Synergistic Effects of Probiotics and Phytobiotics on the Intestinal Microbiota in Young Broiler Chicken
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals, Rearing Conditions, and Experimental Diets
2.3. Sampling
2.4. Bacteria and Media
2.5. Feed Additives
2.6. 16S rDNA Sequencing and qPCR
2.7. Analysis of Bacterial Metabolites
2.8. Ex-vivo Survival of the ESBL-Producing E. coli Model Strain in Crop and Caecum
2.9. Statistical Analysis
3. Results
3.1. Qualitative Determination of the Intestinal Microbiota in Young Broiler Chickens
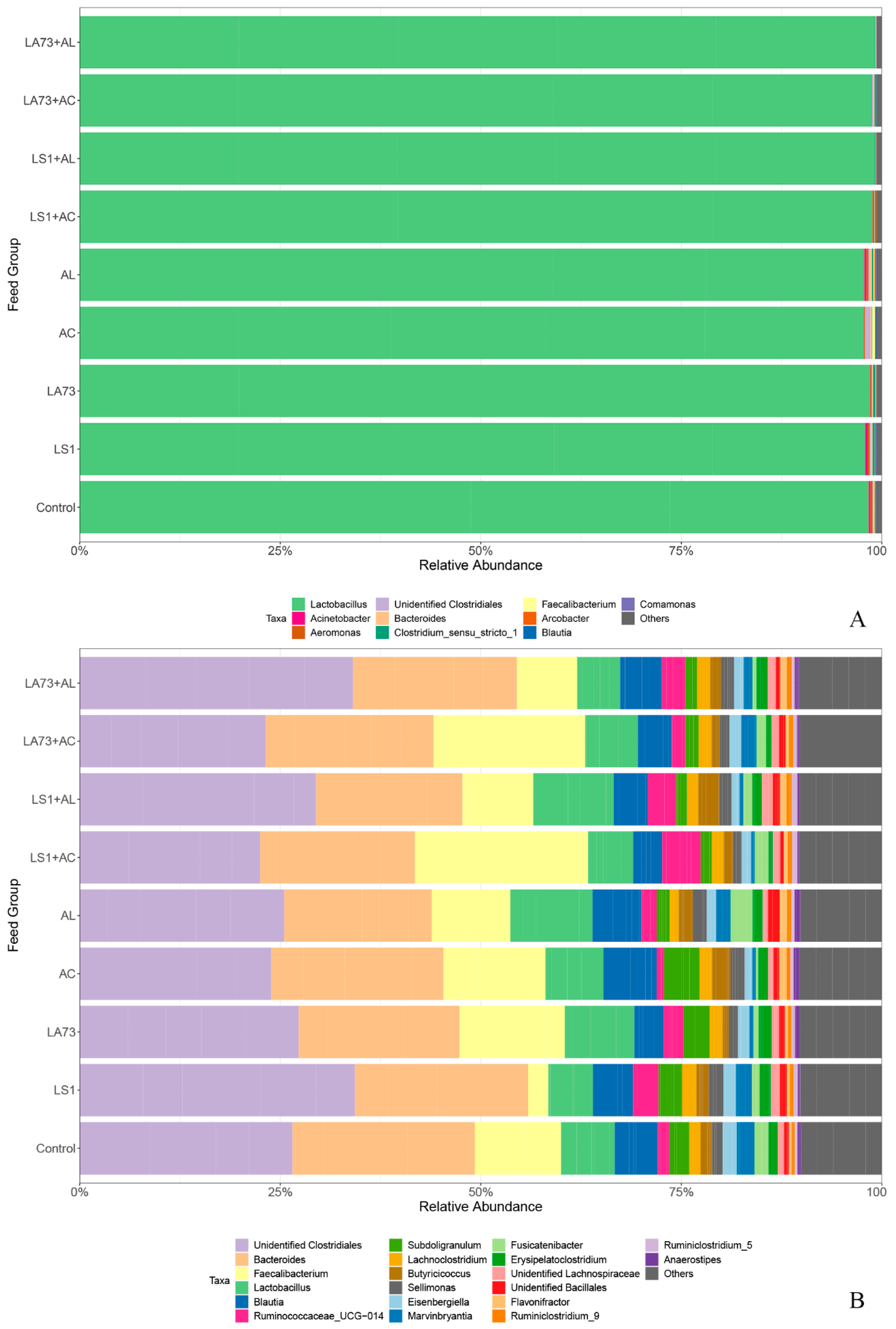
3.2. Impact of Probiotics and Phytobiotics on the Relative Composition of the Crop Microbiota
3.3. Impact of Probiotics and Phytobiotics on the Relative Composition of the Caecal Microbiota
3.4. Quantitative Determination of the Intestinal Microbiota in Young Broiler Chickens
3.5. Impact of Probiotics and Phytobiotics on Dominant Bacterial Groups and Species in the Crop
3.6. Impact of Probiotics and Phytobiotics on Dominant Bacterial Groups and Species in the Caecum
3.7. Bacterial Metabolism of the Intestinal Microbiota in Young Broiler Chickens
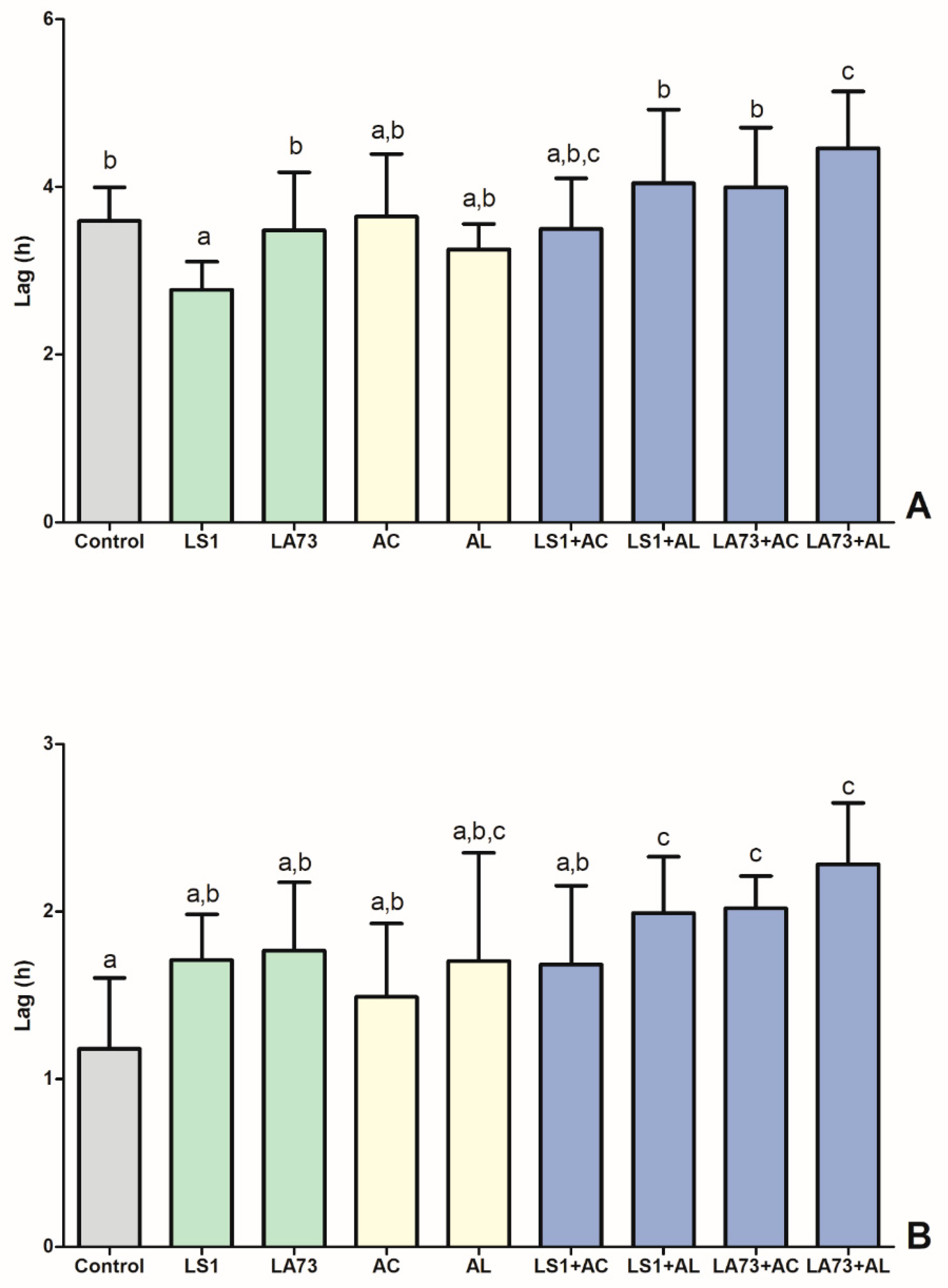
3.8. Ex-vivo Growth Response of an ESBL Producing E. coli Model Strain in Intestinal Samples
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO. Food Outlook Biannual Report on Global Food Markets; FAO: Rome, Italy, 2018; 104p. [Google Scholar]
- Saliu, E.-M.; Vahjen, W.; Zentek, J. Types and prevalence of extended–spectrum beta–lactamase producing Enterobacteriaceae in poultry. Anim. Health Res. Rev. 2017, 18, 46–57. [Google Scholar] [CrossRef]
- Costa, M.C.; Bessegatto, J.A.; Alfieri, A.A.; Weese, J.S.; Filho, J.A.B.; Oba, A. Different antibiotic growth promoters induce specific changes in the cecal microbiota membership of broiler chicken. PLoS ONE 2017, 12, e0171642. [Google Scholar] [CrossRef]
- Castanon, J.I.R. History of the Use of Antibiotic as Growth Promoters in European Poultry Feeds. Poult. Sci 2007, 86, 2466–2471. [Google Scholar] [CrossRef]
- Cheng, G.; Hao, H.; Xie, S.; Wang, X.; Dai, M.; Huang, L.; Yuan, Z. Antibiotic alternatives: The substitution of antibiotics in animal husbandry? Front. Microbiol. 2014, 5, 217. [Google Scholar] [CrossRef]
- Mehdi, Y.; Létourneau-Montminy, M.-P.; Gaucher, M.-L.; Chorfi, Y.; Suresh, G.; Rouissi, T.; Brar, S.K.; Côté, C.; Ramirez, A.A.; Godbout, S. Use of antibiotics in broiler production: Global impacts and alternatives. Anim. Nutr. 2018, 4, 170–178. [Google Scholar] [CrossRef]
- Fallah, R.; Kiani, A.; Azarfar, A. A review of the role of five kinds of alternatives to infeed antibiotics in broiler production. J. Vet. Med. Anim. Health 2013, 5. [Google Scholar]
- Clavijo, V.; Flórez, M.J.V. The gastrointestinal microbiome and its association with the control of pathogens in broiler chicken production: A review. Poult. Sci. 2018, 97, 1006–1021. [Google Scholar] [CrossRef]
- Rubio, L.A. Possibilities of early life programming in broiler chickens via intestinal microbiota modulation. Poult. Sci. 2018, 98, 695–706. [Google Scholar] [CrossRef]
- Robyn, J.; Rasschaert, G.; Messens, W.; Pasmans, F.; Heyndrickx, M. Screening for lactic acid bacteria capable of inhibiting Campylobacter jejuni in in vitro simulations of the broiler chicken caecal environment. Benef. Microbes 2012, 3, 299–308. [Google Scholar] [CrossRef]
