Chemical Interactions at the Interface of Plant Root Hair Cells and Intracellular Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Experimental Conditions
2.3. Histochemical Protocols
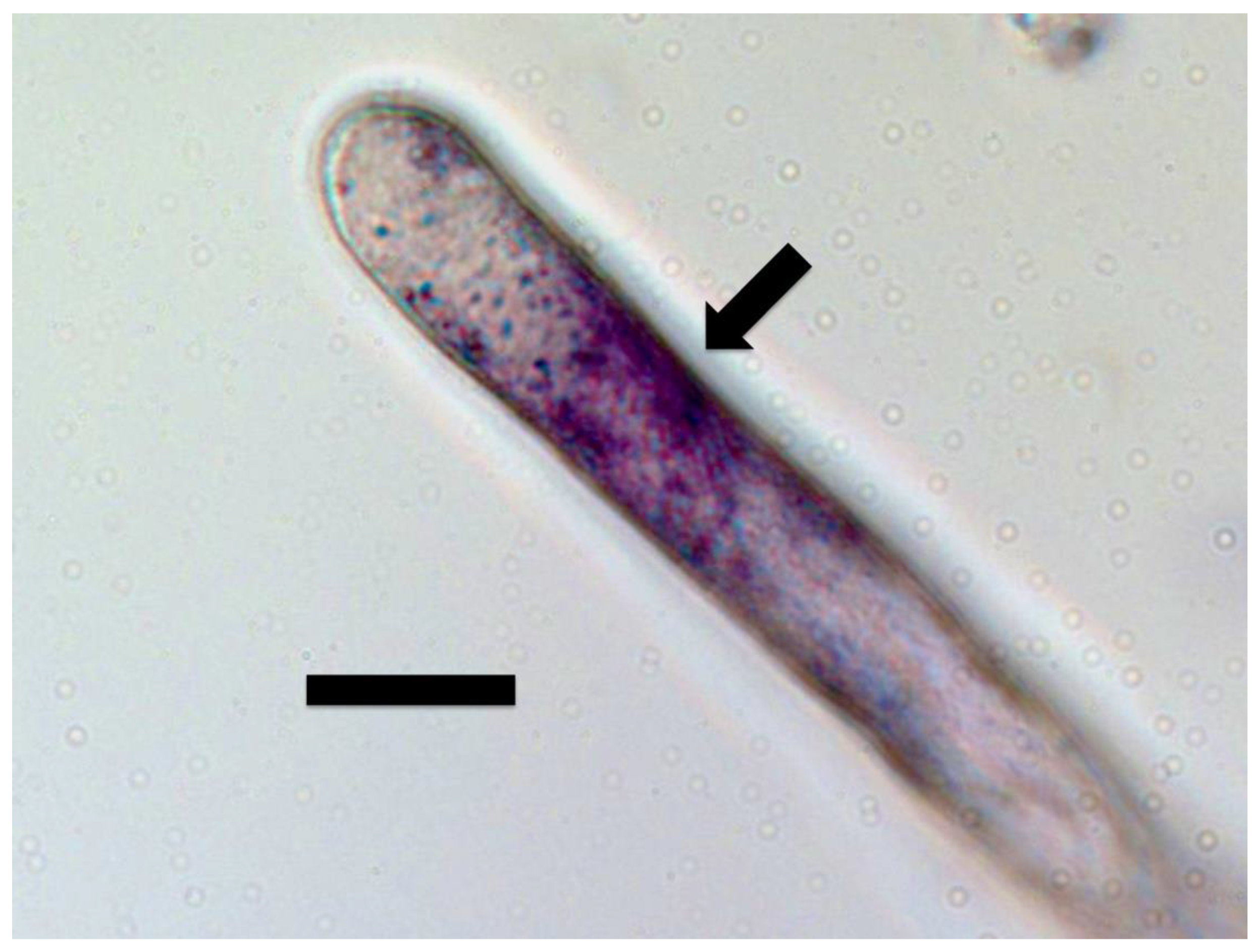
2.3.1. Ethylene Staining
2.3.2. Superoxide Staining
2.3.3. Hydrogen Peroxide Staining
2.3.4. Nitric Oxide Staining
2.3.5. Nitrate Staining
2.4. Microbe and Ethylene Experiments
2.4.1. Microbe Removal and Replacement
2.4.2. Ethylene Gas Experiment
2.5. Experiments with Inhibitors
2.5.1. Inhibition of Superoxide
2.5.2. Inhibition of Ethylene Production by Microbes
2.5.3. Inhibition of Ethylene Synthesis in Plants
2.5.4. Inhibition of Nitric Oxide
2.6. Cynodon dactylon Root Hair Ejection Experiment
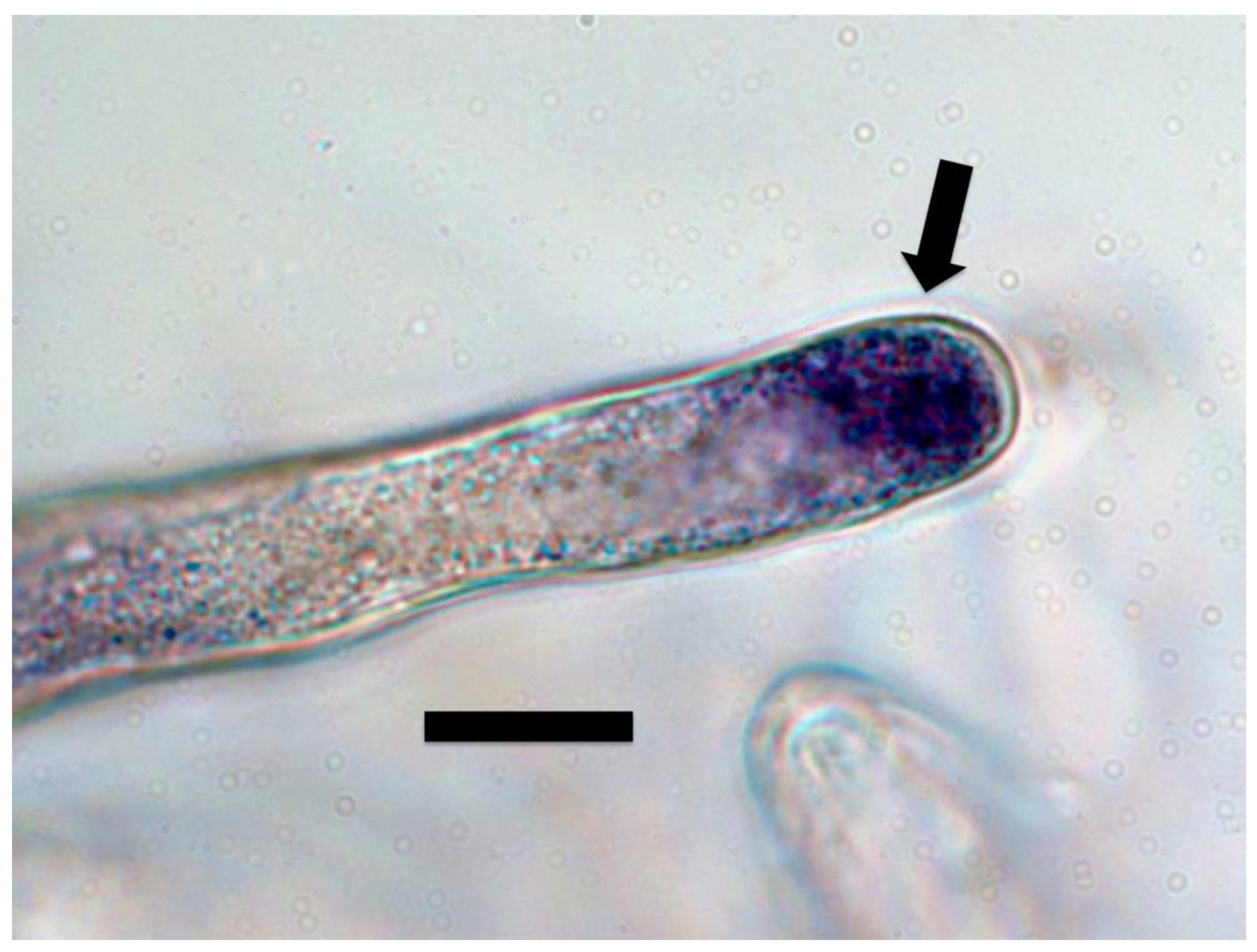
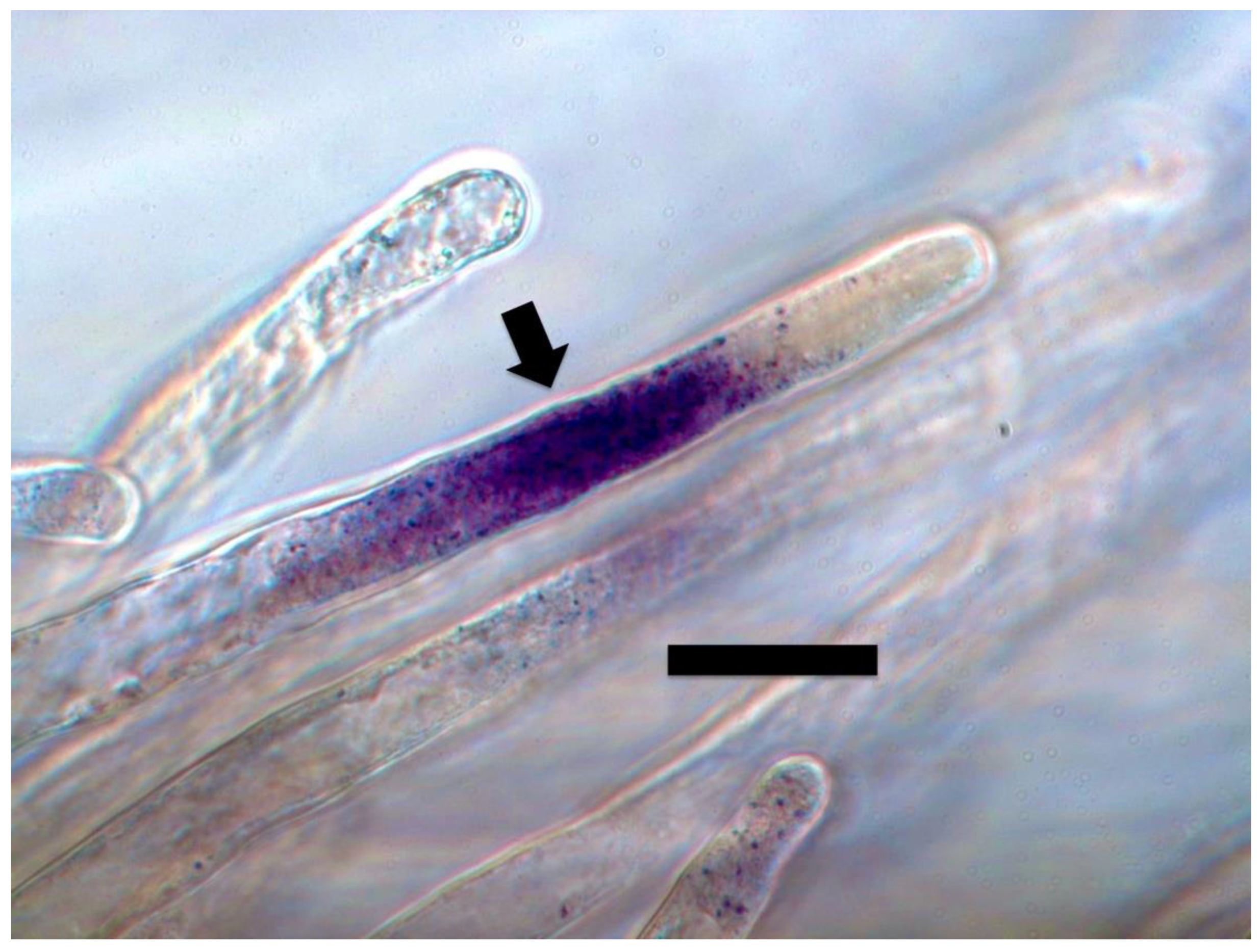
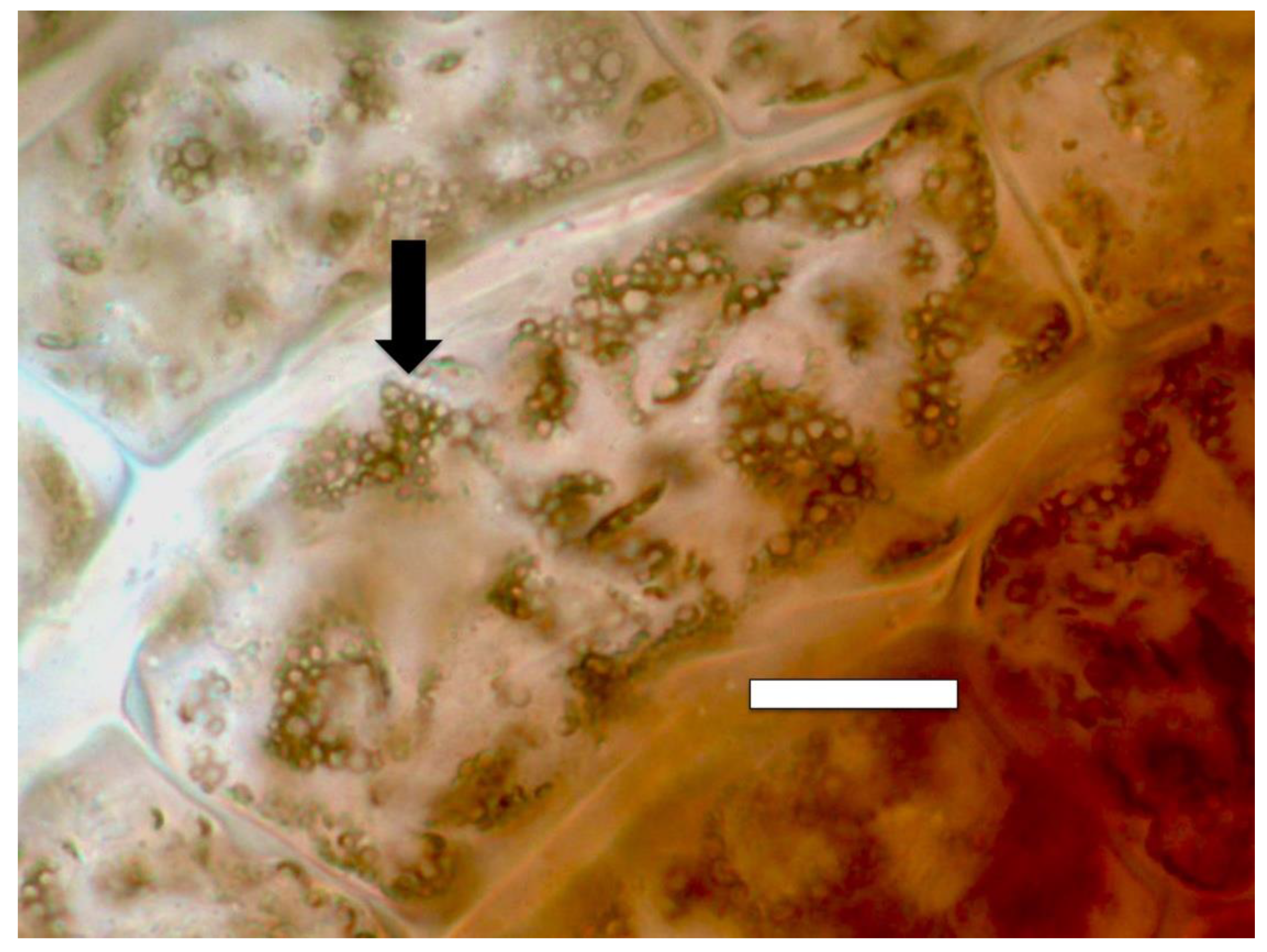
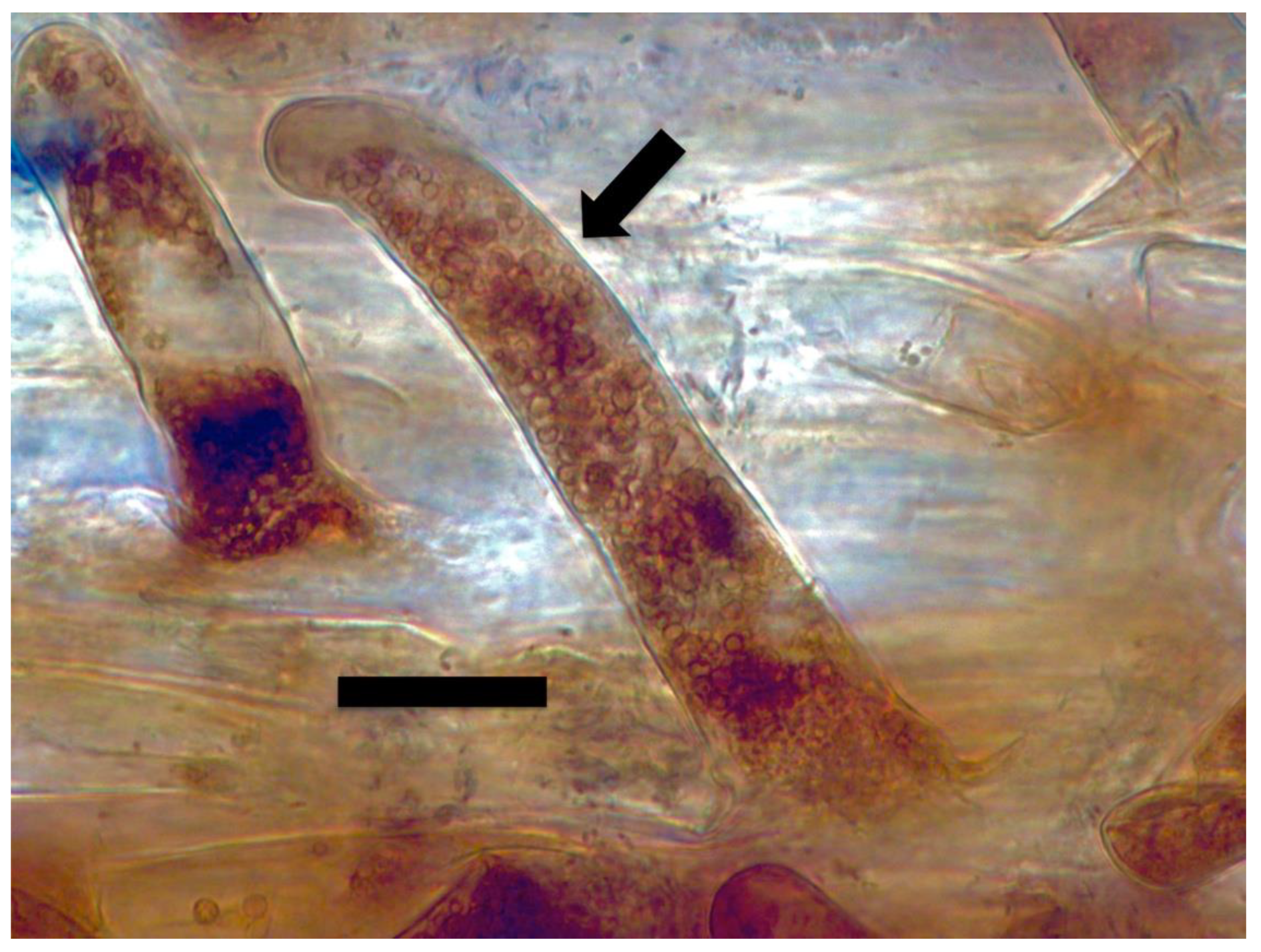
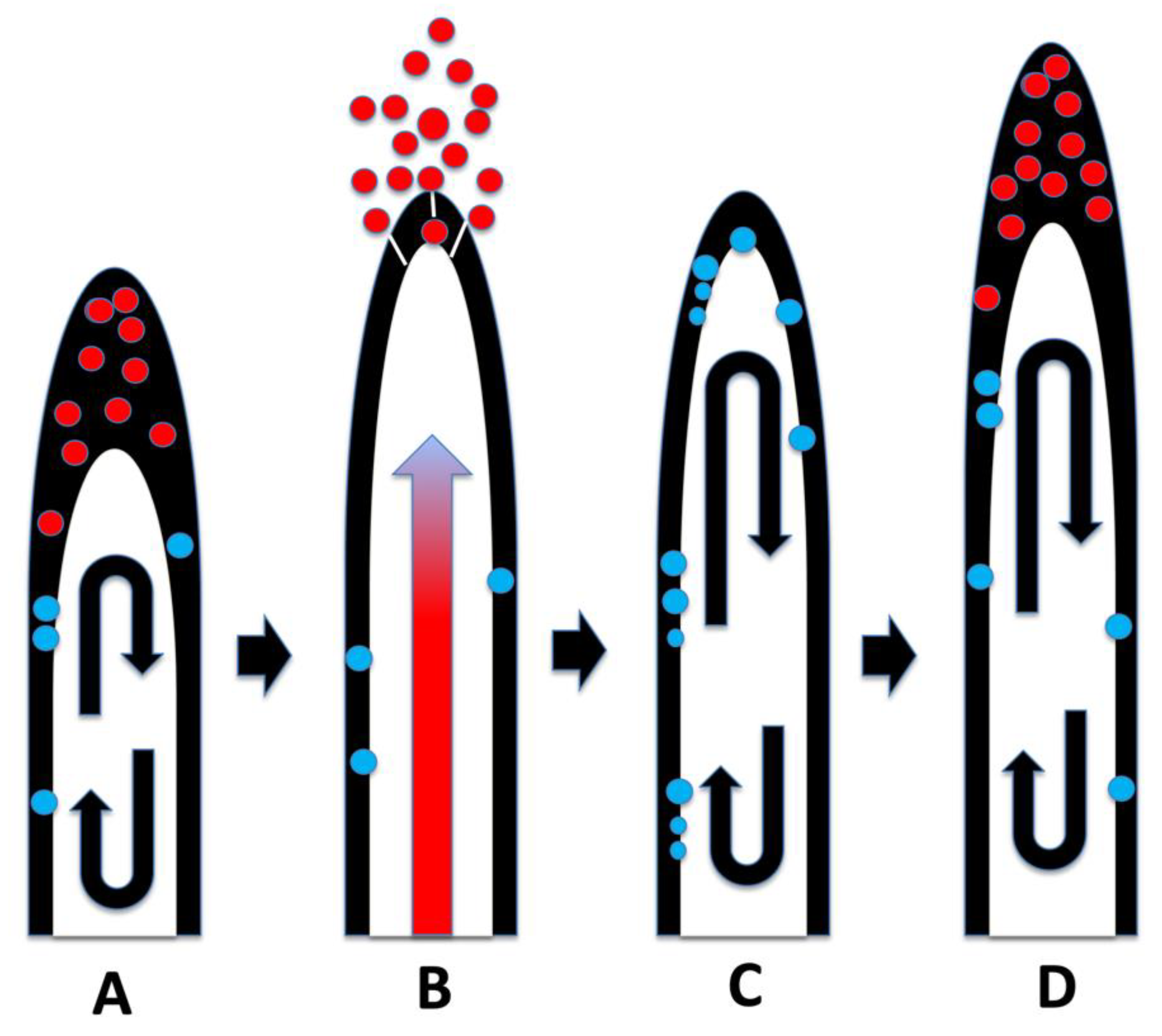
3. Results and Discussion
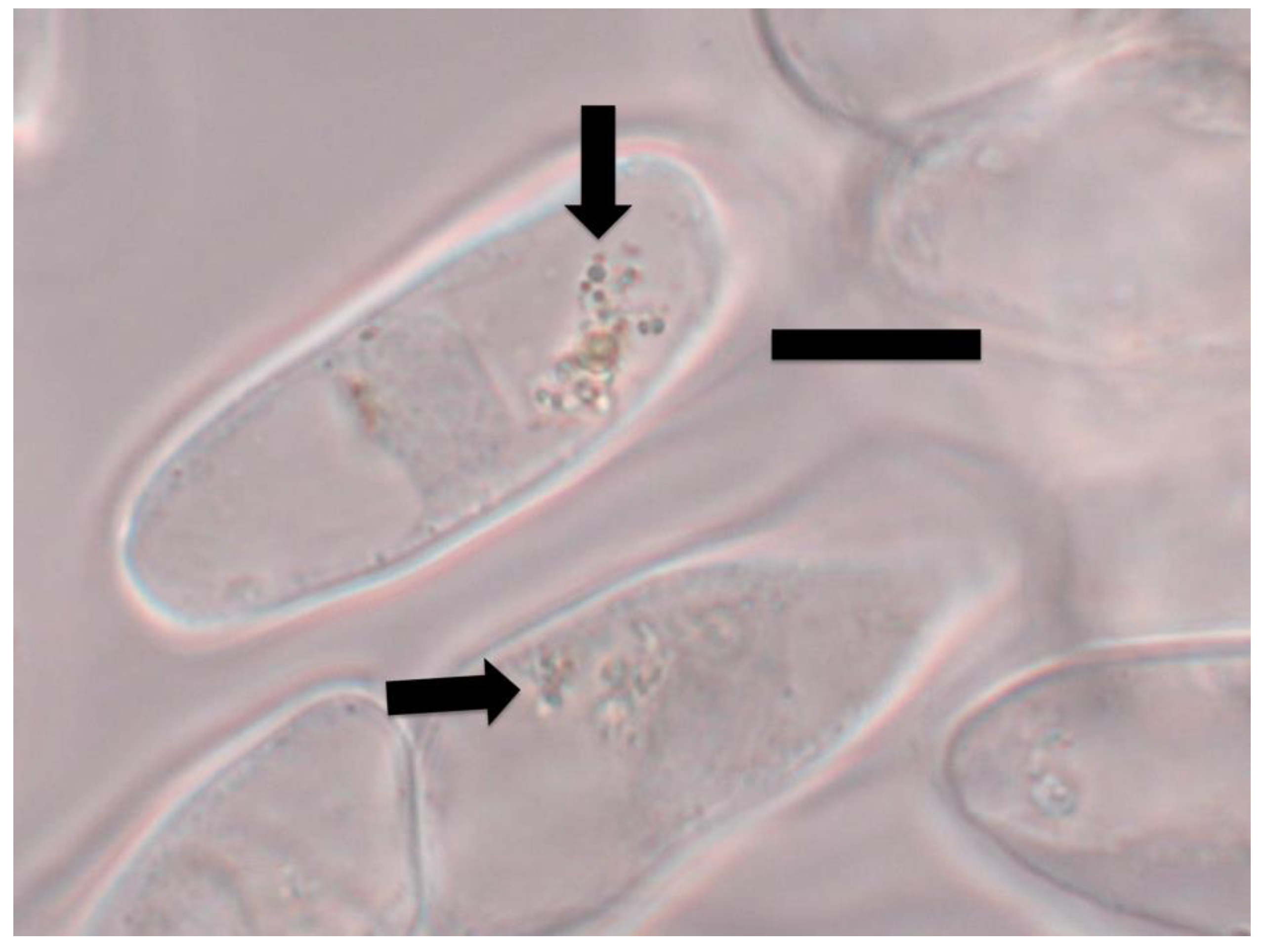
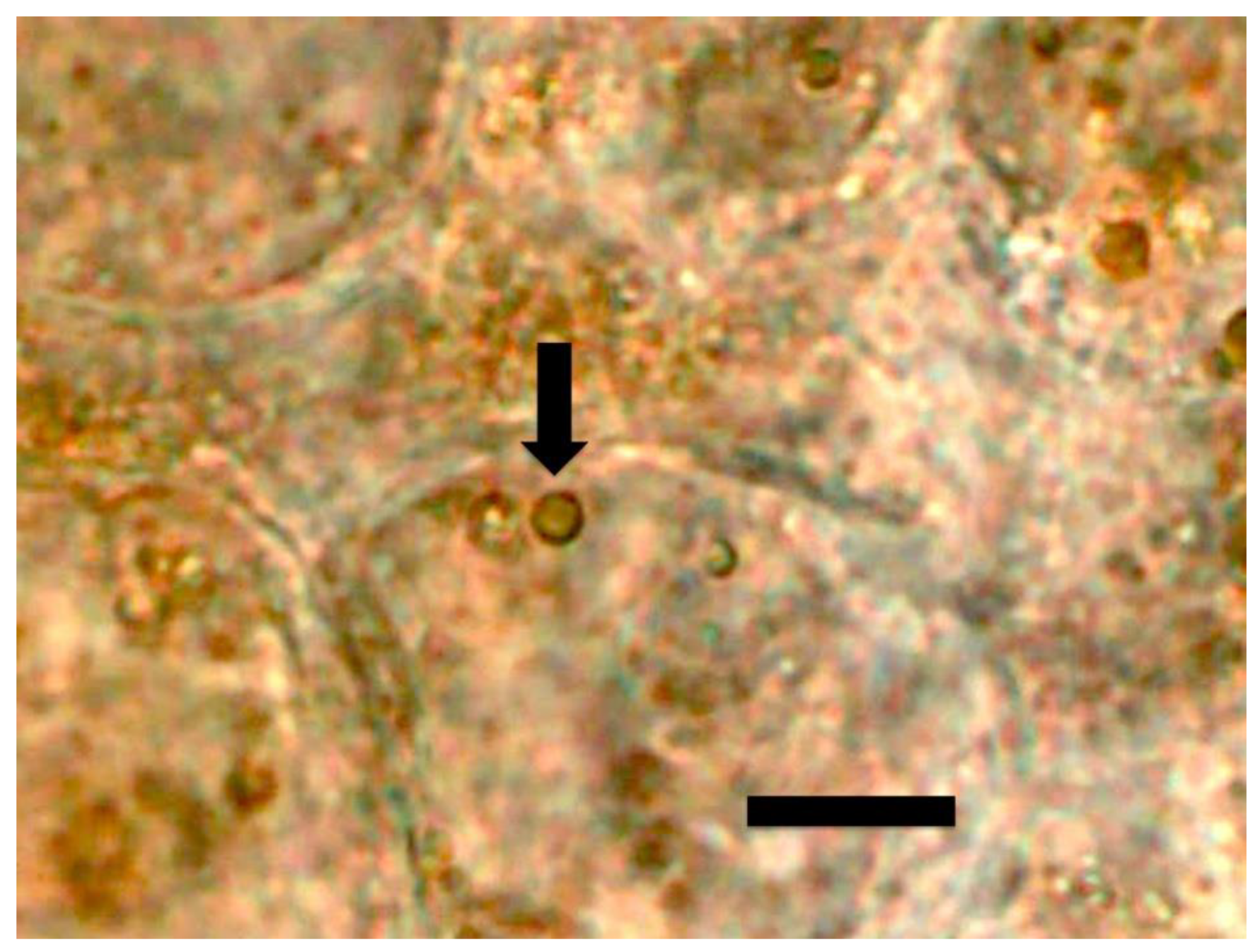
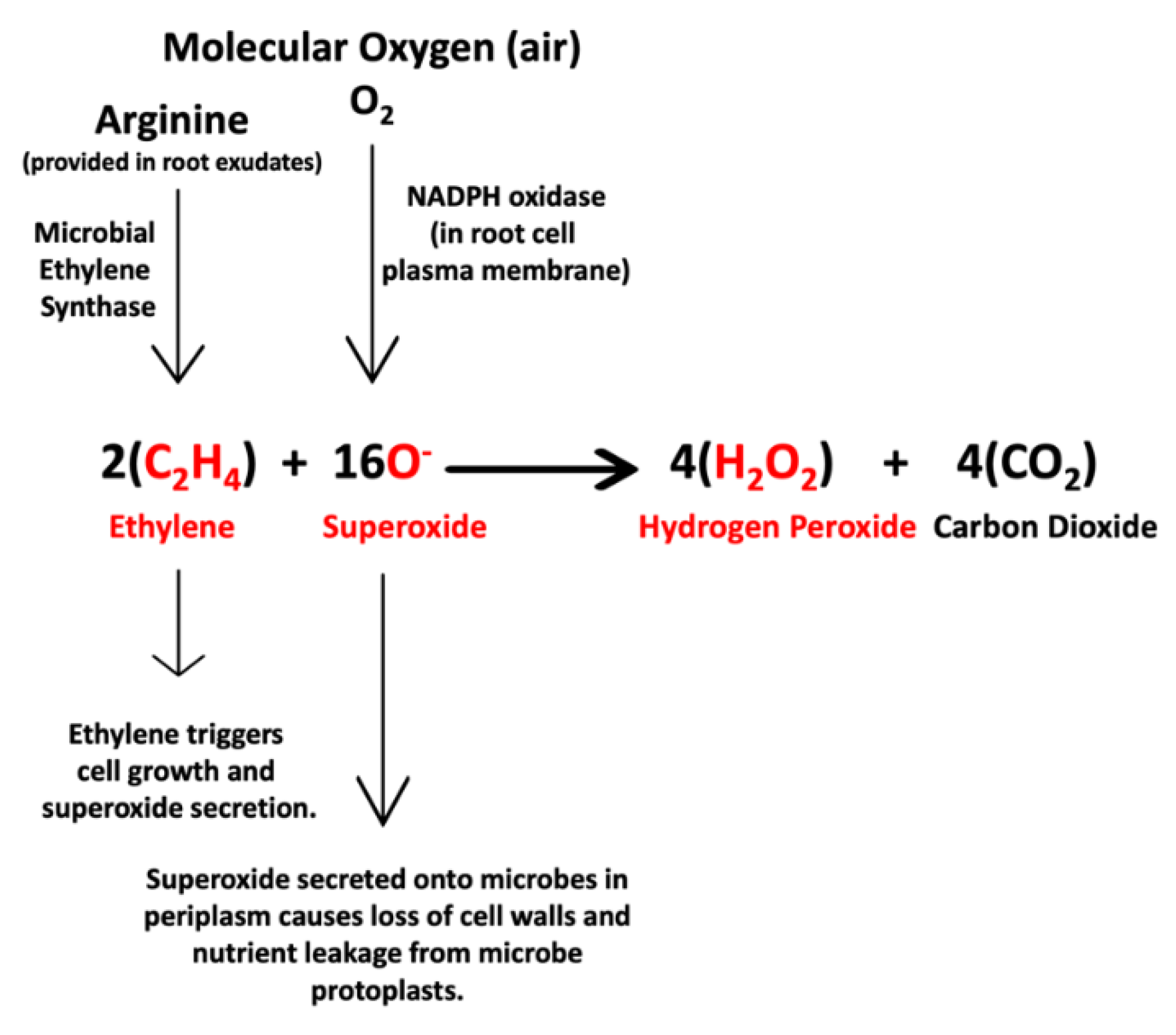
3.1. Evidence for the First Chemical Interaction
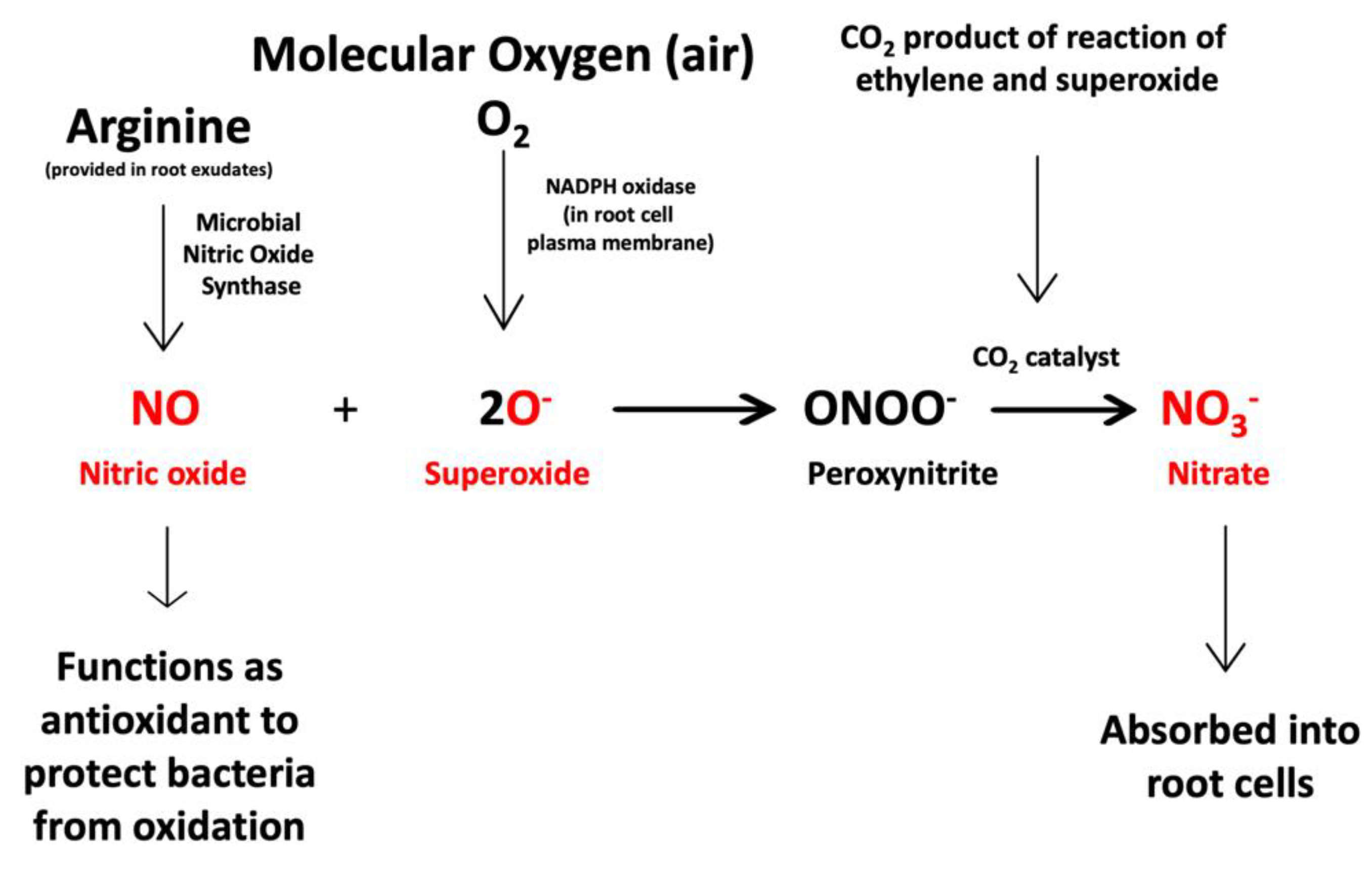
3.2. Evidence for the Second Chemical Interaction
3.3. Evidence Supporting Functions of Components of the First Chemical Interaction
3.4. Evidence Supporting Functions of Components of the Second Chemical Interaction
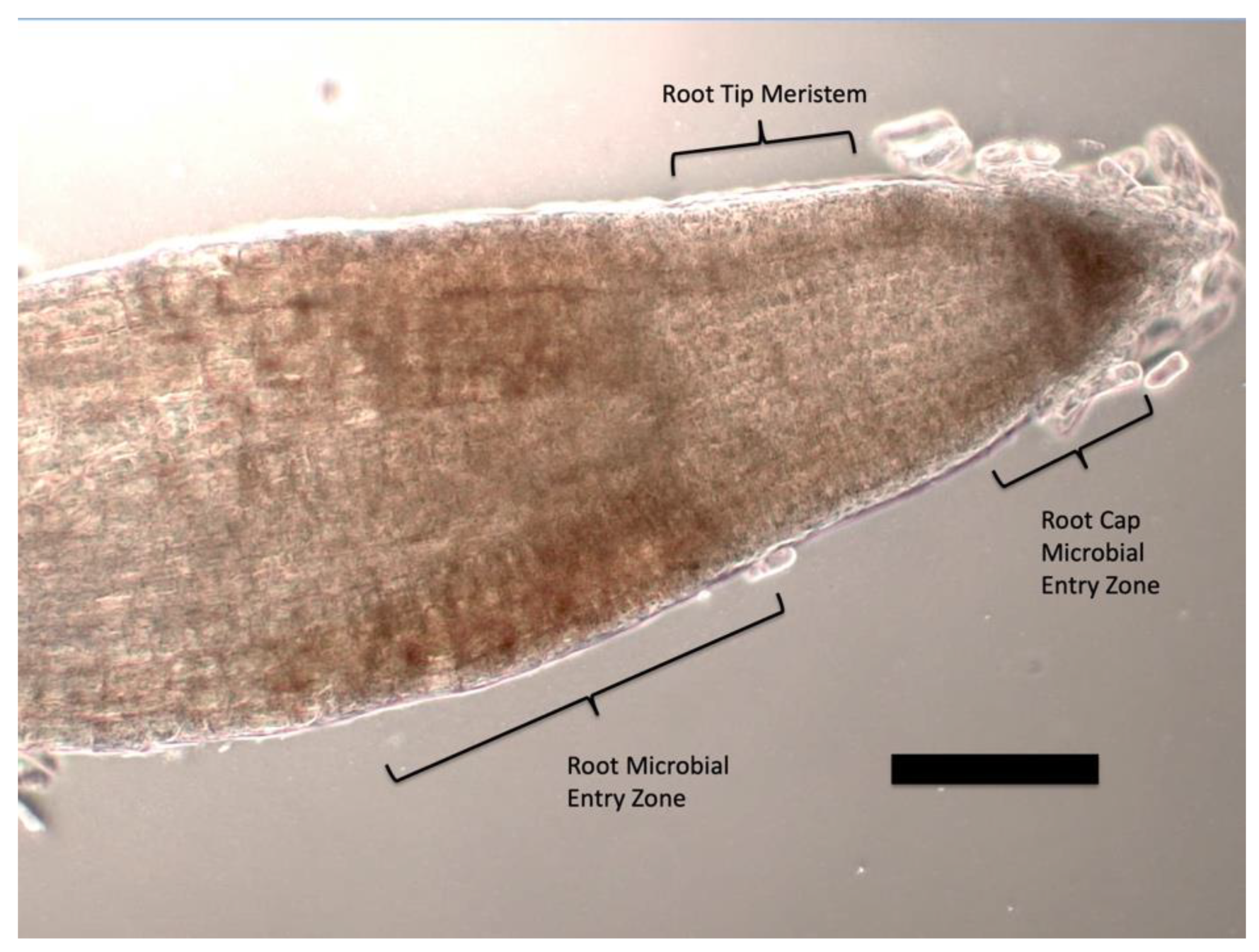
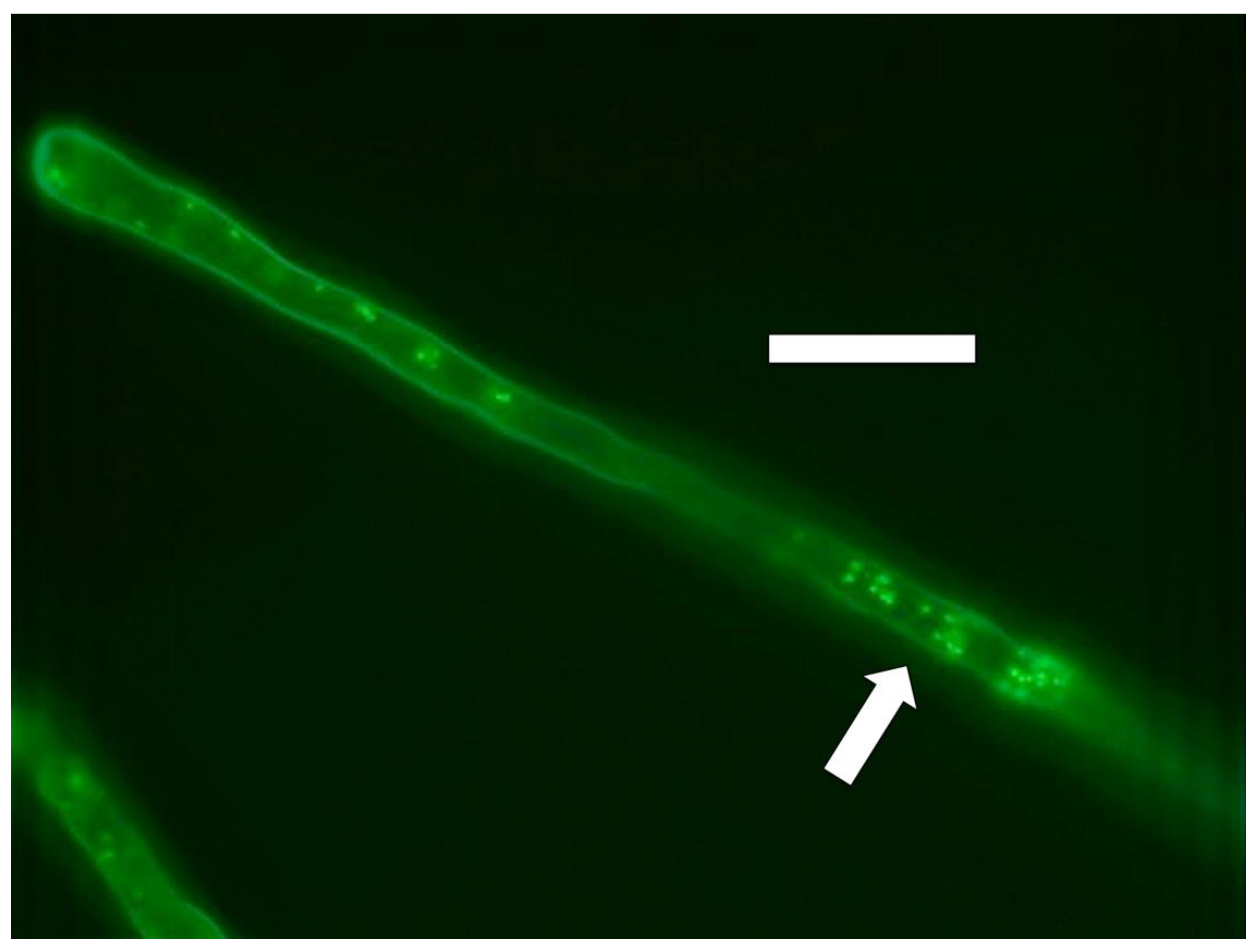
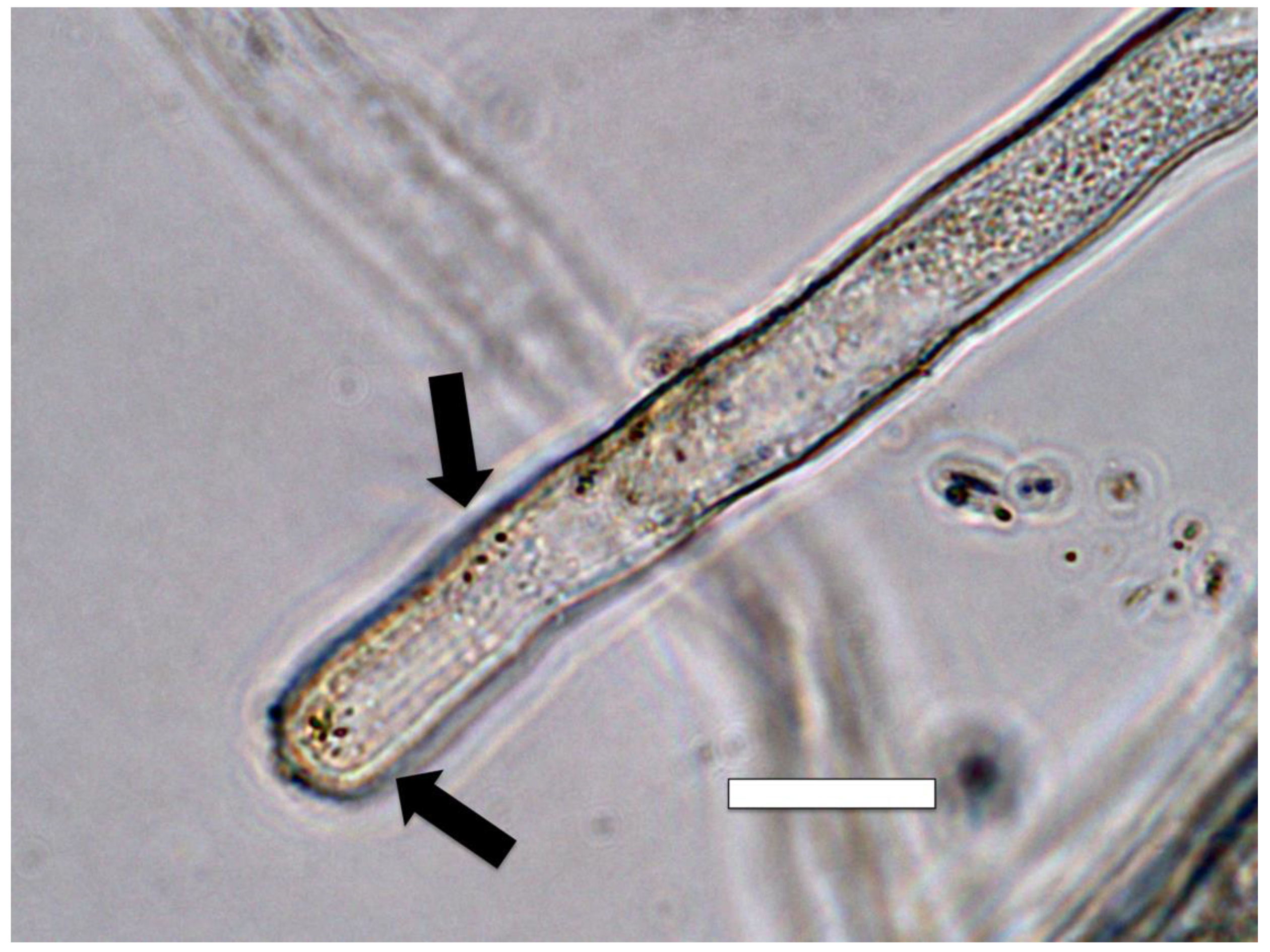
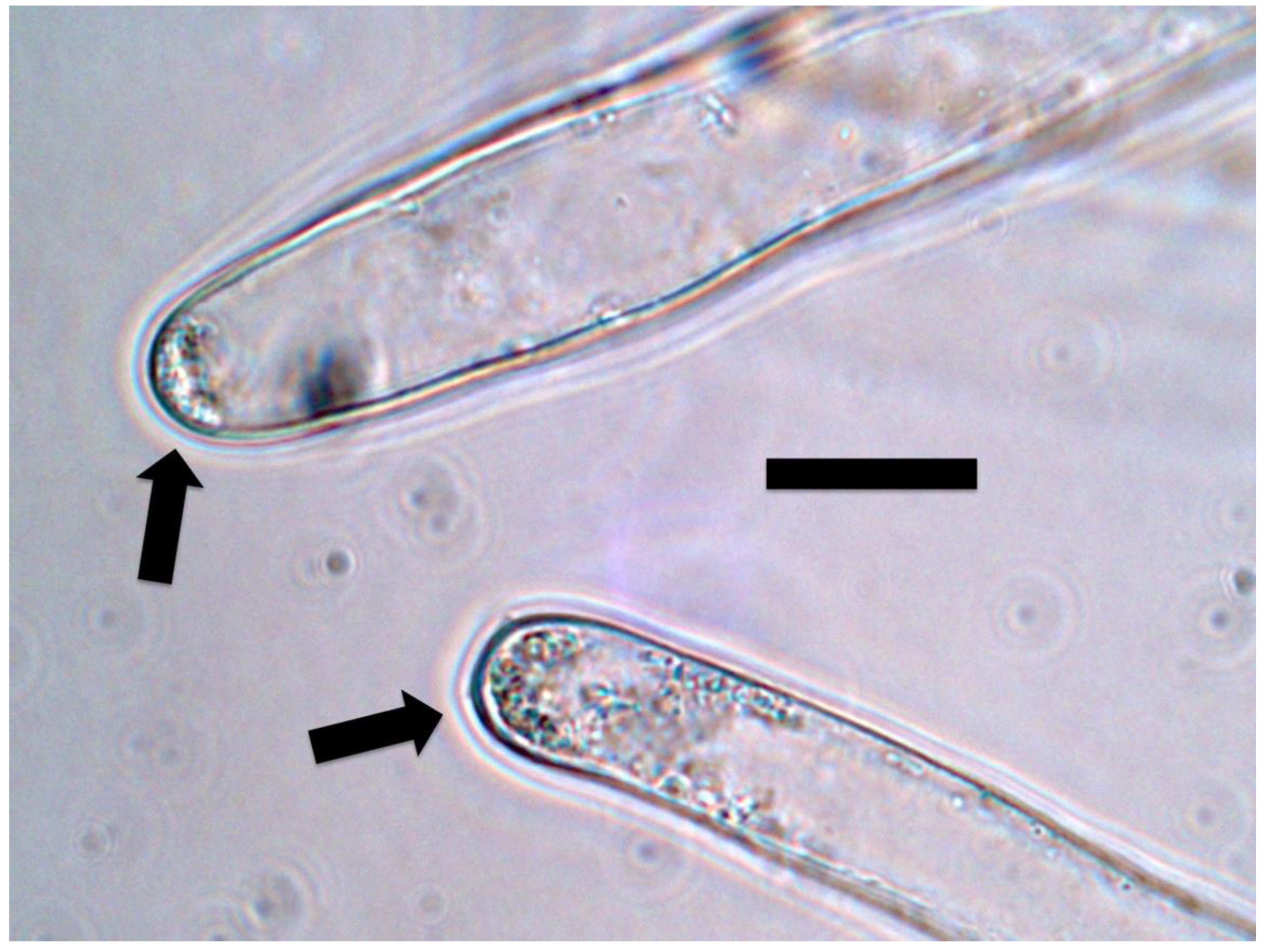
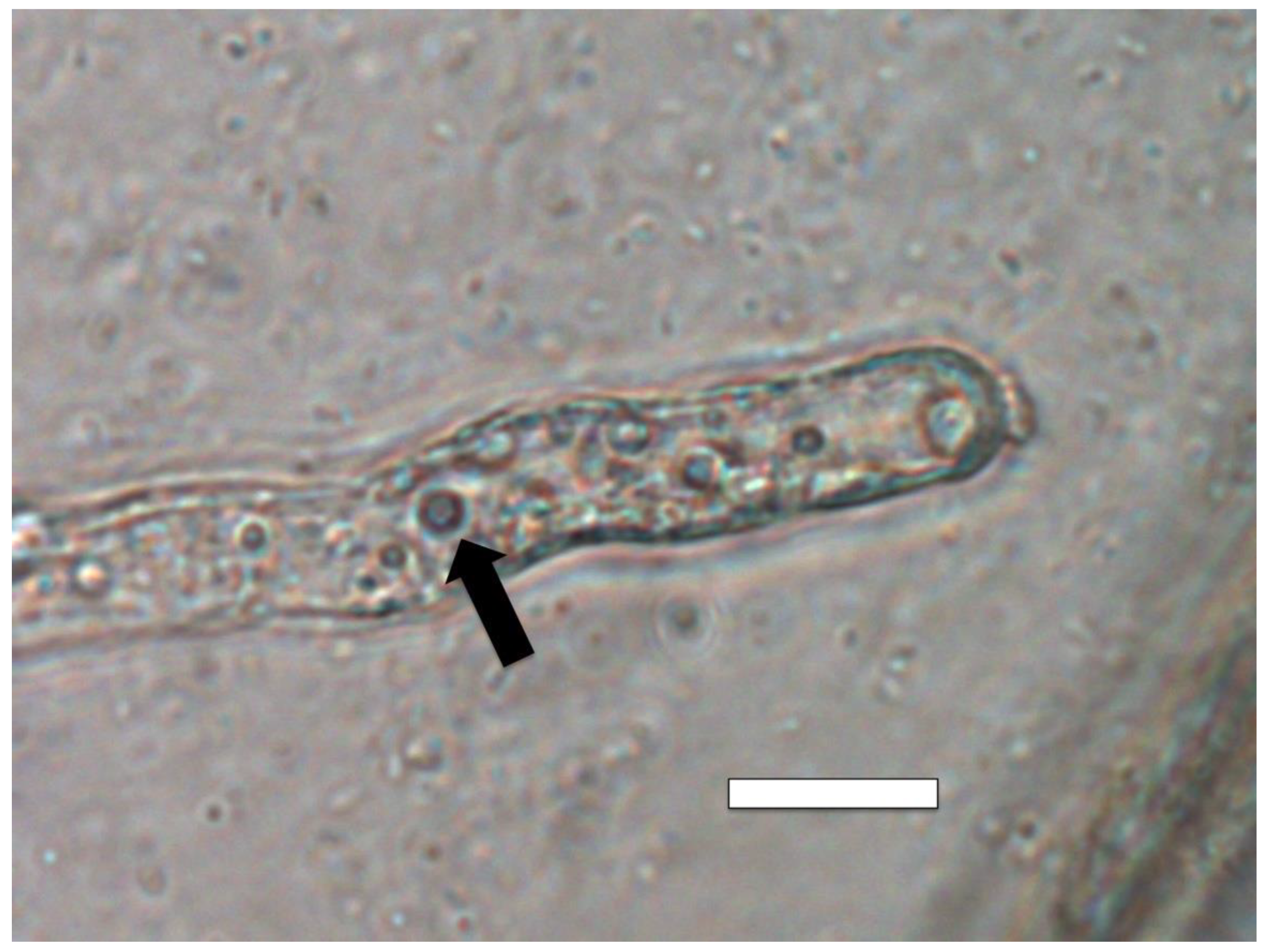
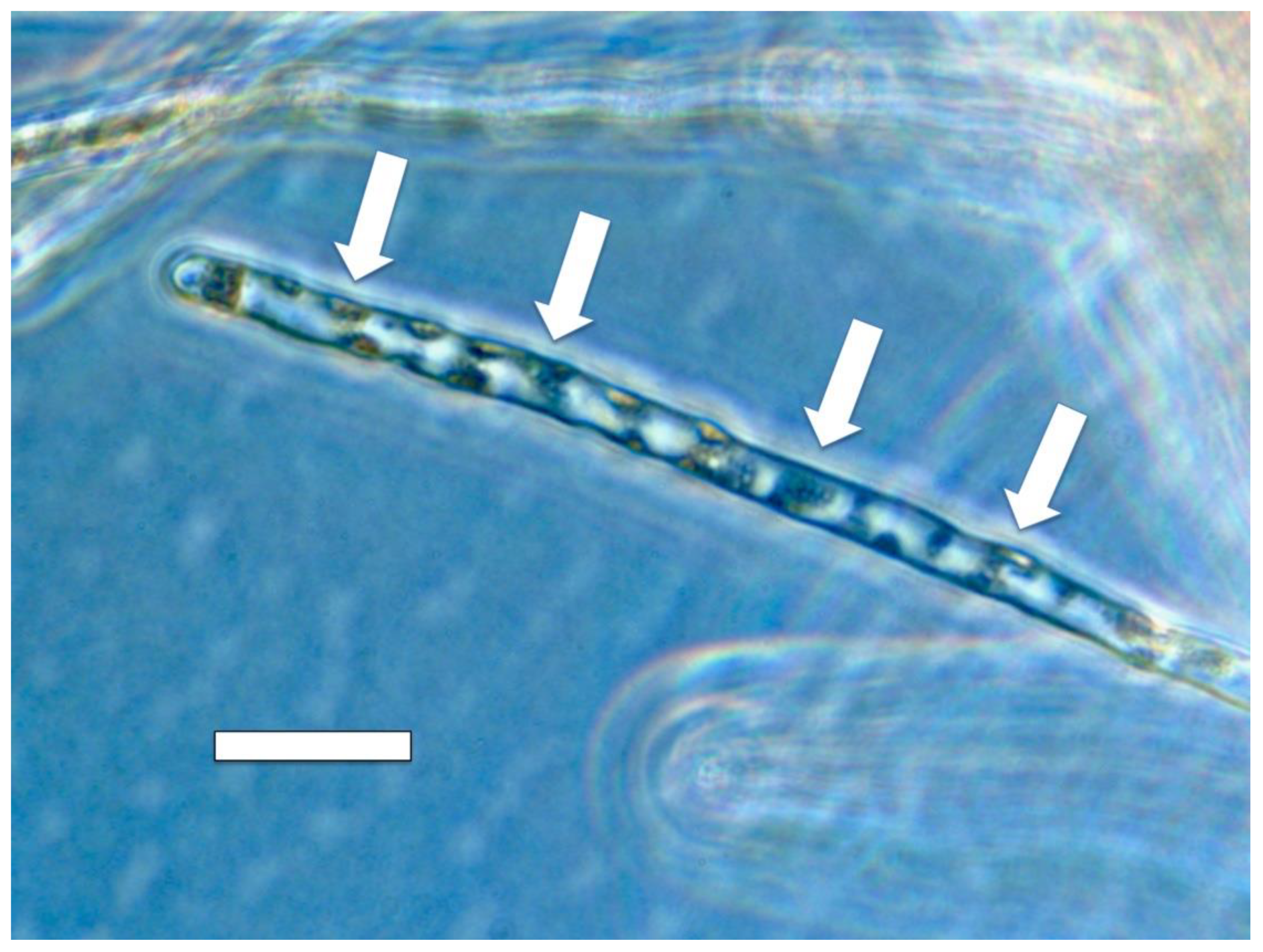
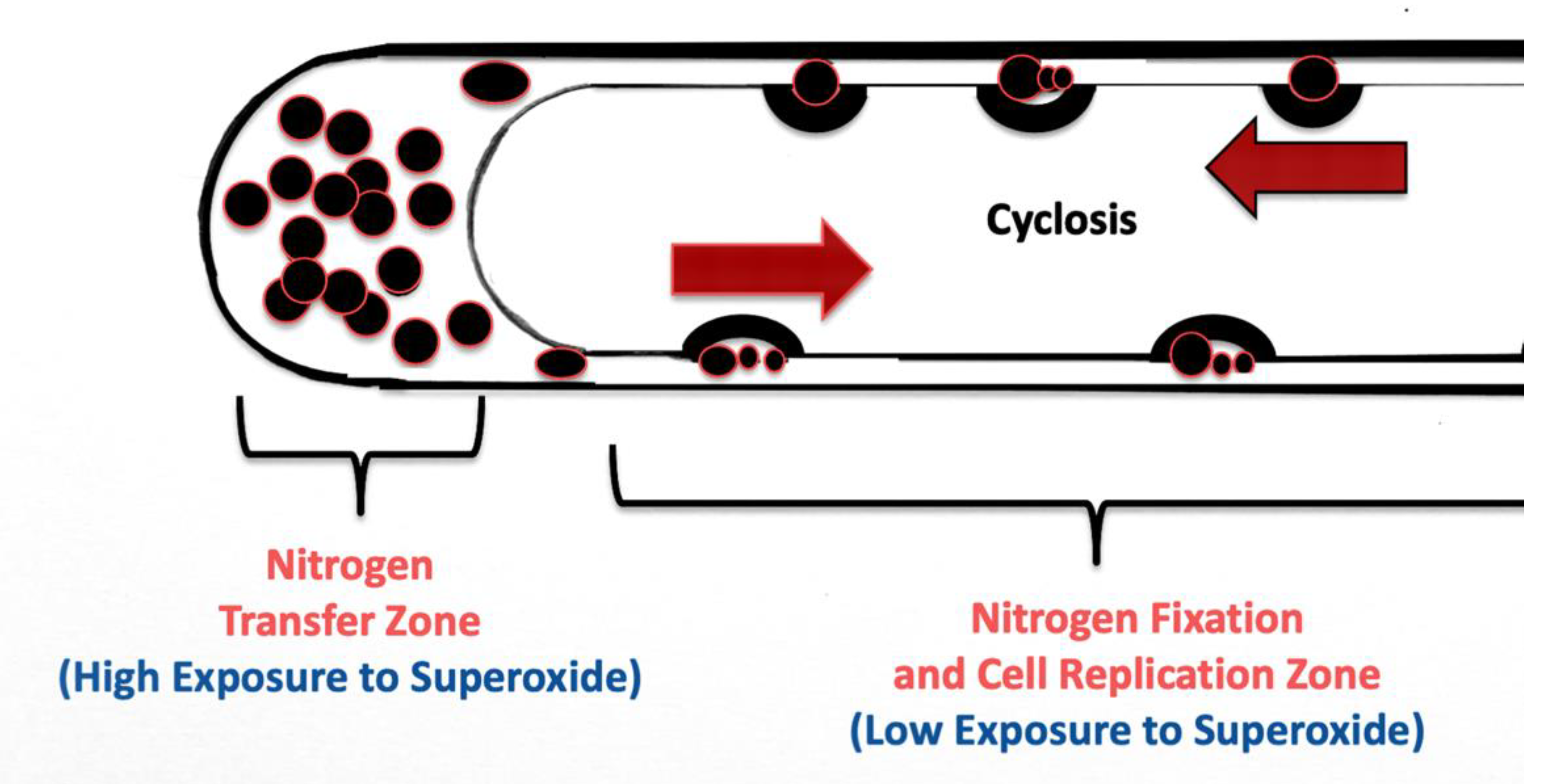
3.5. Nutrient Trap for Intracellular Microbes
3.6. Significance of Ethylene Secretion for Root Growth, Timing of Root Hair Growth Spurts and Ejection of Bacteria from Tips of Root Hairs
3.7. Increased Oxidative Stress Tolerance in Plants
3.8. Role of Microbe-Produced Auxins in Modulating Plant Growth
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Doty, S.L. (Ed.) Endophytic N-fixation: Controversy and a path forward. In Functional Importance of the Plant Microbiome; Springer International Publishing: New York, NY, USA, 2017; pp. 17–58. [Google Scholar]
