Preservation, Characterization and Exploitation of Microbial Biodiversity: The Perspective of the Italian Network of Culture Collections
Abstract
1. Introduction
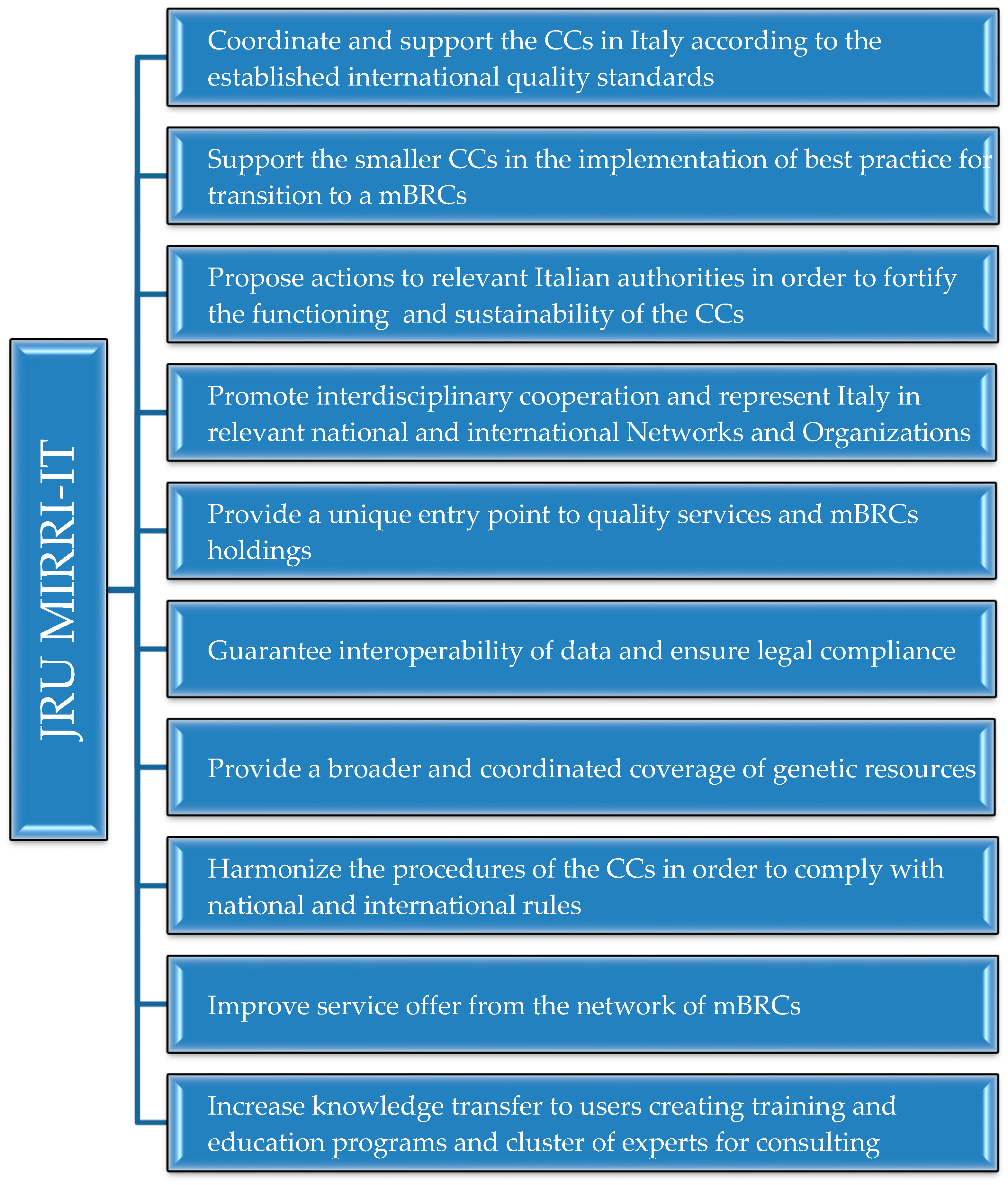
2. Culture Collections and Biological Research Centre Networks: the European and Italian Scenarios
3. Services and Expertise of mBRCs
3.1. Preservation of Biological Resources
3.2. Characterization of Biological Resources
3.3. Databases and Data Sharing
3.3.1. Data Modelling and Interoperability
3.3.2. Recommendations for Collections
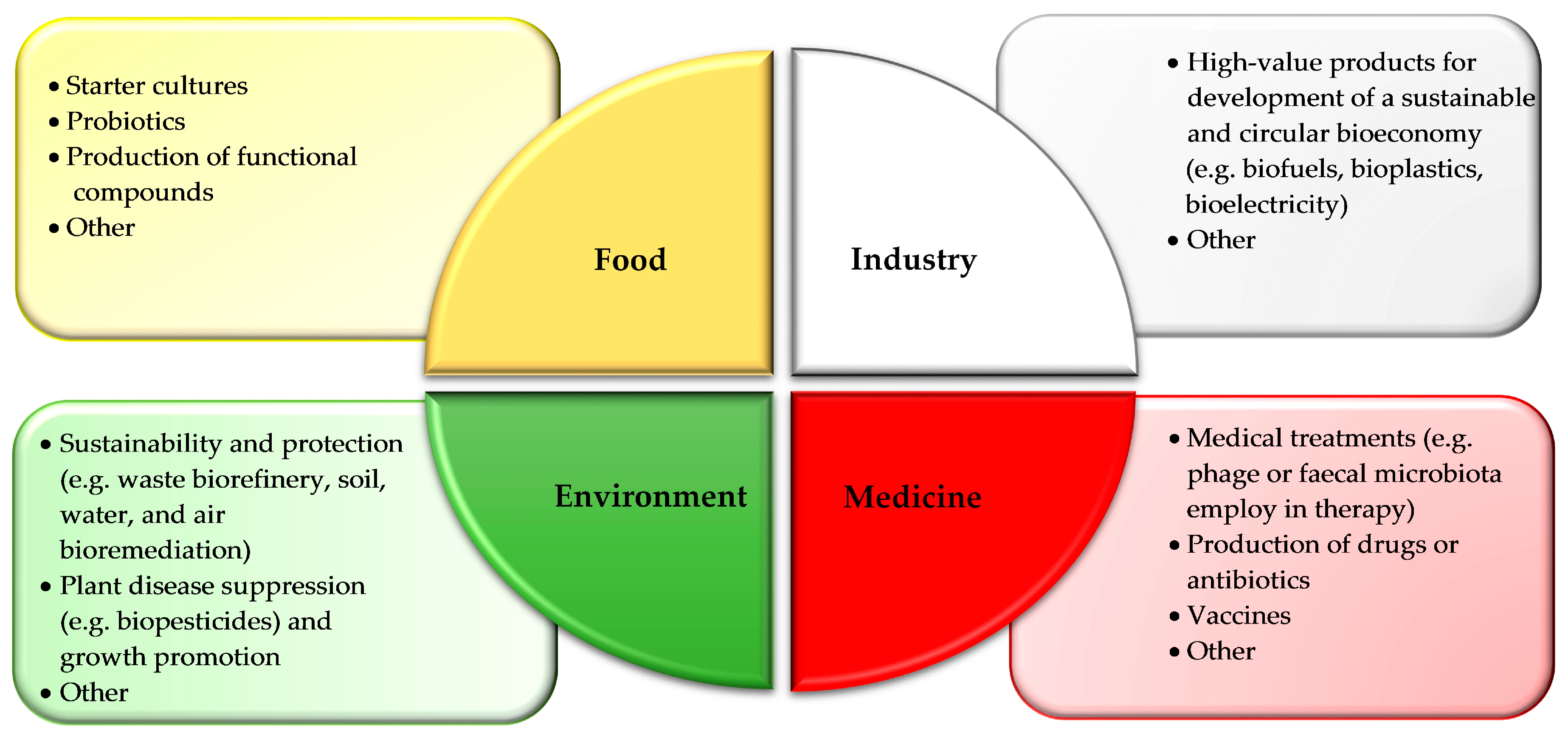
4. Exploitation of Biological Resources and Connection Between Industry and mBRC: A Focus on Food Applications
5. mBRC Regulations and Guidelines
5.1. Quality Management System
5.2. Guidelines on Biosecurity and Biosafety
6. Conclusions and Future Prospects
Funding
Acknowledgments
Conflicts of Interest
References
- Antunes, A.; Stackebrandt, E.; Lima, N. Fueling the Bio-economy: European Culture Collections and Microbiology Education and Training. Trends Microbiol. 2016, 24, 77–79. [Google Scholar] [CrossRef] [PubMed]
- Overmann, J. Significance and future role of microbial resource centers. Syst. Appl. Microbiol. 2015, 38, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Çaktü, K.; Türkoǧlu, E.A. Microbial culture collections: The essential resources for life. Gazi Univ. J. Sci. 2011, 24, 175–180. [Google Scholar]
- Stackebrandt, E.; Smith, D.; Casaregola, S.; Varese, G.C.; Verkleij, G.; Lima, N.; Bridge, P. Deposit of microbial strains in public service collections as part of the publication process to underpin good practice in science. Springerplus 2014, 3, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.; McCluskey, K.; Stackebrandt, E. Investment into the future of microbial resources: Culture collection funding models and BRC business plans for biological resource centres. Springerplus 2014, 3, 81. [Google Scholar] [CrossRef] [PubMed]
- Janssens, D.; Arahal, D.R.; Bizet, C.; Garay, E. The role of public biological resource centers in providing a basic infrastructure for microbial research. Res. Microbiol. 2010, 161, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Boundy-Mills, K.; Hess, M.; Bennett, A.R.; Ryan, M.; Kang, S.; Nobles, D.; Eisen, J.A.; Inderbitzin, P.; Sitepu, I.R.; Torok, T.; et al. The United States Culture Collection Network (USCCN): Enhancing microbial genomics research through living microbe culture collections. Appl. Environ. Microbiol. 2015, 81, 5671–5674. [Google Scholar] [CrossRef]
- McCluskey, K. A Review of Living Collections with Special Emphasis on Sustainability and Its Impact on Research Across Multiple Disciplines. Biopreserv. Biobank. 2017, 15, 20–30. [Google Scholar] [CrossRef]
- Ryan, M.J.; McCluskey, K.; Verkleij, G.; Robert, V.; Smith, D. Fungal biological resources to support international development: Challenges and opportunities. World J. Microbiol. Biotechnol. 2019, 7, 1–13. [Google Scholar] [CrossRef]
- Lee, S.; Jung, P.E.; Lee, Y. Publicly-funded biobanks and networks in East Asia. SpringerPlus 2016, 5, 1080. [Google Scholar] [CrossRef]
- Santos, C.; Durán, P.; Tortela, G.; Barrientos, L.; Briceño, G.; Rodriguez, R.; Godoy, P.; Alvarez-Duarte, E.; Cruz Choappa, R.; Silva, V.; et al. The Chilean Network of Microbial Culture Collections: Establishment and Operation. Bol. Micol. 2016, 31, 44–50. [Google Scholar] [CrossRef]
