Abstract
Lyme borreliosis (LB), caused by spirochetes of the Borrelia burgdorferi sensu lato (s.l.) complex, is one of the most common vector-borne zoonotic diseases in Europe. Knowledge about the enzootic circulation of Borrelia pathogens between ticks and their vertebrate hosts is epidemiologically important and enables assessment of the health risk for the human population. In our project, we focused on the following vertebrate species: European hedgehog (Erinaceus europaeus), Northern white-breasted hedgehog (E. roumanicus), Eurasian red squirrel (Sciurus vulgaris), and Common blackbird (Turdus merula). The cadavers of accidentally killed animals used in this study constitute an available source of biological material, and we have confirmed its potential for wide monitoring of B. burgdorferi s.l. presence and genospecies diversity in the urban environment. High infection rates (90% for E. erinaceus, 73% for E. roumanicus, 91% for S. vulgaris, and 68% for T. merula) were observed in all four target host species; mixed infections by several genospecies were detected on the level of individuals, as well as in particular tissue samples. These findings show the usefulness of multiple tissue sampling as tool for revealing the occurrence of several genospecies within one animal and the risk of missing particular B. burgdorferi s.l. genospecies when looking in one organ alone.
1. Introduction
Lyme borreliosis (LB) is one of the most abundant vector-borne diseases in Europe, with a purported annual number of human disease cases between 65,000 and 85,000 [1,2]. Only rough estimates of the LB incidence in Europe are available because the reporting systems differ among countries, and LB is not a compulsorily notifiable disease in all of them. However, the incidence numbers seem to be underestimated as results of a more recent study calculate ~232,000 new cases per year in Western Europe alone, while several countries with the highest incidence rates on the continent were not included [3,4]. The disease is caused by spirochetes of the Borrelia burgdorferi sensu lato (s.l.) complex [5]. Today, 22 named B. burgdorferi s.l. genospecies are known and well established around the world. As new species and variants are continuously being recognized, the current number of described taxa is apparently not final.
In terms of human sensitivity to B. burgdorferi s.l., the complex of 22 Borrelia genospecies can be divided into two groups:
(a) 10 genospecies with pathogenic potential that have been detected in or isolated from humans: B. afzelii [6], B. bavariensis [7], B. bissettii [8], B. burgdorferi sensu stricto (s.s.) [9], B. garinii [9], B. kurtenbachii [10], B. lusitaniae [11], B. mayonii [12], B. spielmanii [13], and B. valaisiana [14].
(b) Another 12 genospecies that have not been detected in or isolated from humans yet: B.americana [15], B. andersonii [16], B. californiensis [17], B. carolinensis [18], B. chilensis [19], B. finlandensis [20], B. japonica [21], B. lanei [22], B. tanukii [23], B. turdi [23], B. sinica [24], and B. yangtzensis [25].
In Europe, B. burgdorferi s.s., B. afzelii, B. garinii, and B. bavariensis are responsible for the vast majority of human disease cases [5]. Other genospecies, like B. bissettii, B. spielmanii, B. kurtenbachii, B. valaisiana, and B. lusitaniae may also be involved in human infections [26,27,28,29]. Humans become infected when an infectious tick feeds on their blood. In Europe, the common tick (Ixodes ricinus) is the most prevalent vector, ensuring the circulation of the pathogen among its natural vertebrate hosts, as well as transmission to humans [30]. Other tick species may also participate in the natural circulation of the spirochetes (e.g., I. hexagonus) [31,32].
Forests, preferably mixed and deciduous, are considered typical I. ricinus habitats, and, consequently, they are natural circulation areas for tick-borne pathogens. Nevertheless, in the last decade, attention has also been drawn to the occurrence of ticks and tick-borne pathogens in urban and peri-urban areas [33,34]. Human exposure to tick bites in urban areas may be high, thus representing a relevant health risk. People entering urban tick habitat do not pay as much attention to tick bite prevention as when visiting woodland areas; the elderly, disabled persons, and young children visit parks more frequently than woodland areas [35].
Considering the generally low mobility of ticks, birds are important hosts for tick dispersal [36] to urban habitats (i.e., gardens, city parks, cemeteries), which then provide a suitable environment for the existence of permanent tick populations. One of the important prerequisites for tick survival in urban ecosystems is the presence of vertebrate hosts suitable for feeding of all developmental stages. Synurbic populations of small rodents and birds serve as a blood-meal source for larval and nymphal ticks. Adult ticks feed on larger rodents (e.g., squirrels), insectivores (e.g., hedgehogs), or domestic pets; however, lagomorphs and roe deer may also be able to thrive in urban or peri-urban areas [34,37]. There are numerous studies reporting the occurrence of ticks in urban green spaces carrying various tick-borne human pathogens [32,33,38,39,40,41,42,43,44,45]. Nevertheless, the role of wildlife as potential reservoir hosts of tick-borne pathogens in urban areas is rarely addressed.
In our project, we focused on four vertebrate species: The European hedgehog (Erinaceus europaeus), the Northern white-breasted hedgehog (E. roumanicus), the Eurasian red squirrel (Sciurus vulgaris), and the Common blackbird (Turdus merula), which tend towards synurbization, are frequently heavily infested by ticks, and are important reservoir hosts of B. burgdorferi s.l. [46,47,48,49,50,51,52,53]. We used randomly found cadavers of accidentally killed animals or handicapped animals that had died in rescue centers, as they constitute an easy to collect and valuable source of biological material for tick-borne pathogen monitoring [44].
The main aims of the study were: (i) to assess the prevalence and diversity of B. burgdorferi s.l. genospecies in the four target species and (ii) to evaluate the potential of cadavers of accidentally killed vertebrates as a source of biological material for monitoring of zoonotic pathogens in urban environments.
2. Materials and Methods
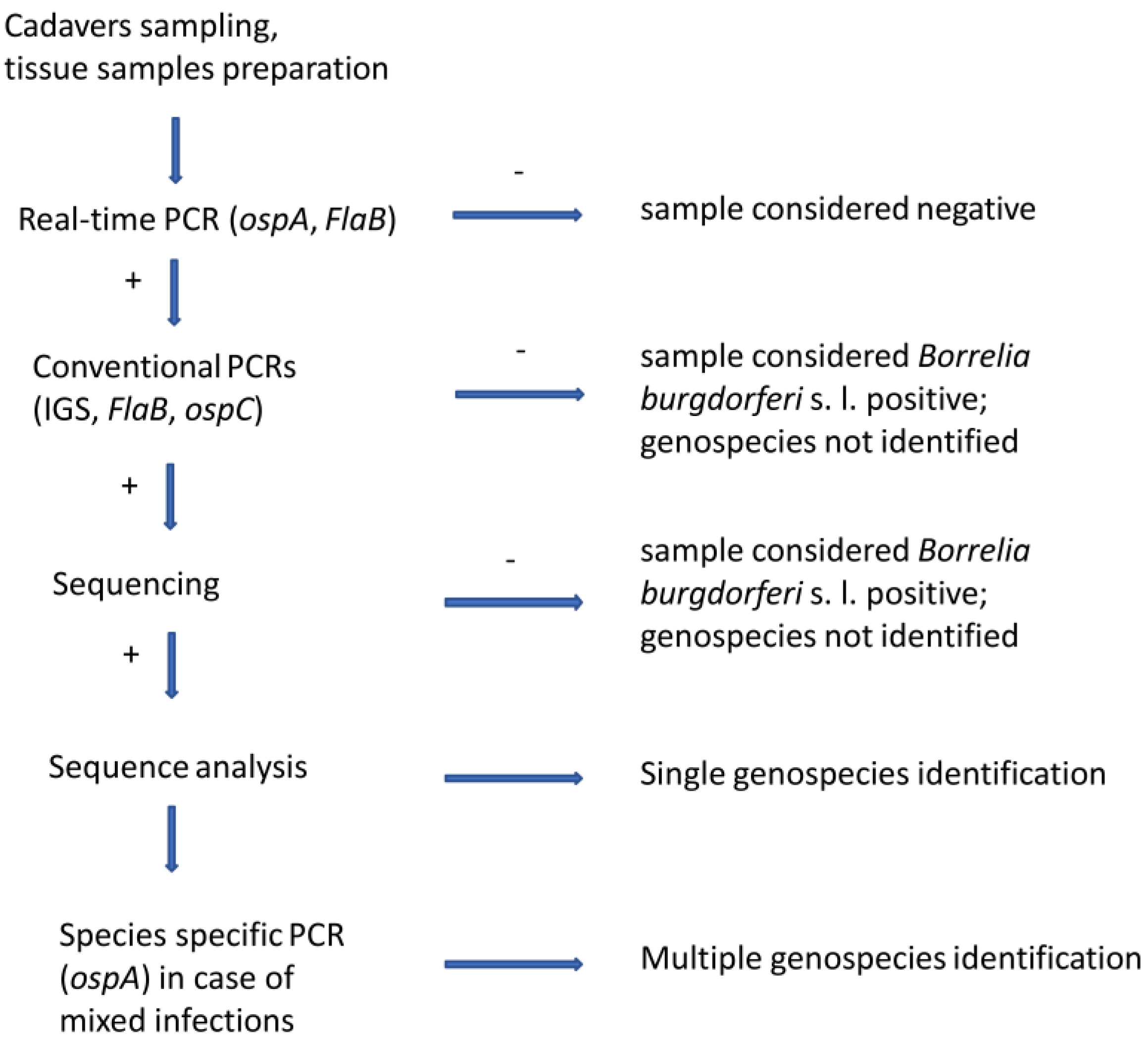
The methodology used in this study consist of cadavers sampling, dissections, tissue samples preparation, DNA isolation, multiple real-time PCR, and a set of conventional PCRs (Figure 1).