- Hardoim, P.R.; van Overbeek, L.S.; Berg, G.; Pirttilä, A.M.; Compant, S.; Campisano, A.; Döring, M.; Sessitsch, A. The hidden world within plants: Ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol. Rev. 2015, 79, 293–320. [Google Scholar] [CrossRef]
- Macedo-Raygoza, G.M.; Valdez-Salas, B.; Prado, F.M.; Prieto, K.R.; Yamaguchi, L.F.; Kato, M.J.; Canto-Canché, B.B.; Carrillo-Beltrán, M.; Di Mascio, P.; White, J.F.; et al. Enterobacter cloacae, an endophyte that establishes a nutrient-transfer symbiosis with banana plants and protects against the black Ssgatoka pathogen. Front. Microbiol. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Prieto, P.; Schilirò, E.; Maldonado-González, M.M.; Valderrama, R.; Barroso-Albarracín, J.B.; Mercado-Blanco, J. Root hairs play a key role in the endophytic colonization of olive roots by Pseudomonas spp. with biocontrol activity. Microb. Ecol. 2011, 62, 435–445. [Google Scholar] [CrossRef]
- Shehata, H.R.; Dumigan, C.; Watts, S.; Raizada, M.N. An endophytic microbe from an unusual volcanic swamp corn seeks and inhabits root hair cells to extract rock phosphate. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Agrawal, M.; Bharathkumar, C.B. Diverse cellular colonizing endophytic bacteria in field shoots and in vitro cultured papaya with physiological and functional implications. Physiol. Plant 2019, 166, 729–747. [Google Scholar] [CrossRef]
- Bacon, C.W.; White, J.F. Functions, mechanisms and regulation of endophytic and epiphytic microbial communities of plants. Symbiosis 2016, 68, 87–98. [Google Scholar] [CrossRef]
- Compant, S.; Duffy, B.; Nowak, J.; Clément, C.; Barka, E.A. Use of plant growth-promoting bacteria for biocontrol of plant diseases: Principles, mechanisms of action, and future prospects. Appl. Environ. Microbiol. 2005, 71, 4951–4959. [Google Scholar] [CrossRef] [PubMed]
- Foreman, J.; Demidchik, V.; Bothwell, J.H.F.; Mylona, P.; Miedema, H.; Angel Torres, M.; Linstead, P.; Costa, S.; Brownlee, C.; Jones, J.D.G.; et al. Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 2013, 422, 442–446. [Google Scholar] [CrossRef]
- Kandel, S.; Joubert, P.; Doty, S. Bacterial endophyte colonization and distribution within plants. Microorganisms 2017, 5, 77. [Google Scholar] [CrossRef]
- Khare, E.; Mishra, J.; Arora, N.K. Multifaceted Interactions between Endophytes and Plant: Developments and Prospects. Front. Microbiol. 2018, 9, 2732. [Google Scholar] [CrossRef]