- Martin, D.; Stackebrandt, E.; Smith, D. MIRRI Promoting Quality Management Systems for Microbiology. Microbiol. Perspect. 2015, 2, 278–287. [Google Scholar]
- Schüngel, M.; Stackebrandt, E. Microbial Resource Research Infrastructure (MIRRI): Infrastructure to foster academic research and biotechnological innovation. Biotechnol. J. 2015, 10, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.; da Silva, M.; Jackson, J.; Lyal, C. Explanation of the Nagoya Protocol on access and benefit sharing and its implication for microbiology. Microbiology 2017, 163, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Boundy-Mills, K.L.; Glantschnig, E.; Roberts, I.N.; Yurkov, A.; Casaregola, S.; Daniel, H.M.; Groenewald, M.; Turchetti, B. Yeast culture collections in the twenty-first century: New opportunities and challenges. Yeast 2016, 33, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Shouche, Y. Microbial Culture Collection (MCC) and International Depositary Authority (IDA) at National Centre for Cell Science, Pune. Indian J. Microbiol. 2014, 54, 129–133. [Google Scholar] [CrossRef]
- Gherna, R. Culture preservation. In Encyclopedia of Industrial Biotechnology: Bioprocess, Bioseparation, and Cell Technology; Flickinger, M.C., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2010; pp. 1–8. [Google Scholar]
- Officer, I.C. World Federation for Culture Collections Guidelines for the Establishment and Operation of Collections of Cultures of 2010; WFCC: Utrecht, The Netherlands, 2010; pp. 1–19. [Google Scholar]
- OECD Best Practice Guidelines for Biological Resource Centres. Available online: http://www.oecd.org/sti/emerging-tech/38777417.pdf (accessed on 30 October 2019).
- Smith, D. Culture Collections. Adv. Appl. Microbiol. 2012, 79, 73–118. [Google Scholar]
- De Paoli, P. Bio-banking in microbiology: From sample collection to epidemiology, diagnosis and research. FEMS Microbiol. Rev. 2005, 29, 897–910. [Google Scholar] [CrossRef]
- D’Elia, L.; Del Mondo, A.; Santoro, M.; De Natale, A.; Pinto, G.; Pollio, A. Microorganisms from harsh and extreme environments: A collection of living strains at ACUF (Naples, Italy). Ecol. Quest. 2018, 29, 63–74. [Google Scholar] [CrossRef]
- Morgan, C.A.; Herman, N.; White, P.A.; Vesey, G. Preservation of micro-organisms by drying; A review. J. Microbiol. Methods 2006, 66, 183–193. [Google Scholar] [CrossRef]
- Smith, D.; Ryan, M. Implementing best practices and validation of cryopreservation techniques for microorganisms. Sci. World J. 2012, 2012, 805659. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.; Ryan, M.J. The impact of OECD best practice on the validation of cryopreservation techniques for microorganisms. CryoLetters 2008, 29, 63–72. [Google Scholar] [PubMed]
- Becker, P.; Bosschaerts, M.; Chaerle, P.; Daniel, H.-M.; Hellemans, A.; Olbrechts, A.; Rigouts, L.; Wilmotte, A.; Hendrickx, M. Public microbial resources centres: Key hubs for FAIR microorganisms and genetic materials. Appl. Environ. Microbiol. 2019, 85, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Prakash, O.; Nimonkar, Y.; Shouche, Y.S. Practice and prospects of microbial preservation. FEMS Microbiol. Lett. 2013, 339, 1–9. [Google Scholar] [CrossRef] [PubMed]
