Postbiotics against Pathogens Commonly Involved in Pediatric Infectious Diseases
Abstract
1. Introduction
2. Potential Mechanisms of Action
2.1. Effects against Pathogenic Bacteria
2.1.1. Escherichia coli
2.1.2. Cronobacter sakazakii
2.1.3. Clostridioides difficile
2.1.4. Salmonella spp.
2.2. Effects against Viruses
2.2.1. Influenza
2.2.2. Rotavirus
2.2.3. Human Immunodeficiency Virus
2.3. Effects against Candida spp.
2.4. Effect against Common Pediatric Infectious Diseases with Unknown Cause
2.5. Effects against Neonatal Necrotizing Enterocolitis
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- UN IGME Levels & Trends in Child Mortality: Report 2019, Estimates Developed by the UN Inter-Agency Group for Child Mortality Estimation; UN: New York, NY, USA, 2019.
- WHO Maternal and Child Epidemiology Estimation Group Child Causes of Death 2000–2017; WHO: Geneva, Switzerland, 2018.
- Troeger, C.; Blacker, B.F.; Khalil, I.A.; Rao, P.C.; Cao, S.; Zimsen, S.R.; Albertson, S.B.; Stanaway, J.D.; Deshpande, A.; Abebe, Z.; et al. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of diarrhoea in 195 countries: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect. Dis. 2018, 18, 1211–1228. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Vuotto, C.; Longo, F.; Donelli, G. Probiotics to counteract biofilm-associated infections: Promising and conflicting data. Int. J. Oral Sci. 2014, 6, 189–194. [Google Scholar] [CrossRef]
- Do Carmo, F.L.R.; Rabah, H.; De Oliveira Carvalho, R.D.; Gaucher, F.; Cordeiro, B.F.; da Silva, S.H.; Le Loir, Y.; Azevedo, V.; Jan, G. Extractable Bacterial Surface Proteins in Probiotic–Host Interaction. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Kolaček, S.; Hojsak, I.; Berni Canani, R.; Guarino, A.; Indrio, F.; Orel, R.; Pot, B.; Shamir, R.; Szajewska, H.; Vandenplas, Y.; et al. Commercial Probiotic Products: A Call for Improved Quality Control. A Position Paper by the ESPGHAN Working Group for Probiotics and Prebiotics. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 117–124. [Google Scholar] [CrossRef]
- Braegger, C.; Chmielewska, A.; Decsi, T.; Kolacek, S.; Mihatsch, W.; Moreno, L.; Pieścik, M.; Puntis, J.; Shamir, R.; Szajewska, H.; et al. Supplementation of infant formula with probiotics and/or prebiotics: A systematic review and comment by the ESPGHAN committee on nutrition. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Skórka, A.; Pieścik-Lech, M.; Kołodziej, M.; Szajewska, H. To add or not to add probiotics to infant formulae? An updated systematic review. Benef. Microbes 2017, 8, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Puccetti, M.; Xiroudaki, S.; Ricci, M.; Giovagnoli, S. Postbiotic-Enabled Targeting of the Host-Microbiota-Pathogen Interface: Hints of Antibiotic Decline? Pharmaceutics 2020, 12, 624. [Google Scholar] [CrossRef] [PubMed]
- Courvalin, P. Antibiotic resistance: The pros and cons of probiotics. Dig. Liver Dis. 2006, 38, S261–S265. [Google Scholar] [CrossRef]
- Zheng, M.; Zhang, R.; Tian, X.; Zhou, X.; Pan, X.; Wong, A. Assessing the Risk of Probiotic Dietary Supplements in the Context of Antibiotic Resistance. Front. Microbiol. 2017, 8, 908. [Google Scholar] [CrossRef] [PubMed]
- Embleton, N.D.; Zalewski, S.; Berrington, J.E. Probiotics for prevention of necrotizing enterocolitis and sepsis in preterm infants. Curr. Opin. Infect. Dis. 2016, 29, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Coviello, C.C.; Corsini, I.I.; Arena, F.; Antonelli, A.; Rossolini, G.M. Lactobacillus Sepsis and Probiotic Therapy in Newborns: Two New Cases and Literature Review. AJP Rep. 2016, 6, e25–e29. [Google Scholar] [CrossRef] [PubMed]
- Lahtinen, S.J. Probiotic viability – does it matter? Microb. Ecol. Health Dis. 2012, 23. [Google Scholar] [CrossRef] [PubMed]
- Ouwehand, A.C.; Tölkkö, S.; Kulmala, J.; Salminen, S.; Salminen, E. Adhesion of inactivated probiotic strains to intestinal mucus. Lett. Appl. Microbiol. 2000, 31, 82–86. [Google Scholar] [CrossRef]
- Corsello, G.; Carta, M.; Marinello, R.; Picca, M.; De Marco, G.; Micillo, M.; Ferrara, D.; Vigneri, P.; Cecere, G.; Ferri, P.; et al. Preventive Effect of Cow’s Milk Fermented with Lactobacillus paracasei CBA L74 on Common Infectious Diseases in Children: A Multicenter Randomized Controlled Trial. Nutrients 2017, 9, 669. [Google Scholar] [CrossRef]
- Del Rigo-Adrover, M.M.; Knipping, K.; Garssen, J.; van Limpt, K.; Knol, J.; Franch, À.; Castell, M.; Rodríguez-lagunas, M.J.; Pérez-Cano, F.J. Prevention of Rotavirus Diarrhea in Suckling Rats by a Specific Fermented Milk Concentrate with Prebiotic Mixture. Nutrients 2019, 11, 189. [Google Scholar] [CrossRef]
- Rudkowski, Z.; Bromirska, J. [Reduction of the duration of salmonella excretion in infants with Hylak forte]. Padiatr. Padol. 1991, 26, 111–114. [Google Scholar]
- Nordeste, R.; Tessema, A.; Sharma, S.; Kovač, Z.; Wang, C.; Morales, R.; Griffiths, M.W. Molecules produced by probiotics prevent enteric colibacillosis in pigs. BMC Vet. Res. 2017, 13. [Google Scholar] [CrossRef]
- Aguilar-Toalá, J.E.; Garcia-Varela, R.; Garcia, H.S.; Mata-Haro, V.; González-Córdova, A.F.; Vallejo-Cordoba, B.; Hernández-Mendoza, A. Postbiotics: An evolving term within the functional foods field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
- Kanauchi, O.; Andoh, A.; AbuBakar, S.; Yamamoto, N. Probiotics and Paraprobiotics in Viral Infection: Clinical Application and Effects on the Innate and Acquired Immune Systems. Curr. Pharm. Des. 2018, 24, 710–717. [Google Scholar] [CrossRef]