- Meimandipour, A.; Shuhaimi, M.; Soleimani, A.F.; Azhar, K.; Hair-Bejo, M.; Kabeir, B.M.; Javanmard, A.; Muhammad Anas, O.; Yazid, A.M. Selected microbial groups and short-chain fatty acids profile in a simulated chicken cecum supplemented with two strains of Lactobacillus. Poult. Sci. 2010, 89, 470–476. [Google Scholar] [CrossRef]
- Baldwin, S.; Hughes, R.J.; Hao Van, T.T.; Moore, R.J.; Stanley, D. At-hatch administration of probiotic to chickens can introduce beneficial changes in gut microbiota. PLoS ONE 2018, 13, e0194825. [Google Scholar] [CrossRef] [PubMed]
- Lan, P.T.N.; Sakamoto, M.; Benno, Y. Effects of Two Probiotic Lactobacillus Strains on Jejunal and Cecal Microbiota of Broiler Chicken under Acute Heat Stress Condition as Revealed by Molecular Analysis of 16S rRNA Genes. Microbiol. Immunol. 2004, 48, 917–929. [Google Scholar] [CrossRef] [PubMed]
- Blajman, J.E.; Olivero, C.A.; Fusari, M.L.; Zimmermann, J.A.; Rossler, E.; Berisvil, A.P.; Romero Scharpen, A.; Astesana, D.M.; Soto, L.P.; Signorini, M.L.; et al. Impact of lyophilized Lactobacillus salivarius DSPV 001P administration on growth performance, microbial translocation, and gastrointestinal microbiota of broilers reared under low ambient temperature. Res. Vet. Sci. 2017, 114, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Saint-Cyr, M.J.; Haddad, N.; Taminiau, B.; Poezevara, T.; Quesne, S.; Amelot, M.; Daube, G.; Chemaly, M.; Dousset, X.; Guyard-Nicodème, M. Use of the potential probiotic strain Lactobacillus salivarius SMXD51 to control Campylobacter jejuni in broilers. Int. J. Food Microbiol. 2017, 247, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Hemarajata, P.; Versalovic, J. Effects of probiotics on gut microbiota: Mechanisms of intestinal immunomodulation and neuromodulation. Therap. Adv. Gastroenterol. 2013, 6, 39–51. [Google Scholar] [CrossRef]
- Prado-Rebolledo, O.F.; Delgado-Machuca, J.d.J.; Macedo-Barragan, R.J.; Garcia-Márquez, L.J.; Morales-Barrera, J.E.; Latorre, J.D.; Hernandez-Velasco, X.; Tellez, G. Evaluation of a selected lactic acid bacteria-based probiotic on Salmonella enterica serovar Enteritidis colonization and intestinal permeability in broiler chickens. Avian Pathol. 2017, 46, 90–94. [Google Scholar] [CrossRef]
- Johnson, N.C.; Kogut, H.M.; Genovese, K.; He, H.; Kazemi, S.; Arsenault, J.R. Administration of a Postbiotic Causes Immunomodulatory Responses in Broiler Gut and Reduces Disease Pathogenesis Following Challenge. Microorganisms 2019, 7, 268. [Google Scholar] [CrossRef]
- Reuter, G. The Lactobacillus and Bifidobacterium microflora of the human intestine: Composition and succession. Curr. Issues Intest. Microbiol. 2001, 2, 43–53. [Google Scholar]
- Yuki, N.; Shimazaki, T.; Kushiro, A.; Watanabe, K.; Uchida, K.; Yuyama, T.; Morotomi, M. Colonization of the stratified squamous epithelium of the nonsecreting area of horse stomach by lactobacilli. Appl. Environ. Microbiol. 2000, 66, 5030–5034. [Google Scholar] [CrossRef]
- Windisch, W.; Schedle, K.; Plitzner, C.; Kroismayr, A. Use of phytogenic products as feed additives for swine and poultry1. J. Anim. Sci. 2008, 86, E140–E148. [Google Scholar] [CrossRef]
- Kim, J.; Marshall, M.R.; Wei, C.-i. Antibacterial activity of some essential oil components against five foodborne pathogens. J. Agric. Food Chem. 1995, 43, 2839–2845. [Google Scholar] [CrossRef]
