Virulence Traits of Inpatient Campylobacter jejuni Isolates, and a Transcriptomic Approach to Identify Potential Genes Maintaining Intracellular Survival
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Polymerase Chain Reaction (PCR)
2.3. Solid-Phase Extracellular Matrix Protein (ECMP) Binding Assay
2.4. INT 407 Binding and Internalization Assay
2.5. Isolation of RNA from the Cultured and INT 407 Cell Invaded C. jejuni
2.6. Sequencing C. jejuni Strain CjTD-119
2.7. Whole Transcriptome Analysis (RNA-Seq)
2.8. Bioinformatics
3. Results
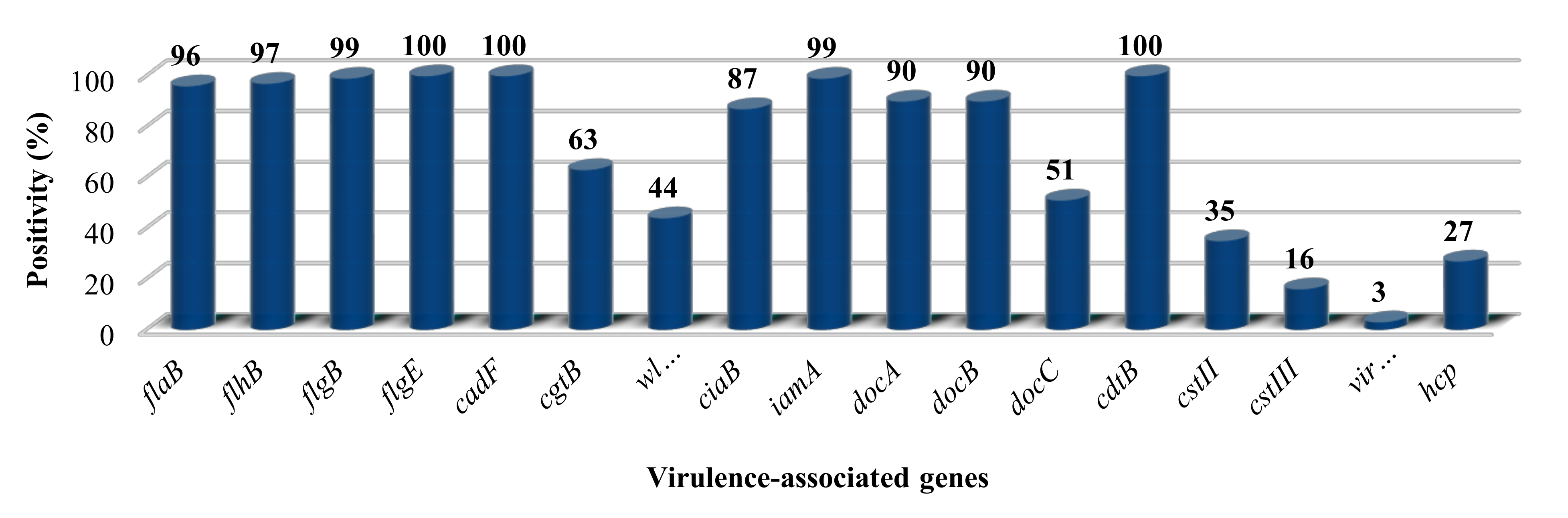
3.1. The Presence of Putative Virulence Genes
3.2. Quantitation of the ECMP Binding Assay
3.3. Adhesion and Invasion Abilities
3.4. Sequence of C. jejuni Strain CjTD-119
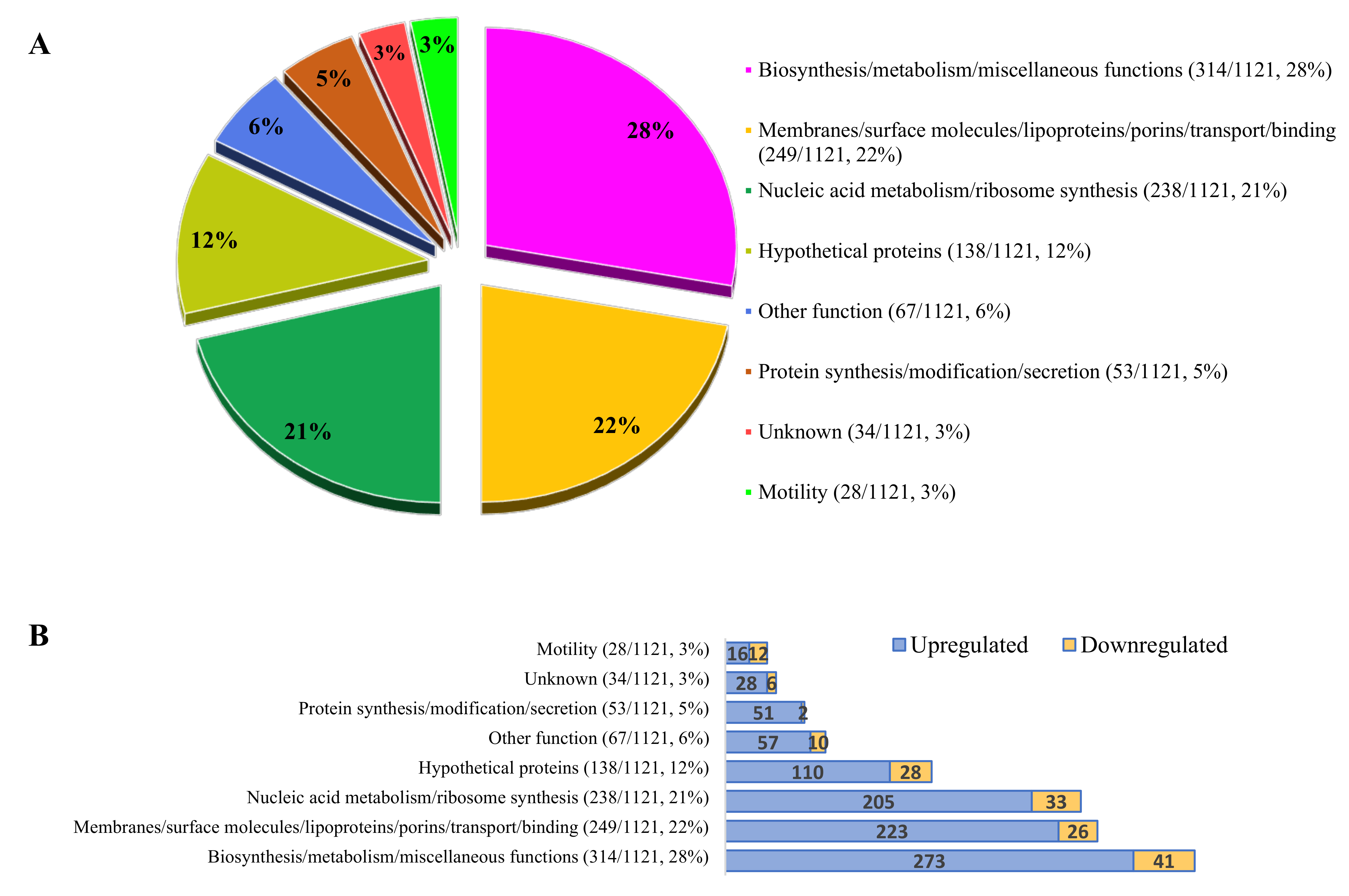
3.5. Bacterial Gene Expression during Invasion
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dasti, J.; Tareen, A.M.; Lugert, R.; Zautner, A.E.; Groß, U. Campylobacter jejuni: A brief overview on pathogenicity-associated factors and disease-mediating mechanisms. Int. J. Med Microbiol. 2010, 300, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Havelaar, A.H.; Ivarsson, S.; Löfdahl, M.; Nauta, M.J. Estimating the true incidence of campylobacteriosis and salmonellosis in the European Union, 2009. Epidemiol. Infect. 2012, 141, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. CDC Health Information for International Travel 2014: The Yellow Book; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Bereswill, S.; Kist, M. Recent developments in Campylobacter pathogenesis. Curr. Opin. Infect. Dis. 2003, 16, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Hughes, R.A.C.; Cornblath, D.R. Guillain-Barré syndrome. Lancet 2005, 366, 1653–1666. [Google Scholar] [CrossRef]
- Datta, S.; Niwa, H.; Itoh, K. Prevalence of 11 pathogenic genes of Campylobacter jejuni by PCR in strains isolated from humans, poultry meat and broiler and bovine faeces. J. Med Microbiol. 2003, 52, 345–348. [Google Scholar] [CrossRef]
- Rozynek, E.; Dzierzanowska-Fangrat, K.; Jóźwiak, P.; Popowski, J.; Korsak, D.; Dzierżanowska, D. Prevalence of potential virulence markers in Polish Campylobacter jejuni and Campylobacter coli isolates obtained from hospitalized children and from chicken carcasses. J. Med Microbiol. 2005, 54, 615–619. [Google Scholar] [CrossRef]
- Biswas, D.; Hannon, S.J.; Townsend, H.G.G.; Potter, A.; Allan, B.J. Genes coding for virulence determinants of Campylobacter jejuni in human clinical and cattle isolates from Alberta, Canada, and their potential role in colonization of poultry. Int. Microbiol. 2011, 14, 25–32. [Google Scholar]
- González-Hein, G.; Huaracán, B.; García, P.; Figueroa, G. Prevalence of virulence genes in strains of Campylobacter jejuni isolated from human, bovine and broiler. Braz. J. Microbiol. 2014, 44, 1223–1229. [Google Scholar] [CrossRef]
- Yao, R.; Burr, D.H.; Doig, P.; Trust, T.J.; Niu, H.; Guerry, P. Isolation of motile and non-motile insertional mutants of Campylobacter jejuni: the role of motility in adherence and invasion of eukaryotic cells. Mol. Microbiol. 1994, 14, 883–893. [Google Scholar] [CrossRef]
- Gilbreath, J.J.; Cody, W.L.; Merrell, D.S.; Hendrixson, D.R. Change Is Good: Variations in Common Biological Mechanisms in the Epsilonproteobacterial Genera Campylobacter and Helicobacter. Microbiol. Mol. Boil. Rev. 2011, 75, 84–132. [Google Scholar] [CrossRef]
