Abstract
Acinetobacter (A.) baumannii has gained global notoriety as a significant nosocomial pathogen because it is frequently associated with multi-drug resistance and hospital-based outbreaks. There is a substantial difference in the incidence of A. baumannii infections between different countries and within Germany. However, its continuous spread within Germany is a matter of concern. A systematic literature search and analysis of the literature published between 2000 and 2018 on A. baumannii in humans was performed. Forty-four studies out of 216 articles met the criteria for inclusion, and were selected and reviewed. The number of published articles is increasing over time gradually. Case reports and outbreak investigations are representing the main body of publications. North Rhine-Westphalia, Hesse and Baden-Wuerttemberg were states with frequent reports. Hospitals in Cologne and Frankfurt were often mentioned as specialized institutions. Multiresistant strains carrying diverse resistance genes were isolated in 13 of the 16 German states. The oxacillinase blaOXA-23-like, intrinsic blaOXA-51-like, blaOXA-58 variant, blaNDM-1, blaGES-11, blaCTX-M and blaTEM are the most predominant resistance traits found in German A. baumannii isolates. Five clonal lineages IC-2, IC-7, IC-1, IC-4 and IC-6 and six sequence types ST22, ST53, ST195, ST218, ST944/ST78 and ST348/ST2 have been reported. Due to multidrug resistance, colistin, tigecycline, aminoglycosides, fosfomycin, ceftazidime/avibactam and ceftolozan/tazobactam were often reported to be the only effective antibiotics left to treat quadruple multi-resistant Gram-negative (4MRGN) A. baumannii. Dissemination and infection rates of A. baumannii are on the rise nationwide. Hence, several aspects of resistance development and pathogenesis are not fully understood yet. Increased awareness, extensive study of mechanisms of resistance and development of alternative strategies for treatment are required. One-Health genomic surveillance is needed to understand the dynamics of spread, to identify the main reservoirs and routes of transmission and to develop targeted intervention strategies.
1. Introduction
Acinetobacter (A.) baumannii is a ubiquitous, Gram-negative, non-motile bacterium associated with hospital-acquired infection globally. It has been classified among the most dangerous multiple drug-resistant (MDR) pathogens worldwide. It has been listed in the “priority 1: critical” pathogens list of the World Health Organization (WHO). The pathogen was found in humans, animals, foods and the environment. A. baumannii is associated with severe infection, high mortality rates and massive economic loss [1]. Since the late 20th century, this bacterium emerged as a major cause of healthcare-associated infection in critically ill patients causing bloodstream infections, ventilator-associated pneumonia, meningitis, endocarditis, wound infection, urinary tract infections, skin and soft tissues infections [2]. Mortality rates can reach 35% in ventilator-associated pneumonia and bloodstream infections [3]. Members of the genus Acinetobacter are usually environmental bacteria. For unclear reasons, A. baumannii emerged as an opportunistic nosocomial pathogen. It has established niches for survival in hospitals as well as in primary animal production systems and the environment [4]. The main vital factors contributing to the extravagant dissemination of A. baumannii in hospitals are a large variety of potential sources of entry, environmental resilience and its ability to develop resistance to the majority of antibiotics, particularly the ability to acquire resistance to carbapenems [1,5]. A. baumannii becomes resistant via reducing membrane permeability, increasing efflux pump activity and production of a wide variety of β-lactamases. It is also able to acquire resistance via mutations and through horizontal gene transfer [6]. A. baumannii exhibits high genetic plasticity, which allows the accumulation of the acquired resistance determinants [7]. Resistance in this pathogen is frequently associated with mobile genetic elements (MGEs) that are transferable between bacteria, thus enabling the rapid dissemination of resistance genes between bacteria of different species and creating a reservoir of antimicrobial-resistance (AMR) genes [7].
For several years, the prevalence of infection with MDR A. baumannii has been increasing dramatically in the European countries that is, from 15.4% in 2004 to 48.5% in 2014 [8]. In the late 20th century, A. baumannii has emerged as a significant nosocomial pathogen in Germany [9,10,11]. Numerous nosocomial outbreaks were reported among inpatients and preterm infants with a considerable mortality rate of up to 24% after bacteremia in intensive care units at a large teaching hospital of Cologne [12]. Since 2000, the number of reported cases and hospital outbreaks was increasing continuously in most German states [13,14,15,16,17]. Isolates with resistance to all β-lactams, including carbapenems [18] and last resort antimicrobial agents such as colistin and tigecycline [19], were reported nationwide and have become a significant cause of worry of the healthcare system. Several studies on A. baumannii have been published from different German states but a systematic review on its spatial and temporal distribution is not yet available. Thus, the current report aims at providing a comprehensive, evidence-based assessment of the literature published from 2000-2018 on A. baumannii in Germany.
2. Materials and Methods
2.1. Data Sources and Search Strategy
A comprehensive search in PubMed, Web of Science, Scopus and the catalogue of the German National Library was performed for articles that include the search terms “Acinetobacter baumannii” and “Germany” in their title and abstract. All national and international studies discussing A. baumannii in Germany published in both English and German language were collected. References cited within these papers were checked to find additional relevant information.
2.2. Selection Criteria and Data Extraction
Studies discussing isolation, epidemiology and antimicrobial resistance of A. baumannii from 2000 through 2018 were included. Review articles, congress abstracts, case reports, letters, commentaries and editorials were included. Abstract analysis for each publication was done and, if selected, the full text was carefully analyzed. The following keywords were used—Acinetobacter baumannii + Germany + human + antimicrobial resistance. Articles describing other Acinetobacter spp., A. baumannii in non-human sources, novel diagnostics, development of PCR and studies published before the year 2000 were excluded. For each study, following information was extracted from the full text—first author, year of publication, sampling date, location of sampling (city and state), study population, type and source of samples, number of strains and cases, type of study, resistance profiles of the recovered strains including phenotypic testing and detection of resistance genes. If available, the sequence and clonal type of strains were included.
2.3. Data Acquisition and Analysis
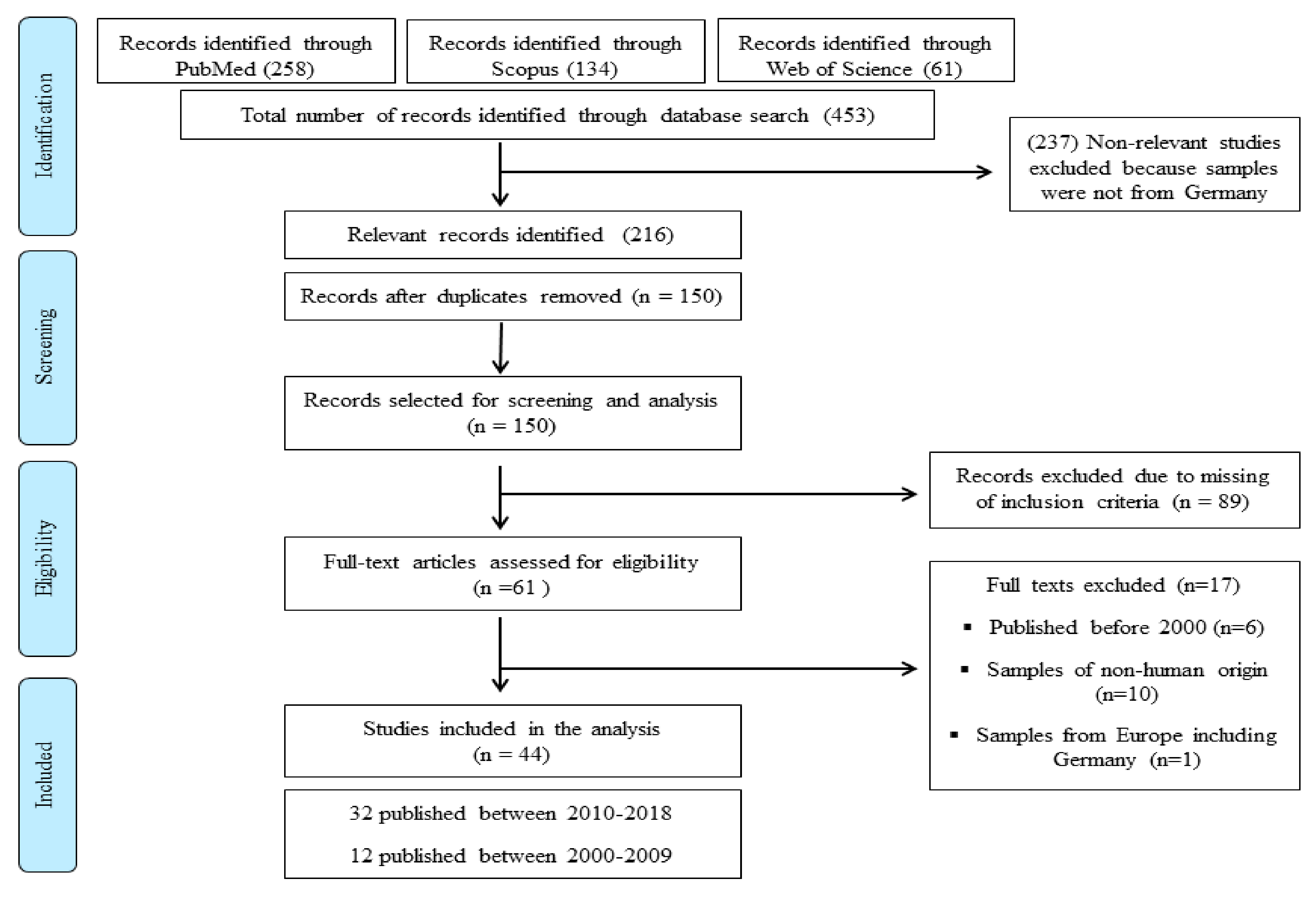
Overall, a total of 216 potentially relevant articles [PubMed (n = 114), Scopus (n = 69) and Web of Science (n = 33)] were identified. Sixty-one articles were selected for screening and forty-four studies were included in the review (Figure 1: PRISMA diagram). The information was categorized starting with the latest one.