- De Vero, L.; Gullo, M.; Giudici, P. Preservation of acetic acid bacteria. In Acetic Acid Bacteria: Fundamentals and Food Applications; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Abingdon, UK, 2017; pp. 193–209. [Google Scholar]
- Prakash, O.; Shouche, Y.; Jangid, K.; Kostka, J.E. Microbial cultivation and the role of microbial resource centers in the omics era. Appl. Microbiol. Biotechnol. 2013, 97, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Nerva, L.; Chitarra, W.; Siciliano, I.; Gaiotti, F.; Ciuffo, M.; Forgia, M.; Varese, C.G.; Turina, M. Mycoviruses mediate mycotoxin regulation in Aspergillus ochraceus. Environ. Microbiol. 2019, 21, 1957–1968. [Google Scholar] [CrossRef]
- Bodelier, P.L.E. Toward understanding, managing, and protecting microbial ecosystems. Front. Microbiol. 2011, 2, 1–8. [Google Scholar] [CrossRef]
- Stackebrandt, E. Towards a strategy to enhance access to microbial diversity. Int. J. Syst. Evol. Microbiol. 2011, 61, 479–481. [Google Scholar] [CrossRef]
- Vartoukian, S.R.; Palmer, R.M.; Wade, W.G. Strategies for culture of “unculturable” bacteria. FEMS Microbiol. Lett. 2010, 309, 1–7. [Google Scholar] [CrossRef]
- Chaudhary, D.K.; Khulan, A.; Kim, J. Development of a novel cultivation technique for uncultured soil bacteria. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Kaeberlein, T.; Lewis, K.; Epstein, S.S. Isolating “uncultivabte” microorganisms in pure culture in a simulated natural environment. Science 2002, 296, 1127–1129. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.H.T.; Kim, J. Cultivation of unculturable soil bacteria. Trends Biotechnol. 2012, 30, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Vries, R.G.; Snippert, H.J.; Van De Wetering, M.; Barker, N.; Stange, D.E.; Van Es, J.H.; Abo, A.; Kujala, P.; Peters, P.J.; et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009, 459, 262–265. [Google Scholar] [CrossRef]
- Sato, T.; Clevers, H. Growing self-organizing mini-guts from a single intestinal stem cell: Mechanism and applications. Science 2013, 340, 1190–1194. [Google Scholar] [CrossRef] [PubMed]
- Khalil, H.A.; Lei, N.Y.; Brinkley, G.; Scott, A.; Wang, J.; Kar, U.K.; Jabaji, Z.B.; Lewis, M.; Martín, M.G.; Dunn, J.C.Y.; et al. A novel culture system for adult porcine intestinal crypts. Cell Tissue Res. 2016, 365, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Derricott, H.; Luu, L.; Fong, W.; Hartley, C.; Johnston, L.; Armstrong, S.; Randle, N.; Duckworth, C.; Campbell, B.; Wastling, J.; et al. Developing a 3D intestinal epithelium model for livestock species. Cell Tissue Res. 2018, 375, 409–424. [Google Scholar] [CrossRef] [PubMed]
- Lanik, W.E.; Mara, M.A.; Mihi, B.; Coyne, C.B.; Good, M. Stem cell-derived models of viral infections in the gastrointestinal tract. Viruses 2018, 10, 124. [Google Scholar] [CrossRef]
- Saxena, K.; Blutt, S.E.; Ettayebi, K.; Zeng, X.-L.; Broughman, J.R.; Crawford, S.E.; Karandikar, U.C.; Sastri, N.P.; Conner, M.E.; Opekun, A.R.; et al. Human intestinal enteroids: a new model to study human rotavirus infection, host restriction, and pathophysiology. J. Virol. 2016, 90, 43–56. [Google Scholar] [CrossRef]