- Delaquis, P.J.; Stanich, K.; Girard, B.; Mazza, G. Antimicrobial activity of individual and mixed fractions of dill, cilantro, coriander and eucalyptus essential oils. Int. J. Food Microbiol. 2002, 74, 101–109. [Google Scholar] [CrossRef]
- Murugesan, G.R.; Syed, B.; Haldar, S.; Pender, C. Phytogenic Feed Additives as an Alternative to Antibiotic Growth Promoters in Broiler Chickens. Front. Vet. Sci. 2015, 2. [Google Scholar] [CrossRef]
- Karadas, F.; Pirgozliev, V.; Rose, S.P.; Dimitrov, D.; Oduguwa, O.; Bravo, D. Dietary essential oils improve the hepatic antioxidative status of broiler chickens. Br. Poult. Sci. 2014, 55, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Song, M.; Yun, W.; Lee, J.; Lee, C.; Kwak, W.; Han, N.; Kim, H.; Cho, J. Effects of oral administration of different dosages of carvacrol essential oils on intestinal barrier function in broilers. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1257–1265. [Google Scholar] [CrossRef]
- Yang, X.; Xin, H.; Yang, C.; Yang, X. Impact of essential oils and organic acids on the growth performance, digestive functions and immunity of broiler chickens. Anim. Nutr. 2018, 4, 388–393. [Google Scholar] [CrossRef]
- Cerisuelo, A.; Marín, C.; Sánchez-Vizcaíno, F.; Gómez, E.A.; de la Fuente, J.M.; Durán, R.; Fernández, C. The impact of a specific blend of essential oil components and sodium butyrate in feed on growth performance and Salmonella counts in experimentally challenged broilers. Poult. Sci. 2014, 93, 599–606. [Google Scholar] [CrossRef]
- Khattak, F.; Ronchi, A.; Castelli, P.; Sparks, N. Effects of natural blend of essential oil on growth performance, blood biochemistry, cecal morphology, and carcass quality of broiler chickens. Poult. Sci. 2014, 93, 132–137. [Google Scholar] [CrossRef]
- Shipradeep; Karmakar, S.; Sahay Khare, R.; Ojha, S.; Kundu, K.; Kundu, S. Development of probiotic candidate in combination with essential oils from medicinal plant and their effect on enteric pathogens: A review. Gastroenterol. Res. Pract. 2012, 2012, 457150. [Google Scholar] [CrossRef]
- de Vrese, M.; Schrezenmeir, J. Probiotics, Prebiotics, and Synbiotics. Adv. Biochem. Eng. Biotechnol. 2008, 111, 1–66. [Google Scholar]
- Smulikowska, S.; Czerwiński, J.; Mieczkowska, A. Effect of an organic acid blend and phytase added to a rapeseed cake-containing diet on performance, intestinal morphology, caecal microflora activity and thyroid status of broiler chickens. J. Anim. Physiol. Anim. Nutr. 2010, 94, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Czerwiński, J.; Højberg, O.; Smulikowska, S.; Engberg, R.M.; Mieczkowska, A. Influence of dietary peas and organic acids and probiotic supplementation on performance and caecal microbial ecology of broiler chickens. Br. Poult. Sci. 2010, 51, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Olukosi, O.A.; Dono, N.D. Modification of digesta pH and intestinal morphology with the use of benzoic acid or phytobiotics and the effects on broiler chicken growth performance and energy and nutrient utilization12. J. Anim. Sci. 2014, 92, 3945–3953. [Google Scholar] [CrossRef] [PubMed]
- Erdoğan, Z.; Erdoğan, S.; Aslantaş, Ö.; Çelik, S. ORIGINAL ARTICLE: Effects of dietary supplementation of synbiotics and phytobiotics on performance, caecal coliform population and some oxidant/antioxidant parameters of broilers. J. Anim. Physiol. Anim. Nutr. 2010, 94, e40–e48. [Google Scholar] [CrossRef]