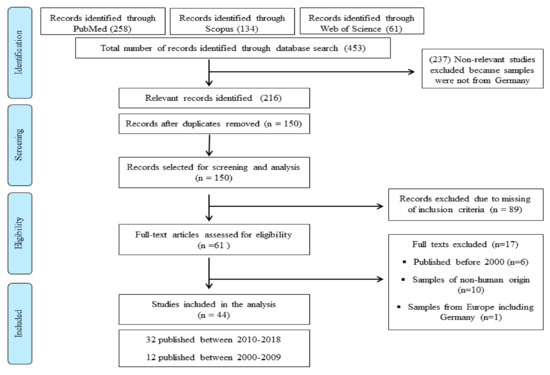
Figure 1.
Systematic review flowchart: Acinetobacter baumannii in Germany, the flow diagram indicates the inclusion and exclusion of publications at each stage of the systematic review process.
2.4. Relative Research Interest
The number of publications that are available on PubMed for each year was extracted and the relative research interest was calculated [20]. It is defined as the quotient of specific articles divided by all articles in this year to consider the overall publication growth per year.
3. Results
3.1. Data Analysis
Case reports represented the majority of the published articles (n = 14), followed by outbreak reports (n = 7) and evaluation of antibiotic activity (n = 6). The remaining studies were surveillance studies, routine microbiological examination and descriptive data analysis. Thirty-seven articles were published in English and seven in German [15,16,17,18,19,20,21]. The sampling date was not reported in 15 studies, while the geographical location of samples was ignored in three studies. Lost information was categorized as “non-determined” and referred to as ‘ND’ in the tables. A. baumannii was most often isolated from the bloodstream, wound infections and respiratory and urinary tract infections as well as from skin and rectal swabs. In outbreak investigations it was also isolated from the patient’s environment, medical devices, patient’s associated belongings, room equipment, infusion pumps and oxygen masks [20,22]. In the catalogue of the German National Library, 148 doctoral theses containing the word Acinetobacter in their titles were identified. Twenty-one of these were published between 2000–2018. Only nine dissertations with emphasis on isolation, molecular and functional typing, distribution and genetic composition of AMR determinants were found (data are not shown). Three of these were done in Bonn, three in Cologne and one each in Berlin, Freiburg and Frankfurt am Main representing the main centres of A. baumannii research.
3.2. Temporal Distribution of the Analyzed Studies
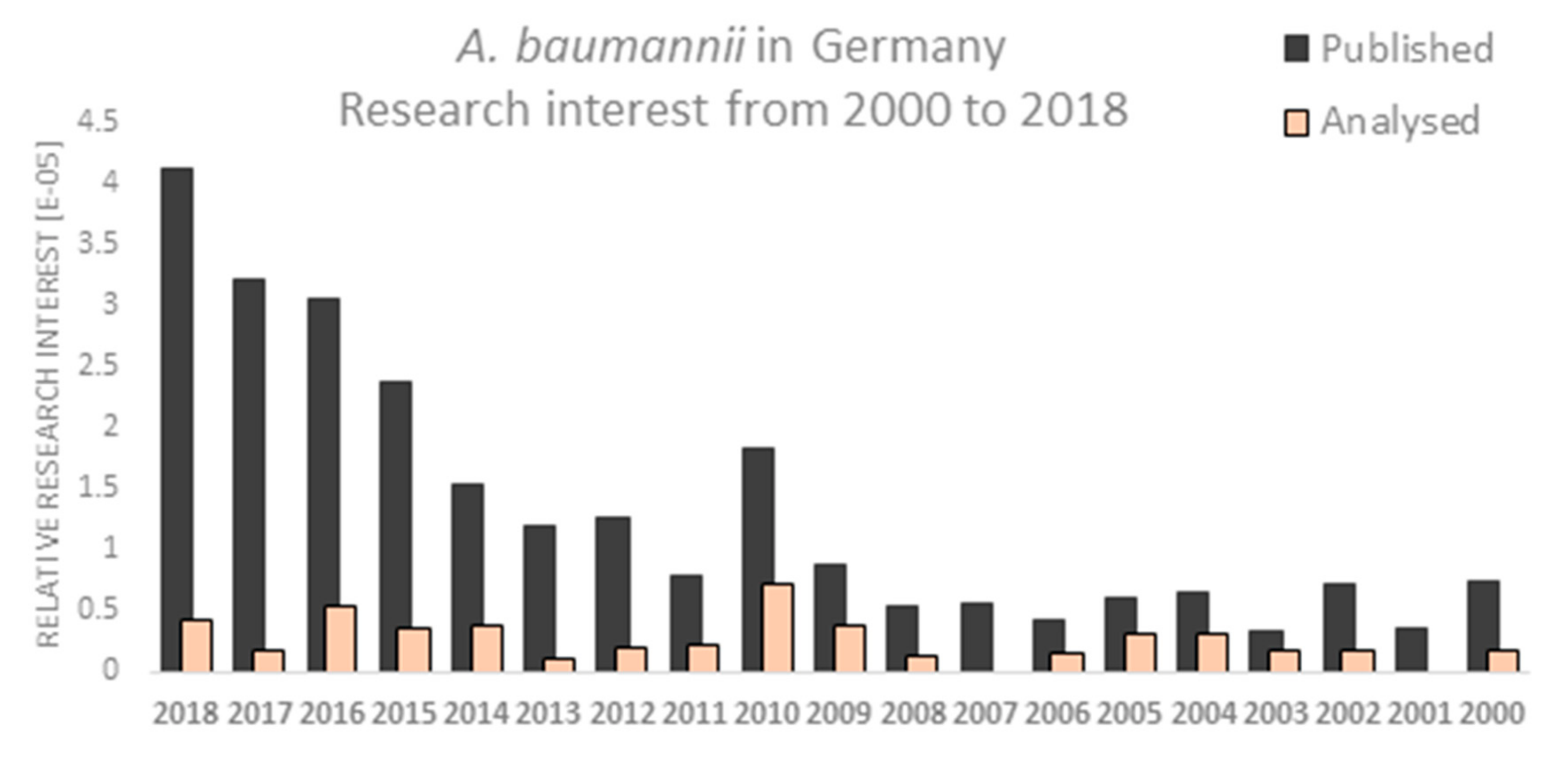
The emergence of MDR in A. baumannii has been steadily increasing over time and the number of published articles reflects this fact. The relative research interest has been growing after 2013. The year 2010 was super crucial for the research on A. baumannii in Germany. PubMed analysis using the search term ‘A. baumannii in Germany’ showed a gradual increase in the number of published studies over time with the highest number of annual articles in 2018 (Figure 2). Finally, 44 articles were analyzed. Approximately two thirds of studies (n = 32) were published in or after 2010, of them 13 articles have been published in the years 2016–2018 (Table 1).
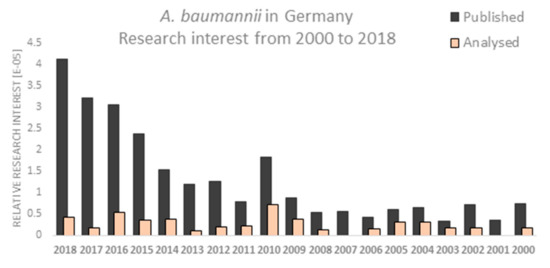
Figure 2.
PubMed analysis showing the relative research interest for A. baumannii in Germany from 2000–2018. Publications analyzed in this work a marked orange.

Table 1.
Spatio-temporal distribution of studies on A. baumannii of human origin in Germany from 2000 to 2018.
3.3. Spatial Distribution of the Analyzed Studies Based on Geographical Location of Sampling
Most of the published articles are regional studies. Only six studies included samples throughout Germany but without the exact geographical origin of samples. North Rhine-Westphalia, Hesse and Baden-Wuerttemberg contributed eleven, eight and five studies, respectively, followed by Bavaria (four studies), Saxony (three studies) and Berlin and Schleswig-Holstein (two studies each). From Thuringia, Hamburg, Lower Saxony, Saxony-Anhalt, Rhineland-Palatinate and Mecklenburg-Vorpommern, only one study was accessible. Brandenburg, Bremen and Saarland are not present at all. Cologne, the largest city of North Rhine-Westphalia, was the most represented city and was mentioned in six studies. Frankfurt am Main, the largest city of Hesse, was mentioned in five studies. Both cities are centers of A. baumannii research. Munich, the largest city of Bavaria, contributed three studies. Leipzig, one of the biggest cities of Saxony, Tuebingen and Freiburg, two cities in Baden-Württemberg, were study areas for two studies each (Table 1).