- Pace, N.R. A molecular view of microbial diversity and the biosphere. Science 1997, 276, 734–740. [Google Scholar] [CrossRef]
- Raja, H.A.; Miller, A.N.; Pearce, C.J.; Oberlies, N.H. Fungal Identification Using Molecular Tools: A Primer for the Natural Products Research Community. J. Nat. Prod. 2017, 80, 756–770. [Google Scholar] [CrossRef]
- Samson, R.A.; Visagie, C.M.; Houbraken, J.; Hong, S.B.; Hubka, V.; Klaassen, C.H.W.; Perrone, G.; Seifert, K.A.; Susca, A.; Tanney, J.B.; et al. Phylogeny, identification and nomenclature of the genus Aspergillus. Stud. Mycol. 2014, 78, 141–173. [Google Scholar] [CrossRef] [PubMed]
- Visagie, C.M.; Houbraken, J.; Frisvad, J.C.; Hong, S.-B.; Klaassen, C.H.W.; Perrone, G.; Seifert, K.A.; Varga, J.; Yaguchi, T.; Samson, R.A. Identification and nomenclature of the genus Penicillium. Stud. Mycol. 2014, 78, 343–371. [Google Scholar] [CrossRef] [PubMed]
- Kurtzman, C.P. Identification of food and beverage spoilage yeasts from DNA sequence analyses. Int. J. Food Microbiol. 2015, 213, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Graf, B.; Adam, T.; Zill, E.; Göbel, U.B. Evaluation of the VITEK 2 system for rapid identification of yeasts and yeast-like organisms. J. Clin. Microbiol. 2000, 38, 1782–1785. [Google Scholar] [PubMed]
- Singhal, N.; Kumar, M.; Kanaujia, P.K.; Virdi, J.S. MALDI-TOF mass spectrometry: An emerging technology for microbial identification and diagnosis. Front. Microbiol. 2015, 6, 1–16. [Google Scholar] [CrossRef]
- Posteraro, B.; Efremov, L.; Leoncini, E.; Amore, R.; Posteraro, P.; Ricciardi, W.; Sanguinetti, M. Are the conventional commercial yeast identification methods still helpful in the era of new clinical microbiology diagnostics? A meta-analysis of their accuracy. J. Clin. Microbiol. 2015, 53, 2439–2450. [Google Scholar] [CrossRef]
- Schumann, P.; Pukall, R. The discriminatory power of ribotyping as automatable technique for differentiation of bacteria. Syst. Appl. Microbiol. 2013, 36, 369–375. [Google Scholar] [CrossRef]
- Morgan, M.C.; Boyette, M.; Goforth, C.; Sperry, K.V.; Greene, S.R. Comparison of the Biolog OmniLog Identification System and 16S ribosomal RNA gene sequencing for accuracy in identification of atypical bacteria of clinical origin. J. Microbiol. Methods 2009, 79, 336–343. [Google Scholar] [CrossRef]
- Hibbett, D.; Glotzer, D. Where are all the undocumented fungal species? A study of Mortierella demonstrates the need for sequence-based classification. New Phytol. 2011, 191, 592–596. [Google Scholar] [CrossRef]
- Jones, D.S.; Albrecht, H.L.; Dawson, K.S.; Schaperdoth, I.; Freeman, K.H.; Pi, Y.; Pearson, A.; MacAlady, J.L. Community genomic analysis of an extremely acidophilic sulfur-oxidizing biofilm. ISME J. 2012, 6, 158–170. [Google Scholar] [CrossRef]
- Rosselló-Móraa, R.; Amann, R. Past and future species definitions for Bacteria and Archaea. Syst. Appl. Microbiol. 2015, 38, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Auch, A.F.; von Jan, M.; Klenk, H.P.; Göker, M. Digital DNA-DNA hybridization for microbial species delineation by means of genome-to-genome sequence comparison. Stand. Genomic Sci. 2010, 2, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Auch, A.F.; Klenk, H.P.; Göker, M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform. 2013, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Houle, D.; Govindaraju, D.R.; Omholt, S. Phenomics: The next challenge. Nat. Rev. Genet. 