- Shenderov, B.A. Metabiotics: Novel idea or natural development of probiotic conception. Microb. Ecol. Health Dis. 2013, 24. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Chandra, V.; Kim, N.-H.; Rai, R.; Kumar, P.; Kim, K.; Aeron, A.; Kang, S.C.; Maheshwari, D.K.; Na, M.; et al. Ghost probiotics with a combined regimen: A novel therapeutic approach against the Zika virus, an emerging world threat. Crit. Rev. Biotechnol. 2018, 38, 438–454. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.-S.; Song, E.-J.; Nam, Y.-D.; Lee, S.-Y. Probiotics in human health and disease: From nutribiotics to pharmabiotics. J. Microbiol. 2018, 56, 773–782. [Google Scholar] [CrossRef]
- Collado, M.C.; Vinderola, G.; Salminen, S. Postbiotics: Facts and open questions. A position paper on the need for a consensus definition. Benef. Microbes 2019, 10, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Żółkiewicz, J.; Marzec, A.; Ruszczyński, M.; Feleszko, W. Postbiotics—A Step Beyond Pre- and Probiotics. Nutrients 2020, 12, 2189. [Google Scholar] [CrossRef]
- Taverniti, V.; Guglielmetti, S. The immunomodulatory properties of probiotic microorganisms beyond their viability (ghost probiotics: Proposal of paraprobiotic concept). Genes Nutr. 2011, 6, 261–274. [Google Scholar] [CrossRef]
- Malagón-Rojas, J.N.; Mantziari, A.; Salminen, S.; Szajewska, H. Postbiotics for Preventing and Treating Common Infectious Diseases in Children: A Systematic Review. Nutrients 2020, 12, 389. [Google Scholar] [CrossRef]
- Jayamani, E.; Mylonakis, E. Effector triggered manipulation of host immune response elicited by different pathotypes of Escherichia coli. Virulence 2014, 5, 733–739. [Google Scholar] [CrossRef][Green Version]
- Kim, Y.K.; Shin, J.-S.; Nahm, M.H. NOD-Like Receptors in Infection, Immunity, and Diseases. Yonsei Med. J. 2016, 57, 5–14. [Google Scholar] [CrossRef]
- Fenini, G.; Contassot, E.; French, L.E. Potential of IL-1, IL-18 and Inflammasome Inhibition for the Treatment of Inflammatory Skin Diseases. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef]
- Kummer, J.A.; Broekhuizen, R.; Everett, H.; Agostini, L.; Kuijk, L.; Martinon, F.; van Bruggen, R.; Tschopp, J. Inflammasome Components NALP 1 and 3 Show Distinct but Separate Expression Profiles in Human Tissues Suggesting a Site-specific Role in the Inflammatory Response. J. Histochem. Cytochem. 2007. [Google Scholar] [CrossRef] [PubMed]
- Warshakoon, H.J.; Hood, J.D.; Kimbrell, M.R.; Malladi, S.; Wu, W.Y.; Shukla, N.M.; Agnihotri, G.; Sil, D.; David, S.A. Potential adjuvantic properties of innate immune stimuli. Hum. Vaccin. 2009, 5, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Chandler, C.E.; Ernst, R.K. Bacterial lipids: Powerful modifiers of the innate immune response. F1000Research 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Petes, C.; Odoardi, N.; Gee, K. The Toll for Trafficking: Toll-Like Receptor 7 Delivery to the Endosome. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Dalpke, A.; Frank, J.; Peter, M.; Heeg, K. Activation of Toll-Like Receptor 9 by DNA from Different Bacterial Species. Infect. Immun. 2006, 74, 940–946. [Google Scholar] [CrossRef]
- Yu, S.; Gao, N. Compartmentalizing Intestinal Epithelial Cell Toll-like Receptors for Immune Surveillance. Cell. Mol. Life Sci. CMLS 2015, 72, 3343–3353. [Google Scholar] [CrossRef]
- Wojdasiewicz, P.; Poniatowski, Ł.A.; Szukiewicz, D. The Role of Inflammatory and Anti-Inflammatory Cytokines in the Pathogenesis of Osteoarthritis. Mediators Inflamm. 2014, 2014. [Google Scholar] [CrossRef]
- Srinivasan, L.; Harris, M.C.; Kilpatrick, L.E. 128-Cytokines and Inflammatory Response in the Fetus and Neonate. In Fetal and Neonatal Physiology, Polin, R.A., Abman, S.H., Rowitch, D.H., Benitz, W.E., Fox, W.W., Eds.; 5th ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1241–1254. ISBN 978-0-323-35214-7. [Google Scholar]
- Azad, M.A.K.; Sarker, M.; Wan, D. Immunomodulatory Effects of Probiotics on Cytokine Profiles. Available online: https://www.hindawi.com/journals/bmri/2018/8063647/ (accessed on 7 April 2019).
- Miyazawa, K.; He, F.; Kawase, M.; Kubota, A.; Yoda, K.; Hiramatsu, M. Enhancement of immunoregulatory effects of Lactobacillus gasseri TMC0356 by heat treatment and culture medium. Lett. Appl. Microbiol. 2011, 53, 210–216. [Google Scholar] [CrossRef]
- Zagato, E.; Mileti, E.; Massimiliano, L.; Fasano, F.; Budelli, A.; Penna, G.; Rescigno, M. Lactobacillus paracasei CBA L74 Metabolic Products and Fermented Milk for Infant Formula Have Anti-Inflammatory Activity on Dendritic Cells In Vitro and Protective Effects against Colitis and an Enteric Pathogen In Vivo. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Wang, H.; Yan, Y.; Wang, J.; Zhang, H.; Qi, W. Production and Characterization of Antifungal Compounds Produced by Lactobacillus plantarum IMAU10014. PLoS ONE 2012, 7, e29452. [Google Scholar] [CrossRef]
- Servin, A.L. Antagonistic activities of lactobacilli and bifidobacteria against microbial pathogens. FEMS Microbiol. Rev. 2004, 28, 405–440. [Google Scholar] [CrossRef] [PubMed]
- Ricke, S.C. Perspectives on the use of organic acids and short chain fatty acids as antimicrobials. Poult. Sci. 2003, 82, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Suiryanrayna, M.V.A.N.; Ramana, J.V. A review of the effects of dietary organic acids fed to swine. J. Anim. Sci. Biotechnol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Slonczewski, J.L.; Fujisawa, M.; Dopson, M.; Krulwich, T.A. Cytoplasmic pH Measurement and Homeostasis in Bacteria and Archaea. In Advances in Microbial Physiology; Poole, R.K., Ed.; Academic Press: Cambridge, MA, USA, 2009; Volume 55, pp. 1–317. [Google Scholar]