3.4. Official Data Concerning the Infection and Epidemiology of Acinetobacter in Germany
Within the framework of the Infection Protection Act in Germany, the Robert Koch Institute (RKI) is responsible for collecting data on notifiable diseases. The number of nosocomial outbreaks and the number of cases in each outbreak, as well as the number of deaths were recorded for 2010 onwards [59]. As shown in Table 2, no information has been documented in 2010 and 2011 and for the years 2012, 2013 and 2016 the highest numbers of nosocomial outbreaks were reported. The annual statistics on reportable diseases of Germany reported 794 and 784 Acinetobacter infections or colonization in 2017 and 2018, with an incidence of 1.0 and 0.9, respectively. North Rhine-Westphalia reported the highest numbers of Acinetobacter infections or colonizations with 405 notifications, followed by Bavaria (n = 205), Hesse (n = 200) and Berlin (n = 196) [60]. In contrast, the lowest incidences were reported for the federal states of Bremen (n = 6) and Saarland (n = 5). It has to be taken into consideration that the numbers mentioned in the RKI statistics do not represent the total number of infections in Germany because only the carbapenem-non-susceptible isolates are reported to the RKI and species of Acinetobacter are not differentiated.

Table 2.
Number of the nosocomial outbreaks of carbapenem-non-susceptible Acinetobacter spp. in Germany based on official data published by the Robert Koch Institute (RKI) (2010–2018).
3.5. Identification of A. baumannii and Antibiotic Susceptibility Testing (AST)
Identification of A. baumannii has been done mostly by the VITEK-2 system, MALDI-TOF [18,26] and detection of the intrinsic blaOXA-51-like gene [30]. Phenotypic antibiotic sensitivity testing (AST) was performed with the VITEK-2 platform, E-test [28], disk diffusion method [24], antibiotic gradient test and micro-broth dilution using Micronaut-S system [22]. The National Committee for Clinical Laboratory Standards (NCCLS) broth microdilution method and the respective breakpoints were used until 2006 [52]. The broth dilution test was performed following the guidelines of the German Institute for Standardization (DIN) in 2010 [46] and 2013 [39]. The phenotypic detection of resistance in most of the published studies was performed following the Clinical and Laboratory Standards Institute (CLSI) recommendations and the European Committee on Antimicrobial Susceptibility Testing (EUCAST). No breakpoint for tigecycline is approved by EUCAST [49,50]. Thus, the breakpoint of EUCAST for Enterobacteriaceae has been used [16].
3.6. Resistance Development of A. baumannii
At the beginning of the 21st century, A. baumannii attracted the attention of researchers due to its rapid spread and its resistance to most of the antibiotics used. In a surveillance study performed from 2001 to 2008 in 53 German intensive care units (ICUs), the resistance to imipenem increased from 1.1% in 2001 to 4.5% in 2008 [61]. From 2002 to 2006, the rate of isolation of A. baumannii increased in university hospitals of six German federal states (Baden-Wuerttemberg, Hesse, Lower Saxony, Saxony, Schleswig–Holstein and Thuringia) from 2.1% in 2002 to 7.9% in 2006 [46] and the proportion of MDR of A. baumannii isolates raised among inpatients [46]. XDR strains resistant to all available antibiotics except colistin have been isolated from wound, skin and respiratory tract samples in an outbreak in 2003 [14]. To overcome the resistance developed against broad-spectrum antibiotics tetracycline, tigecycline a new class of antibiotics derived from tetracycline has been implemented in 2006. Between 2004 and 2007, tigecycline had excellent activity against A. baumannii and other Gram-negative nosocomial bacteria [49,50,51]. Until 2005 carbapenems remained the gold standard of therapy for severe A. baumannii infections and resistance to carbapenems remained low (0.3%) [54]. In 2006, the first outbreak of carbapenem-resistant A. baumannii (CRAb), carrying the blaOXA-23-like gene, was reported in Germany. The strain was resistant to imipenem, meropenem, penicillins, cephalosporins, ciprofloxacin, gentamicin, tobramycin and tigecycline and was related to the pan-European A. baumannii clone II [16]. Since then, carbapenemase-producing strains have been recovered from several outbreaks every year [26,27,30,33,62]. According to the National Reference Laboratory for Gram-negative nosocomial pathogens at the Ruhr-University Bochum, CRAb was found in 96.3% of strains and blaOXA-23 was the most frequent carbapenemase in A. baumannii (81.1%) in 2011 [41]. This high resistance rates can be explained by the practice that only non-susceptible strains are sent to the National Reference Laboratory for clarification. It is worth to mention that, the number of cases of carbapenem insensitive Acinetobacter infection or colonization was 794 in 2017 and 784 in 2018 [60]. In contrast, the ARS data demonstrate that only 4% of A. baumannii ABC complex strains were not carbapenem sensitive in 2015 to 2018 (https://ars.rki.de/Content/Database/ResistanceOverview.aspx).
Susceptibility to tigecycline and colistin was reported in four and nineteen studies, respectively (Table 3). Susceptibility to colistin and tigecycline remained unaffected until December 2013. At this time, the first reported pan drug-resistant (PDR)-CRAb strain was recovered from a patient at Goethe University hospital Frankfurt after a previous hospitalization in Greece. This strain was also resistant to colistin [19]. Until 2018 colistin-resistant strains were detected three times in Germany. One was isolated from a human in 2013 in Frankfurt [19], one from a dog in Giessen in 2011 [63] and one from wastewater in 2017 [64]. It has to be taken in consideration that most of the earlier colistin testing was performed by gradient tests for example, disc diffusion, E-test or with VITEK. EUCAST has revised the recommendation for colistin AST and microdilution technique is considered the only reliable method [65]. Tigecycline resistant strains were reported five times: once in 2016 in Leverkusen [30], once in 2014 from Frankfurt [19], two times in 2010 from the university hospital Frankfurt [47] and from the university hospital Rostock [15] and once in 2009 with undetermined location [16]. Colistin, tigecycline, aminoglycosides, fosfomycin, ceftazidime/avibactam and ceftolozan/tazobactam are often used antibiotics to treat 4MRGN A. baumannii infection in Germany [66]. Information regarding the resistance patterns of A. baumannii are shown in Table 3.

Table 3.
Temporal distribution of resistance profiles of A. baumannii strains from Germany based on studies published from 2000 to 2018.