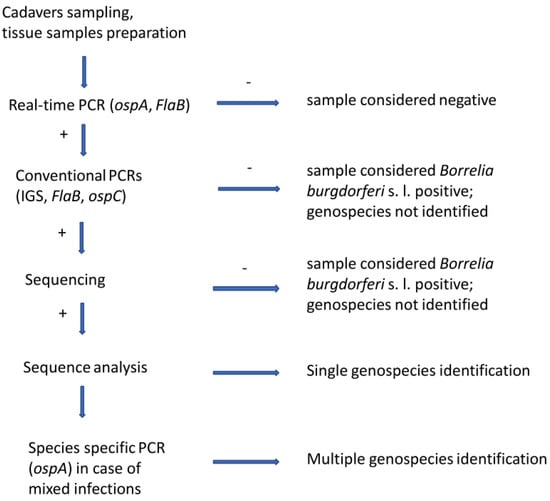
Figure 1.
Methodology scheme. + means positive results; − means negative results.
2.1. Hosts, Dissections, Sampling
European hedgehog (E. europaeus), Northern white-breasted hedgehog (E. roumanicus), Eurasian red squirrel (S. vulgaris), and Common blackbird (T. merula) cadavers were used as the source of biological material. All of these animals were found dead, mostly as road-kill, killed by other animals (above all by dogs and cats), or from glass collisions (birds); or died in animal rescue centers. The cadavers were kept at −20°C until further processing. Thawing of cadaver before necropsy took 4–18 h, based on the cadaver size and thawing temperature, which was 8–20 °C. After thawing, the grade of autolysis was determined (Table S1). Only cadavers with a low (1A,1B) or intermediate (2) grade of autolysis were utilized.
The species of a given animal was identified based on characteristic morphological features [54,55,56]. However, species determination of hedgehogs based solely on morphological characteristics might be biased, especially in road-kill. Therefore, we confirmed the morphological identification using the molecular method based on mitochondrial control region analysis [57,58].
Each cadaver was weighted, its foot length was measured, and an age category was assigned, either juvenile (presence of primary dentice in mammals or juvenile feathers in birds), or adult. The gender of the animal was determined when external or internal sexual organs were present in the cadaver. The ectoparasitic infestation was recorded only as presence/absence due to the bias resulting from differences in the stretch of time from animal death to cadaver collection and the possibility of antiparasitic treatment of individuals acquired from animal rescue centers.
The animal tissues were removed under sterile conditions. Each organ was taken out with sterile surgical instruments treated with PCR Clean™ (Minerva Biolabs, Berlin, Germany) to eliminate DNA and RNA contamination from the surface. A piece of the tissue (~5 × 5 mm) was placed into a 2 mL Eppendorf tube, where it was cut into ~1-mm pieces. We collected ear tissue (or skin in the case of birds or when the ears were absent) and muscle, lung, blood, liver, spleen, urinary bladder, kidney, and brain tissue. A blood coagulum or liquid blood was obtained from the heart or thoracic cavity of the cadaver with a sterile Pasteur pipette, and a volume of 50–500 µL of blood was placed into a 2 mL Eppendorf tube. To improve the preservation of DNA and RNA (used for the detection of flaviviruses [59]), 1 mL of RLT buffer (Qiagen, Hilden, Germany) was added to each Eppendorf tube with the sample of blood or tissue and stored at −70 °C [60] until further analyses were conducted.
2.2. DNA Isolation
Tissue samples were homogenized in RLT buffer (Qiagen, Hilden, Germany) with the addition of beta-mercaptoethanol (10 µL of 14.3M beta-ME per 1 mL of RLT buffer) using sterile stainless-steel beads (Qiagen, Hilden, Germany) in a Tissue Lyzer II. Briefly, samples of ear, skin, muscle, lung, liver, spleen, urinary bladder, kidney, and brain tissue were prepared as 30% (w/v) suspensions. After homogenization, 20 µL of proteinase K (Qiagen, Hilden, Germany) was added, and the samples were incubated for 30 min at 57 °C. The lysate was cleared by centrifugation, and the supernatant was collected in a clean, sterile microtube. The samples of blood (10–100 µL depending on whether it was nucleated or nonnucleated blood and the sample volume obtained from a particular animal) were resuspended in 220 µL of sterile PBS, and the solution was used for DNA isolation using a DNeasy Blood and Tissue kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. The elution volume was 200 µL.
2.3. Screening of the Samples through Multiplex Real-Time PCR
All PCR and real-time PCR reactions were set up in a separate area with all appropriate precautions (separated supplies, equipment, and personal safety items, pre- and post-amplification activities). The extracted DNA samples were screened for B. burgdorferi s.l. using multiplex real-time PCR, which also enabled the detection and identification of B. miyamotoi. For the detection of B. burgdorferi s.l., two primer pairs and two different probes were used (Table S2), targeting outer surface protein (ospA) and flagellin B (FlaB) genes [61]. For the real-time PCR, an iQ Multiplex Powermix PCR reagent kit containing iTaq DNA polymerase (Bio-Rad Laboratories, Hercules, CA, USA) was used. The real-time PCR was performed in a LightCycler 480 Real-Time PCR System (Roche, Base, Switzerland) using the following program: an initial activation of the iTaq DNA polymerase at 95 °C for 5 min, 60 cycles of a 5 s denaturation at 95 °C, followed by a 35 s annealing-extension step at 60 °C (single point measurement at 60 °C), and a cooling cycle of 37 °C for 20 s. The analysis was performed using second derivative calculations for Cp (crossing point) values. A color compensation was conducted for the overflow of fluorescence from the dyes that were used. Curves were assessed visually in LightCycler 480 software. Samples with a positive score for one or both targets (ospA; FlaB) for B. burgdorferi s.l. were considered positive.
2.4. Conventional PCRs and Sequencing
All samples which were positive for B. burgdorferi s.l. according to real-time PCR were subsequently analyzed by three different conventional PCRs, targeting portions of the 5S-23S (rrfA–rrlB) ribosomal RNA intergenic spacer region (IGS), and genes encoding flagellin B (FlaB) and outer surface protein C (ospC), where nested PCR was performed (Table S2). A MasterTaq kit (Eppendorf, Hamburg, Germany) containing recombinant Taq DNA polymerase from Escherichia coli DH1 and a 5× TaqMaster PCR enhancer was used for the amplification of the spirochete sequences. A negative control (no template) and positive control (B. burgdorferi sensu stricto, strain B31 DNA) was added to each amplification reaction. The PCR products were separated using gel electrophoresis, cleaned with ExoSAP-IT™ PCR Product Cleanup Reagent (Applied Biosystems™, Waltham, MA, USA), and Sanger sequencing was provided by BaseClear (Leiden, Netherlands) using internal nested PCR primers. The sequence chromatograms were checked visually and, afterwards, used for the B. burgdorferi s.l. genospecies identification as a comparison to sequences of known genospecies from the NCBI GenBank database.
2.5. Species-Specific PCR
To identify mixed infections with multiple genospecies of B. burgdorferi s.l. complex, the real-time PCR positive samples were screened using species-specific PCR. Primers designed to target the outer surface protein A (ospA) gene of B. burgdorferi s.s. (primer set GI), B. garinii (primer set GII), and B. afzelii (primer set GIII) (Table S2) described by Reference [62] were used with the following modification: PCR was conducted in two steps, amplification and re-amplification. A HotStarTaq Plus Master kit (Qiagen, Hilden, Germany) was used for amplification of species-specific fragments of ospA genes. After the completion of primary amplification, 5 µL of the resultant mixture was transferred into a fresh PCR tube with the HotStarTaq Plus Master mixture and the same PCR primers (20 µL final volume), and re-amplification was conducted under the same conditions but only for 25 cycles. A negative control (no template) and positive control (B. carolinensis DNA) was added to each amplification run. The final PCR products were separated by agarose gel electrophoresis. The genospecies of B. burgdorferi s.l. were determined based on the difference in amplicon size. The use of the modified technique described above allows detection of the presence of multiple spirochete species in a single sample.
2.6. Statistical Analyses
Differences in prevalence rates were tested using contingency tables and chi-square or Fisher’s exact test. To analyze genospecies distribution among host species and tissues, we used the asymptotic (permutation-based) generalized Cochran-Mantel-Haenszel (CMH) test, as described in Reference [63], and implemented in R by the coin package [64]; the CMH statistic tests conditional independence in three-way contingency tables. As all the tests performed rejected the null hypothesis at high p-values, we complemented the analysis with post-hoc McNemar’s tests (to compare proportions in partial tables) with continuity correction and Holm adjusted p-values. Differences with p < 0.05 were considered statistically significant.
3. Results
A total of 157 cadavers (60 European hedgehogs, 15 northern white-breasted hedgehogs, 22 Eurasian red squirrels, and 60 common blackbirds) were collected and dissected, resulting in a total of 862 tissue samples (Table S3). All samples were screened for the presence of Borrelia miyamotoi and B. burgdorferi s.l. DNA by multiplex real-time PCR.
3.1. Prevalence of B. miyamotoi
B. miyamotoi DNA was detected in one or more tissues in three European hedgehog specimens (5.0%) and three Eurasian red squirrel specimens (13.6%) (Table S3).