- Patel, R.M.; Denning, P.W. Therapeutic Use of Prebiotics, Probiotics, and Postbiotics to Prevent Necrotizing Enterocolitis: What is the Current Evidence? Clin. Perinatol. 2013, 40, 11–25. [Google Scholar] [CrossRef]
- Kareem, K.Y.; Hooi Ling, F.; Teck Chwen, L.; May Foong, O.; Anjas Asmara, S. Inhibitory activity of postbiotic produced by strains of Lactobacillus plantarum using reconstituted media supplemented with inulin. Gut Pathog. 2014, 6, 23. [Google Scholar] [CrossRef]
- Sharma, V.; Harjai, K.; Shukla, G. Effect of bacteriocin and exopolysaccharides isolated from probiotic on P. aeruginosa PAO1 biofilm. Folia Microbiol. (Praha) 2018, 63, 181–190. [Google Scholar] [CrossRef]
- Li, P.; Gu, Q. Antimicrobial Effects of Probiotics and Novel Probiotic-Based Approaches for Infectious Diseases. Probiotics Curr. Knowl. Future Prospects 2018. [Google Scholar] [CrossRef]
- Pridmore, R.D.; Pittet, A.-C.; Praplan, F.; Cavadini, C. Hydrogen peroxide production by Lactobacillus johnsonii NCC 533 and its role in anti-Salmonella activity. FEMS Microbiol. Lett. 2008, 283, 210–215. [Google Scholar] [CrossRef]
- Wang, Y.; Gänzle, M.G.; Schwab, C. Exopolysaccharide Synthesized by Lactobacillus reuteri Decreases the Ability of Enterotoxigenic Escherichia coli To Bind to Porcine Erythrocytes. Appl Environ. Microbiol. 2010, 76, 4863–4866. [Google Scholar] [CrossRef]
- Fanning, S.; Hall, L.J.; Cronin, M.; Zomer, A.; MacSharry, J.; Goulding, D.; O’Connell Motherway, M.; Shanahan, F.; Nally, K.; Dougan, G.; et al. Bifidobacterial surface-exopolysaccharide facilitates commensal-host interaction through immune modulation and pathogen protection. Proc. Natl. Acad. Sci. USA 2012, 109, 2108–2113. [Google Scholar] [CrossRef]
- Abdelhamid, A.G.; Esaam, A.; Hazaa, M.M. Cell free preparations of probiotics exerted antibacterial and antibiofilm activities against multidrug resistant E. coli. Saudi Pharm. J. SPJ 2018, 26, 603–607. [Google Scholar] [CrossRef]
- Bondue, P.; Crèvecoeur, S.; Brose, F.; Daube, G.; Seghaye, M.-C.; Griffiths, M.W.; LaPointe, G.; Delcenserie, V. Cell-Free Spent Media Obtained from Bifidobacterium bifidum and Bifidobacterium crudilactis Grown in Media Supplemented with 3′-Sialyllactose Modulate Virulence Gene Expression in Escherichia coli O157:H7 and Salmonella Typhimurium. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Bayoumi, M.A.; Griffiths, M.W. In vitro inhibition of expression of virulence genes responsible for colonization and systemic spread of enteric pathogens using Bifidobacterium bifidum secreted molecules. Int. J. Food Microbiol. 2012, 156, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Chauvière, G.; Coconnier, M.H.; Kerneis, S.; Darfeuille-Michaud, A.; Joly, B.; Servin, A.L. Competitive exclusion of diarrheagenic Escherichia coli (ETEC) from human enterocyte-like Caco-2 cells by heat-killed Lactobacillus. FEMS Microbiol. Lett. 1992, 70, 213–217. [Google Scholar] [CrossRef]
- Coconnier, M.H.; Bernet, M.F.; Chauvière, G.; Servin, A.L. Adhering heat-killed human Lactobacillus acidophilus, strain LB, inhibits the process of pathogenicity of diarrhoeagenic bacteria in cultured human intestinal cells. J. Diarrhoeal Dis. Res. 1993, 11, 235–242. [Google Scholar] [PubMed]
- Muñoz-Quezada, S.; Bermudez-Brito, M.; Chenoll, E.; Genovés, S.; Gomez-Llorente, C.; Plaza-Diaz, J.; Matencio, E.; Bernal, M.J.; Romero, F.; Ramón, D.; et al. Competitive inhibition of three novel bacteria isolated from faeces of breast milk-fed infants against selected enteropathogens. Br. J. Nutr. 2013, 109, S63–S69. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-C.; Lin, P.-P.; Hsieh, Y.-M. Three Lactobacillus strains from healthy infant stool inhibit enterotoxigenic Escherichia coli grown in vitro. Anaerobe 2008, 14, 61–67. [Google Scholar] [CrossRef]
- He, X.; Zeng, Q.; Puthiyakunnon, S.; Zeng, Z.; Yang, W.; Qiu, J.; Du, L.; Boddu, S.; Wu, T.; Cai, D.; et al. Lactobacillus rhamnosus GG supernatant enhance neonatal resistance to systemic Escherichia coli K1 infection by accelerating development of intestinal defense. Sci. Rep. 2017, 7, 43305. [Google Scholar] [CrossRef]
- Gao, J.; Li, Y.; Wan, Y.; Hu, T.; Liu, L.; Yang, S.; Gong, Z.; Zeng, Q.; Wei, Y.; Yang, W.; et al. A Novel Postbiotic From Lactobacillus rhamnosus GG With a Beneficial Effect on Intestinal Barrier Function. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Charchoghlyan, H.; Kwon, H.; Hwang, D.-J.; Lee, J.S.; Lee, J.; Kim, M. Inhibition of Cronobacter sakazakii by Lactobacillus acidophilus n.v. Er2 317/402. Korean J. Food Sci. Anim. Resour. 2016, 36, 635–640. [Google Scholar] [CrossRef]
- Jamwal, A.; Sharma, K.; Chauhan, R.; Bansal, S.; Goel, G. Evaluation of commercial probiotic lactic cultures against biofilm formation by Cronobacter sakazakii. Intest. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dowdell, P.; Chankhamhaengdecha, S.; Panbangred, W.; Janvilisri, T.; Aroonnual, A. Probiotic Activity of Enterococcus faecium and Lactococcus lactis Isolated from Thai Fermented Sausages and Their Protective Effect Against Clostridium difficile. Probiotics Antimicrob. Proteins 2019. [Google Scholar] [CrossRef] [PubMed]
- Ephraim, E.; Schultz, R.D.; Safdar, N. Lactobacillus rhamnosus GG Protects Cells from Clostridium difficile Toxins. Br. Microbiol. Res. J. 2013, 3, 165–175. [Google Scholar] [CrossRef]