- Heak, C.; Sukon, P.; Sornplang, P. Effect of direct-fed microbials on culturable gut microbiotas in broiler chickens: A meta-analysis of controlled trials. Asian-Australas J. Anim. Sci. 2018, 31, 1781–1794. [Google Scholar] [CrossRef]
- Kers, J.G.; Velkers, F.C.; Fischer, E.A.J.; Hermes, G.D.A.; Stegeman, J.A.; Smidt, H. Host and Environmental Factors Affecting the Intestinal Microbiota in Chickens. Front. Microbiol. 2018, 9, 235. [Google Scholar] [CrossRef]
- Hao, R.; Eva-Maria, S.; Jürgen, Z.; Farshad, G.B.; Wilfried, V. Screening of host specific lactic acid bacteria active against Escherichia coli from massive sample pools with a combination of in vitro and ex vivo methods. Front. Microbiol. 2019, 10, 2705. [Google Scholar] [CrossRef]
- Falgenhauer, L.; Imirzalioglu, C.; Ghosh, H.; Gwozdzinski, K.; Schmiedel, J.; Gentil, K.; Bauerfeind, R.; Kämpfer, P.; Seifert, H.; Michael, G.B.; et al. Circulation of clonal populations of fluoroquinolone-resistant CTX-M-15-producing Escherichia coli ST410 in humans and animals in Germany. Int. J. Antimicrob. Agents 2016, 47, 457–465. [Google Scholar] [CrossRef]
- Ren, H.; Zentek, J.; Vahjen, W. Optimization of Production Parameters for Probiotic Lactobacillus Strains as Feed Additive. Molecules 2019, 24, 3286. [Google Scholar] [CrossRef]
- Bushnell, B.; Rood, J.; Singer, E. BBMerge – Accurate paired shotgun read merging via overlap. PLoS ONE 2017, 12, e0185056. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, P.; Parfrey, L.W.; Yarza, P.; Gerken, J.; Pruesse, E.; Quast, C.; Schweer, T.; Peplies, J.; Ludwig, W.; Glöckner, F.O. The SILVA and “All-species Living Tree Project (LTP)” taxonomic frameworks. Nucleic Acids Res. 2014, 42, D643–D648. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581. [Google Scholar] [CrossRef] [PubMed]
- Huse, S.M.; Huber, J.A.; Morrison, H.G.; Sogin, M.L.; Welch, D.M. Accuracy and quality of massively parallel DNA pyrosequencing. Genome Biol. 2007, 8, R143. [Google Scholar] [CrossRef]
- Weiss, S.; Xu, Z.Z.; Peddada, S.; Amir, A.; Bittinger, K.; Gonzalez, A.; Lozupone, C.; Zaneveld, J.R.; Vázquez-Baeza, Y.; Birmingham, A.; et al. Normalization and microbial differential abundance strategies depend upon data characteristics. Microbiome 2017, 5, 27. [Google Scholar] [CrossRef]
- Berthold, M.R.; Cebron, N.; Dill, F.; Gabriel, T.R.; Kötter, T.; Meinl, T.; Ohl, P.; Thiel, K.; Wiswedel, B. KNIME - the Konstanz information miner: Version 2.0 and beyond. ACM SIGKDD Explor. Newsl. 2009, 11, 26–31. [Google Scholar] [CrossRef]
- Kröger, S.; Vahjen, W.; Zentek, J. Influence of lignocellulose and low or high levels of sugar beet pulp on nutrient digestibility and the fecal microbiota in dogs. J. Anim. Sci. 2017, 95, 1598–1605. [Google Scholar] [CrossRef]
- Starke, I.C.; Zentek, J.; Vahjen, W. Ex vivo-growth response of porcine small intestinal bacterial communities to pharmacological doses of dietary zinc oxide. PLoS ONE 2013, 8, e56405. [Google Scholar] [CrossRef]
- Patterson, J.A.; Burkholder, K.M. Application of prebiotics and probiotics in poultry production. Poult. Sci. 2003, 82, 627–631. [Google Scholar] [CrossRef]
- Hadrich, D. Microbiome Research Is Becoming the Key to Better Understanding Health and Nutrition. Front. Genet. 2018, 9, 212. [Google Scholar] [CrossRef]