3.2. Prevalence of B. burgdorferi s.l.
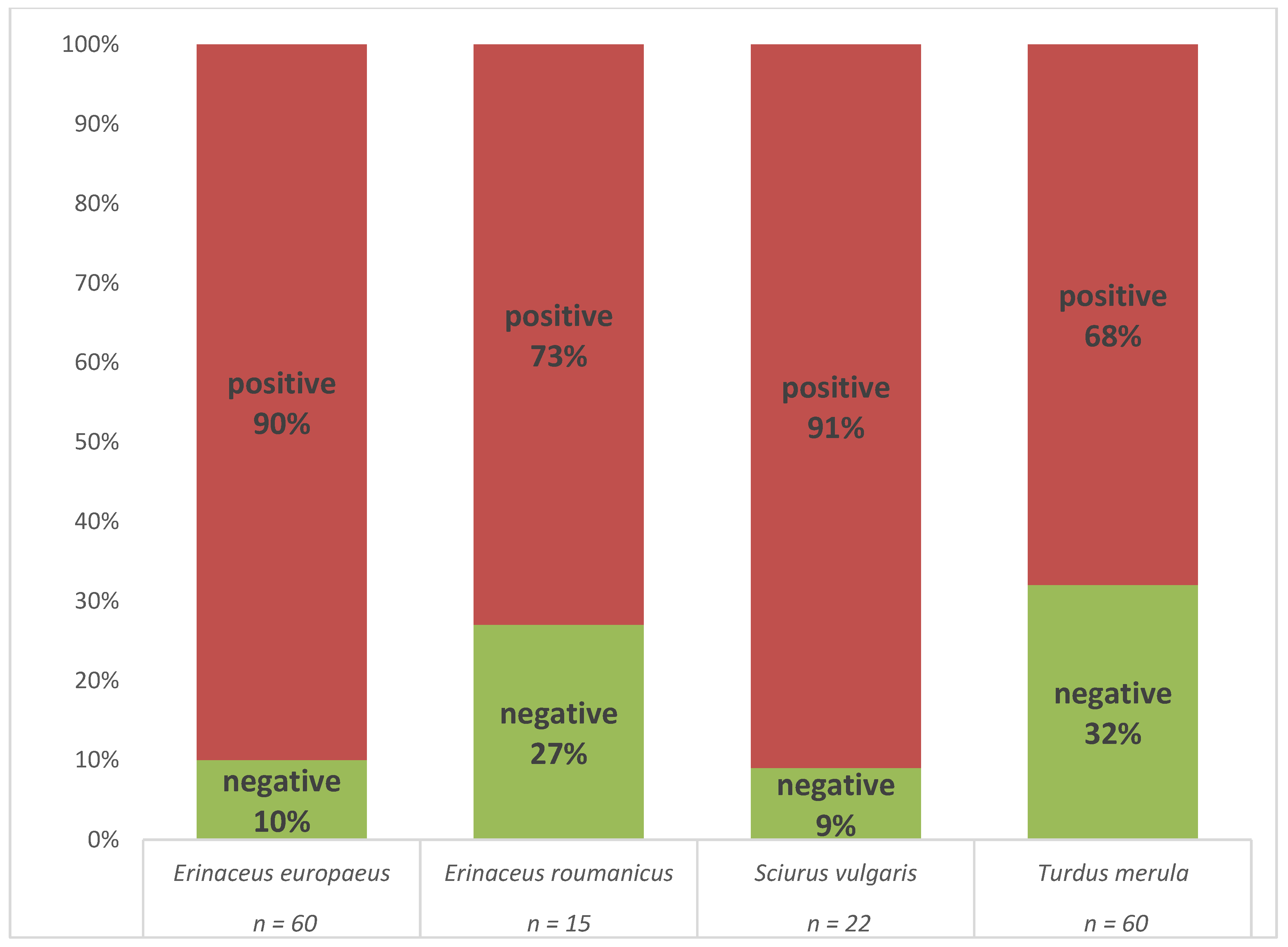
The presence of B. burgdorferi s.l. DNA was confirmed by real-time PCR in 126 (80.2%) of the individuals (Table S4). An individual was considered positive if the spirochete was detected in at least one of the tested tissue samples. A high proportion of positive animals was observed in all four host species (Figure 2).
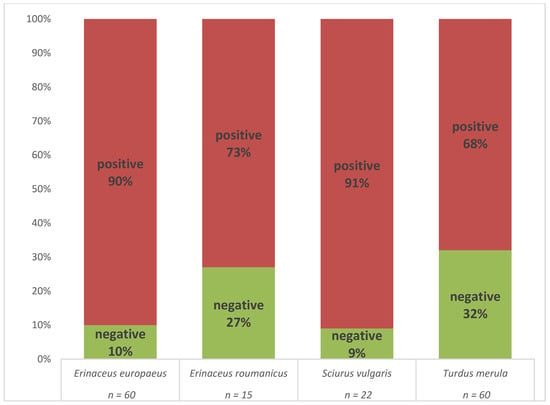
Figure 2.
Comparison of B. burgdorferi s.l. prevalence in four host species as assessed by multiplex real-time PCR.
3.3. Occurrence of Borrelia burgdorferi s.l. in Specific Tissues
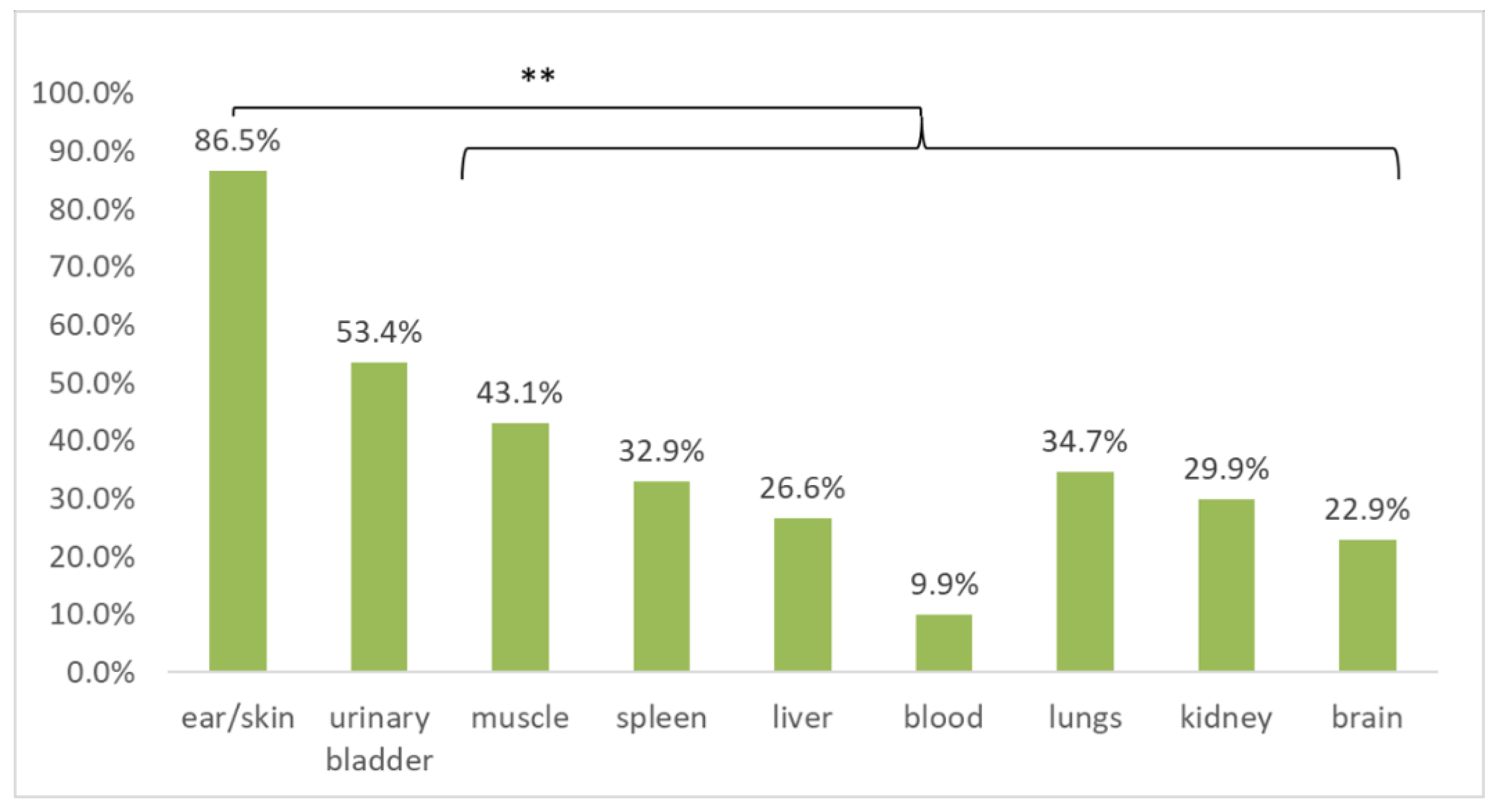
Multiple tissues were sampled from each cadaver. Although some of the tissues were commonly missing, all the tissue samples available from each animal were screened for B. burgdorferi s.l. DNA in order to compare the detection efficiency (Table 1) and to investigate tissue tropism. When compared for all host species, ear/skin (treated as equivalent for this analysis) tissue samples were significantly more frequently positive than all of the other tested tissues (p < 0.01; McNemar’s chi-square test with continuity correction, Holm adjusted p-values), with the exception of urinary bladder tissue (Figure 3).

Table 1.
Comparison of different tissues for the detection efficiency of Borrelia burgdorferi s.l. DNA by real-time PCR. The percentage represents the portion of positive samples of a particular tissue from the total number of B. burgdorferi s.l. positive individuals (with the tissue available).
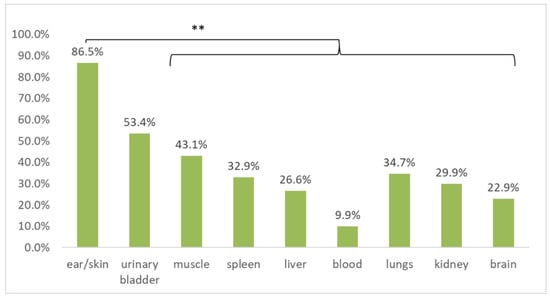
Figure 3.