- Tsilingiri, K.; Barbosa, T.; Penna, G.; Caprioli, F.; Sonzogni, A.; Viale, G.; Rescigno, M. Probiotic and postbiotic activity in health and disease: Comparison on a novel polarised ex-vivo organ culture model. Gut 2012, 61, 1007–1015. [Google Scholar] [CrossRef]
- Bermudez-Brito, M.; Munoz-Quezada, S.; Gomez-Llorente, C.; Matencio, E.; Romero, F.; Gil, A. Lactobacillus paracasei CNCM I-4034 and its culture supernatant modulate Salmonella-induced inflammation in a novel transwell co-culture of human intestinal-like dendritic and Caco-2 cells. Bmc Microbiol. 2015, 15, 79. [Google Scholar] [CrossRef]
- Rather, I.A.; Choi, K.-H.; Bajpai, V.K.; Park, Y.-H. Antiviral mode of action of Lactobacillus plantarum YML009 on Influenza virus H1N1. Bangladesh J. Pharmacol. 2015, 10, 475. [Google Scholar] [CrossRef]
- Seo, B.J.; Rather, I.A.; Kumar, V.J.R.; Choi, U.H.; Moon, M.R.; Lim, J.H.; Park, Y.H. Evaluation of Leuconostoc mesenteroides YML003 as a probiotic against low-pathogenic avian influenza (H9N2) virus in chickens. J. Appl. Microbiol. 2012, 113, 163–171. [Google Scholar] [CrossRef]
- Kaila, M.; Isolauri, E.; Saxelin, M.; Arvilommi, H.; Vesikari, T. Viable versus inactivated lactobacillus strain GG in acute rotavirus diarrhoea. Arch. Dis. Child. 1995, 72, 51–53. [Google Scholar] [CrossRef]
- Martín, V.; Maldonado, A.; Fernández, L.; Rodríguez, J.M.; Connor, R.I. Inhibition of Human Immunodeficiency Virus Type 1 by Lactic Acid Bacteria from Human Breastmilk. Breastfeed. Med. 2010, 5, 153–158. [Google Scholar] [CrossRef]
- Hassan, Z. Anti-Adhesion Activity of Lactic Acid Bacteria Supernatant against Human Pathogenic Candida Species Biofilm. Health Sci. J. 2015, 9. [Google Scholar]
- Parolin, C.; Marangoni, A.; Laghi, L.; Foschi, C.; Palomino, R.A.Ñ.; Calonghi, N.; Cevenini, R.; Vitali, B. Isolation of Vaginal Lactobacilli and Characterization of Anti-Candida Activity. PLoS ONE 2015, 10, e0131220. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.-G.; Lee, S.-H. Inhibitory effects of Lactobacillus rhamnosus and Lactobacillus casei on Candida biofilm of denture surface. Arch. Oral Biol. 2017, 76, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Nocerino, R.; Paparo, L.; Terrin, G.; Pezzella, V.; Amoroso, A.; Cosenza, L.; Cecere, G.; Marco, G.D.; Micillo, M.; Albano, F.; et al. Cow’s milk and rice fermented with Lactobacillus paracasei CBA L74 prevent infectious diseases in children: A randomized controlled trial. Clin. Nutr. 2017, 36, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Thibault, H.; Aubert-Jacquin, C.; Goulet, O. Effects of Long-term Consumption of a Fermented Infant Formula (with Bifidobacterium breve c50 and Streptococcus thermophilus 065) on Acute Diarrhea in Healthy Infants. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 147. [Google Scholar] [CrossRef] [PubMed]
- Romond, M.B.; Ais, A.; Yazourh, A.; Romond, C. Cell-free Wheys From Bifidobacteria Fermented Milks Exert a Regulatory Effect on the Intestinal Microflora of Mice and Humans. Anaerobe 1997, 3, 137–143. [Google Scholar] [CrossRef]
- Arai, S.; Iwabuchi, N.; Takahashi, S.; Xiao, J.; Abe, F.; Hachimura, S. Orally administered heat-killed Lactobacillus paracasei MCC1849 enhances antigen-specific IgA secretion and induces follicular helper T cells in mice. PLoS ONE 2018, 13, e0199018. [Google Scholar] [CrossRef]
- Good, M.; Sodhi, C.P.; Ozolek, J.A.; Buck, R.H.; Goehring, K.C.; Thomas, D.L.; Vikram, A.; Bibby, K.; Morowitz, M.J.; Firek, B.; et al. Lactobacillus rhamnosus HN001 decreases the severity of necrotizing enterocolitis in neonatal mice and preterm piglets: Evidence in mice for a role of TLR9. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 306, G1021–G1032. [Google Scholar] [CrossRef]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.A.P.; Harris, H.M.B.; Mattarelli, P.; O’Toole, P.W.; Pot, B.; Vandamme, P.; Walter, J.; et al. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef]
- Kotloff, K.L.; Nataro, J.P.; Blackwelder, W.C.; Nasrin, D.; Farag, T.H.; Panchalingam, S.; Wu, Y.; Sow, S.O.; Sur, D.; Breiman, R.F.; et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): A prospective, case-control study. Lancet 2013, 382, 209–222. [Google Scholar] [CrossRef]
- Kotloff, K.L.; Nasrin, D.; Blackwelder, W.C.; Wu, Y.; Farag, T.; Panchalingham, S.; Sow, S.O.; Sur, D.; Zaidi, A.K.M.; Faruque, A.S.G.; et al. The incidence, aetiology, and adverse clinical consequences of less severe diarrhoeal episodes among infants and children residing in low-income and middle-income countries: A 12-month case-control study as a follow-on to the Global Enteric Multicenter Study (GEMS). Lancet Glob. Health 2019, 7, e568–e584. [Google Scholar] [CrossRef]
- Liévin-Le Moal, V. A gastrointestinal anti-infectious biotherapeutic agent: The heat-treated Lactobacillus LB. Ther. Adv. Gastroenterol. 2016, 9, 57–75. [Google Scholar] [CrossRef] [PubMed]
- Chauvière, G.; Coconnier, M.H.; Kernéis, S.; Fourniat, J.; Servin, A.L. Adhesion of human Lactobacillus acidophilus strain LB to human enterocyte-like Caco-2 cells. J. Gen. Microbiol. 1992, 138, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Quezada, S.; Chenoll, E.; Vieites, J.M.; Genovés, S.; Maldonado, J.; Bermúdez-Brito, M.; Gomez-Llorente, C.; Matencio, E.; Bernal, M.J.; Romero, F.; et al. Isolation, identification and characterisation of three novel probiotic strains (Lactobacillus paracasei CNCM I-4034, Bifidobacterium breve CNCM I-4035 and Lactobacillus rhamnosus CNCM I-4036) from the faeces of exclusively breast-fed infants. Br. J. Nutr. 2013, 109 (Suppl. 2), S51–S62. [Google Scholar] [CrossRef]