- Loh, T.C.; Thanh, N.T.; Foo, H.L.; Hair-Bejo, M.; Azhar, B.K. Feeding of different levels of metabolite combinations produced by Lactobacillus plantarum on growth performance, fecal microflora, volatile fatty acids and villi height in broilers. Anim. Sci. J. 2010, 81, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Peng, Q.; Zeng, X.F.; Zhu, J.L.; Wang, S.; Liu, X.T.; Hou, C.L.; Thacker, P.A.; Qiao, S.Y. Effects of dietary Lactobacillus plantarum B1 on growth performance, intestinal microbiota, and short chain fatty acid profiles in broiler chickens. Poult. Sci. 2016, 95, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Liu, H.; Wang, S.; Wu, J.; Kluenter, A.-M. Potential of essential oils for poultry and pigs. Anim. Nutr. 2018, 4, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.M.; Lee, T.T. Immunomodulatory effects of phytogenics in chickens and pigs—A review. Asian-Australas J. Anim. Sci. 2018, 31, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Hashemipour, H.; Kermanshahi, H.; Golian, A.; Veldkamp, T. Effect of thymol and carvacrol feed supplementation on performance, antioxidant enzyme activities, fatty acid composition, digestive enzyme activities, and immune response in broiler chickens. Poult. Sci. 2013, 92, 2059–2069. [Google Scholar] [CrossRef]
- Venkitanarayanan, K.; Kollanoor-Johny, A.; Darre, M.J.; Donoghue, A.M.; Donoghue, D.J. Use of plant-derived antimicrobials for improving the safety of poultry products1. Poult. Sci. 2013, 92, 493–501. [Google Scholar] [CrossRef]
- Kohlert, C.; Rensen, I.; Maerz, R.; Schindler, G.; Graefe, E.U.; Veit, M. Bioavailability and Pharmacokinetics of Natural Volatile Terpenes in Animals and Humans. Planta Med. 2000, 66, 495–505. [Google Scholar] [CrossRef]
- Michiels, J.; Missotten, J.; Dierick, N.; Fremaut, D.; Maene, P.; De Smet, S. In vitro degradation and in vivo passage kinetics of carvacrol, thymol, eugenol and trans-cinnamaldehyde along the gastrointestinal tract of piglets. J. Sci. Food Agric. 2008, 88, 2371–2381. [Google Scholar] [CrossRef]
- Svihus, B. Function of the digestive system1. J. Appl. Poult. Res. 2014, 23, 306–314. [Google Scholar] [CrossRef]
- Lu, J.; Idris, U.; Harmon, B.; Hofacre, C.; Maurer, J.J.; Lee, M.D. Diversity and succession of the intestinal bacterial community of the maturing broiler chicken. Appl. Environ. Microbiol. 2003, 69, 6816–6824. [Google Scholar] [CrossRef]
- Fuller, R.; Brooker, B.E. Lactobacilli which attach to the crop epithelium of the fowl. Am. J. Clin. Nutr. 1974, 27, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Abbas Hilmi, H.T.; Surakka, A.; Apajalahti, J.; Saris, P.E.J. Identification of the Most Abundant Lactobacillus Species in the Crop of 1- and 5- Week-Old Broiler Chickens. Appl. Environ. Microbiol. 2007, 73, 7867–7873. [Google Scholar] [CrossRef] [PubMed]
- Peinado, M.J.; Echávarri, A.; Ruiz, R.; Suárez-Pereira, E.; Ortiz Mellet, C.; García Fernández, J.M.; Rubio, L.A. Effects of inulin and di-d-fructose dianhydride-enriched caramels on intestinal microbiota composition and performance of broiler chickens. Animal 2013, 7, 1779–1788. [Google Scholar] [CrossRef] [PubMed]
- Rada, V.; Marounek, M.; Rychlý, I.; Šantrŭčková, D.; Vořišek, K. Effect of Lactobacillus salivarius administration on microflora in the crop and caeca of broiler chickens. J. Anim. Feed Sci 1995, 4, 161–170. [Google Scholar] [CrossRef]
- Huang, T.; Gao, B.; Chen, W.-L.; Xiang, R.; Yuan, M.-G.; Xu, Z.-H.; Peng, X.-Y. Temporal Effects of High Fishmeal Diet on Gut Microbiota and Immune Response in Clostridium perfringens-Challenged Chickens. Front. Microbiol. 2018, 9, 2754. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-Y.; Lee, Y.; Lu, H.; Chou, C.-H.; Wang, C. Analysis of gut microbiota and the effect of lauric acid against necrotic enteritis in Clostridium perfringens and Eimeria side-by-side challenge model. PLoS ONE 2019, 14, e0205784. [Google Scholar] [CrossRef]
