Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii
Abstract
1. Introduction
2. Materials and Methods
2.1. Parasite Growth and Maintenance
2.2. Doubling Assays
2.3. MitoTracker Staining and Fluorescence Microscopy
2.4. Statistical Analyses
3. Results
3.1. Bisphenol Z and Its Derivatives Inhibit T. gondii Replication
3.2. The Parasite Mitochondrion Is Impacted by BPZ and Its Derivatives
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joiner, K.A.; Dubremetz, J.F. Toxoplasma gondii: A protozoan for the nineties. Infect. Immun. 1993, 61, 1169–1172. [Google Scholar] [CrossRef] [PubMed]
- Dubey, J.P. History of the discovery of the life cycle of Toxoplasma gondii. Int. J. Parasitol. 2009, 39, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Pappas, G.; Roussos, N.; Falagas, M.E. Toxoplasmosis snapshots: Global status of Toxoplasma gondii seroprevalence and implications for pregnancy and congenital toxoplasmosis. Int. J. Parasitol. 2009, 39, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Sousa, O.E.; Saenz, R.E.; Frenkel, J.K. Toxoplasmosis in Panama: A 10-year study. Am. J. Trop. Med. Hyg. 1988, 38, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.L.; Parise, M.E.; Fiore, A.E. Neglected parasitic infections in the United States: Toxoplasmosis. Am. J. Trop. Med. Hyg. 2014, 90, 794–799. [Google Scholar] [CrossRef]
- Guo, M.; Mishra, A.; Buchanan, R.L.; Dubey, J.P.; Hill, D.E.; Gamble, H.R.; Jones, J.L.; Pradhan, A.K. A Systematic Meta-Analysis of Toxoplasma gondii Prevalence in Food Animals in the United States. Foodborne Pathog. Dis. 2016, 13, 109–118. [Google Scholar] [CrossRef]
- Sheffield, H.G.; Melton, M.L. Toxoplasma gondii: The Oocyst, Sporozoite, and Infection of Cultured Cells. Science 1970, 167, 892–893. [Google Scholar] [CrossRef]
- Dubey, J.P.; Miller, N.L.; Frenkel, J.K. The Toxoplasma Gondii Oocyst from Cat Feces. J. Exp. Med. 1970, 132, 636–662. [Google Scholar] [CrossRef]
- Hutchison, W.M.; Dunachie, J.F.; Siim, J.C.; Work, K. Life cycle of Toxoplasma gondii. BMJ 1969, 4, 806. [Google Scholar] [CrossRef]
- Dubey, J.P. Advances in the life cycle of Toxoplasma gondii. Int. J. Parasitol. 1998, 28, 1019–1024. [Google Scholar] [CrossRef]