- Zhang, H.; Gao, J.; He, X.; Gong, Z.; Wan, Y.; Hu, T.; Li, Y.; Cao, H. The postbiotic HM0539 from Lactobacillus rhamnosus GG prevents intestinal infection by enterohemorrhagic E. coli O157: H7 in mice. J. South. Med. Univ. 2020, 40, 211–218. [Google Scholar] [CrossRef]
- Agostoni, C.; Axelsson, I.; Goulet, O.; Koletzko, B.; Michaelsen, K.F.; Puntis, J.W.L.; Rigo, J.; Shamir, R.; Szajewska, H.; Turck, D.; et al. Preparation and Handling of Powdered Infant Formula: A Commentary by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 320–322. [Google Scholar] [CrossRef]
- Borali, E.; De Giacomo, C. Clostridium Difficile Infection in Children: A Review. J. Pediatr. Gastroenterol. Nutr. 2016, 63, e130–e140. [Google Scholar] [CrossRef]
- Plants-Paris, K.; Bishoff, D.; Oyaro, M.O.; Mwinyi, B.; Chappell, C.; Kituyi, A.; Nyangao, J.; Mbatha, D.; Darkoh, C. Prevalence of Clostridium difficile infections among Kenyan children with diarrhea. Int. J. Infect. Dis. 2019, 81, 66–72. [Google Scholar] [CrossRef]
- Jangi, S.; Lamont, J. Asymptomatic Colonization by Clostridium difficile in Infants: Implications for Disease in Later Life. J. Pediatr. Gastroenterol. Nutr. 2010, 51, 2–7. [Google Scholar] [CrossRef]
- Schäffler, H.; Breitrück, A. Clostridium difficile–From Colonization to Infection. Front. Microbiol. 2018, 9, 646. [Google Scholar] [CrossRef]
- Fiedoruk, K.; Daniluk, T.; Rozkiewicz, D.; Zaremba, M.L.; Oldak, E.; Sciepuk, M.; Leszczynska, K. Conventional and molecular methods in the diagnosis of community-acquired diarrhoea in children under 5 years of age from the north-eastern region of Poland. Int. J. Infect. Dis. 2015, 37, 145–151. [Google Scholar] [CrossRef][Green Version]
- Dunand, E.; Burns, P.; Binetti, A.; Bergamini, C.; Peralta, G.H.; Forzani, L.; Reinheimer, J.; Vinderola, G. Postbiotics produced at laboratory and industrial level as potential functional food ingredients with the capacity to protect mice against Salmonella infection. J. Appl. Microbiol. 2019, 127, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Lafond, K.E.; Nair, H.; Rasooly, M.H.; Valente, F.; Booy, R.; Rahman, M.; Kitsutani, P.; Yu, H.; Guzman, G.; Coulibaly, D.; et al. Global Role and Burden of Influenza in Pediatric Respiratory Hospitalizations, 1982–2012: A Systematic Analysis. PLoS Med. 2016, 13. [Google Scholar] [CrossRef] [PubMed]
- El Guerche-Séblain, C.; Moureau, A.; Schiffler, C.; Dupuy, M.; Pepin, S.; Samson, S.I.; Vanhems, P.; Schellevis, F. Epidemiology and burden of influenza in healthy children aged 6 to 35 months: Analysis of data from the placebo arm of a phase III efficacy trial. BMC Infect. Dis. 2019, 19, 308. [Google Scholar] [CrossRef] [PubMed]
- de Francisco (Shapovalova), N.; Donadel, M.; Jit, M.; Hutubessy, R. A systematic review of the social and economic burden of influenza in low- and middle-income countries. Vaccine 2015, 33, 6537–6544. [Google Scholar] [CrossRef]
- Thorrington, D.; Balasegaram, S.; Cleary, P.; Hay, C.; Eames, K. Social and Economic Impacts of School Influenza Outbreaks in England: Survey of Caregivers. J. Sch. Health 2017, 87, 209–216. [Google Scholar] [CrossRef]
- Hagbom, M.; Sharma, S.; Lundgren, O.; Svensson, L. Towards a human rotavirus disease model. Curr. Opin. Virol. 2012, 2, 408–418. [Google Scholar] [CrossRef]
- Simakachorn, N.; Pichaipat, V.; Rithipornpaisarn, P.; Kongkaew, C.; Tongpradit, P.; Varavithya, W. Clinical evaluation of the addition of lyophilized, heat-killed Lactobacillus acidophilus LB to oral rehydration therapy in the treatment of acute diarrhea in children. J. Pediatr. Gastroenterol. Nutr. 2000, 30, 68–72. [Google Scholar] [CrossRef]
- Salazar-Lindo, E.; Figueroa-Quintanilla, D.; Caciano, M.; Reto-Valiente, V.; Chauviere, G.; Colin, P. Effectiveness and Safety of Lactobacillus LB in the Treatment of Mild Acute Diarrhea in Children. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 571–576. [Google Scholar] [CrossRef]
- Osano, B.O.; Were, F.; Mathews, S. Mortality among 5-17 year old children in Kenya. Pan Afr. Med. J. 2017, 27. [Google Scholar] [CrossRef]
- Buvé, A.; Bishikwabo-Nsarhaza, K.; Mutangadura, G. The spread and effect of HIV-1 infection in sub-Saharan Africa. Lancet 2002, 359, 2011–2017. [Google Scholar] [CrossRef]
- WHO Antiretroviral Drugs for Treating Pregnant Women and Preventing HIV Infection in Infants. Available online: https://apps.who.int/iris/bitstream/handle/10665/75236/9789241599818_eng.pdf?sequence=1 (accessed on 17 August 2020).
- WHO Guidelines on HIV and Infant Feeding. 2010. Available online: http://www.who.int/maternal_child_adolescent/documents/9789241599535/en/ (accessed on 17 August 2020).
- WHO HIV and Infant Feeding: Update. Available online: https://apps.who.int/iris/bitstream/handle/10665/43747/9789241595964_eng.pdf;jsessionid=3D425244271D458E9F10E85E311DFBCD?sequence=1 (accessed on 17 August 2020).
- White, A.B.; Mirjahangir, J.F.; Horvath, H.; Anglemyer, A.; Read, J.S. Antiretroviral interventions for preventing breast milk transmission of HIV. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Toda, M. Population-Based Active Surveillance for Culture-Confirmed Candidemia — Four Sites, United States, 2012–2016. MMWR Surveill. Summ. 2019, 68. [Google Scholar] [CrossRef] [PubMed]