- Zhan, H.Q.; Dong, X.Y.; Li, L.L.; Zheng, Y.X.; Gong, Y.J.; Zou, X.T. Effects of dietary supplementation with Clostridium butyricum on laying performance, egg quality, serum parameters, and cecal microflora of laying hens in the late phase of production. Poult. Sci. 2018, 98, 896–903. [Google Scholar] [CrossRef]
- Du, E.; Gan, L.; Li, Z.; Wang, W.; Liu, D.; Guo, Y. In vitro antibacterial activity of thymol and carvacrol and their effects on broiler chickens challenged with Clostridium perfringens. J. Anim. Sci. Biotechnol. 2015, 6, 58. [Google Scholar] [CrossRef]
- Sfeir, J.; Lefrançois, C.; Baudoux, D.; Derbré, S.; Licznar, P. In Vitro Antibacterial Activity of Essential Oils against Streptococcus pyogenes. Evid. Based Complement. Alternat. Med. 2013, 2013, 269161. [Google Scholar] [CrossRef]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Lustri, B.C.; Sperandio, V.; Moreira, C.G. Bacterial Chat: Intestinal Metabolites and Signals in Host-Microbiota-Pathogen Interactions. Infect. Immun. 2017, 85, e00476-00417. [Google Scholar] [CrossRef] [PubMed]
- Tekiner, İ.H.; Özpınar, H. Occurrence and characteristics of extended spectrum beta-lactamases-producing Enterobacteriaceae from foods of animal origin. Braz. J. Microbiol. 2016, 47, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Kostrzynska, M.; Bachand, A. Use of microbial antagonism to reduce pathogen levels on produce and meat products: A review. Can. J. Microbiol. 2006, 52, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Rolfe, R.D. The Role of Probiotic Cultures in the Control of Gastrointestinal Health. J. Nutr. 2000, 130, 396S–402S. [Google Scholar] [CrossRef]
- Fluit, A.C.; Schmitz, F.J. Class 1 Integrons, Gene Cassettes, Mobility, and Epidemiology. Eur. J. Clin. Microbiol. Infect. Dis. 1999, 18, 761–770. [Google Scholar] [CrossRef]
- Gallazzi, D.; Giardini, A.; Mangiagalli, G.M.; Marelli, S.; Ferrazzi, V.; Orsi, C.; Cavalchini, G.L. Effects of Lactobacillus acidophilus D2/CSL on laying hen performance. Ital. J. Anim. Sci. 2008, 7, 27–37. [Google Scholar] [CrossRef]
- Ercolini, D.; Fogliano, V. Food Design to Feed the Human Gut Microbiota. J. Agric. Food Chem. 2018, 66, 3754–3758. [Google Scholar] [CrossRef]
- Gao, P.; Ma, C.; Sun, Z.; Wang, L.; Huang, S.; Su, X.; Xu, J.; Zhang, H. Feed-additive probiotics accelerate yet antibiotics delay intestinal microbiota maturation in broiler chicken. Microbiome 2017, 5, 91. [Google Scholar] [CrossRef]
| Ingredient (%) | |
|---|---|
| Soybean Meal (49% CP) | 32.33 |
| Maize | 32.03 |
| Wheat | 24.78 |
| Soy oil | 5.95 |
| Limestone | 1.46 |
| Monocalcium Phosphate | 1.84 |
| Vitamin and Mineral Premix | 1.20 |
| Salt | 0.10 |
| DL-Methionine | 0.18 |
| L-Lysine | 0.13 |
| Nutrient Composition | |
| Crude Protein (%) | 22.00 |
| Crude Fat (%) | 8.19 |
| Crude Fiber (%) | 2.42 |
| Methionine (%) | 0.51 |
| Lysine (%) | 1.28 |
| Threonine (%) | 0.84 |
| Calcium (%) | 0.96 |
| Phosphorus (%) | 0.80 |
| AMEN (MJ/kg)3 | 12.6 |
| Putative species name | Control | LS1 | LA73 | Formulation C | Formulation L | LS1 & Formulation C | LS1 & Formulation L | LA73 & Formulation C | LA73 & Formulation L | Pooled SEM | p-value1 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| L. salivarius | 33.8a,b | 46.5a,b | 24.9a,b | 68.2b,c | 26.3a,b | 57.1b,c | 71.9c | 18.1a | 37.6b | 3.21 | 0.002 |