- Ferrero, R.L.; Lee, A. Motility of Campylobacter jejuni in a viscous environment: Comparison with conventional rod-shaped bacteria. Microbiology 1988, 134, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Frirdich, E.; Vermeulen, J.; Biboy, J.; Soares, F.; Taveirne, M.E.; Johnson, J.; DiRita, V.J.; Girardin, S.E.; Vollmer, W.; Gaynor, E.C. Peptidoglycan LD-carboxypeptidase Pgp2 influences Campylobacter jejuni helical cell shape and pathogenic properties and provides the substrate for the DL-carboxypeptidase Pgp1. J. Boil. Chem. 2014, 289, 8007–8018. [Google Scholar] [CrossRef] [PubMed]
- Esson, D.; Mather, A.E.; Scanlan, E.; Gupta, S.; De Vries, S.P.W.; Bailey, D.; Harris, S.R.; McKinley, T.J.; Méric, G.; Berry, S.K.; et al. Genomic variations leading to alterations in cell morphology of Campylobacter spp. Sci. Rep. 2016, 6, 38303. [Google Scholar] [CrossRef]
- Monteville, M.R.; Yoon, J.E.; Konkel, M.E. Maximal adherence and invasion of INT 407 cells by Campylobacter jejuni requires the CadF outer-membrane protein and microfilament reorganization. Microbiology 2003, 149, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Burucoa, C.; Grignon, B.; Baqar, S.; Huang, X.-Z.; Kopecko, D.J.; Bourgeois, A.L.; Fauchere, J.-L.; Blaser, M.J. Mutation in the peb1A Locus of Campylobacter jejuni Reduces Interactions with Epithelial Cells and Intestinal Colonization of Mice. Infect. Immun. 1998, 66, 938–943. [Google Scholar] [CrossRef]
- Asakura, H.; Yamasaki, M.; Yamamoto, S.; Igimi, S. Deletion ofpeb4gene impairs cell adhesion and biofilm formation in Campylobacter jejuni. FEMS Microbiol. Lett. 2007, 275, 278–285. [Google Scholar] [CrossRef]
- Ashgar, S.S.A.; Oldfield, N.; Wooldridge, K.; Jones, M.; Irving, G.J.; Turner, D.P.; Ala’Aldeen, D.A.A. CapA, an Autotransporter Protein of Campylobacter jejuni, Mediates Association with Human Epithelial Cells and Colonization of the Chicken Gut. J. Bacteriol. 2006, 189, 1856–1865. [Google Scholar] [CrossRef]
- Eucker, T.P.; Konkel, M.E. The cooperative action of bacterial fibronectin-binding proteins and secreted proteins promote maximal Campylobacter jejuni invasion of host cells by stimulating membrane ruffling. Cell. Microbiol. 2011, 14, 226–238. [Google Scholar] [CrossRef]
- Kawai, F.; Paek, S.; Choi, K.-J.; Prouty, M.; Kanipes, M.I.; Guerry, P.; Yeo, H.-J. Crystal structure of JlpA, a surface-exposed lipoprotein adhesin of Campylobacter jejuni. J. Struct. Boil. 2012, 177, 583–588. [Google Scholar] [CrossRef]
- Karlyshev, A.; Ketley, J.; Wren, B. The glycome. FEMS Microbiol. Rev. 2005, 29, 377–390. [Google Scholar] [CrossRef]
- Szymanski, C.M.; Burr, D.H.; Guerry, P. Campylobacter Protein Glycosylation Affects Host Cell Interactions. Infect. Immun. 2002, 70, 2242–2244. [Google Scholar] [CrossRef] [PubMed]
- Hendrixson, D.R.; DiRita, V.J. Identification of Campylobacter jejuni genes involved in commensal colonization of the chick gastrointestinal tract. Mol. Microbiol. 2004, 52, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Karlyshev, A.; Everest, P.; Linton, D.; Cawthraw, S.; Newell, D.G.; Wren, B.W. The Campylobacter jejuni general glycosylation system is important for attachment to human epithelial cells and in the colonization of chicks. Microbiology 2004, 150, 1957–1964. [Google Scholar] [CrossRef] [PubMed]