3.7. Resistance Genes in A. baumannii
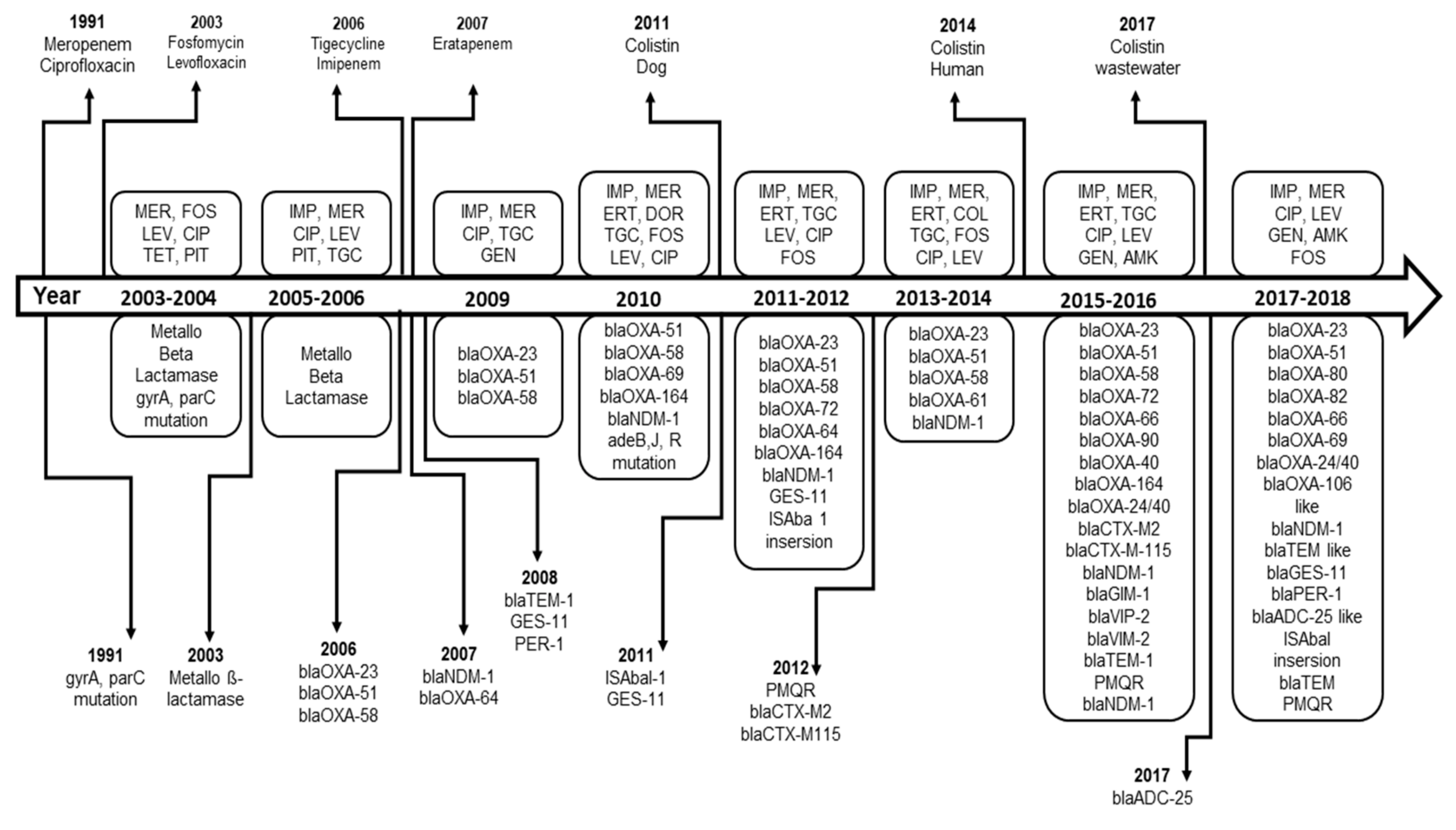
Twenty-eight of the selected studies described AMR genes. As shown in Table 3, the metallo-ß-lactamase was first described in studies published in 2004, 2005 and 2006 [14,52,53]. The oxacillinase blaOXA-23-like, the intrinsic blaOXA-51-like, the New Delhi Metallo-ß-lactamases (blaNDM) and the blaOXA-58-like variant are the most dominant resistance genes that have been described in A. baumannii from Germany (Table 3). The gene blaOXA-23-like, the intrinsic blaOXA-51-like and the blaOXA-58-like variants were found in a CRAb strain for the first time in an outbreak, which occurred at a German university medical center in 2006. However, the location of the sampling was ignored [16]. Those three genes have been reported later on in sixteen, elven and seven studies, respectively. In 2011, the National Reference Laboratory for Gram-negative nosocomial pathogens at Ruhr-University Bochum found that the blaOXA-23 was the most frequent carbapenemase in A. baumannii [41]. The first isolation of a blaNDM-1 strain was reported in 2007 in Frankfurt [47] and then in ten other studies. Additionally, Guiana extended-spectrum β-lactamases (GES-11) [41], Cefotaxime Hydrolyzing Capabilities (CTX-M) β-lactamases [28], Temoneira (TEM) and Plasmid-Mediated Quinolone Resistance (PMQR) [35], German imipenemase (GIM-1), VIP-2 and Verona integron-encoded-metallo-β-lactamase (VIM-2) [32] and blaADC-25-like [18] have been described in A. baumannii (Table 3). Figure 3 represents the chronological emergence of resistance to carbapenems, colistin, tigecycline and quinolones antibiotics as well as the resistance genes according to time of sampling or time of reporting if the time of sampling is not available.
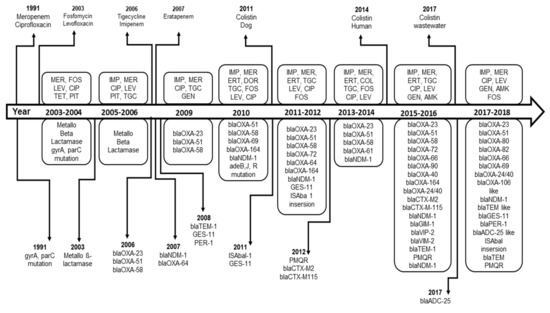
Figure 3.
Chronological emergence of ß-lactam resistance in A. baumannii in Germany.
3.8. Clonality and Sequence Typing
Cluster analysis based on Pulsed-field Gel Electrophoresis (PFGE) and rep-PCR typing identified five clonal lineages of A. baumannii in Germany, IC-2, IC-7, IC-1, IC-4 and IC-6. The international clone IC-2 is the most common clonal lineage widespread in Germany and was present in seven studies. Followed by IC-7 and IC-1, each was reported in two studies, while IC-4 and IC-6, each was identified only in one study. IC-2 was detected in five studies alone [16,19,27,33,42] and in combination with IC-6 once [28] and with IC-1, IC-7 and IC-4 in another study during the investigation of strains collected from different laboratories in the years 2005, 2007 and 2009 [39]. Only six sequence types of A. baumannii have been reported in the reviewed articles. Multilocus sequence typing (MLST) revealed ST22 and ST53 types among 13 strains recovered from an outbreak of seven patients in Rostock [15]. ST195 and ST218 were identified in an outbreak involving ten patients in Leverkusen [30]. ST944/ST78 and ST348/ST2 were determined in three isolates recovered from two patients hospitalized in Bavaria and Hesse [28].
3.9. The Risk Factors Associated with A. baumannii Infections
Investigation and outbreak studies, as well as case reports analyzed in this review, have reported multiple risk factors for the emergence and acquisition of A. baumannii infections. Firstly, travel, medical tourism or contact with health care systems abroad, as well as cross border transfer of patients from highly endemic countries into Germany, lead to the import and emerging presence of A. baumannii in the German hospitals. Between June and December 2015, 143 refugees have been admitted to the University hospital Frankfurt, Hesse. 60.8% were positive for MDR Gram-negative bacteria and CRAb strains harboring blaOXA-23 and OXA-24 genes were reported in 1.4% of clinical samples examined [27]. blaOXA-23 and blaOXA-48, the most frequently reported resistance genes in Germany, were detected with high prevalence among Libyan war casualties admitted to the Northwest Medical Center (NMC) in Frankfurt/Main, Hesse between August 2016 and January 2017 [24]. MDR A. baumannii was recovered from patients in German hospitals after previous hospitalization in Russia [28,34], Thailand [30], Serbia [42,47], Poland [43], China, the United Arabic Emirates and Croatia [37], Greece and Italy [53] and other Mediterranean countries [52]. A patient was infected in Cameroon [14]. First reports on PDR (pan-drug resistant) A. baumannii appeared in 2001 and an outbreak was reported in Spain in 2002 [67]. Subsequently, the first isolation of a PDR strain in Germany belonging to the IC-2 cluster was done from the skin and rectal swab samples of a patient previously hospitalized in Greece [19]. In contrast, a CRAb strain harboring blaOXA-23 was isolated from a hip joint infection in a patient with no history of traveling abroad at the University hospital Bonn [33]. Between 2005 and 2009, 140 CRAb strains were isolated in 15 different medical centers in Germany, only 32% of these strains belonged to one of the known international clonal lineages [39].
Secondly, during a (long-term) ICU stay, A. baumannii can be acquired via mechanical ventilation. An epidemic strain of A. baumannii was transferred from the index patient to other nine patients at the same hospital in Leverkusen, North Rhine-Westphalia within 16 days [30]. Cross-transmission via the hands of health care workers from colonized or infected patients has been described in an epidemic setting in a German university medical center in Berlin [16]. In one year, 44 individual patients acquired infection in a hospital in Cologne. A. baumannii was recovered from the washbasin and fixation bath [18], from medical devices, infusion pumps and oxygen masks [15,16]. An identical macrorestriction pattern (PFGE-type) was detected in isolates of two different hospitals in 2008 in North Rhine-Westphalia confirming that inter-hospital transmission is possible [26]. Moreover, a prolonged hospital stay is frequently associated with exposure to antibiotics [68]. A. baumannii can rapidly acquire resistance by mutation during antimicrobial therapy [44]. Selective pressure due to preceding antimicrobial treatment was a significant factor in acquiring carbapenem-resistance in a study from Hamburg-Eppendorf, Hamburg [21].
Finally, the environmental resilience of A. baumannii and environmental contamination facilitates the spread and dissemination of infection from colonized or infected patients. Examination of environmental samples during an outbreak revealed relevant dissemination of A. baumannii [18]. A. baumannii was isolated from the patients’ environment, patients’ belongings and room equipment [15,16]. A. baumannii can survive in the environment for a prolonged time and it can colonize non-human hosts for example, pet animals with ease [69]. The occurrence of genotypically related clones I–III in animals and humans in Germany raises the concern about the possibility of a spillover of the organisms from humans to animals or vice versa [63]. Strains harboring resistance genes coding for clinically relevant antibiotics can enter the clinical setting via food and community routes [70,71]. Thus, humans can be colonized by A. baumannii through the food chain or from other environmental sources for example, wastewater, hatchery system and dust.
