Abstract
Nutrient limitation and nonfavorable growth conditions have been suggested to be major triggers for the expression of small, cysteine-rich antimicrobial proteins (AMPs) of fungal origin, e.g., the Penicillium chrysogenum antifungal protein (PAF), the Aspergillus giganteus antifungal protein (AFP), the Aspergillus niger antifungal protein (AnAFP). Therefore, these AMPs have been considered to be fungal secondary metabolite products. In contrast, the present study revealed that the expression of the PAF-related AMP P. chrysogenum antifungal protein B (PAFB) is strongly induced under nutrient excess during the logarithmic growth phase, whereas PAFB remained under the detection level in the supernatant of cultures grown under nutrient limitation. The efficiency of the pafB-promoter to induce PAFB expression was compared with that of two P. chrysogenum promoters that are well established for recombinant protein production: the paf-promoter and the xylose-inducible promoter of the xylanase gene, xylP. The inducibility of the pafB-promoter was superior to that of the xylP-promoter yielding comparable PAFB amounts as under the regulation of the paf-promoter. We conclude that (i) differences in the expression regulation of AMPs suggest distinct functional roles in the producer beyond their antifungal activity; and (ii) the pafB-promoter is a promising tool for recombinant protein production in P. chrysogenum, as it guarantees strong gene expression with the advantage of inducibility.
1. Introduction
Fungi with sequenced genomes that belong to the class Eurotiomycetes contain at least one gene coding for antimicrobial proteins (AMPs) [1]. These proteins are small in size (~6.5 kDa), cysteine-rich and amphipathic, and are secreted into the culture broth by their producer strains. Their compact disulfide-stabilized tertiary structure—containing five β-strands—renders them highly stable against adverse environmental conditions [2,3,4,5].
AMPs efficiently inhibit the growth of human and plant pathogenic fungal species when applied at low micromolar concentrations and those acting in a fungicidal way, lower the risk of resistance development. Extensively studied examples are AMPs from Penicillium chrysogenum (P. chrysogenum antifungal protein (PAF) and P. chrysogenum antifungal protein B (PAFB)) [6,7], Neosartorya fischeri (N. fischeri antifungal protein (NFAP) and N. fischeri antifungal protein 2 (NFAP2)) [8,9], Penicillium digitatum (P. digidatum antifungal protein B (AfpB)) [10], Penicillium expansum (P. expansum antifungal proteins A, B, C (PeAfpA, PeAfpB and PeAfpC)) [11], Aspergillus giganteus (A. giganteus antifungal protein (AFP)) [12,13] and Aspergillus niger (A. niger antifungal protein (AnAFP)) [14]. Most of them show no cytotoxicity to mammalian cells in vitro [10,11,15,16,17,18] and in vivo [16,19].
AMPs are therefore considered as promising candidates for the development of novel antifungal treatment strategies in medicine and agriculture [9].
Many studies have been published and provide insight into the AMPs specificity, their structure and their antimicrobial mode of action. However, limited information is available on the expression regulation of the respective encoding genes and the cultivation conditions that trigger production of these biomolecules by their producers. AMP yields vary significantly between different fungal species and cultivation conditions. PAF, for example, is secreted in high amounts by P. chrysogenum [15], whereas the quantities of NFAP appear comparably low in the culture supernatant of N. fischeri [8]. The knowledge how AMP gene expression is regulated is important for two reasons: (i) to understand the AMPs’ function for the host; and (ii) to identify culture conditions to improve AMP production and reach protein yields that are sufficient for their experimental use to investigate their structure and mode of action and for their applicability in vitro and in vivo. For high-yield AMP production, our group has established a P. chrysogenum-based expression system that works under the control of the promoter of the PAF encoding gene (paf) [20]. This gene is strongly induced in minimal medium under nutrient-limiting conditions when the culture has entered the nonexponential growth phase. Nutrient limitation and nonfavorable growth conditions e.g., heat shock or pH-stress were reported to trigger the production of other AMPs, like the A. gigenteus AFP or the A. niger AnAFP [21,22]. This expression regulation led to the hypothesis that ascomycetes AMPs cover—in addition to their growth inhibition activity—additional regulatory functions in the producing fungi; for example, they play a role in development [21,23], apoptosis [24], autolysis and autophagy [25].
In contrast to this assumption, the expression of the PAF-related P. chrysogenum antifungal protein PAFB was found to be not induced under these cultivation conditions [7]. The pafB-mRNA was only slightly expressed in minimal medium and the protein PAFB remained under the detection level in the culture supernatant. This necessitated the application of the P. chrysogenum-based expression system for high-yield and high-quality production of PAFB for structural and functional analyses [7]. PAFB revealed a PAF-like compact β-folded structure, containing five β-strands connected by four flexible loops and an “abcabc” disulphide bond pattern. Functional studies identified human pathogenic fungal species such as Aspergillus fumigatus, Candida albicans or Trichophyton rubrum and model organisms, such as Neurospora crassa, to be highly susceptible against low doses (μM) of PAFB and PAF [7].
In the present study, we searched for cultivation conditions triggering PAFB expression and secretion into the culture broth of P. chrysogenum wild-type (wt). Interestingly and unexpectedly, we found an increased and sustained induction of the pafB gene during the exponential growth phase under high nutrient availability. The differences in the regulation of PAF and PAFB expression let us hypothesize that these two AMPs cover diverse functions in P. chrysogenum beyond their antifungal activity. Applying nutrient abundance, we could purify high amounts of the native PAFB from the culture supernatant. The efficiency of the pafB-promoter to induce PAFB expression was further compared with that of two already well established promoter systems derived from P. chrysogenum: the paf-promoter and the xylose-inducible promoter of the xylanase gene xylP [26], revealing a clear superiority of the pafB-promoter. The inducibility of the pafB-promoter by nutrient excess and the high transcription efficiency suggests this promoter to be a suitable tool for high-yield production of recombinant proteins in P. chrysogenum.
2. Materials and Methods
2.1. Strains, Media and Cultivation Conditions
Media and strains used in this study are listed in Supplementary Tables S1 and S2. Conidia of all P. chrysogenum strains were generated on solid P. chrysogenum minimal medium (PcMM) at 25 °C for 72–96 h and the spores were harvested in spore buffer (0.9% NaCl (w/v), 0.01% Tween 80 (v/v)). To monitor RNA and protein expression and for protein generation 2 × 108 conidia of the respective P. chrysogenum strains were cultivated in 200 mL medium at 25 °C for up to 96 h. PAFB expression was monitored in P. chrysogenum wt grown in PcMM containing up to fourfold molar excess of all compounds (1×, 2×, 3× and 4× PcMM, respectively) and in complete medium (CM) containing onefold and fourfold molar excess of all compounds (1× CM and 4× CM). To determine the impact of the different compounds of PcMM on pafB expression, P. chrysogenum wt was grown in PcMM, in which the single compounds were supplemented in fourfold excess, respectively: KCl (26.8 mM), FeSO4 × 7H2O (0.72 mM), MgSO4 × 7H2O (8 mM), NaNO3 (141.2 mM) or sucrose (232 mM).
For the determination of the promoter efficiency the P. chrysogenum mutant pafBpaf_promoter was cultivated in 1× PcMM containing 2% sucrose, the mutant pafBxylP_promoter in 1× PcMM containing 2% xylose and the P. chrysogenum wt in 4× PcMM containing 8% sucrose as sole carbon source, respectively. Bacillus subtilis was grown on lysogeny broth (LB) agar plates.
2.2. Cloning Vector Construction and Fungal Transformation for Promoter/Terminator Exchange
Oligonucleotides used for cloning are indicated in Supplementary Table S3. The bipartite marker technique was used for generating the pafBpaf_promoter, pafBpaf_terminator and the pafBxylP_promoter mutants, respectively. For each strain, the P. chrysogenum wt was cotransformed with two polymerase chain reaction (PCR) fragments (Figure S1).
For the generation of the pafBpaf_promoter strain, P. chrysogenum wt was cotransformed with the PCR fragments A and B. To generate fragment A, gDNA of P. chrysogenum wt was amplified using the primer pair pafB3′NotI_rev and pafB_BglII_fw. The PCR product was digested using the enzymes BglII and NotI and ligated into the plasmid pSK275pafB [7]. From this plasmid, a fragment containing the paf 5′untranslated region (UTR), the pafB gene and the pafB 3′UTR was released by using the enzymes NotI and XbaI and ligated into the plasmid pD-NAT1 [27], resulting in plasmid pD-NAT1_pafBpaf_promoter. Fragment A (4767 bp) was amplified by the primer pair Onat2 and 3′pafb flanken nested_fw.