- Gilot-Fromont, E.; Lélu, M.; Dardé, M.-L.; Richomme, C.; Aubert, D.; Afonso, E.; Mercier, A.; Gotteland, C.; Villena, I. The Life Cycle of Toxoplasma gondii in the Natural Environment. In Toxoplasmosis - Recent Advances; Djaković, O.D., Ed.; InTechOpen: London, UK, 2012; pp. 3–36. [Google Scholar]
- Montoya, J.G.; Remington, J.S. Management of Toxoplasma gondii infection during pregnancy. Clin. Infect. Dis. 2008, 47, 554–566. [Google Scholar] [CrossRef] [PubMed]
- Krueger, W.S.; Hilborn, E.D.; Converse, R.R.; Wade, T.J. Drinking water source and human Toxoplasma gondii infection in the United States: A cross-sectional analysis of NHANES data. BMC Public Health 2014, 14, 711. [Google Scholar] [CrossRef] [PubMed]
- Wilking, H.; Thamm, M.; Stark, K.; Aebischer, T.; Seeber, F. Prevalence, incidence estimations, and risk factors of Toxoplasma gondii infection in Germany: A representative, cross-sectional, serological study. Sci. Rep. 2016, 6, 22551. [Google Scholar] [CrossRef] [PubMed]
- Belluco, S.; Simonato, G.; Mancin, M.; Pietrobelli, M.; Ricci, A. Toxoplasma gondii infection and food consumption: A systematic review and meta-analysis of case-controlled studies. Crit. Rev. Food Sci. Nutr. 2018, 58, 3085–3096. [Google Scholar] [CrossRef] [PubMed]
- Kervan, U.; Ozdamar, Y.; Yurdakok, O.; Kucuker, S.A.; Pac, M. A rare ocular complication after a heart transplant: Toxoplasma retinitis. Exp. Clin. Transplant. 2014, 12, 78–80. [Google Scholar] [CrossRef]
- Miltgen, G.; Paillot, J.; Vanlemmens, C.; Di Martino, V.; Heyd, B.; Millon, L.; Bellanger, A.-P. Fatal acute respiratory distress by Toxoplasma gondii in a Toxoplasma seronegative liver transplant recipient. New Microbiol. 2016, 39, 160–162. [Google Scholar]
- Betancourt, E.D.; Hamid, B.; Fabian, B.T.; Klotz, C.; Hartmann, S.; Seeber, F. From Entry to Early Dissemination—Toxoplasma gondii’s Initial Encounter with Its Host. Front. Cell. Infect. Microbiol. 2019, 9, 46. [Google Scholar] [CrossRef]