- Rubinchik, S.; Seddon, A.; Karlyshev, A. Molecular mechanisms and biological role of Campylobacter jejuni attachment to host cells. Eur. J. Microbiol. Immunol. 2012, 2, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.; Karwaski, M.-F.; Bernatchez, S.; Young, N.M.; Taboada, E.N.; Michniewicz, J.; Cunningham, A.-M.; Wakarchuk, W. The Genetic Bases for the Variation in the Lipo-oligosaccharide of the Mucosal Pathogen, Campylobacter jejuni. J. Boil. Chem. 2001, 277, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Guerry, P.; Ewing, C.P.; Hickey, T.E.; Prendergast, M.M.; Moran, A.P. Sialylation of Lipooligosaccharide Cores Affects Immunogenicity and Serum Resistance of Campylobacter jejuni. Infect. Immun. 2000, 68, 6656–6662. [Google Scholar] [CrossRef]
- Linton, D.; Karlyshev, A.; Hitchen, P.G.; Morris, H.R.; Dell, A.; Gregson, N.A.; Wren, B.W. Multiple N-acetyl neuraminic acid synthetase (neuB) genes in Campylobacter jejuni: identification and characterization of the gene involved in sialylation of lipo-oligosaccharide. Mol. Microbiol. 2000, 35, 1120–1134. [Google Scholar] [CrossRef]
- Fauchere, J.L.; Rosenau, A.; Veron, M.; Moyen, E.N.; Richard, S.; Pfister, A. Association with HeLa cells of Campylobacter jejuni and Campylobacter coli isolated from human feces. Infect. Immun. 1986, 54, 283–287. [Google Scholar] [CrossRef]
- Everest, P.H.; Goossens, H.; Butzler, J.-P.; Lloyd, D.; Knutton, S.; Ketley, J.M.; Williams, P.H. Differentiated Caco-2 cells as a model for enteric invasion by Campylobacter jejuni and C. coli. J. Med Microbiol. 1992, 37, 319–325. [Google Scholar] [CrossRef]
- Law, B.F.; Adriance, S.M.; Joens, L.A. Comparison ofIn VitroVirulence Factors of Campylobacter jejuni to In Vivo Lesion Production. Foodborne Pathog. Dis. 2009, 6, 377–385. [Google Scholar] [CrossRef]
- Van Spreeuwel, J.P.; Duursma, G.C.; Meijer, C.J.; Bax, R.; Rosekrans, P.C.; Lindeman, J. Campylobacter colitis: histological immunohistochemical and ultrastructural findings. Gut 1985, 26, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Cróinín, T.Ó.; Backert, S. Host Epithelial Cell Invasion by Campylobacter jejuni: Trigger or Zipper Mechanism? Front. Microbiol. 2012, 2, 25. [Google Scholar]
- Monteville, M.R.; E Konkel, M. Fibronectin-Facilitated Invasion of T84 Eukaryotic Cells by Campylobacter jejuni Occurs Preferentially at the Basolateral Cell Surface. Infect. Immun. 2002, 70, 6665–6671. [Google Scholar] [CrossRef] [PubMed]
- Poly, F.; Threadgill, D.; Stintzi, A. Identification of Campylobacter jejuni ATCC 43431-Specific Genes by Whole Microbial Genome Comparisons. J. Bacteriol. 2004, 186, 4781–4795. [Google Scholar] [CrossRef] [PubMed]
- Friis, L.; Pin, C.; Pearson, B.; Wells, J.M. In vitro cell culture methods for investigating Campylobacter invasion mechanisms. J. Microbiol. Methods 2005, 61, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Christensen, J.E.; Pacheco, S.A.; Konkel, M.E. Identification of a Campylobacter jejuni-secreted protein required for maximal invasion of host cells. Mol. Microbiol. 2009, 73, 650–662. [Google Scholar] [CrossRef]
- Negretti, N.M.; Clair, G.C.; Talukdar, P.K.; Gourley, C.R.; Huynh, S.; Adkins, J.N.; Parker, C.T.; Corneau, C.M.; Konkel, M.E. Campylobacter jejuni Demonstrates Conserved Proteomic and Transcriptomic Responses When Co-cultured With Human INT 407 and Caco-2 Epithelial Cells. Front. Microbiol. 2019, 10, 755. [Google Scholar] [CrossRef]