To generate fragment B, gDNA of P. chrysogenum wt was amplified using the primer pair pafB5_BamHI_rev and pafB5_PstI_fw. The fragment was digested and ligated into plasmid pD-NAT1. Fragment B (2883 bp) was amplified from this plasmid by using the primer pair Onat1 and 5′pafb fl nested_fw.
For the generation of the pafBxylP_promoter strain, P. chrysogenum wt was cotransformed with the PCR fragments B & C. Fragment C was generated by amplifying the xylP-promoter sequence from plasmid px-ergA [28] using the primer pair Xylp_XbaI_rev_B and Xylp_BglII_fw_B. The PCR fragment was digested using the enzymes XbaI and BglII and the paf-5′UTR region in plasmid pD-NAT1_pafBpaf_promoter was exchanged by the xylP-promoter sequence ending up in plasmid pD-NAT1_pafxylP_promoter. Fragment C (4729 bp) was amplified by the primer pair Onat2 and 3′pafb flanken nested_rev and fragment B was generated as described above.
The pafBpaf_terminator mutant was generated by cotransforming the P. chrsogenum wt with the fragments D and E. Fragment D was generated by amplifying gDNA of the P. chrysogenum wt strain with the primer pair pafB5_PstI_fw and pafB_SmaI_rev. The purified PCR fragment was digested by PstI and SmaI and ligated into plasmid pSK275pafB [7], resulting in plasmid pSK275pafBpaf3′pafb5′. The plasmid pSK275pafBpaf3′pafb5′ was used in PCR reaction together with the primer pair, pafB5_PstI_fw and 3′paf_BamHI_rev. This fragment was digested using the enzymes PstI and BamHI and ligated into plasmid pD-NAT1, resulting in plasmid pD-NAT1_pafBpaf_terminator. pD-NAT1_pafBpaf_terminator was used as template to generate the transformation fragment D (3677 bp) by using the primers Onat1 and 5′pafb fl nested_fw.
Fragment E was generated by amplifying gDNA of P. chrysogenum wt by the primer pair pafB3_XbaI_fw and pafB3′NotI_rev. After digesting the fragment with the enzymes NotI and XbaI, it was ligated into plasmid pD-NAT1_pafBpaf_terminator, resulting in plasmid pD-NAT1_pafBpaf_terminator/pafB5′UTR. Plasmid pD-NAT1_pafBpaf_terminator/pafB5′UTR was used together with the primer pair, Onat2 and 3′pafb flanken nested_rev, in a PCR reaction to generate fragment E (2348 bp).
PCR fragments A and B, B and C, and D and E shared a 400-bp overlap within the nat1 cassette, which served as a potential recombination site during transformation. The transformation of P. chrysogenum protoplast with 5 µg DNA per fragment was carried out according to Cantoral et al. (1987) [29]. Transformants were single-spored three times on PcMM agar supplemented with 200 μg/mL nourseothricin (Jena Bioscience, Jena, Germany).
Positive transformants were analyzed by Southern blotting for homologous integration (Figure S1).
2.3. Southern Blot Analysis
Southern blot analysis was performed to verify locus specific and single copy integration in the mutants pafBpaf_promoter, pafBxylP_promoter and pafBpaf_terminator. Genomic DNA was extracted according to Zadra et al. (2000) [26].
Per lane, 2 μg gDNA of the respective mutant and the wt—digested using the respective restriction enzymes—was size-fractionated on a 0.8% (w/v) agarose gel. The DNA was transferred onto Hybond-N membranes (Amersham Biosciences, Little Chalfont, UK) and hybridized with digoxigenin (DIG)-labelled probes (Roche Diagnostics, Mannheim, Germany), specific for the nourseothricin-acetyltransferase gene (nat1) and the pafB gene, respectively. The probes were generated from pSK275pafB or pD-NAT1 by PCR using the oligonucleotides opafb_fw/opafb_rev2 or Onat1/Onat2.
2.4. Northern Blot Analysis
Northern blot analysis was used to monitor the mRNA expression of pafB and paf as well as the expression of genes encoding the ribosomal proteins S5 and S6. Total RNA was extracted from mycelia of the respective P. chrysogenum strains using TRI Reagent® (Sigma-Aldrich, Vienna, Austria). Ten µg RNA per lane were loaded on 1.2% formaldehyde-agarose gels, blotted onto Hybond-N membranes (Amersham Biosciences) and hybridized with DIG-labelled probes (Roche). For generation of pafB and paf specific hybridization probes, the primer pairs opafB_without prepro_fw/opafB_rev2 and opaf_without prepro_fw/opaf_rev were used in the PCR reaction, respectively. For probes to specifically detect gene expression of the ribosomal protein S5, the primer pair 40S rib_protein_S5_rev and 40S rib_protein_S5_fw, and for protein S6, the primer pair 40S rib_protein_S6_rev and 40S rib_protein_S6_fw were used.
2.5. Protein Expression, Purification and Verification
Recombinant PAF and PAFB were generated by cultivating the strains P. chrysogenum paf and P. chrysogenum pafB in 1× PcMM, respectively. Purification was performed as recently described [20]. Native PAFB was expressed in 4× PcMM by the P. chrysogenum wt strain and purified as described [7,20]. The higher positive net charge (+5.2 at pH 7) of PAFB enabled the separation of PAFB from PAF by elution at a higher salt concentration (500 mM NaCl).
PAFB expression by the mutant pafBpaf_promoter was performed in 1× PcMM. The PAFB purification strategies are depicted in Supplementary Methods. Electro-spray ionization mass spectrometry (ESI-MS) was used to determine protein mass and to verify the identity of purified PAFB (Protein Micro-Analysis Facility at the Medical University of Innsbruck).
2.6. SDS-PAGE and Western Blotting
For the detection of PAFB and PAF in the culture broth, 25 µL of cell-free culture supernatant were separated on a 18% (w/v) tris-glycine sodium dodecyl sulfate (SDS)-polyacrylamide gel and then transferred to nitrocellulose membranes (Bio Trace, Pall, Port Washington, NY, USA). After blotting, membranes were blocked for 2 h in PBS/0.3% Tween (v/v)/3.0% skim milk powder (w/v) and then incubated overnight at 4 °C with anti-PAFB (α-PAFB) or anti-PAF (α-PAF) antiserum (1:1000 in PBS/0.3% Tween (v/v)/1.0% skim milk powder (w/v)). After washing in PBS/0.3% Tween (3 × 10 min), the membranes were incubated with alkaline phosphatase conjugated goat anti-rabbit secondary antibody (Sigma), diluted 1:10,000 in PBS/0.3% Tween (v/v)/1.0% skimmed milk powder (w/v) for 1 h at room temperature. The membranes were washed and protein immunocomplexes were visualized using nitro blue tetrazolium chloride (NBT)/5-bromo-4-chloro-3-indolyl-phosphate (BCIP) (Promega, Madison, WI, USA).
2.7. Agar Diffusion Test
To monitor the penicillin content in the fungal culture, broth agar diffusion tests were performed. Four colonies of B. subtilis were harvested from overnight agar plates and transferred into 2 mL of LB medium. One-hundred µL of the bacterial suspension were added to 10 mL LB medium and poured on a petri dish (diameter = 13.5 cm) containing 100 mL LB-agar. The plate was incubated for 1 h at 37 °C, then the liquid was removed and holes were punched into the agar.
Cell-free supernatants of 24–120-h-old P. chrysogenum wt cultures grown in 1× PcMM or 4× PcMM were added to the holes. The amount of the loaded culture broth was adjusted to the mycelial dry weight and calculated according to the following formula, 250/gDW × 6. To prove the presence of penicillin, 250 µL of cell-free culture supernatant (1× PcMM, 120 h cultivation) were treated for 1 h at 30 °C with 1000 U penicillinase (TCI). As a control, 2.5 µg and 1.2 µg penicillin G sodium salt (Sigma) were used instead of the culture broth and treated with 1000 U penicillinase in a total volume of 250 µL. The samples were then used in agar diffusion assays. As controls, untreated penicillin (1.2 and 2.5 µg), untreated supernatant (250 µL, 1× PcMM, 120 h cultivation) and 1000 U penicillinase were used, respectively. Plates were incubated overnight at 37 °C and inhibition zones were evaluation.