- McLeod, R.; Berry, P.F.; Marshall, W.H., Jr.; Hunt, S.A.; Ryning, F.W.; Remington, J.S. Toxoplasmosis presenting as brain abscesses. Diagnosis by computerized tomography and cytology of aspirated purulent material. Am. J. Med. 1979, 67, 711–714. [Google Scholar] [CrossRef]
- Luft, B.J.; Remington, J.S. Toxoplasmic encephalitis in AIDS. Clin. Infect. Dis. 1992, 15, 211–222. [Google Scholar] [CrossRef]
- Israelski, D.M.; Remington, J.S. Toxoplasmosis in the non-AIDS immunocompromised host. Curr. Clin. Top. Infect. Dis. 1993, 13, 322–356. [Google Scholar]
- Slavin, M.A.; Meyers, J.D.; Remington, J.S.; Hackman, R.C. Toxoplasma gondii infection in marrow transplant recipients: A 20 year experience. Bone Marrow Transplant. 1994, 13, 549–557. [Google Scholar] [PubMed]
- Gallino, A.; Maggiorini, M.; Kiowski, W.; Martin, X.; Wunderli, W.; Schneider, J.; Turina, M.; Follath, F. Toxoplasmosis in heart transplant recipients. Eur. J. Clin. Microbiol. Infect. Dis. 1996, 15, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.Y.; Remington, J.S. Toxoplasmosis in pregnancy. Clin. Infect. Dis. 1994, 18, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Henriquez, S.A.; Brett, R.; Alexander, J.; Pratt, J.; Roberts, C.W. Neuropsychiatric disease and Toxoplasma gondii infection. Neuroimmunomodulation 2009, 16, 122–133. [Google Scholar] [CrossRef]
- Martinez, V.O.; de Mendonça Lima, F.W.; de Carvalho, C.F.; Menezes-Filho, J.A. Toxoplasma gondii infection and behavioral outcomes in humans: A systematic review. Parasitol. Res. 2018, 117, 3059–3065. [Google Scholar] [CrossRef]
- Ortiz-Guerrero, G.; Gonzalez-Reyes, R.E.; de-la-Torre, A.; Medina-Rincón, G.; Nava-Mesa, M.O. Pathophysiological Mechanisms of Cognitive Impairment and Neurodegeneration by Toxoplasma gondii Infection. Brain Sci. 2020, 10, 369. [Google Scholar] [CrossRef]
- Neville, A.J.; Zach, S.J.; Wang, X.; Larson, J.J.; Judge, A.K.; Davis, L.A.; Vennerstrom, J.L.; Davis, P.H. Clinically Available Medicines Demonstrating Anti-Toxoplasma Activity. Antimicrob. Agents Chemother. 2015, 59, 7161–7169. [Google Scholar] [CrossRef]
- Maldonado, Y.A.; Read, J.S.; Committee on Infectious Diseases. Diagnosis, Treatment, and Prevention of Congenital Toxoplasmosis in the United States. Pediatrics 2017, 139, e20163860. [Google Scholar] [CrossRef]