Comparison of B. burgdorferi sensu lato prevalence as assessed by multiplex real-time PCR in different tissue samples collected in cadavers of European hedgehogs, Northern white-breasted hedgehogs, Eurasian red squirrels, and Common blackbirds. ** indicates statistically significant differences (p < 0.01; McNemar’s chi-square test with continuity correction, Holm adjusted p-values).
Similar trends were observed when tissues within the host species were compared. In a pairwise comparison, B. burgdorferi s.l. DNA was significantly more frequently detected in the skin (87.8% positive of 41 tested) than in other tissues of blackbirds (p < 0.0001). The same was true for ear tissue in European hedgehogs except for muscle and urinary bladder tissue (p < 0.05; McNemar’s chi-square test with continuity correction, Holm adjusted p-values). European hedgehog blood was significantly less infected than ear or urinary bladder tissue (p < 0.05; McNemar’s chi-square test with continuity correction, Holm adjusted p-values). In the case of squirrels, significant differences were found only between ear tissue compared to blood and kidney tissue (p < 0.05; McNemar’s chi-square test with continuity correction, Holm adjusted p-values). No significant differences were found among the tissues of Northern white-breasted hedgehogs.
3.4. Identification of Borrelia burgdorferi s.l. Genospecies
All real-time PCR positive samples were further investigated by four different conventional PCRs to identify the B. burgdorferi s.l. genospecies including mixed infections. A relatively high rate of mixed infections was found on the level of individuals (= different genospecies found in different tissues of the same animal) but also in specific tissue samples. The combinations of genospecies found in mixed infections, as well as their frequencies, are shown in Tables S4–S8. The highest genospecies diversity was observed in the European hedgehog, either as mono-infections or as part of multiple infections. No apparent associations of genospecies were found in the mixed infections (although the relatively low total numbers prevented proper statistical analysis). Unexpectedly, B. bissettii/carolinensis DNA was detected in several samples (GenBank accession numbers: MW297142–297147). None of the methods allowed discrimination between these two genospecies as the DNA was found only in samples with mixed infections, and short ospA sequences obtained from species-specific PCR were not sufficient for distinguishing due to the close genetic relatedness of these two Borrelia genospecies.
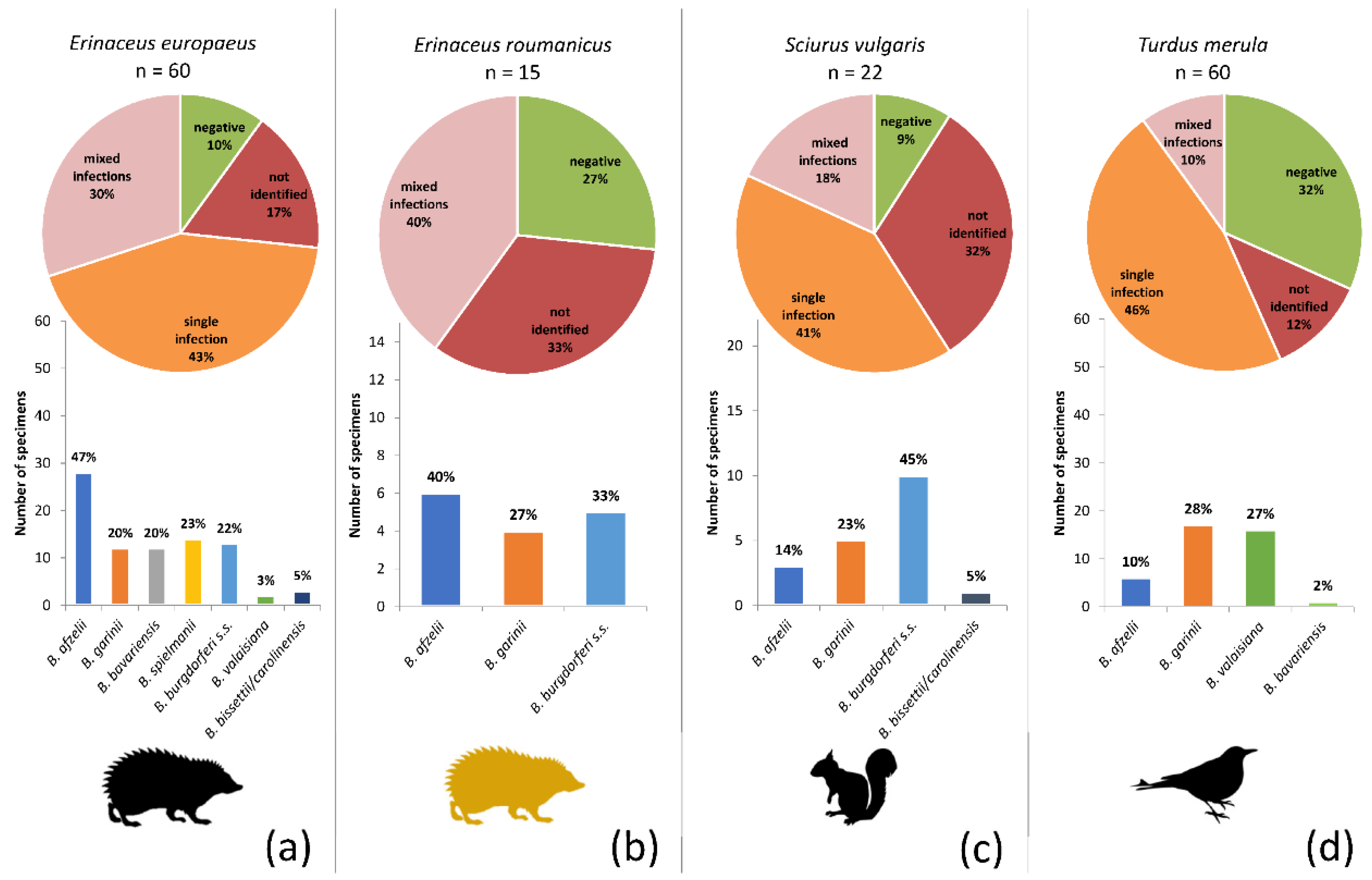
The proportional representation of the genospecies differed significantly among the hosts (Figure 4) (Asymptotic General Independence Test p < 0.0001). Particularly in the case of European hedgehogs, B. afzelii was significantly more frequent than any other genospecies (p < 0.0001; McNemar’s chi-square test with continuity correction, Holm adjusted p-values), and B. bissetii/carolinensis and B. valaisiana were less frequent than all but B. bavariensis and B. garinii (p < 0.05; McNemar’s chi-square test with continuity correction, Holm adjusted p-values). No significant differences were found in a pairwise comparison of the genospecies infecting the Northern white-breasted hedgehog. B. burgdorferi s.s. was significantly more frequent in squirrels than B. bissettii/carolinensis (p < 0.05; McNemar’s chi-square test with continuity correction, Holm adjusted p-values), and B. bavariensis was significantly less frequently found in blackbirds than B. garinii and B. valaisiana (p < 0.05; McNemar’s chi-square test with continuity correction, Holm adjusted p-values).
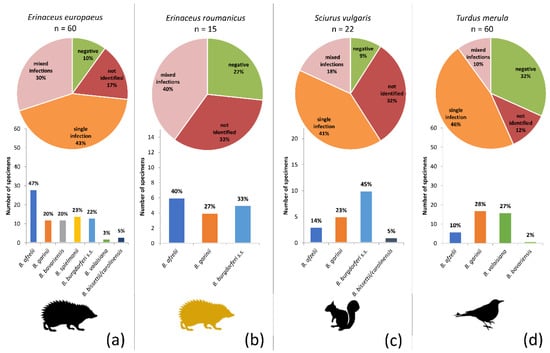
Figure 4.
Prevalence of Borrelia burgdorferi s.l. genospecies among the four target host species: (a) European hedgehog, (b) Northern white-breasted hedgehog, (c) Eurasian red squirrel, and (d) Common blackbird. The pie charts indicate the proportions of individuals not infected (green), infected by a single genospecies (orange), infected by multiple genospecies (pink) and those samples that were positive in B. burgdorferi s.l. real-time PCR, but in which genospecies identification was not possible (dark red). The bar charts show the proportions of the genospecies as found by a combination of sequencing and species-specific PCR (summed for single and multiple infections).
3.5. Comparison of Occurrence of Borrelia burgdorferi s.l. Genospecies in Investigated Tissues
The overall distribution of B. afzelii, B. garinii, and B. valaisiana among the tissues differs significantly (Asymptotic General Independence Test, p < 0.0001) in general analysis ignoring the host species (Table 2). In a pairwise comparison, B. afzelii DNA was significantly more frequently found in ear/skin and muscle tissues than in all other tissues except urinary bladder and kidney tissue (p < 0.05, McNemar’s chi-square test with continuity correction, Holm adjusted p-values). In the case of B. garinii, ear/skin tissue was significantly more frequently infected than blood and brain tissue (p < 0.05, McNemar’s chi-square test). In the case of B. valaisiana, ear/skin tissue was significantly more infected than blood, liver, and muscle tissue (p < 0.05, McNemar’s chi-square test).

Table 2.
Distribution of B. burgdorferi sensu lato genospecies among the tissues of European hedgehog, Northern white-breasted hedgehog, Eurasian red squirrel, and Common blackbird.