2.8. Microscopy and Image Processing
The validation of pellet morphology was done with an inverted Leica DM IL LED microscope (Leica Microsystems, Wetzlar, Germany) and imaging was performed with an AxioCam MR3 camera (Carl Zeiss GmbH, Oberkochen, Germany). Image processing and editing was achieved with the programs Axio Vision software (Carl Zeiss GmbH), GNU Image Manipulation Program (GIMP, version 2.8.201) and Microsoft PowerPoint (Microsoft Corp., Redmond, WA, USA).
2.9. Statistical Analysis
Statistical analysis was performed using Microsoft Excel 2010 software (Microsoft Corp.). A two-sample t-test with equal variance and one-tailed distribution was applied to determine the p-values.
3. Results
3.1. The Expression of pafB Is Induced by Nutrient Excess
We have recently reported that the protein PAFB remained under the limit of detection in the culture supernatant, although the pafB gene expression was induced after 48 h of cultivation in standard PcMM (1× PcMM) [7]. To identify conditions that trigger PAFB expression, we tested different media with variable nutrient compositions. In minimal media containing a two-to-four-fold nutrient excess (2×, 3× and 4× PcMM), PAFB expression was strongly induced and the protein amount in the supernatant correlated with the increasing nutrient concentration (Figure 1). In contrast, paf gene expression peaked after 72 h of cultivation in 1× PcMM [7] and PAF-production could not be further increased by high nutrient availability (Figure 1). It should be noted that the salts added with the trace element solution or the KPO4-buffer remained single-concentrated in 2×–4× PcMM, respectively. Therefore, these components could be excluded to trigger pafB transcription.
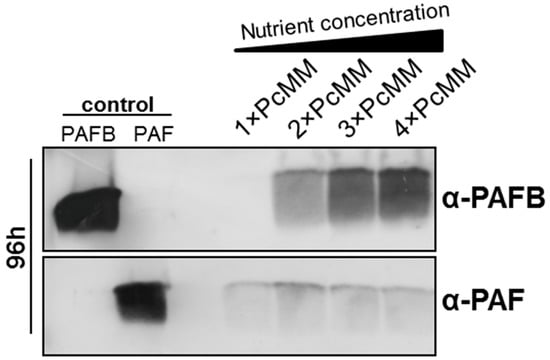
Figure 1.
Expression of the P. chrysogenum antifungal protein (PAF) and P. chrysogenum antifungal protein B (PAFB) in minimal medium (PcMM) containing increasing nutrient concentrations. P. chrysogenum wt was cultivated in 1×, 2×, 3× or 4× PcMM, respectively, at 25 °C for 96 h. Western blot: Culture supernatants (25 μL per lane) were loaded on a 18% (w/v) sodium dodecyl sulfate (SDS) polyacrylamide gel and size fractionated proteins were transferred on a nitrocellulose membrane. Recombinant PAFB and PAF (1 μg per lane) served as controls, respectively. A polyclonal antibody (α-PAFB or α-PAF) was used for specific PAFB and PAF detection in a 1000-fold dilution, respectively.
Based on these findings, we conclude that PAFB expression differs from that of other AMPs like PAF (P. chrysogenum), AFP (A. giganteus) and AnAFP (A. niger), which are strongly induced under nutrient starvation conditions [25].
To evaluate the expression pattern of PAFB on gene transcript and protein level, we performed time-course experiments with P. chrysogenum wt grown in 1× PcMM and 4× PcMM. In 4× PcMM, PAFB could be detected after 48 h of inoculation and the protein amount steadily increased with the cultivation time (up to 96 h) (Figure 2A). Gene transcription preceded protein production, peaked after 48 h of cultivation and remained elevated during the applied time course (up to 96 h). In contrast the pafB-mRNA signal from 1× PcMM culture conditions was much weaker at the cultivation time of 48 h and disappeared 24 h later (Figure 2B). For comparison, the PAF content in the supernatant differed not significantly between high (4× PcMM) and low (1× PcMM) nutrient conditions (Figure 2A). The paf-mRNA expression reached a maximum after 72 h of cultivation in 1× PcMM, whereas in 4× PcMM, gene expression was delayed for 24 h (Figure 2B).
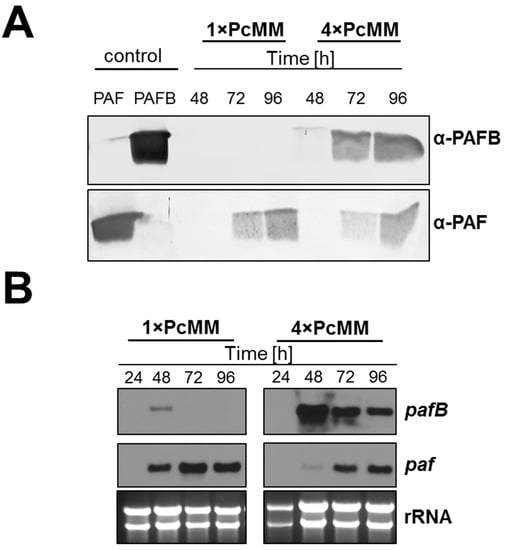
Figure 2.
Comparison of PAFB and PAF expression on mRNA and protein level in P. chrysognum wt under low nutrient (1× PcMM) and nutrient excess conditions (4× PcMM). (A) Western blot: Samples were taken after 48, 72 and 96 h of cultivation at 25 °C. Culture supernatants (25 μL per lane) were loaded on a 18% (w/v) SDS polyacrylamide gel and size fractionated proteins were transferred on a nitrocellulose membrane. A polyclonal antibody (α-PAFB or α-PAF) was used for specific PAFB and PAF detection in a 1000-fold dilution. As control, 1 µg purified PAFB or PAF were loaded. (B) Northern blot: Samples to monitor mRNA expression were taken after 24, 48, 72 and 96 h of incubation at 25 °C. Ten μg total RNA were loaded per lane on a 1.2% denaturing agarose gel, blotted and hybridized with a pafB or paf-specific digoxigenin (DIG)-labelled probe. Ethidium bromide-stained rRNA (26S and 18S) is shown as loading control.
Next, we wanted to identify which components of PcMM induce pafB expression in P. chrysogenum wt. To answer this question, we supplemented 1× PcMM with each of the following compounds, KCl, MgSO4, FeSO4, sucrose or NaNO3 in 4-fold concentration, respectively.
Western blot analysis revealed that increased nitrogen availability triggered PAFB production (96 h cultivation), although the protein amount in the supernatant remained significantly lower compared to that in 4× PcMM. The excess of the other compounds did not induce protein production (Figure 3A). We therefore monitored pafB-mRNA expression under increased nitrogen availability in a time course of 48–96 h of cultivation. Transcription induction after 48 h of cultivation could be observed with fourfold excess of NaNO3; however, the amount of pafB-mRNA rapidly decreased after this time point and resulted in only a mild accumulation of PAFB in the supernatant after 96 h. In contrast, high and sustained amounts of pafB transcripts were detectable in 4× PcMM, which was reflected in a significantly increased PAFB amount in the culture broth (Figure 3B). We therefore conclude, that not only the upregulation of transcription but also a sustained transcription over time are necessary to reach high PAFB yields in the culture broth.
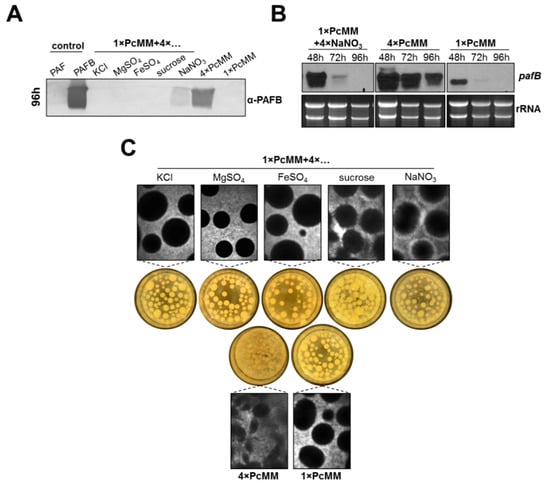
Figure 3.