- Paungfoo-Lonhienne, C.; Rentsch, D.; Robatzek, S.; Webb, R.I.; Sagulenko, E.; Näsholm, T.; Schmidt, S.; Lonhienne, T.G.A. Turning the table: Plants consume microbes as a source of nutrients. PLoS ONE 2010, 5, e0011915. [Google Scholar] [CrossRef]
- Paungfoo-Lonhienne, C.; Schmidt, S.; Webb, R.I.; Lonhienne, T.G.A. Rhizophagy-A New Dimension of Plant-Microbe Interactions. In Molecular Microbial Ecology of the Rhizosphere; John Wiley and Sons: Hoboken, NJ, USA, 2013; pp. 1199–1207. [Google Scholar]
- White, J.; Kingsley, K.; Verma, S.; Kowalski, K. Rhizophagy Cycle: An Oxidative Process in Plants for Nutrient Extraction from Symbiotic Microbes. Microorganisms 2018, 6, 95. [Google Scholar] [CrossRef]
- White, J.F.; Kingsley, K.L.; Zhang, Q.; Verma, R.; Obi, N.; Dvinskikh, S.; Elmore, M.T.; Verma, S.K.; Gond, S.K.; Kowalski, K.P. Review: Endophytic microbes and their potential applications in crop management. Pest Manag. Sci. 2019, 75, 2558–2565. [Google Scholar] [CrossRef]
- Atsatt, P.R.; Whiteside, M.D. Novel symbiotic protoplasts formed by endophytic fungi explain their hidden existence, lifestyle switching, and diversity within the plant kingdom. PLoS ONE 2014, 9, e0095266. [Google Scholar] [CrossRef]
- Errington, J. L-form bacteria, cell walls and the origins of life. Open Biol. 2013, 3, 120143. [Google Scholar] [CrossRef]
- White, J.F.; Chen, Q.; Torres, M.S.; Mattera, R.; Irizarry, I.; Tadych, M.; Bergen, M. Collaboration between grass seedlings and rhizobacteria to scavenge organic nitrogen in soils. Aob Plants 2015, 7, 1–11. [Google Scholar] [CrossRef]
- Kawai, Y.; Mercier, R.; Wu, L.J.; Domínguez-Cuevas, P.; Oshima, T.; Errington, J. Cell growth of wall-free L-Form bacteria is limited by oxidative damage. Curr. Biol. 2015, 25, 1613–1618. [Google Scholar] [CrossRef]
- Verma, S.K.; Kharwar, R.N.; White, J.F. The role of seed-vectored endophytes in seedling development and establishment. Symbiosis 2019, 78, 107–113. [Google Scholar] [CrossRef]
- Verma, S.K.; Kingsley, K.; Irizarry, I.; Bergen, M.; Kharwar, R.N.; White, J.F. Seed-vectored endophytic bacteria modulate development of rice seedlings. J. Appl. Microbiol. 2017, 122, 1680–1691. [Google Scholar] [CrossRef]
- White, J.F.; Kingsley, K.I.; Kowalski, K.P.; Irizarry, I.; Micci, A.; Soares, M.A.; Bergen, M.S. Disease protection and allelopathic interactions of seed-transmitted endophytic pseudomonads of invasive reed grass (Phragmites australis). Plant Soil 2017, 422, 195–208. [Google Scholar] [CrossRef]