Distribution of B. burgdorferi s.l. genospecies also differs among the tissues within each of the four host species (Table 3). In a pairwise comparison (McNemar´s test), significant differences were found only for European hedgehog ear and muscle tissue and Common blackbird skin tissue. In European hedgehog ear tissue, B. afzelii was significantly more frequently detected than any other species except for B. spielmanii and B. bavariensis (p < 0.01, McNemar’s chi-square test with continuity correction, Holm adjusted p-values). The same genospecies were also more frequent in European hedgehog muscle tissue than all other genospecies except for B. bavariensis, B. burgdorferi s.s., and B. bissettii/carolinensis (p < 0.01, McNemar’s chi-square test with continuity correction, Holm adjusted p-values). In Common blackbird skin tissue, B. garinii and B. valaisiana were significantly more frequent than all other genospecies except for B. afzelii (p < 0.01, McNemar’s chi-square test with continuity correction, Holm adjusted p-values).

Table 3.
Distribution of B. burgdorferi sensu lato genospecies among the different tissues in European hedgehog (E. europaeus), Northern white-breasted hedgehog (E. roumanicus), Eurasian red squirrel (S. vulgaris), and Common blackbird (T. merula).
4. Discussion
Borrelia miyamotoi, a causative agent of human nonspecific febrile illness, has been previously detected in various host species, mostly rodents, but also other vertebrates, e.g., passerine birds and hedgehogs (reviewed in Reference [65]). Although our study was primarily focused on B. burgdorferi s.l., using a multiplex real-time PCR protocol that includes B. miyamotoi specific primers and probe, we detected the DNA of this Borrelia in a few European hedgehog and Eurasian red squirrel individuals. These findings support the previous reports on B. myiamotoi in Europe [66,67,68] and suggest that squirrels and hedgehogs contribute to the natural transmission cycle of this pathogen.
Although three vertebrate species chosen for this study (E. europaeus, S. vulgaris, T. merula) have been listed as reservoir hosts of B. burgdorferi s.l. for a long time [32,49,50], and European hedgehog was reported as a potential reservoir host [52], there is only a limited number of studies focused on the prevalence of B. burgdorferi s.l. or genospecies diversity in these hosts. Moreover, most of the studies (especially in the case of blackbirds) investigated the attached ticks not the host blood or tissues. Using cadavers as a source of biological material of different tissues enabled us to detect various genospecies of B. burgdorferi s.l. in all target host species and gain insight into prevalence, tissue specificity patterns and mixed infections.
We detected DNA of four different genospecies in Eurasian red squirrels: B. burgdorferi s.s., B. garinii, B. afzelii, and, for the first time, also B. bissettii/carolinensis in one specimen. B. burgdorferi s.s., B. afzelii, and B. garinii have been repeatedly observed in Eurasian red squirrels [49,69,70,71,72], and Borrelia genospecies mixed infections in analyses based on skin or ear tissue samples have also been reported previously [49,70]. In comparison to most of the published studies, we observed a high prevalence (91%), similar to results from Norway (88%) where a real-time PCR detection method and a similar number of tested animals were used [72]. B. burgdorferi s.l. DNA was most frequently found in ear/skin tissue (with significant differences compared to blood and kidney tissue). However, screening of multiple host tissues revealed the presence of mixed infections, and, among them, DNA of B. bissettii/carolinensis was also detected.
The European hedgehog is a well-recognized reservoir host for several Borrelia genospecies: B. afzelii, B. spielmanii, B. bavariensis, B. garinii, and B. burgdorferi s.s. have been repeatedly reported [32,52,73,74,75], and the role of European hedgehog in Borrelia transmission has also been proven experimentally [32]. The vast majority of European hedgehog specimens tested in our study (90%) were found to be positive for at least one of the previously reported Borrelia genospecies (the infection rate was higher when compared to other studies). DNA of B. valaisiana and B. bissettii/carolinensis was detected for the first time in the tissues of these insectivores. While Borrelia genospecies mixed infections have been observed in European hedgehogs before [32,52], we detected the co-occurrence of up to five genospecies in particular individuals. Our results strongly support the hypothesis that European hedgehog plays an important role in the maintenance of various Borrelia genospecies in natural cycles, including the urban environment.
Of all of the target host species, data about Borrelia infections in the Northern white-breasted hedgehog are the scarcest. Despite the fact that this hedgehog species seems to be an important host of the nidicolous hedgehog tick, I. hexagonus, as well as I. ricinus, its role in harboring zoonotic agents has rarely been examined [37,52,76,77]. According to our knowledge, there have only been two B. burgdorferi s.l. genospecies described from Northern white-breasted hedgehog to date, B. afzelii and B. bavariensis [44,52,76]. In our study, more than 70% of the tested specimens were Borrelia-positive, and, interestingly, mixed infections of two or three genospecies were recorded considerably often, thanks to the parallel analysis of several tissue samples per individual. We detected a total of three genospecies: B. afzelii, B. burgdorferi s.s., and B. garinii; to our knowledge, this was the first detection of two of them, namely B. burgdorferi s.s. and B. garinii, in this vertebrate species.
Birds, especially passerine birds, are known as important hosts of zoonotic tick-borne pathogens, which they can transport across geographical barriers (e.g., Reference [78]). The potential of Common blackbirds to serve as reservoir hosts for B. burgdorferi s.l. was proven more than twenty years ago [50]. Since that time, several studies have been performed to describe Borrelia genospecies diversity and prevalence in this avian species, and three of them, B. garinii, B. valaisiana, and B. turdi, are considered to be more closely associated with the Common blackbird [53,78,79,80]. In our set of tested Common blackbirds, B. garinii and B. valaisiana were found most frequently, in line with our expectations. B. bavariensis, a rodent-associated genospecies closely related to B. garinii [81], was found in a single individual. In six (10%) Common blackbird individuals, DNA of B. afzelii was detected in the skin or muscle tissue. These findings can contribute to the discussion about its strict mammal-host specificity. There are several reports of B. afzelii detected in ticks attached to birds (including Common blackbirds), in the vast majority nymphs, where other means of infection may play a role [79,82], but also in tick larvae [83]. Since transovarial transmission of B. burgdorferi s.l. from female ticks to larvae has not been proven to date, that and transmission among co-feeding ticks seems not to be the most likely explanation [82,83], while the hypothesis of birds serving as possible hosts for this Borrelia genospecies is still under discussion [83]. On the other hand, a recent experimental infection study concluded that avian blood most likely has a borreliacidal effect on B. afzelii [82,84]. According to our observations, skin tissue analysis seems to be the most suitable method for Borrelia infection testing in birds, supporting the results of a recent study [85] proving the dermatropism of B. garinii and B. valaisiana in common blackbirds.
Overall, the use of cadavers (and examination of multiple tissue samples) of all target host species seems to be one of the most suitable approaches for studying the prevalence and Borrelia genospecies diversity in urban and periurban areas. We were able to detect the vast majority of Borrelia genospecies previously described in the given host species. A considerably high proportion of real-time PCR positive samples could not be amplified in conventional PCRs or (more often) poor sequences were obtained, which reduced the genospecies identification success rate. Nevertheless, conventional species-specific PCR enabled us to detect some of the previously unidentified genospecies, and, furthermore, we were able to distinguish mixed infections with multiple genospecies in many cases. Successful genospecies identification has shown to be difficult also in Borrelia focused studies in the past, and we suppose that the higher sensitivity of real-time PCR assay compared to conventional PCRs is the most likely explanation for this phenomenon [72,75].
5. Conclusions
A total of 157 cadavers (60 European hedgehogs, 15 Northern white-breasted hedgehogs, 22 Eurasian red squirrels, and 60 Common blackbirds) were collected and dissected resulting in a total of 862 tissue samples. All of the samples were screened for the presence of Borrelia miyamotoi and B. burgdorferi s.l. by multiplex real-time PCR. Several individuals (3 specimens of E. europaeus (5%) and 3 specimens of S. vulgaris (14%)) were found to be B. miyamotoi-positive in one or multiple tissues. High infection rates (90% for E. europaeus, 73% for E. roumanicus, 91% for S. vulgaris, and 68% for T. merula) were observed in all four target host species for B. burgdorferi s.l. The methodological approach using DNA from different tissue samples and a set of conventional PCRs enabled us to detect a wide spectrum of various B. burgdorferi s.l. genospecies in all four vertebrate hosts and revealed a considerably high occurrence of mixed infections on the level of individuals, as well as in particular tissues. According to our knowledge, we are the first to report the detection of B. bissettii/carolinensis DNA in tissue samples of Eurasian red squirrels and European hedgehogs, B. burgdorferi s.s. and B. garinii in Northern white-breasted hedgehogs, B. valaisiana in European hedgehogs, and B. bavariensis, as well as B. afzelii, in Common blackbirds. We conclude that all of the four target vertebrate species contribute to the maintenance of zoonotic Borrelia species, and the cadavers of accidentally killed vertebrates are a valuable source of biological material for monitoring of B. burgdorferi s.l. presence and diversity.
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-2607/8/12/1908/s1, Table S1: Grade of autolysis, Table S2: PCR primers and protocols, Table S3: Samples database, Table S4: Borrelia burgdorferi s.l.-positive animals database, Table S5: Erinaceus europaeus–combinations of Borrelia burgdorferi s.l. genospecies found in animals with mixed infections, Table S6: Erinaceus roumanicus–combinations of Borrelia burgdorferi s.l. genospecies found in animals with mixed infections, Table S7: Sciurus vulgaris–combinations of Borrelia burgdorferi s.l. genospecies found in animals with mixed infections, Table S8: Turdus merula–combinations of Borrelia burgdorferi s.l. genospecies found in animals with mixed infections.