Impact of PcMM compounds on pafB expression and fungal culture morphology. P. chrysogenum wt was cultivated in 1× PcMM, 4× PcMM and 1× PcMM supplemented with 4× KCl, 4× MgSO4, 4× FeSO4, 4× sucrose or 4× NaNO3, respectively. (A) Western blot: PAFB secretion into culture supernatant was monitored after 96 h of cultivation. Culture supernatants (25 μL per lane) were loaded on a 18% (w/v) SDS polyacrylamide gel and size fractionated proteins were transferred on a nitrocellulose membrane. A polyclonal antibody (α-PAFB) was used for specific PAFB detection in a 1000-fold dilution. As control, 1 µg purified PAFB and PAF were loaded. (B) Northern blot: mRNA expression in P. chrysogenum wt grown in 1× PcMM + 4× NaNO3 was compared to that cultivated in 1× PcMM and 4× PcMM. Expression was analyzed after 48, 72 and 96 h. Ten μg total RNA per lane were loaded on a 1.2% denaturing agarose gel, blotted and hybridized with a pafB-specific DIG-labelled probe. Ethidium bromide-stained rRNA (26S and 18S) is shown as loading control. (C) Morphology of fungal pellets after 96 h of cultivation under low and high nutrient conditions.
Interestingly, the morphology of the submerse culture grown in 4× PcMM markedly differed from that grown under 1× PcMM and 1× PcMM supplemented with 4-fold excess of the single compounds, respectively. In 4× PcMM the pellets appeared less dense, in comparison to the other two growth conditions where more defined and compact pellets were formed (Figure 3C).
Our results let us assume that not only the excess of nutrients but also the combination of compounds in the medium is needed for the induction of prolonged pafB expression. We were therefore interested if pafB is expressed in a nutrient-rich complete medium (CM) like in 4× PcMM. In 1× CM, we could neither identify PAFB in the culture supernatant after 96 h of cultivation nor detect pafB gene transcripts. In contrast, in 4× CM, PAFB was secreted into the culture supernatant in similar amounts as detected in 4× PcMM (Figure 4A). This result correlated with the increased amount of pafB-mRNA over the investigated time (48–96 h) (Figure 4B). Interestingly, the fungal morphology in 4× CM was similar to that in 4× PcMM, where fluffy mycelium was formed, instead of dense pellets as observed in the single concentrated media, respectively (Figure 4C).
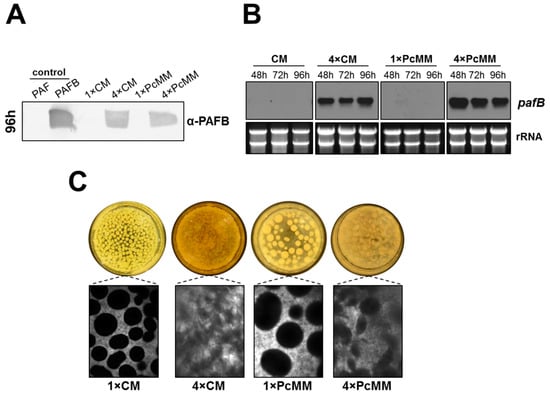
Figure 4.
Impact of complete medium on pafB expression and fungal culture morphology. The pafB expression was compared between P. chrysogenum wt grown up to 96 h at 25 °C in 1× CM, 4× CM, 1× PcMM and 4× PcMM, respectively. (A) Western blot: PAFB secretion into the culture supernatant after 96 h of cultivation. Culture supernatants (25 μL per lane) were loaded on a 18% (w/v) SDS polyacrylamide gel and size fractionated proteins were transferred on a nitrocellulose membrane. A polyclonal antibody was used for specific PAFB detection in a 1000-fold dilution. As control, 1 µg purified PAFB and PAF were loaded. (B) Northern blot: pafB-mRNA expression. Samples were taken after 48, 72 und 96 h of cultivation. Ten μg total RNA per lane were loaded on a 1.2% denaturing agarose gel, blotted and hybridized with a pafB-specific DIG-labelled probe. Ethidium bromide-stained rRNA (26S and 18S) is shown as loading control. (C) Morphology of fungal pellets after 96 h of cultivation in the respective medium.
3.2. PAFB Is Expressed during Exponential Growth
To provide evidence for the fitness of the fungal culture under pafB-inducing vs. noninducing conditions, we evaluated parameters such as biomass, secondary metabolite production and expression of genes encoding ribosomal proteins.
We determined the fungal biomass and observed an increase of dry weight under pafB-inducing conditions (4× PcMM) from 48 to 120 h, reflecting the exponential growth phase during this cultivation period. Under pafB noninducing conditions (1× PcMM), in contrast, the biomass reached a maximum after 72 h of cultivation and then diminished with prolonged incubation time indicating the start of the stationary growth phase (Figure 5A). This paralleled well with significantly more extracellular proteins secreted into 4× PcMM supernatant compared to 1× PcMM culture broth over a time periode of 120 h. These results pointed at a highly active fungal metabolism under pafB-inducing culture conditions (Figure 5B).
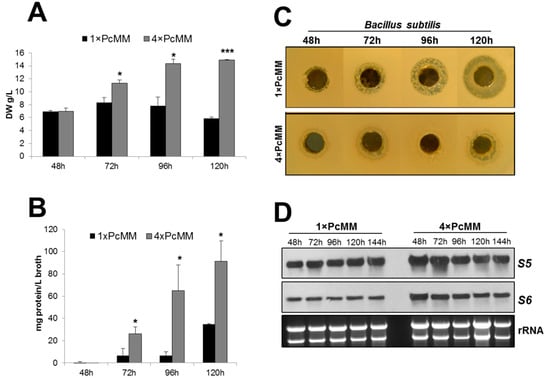
Figure 5.
Parameters reflecting the fungal fitness under pafB-inducing and noninducing conditions. P. chrysogenum wt was grown in 1× PcMM and 4× PcMM at 25 °C, respectively. Samples were taken after 48, 72, 96 and 120 h to monitor biomass, extracellular protein and penicillin production. Expression of genes coding for ribosomal protein S5 and S6 was analyzed up to 144 h. (A) Fungal biomass: Biomass is indicated as gram dry weight per liter (DW g/L). Values are given as mean ± SD (n = 3). (B) Extracellular protein production: Secretion of extracellular proteins is indicated as mg protein per liter culture broth (mg protein/L broth). Values are given as mean ± SD (n = 3). (C) Penicillin production: Penicillin production was tested on B. subtilis. The amount of culture broth tested, was related to the dry weight of the biomass. Plates were evaluated after 24 h of incubation (D) Northern blot: mRNA expression of ribosomal proteins S5 and S6. Ten μg total RNA per lane were loaded on a 1.2% denaturing agarose gel, blotted and hybridized with S5 and S6 specific DIG-labelled probes, respectively. Ethidium bromide-stained rRNA (26S and 18S) is shown as loading control. P-values in (A,B) were determined to compare the biomass production and the extracellular protein level between 1× PcMM and 4× PcMM conditions, respectively. * p ≤ 0.05, *** p ≤ 0.0005.
To further substantiate our assumptions that PAFB is produced under favourable growth conditions, we investigated the production of secondary metabolites like penicillins, which are strongly induced under stress conditions when resources are low [30,31]. We therefore compared the penicillin production of P. chrysogenum grown in 1× PcMM and 4× PcMM. For this, we applied an agar diffusion test and exposed the penicillin-sensitive gram positive B. subtilis to conditioned cell-free supernatant collected at various time points from the respective P. chrysogenum cultures. The growth inhibition zones around the holes containing the conditioned 1× PcMM supernatant indicated the presence of a compound that inhibits the growth of B. subtilis. The strongest inhibition was observed with the sample collected from pafB-repressing condition after 120 h of cultivation. The inhibition zone diameter reached 20 mm. The supernatnat from cultures grown in 4× PcMM, in contrast, did not cause any distinct inhibition zones (Figure 5C). To verify the presence of penicillin in the culture broth, we treated the P. chrysogenum conditioned cell-free supernatant with β-lactamase—an enzyme that specifically degrades penicillins [32], before applying it in the agar diffusion assay. Indeed, the supernatant had lost antibacterial activity after β-lactamase treatment (Figure S2). The absence of antibiotic production under pafB-inducing conditions further substantiate that the fungal culture had not entered the stationary growth phase and has therefore not reached starvation conditions.
Finally, we tested the expression of genes encoding the ribosomal proteins S5 and S6 to evaluate the physiological state of the culture. It is well known that nutrient starvation may lead to the induction of autophagy, which is reflected by the downregulation of ribosomal genes [33]. When comparing the expression of genes coding for the ribosomal proteins S5 and S6 in 1× PcMM and 4× PcMM, we could not detect any substantial change in the transcriptional levels of both genes indicating the absence of autophagy under both cultivation conditions (Figure 5D).