- Hill, P.W.; Marsden, K.A.; Jones, D.L. How significant to plant N nutrition is the direct consumption of soil microbes by roots? New Phytol. 2013, 199, 948–955. [Google Scholar] [CrossRef]
- White, J.F.; Kingsley, K.; Harper, C.J.; Verma, S.K.; Brindisi, L.; Chen, Q.; Chang, X.; Micci, A.; Bergen, M. Reactive Oxygen Defense Against Cellular Endoparasites and the Origin of Eukaryotes. In Transformative Paleobotany; Elsevier: Amsterdam, The Netherlands, 2018; pp. 439–460. [Google Scholar]
- White, J.F.; Torres, M.S.; Verma, S.K.; Elmore, M.T.; Kowalski, K.P.; Kingsley, K.L. Evidence for widespread microbivory of endophytic bacteria in roots of vascular plants through oxidative degradation in root cell periplasmic spaces. In PGPR Amelioration in Sustainable Agriculture; Elsevier: Amsterdam, The Netherlands, 2019; pp. 167–193. [Google Scholar]
- White, J.F.; Crawford, H.; Torres, M.S.; Mattera, R.; Irizarry, I.; Bergen, M. A proposed mechanism for nitrogen acquisition by grass seedlings through oxidation of symbiotic bacteria. Symbiosis 2012, 57, 161–171. [Google Scholar] [CrossRef]
- White, J.F.; Torres, M.S. Is plant endophyte-mediated defensive mutualism the result of oxidative stress protection? Physiol. Plant. 2010, 138, 440–446. [Google Scholar] [CrossRef]
- White, J.F.; Torres, M.S.; Somu, M.P.; Johnson, H.; Irizarry, I.; Chen, Q.; Zhang, N.; Walsh, E.; Tadych, M.; Bergen, M. Hydrogen peroxide staining to visualize intracellular bacterial infections of seedling root cells. Microsc. Res. Tech. 2014, 77, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Lang, C.; Hübert, T. A colour ripeness indicator for apples. Food Bioprocess Technol. 2012, 5, 3244–3249. [Google Scholar] [CrossRef]
- Chae, H.S.; Lee, W.S. Ethylene- and enzyme-mediated superoxide production and cell death in carrot cells grown under carbon starvation. Plant Cell Rep. 2001, 20, 256–261. [Google Scholar] [CrossRef]
- Schwendemann, J.; Sehringer, B.; Noethling, C.; Zahradnik, H.P.; Schaefer, W.R. Nitric oxide detection by DAF (diaminofluorescein) fluorescence in human myometrial tissue. Gynecol. Endocrinol. 2008, 24, 306–311. [Google Scholar] [CrossRef]
- Holtzclaw, H.; Robinson, W. College Chemistry with Qualitative Analysis, 8th ed.; D.C. Heath: Lexington, MA, USA, 1988; pp. 1006–1007. [Google Scholar]