Author Contributions
D.M., J.V., D.R., V.H., and K.M.; methodology, H.S., M.F., N.R., M.G., B.Č.B., P.H., L.H., M.H., and V.H.; formal analysis, K.M., V.H., and M.H.; investigation, V.H., K.M., M.F., L.H., P.P., N.R., M.G., M.H., and B.Č.B.; resources, K.M., J.V., L.H., P.P., V.H., and D.M.; data curation, K.M.,V.H., and L.H.; writing—original draft preparation, K.M., V.H., N.R., and L.H.; writing—review and editing, D.R., J.V., D.M., H.S., M.H., B.Č.B., and P.H.; visualization, K.M. and V.H.; supervision, D.M., J.V., and D.R.; project administration, L.H. and D.M.; funding acquisition, J.V., L.H., D.M., H.S., N.R., and P.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Czech Science Foundation, grant number 17-16009S and the project CePaViP (CZ.02.1.01/16_019/0000759). Hein Sprong and Manoj Fonville were financially supported by the Dutch Ministry of Health, Welfare and Sport (VWS), and by a grant from the European Interreg North Sea Region program, as part of the NorthTick project. Selected analyses of the tested samples were supported by grant QK1920258 from the Ministry of Agriculture of the Czech Republic. Hedgehog species determination was funded by the Czech University of Life Sciences (IGA 20205007 and CIGA 20185006).
Acknowledgments
The authors would like to thank all of the volunteers who participated in cadaver reporting and collection.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lindgren, E.; Jaenson, T. Lyme Borreliosis in Europe: Influences of Climate and Climate Change, Epidemiology, Ecology and Adaptation Measures; WHO Regional Office for Europe: Copenhagen, Denmark, 2006. [Google Scholar]
- Hubálek, Z. Epidemiology of lyme borreliosis. Curr. Probl. Dermatol. 2009, 37, 31–50. [Google Scholar] [PubMed]
- Sykes, R.A.; Makiello, P. An estimate of Lyme borreliosis incidence in Western Europe. J. Public Health 2016, 39, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Petrulioniené, A.; Radzišauskienè, D.; Ambrozaitis, A.; Čaplinskas, S.; Paulauskas, A.; Venalis, A. Epidemiology of Lyme Disease in a highly endemic European zone. Medicina 2020, 56, 115. [Google Scholar] [CrossRef] [PubMed]
- Stanek, G.; Strle, F. Lyme borreliosis–from tick bite to diagnosis and treatment. FEMS Microbiol. Rev. 2018, 42, 233–258. [Google Scholar] [CrossRef]
- Canica, M.M.; Nato, F.; du Merle, L.; Mazie, J.C.; Baranton, G.; Postic, D. Monoclonal antibodies for identification of Borrelia afzelii sp. nov. associated with late cutaneous manifestations of Lyme borreliosis. Scand. J. Infect. Dis. 1993, 25, 441–448. [Google Scholar] [CrossRef]
- Margos, G.; Vollmer, S.A.; Cornet, M.; Garnier, M.; Fingerle, V.; Wilske, B.; Bormane, A.; Vitorino, L.; Collares-Pereira, M.; Drancourt, M.; et al. A new Borrelia species defined by multilocus sequence analysis of housekeeping genes. Appl. Environ. Microbiol. 2009, 75, 5410–5416. [Google Scholar] [CrossRef]
- Postic, D.; Ras, N.M.; Lane, R.S.; Hendson, M.; Baranton, G. Expanded diversity among Californian Borrelia isolates and description of Borrelia bissettii sp. nov. (formerly Borrelia group DN127). J. Clin. Microbiol. 1998, 36, 3497–3504. [Google Scholar] [CrossRef]
- Baranton, G.; Postic, D.; Saint Girons, I.; Boerlin, P.; Piffaretti, J.C.; Assous, M.; Grimont, P.A. Delineation of Borrelia burgdorferi sensu stricto, Borrelia garinii sp. nov., and group VS461 associated with Lyme borreliosis. Int. J. Syst. Bacteriol. 1992, 42, 378–383. [Google Scholar] [CrossRef]
- Margos, G.; Hojgaard, A.; Lane, R.S.; Cornet, M.; Fingerle, V.; Rudenko, N.; Ogden, N.; Aanensen, D.M.; Fish, D.; Piesman, J. Multilocus sequence analysis of Borrelia bissettii strains from North America reveals a new Borrelia species, Borrelia kurtenbachii. Ticks Tick Borne Dis. 2010, 1, 151–158. [Google Scholar] [CrossRef]
- Le Fleche, A.; Postic, D.; Girardet, K.; Peter, O.; Baranton, G. Characterization of Borrelia lusitaniae sp. nov. by 16S ribosomal DNA sequence analysis. Int. J. Syst. Bacteriol. 1997, 47, 921–925. [Google Scholar] [CrossRef]
- Pritt, B.S.; Respicio-Kingry, L.B.; Sloan, L.M.; Schriefer, M.E.; Replogle, A.J.; Bjork, J.; Liu, G.; Kingry, L.C.; Mead, P.S.; Neitzel, D.F.; et al. Borrelia mayonii sp. nov., a member of the Borrelia burgdorferi sensu lato complex, detected in patients and ticks in the upper midwestern United States. Int. J. Syst. Evol. Microbiol. 2016, 66, 4878–4880. [Google Scholar] [CrossRef]
- Richter, D.; Postic, D.; Sertour, N.; Livey, I.; Matuschka, F.R.; Baranton, G. Delineation of Borrelia burgdorferi sensu lato species by multilocus sequence analysis and confirmation of the delineation of Borrelia spielmanii sp. nov. Int. J. Syst. Evol. Microbiol. 2006, 56, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; van Dam, A.P.; Le Fleche, A.; Postic, D.; Peter, O.; Baranton, G.; de Boer, R.; Spanjaard, L.; Dankert, J. Genetic and phenotypic analysis of Borrelia valaisiana sp. nov. (Borrelia genomic groups VS116 and M19). Int. J. Syst. Bacteriol. 1997, 47, 926–932. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Lin, T.; Gao, L.; Grubhoffer, L.; Oliver, J.H., Jr. Delineation of a new species of the Borrelia burgdorferi Sensu Lato Complex, Borrelia americana sp. nov. J. Clin. Microbiol. 2009, 47, 3875–3880. [Google Scholar] [CrossRef] [PubMed]
- Marconi, R.T.; Liveris, D.; Schwartz, I. Identification of novel insertion elements, restriction fragment length polymorphism patterns, and discontinuous 23S rRNA in Lyme disease spirochetes: Phylogenetic analyses of rRNA genes and their intergenic spacers in Borrelia japonica sp. nov. and genomic group 21038 (Borrelia andersonii sp. nov.) isolates. J. Clin. Microbiol. 1995, 33, 2427–2434. [Google Scholar] [PubMed]
- Postic, D.; Garnier, M.; Baranton, G. Multilocus sequence analysis of atypical Borrelia burgdorferi sensu lato isolates--description of Borrelia californiensis sp. nov., and genomospecies 1 and 2. Int. J. Med. Microbiol. 2007, 297, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Borrelia carolinensis sp. nov., a new (14th) member of the Borrelia burgdorferi Sensu Lato complex from the southeastern region of the United States. J Clin. Microbiol. 2009, 47, 134–141. [Google Scholar] [CrossRef]
- Ivanova, L.B.; Tomova, A.; González-Acuña, D.; Murúa, R.; Moreno, C.X.; Hernández, C.; Cabello, J.; Cabello, C.; Daniels, T.J.; Godfrey, H.P.; et al. Borrelia chilensis, a new member of the Borrelia burgdorferi sensu lato complex that extends the range of this genospecies in the Southern Hemisphere. Environ Microbiol. 2014, 16, 1069–1080. [Google Scholar] [CrossRef]
- Casjens, S.R.; Fraser-Liggett, C.M.; Mongodin, E.F.; Qiu, W.G.; Dunn, J.J.; Luft, B.J.; Schutzer, S.E. Whole genome sequence of an unusual Borrelia burgdorferi sensu lato isolate. J/ Bacteriol. 2011, 193, 1489–1490. [Google Scholar] [CrossRef]
- Kawabata, H.; Masuzawa, T.; Yanagihara, Y. Genomic analysis of Borrelia japonica sp. nov. isolated from Ixodes ovatus in Japan. Microbiol/ Immunol. 1993, 37, 843–848. [Google Scholar] [CrossRef]
- Margos, G.; Fedorova, N.; Kleinjan, J.E.; Hartberger, C.; Schwan, T.G.; Sing, A.; Fingerle, V. Borrelia lanei sp. nov. extends the diversity of Borrelia species in California. Int. J. Syst. Evol. Microbiol. 2017, 67, 3872–3876. [Google Scholar] [CrossRef] [PubMed]
- Fukunaga, M.; Hamase, A.; Okada, K.; Nakao, M. Borrelia tanukii sp. nov. and Borrelia turdae sp. nov. found from ixodid ticks in Japan: Rapid species identification by 16S rRNA gene-targeted PCR analysis. Microbiol Immunol. 1996, 40, 877–881. [Google Scholar] [CrossRef] [PubMed]