- Konkel, M.E.; Klena, J.D.; Rivera-Amill, V.; Monteville, M.R.; Biswas, D.; Raphael, B.; Mickelson, J. Secretion of Virulence Proteins from Campylobacter jejuni Is Dependent on a Functional Flagellar Export Apparatus. J. Bacteriol. 2004, 186, 3296–3303. [Google Scholar] [CrossRef]
- Parkhill, J.; Wren, B.W.; Mungall, K.; Ketley, J.M.; Churcher, C.; Basham, D.; Chillingworth, T.; Davies, R.M.; Feltwell, T.; Holroyd, S.; et al. The genome sequence of the food-borne pathogen Campylobacter jejuni reveals hypervariable sequences. Nature 2000, 403, 665–668. [Google Scholar] [CrossRef]
- Poly, F.; Guerry, P. Pathogenesis of Campylobacter. Curr. Opin. Gastroenterol. 2008, 24.1, 27–31. [Google Scholar] [CrossRef]
- Carrillo, C.D.; Taboada, E.N.; Nash, J.; Lanthier, P.; Kelly, J.; Lau, P.C.; Verhulp, R.; Mykytczuk, O.; Sy, J.; Findlay, W.A.; et al. Genome-wide Expression Analyses of Campylobacter jejuni NCTC11168 Reveals Coordinate Regulation of Motility and Virulence byflhA. J. Boil. Chem. 2004, 279, 20327–20338. [Google Scholar] [CrossRef] [PubMed]
- Samuelson, D.; Eucker, T.P.; Bell, J.A.; Dybas, L.; Mansfield, L.S.; Konkel, M.E. The Campylobacter jejuni CiaD effector protein activates MAP kinase signaling pathways and is required for the development of disease. Cell Commun. Signal. 2013, 11, 79. [Google Scholar] [CrossRef] [PubMed]
- Man, S.M. The clinical importance of emerging Campylobacter species. Nat. Rev. Gastroenterol. Hepatol. 2011, 8, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Scuron, M.D.; Boesze-Battaglia, K.; Dlakić, M.; Shenker, B.J. The cytolethal distending toxin contributes to microbial virulence and disease pathogenesis by acting as a tri-perditious toxin. Front. Cell. Infect. Microbiol. 2016, 6, 168. [Google Scholar] [CrossRef] [PubMed]
- Hickey, T.E.; McVeigh, A.L.; Scott, D.A.; Michietutti, R.E.; Bixby, A.; Carroll, S.A.; Bourgeois, A.L.; Guerry, P. Campylobacter jejuni cytolethal distending toxin mediates release of interleukin-8 from intestinal epithelial cells. Infect. Immun. 2000, 68, 6535–6541. [Google Scholar] [CrossRef]
- Lara-Tejero, M. A Bacterial Toxin That Controls Cell Cycle Progression as a Deoxyribonuclease I-Like Protein. Science 2000, 290, 354–357. [Google Scholar] [CrossRef]
- Bang, D.D.; Scheutz, F.; Ahrens, P.; Pedersen, K.; Blom, J.; Madsen, M. Prevalence of cytolethal distending toxin (cdt) genes and CDT production in Campylobacter spp. isolated from Danish broilers. J. Med Microbiol. 2001, 50, 1087–1094. [Google Scholar] [CrossRef]
- Lee, R.B.; Hassane, D.; Cottle, D.L.; Pickett, C.L. Interactions of Campylobacter jejuni Cytolethal Distending Toxin Subunits CdtA and CdtC with HeLa Cells. Infect. Immun. 2003, 71, 4883–4890. [Google Scholar] [CrossRef]
- Hickey, T.E.; Majam, G.; Guerry, P. Intracellular Survival of Campylobacter jejuni in Human Monocytic Cells and Induction of Apoptotic Death by Cytholethal Distending Toxin. Infect. Immun. 2005, 73, 5194–5197. [Google Scholar] [CrossRef]
- Oelschlaeger, T.A.; Guerry, P.; Kopecko, D.J. Unusual microtubule-dependent endocytosis mechanisms triggered by Campylobacter jejuni and Citrobacter freundii. Proc. Natl. Acad. Sci. USA 1993, 90, 6884–6888. [Google Scholar] [CrossRef]
- Watson, R.O.; Galán, J.E. Campylobacter jejuni Survives within Epithelial Cells by Avoiding Delivery to Lysosomes. PLOS Pathog. 2008, 4, e14.