- Noni, M.; Stathi, A.; Vaki, I.; Velegraki, A.; Zachariadou, L.; Michos, A. Changing Epidemiology of Invasive Candidiasis in Children during a 10-Year Period. J. Fungi 2019, 5, 19. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, D.; Boyle, R.; Hazen, K.C.; Patrie, J.T.; Robinson, M.; Donowitz, L.G. Fluconazole prophylaxis against fungal colonization and infection in preterm infants. N. Engl. J. Med. 2001, 345, 1660–1666. [Google Scholar] [CrossRef]
- Gephart, S.M.; McGrath, J.M.; Effken, J.A.; Halpern, M.D. Necrotizing Enterocolitis Risk: State of the Science. Adv. Neonatal Care 2012, 12, 77–87. [Google Scholar] [CrossRef]
- Coggins, S.A.; Wynn, J.L.; Weitkamp, J.-H. Infectious causes of necrotizing enterocolitis. Clin. Perinatol. 2015, 42, 133. [Google Scholar] [CrossRef]
- Wu, W.; Wang, Y.; Zou, J.; Long, F.; Yan, H.; Zeng, L.; Chen, Y. Bifidobacterium adolescentis protects against necrotizing enterocolitis and upregulates TOLLIP and SIGIRR in premature neonatal rats. BMC Pediatr. 2017, 17, 1. [Google Scholar] [CrossRef]
- Schönherr-Hellec, S.; Klein, G.L.; Delannoy, J.; Ferraris, L.; Rozé, J.C.; Butel, M.J.; Aires, J. Clostridial Strain-Specific Characteristics Associated with Necrotizing Enterocolitis. Appl Environ. Microbiol 2018, 84, e02428-17. [Google Scholar] [CrossRef]
- Alfa, M.J.; Robson, D.; Davi, M.; Bernard, K.; Caeseele, P.V.; Harding, G.K.M. An Outbreak of Necrotizing Enterocolitis Associated with a Novel Clostridium Species in a Neonatal Intensive Care Unit. Clin. Infect. Dis. 2002, 35, S101–S105. [Google Scholar] [CrossRef]
- Denning, N.-L.; Prince, J.M. Neonatal intestinal dysbiosis in necrotizing enterocolitis. Mol. Med. 2018, 24, 4. [Google Scholar] [CrossRef]
- Land, M.H.; Rouster-Stevens, K.; Woods, C.R.; Cannon, M.L.; Cnota, J.; Shetty, A.K. Lactobacillus Sepsis Associated With Probiotic Therapy. Pediatrics 2005, 115, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.M.; Underwood, M.A. Probiotics and Necrotizing Enterocolitis. Semin. Pediatr. Surg. 2018, 27, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Chiang, M.-C.; Chen, C.-L.; Feng, Y.; Chen, C.-C.; Lien, R.; Chiu, C.-H. Lactobacillus rhamnosus sepsis associated with probiotic therapy in an extremely preterm infant: Pathogenesis and a review for clinicians. J. Microbiol. Immunol. Infect. 2020. [Google Scholar] [CrossRef] [PubMed]
- Su, G.L.; Ko, C.W.; Bercik, P.; Falck-Ytter, Y.; Sultan, S.; Weizman, A.V.; Morgan, R.L. AGA Clinical Practice Guidelines on the Role of Probiotics in the Management of Gastrointestinal Disorders. Gastroenterology 2020. [Google Scholar] [CrossRef]
- van den Akker, C.H.P.; van Goudoever, J.B.; Shamir, R.; Domellöf, M.; Embleton, N.D.; Hojsak, I.; Lapillonne, A.; Mihatsch, W.A.; Berni Canani, R.; Bronsky, J.; et al. Probiotics and Preterm Infants: A Position Paper by the European Society for Paediatric Gastroenterology Hepatology and Nutrition Committee on Nutrition and the European Society for Paediatric Gastroenterology Hepatology and Nutrition Working Group for Probiotics and Prebiotics. J. Pediatr. Gastroenterol. Nutr. 2020, 70, 664–680. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.T.; Fukumori, C.; Ferreira, C.M. New insights into therapeutic strategies for gut microbiota modulation in inflammatory diseases. Clin. Transl. Immunol. 2016, 5, e87. [Google Scholar] [CrossRef] [PubMed]
- Mosca, F.; Gianni, M.L.; Rescigno, M. Can Postbiotics Represent a New Strategy for NEC? Adv. Exp. Med. Biol. 2019. [Google Scholar] [CrossRef]
- Campeotto, F.; Suau, A.; Kapel, N.; Magne, F.; Viallon, V.; Ferraris, L.; Waligora-Dupriet, A.-J.; Soulaines, P.; Leroux, B.; Kalach, N.; et al. A fermented formula in pre-term infants: Clinical tolerance, gut microbiota, down-regulation of faecal calprotectin and up-regulation of faecal secretory IgA. Br. J. Nutr. 2011, 105, 1843–1851. [Google Scholar] [CrossRef]
- Ehrenkranz, R.A.; Dusick, A.M.; Vohr, B.R.; Wright, L.L.; Wrage, L.A.; Poole, W.K. Growth in the Neonatal Intensive Care Unit Influences Neurodevelopmental and Growth Outcomes of Extremely Low Birth Weight Infants. Pediatrics 2006, 117, 1253–1261. [Google Scholar] [CrossRef]
- Jeong, S.J. The role of fecal calprotectin in pediatric disease. Korean J. Pediatr. 2019, 62, 287–291. [Google Scholar] [CrossRef]
- Mullié, C.; Yazourh, A.; Thibault, H.; Odou, M.-F.; Singer, E.; Kalach, N.; Kremp, O.; Romond, M.-B. Increased Poliovirus-Specific Intestinal Antibody Response Coincides with Promotion of Bifidobacterium longum-infantis and Bifidobacterium breve in Infants: A Randomized, Double-Blind, Placebo-Controlled Trial. Pediatr. Res. 2004, 56, 791–795. [Google Scholar] [CrossRef] [PubMed]
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Barczyńska, R.; Litwin, M.; Sliżewska, K.; Szalecki, M.; Berdowska, A.; Bandurska, K.; Libudzisz, Z.; Kapuśniak, J. Bacterial Microbiota and Fatty Acids in the Faeces of Overweight and Obese Children. Pol. J. Microbiol. 2018, 67, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Riva, A.; Borgo, F.; Lassandro, C.; Verduci, E.; Morace, G.; Borghi, E.; Berry, D. Pediatric obesity is associated with an altered gut microbiota and discordant shifts in F irmicutes populations. Environ. Microbiol. 2017, 19, 95–105. [Google Scholar] [CrossRef]
- Ibrahim, M.K.; Zambruni, M.; Melby, C.L.; Melby, P.C. Impact of Childhood Malnutrition on Host Defense and Infection. Clin. Microbiol. Rev. 2017, 30, 919–971. [Google Scholar] [CrossRef]