4. Discussion
Acinetobacter baumannii is one of the most threatening emerging environmental pathogens, causing particularly nosocomial infections in humans in Germany. The number of published studies concerning A. baumannii is dramatically increasing worldwide and also in Germany. This systematic review investigated the spatial and temporal distribution of A. baumannii in humans in Germany based on published data from 2000 to 2018. The pathogen was extensively studied in the last few years. This increase of interest reflects the increasing clinical impact and its ability to acquire resistance against the majority of antibiotics. A. baumannii MDR strains that is, strains with resistance to all β-lactams including carbapenems and production of β-lactamases of several types, were reported nationwide. A prominent number of published articles originates from North Rhine-Westphalia. This state is located in the West of Germany and has the highest population of German states. A considerable number of reports also describe the situation of Hesse, which is a central German state. The cities Cologne and Frankfurt-am-Main are centres of A. baumannii research and are home to tertiary care clinics and hospitals which treat a relatively high numbers of patients in ICUs. These hospitals also run large highly specialized treatment units that received patients from abroad [18,24]. The obtained results are in harmony with the official data of the Robert Koch Institute (RKI) considering numbers of infection or colonization of carbapenem non-susceptible Acinetobacter spp. and incidence [60]. The official data published by RKI were included to avoid loss of information, aware the shortcoming of these data that is, no differentiation between the different species was made and that numbers do not present the total number of infections in Germany, as only carbapenem non-susceptible strains have to be notified. Information published by the ARS project cover data for the whole A. baumannii complex group.
The identification of A. baumannii was mostly based on the VITEK-2 system, MALDI-TOF and PCR. The failure of the semi-automated VITEK-2 system to correctly identify A. baumannii and predict carbapenem susceptibility is well known [44]. MALDI-TOF log score values >2000 were accepted in some laboratories for species identification [72]. However, evaluation of species-specific score cut-off values of clinical A. baumannii revealed that a score value > 2.3 is the valid score value for species identification [73]. Species misidentification and false AST may negatively influence clinical outcome. A. baumannii is part of the ACB complex and it is difficult to distinguish A. baumannii from other species phenotypically. A proper, accurate and reliable molecular identification of A. baumannii to species level is needed and a typing method with high discriminatory power is crucial [18,44] for diagnosis, epidemiological trace back and vaccine production. Rates of colonization and infection with triple (3MRGN) and quadruple (4MRGN) A. baumannii are on the rise in Germany [17,29,36]. However, this type of classification is specific for Germany only. Carbapenems are the gold standard to date for the treatment of severe infections with MDR strains. However, the appearance of CRAb poses new challenges for treatment strategies [66]. Nevertheless, colistin and tigecycline are therapeutic options and showed efficacy in the treatment of MDR strains [15,25,37,51]. The appearance of colistin and tigecycline resistant [47] and colistin-resistant strains carrying the mcr-1 gene in non-human sources [64] will result in limited effectiveness in the near future. Several methods have been used to detect colistin and tigecycline resistant strains. The lack of EUCAST and CLSI breakpoints for tigecycline hampers the detection of tigecycline resistance and is a big challenge for clinicians [74]. Accordingly, the breakpoint for Enterobacteriaceae was recommended and used in different studies in the past [16]. However, caution is needed during interpretation. A. baumannii is intrinsically resistant to fosfomycin [75]. Only six articles were found discussing resistance to fosfomycin. The first fosfomycin resistant A. baumannii isolate was reported from an outbreak at the university hospital Tuebingen in 2004 [14] and then MDR strains resistant to fosfomycin were reported again in 2010, 2011, 2014 and 2017 [19,25,43,44]. According to the Surveillance of Antibiotic Use and Resistance in Intensive Care Units (SARI-ICUs) project, that was initiated in Germany in 2000, the resistance rate of MRGN pathogens increased markedly from 2001 to 2015 [76]. The increase in A. baumannii isolates with resistance to imipenem has been reported from 2005 onwards with a pronounced trend. The resistance rate has more than doubled over recent years and reach to 43% in 2015 [76]. The antibiotic resistance surveillance (ARS) project revealed that, the level of resistance in A. baumannii isolates obtained from patients in ICUs is about three times that higher than of isolates obtained from patients in normal and outpatient settings. 7.6% of A. baumannii strains were carbapenem resistant in 2014 and 4% in 2017 and 2018. However, it has to be kept in mind that this information is not restricted to A. baumannii but include all of the A. baumannii complex group (https://ars.rki.de/Content/Database/ResistanceOverview.aspx).
The blaOXA-23, blaOXA-51-like and blaOXA-58-like, as well as blaNDM-1 were the resistance genes most often found in A. baumannii in the last ten years followed by blaGES-11, blaCTX-M and blaVIM. Since the first description of the blaOXA-23 in a clinical isolate of A. baumannii in Scotland in 1995 [77], A. baumannii harboring the blaOXA-23 gene spread to hospitals worldwide and were also detected in companion animals, livestock, environment and ectoparasites [78]. It has become the most frequent carbapenemase gene among A. baumannii in most countries. Transfer of resistance genes from bacteria of other genera is a predominant mechanism of acquiring resistance to carbapenems in A. baumannii. The blaNDM-1 was identified mainly in E. coli and K. pneumoniae and to a lower extent in Pseudomonas and Acinetobacter. The first occurrence of A. baumannii carrying blaNDM-1 in Germany dates bake to the year 2007 [47,48]. Hence, current PCR protocols only identify resistance-coding genes but provide no in-depth information regarding the functions and mechanisms of resistance development or actual clinical resistance. Detection and characterization of AMR genes are gradually moving from PCR to high throughput identification via sequencing and in-silico detection utilizing several databases now. However, the use of next-generation sequencing (NGS) to investigate AMR in A. baumannii is still rare and has just begun to start in Germany.
This discriminatory power enables transmission-chain analysis, optimizes surveillance and promotes suitable containment measures. It also offers the traits to identify transmission dynamics [79]. Molecular characterization of AMR in A. baumannii often has been performed by using conventional methods and very few studies utilizing the WGS technology have been performed [18,31,35]. For outbreak investigation, PFGE has been considered the gold standard for a long time but it has been replaced by NGS technology due to its higher discriminatory properties. Most of the WGS based studies on A. baumannii were performed in outbreak settings and there is little data on WGS in the context of surveillance.
International travel of colonized patients, especially from countries with high prevalence for example, Mediterranean and Asian countries has resulted in and still resulting in the introduction and later spread of MDR A. baumannii to Europe and subsequently into Germany [53]. Following a fatal outbreak of six patients with A. baumannii infection-related death at a tertiary care facility in the northwest United States, comparative genomics of strains showed a close relation to strains isolated from Germany in 2003 [80]. Isolates from Germany were found to share the same rep-PCR pattern with isolates from Turkey and the USA [81] and identical to those from isolates from the UK [82]. Thus, cross border transmission of MDR A. baumannii or its resistance genes have to be considered and the colonization status of patients treated abroad should be handled with care to avoid the spread of this pathogen into countries with low incidence rates [53]. WGS can be the tool of choice to assist public health officer and clinicians to contain spread and to initiate well timed start of treatment with the best medication.
5. Conclusions
In conclusion, the published knowledge on the existence and general distribution of A. baumannii in the German population is gradually increasing over time but is mainly based on regional studies, outbreak investigations and case reports. Only few countrywide studies have been done yet. Several aspects of resistance development remain cryptic. Large cities with tertiary care hospitals and vital tourism are hot spots for research and publication activity on A. baumannii. MDR strains harboring diverse resistance genes were found in most of the federal states. Five clonal lineages and six sequence types of A. baumannii have been identified within the last 18 years in Germany. A. baumannii is part of the ACB complex and it is difficult to distinguish it from other species. Due to clinical needs, its differentiation from other members of the ACB complex species, particularly the closely related A. pittii and A. nosocomialis is required. Accurate and safe diagnostics for species identification as well as the definition of breakpoints for all antibiotics used needs to be established. Several risk factors contributing to the spread and dissemination of infection have been identified for example, travelling, medical tourisms, long-term stay at ICUs and the environmental contamination. Dissemination and infection rates of MDR (particularly those known in Germany as 3MRGN-Ab and 4MRGN-Ab) are on the rise nationwide and need to be considered a threat to public health.
Due to the nature of the data reported to RKI they cannot differentiate between the different species causing infection or colonization. The ARS project shaves this limitation as it collects data on the A. baumannii complex following a holistic approach. The reported data published by the National Reference Laboratory and the RKI do not sum up the total number of infections or colonizations in Germany because only carbapenem non-susceptible Acinetobacter strains need to be reported to the NRL and RKI. The nationwide One-Health genomic surveillance of A. baumannii using NGS technology to understand the dynamics of resistance and mobile genetic elements (MGEs) has to be in the center of public efforts, now the NGS is afford and available. Especially pets, livestock, food and the environment are neglected reservoirs and isolates from theses niches need to be investigated to assess their impact on human health and economy. Thus, identification of novel and alternative strategies for treatment, collaboration between the veterinary and human health sectors and the increase of awareness in the public and in the risk group are fields identified to be improved.