- Porter, S.B.; Sande, M.A. Toxoplasmosis of the central nervous system in the acquired immunodeficiency syndrome. N. Engl. J. Med. 1992, 327, 1643–1648. [Google Scholar] [CrossRef]
- McLeod, R.; Khan, A.R.; Noble, G.A.; Latkany, P.; Jalbrzikowski, J.; Boyer, K. Severe sulfadiazine hypersensitivity in a child with reactivated congenital toxoplasmic chorioretinitis. Pediatric Infect. Dis. J. 2006, 25, 270–272. [Google Scholar] [CrossRef]
- Alday, P.H.; Doggett, J.S. Drugs in development for toxoplasmosis: Advances, challenges, and current status. Drug Des. Devel. Ther. 2017, 11, 273–293. [Google Scholar] [CrossRef] [PubMed]
- Doliwa, C.; Escotte-Binet, S.; Aubert, D.; Sauvage, V.; Velard, F.; Schmid, A.; Villena, I. Sulfadiazine resistance in Toxoplasma gondii: No involvement of overexpression or polymorphisms in genes of therapeutic targets and ABC transporters. Parasite 2013, 20, 19. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, M.; Mehrzadi, S.; Sharif, M.; Sarvi, S.; Tanzifi, A.; Aghayan, S.A.; Daryani, A. Drug Resistance in Toxoplasma gondii. Front. Microbiol. 2018, 9, 2587. [Google Scholar] [CrossRef] [PubMed]
- Stitzlein, L.M.; Stang, C.R.T.; Inbody, L.R.; Rao, P.S.S.; Schneider, R.A.; Dudley, R.W. Design, synthesis, and biological evaluation of lipophilically modified bisphenol Z derivatives. Chem. Biol. Drug Des. 2019, 94, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Salin, N.H.; Noordin, R.; Al-Najjar, B.O.; Kamarulzaman, E.E.; Yunus, M.H.; Karim, I.Z.A.; Nasim, N.N.M.; Zakaria, I.I.; Wahab, H.A. Identification of potential dual -targets anti- Toxoplasma gondii compounds through structure-based virtual screening and in-vitro studies. PLoS ONE 2020, 15, e0225232. [Google Scholar] [CrossRef]
- Pfefferkorn, E.R.; Borotz, S.E.; Nothnagel, R.F. Mutants of Toxoplasma gondii Resistant to Atovaquone (566C80) or Decoquinate. J. Parasitol. 1993, 79, 559. [Google Scholar] [CrossRef]