Taken together, our results indicate that PAFB is produced in P. chrysogenum wt cultures with high metabolic activity. This stands in contrast to the reported culture conditions under which the P. chrysogenum PAF, the A. giganteus AFP and the A. niger AnAFP are produced, and for which a role in nutrition, apoptosis or autophagy has been suggested [24,25].
3.3. Increased and Sustained pafB Gene Expression Is Promoter-Regulated
The low amount of pafB transcripts in 1× PcMM could result from low transcription efficiency due to promoter-based gene repression or from mRNA instability regulated by the pafB 3′UTR region. We therefore exchanged the pafB-promoter by the paf-promoter and the pafB 3′UTR by the paf 3′UTR, respectively, generating the mutants P. chrysogenum pafBpaf_promoter and P. chrysogenum pafBpaf_terminator. Both mutants were verified to contain a pafB-locus specific single integration of the respective transforming DNA, replacing the wt pafB gene (Supplementary Figure S1a,c). The pafB expression in these mutants was compared with that of the P. chrysogenum wt. The exchange of the pafB-promoter by the paf-promoter resulted in a pafB transcription pattern resembling that of paf. The significantly induced pafB-mRNA transcription peaked in 72 h cultures of P. chrysogenum pafBpaf_promoter compared to the wt strain (Figure 6A). The dramatically increased and prolonged pafB transcription time resulted in the detection of high amounts of secreted PAFB in the culture broth after 72 h of incubation (Figure 6B). In contrast, the use of the paf-terminator mildly increased the signal for pafB-mRNA after 48 h of cultivation but did not prolong the gene expression. No PAFB could be detected in the supernatant of this mutant (Figure 6A,B).
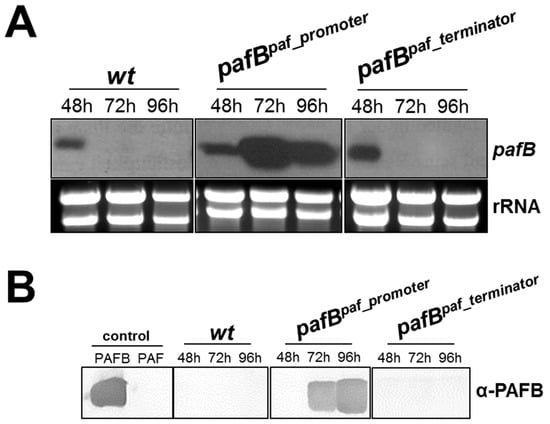
Figure 6.
Expression of pafB in P. chrysogenum wt and in the mutants pafBpaf_promoter and pafBpaf_terminator. Strains were cultivated in 1× PcMM up to 96 h at 25 °C. Samples were analyzed after 48, 72 and 96 h. (A) Northern blot: pafB-mRNA expression in the mutants and the wt. Ten μg total RNA per lane were loaded on a 1.2% denaturing agarose gel, blotted and hybridized with a pafB-specific DIG-labelled probe. Ethidium bromide-stained rRNA (26S and 18S) is shown as loading control. (B) Western blot: Secretion of PAFB into the culture supernatant. Culture supernatants (25 μL per lane) were loaded on a 18% (w/v) SDS polyacrylamide gel and size fractionated proteins were transferred on a nitrocellulose membrane. A polyclonal antibody was used for specific PAFB detection in a 1000-fold dilution. As control, 1 µg purified PAFB and PAF were loaded.
When comparing the biomass of the P. chrysogenum pafBpaf_promoter with that of the wt, the dry weight was less when the strains were grown for 72–96 h in 1× PcMM than in 4× PcMM, but there was no strain-dependent difference when cultivated in either of the two media (Figure S3a). Similarly, no morphological variation from the wt strain could be observed (data not shown).
Interestingly, pafB-mRNA transcription was strongly induced in P. chrysogenum wt surface cultures after 72 h and 96 h, irrespective of the nutrient availability, though gene expression was slightly higher in 4× PcMM compared to 1× PcMM (Figure S3b). This indicates that PAFB might play a greater role under surface than under liquid growth conditions, where pafB was hardly expressed in 1× PcMM. Notably, the colonies showed intensive sporulation on 1× PcMM agar whereas sporulation of the colonies on 4× PcMM agar was delayed (Supplementary Figure S3c). This was also reflected in the conidial counts (Figure S3d). Comparison of the colonies of the P. chrysogenum wt and the pafBpaf_promoter strain, however, showed no difference in phenotype (Figure S3a,c). This was also true, when the colony establishment of these two strains was investigated. Neither germination efficiency nor germ tube length were affected by the deregulation of the pafB expression (data not shown).
Taken together, our results indicate that regulatory elements in the pafB-promoter determine the amount and duration of pafB gene transcription. Deregulation of pafB expression does not cause any significant changes in vegetative growth, conidiation and morphology in P. chrysogenum.
3.4. The pafB-Promoter Is Strong and Inducible
Since we were able to identify conditions under which PAFB is expressed in P. chrysogenum wt, we tested the efficiency of the pafB-promoter with that of two other well established promoter-systems derived from P. chrysogenum. We therefore evaluated the expression of pafB under the regulation of the paf-promoter [6,20] and the xylose-inducible promoter of the xylanase gene (xylP) [26]. For this purpose, we used (i) the P. chrysogenum pafBpaf_promoter and (ii) created a new P. chrysogenum pafBxylP_promoter mutant where the promoter of the pafB gene was replaced by the xylP-promoter. The P. chrysogenum mutant pafBxylP_promoter was verified to carry one single integration of this expression cassette at the pafB locus (Figure S1b). The performance of PAFB production in these two mutants was compared to that of the wt strain, in which the expression of the endogenous pafB was under the regulation of its own promoter. The P. chrysogenum mutants and wt strain were cultivated under the conditions that induce the respective promoters.
After 96 h of cultivation, the highest PAFB-amounts in the culture broth were detected for the P. chrysogenum wt strain grown in 4× PcMM and the pafBpaf_promoter mutant grown in 1× PcMM. The pafBxylP_promoter mutant in contrast, produced significantly less PAFB (Figure 7A).
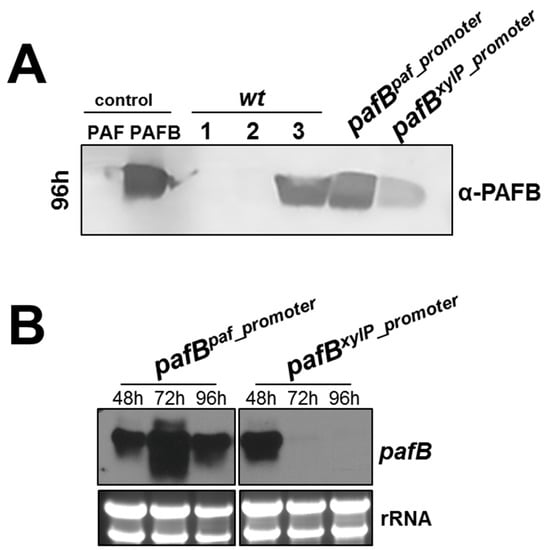
Figure 7.
Comparison of pafB expression under different promoters derived from P. chrysogenum. PAFB expression was monitored under the paf-promoter, the xylP-promoter and the pafB-promoter. The mutant pafBpaf_promoter was grown in 1× PcMM (2% sucrose), the mutant pafBxylP_promoter was grown in 1× PcMM (2% xylose) and the P. chrysogenum wt was grown in 4× PcMM. As control the wt strain was grown in 1× PcMM with either sucrose or xylose as sole carbon source. (A) Western blot to monitor PAFB secretion into the culture supernatant after 96 h of cultivation. Culture supernatants (25 μL per lane) were loaded on a 18% (w/v) SDS polyacrylamide gel and size fractionated proteins were transferred on a nitrocellulose membrane. A polyclonal antibody was used for specific PAFB detection in a 1000-fold dilution. 1, P. chrysogenum wt grown in 1× PcMM (2% sucrose); 2, P. chrysogenum wt grown in 1× PcMM (2% xylose); 3, P. chrysogenum wt grown in 4× PcMM (8% sucrose). As control, 1 µg purified PAFB and PAF were loaded. (B) Northern blot: Samples to monitor mRNA expression were taken after 48, 72 and 96 h of incubation at 25 °C. Ten μg total RNA per lane were loaded on a 1.2% denaturing agarose gel, blotted and hybridized with a pafB-specific DIG-labelled probe. Ethidium bromide-stained rRNA is shown as loading control.