| L. agilis | 16.1a,b | 9.1a | 38.1b | 4.5a | 38.3b | 13.3a,b | 5.7a | 49.4c | 20.8a,b | 0.97 | 0.003 |
| L. crispatus | 18.2c | 10.3b,c | 9.9b,c | 8.7b,c | 5.7a | 7.1a | 3.2a | 13.8b,c | 8.4b | 0.68 | 0.049 |
| L. reuteri | 15.3 | 18.0 | 15.2 | 6.2 | 14.0 | 10.9 | 8.2 | 7.2 | 16.5 | 0.61 | 0.425 |
| L. johnsonii | 8.7 | 9.8 | 8.0 | 9.5 | 11.6 | 9.8 | 8.7 | 5.2 | 11.1 | 1.39 | 0.593 |
| L. kitasatonis | 4.4 | 4.3 | 1.1 | 0.6 | 1.9 | 0.4 | 1.2 | n.d. | 0.7 | 3.73 | 0.814 |
| L. vaginalis | 3.4 | 1.9 | 2.9 | 2.4 | 2.1 | 1.5 | 1.1 | 6.3 | 4.7 | 0.43 | 0.120 |
| Control | LS1 | LA73 | Formulation C | Formulation L | LS1 & Formulation C | LS1 & Formulation L | LA73 & Formulation C | LA73 & Formulation L | Pooled SEM | p- value1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Lactobacillus spp. | 9.84 | 9.81 | 10.07 | 9.81 | 9.77 | 9.81 | 10.01 | 9.89 | 10.00 | 0.04 | 0.167 |
| L. salivarius | 9.14a | 9.55b | 9.39a.b | 9.58b.c | 9.25a | 9.6b.c | 9.92c | 9.20a | 9.53a.b | 0.05 | 0.016 |
| L. agilis | 8.87b | 8.83b | 9.30d | 8.19a | 9.14c | 8.73b | 8.61b | 9.40d | 9.13c | 0.11 | 0.016 |
| L. reuteri | 9.19 | 9.23 | 9.61 | 8.91 | 9.08 | 9.11 | 9.23 | 9.29 | 9.52 | 0.07 | 0.338 |
| L. acidophilus | 8.45 | 8.33 | 8.58 | 7.99 | 8.12 | 7.98 | 8.08 | 7.91 | 8.10 | 0.11 | 0.768 |
| Clostridial Cluster XIVa | 8.38b | 8.41b | 8.45b | 8.60b | 8.43b | 7.76a | 7.88a | 8.49b | 8.22b | 0.09 | 0.040 |
| Clostridial Cluster IV | 8.08 | 7.89 | 8.11 | 8.16 | 7.86 | 7.36 | 7.43 | 7.92 | 7.72 | 0.09 | 0.206 |
| Clostridial Cluster I | 7.41b | 7.32b | 7.81c | 6.95a.b | 7.27b | 6.99b | 7.27b | 7.03b | 6.62a | 0.08 | 0.016 |
| BPP-Cluster2 | 8.48 | 8.36 | 8.56 | 8.22 | 8.37 | 7.95 | 8.16 | 8.29 | 7.82 | 0.07 | 0.338 |
| Enterobacteria3 | 8.83a.b | 8.42a | 8.94b | 8.64a | 8.38a | 8.30a | 8.47a | 8.62a.b | 8.45a | 0.04 | 0.004 |
| Escherichia group | 6.89a | 6.66a.b | 7.28c | 6.93a.b | 6.27a | 6.06a | 6.62a | 7.01b.c | 6.40a | 0.08 | 0.018 |
| Int14 | 7.77b | 7.37b | 7.98c | 7.27b | 7.34b | 7.17b | 7.27b | 6.99a.b | 6.72a | 0.08 | 0.042 |
| Control | LS1 | LA73 | Formulation C | Formulation L | LS1 & Formulation C | LS1 & Formulation L | LA73 & Formulation C | LA73 & Formulation L | Pooled SEM | p- Value1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Clostridial Cluster XIVa | 10.96 | 10.97 | 10.85 | 11.15 | 11.09 | 11.08 | 11.17 | 10.93 | 11.11 | 0.05 | 0.639 |
| Clostridial Cluster IV | 10.39 | 10.28 | 10.51 | 10.72 | 10.61 | 10.73 | 10.67 | 10.47 | 10.30 | 0.05 | 0.134 |
| Clostridial Cluster I | 6.45 | 6.13 | 5.93 | 6.33 | 6.15 | 6.76 | 6.70 | 5.60 | 6.03 | 0.14 | 0.383 |
| BPP-Cluster2 | 10.72 | 10.77 | 10.72 | 10.86 | 10.72 | 10.72 | 10.86 | 10.66 | 10.69 | 0.03 | 0.650 |
| Lactobacillus spp. | 9.63 | 9.61 | 9.82 | 9.73 | 9.83 | 9.69 | 9.94 | 9.61 | 9.67 | 0.04 | 0.605 |
| Enterobacteria3 | 10.51 | 10.49 | 10.43 | 10.43 | 10.36 | 10.35 | 10.75 | 10.30 | 10.53 | 0.06 | 0.857 |
| Escherichia group | 8.45 | 8.43 | 8.32 | 8.21 | 8.53 | 8.13 | 8.13 | 8.20 | 8.43 | 0.07 | 0.910 |
| L. salivarius | 8.78 | 8.94 | 8.95 | 9.20 | 9.14 | 9.17 | 9.32 | 8.70 | 8.94 | 0.05 | 0.130 |
| L. agilis | 9.42 | 9.17 | 9.54 | 9.44 | 9.47 | 9.50 | 9.75 | 9.21 | 9.51 | 0.07 | 0.392 |
| L. reuteri | 8.79 | 8.71 | 9.10 | 8.27 | 8.86 | 8.18 | 8.12 | 9.14 | 9.05 | 0.07 | 0.774 |