2010, 11, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Moore, L.R.; Blank, C.E.; Wu, E.H.H.; Ackerman, M.; Ranade, S.; Cui, H. Microbial phenomics information extractor (MicroPIE): A natural language processing tool for the automated acquisition of prokaryotic phenotypic characters from text sources. BMC Bioinform. 2016, 17, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.M.A.; Chu, K.; Palaniappan, K.; Pillay, M.; Ratner, A.; Huang, J.; Huntemann, M.; Varghese, N.; White, J.R.; Seshadri, R.; et al. IMG/M v.5.0: An integrated data management and comparative analysis system for microbial genomes and microbiomes. Nucleic Acids Res. 2019, 47, D666–D677. [Google Scholar] [CrossRef]
- Adiba, M.; Espinosa, J.A.; Adiba, M.; Alfonso, J.; Oviedo, E.; Vargas-Solar, G.; Zechinelli-Martini, J.-L. Big Data Management Challenges, Approaches, Tools and Their Limitations: To Cite This Version: HAL Id: Hal-01270335; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Gams, W.; Hennebert, L.; Stalpers, A.; Janssens, D.; Schipper, M.; Smith, J.; Yarrow, D.; Hawksworth, D. Structuring Strain Data for Storage and Retrieval of Information on Bacteria in MINE, the Microbial Information Network Europe. Syst. Appl. Microbiol. 1990, 13, 92–103. [Google Scholar]
- Rohde, C.; Smith, D.; Martin, D.; Fritze, D.; Stalpers, J. Code of conduct on biosecurity for biological resource centres: Procedural implementation. Int. J. Syst. Evol. Microbiol. 2013, 63, 2374–2382. [Google Scholar] [CrossRef]
- Romano, P.; Marra, D.; Milanesi, L. Web services and workflow management for biological resources. BMC Bioinform. 2005, 6, 1–9. [Google Scholar] [CrossRef]
- Romano, P. Automation of in-silico data analysis processes through workflow management systems. Brief. Bioinform. 2008, 9, 57–68. [Google Scholar] [CrossRef]
- Overmann, J.; Abt, B.; Sikorski, J. Present and Future of Culturing Bacteria. Annu. Rev. Microbiol. 2017, 71, 711–730. [Google Scholar] [CrossRef] [PubMed]
- Franzetti, A.; Gandolfi, I.; Bestetti, G.; Padoa Schioppa, E.; Canedoli, C.; Brambilla, D.; Cappelletti, D.; Sebastiani, B.; Federici, E.; Papacchini, M.; et al. Plant-microorganisms interaction promotes removal of air pollutants in Milan (Italy) urban area. J. Hazard. Mater. 2019, 384, 121021. [Google Scholar] [CrossRef] [PubMed]
- Villena, J.; Kitazawa, H. Probiotic Microorganisms: A Closer Look. Microorganisms 2017, 5, 17. [Google Scholar] [CrossRef] [PubMed]
- Reid, G.; Gadir, A.A.; Dhir, R. Probiotics: Reiterating what they are and what they are not. Front. Microbiol. 2019, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Liu, Y.; Latta, M.; Ma, W.; Wu, Z.; Chen, P. Probiotics database: A potential source of fermented foods. Int. J. Food Prop. 2019, 22, 197–216. [Google Scholar] [CrossRef]
- Venkata Mohan, S.; Nikhil, G.N.; Chiranjeevi, P.; Nagendranatha Reddy, C.; Rohit, M.V.; Kumar, A.N.; Sarkar, O. Waste biorefinery models towards sustainable circular bioeconomy: Critical review and future perspectives. Bioresour. Technol. 2016, 215, 2–12. [Google Scholar] [CrossRef]
- Rohde, C.; Wittmann, J.; Kutter, E. Bacteriophages: A therapy concept against multi-drug-resistant bacteria. Surg. Infect. 2018, 19, 737–744. [Google Scholar] [CrossRef]