- Basto, A.P.; Müller, J.; Rubbiani, R.; Stibal, D.; Giannini, F.; Süss-Fink, G.; Balmer, V.; Hemphill, A.; Gasser, G.; Furrer, J. Characterization of the Activities of Dinuclear Thiolato-Bridged Arene Ruthenium Complexes against Toxoplasma gondii. Antimicrob. Agents Chemother. 2017, 61, e01031-17. [Google Scholar] [CrossRef]
- Strobl, J.S.; Seibert, C.W.; Li, Y.; Nagarkatti, R.; Mitchell, S.M.; Rosypal, A.C.; Rathore, D.; Lindsay, D.S. Inhibition of Toxoplasma gondii and Plasmodium falciparum infections in vitro by NSC3852, a redox active antiproliferative and tumor cell differentiation agent. J. Parasitol. 2009, 95, 215–223. [Google Scholar] [CrossRef]
- Chen, Q.-W.; Dong, K.; Qin, H.-X.; Yang, Y.-K.; He, J.-L.; Li, J.; Zheng, Z.-W.; Chen, D.-L.; Chen, J.-P. Direct and Indirect Inhibition Effects of Resveratrol against Toxoplasma gondii Tachyzoites In Vitro. Antimicrob. Agents Chemother. 2019, 63, e01233-18. [Google Scholar] [CrossRef]
- Nishi, M.; Hu, K.; Murray, J.M.; Roos, D.S. Organellar dynamics during the cell cycle of Toxoplasma gondii. J. Cell Sci. 2008, 121, 1559–1568. [Google Scholar] [CrossRef] [PubMed]
- Garbuz, T.; Arrizabalaga, G. Lack of mitochondrial MutS homolog 1 in Toxoplasma gondii disrupts maintenance and fidelity of mitochondrial DNA and reveals metabolic plasticity. PLoS ONE 2017, 12, e0188040. [Google Scholar] [CrossRef] [PubMed]
- Melo, E.J.L.; Attias, M.; De Souza, W. The Single Mitochondrion of Tachyzoites of Toxoplasma gondii. J. Struct. Biol. 2000, 130, 27–33. [Google Scholar] [CrossRef]
- Judge, D.M.; Anderson, M.S. Ultrastructure of trypanosoma lewisi. J. Parasitol. 1964, 50, 757–762. [Google Scholar] [CrossRef]
- Rudzinska, M.A.; Trager, W. Electron-microscope studies on malaria parasites. Triangle 1959, 4, 97–104. [Google Scholar] [PubMed]
- Charvat, R.A.; Arrizabalaga, G. Oxidative stress generated during monensin treatment contributes to altered Toxoplasma gondii mitochondrial function. Sci. Rep. 2016, 6, 22997. [Google Scholar] [CrossRef]
- Jeffers, V.; Kamau, E.T.; Srinivasan, A.R.; Harper, J.; Sankaran, P.; Post, S.E.; Varberg, J.M.; Sullivan, W.J., Jr.; Boyle, J.P. TgPRELID, a Mitochondrial Protein Linked to Multidrug Resistance in the Parasite. mSphere 2017, 2, e00229-16. [Google Scholar] [CrossRef]