Gene expression monitored in the time course of 48–96 h after inoculation revealed, that the paf-promoter efficiency was similar to that of the pafB-promoter in the wt, but superior to that of the xylP-promoter, as the pafB transcription ceased after 48 h of xylose induction (Figure 7B). These findings strengthen our assumption that the yield of PAFB in the culture supernatant depends on the amount and duration of the pafB transcription.
3.5. Secreted PAFB Exhibits Different N-Termini
For quantification of the PAFB-yield that can be achieved with the two most efficient promoters, we purified PAFB from the supernatants of 96 h old P. chrysogenum wt strain and pafBpaf_promoter mutant. Under inducible pafB-promoter condition (4× PcMM) we purified up to 5 mg/L native PAFB. The pafBpaf_promoter mutant grown in 1× PcMM produced up to 3 mg/L PAFB. This indicates that the protein yield from a single-integrated pafB gene copy regulated by the pafB- and the paf-promoter is very similar.
In our previous study, we generated high amounts of recombinant PAFB for structural and functional studies by the multicopy integration of an expression plasmid that induces transcription of pafB via the paf-promoter [7]. Mass spectrometry identified three different N-terminal variants of PAFB, the full-length form of the mature protein and two truncated forms lacking either the last one leucine (-L) or the last two N-terminal amino acids leucine and serine (-LS) [7]. At that time, the question remained unanswered if the N-terminal variation of PAFB resulted from defective cleavage of the pro-sequence from the mature protein under overproducing conditions. We were now able to address this issue by the analysis of native PAFB produced by P. chrysogenum wt in 4× PcMM and the PAFB produced by the pafBpaf_promoter mutant. The ESI-MS analysis indicated the presence of the full length mature protein form (6.490 kDa) and the form lacking the last two N-terminal amino acids leucine and serine (6.289 kDa), which were also found for recombinant PAFB [7]. The N-terminal variant lacking only the last N-terminal leucine (6.376 kDa) was detectable in very low amounts (Figure 8A). PAFB generated by the pafBpaf_promoter mutant consisted of a mixture of all three N-terminal variants that were identified previously for recombinant PAFB (Figure 8B).
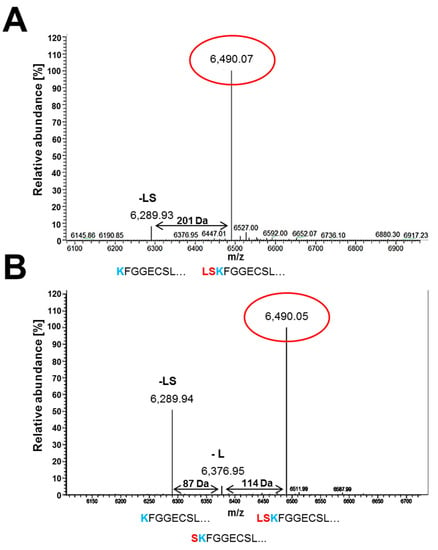
Figure 8.
Electrospray ionization mass spectrometry (ESI-MS) analysis of purified PAFB from (A) P. chrysogenum wt and (B) pafBpaf_promoter mutant. The x-axis represents the ratio between mass and charge and the y-axis represents the relative abundance of isotopes. The masses correspond to the different N-terminally truncated PAFB forms: the full length (6490.03 Da), the truncated form lacking the amino acid leucine (6376 Da, -L) and the truncated variant lacking two amino acids leucine and serine (6289.91 Da, -LS), respectively. The aa sequences below the peaks represent the N-terminus variations of PAFB.
4. Discussion
Studies on AMPs from A. giganteus [21] and P. chrysogenum [34] suggest afp-like genes to be highly expressed in submerged cultures under nonfavorable growth conditions during the stationary growth phase. The A. giganteus AFP has been found mostly secreted under carbon starvation, heat shock and pH stress [21], PAF from P. chrysogenum accumulates in the culture broth under carbon limitation [34] and the gene transcription level of anafp is induced during environmental stress conditions [22]. Therefore, AMPs from Ascomycetes were suggested to be important molecules involved in survival under unfavorable growth conditions [25].
Our attempts to identify PAFB in the culture broth of P. chrysogenum when cultivated under nutrient limiting conditions had failed, so far. The existence of the gene-encoded PAFB remained questionable and we produced recombinant PAFB applying a P. chrysogenum-based expression system [7].
Here, we provide a comprehensive study on the culture conditions favoring the expression of the antimicrobial protein PAFB in the P. chrysogenum wt strain. We tested media with up to 4-fold concentrated nutrients (4× PcMM, 4× CM), where PAFB reached the highest amounts in the culture broth (Figure 2, Figure 4). To the best of our knowledge, this is the first report on the expression of a fungal AMP under nutrient excess conditions.
In comparison to PAFB, PAF was strongly induced in 1× PcMM under nutrient starvation, as reported earlier [34]. However, PAF expression was mildly repressed under nutrient excess (4× PcMM) compared to the limiting conditions, but still present in sufficient amounts. This observation points towards a more complex regulation, excluding nutrient starvation to be the only trigger for PAF expression as assumed so far [6,25] (Figure 2, Figure 4).
To further investigate the expression regulation of pafB, we analyzed the impact of the PcMM compounds on protein production. No protein was detectable when potassium, iron, magnesium or sucrose were added in 4-fold amounts to the 1× PcMM, respectively. Contrary, increased nitrogen availability triggered PAFB secretion. However, the protein amounts produced were significantly lower than in 4× PcMM (Figure 3). These findings suggest that the nutrient combination triggers the maximum expression of PAFB. A promoter expression analysis planned in the near future will help to dissect in detail regulatory elements involved in the pafB gene expression.
Interestingly, the morphology of the fungal biomass markedly differed between PAFB-inducing conditions (fluffy mycelium) and noninducing conditions (dense pellets). Some studies describe the impact of fungal morphology on the yield of fungal products [35]. It has been shown that the formation of small pellets support the production of, e.g., citric acid with A. niger [36], whereas enzymes like amylases or neo-fructosyl-transferases are more efficiently produced from dispersed mycelia [37]. Obviously, culture conditions that favor mycelial growth of P. chrysogenum enhance the PAFB production and secretion into the supernatant.
AMP expression requires distinct environmental stimuli and is under tight temporal control. Not only nutrient limitation or unfavorable growth conditions, as originally assumed, but also nutrient excess could induce the production of certain AMPs as shown in our study. It would therefore be interesting to test whether these nutrient excess conditions induce the production of other AMPs that have remained undetectable in the culture supernatant so far, e.g., AfpB from P. digitatum [38] or NFAP from N. fischeri [8].
Considering that AMPs are secreted into the supernatant of a submerse culture in the late exponential or stationary growth phase (>48 h) and importantly, in the absence of invading microbes, it is likely that AMPs have a biological function that goes beyond their antifungal activity. P. chrysogenum harbors more than one gene coding for AMPs [1]. The different expression regulation of PAFB and PAF let us therefore conclude that both proteins cover distinct functional roles in the producer.
For PAF an intrinsic function has been already demonstrated. The deletion of the paf gene resulted in a P. chrysogenum mutant with impaired conidiogenesis and therefore this protein was assumed to be a mediator of asexual development in P. chrysogenum [23]. Interestingly, apoptosis rates and expression of autophagy related genes were lower under carbon limitation in a paf-null mutant than in the wt strain [24]. Data generated via a transcriptome meta-analysis suggested a role of AnAFP during nutrient starvation and autophagic processes in A. niger [22].
PAFB seems to cover a function distinct from PAF, as it is produced under favorable growth conditions and during exponential growth of P. chrysogenum, that showed no signs of nutrient limitation or autophagy (Figure 5). Notably, our data also revealed no significant repression of ribosomal gene expression (S5 and S6), which would have been expected when autophagy was triggered by TOR signaling under PAF-inducing nonfavorable culture conditions (1× PcMM). This stands in contrast to previous reports that assign PAF a role in nutrient recycling under stress conditions [34]. Differences in cultivation conditions could explain these divergent observations, which need further investigations.
At the moment, we cannot assign a specific biological function to PAFB for P. chrysogenum as overproduction of PAFB resulted in no significant phenotype concerning morphology, biomass and conidiation (Figure S3). The generation of a P. chrysogenum strain defective in pafB production is in progress and will shed light on this question.