- Coldwell, B.B.; McLean, S.R. The reaction between diphenylamine and nitrates in ultraviolet light. Can. J. Chem. 1959, 37, 1637–1643. [Google Scholar] [CrossRef]
- Bolevich, S.; Alekandr Haritonovic Kogan, H.; Zivkovic, V.; Djuric, D.; Aleksey Novikov, A.; Sergey Vorobyev, I.; Jakovljevic, V. Protective role of carbon dioxide (CO2) in generation of reactive oxygen species. Mol. Cell. Biochem. 2016, 411, 317–330. [Google Scholar] [CrossRef]
- Binder, B.M.; Eric Schaller, G. (Eds.) Ethylene Signaling; Springer: New York, NY, USA, 2017. [Google Scholar]
- Baker, J.E.; Anderson, J.D.; Adams, D.O.; Apelbaum, A.; Lieberman, M. Biosynthesis of ethylene from methionine in aminoethoxyvinylglycine-resistant avocado tissue. Plant Physiol. 1982, 69, 93–97. [Google Scholar] [CrossRef]
- Mayer, B.; Brunner, F.; Schmidt, K. Inhibition of nitric oxide synthesis by methylene blue. Biochem. Pharm. 1993, 45, 367–374. [Google Scholar] [CrossRef]
- Xia, J.; Yamaji, N.; Che, J.; Shen, R.F.; Ma, J.F. Normal root elongation requires arginine produced by argininosuccinate lyase in rice. Plant J. 2014, 78, 215–226. [Google Scholar] [CrossRef]
- Feng, Y.; Xu, P.; Li, B.; Li, P.; Wen, X.; An, F.; Gong, Y.; Xin, Y.; Zhu, Z.; Wang, Y.; et al. Ethylene promotes root hair growth through coordinated EIN3/EIL1 and RHD6/RSL1 activity in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, 13834–13839. [Google Scholar] [CrossRef]
- Lynch, J.; Brown, K.M. Ethylene and plant responses to nutritional stress. Physiol. Plant. 1997, 100, 613–619. [Google Scholar] [CrossRef]
- Pitts, R.J.; Cernac, A.; Estelle, M. Auxin and ethylene promote root hair elongation inArabidopsis. Plant J. 1998, 16, 553–560. [Google Scholar] [CrossRef]
- Trobacher, C.P. Ethylene and programmed cell death in plants. Botany 2009, 87, 757–769. [Google Scholar] [CrossRef]
- Chaudhari, S.S.; Kim, M.; Lei, S.; Razvi, F.; Alqarzaee, A.A.; Hutfless, E.H.; Powers, R.; Zimmerman, M.C.; Fey, P.D.; Thomas, V.C. Nitrite derived from endogenous bacterial nitric oxide synthase activity promotes aerobic respiration. MBio 2017, 8. [Google Scholar] [CrossRef]
- Crane, B.R.; Sudhamsu, J.; Patel, B.A. Bacterial nitric oxide synthases. Annu. Rev. Biochem. 2010, 79, 445–470. [Google Scholar] [CrossRef]