- Kelly, D.J. Complexity and Versatility in the Physiology and Metabolism of Campylobacter jejuni. In Campylo bacter; American Society for Microbiology: Washington DC, USA, 2008; pp. 41–61. [Google Scholar]
- Kern, M.; Simon, J. Electron transport chains and bioenergetics of respiratory nitrogen metabolism in Wolinella succinogenes and other Epsilonproteobacteria. Biochim Biophys Acta. 2009, 1787, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Burnham, P.M.; Hendrixson, D.R. Campylobacter jejuni: collective components promoting a successful enteric lifestyle. Nat. Rev. Genet. 2018, 16, 551–565. [Google Scholar] [CrossRef] [PubMed]
- Young, K.T.; Davis, L.M.; DiRita, V.J. Campylobacter jejuni: molecular biology and pathogenesis. Nat. Rev. Genet. 2007, 5, 665–679. [Google Scholar] [CrossRef] [PubMed]
- Steinbrueckner, B.; Haerter, G.; Pelz, K.; Kist, M. Routine identification of Campylobacter jejuni and Campylobacter coli from human stool samples. FEMS Microbiol. Lett. 1999, 179, 227–232. [Google Scholar] [CrossRef]
- Kuusela, P.; Moran, A.P.; Vartio, T.; Kosunen, T.U. Interaction of Campylobacter jejuni with extracellular matrix components. Biochim. et Biophys. Acta (BBA) - Gen. Subj. 1989, 993, 297–300. [Google Scholar] [CrossRef]
- Backert, S.; Hofreuter, D. Molecular methods to investigate adhesion, transmigration, invasion and intracellular survival of the foodborne pathogen Campylobacter jejuni. J. Microbiol. Methods 2013, 95, 8–23. [Google Scholar] [CrossRef]
- Liao, Y.-C.; Lin, H.-H.; Sabharwal, A.; Haase, E.; Scannapieco, F.A. MyPro: A seamless pipeline for automated prokaryotic genome assembly and annotation. J. Microbiol. Methods 2015, 113, 72–74. [Google Scholar] [CrossRef]
- Du Plessis, D.J.; Nouwen, N.; Driessen, A.J. The Sec translocase. Biochim. et Biophys. Acta (BBA) - Biomembr. 2011, 1808, 851–865. [Google Scholar] [CrossRef]
- Hands, S.L.; Holland, L.E.; Vankemmelbeke, M.; Fraser, L.; Macdonald, C.J.; Moore, G.R.; James, R.; Penfold, C. Interactions of TolB with the Translocation Domain of Colicin E9 Require an Extended TolB Box. J. Bacteriol. 2005, 187, 6733–6741. [Google Scholar] [CrossRef][Green Version]
- Confer, A.W.; Ayalew, S. The OmpA family of proteins: Roles in bacterial pathogenesis and immunity. Vet. Microbiol. 2013, 163, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Malik-Kale, P.; Parker, C.; Konkel, M.E. Culture of Campylobacter jejuni with Sodium Deoxycholate Induces Virulence Gene Expression. J. Bacteriol. 2008, 190, 2286–2297. [Google Scholar] [CrossRef] [PubMed]
- Oakland, M.; Jeon, B.; Sahin, O.; Shen, Z.; Zhang, Q. Functional Characterization of a Lipoprotein-Encoding Operon in Campylobacter jejuni. PLoS ONE 2011, 6, e20084. [Google Scholar] [CrossRef][Green Version]
- Tareen, A.M.; Lüder, C.G.K.; Zautner, A.E.; Groß, U.; Heimesaat, M.M.; Bereswill, S.; Lugert, R. The Campylobacter jejuni Cj0268c Protein Is Required for Adhesion and Invasion In Vitro. PLoS ONE 2013, 8, e81069. [Google Scholar] [CrossRef] [PubMed]
- Heimesaat, M.M.; Lugert, R.; Fischer, A.; Alutis, M.; Kühl, A.A.; Zautner, A.E.; Tareen, A.M.; Göbel, U.B.; Bereswill, S. Impact of Campylobacter jejuni cj0268c Knockout Mutation on Intestinal Colonization, Translocation, and Induction of Immunopathology in Gnotobiotic IL-10 Deficient Mice. PLoS ONE 2014, 9, e90148. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, R.C.; Neal-McKinney, J.M.; Dhillon, A.S.; Miller, W.G.; Konkel, M.E. Examination of Campylobacter jejuni Putative Adhesins Leads to the Identification of a New Protein, Designated FlpA, Required for Chicken Colonization. Infect. Immun. 2009, 77, 2399–2407. [Google Scholar] [CrossRef]
- Sałamaszyńska-Guz, A.; Klimuszko, D. Functional Analysis of the Campylobacter jejuni cj0183 and cj0588 Genes. Curr. Microbiol. 2008, 56, 592–596. [Google Scholar] [CrossRef]
- Pei, Z.H.; Ellison, R.T.; Blaser, M.J. Identification, purification, and characterization of major antigenic proteins of Campylobacter jejuni. J. Boil. Chem. 1991, 266, 16363–16369. [Google Scholar]
- Doble, A.C.; Bulmer, D.M.; Kharraz, L.; Karavolos, M.H.; Khan, C.A. The function of the bacterial cytoskeleton in Salmonella pathogenesis. Virulence 2012, 3, 446–449. [Google Scholar] [CrossRef][Green Version]
- Karlyshev, A.; Linton, D.; Gregson, N.A.; Lastovica, A.J.; Wren, B.W. Genetic and biochemical evidence of a Campylobacter jejuni capsular polysaccharide that accounts for Penner serotype specificity. Mol. Microbiol. 2002, 35, 529–541. [Google Scholar] [CrossRef]
- Hitchcock, A.; Hall, S.; Myers, J.D.; Mulholland, F.; Jones, M.; Kelly, D.J. Roles of the twin-arginine translocase and associated chaperones in the biogenesis of the electron transport chains of the human pathogen Campylobacter jejuni. Microbiology 2010, 156, 2994–3010. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Javed, M.A.; Grant, A.J.; Bagnall, M.C.; Maskell, D.J.; Newell, D.G.; Manning, G. Transposon mutagenesis in a hyper-invasive clinical isolate of Campylobacter jejuni reveals a number of genes with potential roles in invasion. Microbiology 2010, 156, 1134–1143. [Google Scholar] [CrossRef]
- Cohn, M.T.; Ingmer, H.; Mulholland, F.; Jørgensen, K.; Wells, J.M.; Brøndsted, L. Contribution of Conserved ATP-Dependent Proteases of Campylobacter jejuni to Stress Tolerance and Virulence. Appl. Environ. Microbiol. 2007, 73, 7803–7813. [Google Scholar] [CrossRef]