- Rocha-Ramírez, L.M.; Hernández-Ochoa, B.; Gómez-Manzo, S.; Marcial-Quino, J.; Cárdenas-Rodríguez, N.; Centeno-Leija, S.; García-Garibay, M. Impact of Heat-Killed Lactobacillus casei Strain IMAU60214 on the Immune Function of Macrophages in Malnourished Children. Nutrients 2020, 12, 2303. [Google Scholar] [CrossRef]
- van der Lelie, D.; Taghavi, S. COVID-19 and the Gut Microbiome: More than a Gut Feeling. mSystems 2020, 5, e00453-20. [Google Scholar] [CrossRef]
- Gou, W.; Fu, Y.; Yue, L.; Chen, G.; Cai, X.; Shuai, M.; Xu, F.; Yi, X.; Chen, H.; Zhu, Y.J.; et al. Gut microbiota may underlie the predisposition of healthy individuals to COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Ward, S.A.; Kalantar-Zadeh, K.; El-Omar, E.M. Considering the Effects of Microbiome and Diet on SARS-CoV-2 Infection: Nanotechnology Roles. ACS Nano 2020, 14, 5179–5182. [Google Scholar] [CrossRef]
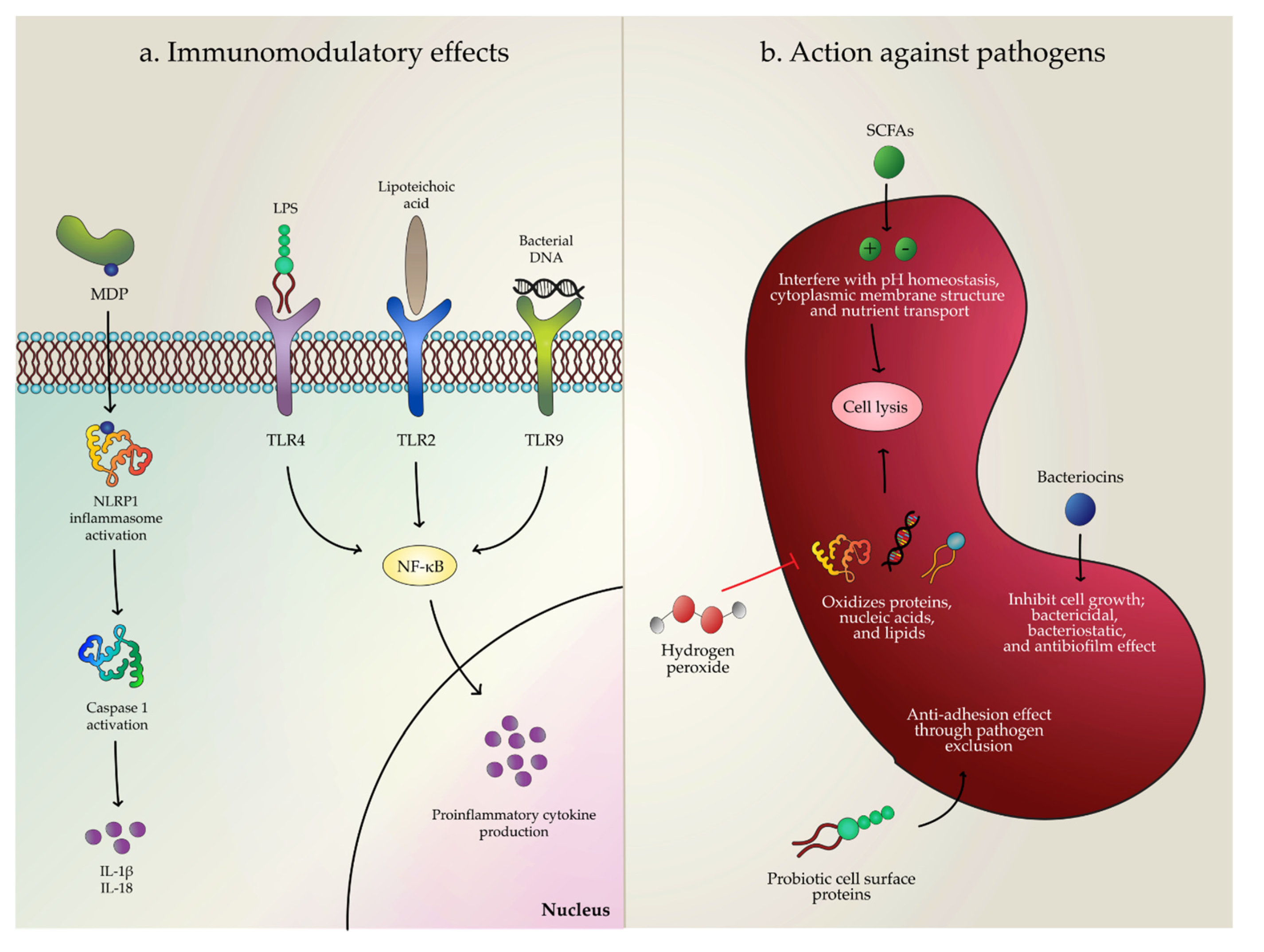
| Targeted Pathogen | Probiotic Bacteria | Preparation | Type of Study | Bioactivity or Effect | Reference |
|---|---|---|---|---|---|
| Escherichia coli | Lactobacillus acidophilus EMCC 1324 (La) *, Lactobacillus helveticus EMCC 1654 (Lh), Lactobacillus plantarum ss. plantarum EMCC 1027 (Lp), Lactobacillus rhamnosus EMCC 1105 (Lr), Bifidobacterium longum EMCC 1547 (BL), Bifidobacterium bifidum EMCC 1334 (Bb) | Cell-free supernatant (CFS; filtered) of MRS or milk fermented by probiotics | In vitro | CFS of MRS fermented by all probiotics resulted in inhibition of two multiresistant E. coli (WW1 and IC2) biofilm formation; CFSM of milk fermented by B. longum and L. helveticus reduced E. coli WW1 biofilm | [56] |
| Bifidobacterium bifidum BBA1 and Bifidobacterium crudilactis FR/62/B/3 | Cell-free supernatant (CFS; filtered) from probiotics grown in media supplemented with 3′-sialyllactose (major bovine milk oligosaccharide) | In vitro | Both CFSs induced a significant decrease in virulence gene expression of E. coli O157:H7 | [57] | |
| Bifidobacterium bifidum ATCC 29521 | Cell-free supernatant (CFS; filtered) of modified MRS fermented by probiotic | HeLa and macrophage cell lines | Downregulated virulence genes in E. coli O157:H7; inhibited its adhesion to HeLa cell line | [58] | |
| Lactobacillus acidophilus LB | Heat-inactivated probiotic with its spent culture supernatant (SCS) | Caco-2 cell culture | Inhibited E. coli ETEC adhesion | [59] | |
| Lactobacillus acidophilus LB | Heat-inactivated probiotic with its spent culture supernatant (SCS) | Caco-2 cell culture | Inhibited cell association and cell invasion of E. coli ETEC, DAEC, and EPEC | [60] | |
| Lactobacillus paracasei CNCM I-4034, Bifidobacterium breve CNCM I-4035, Lactobacillus rhamnosus CNCM I-4036) | Cell-free supernatant (CFS; filtered) of MRS fermented by probiotics | Monitoring bacterial growth | Inhibited the growth of E. coli ETEC and EPEC | [61] | |
| Lactobacillus acidophilus RY2, Lactobacillus salivarius MM1, Lactobacillus paracasei En4 | Spent culture supernatant (SCS) from bacteria grown in MRS + cystein | Colony count assay | Inhibited growth of E. coli ETEC | [62] | |
| Lactobacillus rhamnosus GG | Cell-free supernatant (CFS; filtered) of MRS fermented by probiotic | Caco-2 cell culture | Blocked adhesion, invasion, and translocation of E. coli K1 | [63] | |
| Lactobacillus rhamnosus GG | Novel secreted protein (HM0539) present in the CFS | Infected Sprague-Dawley neonatal rats | Promoted the development of neonatal intestinal defense and prevented against E. coli K1 pathogenesis | [64] | |
| Cronobacter sakazakii | Lactobacillus acidophilus INMIA 9602 Er 317/402 strain Narine | Heat-inactivated probiotic | Agar well diffusion method | Inhibited the growth of C. sakazakii in contaminated reconstructed powdered infant formula | [65] |
| Lactobacillus casei strain Shirota (Yakult); Lactobacillus sporongenes, Streptococcus faecalis, Clostridium butyricum, Bacillus mesentericus (Bifilac; cells); Streptococcus faecalis, Lactobacillus sporongenes, Clostridium butyricum, Bacillus mesentericus (Vibact; spores); Lactobacillus sporongenes (Caplac; spores) | Cell-free supernatant (CFS; filtered) from isolated probiotics | In vitro | Four probiotic-derived CFS (filtered and filtered + heat inactivated) possessed antimicrobial activity against C. sakazakii; a higher biofilm inhibitory activity (> 80%) was observed at initial stages of biofilm formation | [66] | |
| Clostridioides difficile | Enterococcus faecium, Lactococcus lactis | Cell-free supernatant (CFS; filtered) of MRS fermented by lactic acid bacteria | Caco-2 cell culture | Diminished the expression level of proinflammatory cytokines induced by the cell-free supernatant of C. difficile | [67] |
| Lactobacillus rhamnosus GG | Cell-free supernatant (CFS; filtered) and cell lysate (sonicated and filtered) of co-cultured toxigenic C. difficile with LGG | Vero cell culture | Decreased the cytotoxic effect of both extracellular and intracellular clostridial toxins resulting in up to 30% increase in cell viability | [68] | |
| Salmonella spp. | Bifidobacterium bifidum ATCC 29521 | Cell-free supernatant (CFS; filtered) of modified MRS fermented by probiotic | HeLa and macrophage cell lines | Downregulated virulence genes in S. Typhimurium; inhibited its adhesion to HeLa cell line; diminished the ability of Salmonella to survive and multiply within macrophages | [58] |
| Lactobacillus acidophilus LB | Heat-inactivated probiotic with its spent culture supernatant (SCS) | Caco-2 cell culture | Inhibited cell association and cell invasion of S. typhimurium | [60] | |