Author Contributions
G.W. performed the online database query. G.W. and L.D.S. read abstracts and selected studies for full text review. C.B. carried out the relative research interest and associated figure. G.W. wrote the manuscript. H.N. and M.W.P. supervised the review. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by internal funding of the Friedrich-Loeffler-Institute. C.B. and M.W.P. were co-funded by grants from the Federal Ministry of Education and Research (Germany), Grant Numbers 13GW0096D and 01KI1501.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lin, M.F.; Lan, C.Y. Antimicrobial resistance in Acinetobacter baumannii: From bench to bedside. World J. Clin. Cases 2014, 2, 787–814. [Google Scholar] [CrossRef] [PubMed]
- Howard, A.; O'Donoghue, M.; Feeney, A.; Sleator, R.D. Acinetobacter baumannii: An emerging opportunistic pathogen. Virulence 2012, 3, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Antunes, L.C.; Visca, P.; Towner, K.J. Acinetobacter baumannii: Evolution of a global pathogen. Pathog. Dis. 2014, 71, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Yakkala, H.; Samantarrai, D.; Gribskov, M.; Siddavattam, D. Comparative genome analysis reveals niche-specific genome expansion in Acinetobacter baumannii strains. PLoS ONE 2019, 14, e0218204. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Murray, G.L.; Peleg, A.Y. Acinetobacter baumannii: Evolution of antimicrobial resistance-treatment options. Semin. Respir. Crit. Care Med. 2015, 36, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Lupo, A.; Haenni, M.; Madec, J.Y. Antimicrobial resistance in Acinetobacter spp. and Pseudomonas spp. Microbiol. Spectr. 2018, 6, 377–393. [Google Scholar] [CrossRef]
- Pagano, M.; Martins, A.F.; Barth, A.L. Mobile genetic elements related to carbapenem resistance in Acinetobacter baumannii. Braz. J. Microbiol. 2016, 47, 785–792. [Google Scholar] [CrossRef]
- Rodloff, A.C.; Dowzicky, M.J. Antimicrobial susceptibility among European Gram-negative and Gram-positive isolates collected as part of the Tigecycline Evaluation and Surveillance Trial (2004–2014). Chemotherapy 2017, 62, 1–11. [Google Scholar] [CrossRef]
- Graser, Y.; Klare, I.; Halle, E.; Gantenberg, R.; Buchholz, P.; Jacobi, H.D.; Presber, W.; Schonian, G. Epidemiological study of an Acinetobacter baumannii outbreak by using polymerase chain reaction fingerprinting. J. Clin. Microbiol. 1993, 31, 2417–2420. [Google Scholar] [CrossRef]
- Seifert, H.; Baginski, R.; Schulze, A.; Pulverer, G. The distribution of Acinetobacter species in clinical culture materials. Zent. Bakteriol. Int. J. Med Microbiol. 1993, 279, 544–552. [Google Scholar] [CrossRef]
- Thurm, V.; Ritter, E. Genetic diversity and clonal relationships of Acinetobacter baumannii strains isolated in a neonatal ward: Epidemiological investigations by allozyme, whole-cell protein and antibiotic resistance analysis. Epidemiol. Infect. 1993, 111, 491–498. [Google Scholar] [CrossRef]
- Seifert, H.; Baginski, R. The clinical significance of Acinetobacter baumannii in blood cultures. Zent. Bakteriol. Int. J. Med Microbiol. 1992, 277, 210–218. [Google Scholar] [CrossRef]
- Wieland, K.; Chhatwal, P.; Vonberg, R.P. Nosocomial outbreaks caused by Acinetobacter baumannii and Pseudomonas aeruginosa: Results of a systematic review. Am. J. Infect. Control 2018, 46, 643–648. [Google Scholar] [CrossRef]
- Borgmann, S.; Wolz, C.; Grobner, S.; Autenrieth, I.B.; Heeg, P.; Goerke, C.; Muller, K.; Kyme, P.; Marschal, M. Metallo-beta-lactamase expressing multi-resistant Acinetobacter baumannii transmitted in the operation area. J. Hosp. Infect. 2004, 57, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Frickmann, H.; Crusius, S.; Walter, U.; Podbielski, A. Management eines Ausbruchs nosokomialer Pneumonien durch einen neuen multiresistenten Acinetobacter baumannii-Klon. Pneumologie 2010, 64, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Kohlenberg, A.; Brummer, S.; Higgins, P.G.; Sohr, D.; Piening, B.C.; de Grahl, C.; Halle, E.; Ruden, H.; Seifert, H. Outbreak of carbapenem-resistant Acinetobacter baumannii carrying the carbapenemase OXA-23 in a German university medical centre. J. Med Microbiol. 2009, 58, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Siemers, F.; Fanghanel, S.; Bergmann, P.A.; Tamouridis, G.; Stuttmann, R.; Stolze, B.; Hofmann, G.O. Management eines Ausbruches mit 4 MRGN Acinetobacter baumannii in einem Brandverletzten-zentrum. Handchir. Mikrochir. Plast. Chir. 2014, 46, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Wendel, A.F.; Malecki, M.; Otchwemah, R.; Tellez-Castillo, C.J.; Sakka, S.G.; Mattner, F. One-year molecular surveillance of carbapenem-susceptible A. baumannii on a German intensive care unit: Diversity or clonality. Antimicrob. Resist. Infect. Control 2018, 7, 145. [Google Scholar] [CrossRef]
- Gottig, S.; Gruber, T.M.; Higgins, P.G.; Wachsmuth, M.; Seifert, H.; Kempf, V.A. Detection of pan drug-resistant Acinetobacter baumannii in Germany. J. Antimicrob. Chemother. 2014, 69, 2578–2579. [Google Scholar] [CrossRef]
- Brandt, C.; Makarewicz, O.; Fischer, T.; Stein, C.; Pfeifer, Y.; Werner, G.; Pletz, M.W. The bigger picture: The history of antibiotics and antimicrobial resistance displayed by scientometric data. Int. J. Antimicrob. Agents 2014, 44, 424–430. [Google Scholar] [CrossRef]
- Katchanov, J.; Asar, L.; Klupp, E.M.; Both, A.; Rothe, C.; Konig, C.; Rohde, H.; Kluge, S.; Maurer, F.P. Carbapenem-resistant Gram-negative pathogens in a German university medical center: Prevalence, clinical implications and the role of novel beta-lactam/beta-lactamase inhibitor combinations. PLoS ONE 2018, 13, e0195757. [Google Scholar] [CrossRef]
- Kerz, T.; von Loewenich, F.D.; Roberts, J.; Neulen, A.; Ringel, F. Cerebrospinal fluid penetration of very high-dose meropenem: A case report. Ann. Clin. Microbiol. Antimicrob. 2018, 17, 47. [Google Scholar] [CrossRef]
- Papan, C.; Meyer-Buehn, M.; Laniado, G.; Huebner, J. Evaluation of the multiplex PCR based assay Unyvero implant and tissue infection application for pathogen and antibiotic resistance gene detection in children and neonates. Infection 2018, 47, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Lohr, B.; Pfeifer, Y.; Heudorf, U.; Rangger, C.; Norris, D.E.; Hunfeld, K.P. High prevalence of multidrug-resistant bacteria in Libyan War Casualties admitted to a tertiary care hospital, Germany. Microb. Drug Resist. 2018, 24, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Tafelski, S.; Wagner, L.; Angermair, S.; Deja, M. Therapeutic drug monitoring for colistin therapy in severe multi-resistant Acinetobacter intracerebral abscess: A single case study with high-dose colistin and review of literature. SAGE Open Med Case Rep. 2017, 5, 2050313x17711630. [Google Scholar] [CrossRef] [PubMed]
- Rieber, H.; Frontzek, A.; Pfeifer, Y. Molecular investigation of carbapenem-resistant Acinetobacter spp. from hospitals in North Rhine-Westphalia, Germany. Microb. Drug Resist. 2017, 23, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Reinheimer, C.; Kempf, V.A.; Gottig, S.; Hogardt, M.; Wichelhaus, T.A.; O’Rourke, F.; Brandt, C. Multidrug-resistant organisms detected in refugee patients admitted to a University Hospital, Germany June to December 2015. Eurosurveillance 2016, 21, 30110. [Google Scholar] [CrossRef]
- Pfeifer, Y.; Hunfeld, K.P.; Borgmann, S.; Maneg, D.; Blobner, W.; Werner, G.; Higgins, P.G. Carbapenem-resistant Acinetobacter baumannii ST78 with OXA-72 carbapenemase and ESBL gene blaCTX-M-115. J. Antimicrob. Chemother. 2016, 71, 1426–1428. [Google Scholar] [CrossRef]
- Huenges, K.; Reinecke, A.; Bewig, B.; Haneya, A.; Cremer, J. Lung transplantation in a multidrug-resistant Gram-negative Acinetobacter baumannii-colonized aatient: A case report. Thorac. Cardiovasc. Surg. Rep. 2016, 5, 16–17. [Google Scholar] [CrossRef]
- Molter, G.; Seifert, H.; Mandraka, F.; Kasper, G.; Weidmann, B.; Hornei, B.; Ohler, M.; Schwimmbeck, P.; Kroschel, P.; Higgins, P.G.; et al. Outbreak of carbapenem-resistant Acinetobacter baumannii in the intensive care unit: A multi-level strategic management approach. J. Hosp. Infect. 2016, 92, 194–198. [Google Scholar] [CrossRef]