- Lavine, M.D.; Arrizabalaga, G. Analysis of monensin sensitivity in Toxoplasma gondii reveals autophagy as a mechanism for drug induced death. PLoS ONE 2012, 7, e42107. [Google Scholar] [CrossRef]
- Desoti, V.C.; Lazarin-Bidóia, D.; Sudatti, D.B.; Pereira, R.C.; Alonso, A.; Ueda-Nakamura, T.; Dias Filho, B.P.; Nakamura, C.V.; de Oliveira Silva, S. Trypanocidal action of (-)-elatol involves an oxidative stress triggered by mitochondria dysfunction. Mar. Drugs 2012, 10, 1631–1646. [Google Scholar] [CrossRef]
- Garrison, E.M.; Arrizabalaga, G. Disruption of a mitochondrial MutS DNA repair enzyme homologue confers drug resistance in the parasite Toxoplasma gondii. Mol. Microbiol. 2009, 72, 425–441. [Google Scholar] [CrossRef]
- McFadden, D.C.; Tomavo, S.; Berry, E.A.; Boothroyd, J.C. Characterization of cytochrome b from Toxoplasma gondii and Q(o) domain mutations as a mechanism of atovaquone-resistance. Mol. Biochem. Parasitol. 2000, 108, 1–12. [Google Scholar] [CrossRef]
- Srivastava, I.K.; Rottenberg, H.; Vaidya, A.B. Atovaquone, a Broad Spectrum Antiparasitic Drug, Collapses Mitochondrial Membrane Potential in a Malarial Parasite. J. Biol. Chem. 1997, 272, 3961–3966. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lendahl, U.; Nistér, M. Regulation of mitochondrial dynamics: Convergences and divergences between yeast and vertebrates. Cell. Mol. Life Sci. 2013, 70, 951–976. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, K.; Charvat, R.; Arrizabalaga, G. Identification of Fis1 interactors in Toxoplasma gondii reveals a novel protein required for peripheral distribution of the mitochondrion. Mbio 2020, 11, e02732-19. [Google Scholar] [CrossRef] [PubMed]
- Heredero-Bermejo, I.; Varberg, J.M.; Charvat, R.; Jacobs, K.; Garbuz, T.; Sullivan, W.J., Jr.; Arrizabalaga, G. TgDrpC, an atypical dynamin-related protein in Toxoplasma gondii, is associated with vesicular transport factors and parasite division. Mol. Microbiol. 2019, 111, 46–64. [Google Scholar] [CrossRef] [PubMed]
- Melatti, C.; Pieperhoff, M.; Lemgruber, L.; Pohl, E.; Sheiner, L.; Meissner, M. A unique dynamin-related protein is essential for mitochondrial fission in Toxoplasma gondii. PLoS Pathog. 2019, 15, e1007512. [Google Scholar] [CrossRef]
