | p-Value | F | p-Value |
| Irrigation (I) | FI vs. HI | 4.727 | 0.001 | 6.480 | 0.001 |
| AMF inoculation (M) | NI vs. I | 3.528 | 0.002 | 5.446 | 0.005 |
| Time course (T) | T0 vs. T1 | 9.688 | 0.001 | 26.762 | 0.001 |
| I × M | FINI vs. FII vs. HINI vs. HII | 3.424 | 0.001 | 4.598 | 0.001 |
| I × T | FI_T0 vs. FI_T1 vs. HI_T0 vs. HI_T1 | 8.788 | 0.001 | 14.134 | 0.001 |
| M × T | I_T0 vs. I_T1 vs. NI_T0 vs. NI_T1 | 5.312 | 0.001 | 14.092 | 0.001 |
| I × M × T | FINI_T0 vs. FINI_T1 vs. HINI_T0 vs. HINI_T1 vs. FII_T0 vs. FII_T1 vs. HII_T0 vs. HII_T1 | 4.879 | 0.001 | 7.615 | 0.001 |
| Soil pH | Relative Humidity (RH) | Soil Evaporation (mmol·m−2 s−1) | Soil Temperature (°C) | SWC (g·g−1) | Bulk Density (g·cm−3) | |
|---|---|---|---|---|---|---|
| FI | 5.90 ± 0.10 | 17.4 ± 1.2 | 0.41 ± 0.03 a | 39.1 ± 1.0 | 0.06 ± 0.01 a | 1.11 ± 0.02 |
| HI | 5.74 ± 0.09 | 16.1 ± 0.5 | 0.24 ± 0.02 b | 39.2 ± 0.5 | 0.05 ± 0.01 b | 1.12 ± 0.01 |
| NI | 5.74 ± 0.05 | 16.2 ± 0.9 | 0.29 ± 0.03 b | 39.7 ± 0.5 | 0.05 ± 0.01 | 1.11 ± 0.02 |
| I | 5.89 ± 0.12 | 17.3 ± 1.0 | 0.36 ± 0.04 a | 38.6 ± 0.9 | 0.06 ± 0.01 | 1.11 ± 0.02 |
| LMEM | ||||||
| Irrigation (I) | 0.194 | 0.232 | 0.0001 | 0.862 | 0.029 | 0.591 |
| AMF inoculation (M) | 0.155 | 0.267 | 0.017 | 0.199 | 0.094 | 0.935 |
| I × M | 0.267 | 0.780 | 0.413 | 0.645 | 0.331 | 0.264 |
| Not Treated (NT) | After Treatment | ||
|---|---|---|---|
| Positive connections | |||
| Total | 615 | 795 | |
| Bac–Bac | 281 | 350 | |
| Fun–Fun | 126 | 205 | |
| Bac–Fun | 208 | 240 | |
| Negative connections | |||
| Total | 358 | 254 | |
| Bac–Bac | 93 | 85 | |
| Fun–Fun | 85 | 18 | |
| Bac–Fun | 180 | 151 | |
| Total connections | |||
| Total | 973 | 1049 | |
| Bac–Bac | 374 | 435 | |
| Fun–Fun | 211 | 223 | |
| Bac–Fun | 388 | 391 | |
| Total analyzed pairs | 141,796 | 135,356 | |
| Percentage of non-random | 0.7 | 0.8 | |
| FI | HI | NI | I | ||
|---|---|---|---|---|---|
| Positive connections | |||||
| Total | 130 | 231 | 176 | 196 | |
| Bac–Bac | 72 | 52 | 97 | 66 | |
| Fun–Fun | 24 | 81 | 26 | 62 | |
| Bac–Fun | 34 | 98 | 53 | 68 | |
| Negative connections | |||||
| Total | 99 | 69 | 95 | 67 | |
| Bac–Bac | 30 | 24 | 25 | 28 | |
| Fun–Fun | 21 | 0 | 17 | 0 | |
| Bac–Fun | 48 | 45 | 53 | 39 | |
| Total connections | |||||
| Total | 229 | 300 | 271 | 263 | |
| Bac–Bac | 102 | 76 | 122 | 94 | |
| Fun–Fun | 45 | 81 | 43 | 62 | |
| Bac–Fun | 82 | 143 | 106 | 107 | |
| Total analyzed pairs | 120,299 | 128,975 | 128,097 | 126,817 | |
| Percentage of non-random | 0.2 | 0.8 | 0.2 | 0.2 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, N.; Yu, R.; Kurtural, S.K. Inoculation with Mycorrhizal Fungi and Irrigation Management Shape the Bacterial and Fungal Communities and Networks in Vineyard Soils. Microorganisms 2021, 9, 1273. https://doi.org/10.3390/microorganisms9061273
Torres N, Yu R, Kurtural SK. Inoculation with Mycorrhizal Fungi and Irrigation Management Shape the Bacterial and Fungal Communities and Networks in Vineyard Soils. Microorganisms. 2021; 9(6):1273. https://doi.org/10.3390/microorganisms9061273
Chicago/Turabian StyleTorres, Nazareth, Runze Yu, and S. Kaan Kurtural. 2021. "Inoculation with Mycorrhizal Fungi and Irrigation Management Shape the Bacterial and Fungal Communities and Networks in Vineyard Soils" Microorganisms 9, no. 6: 1273. https://doi.org/10.3390/microorganisms9061273
APA StyleTorres, N., Yu, R., & Kurtural, S. K. (2021). Inoculation with Mycorrhizal Fungi and Irrigation Management Shape the Bacterial and Fungal Communities and Networks in Vineyard Soils. Microorganisms, 9(6), 1273. https://doi.org/10.3390/microorganisms9061273