- He, H.; Oo, T.L.; Huang, W.; He, L.F.; Gu, M. Nitric oxide acts as an antioxidant and inhibits programmed cell death induced by aluminum in the root tips of peanut (Arachis hypogaea L.). Sci. Rep. 2019, 9, 9516. [Google Scholar] [CrossRef]
- Hummel, S.G.; Fischer, A.J.; Martin, S.M.; Schafer, F.Q.; Buettner, G.R. Nitric oxide as a cellular antioxidant: A little goes a long way. Free Radic. Biol. Med. 2006, 40, 501–506. [Google Scholar] [CrossRef]
- Wink, D.A.; Miranda, K.M.; Espey, M.G.; Pluta, R.M.; Hewett, S.J.; Colton, C.; Vitek, M.; Feelisch, M.; Grisham, M.B. Mechanisms of the antioxidant effects of nitric oxide. Antioxid. Redox Signal. 2001, 3, 203–213. [Google Scholar] [CrossRef]
- Hayashi, Y.; Sawa, Y.; Nishimura, M.; Fukuyama, N.; Ichikawa, H.; Ohtake, S.; Nakazawa, H.; Matsuda, H. Peroxynitrite, a product between nitric oxide and superoxide anion, plays a cytotoxic role in the development of post-bypass systemic inflammatory response. Eur. J. Cardio Thorac. Surg. 2004, 26, 276–280. [Google Scholar] [CrossRef][Green Version]
- Meli, R.; Nauser, T.; Latal, P.; Koppenol, W.H. Reaction of peroxynitrite with carbon dioxide: Intermediates and determination of the yield of CO3•− and NO2•. J. Biol. Inorg. Chem. 2002, 7, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Pryor, W.A.; Lemercier, J.N.; Zhang, H.; Uppu, R.M.; Squadrito, G.L. The catalytic role of carbon dioxide in the decomposition of peroxynitrite. Free Radic. Biol. Med. 1997, 23, 331–338. [Google Scholar] [CrossRef]
- Irizarry, I.; White, J.F. Bacillus amyloliquefaciens alters gene expression, ROS production and lignin synthesis in cotton seedling roots. J. Appl. Microbiol. 2018, 124, 1589–1603. [Google Scholar] [CrossRef]
- Monshausen, G.B.; Bibikova, T.N.; Messerli, M.A.; Shi, C.; Gilroy, S. Oscillations in extracellular pH and reactive oxygen species modulate tip growth of Arabidopsis root hairs. Proc. Natl. Acad. Sci. USA 2007, 104, 20996–21001. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.R.; Kramer, E.M.; Knox, K.; Swarup, R.; Bennett, M.J.; Lazarus, C.M.; Leyser, H.M.O.; Grierson, C.S. Auxin transport through non-hair cells sustains root-hair development. Nat. Cell Biol. 2009, 11, 78–84. [Google Scholar] [CrossRef]
- Sudhamsu, J.; Crane, R.B. Bacterial nitric oxide synthases: What are they good for? Trends Microbiol. 2019, 17, 212–218. [Google Scholar] [CrossRef]