- Rathbun, K.; E Hall, J.; Thompson, S.A. Cj0596 is a periplasmic peptidyl prolyl cis-trans isomerase involved in Campylobacter jejuni motility, invasion, and colonization. BMC Microbiol. 2009, 9, 160. [Google Scholar] [CrossRef] [PubMed]
- Elliott, K.; Zhulin, I.B.; Stuckey, J.A.; DiRita, V.J. Conserved Residues in the HAMP Domain Define a New Family of Proposed Bipartite Energy Taxis Receptors. J. Bacteriol. 2008, 191, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Ziprin, R.L.; Young, C.; Byrd, J.A.; Stanker, L.H.; Hume, M.E.; Gray, S.A.; Kim, B.J.; E Konkel, M. Role of Campylobacter jejuni potential virulence genes in cecal colonization. Avian Dis. 2001, 45, 549. [Google Scholar] [CrossRef] [PubMed]
- Bingham-Ramos, L.K.; Hendrixson, D.R. Characterization of Two Putative Cytochrome c Peroxidases of Campylobacter jejuni Involved in Promoting Commensal Colonization of Poultry. Infect. Immun. 2007, 76, 1105–1114. [Google Scholar] [CrossRef]
- MacKichan, J.; Gaynor, E.C.; Chang, C.; Cawthraw, S.; Newell, D.G.; Miller, J.F.; Falkow, S. The Campylobacter jejuni dccRS two-component system is required for optimal in vivo colonization but is dispensable for in vitro growth. Mol. Microbiol. 2004, 54, 1269–1286. [Google Scholar] [CrossRef]
- Xie, Y.; He, Y.; Irwin, P.L.; Jin, T.; Shi, X. Antibacterial Activity and Mechanism of Action of Zinc Oxide Nanoparticles against Campylobacter jejuni. Appl. Environ. Microbiol. 2011, 77, 2325–2331. [Google Scholar] [CrossRef]
- I Smith, S.; Olukoya, D.K.; Fox, A.J.; Coker, A.O. Flagellin gene polymorphism analysis of Campylobacter compared with antigen serotyping. Zeitschrift für Naturforschung C 1999, 54, 946–951. [Google Scholar] [CrossRef]
- Müller, J.; Schulze, F.; Müller, W.; Hänel, I. PCR detection of virulence-associated genes in Campylobacter jejuni strains with differential ability to invade Caco-2 cells and to colonize the chick gut. Veter- Microbiol. 2006, 113, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.G.; Rogers, A.B.; Whary, M.T.; Ge, Z.; Taylor, N.S.; Xu, S.; Horwitz, B.; Erdman, S.E. Gastroenteritis in NF-kappaB-deficient mice is produced with wild-type Camplyobacter jejuni but not with C. jejuni lacking cytolethal distending toxin despite persistent colonization with both strains. Infect. Immun. 2004, 72, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Prasad, K.N.; Sinha, S.; Husain, N. Differences in virulence attributes between cytolethal distending toxin positive and negative Campylobacter jejuni strains. J. Med Microbiol. 2008, 57, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Findik, A.; Iça, T.; Onuk, E.E.; Percin, D.; Kevenk, T.O.; Çiftci, A. Molecular typing and cdt genes prevalence of Campylobacter jejuni isolates from various sources. Trop. Anim. Heal. Prod. 2010, 43, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Meng, J.; Zhao, S.; Singh, R.; Song, W. Campylobacter-Induced Interleukin-8 Secretion in Polarized Human Intestinal Epithelial Cells Requires Campylobacter-Secreted Cytolethal Distending Toxin- and Toll-Like Receptor-Mediated Activation of NF-κB. Infect. Immun. 2008, 76, 4498–4508. [Google Scholar] [CrossRef]
- Mortensen, N.P.; Schiellerup, P.; Boisen, N.; Klein, B.M.; Locht, H.; AbuOun, M.; Newell, D.; Krogfelt, K.A. The role of Campylobacter jejuni cytolethal distending toxin in gastroenteritis: toxin detection, antibody production, and clinical outcome. APMIS 2011, 119, 626–634. [Google Scholar] [CrossRef]
- Van Deun, K.; Haesebrouck, F.; Heyndrickx, M.; Favoreel, H.; Dewulf, J.; Ceelen, L.; Dumez, L.; Messens, W.; Leleu, S.; Van Immerseel, F.; et al. Virulence properties of Campylobacter jejuni isolates of poultry and human origin. J. Med Microbiol. 2007, 56, 1284–1289. [Google Scholar] [CrossRef]
- Sanad, Y.; Kassem, I.I.; Liu, Z.; Lin, J.; Lejeune, J.; Rajashekara, G. Occurrence of the invasion associated marker (iam) in Campylobacter jejuni isolated from cattle. BMC Res. Notes 2011, 4, 570. [Google Scholar] [CrossRef]
- Harrison, J.W.; Dung, T.T.N.; Siddiqui, F.; Korbrisate, S.; Bukhari, H.; Tra, M.P.V.; Hoang, N.V.M.; Carrique-Mas, J.; Bryant, J.; Campbell, J.I.; et al. Identification of Possible Virulence Marker from Campylobacter jejuni Isolates. Emerg. Infect. Dis. 2014, 20, 1026–1029. [Google Scholar] [CrossRef]
- Corcionivoschi, N.; Gundogdu, O.; Moran, L.; Kelly, C.; Scates, P.; Stef, L.; Cean, A.; Wren, B.W.; Dorrell, N.; Madden, R.H. Virulence characteristics of hcp (+) Campylobacter jejuni and Campylobacter coli isolates from retail chicken. Gut Pathog. 2015, 7, 20. [Google Scholar] [CrossRef]
- Al-Mahmeed, A.; Senok, A.C.; Ismaeel, A.Y.; Bindayna, K.M.; Tabbara, K.; Botta, G.A. Clinical relevance of virulence genes in Campylobacter jejuni isolates in Bahrain. J. Med Microbiol. 2006, 55, 839–843. [Google Scholar] [CrossRef] [PubMed]
- Pankov, R. Fibronectin at a glance. J. Cell Sci. 2002, 115, 3861–3863. [Google Scholar] [CrossRef] [PubMed]