P. chrysogenum has been extensively used in biotechnology as “cell factory” for the production of biomolecules [39] and therefore advanced technologies for large-scale cultivation are well established. However, one of the major players in any efficient expression system is a strong promoter and the knowledge about the conditions enabling its best performance [6]. A strong promoter derived from P. chrysogenum is the paf-promoter [6]. The P. chrysogenum-based expression system—developed in our lab—uses the paf-promoter for the high yield production of AMPs in defined minimal medium for structural and functional analysis [20].
Another very prominent example for a potent fungi-derived promoter is the xylose-inducible xylP-promoter from P. chrysogenum [26,40]. This promoter has the advantage to be inducible by xylose as the sole carbon source and can be silenced by the presence of glucose. This allows the generation of biomass prior to protein expression when the promoter is silenced. Then, the mycelium can be shifted to a medium containing xylose to induce transcription and protein production. Therefore, the xylP-promoter is an excellent tool for the generation of products that are toxic for the producing microorganism.
In this study, we have identified a new promoter from P. chrysogenum with high potential for biotechnological use in protein production. The pafB-promoter is highly efficient when induced by nutrient excess conditions. By comparing the performance of the pafB-promoter to that of the xylP- and the paf-promotor in protein yield, the pafB-promoter turned out to be similarly efficient as the paf-promoter and superior to the xylP-promoter (Figure 7). The observed high yields of recombinant proteins achieved with the multi-copy integrations of the paf-promoter-based protein expression cassettes in P. chrysogenum [20], promises a significantly improved amount of recombinant gene products when using the pafB-promoter combined with multiple genomic integrations of the respective expression cassettes, but with the advantage of an inducible promoter.
Finally, the identification of culture conditions that promote PAFB expression allowed us to clarify that the variable N-termini of PAFB occur naturally and do not result from protein overexpression (Figure 8) [7]. The quantities of the respective N-terminal PAFB variants, however, differed between the respective cultivation conditions applied. It is likely that the media composition might be responsible for this observation as the amount of carbon and nitrogen, and the pH influence the presence of proteases in the culture broth [41].
5. Conclusions
We have identified conditions triggering the expression of the native PAFB protein in P. chrysogenum that differ from the reported ones and from those assumed for the so-far uncharacterized AMPs. Our results indicate that PAFB is most efficiently induced under favorable growth conditions when nutrient availability in the culture broth is high. If AMPs fulfill additional roles for their producing organisms beside their antimicrobial functions, these functions differ between AMPs originating from the same producing strain. AMP function might not be restricted to growth control by autolysis, autophagy or apoptosis under starvation conditions, as suggested so far. Further investigations are necessary to unravel these functions.
Based on the identification of culture conditions that trigger pafB expression, we propose that the pafB-promoter a perfect tool for recombinant protein expression. Furthermore, its inducibility by nutrient excess in minimal medium allows the production of toxic proteins in P. chrysogenum.
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-2607/7/12/654/s1. Figure S1: Scheme of construction and verification of the mutants (a) pafBpaf_promoter (b) pafBxylP_promoter (c) pafBpaf_terminator in comparison to the P. chrysogenum wt. Figure S2: Verification of penicillin production in the culture broth of P. chrysogenum. Figure S3: Phenotypical characterization of the P. chrysogenum pafBpaf_promoter strain in comparison to the P. chrysogenum wt. Table S1: Media used in this study. Table S2: Microbial strains used in this study. Table S3: Oligonucleotides used in this study.
Author Contributions
Conceptualization, A.H., F.M.; funding acquisition, F.M.; investigation, A.H., H.L.; methodology, A.H.; project administration, F.M.; supervision, F.M.; validation, A.H., F.M.; writing—original draft, A.H., F.M.; writing—review & editing, A.H., F.M.
Funding
This research was funded by the Austrian Science Fund FWF, grant number I 1644-B20 to F.M.
Acknowledgments
We thank Carmen Kandelbauer and Doris Bratschun-Khan for technical assistance. We are grateful to Ivo Zadra for providing the plasmid px-ergA. Open Access Funding by the Austrian Science Fund (FWF).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sonderegger, C.; Váradi, G.; Galgóczy, L.; Kocsubé, S.; Posch, W.; Borics, A.; Dubrac, S.; Tóth, G.K.; Wilflingseder, D.; Marx, F. The evolutionary conserved gamma-core motif influences the anti-Candida activity of the Penicillium chrysogenum antifungal protein PAF. Front. Microbiol. 2018, 9, 1655. [Google Scholar] [CrossRef] [PubMed]
- Hajdu, D.; Huber, A.; Czajlik, A.; Tóth, L.; Kele, Z.; Kocsubé, S.; Fizil, Á.; Marx, F.; Galgóczy, L.; Batta, G. Solution structure and novel insights into phylogeny and mode of action of the Neosartorya (Aspergillus) fischeri antifungal protein (NFAP). Int. J. Biol. Macromol. 2019, 129, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Galgóczy, L.; Borics, A.; Virágh, M.; Ficze, H.; Váradi, G.; Kele, Z.; Marx, F. Structural determinants of Neosartorya fischeri antifungal protein (NFAP) for folding, stability and antifungal activity. Sci. Rep. 2017, 7, 1963. [Google Scholar] [CrossRef] [PubMed]
- Batta, G.; Barna, T.; Gáspári, Z.; Sándor, S.; Kövér, K.E.; Binder, U.; Sarg, B.; Kaiserer, L.; Chhillar, A.K.; Eigentler, A.; et al. Functional aspects of the solution structure and dynamics of PAF--a highly-stable antifungal protein from Penicillium chrysogenum. FEBS J. 2009, 276, 2875–2890. [Google Scholar] [CrossRef]
- Fizil, Á.; Gáspári, Z.; Barna, T.; Marx, F.; Batta, G. “Invisible” conformers of an antifungal disulfide protein revealed by constrained cold and heat unfolding, CEST-NMR experiments, and molecular dynamics calculations. Chemistry 2015, 21, 5136–5144. [Google Scholar] [CrossRef]
- Marx, F.; Haas, H.; Reindl, M.; Stöffler, G.; Lottspeich, F.; Redl, B. Cloning, structural organization and regulation of expression of the Penicillium chrysogenum paf gene encoding an abundantly secreted protein with antifungal activity. Gene 1995, 167, 167–171. [Google Scholar] [CrossRef]
- Huber, A.; Hajdu, D.; Bratschun-Khan, D.; Gáspári, Z.; Varbanov, M.; Philippot, S.; Fizil, Á.; Czajlik, A.; Kele, Z.; Sonderegger, C.; et al. New antimicrobial potential and structural properties of PAFB: A cationic, cysteine-rich protein from Penicillium chrysogenum Q176. Sci. Rep. 2018, 8, 1751. [Google Scholar] [CrossRef]
- Kovács, L.; Virágh, M.; Takó, M.; Papp, T.; Vágvölgyi, C.; Galgóczy, L. Isolation and characterization of Neosartorya fischeri antifungal protein (NFAP). Peptides 2011, 32, 1724–1731. [Google Scholar] [CrossRef]
- Tóth, L.; Kele, Z.; Borics, A.; Nagy, L.G.; Váradi, G.; Virágh, M.; Takó, M.; Vágvölgyi, C.; Galgóczy, L. NFAP2, a novel cysteine-rich anti-yeast protein from Neosartorya fischeri NRRL 181: Isolation and characterization. AMB Express 2016, 6, 75. [Google Scholar] [CrossRef]
- Garrigues, S.; Gandía, M.; Popa, C.; Borics, A.; Marx, F.; Coca, M.; Marcos, J.F.; Manzanares, P. Efficient production and characterization of the novel and highly active antifungal protein AfpB from Penicillium digitatum. Sci. Rep. 2017, 7, 14663. [Google Scholar] [CrossRef]