- Konturek, P.C.; Haziri, D.; Brzozowski, T.; Hess, T.; Heyman, S.; Kwiecien, S.; Konturek, S.J.; Koziel, J. Emerging role of fecal microbiota therapy in the treatment of gastrointestinal and extra-gastrointestinal diseases. J. Physiol. Pharmacol. 2015, 66, 483–491. [Google Scholar]
- Kumar, P.; Kundu, A.; Kumar, M.; Solanki, R.; Kapur, M.K. Exploitation of potential bioactive compounds from two soil derived actinomycetes, Streptomyces sp. strain 196 and RI.24. Microbiol. Res. 2019, 229, 126312. [Google Scholar] [CrossRef]
- Martín, J.F.; Liras, P. Harnessing microbiota interactions to produce bioactive metabolites: Communication signals and receptor proteins. Curr. Opin. Pharmacol. 2019, 48, 8–16. [Google Scholar] [CrossRef]
- Varese, G.C.; Angelini, P.; Bencivenga, M.; Buzzini, P.; Donnini, D.; Gargano, M.L.; Maggi, O.; Pecoraro, L.; Persiani, A.M.; Savino, E.; et al. The current status of fungal biodiversity in Italy: Ex situ conservation and exploitation of fungi in Italy. Plant Biosyst. 2011, 145, 997–1005. [Google Scholar] [CrossRef]
- Floris, R.; Scanu, G.; Fois, N.; Rizzo, C.; Malavenda, R.; Spanò, N.; Lo Giudice, A. Intestinal bacterial flora of Mediterranean gilthead sea bream (Sparus aurata Linnaeus) as a novel source of natural surface active compounds. Aquac. Res. 2018, 49, 1262–1273. [Google Scholar] [CrossRef]
- Meena, M.; Swapnil, P.; Zehra, A.; Aamir, M.; Dubey, M.; Goutam, J.; Upadhyay, R. Plant-Microbe Interactions in Agro-Ecological Perspectives; Springer: Singapore, 2017; Volume 2, ISBN 9789811065934. [Google Scholar]
- Ghisalba, O.; Meyer, H.P.; Wohlgemuth, R. Industrial Biotransformation. In Encyclopedia of Industrial Biotechnology; Flickinger, M.C., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- OECD 2009. The Bioeconomy to 2030 Main Findings and Policy Conclusions. Available online: https://www.oecd.org/futures/long-termtechnologicalsocietalchallenges/42837897.pdf (accessed on 30 October 2019).
- Alexandraki, V.; Tsakalidou, E.; Papadimitriou, K.; Holzapfel, W. Commission on Genetic Resources; Fao: Rome, Italy, 2013; pp. 1–172. [Google Scholar]
- CNBBSV. National Commette on Biosafety, Biotechnology and Life Sciences. Italian Microbiome Initiative for Improved Human Health and Agri-food Production. Available online: http://cnbbsv.palazzochigi.it/media/1712/microbioma-2019.pdf (accessed on 30 October 2019).
- Melini, F.; Melini, V.; Luziatelli, F.; Ficca, A.G.; Ruzzi, M. Health-promoting components in fermented foods: An up-to-date systematic review. Nutrients 2019, 11, 1189. [Google Scholar] [CrossRef] [PubMed]
- Tamang, J.P.; Shin, D.H.; Jung, S.J.; Chae, S.W. Functional properties of microorganisms in fermented foods. Front. Microbiol. 2016, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Guerzoni, M.E. Human food chain and microorganisms: A case of co-evolution. Front. Microbiol. 2010, 1, 1–2. [Google Scholar] [CrossRef]
- Campus, M.; Degirmencioglu, N.; Comunian, R. Technologies and trends to improve table olive quality and safety. Front. Microbiol. 2018, 9, 617. [Google Scholar] [CrossRef]
- CBD. Convention on Biological Diversity. United Nations 1992. Available online: https://www.cbd.int/doc/legal/cbd-en.pdf (accessed on 30 October 2019).