- Willems, S.; Kampmeier, S.; Bletz, S.; Kossow, A.; Kock, R.; Kipp, F.; Mellmann, A. Whole genome sequencing elucidates epidemiology of nosocomial clusters of Acinetobacter baumannii. J. Clin. Microbiol. 2016, 54, 2391–2394. [Google Scholar] [CrossRef] [PubMed]
- Ghebremedhin, B.; Halstenbach, A.; Smiljanic, M.; Kaase, M.; Ahmad-Nejad, P. MALDI-TOF MS based carbapenemase detection from culture isolates and from positive blood culture vials. Ann. Clin. Microbiol. Antimicrob. 2016, 15, 5. [Google Scholar] [CrossRef] [PubMed]
- Hischebeth, G.T.; Wimmer, M.D.; Molitor, E.; Seifert, H.; Gravius, S.; Bekeredjian-Ding, I. Multidrug resistant Acinetobacter baumannii reaches a new frontier: Prosthetic hip joint infection. Infection 2015, 43, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Dersch, R.; Robinson, E.; Beume, L.; Rauer, S.; Niesen, W.D. Full remission in a patient with catheter-associated ventriculitis due to Acinetobacter baumannii treated with intrathecal and intravenous colistin besides coinfections with other multidrug-resistant bacteria. Neurol. Sci. 2015, 36, 633–634. [Google Scholar] [CrossRef]
- Leistner, R.; Denkel, L.A.; Gastmeier, P.; Werner, G.; Layer, F.; Pfeifer, Y. Prevalence of MRSA and Gram-negative bacteria with ESBLs and carbapenemases in patients from Northern Africa at a German hospital. J. Antimicrob. Chemother. 2015, 70, 3161–3164. [Google Scholar] [CrossRef]
- Hauri, A.M.; Kaase, M.; Hunfeld, K.P.; Heinmüller, P.; Imirzalioglu, C.; Wichelhaus, T.A.; Heudorf, U.; Bremer, J.; Wirtz, A. Meldepflicht für Carbapenem-resistente gramnegative Erreger: Eine Public Health-Priorität? Hyg. Med. 2015, 40, 26–35. [Google Scholar]
- Lahmer, T.; Messer, M.; Schnappauf, C.; Schmidt, A.; Schmid, R.M.; Huber, W. Acinetobacter baumannii sepsis is fatal in medical intensive care unit patients: Six cases and review of literature. Anaesth. Intensive Care 2014, 42, 666–668. [Google Scholar]
- Kleinkauf, N.; Hausemann, A.; Kempf, V.A.J.; Gottschalk, R.; Heudorf, U. Burden of carbapenem-resistant organisms in the Frankfurt/Main Metropolitan Area in Germany 2012/2013—First results and experiences after the introduction of legally mandated reporting. BMC Infect. Dis. 2014, 14, 446. [Google Scholar] [CrossRef]
- Schleicher, X.; Higgins, P.G.; Wisplinghoff, H.; Korber-Irrgang, B.; Kresken, M.; Seifert, H. Molecular epidemiology of Acinetobacter baumannii and Acinetobacter nosocomialis in Germany over a 5-year period (2005-2009). Clin. Microbiol. Infect. 2013, 19, 737–742. [Google Scholar] [CrossRef]
- Kaase, M. Carbapenemasen bei Acinetobacter baumannii: Molekulare epidemiologie in Deutschland und Hinweise zur Diagnostik. Hyg. Med. 2012, 37, 16–19. [Google Scholar]
- Kaase, M. Carbapenemasen bei gramnegativen Erregern in Deutschland. Daten des Nationalen Referenzzentrums fur gramnegative Krankenhauserreger. Bundesgesundheitsblatt Gesundh. Gesundh. 2012, 55, 1401–1404. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, Y.; Wilharm, G.; Zander, E.; Wichelhaus, T.A.; Gottig, S.; Hunfeld, K.P.; Seifert, H.; Witte, W.; Higgins, P.G. Molecular characterization of blaNDM-1 in an Acinetobacter baumannii strain isolated in Germany in 2007. J. Antimicrob. Chemother. 2011, 66, 1998–2001. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.A.; Nenoff, P.; Handrick, W.; Renner, R.; Simon, J.; Treudler, R. Nekrotisierende Fasziitis durch Acinetobacter baumannii: Ein Fallbericht. Hautarzt 2011, 62, 128–130. [Google Scholar] [CrossRef] [PubMed]
- Higgins, P.G.; Schneiders, T.; Hamprecht, A.; Seifert, H. In vivo selection of a missense mutation in adeR and conversion of the novel blaOXA-164 gene into blaOXA-58 in carbapenem-resistant Acinetobacter baumannii isolates from a hospitalized patient. Antimicrob. Agents Chemother. 2010, 54, 5021–5027. [Google Scholar] [CrossRef] [PubMed]
- Aivazova, V.; Kainer, F.; Friese, K.; Mylonas, I. Acinetobacter baumannii infection during pregnancy and puerperium. Arch. Gynecol. Obstet. 2010, 281, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Wadl, M.; Heckenbach, K.; Noll, I.; Ziesing, S.; Pfister, W.; Beer, J.; Schubert, S.; Eckmanns, T. Increasing occurrence of multidrug-resistance in Acinetobacter baumannii isolates from four German University Hospitals, 2002–2006. Infection 2010, 38, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Gottig, S.; Pfeifer, Y.; Wichelhaus, T.A.; Zacharowski, K.; Bingold, T.; Averhoff, B.; Brandt, C.; Kempf, V.A. Global spread of New Delhi metallo-beta-lactamase 1. Lancet. Infect. Dis. 2010, 10, 828–829. [Google Scholar] [CrossRef]
- Pfeifer, Y. Zum Auftreten multiresistenter NDM-1-carbapenemase bildender Escherichia coli und Acinetobacter baumannii in Deutschland. Hyg. Med. 2010, 35, 326–327. [Google Scholar]
- Seifert, H.; Dowzicky, M.J. A longitudinal analysis of antimicrobial susceptibility in clinical institutions in Germany as part of the Tigecycline Evaluation and Surveillance Trial (2004–2007). Chemotherapy 2009, 55, 241–252. [Google Scholar] [CrossRef]
- Kresken, M.; Leitner, E.; Seifert, H.; Peters, G.; von Eiff, C. Susceptibility of clinical isolates of frequently encountered bacterial species to tigecycline one year after the introduction of this new class of antibiotics: Results of the second multicentre surveillance trial in Germany (G-TEST II, 2007). Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 1007–1011. [Google Scholar] [CrossRef]
- Rodloff, A.C.; Leclercq, R.; Debbia, E.A.; Canton, R.; Oppenheim, B.A.; Dowzicky, M.J. Comparative analysis of antimicrobial susceptibility among organisms from France, Germany, Italy, Spain and the UK as part of the Tigecycline Evaluation and Surveillance Trial. Clin. Microbiol. Infect. 2008, 14, 307–314. [Google Scholar] [CrossRef]
- Weyrich, P.; Borgmann, S.; Mayer, F.; Heeg, P.; Riessen, R.; Kotter, I. Fatal multidrug-resistant Acinetobacter baumannii sepsis in a patient with travel history and recent onset of systemic lupus erythematosus: A case report. Int. J. Hyg. Environ. Health 2006, 209, 581–583. [Google Scholar] [CrossRef]
- Schulte, B.; Goerke, C.; Weyrich, P.; Grobner, S.; Bahrs, C.; Wolz, C.; Autenrieth, I.B.; Borgmann, S. Clonal spread of meropenem-resistant Acinetobacter baumannii strains in hospitals in the Mediterranean region and transmission to South-West Germany. J. Hosp. Infect. 2005, 61, 356–357. [Google Scholar] [CrossRef]
- Brauers, J.; Frank, U.; Kresken, M.; Rodloff, A.C.; Seifert, H. Activities of various beta-lactams and beta-lactam/beta-lactamase inhibitor combinations against Acinetobacter baumannii and Acinetobacter DNA group 3 strains. Clin. Microbiol. Infect. 2005, 11, 24–30. [Google Scholar] [CrossRef][Green Version]
- Higgins, P.G.; Wisplinghoff, H.; Stefanik, D.; Seifert, H. Selection of topoisomerase mutations and overexpression of adeB mRNA transcripts during an outbreak of Acinetobacter baumannii. J. Antimicrob. Chemother. 2004, 54, 821–823. [Google Scholar] [CrossRef]
- Frank, U.; Mutter, J.; Schmidt-Eisenlohr, E.; Daschner, F.D. Comparative in vitro activity of piperacillin, piperacillin-sulbactam and piperacillin-tazobactam against nosocomial pathogens isolated from intensive care patients. Clin. Microbiol. Infect. 2003, 9, 1128–1132. [Google Scholar] [CrossRef][Green Version]
- Andermahr, J.; Greb, A.; Hensler, T.; Helling, H.J.; Bouillon, B.; Sauerland, S.; Rehm, K.E.; Neugebauer, E. Pneumonia in multiple injured patients: A prospective controlled trial on early prediction using clinical and immunological parameters. Inflamm. Res. 2002, 51, 265–272. [Google Scholar] [CrossRef]
- Heinemann, B.; Wisplinghoff, H.; Edmond, M.; Seifert, H. Comparative activities of ciprofloxacin, clinafloxacin, gatifloxacin, gemifloxacin, levofloxacin, moxifloxacin and trovafloxacin against epidemiologically defined Acinetobacter baumannii strains. Antimicrob. Agents Chemother. 2000, 44, 2211–2213. [Google Scholar] [CrossRef]
- Robert Koch Institiute (RKI). Infection epidemiological yearbook (2006–2018). Available online: https://www.rki.de/DE/Content/Infekt/Jahrbuch/jahrbuch_node.html (accessed on 5 March 2020).