- Thibodeau, A.; Fravalo, P.; Taboada, E.N.; Lewandowski, S.L.-; Guévremont, E.; Quessy, S.; Letellier, A. Extensive characterization of Campylobacter jejuni chicken isolates to uncover genes involved in the ability to compete for gut colonization. BMC Microbiol. 2015, 15, 97. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bronnec, V.; Turonova, H.; Bouju, A.; Cruveiller, S.; Rodrigues, R.; Demnerova, K.; Tresse, O.; Haddad, N.; Zagorec, M. Adhesion, Biofilm Formation, and Genomic Features of Campylobacter jejuni Bf, an Atypical Strain Able to Grow under Aerobic Conditions. Front. Microbiol. 2016, 7, 278. [Google Scholar] [CrossRef]
- Louwen, R.; Heikema, A.; Van Belkum, A.; Ott, A.; Gilbert, M.; Ang, W.; Endtz, H.P.; Bergman, M.P.; Nieuwenhuis, E.E. The Sialylated Lipooligosaccharide Outer Core in Campylobacter jejuni Is an Important Determinant for Epithelial Cell Invasion. Infect. Immun. 2008, 76, 4431–4438. [Google Scholar] [CrossRef]
- Habib, I.; Louwen, R.; Uyttendaele, M.; Houf, K.; Vandenberg, O.; Nieuwenhuis, E.E.; Miller, W.G.; Van Belkum, A.; De Zutter, L. Correlation between Genotypic Diversity, Lipooligosaccharide Gene Locus Class Variation, and Caco-2 Cell Invasion Potential of Campylobacter jejuni Isolates from Chicken Meat and Humans: Contribution to Virulotyping. Appl. Environ. Microbiol. 2009, 75, 4277–4288. [Google Scholar] [CrossRef]
- Mortensen, N.P.; Kuijf, M.; Ang, C.W.; Schiellerup, P.; Krogfelt, K.A.; Jacobs, B.C.; Van Belkum, A.; Endtz, H.P.; Bergman, M.P. Sialylation of Campylobacter jejuni lipo-oligosaccharides is associated with severe gastro-enteritis and reactive arthritis. Microbes Infect. 2009, 11, 988–994. [Google Scholar] [CrossRef]
- Parker, C.; Horn, S.T.; Gilbert, M.; Miller, W.G.; Woodward, D.L.; Mandrell, R.E. Comparison of Campylobacter jejuni Lipooligosaccharide Biosynthesis Loci from a Variety of Sources. J. Clin. Microbiol. 2005, 43, 2771–2781. [Google Scholar] [CrossRef]
- Ellström, P.; Feodoroff, B.; Hanninen, M.-L.; Rautelin, H.; Allerberger, F. Lipooligosaccharide locus class of Campylobacter jejuni: sialylation is not needed for invasive infection. Clin. Microbiol. Infect. 2013, 20, 524–529. [Google Scholar] [CrossRef]
- Zheng, J.; Meng, J.; Zhao, S.; Singh, R.; Song, W. Adherence to and Invasion of Human Intestinal Epithelial Cells by Campylobacter jejuni and Campylobacter coli Isolates from Retail Meat Products. J. Food Prot. 2006, 69, 768–774. [Google Scholar] [CrossRef]
- Hu, L.; Kopecko, D.J. Interactions of Campylobacter with eukaryotic cells: gut luminal colonization and mucosal invasion mechanisms. Campylobacter 2000, 2, 191–215. [Google Scholar]
- Konkel, M.E.; Joens, L.A.; Mixter, P.F. Molecular characterization of Campylobacter jejuni virulence determinants. Campylobacter 2000, 2, 217–240. [Google Scholar]
- E Konkel, M.; A Joens, L. Adhesion to and invasion of HEp-2 cells by Campylobacter spp. Infect. Immun. 1989, 57, 2984–2990. [Google Scholar] [CrossRef]
- Newell, D.G.; McBride, H.; Saunders, F.; Dehele, Y.; Pearson, A.D. The virulence of clinical and environmental isolates of Campylobacter jejuni. J. Hyg. 1985, 94, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.C.T.; Ruiz-Palacios, G.M.; Ramos-Cervantes, P.; Cervantes, L.-E.; Jiang, X.; Pickering, L.K. Molecular Characterization of Invasive and Noninvasive Campylobacter jejuni and Campylobacter coli Isolates. J. Clin. Microbiol. 2001, 39, 1353–1359. [Google Scholar] [CrossRef]
- Gundogdu, O.; Da Silva, D.T.; Mohammad, B.; Elmi, A.; Mills, D.C.; Wren, B.W.; Dorrell, N. The Campylobacter jejuni MarR-like transcriptional regulators RrpA and RrpB both influence bacterial responses to oxidative and aerobic stresses. Front. Microbiol. 2015, 6, 724. [Google Scholar] [CrossRef]
- Buelow, D.R.; Christensen, J.E.; Neal-McKinney, J.M.; Konkel, M.E. Campylobacter jejuni survival within human epithelial cells is enhanced by the secreted protein CiaI. Mol. Microbiol. 2011, 80, 1296–1312. [Google Scholar] [CrossRef] [PubMed]
- Day, W.A.; Sajecki, J.L.; Pitts, T.M.; Joens, L.A. Role of Catalase in Campylobacter jejuni Intracellular Survival. Infect. Immun. 2000, 68, 6337–6345. [Google Scholar] [CrossRef]
- Frirdich, E.; Biboy, J.; Adams, C.; Lee, J.; Ellermeier, J.; Gielda, L.D.; DiRita, V.J.; Girardin, S.E.; Vollmer, W.; Gaynor, E.C. Peptidoglycan-modifying enzyme Pgp1 is required for helical cell shape and pathogenicity traits in Campylobacter jejuni. PLoS Pathog. 2012, 8, e1002602. [Google Scholar] [CrossRef]
- Vijayan, S.; Mallick, S.; Dutta, M.; Narayani, M.; Ghosh, A.S. PBP Deletion Mutants of Escherichia coli Exhibit Irregular Distribution of MreB at the Deformed Zones. Curr. Microbiol. 2013, 68, 174–179. [Google Scholar] [CrossRef]
- Guerry, P.; Poly, F.; Riddle, M.; Maue, A.C.; Chen, Y.-H.; Monteiro, M.A. Campylobacter Polysaccharide Capsules: Virulence and Vaccines. Front. Microbiol. 2012, 2, 7. [Google Scholar] [CrossRef]
- Karlyshev, A.; Moran, A.P.; Wren, B.W. Campylobacter jejuni Capsular Polysaccharide. In Campylobacter; American Society for Microbiology: Washington, DC, USA, 2008; pp. 505–521. [Google Scholar]
- Rubinchik, S.; Seddon, A.M.; Karlyshev, A. A negative effect of Campylobacter capsule on bacterial interaction with an analogue of a host cell receptor. BMC Microbiol. 2014, 14, 141. [Google Scholar] [CrossRef][Green Version]
- Bacon, D.J.; Szymanski, C.M.; Burr, D.H.; Silver, R.P.; Alm, R.A.; Guerry, P. A phase-variable capsule is involved in virulence of Campylobacter jejuni 81-176. Mol. Microbiol. 2001, 40, 769–777. [Google Scholar] [CrossRef]