- Masuzawa, T.; Takada, N.; Kudeken, M.; Fukui, T.; Yano, Y.; Ishiguro, F.; Kawamura, Y.; Imai, Y.; Ezaki, T. Borrelia sinica sp. nov., a lyme disease-related Borrelia species isolated in China. Int. J. Syst. Evol. Microbiol. 2001, 51, 1817–1824. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.Y.; Liu, W.; Jiang, B.G.; Wang, D.M.; Jiang, W.J.; Zhao, Q.M.; Zhang, P.H.; Wang, Z.X.; Tang, G.P.; Yang, H.; et al. Novel genospecies of Borrelia burgdorferi sensu lato from rodents and ticks in southwestern China. J. Clin. Microbiol. 2008, 46, 3130–3133. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Mokráček, A.; Piskunová, N.; Růžek, D.; Mallatová, N.; Grubhoffer, L. Detection of Borrelia bissettii in cardiac valve tissue of a patient with endocarditis and aortic valve stenosis in the Czech Republic. J. Clin. Microbiol. 2008, 46, 3540–3543. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Updates on Borrelia burgdorferi sensu lato complex with respect to public health. Ticks Tick Borne Dis. 2011, 2, 123–128. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Vancová, M.; Clark, K.; Grubhoffer, L.; Oliver, J.H., Jr. Isolation of live Borrelia burgdorferi sensu lato spirochaetes from patients with undefined disorders and symptoms not typical for Lyme borreliosis. Clin. Microbiol. Infect. 2016, 22, 267.e9–267.e15. [Google Scholar] [CrossRef]
- Stanek, G.; Reiter, M. The expanding Lyme Borrelia complex--clinical significance of genomic species? Clin. Microbiol. Infect. 2011, 17, 487–493. [Google Scholar] [CrossRef]
- Gern, L. Borrelia burgdorferi sensu lato, the agent of lyme borreliosis: Life in the wilds. Parasite 2008, 15, 244–247. [Google Scholar] [CrossRef]
- Estrada- Peña, A.; Jongejan, F. Ticks feeding on humans: A review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp. Appl. Acarol. 1999, 23, 685–715. [Google Scholar] [CrossRef]
- Gern, L.; Rouvinez, E.; Toutoungi, L.N.; and Godfroid, E. Transmission cycles of Borrelia burgdorferi sensu lato involving Ixodes ricinus and/or I. hexagonus ticks and the European hedgehog, Erinaceus europaeus, in suburban and urban areas in Switzerland. Folia Parasitol 1997, 44, 309–314. [Google Scholar]
- Kybicová, K.; Baštová, K.; Malý, M. Detection of Borrelia burgdorferi sensu lato and Anaplasma phagocytophilum in questing ticks Ixodes ricinus from the Czech Republic. Ticks Tick Borne Dis. 2017, 8, 483–487. [Google Scholar] [CrossRef]
- Rizzoli, A.; Silaghi, C.; Obiegala, A.; Rudolf, I.; Hubálek, Z.; Földvári, G.; Plantard, O.; Vayssier-Taussat, M.; Bonnet, S.; Špitalská, E.; et al. Ixodes ricinus and its transmitted pathogens in urban and peri-urban areas in Europe: New hazards and relevance for public health. Front. Public Health 2014, 2, 251. [Google Scholar] [CrossRef] [PubMed]
- Gassner, F.; Hansford, K.M.; Medlock, J.M. Greener cities, a wild card for ticks? In Ecology and Prevention of Lyme Borreliosis. Ecology and Control of Vector-Borne Diseases Volume 4; Braks, M.A.H., van Wieren, S.E., Takken, W., Sprong, H., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; pp. 187–203. [Google Scholar]
- Uspensky, I. Tick pests and vectors (Acari: Ixodoidea) in European towns: Introduction, persistence and management. Ticks Tick Borne Dis 2014, 5, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Földvári, G.; Rigó, K.; Jablonszky, M.; Biró, N.; Majoros, G.; Molnár, V.; Tóth, M. Ticks and the city: Ectoparasites of the Northern white-breasted hedgehog (Erinaceus roumanicus) in an urban park. Ticks Tick Borne Dis. 2011, 2, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Heylen, D.; Lasters, R.; Adriaensen, F.; Fonville, M.; Sprong, H.; Matthysen, E. Ticks and tick-borne diseases in the city: Role of landscape connectivity and green space characteristics in a metropolitan area. Sci. Total Environ. 2019, 670, 941–949. [Google Scholar] [CrossRef]
- Hornok, S.; Meli, M.L.; Gönczi, E.; Halász, E.; Takács, N.; Farkas, R.; Hofmann-Lehmann, R. Occurrence of ticks and prevalence of Anaplasma phagocytophilum and Borrelia burgdorferi s.l. in three types of urban biotopes: Forests, parks and cemeteries. Ticks Tick Borne Dis. 2014, 5, 785–789. [Google Scholar] [CrossRef]
- Kubiak, K.; Sielawa, H.; Dziekońska-Rynko, J.; Kubiak, D.; Rydzewska, M.; Dzika, E. Dermacentor reticulatus ticks (Acari: Ixodidae) distribution in north-eastern Poland: An endemic area of tick-borne diseases. Exp. Appl. Acarol. 2018, 75, 289–298. [Google Scholar] [CrossRef]
- Kubiak, K.; Dziekońska-Rynko, J.; Szymańska, H.; Kubiak, D.; Dmitryjuk, M.; Dzika, E. Questing Ixodes ricinus ticks (Acari, Ixodidae) as a vector of Borrelia burgdorferi sensu lato and Borrelia miyamotoi in an urban area of north-eastern Poland. Exp. Appl. Acarol. 2019, 78, 113–126. [Google Scholar] [CrossRef]
- Maetzel, D.; Maier, W.A.; Kampen, H. Borrelia burgdorferi infection prevalences in questing Ixodes ricinus ticks (Acari: Ixodidae) in urban and suburban Bonn, western Germany. Parasitol. Res. 2005, 95, 5–12. [Google Scholar] [CrossRef]
- Mancini, F.; Di Luca, M.; Toma, L.; Vescio, F.; Bianchi, R.; Khoury, C.; Marini, L.; Rezza, G.; and Ciervo, A. Prevalence of tick-borne pathogens in an urban park in Rome, Italy. Ann. Agric. Environ. Med. 2014, 21, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Szekeres, S.; van Leeuwen, A.D.; Tóth, E.; Majoros, G.; Sprong, H.; Földvári, G. Road-killed mammals provide insight into tick-borne bacterial pathogen communities within urban habitats. Transbound. Emerg. Dis. 2019, 66, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Venclíková, K.; Betášová, L.; Sikutová, S.; Jedličková, P.; Hubálek, Z.; Rudolf, I. Human pathogenic borreliae in Ixodes ricinus ticks in natural and urban ecosystem (Czech Republic). Acta Parasitol. 2014, 59, 717–720. [Google Scholar] [CrossRef] [PubMed]
- Evans, K.L.; Gaston, K.J.; Frantz, A.C.; Dimon, M.T.; Sharp, S.P.; McGowan, A.; Dawson, D.A.; Walasz, K.; Partecke, J.; Burke, T.; et al. Independent colonization of multiple urban centres by a formerly forest specialist bird species. Proc. Biol. Sci. 2009, 276, 2403–2410. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.S.; Kahl, O.; Janetzki-Mittman, C.; Stein, J.; Guy, E. Acquisition of Borrelia burgdorferi by Ixodes ricinus ticks fed on the European hedgehog, Erinaceus europaeus L. Exp. Appl. Acarol. 1994, 18, 485–491. [Google Scholar] [CrossRef]
- Hubert, P.; Julliard, R.; Biagianti, S.; Poulle, M.L. Ecological factors driving the higher hedgehog (Erinaceus europeaus) density in an urban area compared to the adjacent rural area. Landsc. Urban Plan 2011, 103, 34–43. [Google Scholar] [CrossRef]
- Humair, P.F.; Gern, L.E. Relationship between Borrelia burgdorferi sensu lato species, Eurasian red squirrels (Sciurus vulgaris) and Ixodes ricinus in enzootic areas in Switzerland. Acta Trop. 1998, 69, 213–227. [Google Scholar] [CrossRef]
- Humair, P.F.; Postic, D.; Wallich, R.; Gern, L. An avian reservoir (Turdus merula) of the Lyme borreliosis spirochetes. Zentralbl. Bakteriol. 1998, 287, 521–538. [Google Scholar] [CrossRef]
- Skuballa, J.; Oehme, R.; Hartelt, K.; Petney, T.; Bücher, T.; Kimmig, P.; Taraschewski, H. European hedgehogs as hosts for Borrelia spp., Germany. Emerg. Infect Dis. 2007, 13, 952–953. [Google Scholar] [CrossRef] [PubMed]
- Skuballa, J.; Petney, T.; Pfäffle, M.; Oehme, R.; Hartelt, K.; Fingerle, V.; Kimmig, P.; Taraschewski, H. Occurrence of different Borrelia burgdorferi sensu lato genospecies including B. afzelii, B. bavariensis, and B. spielmanii in hedgehogs (Erinaceus spp.) in Europe. Ticks Tick Borne Dis. 2012, 3, 8–13. [Google Scholar] [CrossRef]
- Taragel’ová, V.; Koči, J.; Hanincová, K.; Kurtenbach, K.; Derdáková, M.; Ogden, N.H.; Literák, I.; Kocianová, E.; Labuda, M. Blackbirds and song thrushes constitute a key reservoir of Borrelia garinii, the causative agent of borreliosis in Central Europe. Appl. Environ. Microbiol. 2008, 74, 1289–1293. [Google Scholar] [CrossRef] [PubMed]
- Hudec, K.; Šťastný, K. Fauna ČR Ptáci III/I (pěvci), 2nd ed.; Academia: Prague, Czech Republic, 2011; pp. 278–289. [Google Scholar]
- Wilson, D.E.; Lacher, T.E.; Mittermeier, R.A. Handbook of the Mammals of the World. Volume 6: Lagomorphs and Rodents I; Lynx Edicions and IUCN: Barcelona, Spain, 2016; p. 987. [Google Scholar]
- Wilson, D.E.; Mittermeier, R.A. Handbook of the Mammals of the World. Volume 8: Insectivores, Sloths and Colugos; Lynx Edicions and IUCN: Barcelona, Spain, 2018; p. 710. [Google Scholar]