- Nagoya Protocol on Access to Genetic Resources and the Fair and Equitable Sharing of Benefits Arising from Their Utilizaton to the Convention on Biological Diversity. 2010. Available online: https://www.cbd.int/abs/doc/protocol/nagoya-protocol-en.pdf (accessed on 30 October 2019).
- Regulation (EU) No 511/2014 of the European Parliament and of the Council of 16 April 2014 on Compliance Measures for Users from the Nagoya Protocol on Access to Genetic Resources and the Fair and Equitable Sharing of Benefits Arising from Their Utilization in the Union Text with EEA Relevance. Available online: https://op.europa.eu/en/publication-detail/-/publication/6b16d48a-dff0-11e3-8cd4-01aa75ed71a1/language-en (accessed on 30 October 2019).
- Council Directive 90/219/EEC of 23 April 1990 on the Contained Use of Genetically Modified Micro-organisms. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:31990L0219&from=EN (accessed on 6 December 2019).
- Council Directive 98/81/EC of 26 October 1998 Amending Directive 90/219/EEC on the Contained Use of Genetically Modified Micro-organisms. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:31998L0081&from=GA (accessed on 6 December 2019).
- Regulation (EU) 2016/679 of the European Parliament and of the Council of 27 April 2016 on the Protection of Natural Persons with Regard to the Processing of Personal Data and on the Free Movement of Such Data, and Repealing Directive 95/46/EC (General Data Protection Regulation). Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32016R0679&from=EN (accessed on 6 December 2019).
- Simões, M.; Dias, N.; Santos, C.; Lima, N.; Simões, M.F.; Dias, N.; Santos, C.; Lima, N. Establishment of a Quality Management System Based on ISO 9001 Standard in a Public Service Fungal Culture Collection. Microorganisms 2016, 4, 21. [Google Scholar] [CrossRef]
- Smith, D. Culture Collections, 1st ed.; Elsevier Inc.: Amsterdam, The Netherland, 2012; Volume 79, ISBN 9780123943187. [Google Scholar]
- Martins, A.; Lima, N.; Sampaio, P. A standard proposal for biological resources centres. Int. J. Qual. Reliab. Manag. 2017, 34, 147–162. [Google Scholar] [CrossRef]
- Bendapudi, N.; Berry, L.L. Customers’ motivations for maintaining relationships with service providers. J. Retail. 1997, 73, 15–37. [Google Scholar] [CrossRef]
- Souza-Poza, A.; Altinkilinc, M.; Searcy, C. Implementing a functional ISO 9001 quality management system in small and medium-sized enterprises. Int. J. Eng. 2009, 3, 220. [Google Scholar]
- Smith, D.; Martin, D.; Novossiolova, T. Microorganisms: Good or Evil, MIRRI Provides Biosecurity Awareness. Curr. Microbiol. 2017, 74, 299–308. [Google Scholar] [CrossRef] [PubMed]
| MIRRI-IT* Partners 1 and Associated Members 2 | Culture Collections | Biological Resources (Main Taxa) | Isolation Sources (Category) |
|---|---|---|---|
| Research Centres: | |||
| Agenzia nazionale per le nuove tecnologie, l’energia e lo sviluppo economico sostenibile (ENEA) 2 | ENEA collection | Bacteria, Fungi, Microalgae, Microbial consortia, Virus | Contaminated sites, Hypogea and archaeological sites, Food, Lake sediments, Sea, Soil, Rhizosphere, Water |
| Agenzia regionale per la ricerca in agricoltura, (Agris Sardegna) 2 | Bonassai (BNSS) | Bacteria (Actinobacteria, Bacteroidetes, Firmicutes, Proteobacteria) | Animals, Food |
| Consiglio Nazionale delle Ricerche (CNR) 1 | Agro-Food Microbial Culture Collection (ITEM) | Bacteria, Filamentous fungi, Yeasts | Air, Insects, Food, Plants, Soil |
| Plant Viruses Italy (PLAVIT) | Mycoviruses, Phytoplasmas, Phages Plant viruses | Fungi, Plants | |
| IRCCS Ospedale Policlinico San Martino of Genoa 1 | Interlab Cell Line Collection (ICLC) | Cell lines | |
| Istituto Nazionale Malattie Infettive (INMI) “Lazzaro Spallanzani”, Roma 2 | INMI collection | Bacteria, Yeasts | Humans, Clinical isolates |
| Istituto Nazionale di Oceanografia e di Geofisica Sperimentale, Trieste (OGS) 2 | Collection of Sea Microorganisms (COSMI) | Microalgae (predominantly Bacillariophyceae, Dinophyceae,) | Marine environments |
| Istituto Zooprofilattico Sperimentale della Lombardia e dell’Emilia Romagna (IZSLER) 2 | Biobank of Veterinary Resources (BVR) | Cell lines, Bacteria, Fungi, Parasite, Virus | Animals, Feed, Food, Environments |