- Garrigues, S.; Gandía, M.; Castillo, L.; Coca, M.; Marx, F.; Marcos, J.F.; Manzanares, P. Three antifungal proteins from Penicillium expansum: Different patterns of production and antifungal activity. Front. Microbiol. 2018, 9, 2370. [Google Scholar] [CrossRef] [PubMed]
- Lacadena, J.; Martínez del Pozo, A.; Gasset, M.; Patiño, B.; Campos-Olivas, R.; Vázquez, C.; Martínez-Ruiz, A.; Mancheño, J.M.; Oñaderra, M.; Gavilanes, J.G. Characterization of the antifungal protein secreted by the mould Aspergillus giganteus. Arch. Biochem. Biophys. 1995, 324, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Wnendt, S.; Ulbrich, N.; Stahl, U. Molecular-cloning, sequence-analysis and expression of the gene encoding an antifungal-protein from Aspergillus giganteus. Curr. Genet. 1994, 25, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.G.; Shin, S.Y.; Maeng, C.Y.; Jin, Z.Z.; Kim, K.L.; Hahm, K.S. Isolation and characterization of a novel antifungal peptide from Aspergillus niger. Biochem. Biophys. Res. Commun. 1999, 263, 646–651. [Google Scholar] [CrossRef]
- Marx, F.; Binder, U.; Leiter, E.; Pócsi, I. The Penicillium chrysogenum antifungal protein PAF, a promising tool for the development of new antifungal therapies and fungal cell biology studies. Cell. Mol. Life Sci. 2008, 65, 445–454. [Google Scholar] [CrossRef]
- Kovács, R.; Holzknecht, J.; Hargital, Z.; Papp, C.; Farkas, A.; Borics, A.; Tóth, L.; Váradi, G.; Tóth, G.K.; Kovács, I.; et al. In Vivo applicability of Neosartorya fischeri antifungal Protein 2 (NFAP2) in treatment of vulvovaginal candidiasis. Antimicrob. Agents Chemother. 2019, 63, e01777-18. [Google Scholar] [CrossRef]
- Szappanos, H.; Szigeti, G.P.; Pál, B.; Rusznák, Z.; Szucs, G.; Rajnavölgyi, E.; Balla, J.; Balla, G.; Nagy, E.; Leiter, E.; et al. The antifungal protein AFP secreted by Aspergillus giganteus does not cause detrimental effects on certain mammalian cells. Peptides 2006, 27, 1717–1725. [Google Scholar] [CrossRef]
- Szappanos, H.; Szigeti, G.P.; Pál, B.; Rusznák, Z.; Szucs, G.; Rajnavölgyi, E.; Balla, J.; Balla, G.; Nagy, E.; Leiter, E.; et al. The Penicillium chrysogenum-derived antifungal peptide shows no toxic effects on mammalian cells in the intended therapeutic concentration. Naunyn-Schmiedebergs Arch. Pharmacol. 2005, 371, 122–132. [Google Scholar] [CrossRef]
- Palicz, Z.; Jenes, A.; Gáll, T.; Miszti-Blasius, K.; Kollár, S.; Kovács, I.; Emri, M.; Márián, T.; Leiter, E.; Pócsi, I.; et al. In Vivo application of a small molecular weight antifungal protein of Penicillium chrysogenum (PAF). Toxicol. Appl. Pharmacol. 2013, 269, 8–16. [Google Scholar] [CrossRef]
- Sonderegger, C.; Galgóczy, L.; Garrigues, S.; Fizil, Á.; Borics, A.; Manzanares, P.; Hegedüs, N.; Huber, A.; Marcos, J.F.; Batta, G.; et al. A Penicillium chrysogenum-based expression system for the production of small, cysteine-rich antifungal proteins for structural and functional analyses. Microb. Cell Fact. 2016, 15, 192. [Google Scholar] [CrossRef]
- Meyer, V.; Wedde, M.; Stahl, U. Transcriptional regulation of the antifungal protein in Aspergillus giganteus. Mol. Genet. Genomics 2002, 266, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Paege, N.; Jung, S.; Schäpe, P.; Müller-Hagen, D.; Ouedraogo, J.P.; Heiderich, C.; Jedamzick, J.; Nitsche, B.M.; van den Hondel, C.A.; Ram, A.F.; et al. A transcriptome meta-analysis proposes novel biological roles for the antifungal protein AnAFP in Aspergillus niger. PLoS ONE 2016, 11, e0165755. [Google Scholar] [CrossRef] [PubMed]
- Hegedüs, N.; Sigl, C.; Zadra, I.; Pócsi, I.; Marx, F. The paf gene product modulates asexual development in Penicillium chrysogenum. J. Basic Microbiol. 2011, 51, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Kovács, B.; Hegedüs, N.; Bálint, M.; Szabó, Z.; Emri, T.; Kiss, G.; Antal, M.; Pócsi, I.; Leiter, E. Penicillium antifungal protein (PAF) is involved in the apoptotic and autophagic processes of the producer Penicillium chrysogenum. Acta Microbiol. Immunol. Hung. 2014, 61, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V.; Jung, S. Antifungal peptides of the AFP family revisited: Are these cannibal toxins? Microorganisms 2018, 6, 50. [Google Scholar] [CrossRef]
- Zadra, I.; Abt, B.; Parson, W.; Haas, H. XylP promoter-based expression system and its use for antisense downregulation of the Penicillium chrysogenum nitrogen regulator NRE. Appl. Environ. Microbiol. 2000, 66, 4810–4816. [Google Scholar] [CrossRef]
- Kück, U.; Hoff, B. Application of the nourseothricin acetyltransferase gene (nat1) as dominant marker for the transformation of filamentous fungi. Fungal Genet. Rep. 2006, 53, 3. [Google Scholar] [CrossRef]
- Sigl, C.; Handler, M.; Sprenger, G.; Kürnsteiner, H.; Zadra, I. A novel homologous dominant selection marker for genetic transformation of Penicillium chrysogenum: Overexpression of squalene epoxidase-encoding ergA. J. Biotechnol. 2010, 150, 307–311. [Google Scholar] [CrossRef]
- Cantoral, J.M.; Díez, B.; Barredo, J.L.; Alvarez, E.; Martín, J.F. High-frequency transformation of Penicillium chrysogenum. Nat. Biotechnol. 1987, 5, 494–497. [Google Scholar] [CrossRef]
- Barreiro, C.; García-Estrada, C. Proteomics and Penicillium chrysogenum: Unveiling the secrets behind penicillin production. J. Proteom. 2019, 198, 119–131. [Google Scholar] [CrossRef]
- Pandey, S.; Ahmad, T.; Aryal, S.; Rana, B.; Sapkota, B. Penicillin production and history: An overview. IJOMAS 2014, 1, 103–108. [Google Scholar]
- Abraham, E.P.; Chain, E. An enzyme from bacteria able to destroy penicillin. Rev. Infect. Dis. 1988, 10, 677–678. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Troya, S.; Pérez-Pérez, M.E.; Florencio, F.J.; Crespo, J.L. The role of TOR in autophagy regulation from yeast to plants and mammals. Autophagy 2008, 4, 851–865. [Google Scholar] [CrossRef]
- Marx, F. Small, basic antifungal proteins secreted from filamentous ascomycetes: A comparative study regarding expression, structure, function and potential application. Appl. Microbiol. Biotechnol. 2004, 65, 133–142. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, J. The filamentous fungal pellet and forces driving its formation. Crit. Rev. Biotechnol. 2016, 36, 1066–1077. [Google Scholar] [CrossRef]
- Gómez, R.; Schnabel, I.; Garrido, J. Pellet growth and citric-acid yield of Aspergillus niger 110. Enzym. Microb. Technol. 1988, 10, 188–191. [Google Scholar] [CrossRef]
- Driouch, H.; Sommer, B.; Wittmann, C. Morphology engineering of Aspergillus niger for improved enzyme production. Biotechnol. Bioeng. 2010, 105, 1058–1068. [Google Scholar] [CrossRef]
- Garrigues, S.; Gandía, M.; Marcos, J.F. Occurrence and function of fungal antifungal proteins: A case study of the citrus postharvest pathogen Penicillium digitatum. Appl. Microbiol. Biotechnol. 2016, 100, 2243–2256. [Google Scholar] [CrossRef]
- Jami, M.S.; Barreiro, C.; García-Estrada, C.; Martín, J.F. Proteome analysis of the penicillin producer Penicillium chrysogenum: Characterization of protein changes during the industrial strain improvement. Mol. Cell Proteom. 2010, 9, 1182–1198. [Google Scholar] [CrossRef]
- Marx, F.; Salvenmoser, W.; Kaiserer, L.; Graessle, S.; Weiler-Görz, R.; Zadra, I.; Oberparleiter, C. Proper folding of the antifungal protein PAF is required for optimal activity. Res. Microbiol. 2005, 156, 35–46. [Google Scholar] [CrossRef]
- Ire, F.S.; Okolo, B.N.; Moneke, A.N.; Odibo, F.J.C. Influence of cultivation conditions on the production of a protease from Aspergillus carbonarius using submerged fermentation. Afr. J. Food Sci. 2011, 5, 353–365. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).