| Lactobacillus paracasei CNCM I-4034, Bifidobacterium breve CNCM I-4035, Lactobacillus rhamnosus CNCM I-4036) | Cell-free supernatant (CFS; filtered) of MRS fermented by probiotics | Monitoring bacterial growth | Inhibited the growth of Salmonella typhimurium and/or Salmonella typhi | [61] | |
| Lactobacillus paracasei B21060 | Supernatant of lactic acid bacteria grown in MRS | Ex vivo organ culture model | Inhibited the inflammatory potential of Salmonella and conditioned the epithelium against Salmonella invasion | [69] | |
| Lactobacillus paracasei CNCM I-4034 | Cell-free supernatant (CFS; filtered) of MRS fermented by probiotics | Coculture of dendritic and Caco-2 cells | Increased the production of pro-inflammatory cytokines in the presence of Salmonella typhi | [70] | |
| Influenza | Lactobacillus plantarum YML009 | Cell-free supernatant (CFS; filtered) of MRS fermented by lactic acid bacteria | MDCK cells and hemagglutination assay | Displayed a significant antiviral activity against influenza virus H1N1 and was more effective than Tamiflu | [71] |
| Leuconostoc mesenteroides YML003 | Cell-free supernatant (CFS; filtered) of MRS fermented by lactic acid bacteria | MDCK cells and hemagglutination assay | Demonstrated antiviral activity against low-pathogenic avian influenza (H9N2) | [72] | |
| Rotavirus | Bifidobacterium breve C50 and Streptococcus thermophilus 065 | Heat-treated fermented milk containing the prebiotic mixture scGOS/lcFOS | Rotavirus-infected Lewis rats | Reduced the incidence and severity of the rotaviral diarrhea; enhanced the host’s immune system | [18] |
| Lactobacilus rhamnosus GG | Orally administered heat-inactivated probiotic | Children under the age of 4 with rotavirus diarrhea | Rotavirus diarrhea recovery was equal for both viable and heat-inactivated probiotic-receiving groups | [73] | |
| Human immunodeficiency virus | Lactic acid bacteria isolated from human milk | Heat-inactivated lactic acid bacteria | TZM-bl cells | Significantly inhibited R5-tropic HIV-1 infection | [74] |
| Candida spp. | Four LAB isolated from honey (Lactobacillus plantarum HS, Pediococcus acidilactici HC, Lactobacillus curvatus HH, and Pediococcus pentosaceus HM) | Cell-free supernatant (CFS; filtered) of MRS fermented by probiotics | Microtiter plate method | Decreased the biofilm formation by Candida spp. | [75] |
| Lactobacillus crispatus B1-BC8, Lactobacillus gasseri BC9-BC14, Lactobacillus vaginalis BC15-BC17 | Cell-free supernatant (CFS; filtered) of MRS fermented by probiotics | HeLa cells | Reduced the adhesion of Candida spp. | [76] | |
| Lactobacillus casei ATCC 334, Lactobacilus rhamnosus GG (ATCC 53103) | Cell-free supernatant (CFS; filtered) of MRS fermented by lactic acid bacteria | Kirby–Bauer disk diffusion susceptibility test | Exhibited antifungal activity against blastoconidia and biofilm of Candida albicans | [77] | |
| Common pediatric infectious diseases with unknown cause | L. paracasei CBA L74 | Heat-treated fermented cow’s skim milk powder | Healthy children aged 1–4 years in daycare or preschool | Consumption was associated with a reduction of common infectious disease | [17] |
| L. paracasei CBA L74 | Heat-treated fermented cow’s milk or rice | Healthy children aged 1–4 years attending daycare or preschool | Prevented common pediatric infectious diseases in children after 3 months of consumption | [78] | |
| Bifidobacterium breve C50, Streptococcus thermophilus 065 | Heat-treated fermented infant formula | Healthy children aged 4–6 months | Did not prevent the incidence of acute diarrhea in healthy infants but reduced its severity | [79] | |
| Necrotizing enterocolitis | Six human strains of Bifidobacterium breve | Whey retentate from fermented cow’s milk | SPF C3H male mice, healthy volunteers aged 21–35 years | Led to a decrease in clostridia, bacilli, B. fragilis, and fecal pH, as well as to an increase in bifidobacteria | [80] |
| Lactobacillus paracasei CBA L74 | Heat-treated fermented milk powder | C57/BL6 mice | Strong anti-inflammatory activity in vitro and protected against colitis or enteric pathogens in vivo | [43] | |
| Lactobacillus paracasei MCC1849 | Orally administered heat-inactivated probiotic | Male SPF BALB/c mice | Enhanced antigen-specific IgA secretion and induced follicular helper T cells | [81] | |
| Lactobacillus rhamnosus HN001 | Orally administered UV-killed probiotic | Newborn mice and premature piglets | Attenuated necrotizing enterocolitis severity | [82] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantziari, A.; Salminen, S.; Szajewska, H.; Malagón-Rojas, J.N. Postbiotics against Pathogens Commonly Involved in Pediatric Infectious Diseases. Microorganisms 2020, 8, 1510. https://doi.org/10.3390/microorganisms8101510
Mantziari A, Salminen S, Szajewska H, Malagón-Rojas JN. Postbiotics against Pathogens Commonly Involved in Pediatric Infectious Diseases. Microorganisms. 2020; 8(10):1510. https://doi.org/10.3390/microorganisms8101510
Chicago/Turabian StyleMantziari, Anastasia, Seppo Salminen, Hania Szajewska, and Jeadran Nevardo Malagón-Rojas. 2020. "Postbiotics against Pathogens Commonly Involved in Pediatric Infectious Diseases" Microorganisms 8, no. 10: 1510. https://doi.org/10.3390/microorganisms8101510
APA StyleMantziari, A., Salminen, S., Szajewska, H., & Malagón-Rojas, J. N. (2020). Postbiotics against Pathogens Commonly Involved in Pediatric Infectious Diseases. Microorganisms, 8(10), 1510. https://doi.org/10.3390/microorganisms8101510