| Istituto Zooprofilattico Sperimentale delle Venezie (IZSVE) 2 | European Union Reference Laboratory (EURL) Biobank | Viruses, Antigens, Polyclonal antisera | Animals |
| Universities: | |||
| Sapienza University of Rome, Department of Environmental Biology 2 | Fungal Biodiversity Laboratory (FBL) | Saprotrophic fungi | Litter, Contaminated sites, Soil, Water |
| University of Cagliari 2 | DSMSP Collection | Bacteria, Moulds, Yeasts | Beverages, Environments, Food |
| University of Basilicata, School of Agricultural, Forestry, Food and Environmental Sciences 2 | UNIBAS Yeast Collection (UBYC) | Yeasts | Beverages, Food |
| University of Genoa 2 | Collection of DISTAV (ColD) | Bacteria, Fungi, Yeasts | Extreme environments, Human cadaver |
| University of Milano-Bicocca 2 | MicroMiB collection (MicroMib) | Bacteria, Yeasts, Virus | Humans, Environments, Food |
| University of Modena and Reggio Emilia 1 | Unimore Microbial Culture Collection (UMCC) | Bacteria (Lactic Acid Bacteria, Proteobacteria), Yeasts (Ascomycota) | Beverages, Food |
| University of Naples “Federico II”, Department of Biology 2 | Algal Collection University Federico II (ACUF) | Cyanobacteria, Microalgae | Aeroterrestrial habitats, Environments |
| University of Palermo, Department of Agricultural, Food and Forest Sciences 2 | Herbarium SAF (SAF) | Macrofungi | Ascomata, Basidiomata |
| University of Pavia, Department of Earth and Environmental Sciences 2 | Amico Fungo | Filamentous Fungi, Yeasts | Soil, Contaminated sites |
| University of Perugia 1 | Industrial Yeasts Collection (DBVPG) | Yeasts, Yeast-like organisms | Beverages, Environments, Food |
| University of Sassari 2 | Microbial Culture Collection of University of Sassari (UNISS) | Bacteria (Firmicutes, Proteobacteria), Filamentous fungi, Yeasts (Ascomycota, Basidiomycota) | Agricultural by-products, Animals, Beverages, Environments, Food, Plants, Humans |
| University of Turin 1 | Turin University Culture Collection (TUCC) including Mycotheca Universitatis Taurinenesis (MUT) | Bacteria (Firmicutes, Lactic Acid bacteria, Proteobacteria) Filamentous fungi (Ascomycota, Basidiomycota, Mucoromycota), Yeasts | Animal and human clinical Samples, Extreme environments, Food |
| University of Tuscia, Department of Ecological and Biological Sciences 2 | Culture Collection of Fungi from Extreme Environments (CCFEE) | Filamentous and Meristematic fungi, Lichens, Yeasts | Monuments, Contaminated sites, Rocks, Soil |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Vero, L.; Boniotti, M.B.; Budroni, M.; Buzzini, P.; Cassanelli, S.; Comunian, R.; Gullo, M.; Logrieco, A.F.; Mannazzu, I.; Musumeci, R.; et al. Preservation, Characterization and Exploitation of Microbial Biodiversity: The Perspective of the Italian Network of Culture Collections. Microorganisms 2019, 7, 685. https://doi.org/10.3390/microorganisms7120685
De Vero L, Boniotti MB, Budroni M, Buzzini P, Cassanelli S, Comunian R, Gullo M, Logrieco AF, Mannazzu I, Musumeci R, et al. Preservation, Characterization and Exploitation of Microbial Biodiversity: The Perspective of the Italian Network of Culture Collections. Microorganisms. 2019; 7(12):685. https://doi.org/10.3390/microorganisms7120685
Chicago/Turabian StyleDe Vero, Luciana, Maria Beatrice Boniotti, Marilena Budroni, Pietro Buzzini, Stefano Cassanelli, Roberta Comunian, Maria Gullo, Antonio F. Logrieco, Ilaria Mannazzu, Rosario Musumeci, and et al. 2019. "Preservation, Characterization and Exploitation of Microbial Biodiversity: The Perspective of the Italian Network of Culture Collections" Microorganisms 7, no. 12: 685. https://doi.org/10.3390/microorganisms7120685
APA StyleDe Vero, L., Boniotti, M. B., Budroni, M., Buzzini, P., Cassanelli, S., Comunian, R., Gullo, M., Logrieco, A. F., Mannazzu, I., Musumeci, R., Perugini, I., Perrone, G., Pulvirenti, A., Romano, P., Turchetti, B., & Varese, G. C. (2019). Preservation, Characterization and Exploitation of Microbial Biodiversity: The Perspective of the Italian Network of Culture Collections. Microorganisms, 7(12), 685. https://doi.org/10.3390/microorganisms7120685