- Robert Koch Institiute (RKI). Infektionsepidemiologisches Jahrbuch meldepflichtiger Krankheiten für 2018. Reported cases of Acinetobacter infection or colonization with carbapenem insensitivity by category the case definition, Germany, 2017 and 2018. Available online: https://www.rki.de/DE/Content/Infekt/Jahrbuch/Jahrbuch_2018.pdf?__blob=publicationFile (accessed on 5 March 2020).
- Meyer, E.; Schwab, F.; Schroeren-Boersch, B.; Gastmeier, P. Dramatic increase of third-generation cephalosporin-resistant E. coli in German intensive care units: Secular trends in antibiotic drug use and bacterial resistance, 2001 to 2008. Crit. Care Lond. Engl. 2010, 14, R113. [Google Scholar] [CrossRef]
- Pfeifer, Y.; Cho, S.; Higgins, P.; Fahr, A.; Wichelhaus, T.; Hunfeld, K.; Martin, M.; Witte, W. Molecular characterisation and outbreak analysis of multidrug-resistant Acinetobacter baumannii from German hospitals. Clin. Microbiol. Infect. 2010, 6 (Suppl. S2), P797. [Google Scholar] [CrossRef]
- Zordan, S.; Prenger-Berninghoff, E.; Weiss, R.; van der Reijden, T.; van den Broek, P.; Baljer, G.; Dijkshoorn, L. Multidrug-resistant Acinetobacter baumannii in veterinary clinics, Germany. Emerg. Infect. Dis. 2011, 17, 1751–1754. [Google Scholar] [CrossRef]
- Hembach, N.; Schmid, F.; Alexander, J.; Hiller, C.; Rogall, E.T.; Schwartz, T. Occurrence of the mcr-1 colistin resistance gene and other clinically relevant antibiotic resistance genes in microbial populations at different municipal wastewater treatment plants in Germany. Front. Microbiol. 2017, 8, 1282. [Google Scholar] [CrossRef]
- Matuschek, E.; Ahman, J.; Webster, C.; Kahlmeter, G. Antimicrobial susceptibility testing of colistin—Evaluation of seven commercial MIC products against standard broth microdilution for Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter spp. Clin. Microbiol. Infect. 2018, 24, 865–870. [Google Scholar] [CrossRef]
- Fritzenwanker, M.; Imirzalioglu, C.; Herold, S.; Wagenlehner, F.M.; Zimmer, K.P.; Chakraborty, T. Treatment options for carbapenem-resistant Gram-negative infections. Dtsch. Arztebl. Int. 2018, 115, 345–352. [Google Scholar] [CrossRef]
- Valencia, R.; Arroyo, L.A.; Conde, M.; Aldana, J.M.; Torres, M.J.; Fernandez-Cuenca, F.; Garnacho-Montero, J.; Cisneros, J.M.; Ortiz, C.; Pachon, J.; et al. Nosocomial outbreak of infection with pan-drug-resistant Acinetobacter baumannii in a tertiary care university hospital. Infect. Control Hosp. Epidemiol. 2009, 30, 257–263. [Google Scholar] [CrossRef]
- Ballouz, T.; Aridi, J.; Afif, C.; Irani, J.; Lakis, C.; Nasreddine, R.; Azar, E. Risk Factors, Clinical presentation and outcome of Acinetobacter baumannii bacteremia. Front. Cell. Infect. Microbiol. 2017, 7, 156. [Google Scholar] [CrossRef]
- Wareth, G.; Neubauer, H.; Sprague, L.D. Acinetobacter baumannii—A neglected pathogen in veterinary and environmental health in Germany. Vet. Res. Commun. 2019, 43, 1–6. [Google Scholar] [CrossRef]
- Cho, G.S.; Li, B.; Rostalsky, A.; Fiedler, G.; Rosch, N.; Igbinosa, E.; Kabisch, J.; Bockelmann, W.; Hammer, P.; Huys, G.; et al. Diversity and antibiotic susceptibility of Acinetobacter strains from milk powder produced in Germany. Front. Microbiol. 2018, 9, 536. [Google Scholar] [CrossRef]
- Wilharm, G.; Skiebe, E.; Higgins, P.G.; Poppel, M.T.; Blaschke, U.; Leser, S.; Heider, C.; Heindorf, M.; Brauner, P.; Jackel, U.; et al. Relatedness of wildlife and livestock avian isolates of the nosocomial pathogen Acinetobacter baumannii to lineages spread in hospitals worldwide. Environ. Microbiol. 2017, 19, 4349–4364. [Google Scholar] [CrossRef]
- Klotz, P.; Higgins, P.G.; Schaubmar, A.R.; Failing, K.; Leidner, U.; Seifert, H.; Scheufen, S.; Semmler, T.; Ewers, C. Seasonal occurrence and carbapenem susceptibility of bovine Acinetobacter baumannii in Germany. Front. Microbiol. 2019, 10, 272. [Google Scholar] [CrossRef]
- Szabados, F.; Tix, H.; Anders, A.; Kaase, M.; Gatermann, S.G.; Geis, G. Evaluation of species-specific score cutoff values of routinely isolated clinically relevant bacteria using a direct smear preparation for matrix-assisted laser desorption/ionization time-of-flight mass spectrometry-based bacterial identification. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1109–1119. [Google Scholar] [CrossRef]
- Osei Sekyere, J.; Govinden, U.; Bester, L.A.; Essack, S.Y. Colistin and tigecycline resistance in carbapenemase-producing Gram-negative bacteria: Emerging resistance mechanisms and detection methods. J. Appl. Microbiol. 2016, 121, 601–617. [Google Scholar] [CrossRef]
- Gil-Marques, M.L.; Moreno-Martinez, P.; Costas, C.; Pachon, J.; Blazquez, J.; McConnell, M.J. Peptidoglycan recycling contributes to intrinsic resistance to fosfomycin in Acinetobacter baumannii. J. Antimicrob. Chemother. 2018, 73, 2960–2968. [Google Scholar] [CrossRef]
- Remschmidt, C.; Schneider, S.; Meyer, E.; Schroeren-Boersch, B.; Gastmeier, P.; Schwab, F. Surveillance of antibiotic use and resistance in intensive care units (SARI). Dtsch. Arztebl. Int. 2017, 114, 858–865. [Google Scholar] [CrossRef]
- Scaife, W.; Young, H.K.; Paton, R.H.; Amyes, S.G. Transferable imipenem-resistance in Acinetobacter species from a clinical source. J. Antimicrob. Chemother. 1995, 36, 585–586. [Google Scholar] [CrossRef]
- Guerra, B.; Fischer, J.; Helmuth, R. An emerging public health problem: Acquired carbapenemase-producing microorganisms are present in food-producing animals, their environment, companion animals and wild birds. Vet. Microbiol. 2014, 171, 290–297. [Google Scholar] [CrossRef]
- Eigenbrod, T.; Reuter, S.; Gross, A.; Kocer, K.; Gunther, F.; Zimmermann, S.; Heeg, K.; Mutters, N.T.; Nurjadi, D. Molecular characterization of carbapenem-resistant Acinetobacter baumannii using WGS revealed missed transmission events in Germany from 2012-15. J. Antimicrob. Chemother. 2019, 74, 3473–3480. [Google Scholar] [CrossRef]
- Jones, C.L.; Clancy, M.; Honnold, C.; Singh, S.; Snesrud, E.; Onmus-Leone, F.; McGann, P.; Ong, A.C.; Kwak, Y.; Waterman, P.; et al. Fatal outbreak of an emerging clone of extensively drug-resistant Acinetobacter baumannii with enhanced virulence. Clin. Infect. Dis. 2015, 61, 145–154. [Google Scholar] [CrossRef]
- Higgins, P.G.; Dammhayn, C.; Hackel, M.; Seifert, H. Global spread of carbapenem-resistant Acinetobacter baumannii. J. Antimicrob. Chemother. 2010, 65, 233–238. [Google Scholar] [CrossRef]
- Wisplinghoff, H.; Hippler, C.; Bartual, S.G.; Haefs, C.; Stefanik, D.; Higgins, P.G.; Seifert, H. Molecular epidemiology of clinical Acinetobacter baumannii and Acinetobacter genomic species 13TU isolates using a multilocus sequencing typing scheme. Clin. Microbiol. Infect. 2008, 14, 708–715. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).