- Bolfíková, B.; Hulva, P. Microevolution of sympatry: Landscape genetics of hedgehogs Erinaceus europaeus and E. roumanicus in Central Europe. Heredity (Edinb) 2012, 108, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Černá Bolfíková, B.; Eliášová, K.; Loudová, M.; Kryštufek, B.; Lymberakis, P.; Sándor, A.D.; Hulva, P. Glacial allopatry vs. postglacial parapatry and peripatry: The case of hedgehogs. PeerJ 2017, 5, e3163. [Google Scholar] [CrossRef] [PubMed]
- Hönig, V.; Palus, M.; Kašpar, T.; Zemanová, M.; Majerová, K.; Hofmannová, L.; Papežík, P.; Šikutová, S.; Rettich, F.; Hubálek, Z.; et al. Multiple Lineages of Usutu Virus (Flaviviridae, Flavivirus) in Blackbirds (Turdus merula) and Mosquitoes (Culex pipiens, Cx. modestus) in the Czech Republic (2016–2019). Microorganisms 2019, 7, 568. [Google Scholar]
- Kasahara, T.; Miyazaki, T.; Nitta, H.; Ono, A.; Miyagishima, T.; Nagao, T.; Urushidani, T. Evaluation of methods for duration of preservation of RNA quality in rat liver used for transcriptome analysis. J. Toxicol. Sci. 2006, 31, 509–519. [Google Scholar] [CrossRef]
- Heylen, D.J.A.; Tijsse, E.; Fonville, M.; Matthysen, E.; Sprong, H. Transmission dynamics of Borrelia burgdorferi s.l. in a bird tick community. Environ. Microbiol. 2013, 15, 663–673. [Google Scholar] [CrossRef]
- Demaerschalck, I.; Ben Messaoud, A.; De Kesel, M.; Hoyois, B.; Lobet, Y.; Hoet, P.; Bigaignon, G.; Bollen, A.; Godfroid, E. Simultaneous presence of different Borrelia burgdorferi genospecies in biological fluids of Lyme disease patients. J. Clin. Microbiol. 1995, 33, 602–608. [Google Scholar] [CrossRef]
- Agresti, A. Categorical Data Analysis, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2002. [Google Scholar]
- Hothorn, T.; Hornik, K.; van de Wiel, M.A.; Zeileis, A. Implementing a class of permutation tests: The coin package. J. Stat. Softw. 2008, 28, 1–23. [Google Scholar] [CrossRef]
- Cutler, S.; Vayssier-Taussat, M.; Estrada-Peña, A.; Potkonjak, A.; Mihalca, A.D.; Zeller, H. A new Borrelia on the block: Borrelia miyamotoi—A human health risk? Euro Surveill 2019, 24, 1800170. [Google Scholar] [CrossRef]
- Crowder, C.D.; Carolan, H.E.; Rounds, M.A.; Hönig, V.; Mothes, B.; Haag, H.; Nolte, O.; Luft, B.J.; Grubhoffer, L.; Ecker, D.J.; et al. Prevalence of Borrelia miyamotoi in Ixodes Ticks in Europe and the United States. Emerg. Infect. Dis. 2014, 20, 1678–1682. [Google Scholar] [CrossRef]
- Page, S.; Daschkin, C.; Anniko, S.; Krey, V.; Nicolaus, C.; Maxeiner, H.G. First report of Borrelia miyamotoi in an Ixodes ricinus tick in Augsburg, Germany. Exp. Appl. Acarol. 2018, 74, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Răileanu, C.; Tauchmann, O.; Vasić, A.; Wöhnke, E.; Silaghi, C. Borrelia miyamotoi and Borrelia burgdorferi (sensu lato) identification and survey of tick-borne encephalitis virus in ticks from north-eastern Germany. Parasit. Vectors 2020, 13, 106. [Google Scholar] [CrossRef] [PubMed]
- Morán Cadenas, F.; Rais, O.; Humair, P.F.; Douet, V.; Moret, J.; Gern, L. Identification of host bloodmeal source and Borrelia burgdorferi sensu lato in field-collected Ixodes ricinus ticks in Chaumont (Switzerland). J. Med. Entomol. 2007, 44, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Pisanu, B.; Chapuis, J.L.; Dozières, A.; Basset, F.; Poux, V.; Vourc´h, G. High prevalence of Borrelia burgdorferi s. l. in the European res squirrel in France. Ticks Tick Borne Dis. 2014, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ruyts, S.C.; Frazer-Mendelewska, E.; Van Den Berge, K.; Verheyen, K.; Sprong, H. Molecular detection of tick-borne pathogens Borrelia afzelii, Borrelia miyamotoi and Anaplasma phagocytophilum in Eurasian red squirrels (Sciurus vulgaris). Eur. J. Wildl. Res. 2017, 63, 43. [Google Scholar] [CrossRef]
- Mysterud, A.; Stigum, V.M.; Jaarsma, R.I.; Sprong, H. Genospecies of Borrelia burgdorferi s. l. detected in 16 mammal species and questing ticks from northern Europe. Sci. Rep. 2019, 9, 5088. [Google Scholar] [CrossRef]
- Krawczyk, A.I.; van Leeuwen, A.D.; Jacobs-Reitsma, W.; Wijnands, L.M.; Bouw, E.; Jahfari, S.; van Hoek, A.H.; van der Giessen, J.W.; Roelfsema, J.H.; Kroes, M.; et al. Presence of zoonotic agents in engorged ticks and hedgehog faeces from Erinaceus europaeus in (sub) urban areas. Parasit. Vectors 2015, 8, 210. [Google Scholar] [CrossRef]
- Szekeres, S.; Majláthová, V.; Majláth, I.; Földvári, G. Neglected hosts: The role of lacertid lizards and medium-sized mammals in the eco-epidemiology of Lyme borreliosis. In Ecology and Prevention of Lyme Borreliosis. Ecology and Control of Vector-Borne Diseases Volume 4; Braks, M.A.H., van Wieren, S.E., Takken, W., Sprong, H., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; pp. 103–126. [Google Scholar]
- Jahfari, S.; Ruyts, S.C.; Frazer-Mendelewska, E.; Jaarsma, R.; Verheyen, K.; Sprong, H. Melting pot of tick-borne zoonoses: The European hedgehog contributes to the maintenance of various tick-borne diseases in natural cycles urban and suburban areas. Parasit. Vectors 2017, 10, 134. [Google Scholar] [CrossRef]
- Dumitrache, M.O.; Paştiu, A.I.; Kalmár, Z.; Mircean, V.; Sándor, A.D.; Gherman, C.M.; Peştean, C.; Mihalca, A.D.; Cozma, V. Northern white-breasted hedgehogs Erinaceus roumanicus as hosts for ticks infected with Borrelia burgdorferi sensu lato and Anaplasma phagocytophilum in Romania. Ticks Tick Borne Dis. 2013, 4, 214–217. [Google Scholar] [CrossRef]
- Dziemian, S.; Sikora, B.; Pilacinska, P.; Michalik, J.; Zwolak, R. Ectoparasite loads in sympatric urban populations of the Northern white-breasted and the European hedgehog. Parasitol. Res. 2015, 114, 2317–2323. [Google Scholar] [CrossRef]
- Heylen, D.J.A. Ecological interactions between songbirds, ticks, and Borrelia burfdorferi s.l. In Europe. In Ecology and Prevention of Lyme Borreliosis. Ecology and Control of Vector-Borne Diseases Volume 4; Braks, M.A.H., van Wieren, S.E., Takken, W., Sprong, H., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; pp. 91–101. [Google Scholar]
- Mtierová, Z.; Derdáková, M.; Chvostáč, M.; Didyk, Y.M.; Mangová, B.; Rusňáková Tarageľová, V.; Selyemová, D.; Šujanová, A.; Václav, R. Local population structure and seasonal variability of Borrelia garinii genotypes in Ixodes ricinus ticks, Slovakia. Int. J. Environ. Res. Public Health 2020, 17, 3607. [Google Scholar] [CrossRef] [PubMed]
- Norte, A.C.; Margos, G.; Becker, N.S.; Ramos, J.A.; Núncio, M.S.; Fingerle, V.; Araújo, P.M.; Adamík, P.; Alivizatos, H.; Barba, E.; et al. Host dispersal shapes the population structure of a tick-borne bacterial pathogen. Mol. Ecol. 2020, 29, 485–501. [Google Scholar] [CrossRef] [PubMed]
- Margos, G.; Fingerle, V.; Reynolds, S. Borrelia bavariensis: Vector switch, niche invasion, and geographical spread of a tick-borne bacterial parasite. Front. Ecol. Evol. 2019, 7, 401. [Google Scholar] [CrossRef]
- Heylen, D.J.A.; Sprong, H.; Krawczyk, A.; Van Houtte, N.; Genné, D.; Gomez-Chamorro, A.; van Oers, K.; Voordouw, M.J. Inefficient co-feeding transmission of Borrelia afzelii in two common European songbirds. Sci. Rep. 2017, 7, 39596. [Google Scholar] [CrossRef]
- Franke, J.; Moldenhauer, A.; Hildebrandt, A.; Dorn, W. Are birds reservoir hosts for Borrelia afzelii? Ticks Tick Borne Dis. 2010, 1, 109–112. [Google Scholar] [CrossRef]
- Tichá, L.; Golovchenko, M.; Oliver, J.H.; Grubhoffer, L.; Rudenko, N. Sensitivity of Lyme borreliosis spirochetes to serum complement of regular Zoo animals: Potential reservoir competence of some exotic vertebrates. Vector Borne Zoonotic Dis 2016, 16, 13–19. [Google Scholar] [CrossRef]
- Norte, A.C.; de Carvalho, I.L.; Núncio, M.S.; Araújo, P.M.; Matthysen, E.; Ramos, J.A.; Sprong, H.; Heylen, D. Getting under the bird´s skin-tissue tropism of Borrelia burgdorferi s.l. in naturally and experimentally infected avian hosts. Microb. Ecol. 2020, 79, 756–769. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).