- Hutfless, E.H.; Chaudhari, S.S.; Thomas, V.C. Emerging roles of nitric oxide synthase in bacterial physiology. In Advances in Microbial Physiology; Academic Press: Cambridge, MA, USA, 2018; pp. 147–191. [Google Scholar]
- Santana, M.M.; Gonzalez, J.M.; Cruz, C. Nitric oxide accumulation: The evolutionary trigger for phytopathogenesis. Front. Microbiol. 2017, 8, 1947. [Google Scholar] [CrossRef]
- Yarullina, D.R.; Il’inskaya, O.N.; Aganov, A.V.; Silkin, N.I.; Zverev, D.G. Alternative pathways of nitric oxide formation in Lactobacilli: Evidence for nitric oxide synthase activity by EPR. Microbiology 2006, 75, 634–638. [Google Scholar] [CrossRef]
- Jones, M.A.; Raymond, M.J.; Yang, Z.; Smirnoff, N. NADPH oxidase-dependent reactive oxygen species formation required for root hair growth depends on ROP GTPase. J. Exp. Bot. 2007, 58, 1261–1270. [Google Scholar] [CrossRef]
- Wang, S.S.; Zhu, X.N.; Lin, J.X.; Zheng, W.J.; Zhang, B.T.; Zhou, J.Q.; Ni, J.; Pan, Z.C.; Zhu, S.H.; Ding, W.N. OsNOX3, encoding a NADPH oxidase, regulates root hair initiation and elongation in rice. Biol. Plant. 2018, 6, 732–740. [Google Scholar] [CrossRef]
- Lombardo, M.C.; Graziano, M.; Polacco, J.C.; Lamattina, L. Nitric oxide functions as a positive regulator of root hair development. Plant Signal. Behav. 2006, 1, 28–33. [Google Scholar] [CrossRef]
- Lombardo, M.C.; Lamattina, L. Nitric oxide is essential for vesicle formation and trafficking in Arabidopsis root hair growth. J. Exp. Bot. 2012, 63, 4875–4885. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, H.; Fang, X.; Zhang, Y.; Jin, C. Auxin acts downstream of ethylene and nitric oxide to regulate magnesium deficiency-induced root hair development in Arabidopsis thaliana. Plant Cell Physiol. 2018, 59, 1452–1465. [Google Scholar] [CrossRef] [PubMed]
- Carvalhais, L.; Dennis, P.; Fedoseyenko, D.; Hajirezaei, M.-R.; Borriss, R.; von Wiren, N. Root exudation of sugars, amino acids, and organic acids by maize as affected by nitrogen, phosphorus, potassium, and iron deficiency. J. Plant Nutr. Soil Sci. 2010, 174, 3–11. [Google Scholar] [CrossRef]
- Grierson, C.; Schiefelbein, J. Root hairs. Arab. Book 2002, 1, e0060. [Google Scholar] [CrossRef]
- Mendarte-Alquisira, C.; Gutiérrez-Rojas, M.; González-Márquez, H.; Volke-Sepúlveda, T. Improved growth and control of oxidative stress in plants of Festuca arundinacea exposed to hydrocarbons by the endophytic fungus Lewia sp. Plant Soil 2017, 411, 347–358. [Google Scholar] [CrossRef]