| L. acidophilus | 8.26 | 7.48 | 8.21 | 8.12 | 8.49 | 8.27 | 8.27 | 7.67 | 7.79 | 0.11 | 0.620 |
| Int14 | 7.94 | 7.69 | 7.35 | 7.76 | 7.75 | 7.80 | 7.27 | 7.58 | 7.95 | 0.11 | 0.177 |
| Control | LS1 | LA73 | Formulation C | Formulation L | LS1 & Formulation C | LS1 & Formulation L | LA73 & Formulation C | LA73 & Formulation L | Pooled SEM | p-Value1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| L-lactate | 19.8b | 15.2a,b | 21.1b | 11.2a | 14.2a | 19.5b | 25.0b | 16.9b | 29.5c | 1.10 | 0.008 |
| D-lactate | 11.5b | 3.9a | 6.6a,b,c | 2.2a | 4.2 a | 4.9a,b | 7.6b | 4.8a | 13.2c | 0.70 | 0.029 |
| total Lactate | 31.3b.c | 19.1b | 27.8b,c | 13.4a | 18.3a | 24.4a,b | 32.6b | 21.7b | 42.7c | 1.69 | 0.010 |
| Acetate | 5.9 | 4.4 | 6.0 | 2.4 | 3.3 | 4.2 | 4.1 | 4.1 | 6.7 | 0.49 | 0.638 |
| Propionate | 1.5 | 1.8 | 2.1 | 2.0 | 1.9 | 1.9 | 1.9 | 1.7 | 1.7 | 0.04 | 0.218 |
| n-butyrate | 0.1 | n.d.2 | 0.1 | n.d. | 1.8 | n.d. | n.d. | n.d. | n.d. | 0.19 | 0.332 |
| i-valerate | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.317 | |
| n-valerate | n.d. | n.d. | 0.02 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | 0.277 | |
| Total SCFA | 7.5 | 6.3 | 8.2 | 4.3 | 6.1 | 6.1 | 6.0 | 5.8 | 8.4 | 0.51 | 0.719 |
| Total Metabolites3 | 40.4b | 25.4a,b | 35.9b | 18.9a | 24.5a,b | 33.1b | 39.5b | 29.0a,b | 53.7c | 2.20 | 0.020 |
| Control | LS1 | LA73 | Formulation C | Formulation L | LS1 & Formulation C | LS1 & Formulation L | LA73 & Formulation C | LA73 & Formulation L | Pooled SEM | p-Value1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetate | 39.9b | 55.8c | 32.1a | 30.7a | 34.8a,b | 53.1c | 43.6b | 42.6b | 44.0b | 2.01 | 0.044 |
| Propionate | 5.0 | 7.8 | 6.1 | 5.2 | 4.2 | 5.7 | 5.2 | 5.9 | 7.0 | 0.28 | 0.142 |
| i-butyrate | 1.9 | 2.1 | 0.8 | 0.6 | 1.2 | 1.1 | 0.4 | 0.5 | 0.6 | 0.22 | 0.963 |
| n-butyrate | 8.7 | 7.7 | 5.8 | 7.7 | 6.0 | 9.4 | 8.7 | 10.1 | 6.2 | 0.50 | 0.548 |
| i-valerate | 0.5 | 0.3 | 0.3 | 0.4 | 0.2 | 0.4 | 0.2 | 0.3 | 0.2 | 0.03 | 0.684 |
| n-valerate | 0.4 | 0.4 | 0.6 | 0.6 | 0.3 | 0.4 | 0.3 | 0.2 | 0.3 | 0.04 | 0.504 |
| BCFA2 | 2.0 | 2.4 | 1.1 | 0.8 | 1.2 | 1.5 | 0.6 | 0.6 | 0.7 | 0.21 | 0.873 |
| Total SCFA | 55.9 | 74.1 | 45.6 | 44.9 | 46.4 | 70.1 | 58.4 | 59.3 | 58.2 | 2.50 | 0.103 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, H.; Vahjen, W.; Dadi, T.; Saliu, E.-M.; Boroojeni, F.G.; Zentek, J. Synergistic Effects of Probiotics and Phytobiotics on the Intestinal Microbiota in Young Broiler Chicken. Microorganisms 2019, 7, 684. https://doi.org/10.3390/microorganisms7120684
Ren H, Vahjen W, Dadi T, Saliu E-M, Boroojeni FG, Zentek J. Synergistic Effects of Probiotics and Phytobiotics on the Intestinal Microbiota in Young Broiler Chicken. Microorganisms. 2019; 7(12):684. https://doi.org/10.3390/microorganisms7120684
Chicago/Turabian StyleRen, Hao, Wilfried Vahjen, Temesgen Dadi, Eva-Maria Saliu, Farshad Goodarzi Boroojeni, and Jürgen Zentek. 2019. "Synergistic Effects of Probiotics and Phytobiotics on the Intestinal Microbiota in Young Broiler Chicken" Microorganisms 7, no. 12: 684. https://doi.org/10.3390/microorganisms7120684
APA StyleRen, H., Vahjen, W., Dadi, T., Saliu, E.-M., Boroojeni, F. G., & Zentek, J. (2019). Synergistic Effects of Probiotics and Phytobiotics on the Intestinal Microbiota in Young Broiler Chicken. Microorganisms, 7(12), 684. https://doi.org/10.3390/microorganisms7120684