- Bachtiar, B.M.; Coloe, P.; Fry, B.N. Knockout mutagenesis of the kpsE gene of Campylobacter jejuni 81116 and its involvement in bacterium–host interactions. FEMS Immunol. Med Microbiol. 2007, 49, 149–154. [Google Scholar] [CrossRef]
- Corcionivoschi, N.; Clyne, M.; Lyons, A.; Elmi, A.; Gundogdu, O.; Wren, B.W.; Dorrell, N.; Karlyshev, A.; Bourke, B. Campylobacter jejuni Cocultured with Epithelial Cells Reduces Surface Capsular Polysaccharide Expression. Infect. Immun. 2009, 77, 1959–1967. [Google Scholar] [CrossRef]
- Keo, T.; Collins, J.; Kunwar, P.; Blaser, M.J.; Iovine, N.M. Campylobacter capsule and lipooligosaccharide confer resistance to serum and cationic antimicrobials. Virulence 2011, 2, 30–40. [Google Scholar] [CrossRef]
- Jeon, B.; Muraoka, W.T.; Zhang, Q. Advances in Campylobacter biology and implications for biotechnological applications. Microb. Biotechnol. 2010, 3, 242–258. [Google Scholar] [CrossRef]
- Oh, E.; Jeon, B. Contribution of surface polysaccharides to the resistance of Campylobacter jejuni to antimicrobial phenolic compounds. J. Antibiot. 2015, 68, 591–593. [Google Scholar] [CrossRef]
- Brandt, U. Energy Converting NADH: Quinone Oxidoreductase (Complex I). Annu. Rev. Biochem. 2006, 75, 69–92. [Google Scholar] [CrossRef]
- Rollins, D.M.; Colwell, R.R. Viable but nonculturable stage of Campylobacter jejuni and its role in survival in the natural aquatic environment. Appl. Environ. Microbiol. 1986, 52, 531–538. [Google Scholar] [CrossRef]
- Skaar, E.P. The Battle for Iron between Bacterial Pathogens and Their Vertebrate Hosts. PLOS Pathog. 2010, 6, e1000949. [Google Scholar] [CrossRef]
| Group | Different Adhesion/Invasion Potential | % of Strains |
|---|---|---|
| Group 1 | High adhesion/low invasion | 5.3 |
| Group 2 | Low adhesion/high invasion | 65.8 |
| Group 3 | High adhesion/high invasion | 3.7 |
| Group 4 | Low adhesion/low invasion | 25.2 |
| Functional Group | Significantly Upregulated Genes |
|---|---|
| Transmembrane Proteins | |
| Sec protein system | secE, secY, secG, secD, secF, yidC |
| Tol-dependent translocation system | tolB |
| Pore forming channel proteins | ompA, omp50 |
| Cme efflux pump proteins | cmeAB |
| Other transmembrane proteins | cj0268c, lspA |
| Adhesion Proteins | |
| Autotransporter proteins | capA, capB |
| Fibronectin binding adhesion proteins | flpA, cj1349c |
| Other adhesion proteins | jlpA, cj0090, cj0091, cj0588, cadF |
| Bacterial Shape Determinant Genes | |
| Mre-based bacterial cytoskeleton proteins | pbpC, mreC, pbpB, rodA, mreB |
| Surface-Associated Saccharides | |
| Capsular polysaccharide (CPS) | kpsM, kpsT, kpsE, kpsC |
| Lipooligosaccharide (LOS) | galE, waaF, Cj1135, gmhE, lpxB |
| Invasion Proteins | |
| ATP-dependent protease | lon, clpP |
| energy taxis receptors | cetAB |
| Other invasion proteins | cipAB, htrA, cj0497 |
| Iron Acquisition | |
| Iron-uptake ABC transporter ATP-binding proteins | cfbpABC |
| The ferrous iron transport proteins | feoAB |
| Hemin uptake system proteins | chuBCD |
| Siderophore-mediated iron uptake system | ceuCDE |
| Colonisation | |
| Liv-system | livJ, livK |
| Other colonisation | Cj0561c, dnaJ, pldA, Cj0379c, docA |
| Regulatory Systems | |
| Two-component regulatory (TCM) systems | dccS |
| RacR–RacS system | racS |
| FlgSR system | flgR |
| Non TCM-system regulators | spoT |
| Chemotaxis Genes | cheB, cheW, cheV, cheR, cheA |
| Flagellar Machinery | |
| Stator proteins | motAB |
| Flagellar transport T3SS system | flhAB, fliIPR |
| Motor switch proteins | fliY |
| Energy Metabolism | |
| Glycosylation System | |
| “O”-linked glycosylation | hisH, fabH2 |
| “N”-linked glycosylation | pglAEFK, wlaDJ |
| Respiration | nuoACGIJKL |
| Protein Synthesis/Modification/Secretion | ssrA, tilS |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovács, J.K.; Cox, A.; Schweitzer, B.; Maróti, G.; Kovács, T.; Fenyvesi, H.; Emődy, L.; Schneider, G. Virulence Traits of Inpatient Campylobacter jejuni Isolates, and a Transcriptomic Approach to Identify Potential Genes Maintaining Intracellular Survival. Microorganisms 2020, 8, 531. https://doi.org/10.3390/microorganisms8040531
Kovács JK, Cox A, Schweitzer B, Maróti G, Kovács T, Fenyvesi H, Emődy L, Schneider G. Virulence Traits of Inpatient Campylobacter jejuni Isolates, and a Transcriptomic Approach to Identify Potential Genes Maintaining Intracellular Survival. Microorganisms. 2020; 8(4):531. https://doi.org/10.3390/microorganisms8040531
Chicago/Turabian StyleKovács, Judit K., Alysia Cox, Bettina Schweitzer, Gergely Maróti, Tamás Kovács, Hajnalka Fenyvesi, Levente Emődy, and György Schneider. 2020. "Virulence Traits of Inpatient Campylobacter jejuni Isolates, and a Transcriptomic Approach to Identify Potential Genes Maintaining Intracellular Survival" Microorganisms 8, no. 4: 531. https://doi.org/10.3390/microorganisms8040531
APA StyleKovács, J. K., Cox, A., Schweitzer, B., Maróti, G., Kovács, T., Fenyvesi, H., Emődy, L., & Schneider, G. (2020). Virulence Traits of Inpatient Campylobacter jejuni Isolates, and a Transcriptomic Approach to Identify Potential Genes Maintaining Intracellular Survival. Microorganisms, 8(4), 531. https://doi.org/10.3390/microorganisms8040531