- Ovciarikova, J.; Lemgruber, L.; Stilger, K.L.; Sullivan, W.J.; Sheiner, L. Mitochondrial behaviour throughout the lytic cycle of Toxoplasma gondii. Sci. Rep. 2017, 7, 42746. [Google Scholar] [CrossRef]
- Wildführ, W. Electron microscopic studies on the morphology and reproduction of Toxoplasma gondii. II. Observations on the reproduction of Toxoplasma gondii (endodyogeny). Zentralbl. Bakteriol. Orig. 1966, 201, 110–130. [Google Scholar]
- Black, M.W.; Boothroyd, J.C. Lytic cycle of Toxoplasma gondii. Microbiol. Mol. Biol. Rev. 2000, 64, 607–623. [Google Scholar] [CrossRef]
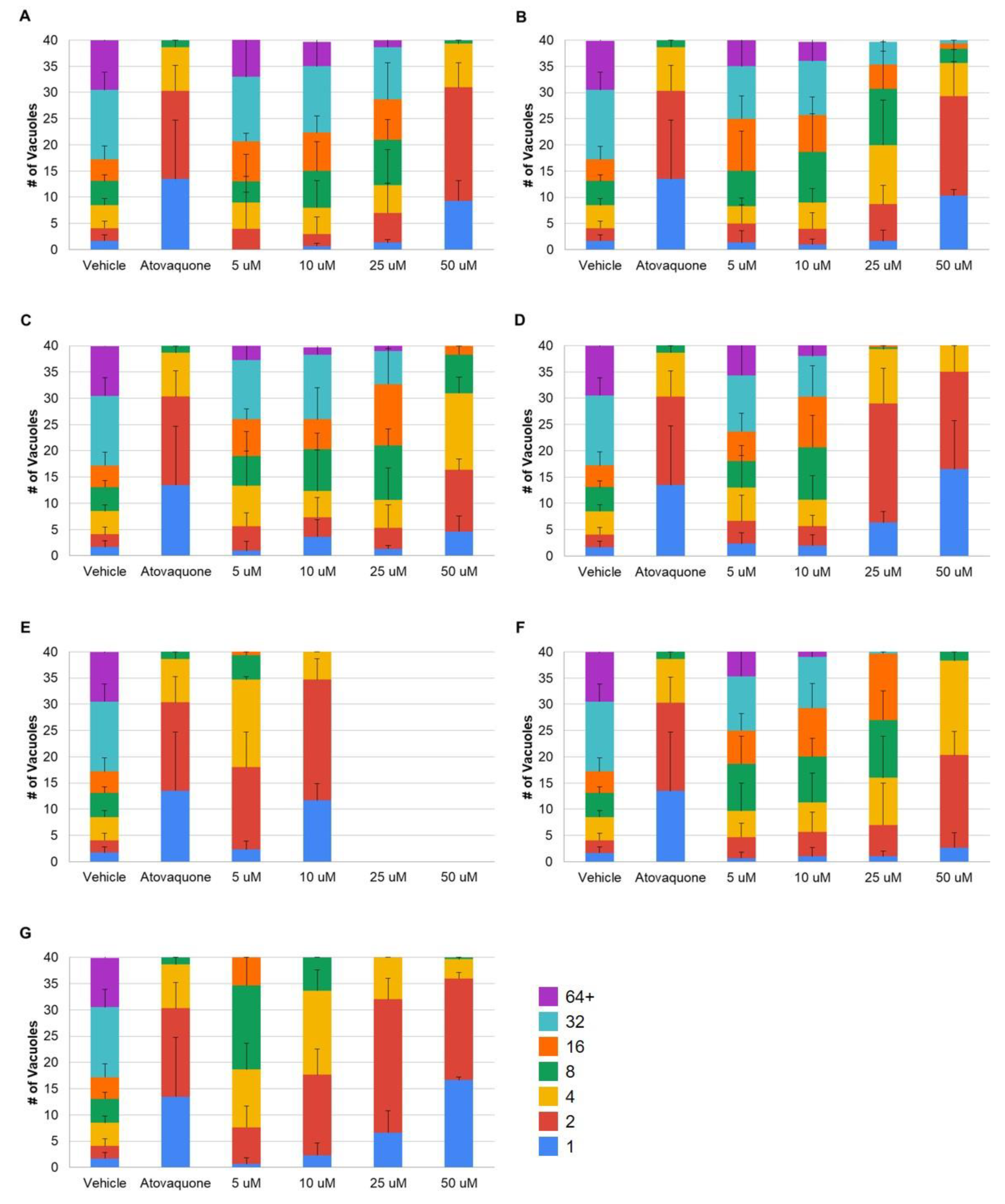
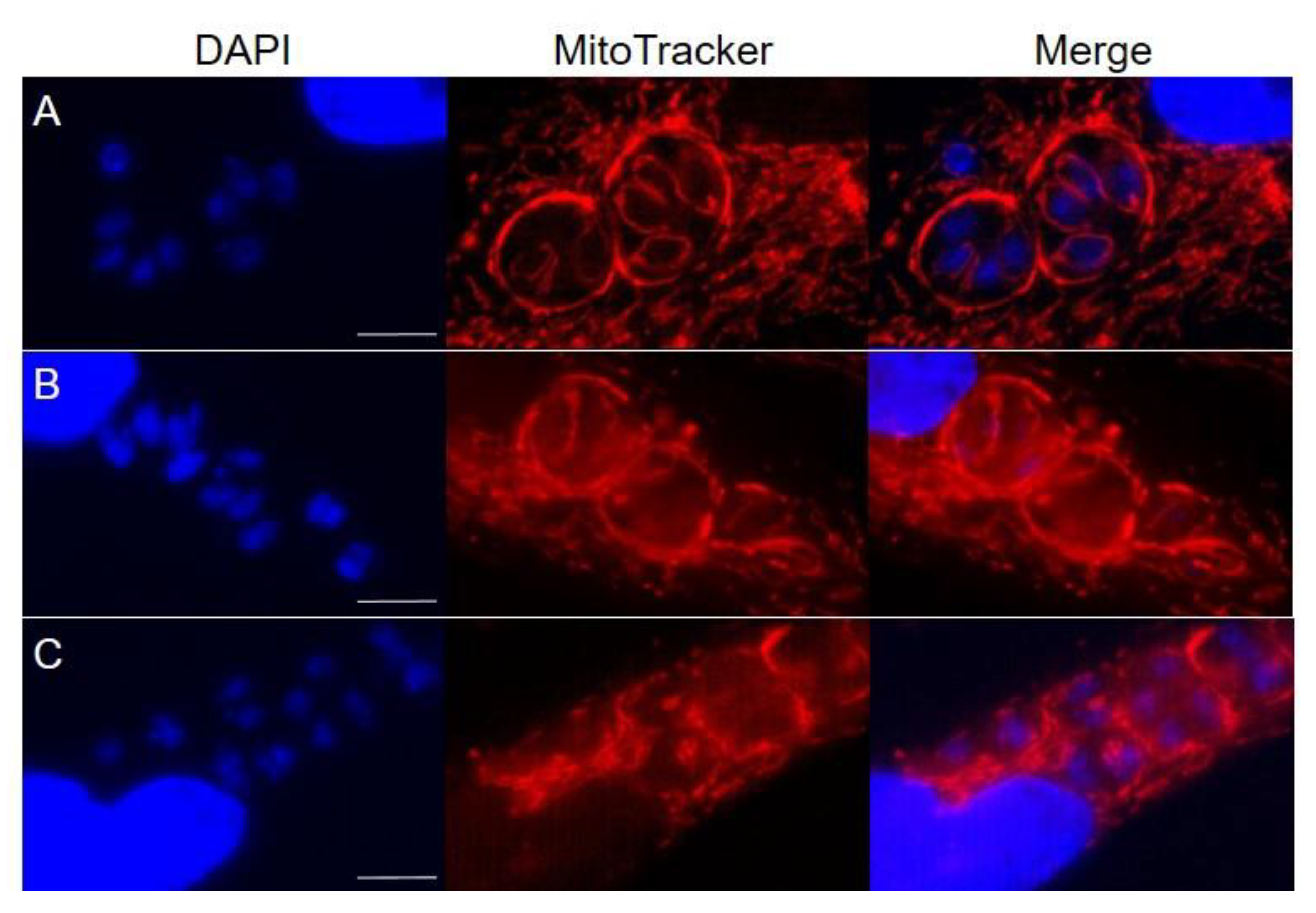
| Treatment | 5 µM | 10 µM | 25 µM | 50 µM |
|---|---|---|---|---|
| DMSO 1 | 28.84 | |||
| atovaquone 2 | 2.28 | |||
| BPZ | 26.17 | 22.57 | 15.78 | 2.28 |
| amino-BPZ | 22.95 | 24.21 | 22.28 | 16.42 |
| methyl-BPZ | 21.88 | 19.54 | 8.99 | 3.31 |
| palmitoyl-BPZ | 18.29 | 16.64 | 14.17 | 4.30 |
| ethyl-BPZ | 21.78 | 15.93 | 2.53 | 1.84 |
| isopropyl-BPZ | 3.71 | 1.98 | - 3 | - |
| hexanoyl-BPZ | 20.78 | 15.63 | 8.76 | 3.08 |
| hydroxyl-BPZ | 25.58 | 20.34 | 22.50 | 20.87 |
| dansyl-BPZ | 6.80 | 3.69 | 2.23 | 1.82 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hatton, O.; Stitzlein, L.; Dudley, R.W.; Charvat, R.A. Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii. Microorganisms 2020, 8, 1159. https://doi.org/10.3390/microorganisms8081159
Hatton O, Stitzlein L, Dudley RW, Charvat RA. Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii. Microorganisms. 2020; 8(8):1159. https://doi.org/10.3390/microorganisms8081159
Chicago/Turabian StyleHatton, Olivia, Lea Stitzlein, Richard W. Dudley, and Robert A. Charvat. 2020. "Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii" Microorganisms 8, no. 8: 1159. https://doi.org/10.3390/microorganisms8081159
APA StyleHatton, O., Stitzlein, L., Dudley, R. W., & Charvat, R. A. (2020). Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii. Microorganisms, 8(8), 1159. https://doi.org/10.3390/microorganisms8081159