- Verma, S.K.; Sahu, P.K.; Kumar, K.; Pal, G.; Gond, S.K.; Kharwar, R.N.; White, J.F. Endophyte roles in nutrient acquisition, root system architecture development and oxidative stress tolerance. J. Appl. Microbiol. 2021. [Google Scholar] [CrossRef]
- López-Bucio, J.; Campos-Cuevas, J.C.; Hernández-Calderón, E.; Velásquez-Becerra, C.; Farías-Rodríguez, R.; Macías-Rodríguez, L.I.; Valencia-Cantero, E. Bacillus megaterium rhizobacteria promote growth and alter root-system architecture through an auxin- and ethylene-independent signaling mechanism in Arabidopsis thaliana. Mol. Plant Microbe Interact. 2007, 20, 207–217. [Google Scholar] [CrossRef]
- Verbon, E.H.; Liberman, L.M. Beneficial microbes affect endogenous mechanisms controlling root development. Trends Plant Sci. 2016, 21, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wu, C.; Oelmüller, R.; Zhang, W. Role of Phytohormones in Piriformospora indica-Induced Growth Promotion and Stress Tolerance in Plants: More Questions Than Answers. Front. Microbiol. 2018, 9, 1646. [Google Scholar] [CrossRef]
- Hu, L.; Robert, C.A.M.; Cadot, S.; Zhang, X.; Ye, M.; Li, B.; Manzo, D.; Chervet, N.; Steinger, T.; Van Der Heijden, M.G.A.; et al. Root exudate metabolites drive plant-soil feedbacks on growth and defense by shaping the rhizosphere microbiota. Nat. Commun. 2018, 9, 1–13. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, X.; Kingsley, K.L.; White, J.F. Chemical Interactions at the Interface of Plant Root Hair Cells and Intracellular Bacteria. Microorganisms 2021, 9, 1041. https://doi.org/10.3390/microorganisms9051041
Chang X, Kingsley KL, White JF. Chemical Interactions at the Interface of Plant Root Hair Cells and Intracellular Bacteria. Microorganisms. 2021; 9(5):1041. https://doi.org/10.3390/microorganisms9051041
Chicago/Turabian StyleChang, Xiaoqian, Kathryn L. Kingsley, and James F. White. 2021. "Chemical Interactions at the Interface of Plant Root Hair Cells and Intracellular Bacteria" Microorganisms 9, no. 5: 1041. https://doi.org/10.3390/microorganisms9051041
APA StyleChang, X., Kingsley, K. L., & White, J. F. (2021). Chemical Interactions at the Interface of Plant Root Hair Cells and Intracellular Bacteria. Microorganisms, 9(5), 1041. https://doi.org/10.3390/microorganisms9051041






